Abstract
Purpose
To investigate the long‐term outcomes of patients who underwent vitrectomy for proliferative diabetic retinopathy.
Methods
Cumulative incidences were calculated for low vision (<0.3), re‐vitrectomy in the study eye and fellow eye vitrectomy. To identify potential prognostic factors that associate with these outcomes, we used multivariable Cox regression models.
Results
In a total of 217 patients, we found 1‐, 5‐ and 10‐year cumulative incidences of low vision in the study eye of 24%, 31% and 39%, respectively. For both eyes, these rates were, respectively, 10%, 14% and 14%. Low vision in both eyes was associated with higher age and worse contralateral visual acuity. The 1‐, 5‐ and 10‐year cumulative incidences for re‐vitrectomy in the study eye were 16%, 27% and 27%, respectively, and for a vitrectomy in the fellow eye 24%, 40% and 54%, respectively. Re‐vitrectomy of the study eye was associated with worse contralateral visual acuity, while vitrectomy of the fellow eye was associated with shorter diabetes duration, worse contralateral visual acuity, higher HbA1c level and worse diabetic retinopathy severity stage of the fellow eye.
Conclusion
Functional visual acuity in at least one eye was achieved or preserved in most patients. After 10 years, about a quarter of all patients underwent a re‐vitrectomy, while more than half of the patients needed a vitrectomy of the fellow eye. Knowledge of these long‐term outcomes is essential when counselling patients for a vitrectomy.
Keywords: diabetic retinopathy, vitrectomy, long‐term outcomes
Introduction
Proliferative diabetic retinopathy (PDR) is a sight‐threatening complication of diabetes mellitus (DM), affecting 7% of all 425 million people living with DM (Yau et al. 2012). The relative incidence of PDR and associated blindness has declined over the past decades due to improvements in diabetes care. These include a tighter grip on blood glucose levels through self‐monitoring devices and various modern types of insulin, implants with real‐time analysis and metered insulin delivery, as well as expanded screening programmes for the ophthalmic complications of diabetes. Moreover, the introduction of anti‐vascular endothelial growth factors (VEGFs) and the refinement of surgical techniques have greatly improved diabetic eye care, including PDR (Antonetti et al. 2012).
Unfortunately, the current diabetic epidemic with its steep incline of diabetic patients still leads to a rising prevalence of PDR. PDR therefore continues to be a major burden on public health (Antonetti et al. 2012). If severe consequences of PDR develop, a vitrectomy can be required to maintain and/or restore visual function. The indications according to the American Academy of Ophthalmology (AAO) for a vitrectomy include the following: non‐clearing vitreous haemorrhage; significant recurring vitreous haemorrhage, despite use of maximal panretinal photocoagulation; dense premacular subhyaloid haemorrhage; tractional retinal detachment involving or threatening the macula; combined tractional and rhegmatogenous retinal detachment; red blood cell‐induced glaucoma and ‘ghost cell’ glaucoma; and anterior segment neovascularization with media opacities preventing panretinal photocoagulation (McCannel et al. 2018). The yearly incidence of a vitrectomy in patients with PDR is estimated to be 6% (Vaideanu et al. 2014).
The majority of the patients that require a vitrectomy for PDR are relatively young and have jobs and families to maintain. Vision loss for these individuals has a large impact on their everyday life and society as a whole. As PDR is a bilateral disorder, the long‐term visual prognosis in these patients is a major concern. Most studies investigating the outcomes of vitrectomy for PDR report, however, short‐term results and largely focus on the operated eye. Arguably, for diabetic patients, the long‐term prognosis for bilateral visual function is important. A recent study by Ostri et al. reported that two thirds of the patients undergoing vitrectomy for PDR obtained a visual acuity of ≥0.3 in the operated eye after 10 years (Ostri et al. 2014). Studies on bilateral visual outcomes after vitrectomy date back at least fifteen years ago and report that 80% of the patients maintained or obtained a visual acuity of ≥0.1 in at least one eye, while 43% underwent fellow eye vitrectomy (Smiddy & Feuer 2004; Vote et al. 2004). Recent studies on bilateral long‐term outcomes of vitrectomy are lacking, and little studies cover a study period of 5 years or longer (Blankenship & Machemer 1985; Ostri et al. 2014). The aim of this study was therefore to study the long‐term outcome of eyes following vitrectomy for PDR as well as the visual prognosis in broader sense. In addition, we studied candidate prognostic factors that may be used in future risk stratification.
Methods
Study population
This study included all patients who underwent primary pars plana vitrectomy for complications of PDR at the Radboud University Medical Centre Nijmegen, The Netherlands, between September 2006 and June 2012. We employed the following exclusion criteria: a follow‐up duration of less than 6 months, prior vitrectomy in either eye, missing intraoperative data of the primary vitrectomy and pre‐existing low vision in the operated eye due to non‐diabetic comorbidity. Low vision was defined as a best‐corrected visual acuity (BCVA) of <0.3 and blindness as a BCVA of <0.05, according to the standards of the World Health Organization (WHO 2015). Follow‐up data were collected both from the tertiary healthcare centre where the vitrectomy was performed and from the referring ophthalmologist. This study was approved by the medical ethical committee of the Radboud University Medical Center Nijmegen and adhered to the tenets of Helsinki.
Surgical procedures
A 20‐ or 23‐gauge 3‐port pars plana vitrectomy was performed in all eyes. First, a core vitrectomy was performed to remove vitreous haemorrhage or opacified vitreous. If present, anterior–posterior vitreoretinal traction was released and vasoproliferative membranes were dissected. Subsequently, the peripheral vitreous was removed as completely as possible using scleral indentation. After fluid–air exchange, photocoagulation for retinal breaks and/or panretinal photocoagulation was applied in case of insufficient previous panretinal laser treatment. If needed, photocoagulation with indentation was applied to treat the ischaemic retina in the far periphery. An air, SF6 gas, C3F8 gas or silicon oil tamponade was employed, depending on the aspect of the retina.
Study outcomes
The primary outcomes of this study were the occurrence of low vision and blindness in either eye and for both eyes, the need for a re‐vitrectomy in the study eye and the need for a vitrectomy in the fellow eye. Removal of silicon oil tamponade was not considered as a re‐vitrectomy. Secondary outcome measures were survival rate, survival of the crystalline lens, the occurrence of postoperative complications and the associations of baseline variables with low vision in both eyes.
Data collection
Preoperative and annual postoperative decimal BCVA in both eyes was collected, and a patient was classified as having low vision if two consecutive BCVA measurements were <0.3. General patient characteristics and preoperative variables that were recorded included sex, age, age of onset of diabetes, diabetes type, duration of diabetes, the presence of nephropathy, amputation or ischaemic heart disease, glycated haemoglobin (HbA1c, mmol/mol), body mass index (BMI, kg/m2) and mean arterial pressure (MAP, mmHg), calculated by the equation: MAP ≃ diastolic blood pressure + ⅓ (systolic blood pressure – diastolic blood pressure). In addition, the following ophthalmological parameters were collected: lens status, indication for vitrectomy, diabetic retinopathy (DR) stage in the fellow eye during the time of vitrectomy and prior treatment with panretinal photocoagulation or anti‐VEGF. Mortality data for all patients were retrieved from the Dutch population register.
Statistical analysis
Decimal BCVA was converted to the logarithm of the maximum angle of resolution (logMAR) for statistical analysis. Patients with vision loss resulting in the non‐numerical vision of perception of light or no perception of light were assigned a logMAR value of 2.78, which is the approximate equivalent of a Snellen VA of 1/300, the lowest VA observed in the cohort (Holladay 1997; Schulze‐Bonsel et al. 2006). Continuous variables were displayed as mean ± standard deviation (SD) when normally distributed, and as median with interquartile range (IQR) when the distribution was skewed. Categorical variables were displayed as a proportion with corresponding percentage. A paired t‐test was performed to investigate the difference in visual acuity before and after vitrectomy. Differences in complication rate for 20‐ or 23‐gauge instruments were tested using a Pearson chi‐square test. Kaplan–Meier survival analysis was used to generate survival curves and cumulative incidences for the occurrence of low vision, re‐vitrectomy of the study eye, vitrectomy of the fellow eye, survival and cataract surgery. To compare the mortality in our population to the general population, we simulated age‐ and sex‐matched survival rates retrieved from the public registry Statistics Netherlands with 100 iterations.
Randomly missing data were imputed using multiple imputation with twenty iterations. We used multivariable Cox regression analysis with backwards stepwise elimination of variables with p < 0.05 to identify variables that were associated with low vision in the best eye, re‐vitrectomy of the study eye and vitrectomy of the fellow eye. Variables were checked for multicollinearity using Pearson's coefficient for parametric distributions and Spearman´s rank for non‐parametric distributions. Hazard ratios with corresponding 95% confidence intervals are displayed for significantly associated variables. Analyses were limited to those patients who had a follow‐up duration of at least one year. A p‐value < 0.05 was considered statistically significant. All analyses were conducted using SPSS version 20 (SPSS, Chicago, IL, USA).
Results
Patient demographics and follow‐up
A total of 273 patients underwent primary vitrectomy for PDR during the study period, of which 40 were excluded due to a follow‐up duration of <6 months, nine due to missing intraoperative data and seven due to comorbidity causing pre‐existing low vision. Subsequently, a total of 217 patients were included in the current study. In 59 patients (27%), 23‐gauge vitrectomy was performed, and the remaining 158 patients (73%) underwent 20‐gauge vitrectomy. Silicon oil tamponade was used in 36 patients (13%). Intraoperative (additional) panretinal photocoagulation was performed in 208 patients (96%), with a mean number of 1193 coagulates. In 4 patients (2%), focal laser or laser around a retinal break was performed. Another 5 patients (2%) did not receive endolaser during the procedure. The course of follow‐up during the study period is displayed in Fig. 1. The mean follow‐up duration for all patients was 5.4 years (SD 2.9 years). Patient characteristics at baseline are shown in Table 1. In 2/3 of the patients, vitrectomy was performed because of a non‐clearing or recurrent vitreous haemorrhage. In 7% of the patients, vitreous haemorrhage was associated with a tractional retinal detachment involving or threatening the macula. Sight‐threatening tractional retinal detachment was present in 13% of patients in the absence of a vitreous haemorrhage. Neovascular glaucoma was a rare (1%) indication for surgery. A relatively common indication that did not meet with the predefined indications for diabetic vitrectomy was severe fibrovascular proliferation, unresponsive to laser therapy (12%). In these patients, surgery is considered a last resort to restrain neovascular activity. Almost half of all patients (46%) had a severe non‐proliferative or proliferative stage of DR in the fellow eye, mild stage or absent DR was present in the fellow eyes of 62 (29%) patients.
Fig. 1.
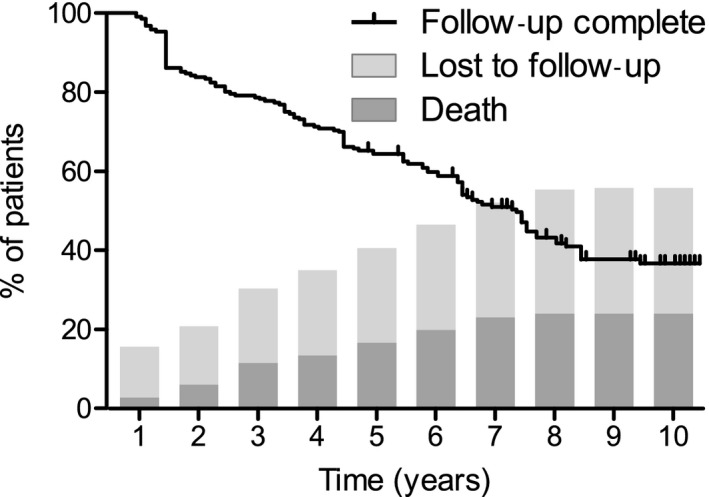
Course of follow‐up during the study period. Follow‐up completion was estimated by Kaplan–Meier survival analysis, censoring patients with complete follow‐up. Yearly cumulative incidence of deceased patients and patients lost to follow‐up were simultaneously plotted on the x‐axis.
Table 1.
Patient characteristics at baseline.
| N = 217 | |
|---|---|
| Demographics | |
| Age, years, mean ± SD [range] | 55 ± 16 [22–95] |
| Sex, n (%) | |
| Male | 122 (56%) |
| Female | 95 (44%) |
| DM Type, n (%) | |
| Type 1 | 80 (38%) |
| Type 2 | 133 (62%) |
| Duration of DM, years, mean ± SD [range] | 21 ± 12 [0–72] |
| Age of onset DM, years, mean ± SD [range] | |
| Type 1 DM | 14 ± 12 [0–41] |
| Type 2 DM | 46 ± 12 [18–76] |
| Systemic characteristics | |
| HbA1c, mmol/mol, mean ± SD | 63 ± 15 [31–103] |
| Mean arterial pressure, mmHg, mean ± SD | 101 ± 14 [76–145] |
| Body mass index, kg/m2, mean ± SD | 28.3 ± 6.6 [17–57] |
| Nephropathy, n (%) | 56 (27%) |
| Amputation, n (%) | 17 (9%) |
| Ischaemic heart disease, n (%) | 52 (24%) |
| Ophthalmological characteristics | |
| Indication for vitrectomy | |
| Non‐clearing or recurrent vitreous haemorrhage | 145 (67%) |
| Non‐clearing or recurrent vitreous haemorrhage with tractional detachment threatening or involving the macula | 16 (7%) |
| Tractional detachment threatening or involving the macula without vitreous haemorrhage | 28 (13%) |
| Neovascular glaucoma unresponsive to laser therapy | 3 (1%) |
| Extensive fibrovascular proliferation unresponsive to laser therapy | 26 (12%) |
| Other | 5 (2%) |
| Preoperative BCVA, decimals, median (IQR) [range] | |
| Study eye | 0.02 (0.003–0.20) [0–1] |
| Fellow eye | 0.50 (0.25–0.79) [0–1] |
| Duration of DR, years, median (IQR) [range] | 6 (2–12) [0–36] |
| Prior treatment for DR, n (%) | |
| None | 25 (12%) |
| Photocoagulation | 174 (83%) |
| Intravitreal anti‐VEGF | 35 (17%) |
| Lens status study eye, n (%) | |
| Phakic | 167 (77%) |
| Pseudophakic | 50 (23%) |
| DR stage fellow eye, n (%) | |
| None | 34 (16%) |
| Mild | 28 (13%) |
| Moderate | 51 (24%) |
| Severe | 13 (6%) |
| Proliferative | 85 (40%) |
Abbreviations: anti‐VEGF = anti‐vascular endothelial growth factor; BCVA = best‐corrected visual acuity; DM = diabetes mellitus; DR = diabetic retinopathy; IQR = interquartile range; n = number; SD = standard deviation.
Vision outcomes
The cumulative incidence of low vision in the study eye was 31% at 5 years, and 39% at 10 years, as is shown in Fig. 2A. For low vision in the fellow eye, cumulative incidences at 5 and 10 years were 25% and 29%, respectively, while preoperative low vision in the fellow eye was present in 55 eyes (20%). The cumulative incidence of low vision in both eyes was 14% at 5 years. In the follow‐up >5 years, we observed no new cases of bilateral low vision.
Fig. 2.

Kaplan–Meier survival plots for (A) low vision and (B) the need for additional vitrectomy. The tables represent cumulative incidences of low vision and additional vitrectomy after 5 and 10 years with the corresponding 95% confidence intervals. Abbreviations: y = years.
For blindness, the 5‐ and 10‐year cumulative incidences in the study eye were 12% and 16%. In the fellow eye, preoperative blindness was present in 18 eyes (9%), and after 5 and 10 years, we found cumulative incidences of blindness of 10% and 11%, respectively. A total of six persons (3%) suffered from blindness in both eyes after a median of 2 years [IQR 1–3] and were considered legally blind. The course of pre‐ and postoperative visual acuity of both eyes is displayed in Fig. S1.
Further surgical interventions
In total, 42 patients (21%) required a re‐vitrectomy in their study eye. If patients needed a re‐vitrectomy, the median interval was 6 months [IQR 2–15], and in 98% of the cases, the procedure was performed within three years after the first vitrectomy in the study eye, resulting in a 5‐year cumulative incidence of 27% (Fig. 2B). In one patient, an evisceration of the study eye was performed after 10 months of follow‐up, because of unbearable and uncontrollable ocular pain due to glaucoma as result of chamber angle neovascularization. In the fellow eye, a total of 81 patients (40%) required a vitrectomy, most often for the indication of proliferative DR with a non‐clearing vitreous haemorrhage (55% of all vitrectomies in the fellow eye). Of the 85 patients with proliferative DR in their fellow eye at baseline, 50 (59%) required a fellow eye vitrectomy at some time‐point during follow‐up, versus 10 out of 62 (16%) in the patients with no or mild DR in their fellow eye at baseline. The 1‐, 5‐ and 10‐year cumulative incidences for a vitrectomy in the fellow eye were 24%, 40% and 54%, respectively (Fig. 2B). Another eight people needed a re‐vitrectomy in their fellow eye, and one person needed a second re‐vitrectomy.
Prognostic factors
We subsequently studied factors predictive for low vision in both eyes, re‐vitrectomy of the study eye and vitrectomy of the fellow eye using multivariable Cox regression analysis. Low vision in both eyes in the follow‐up period was significantly associated with higher age and a lower preoperative BCVA in the fellow eye (Table S1). Lower BCVA in the fellow eye was the only significant predictor for re‐vitrectomy in the study eye (Table S1). Shorter DM duration, higher HbA1c, lower BCVA in the fellow eye and more severe stages of DR in the fellow eye were associated with the need for vitrectomy in the fellow eye (Table S1).
Survival
We then looked at the secondary outcome measures of this study. A total of 80 patients (31%) died during the period of follow‐up. The 5‐ and 10‐year survival rates were 81% and 67%, respectively, as shown in Fig. 3A. In the simulated normal population, the 5‐ and 10‐year survival rates were 94% and 88%.
Fig. 3.

Kaplan–Meier survival plots for (A) life and (B) survival of the crystalline lens. The tables represent cumulative incidences after 5 and 10 years with the corresponding 95% confidence intervals. Abbreviations: y = years. *Because the maximum follow‐up duration in the group with age >50 years was 8 years, the cumulative incidence at 8 year is given.
Cataract surgery
Prior to surgery, 50 people (23%) were pseudophakic and 167 (77%) were phakic. A total of 81 (49%) of all phakic patients at baseline underwent cataract surgery, after a median interval of 16 months [IQR 5–31]. Besides vitrectomy, age is an important factor in the development of cataract. We therefore stratified the cohort according to age and analysed patients older and those younger than 50 years separately. In patients below the age of 50, the cumulative incidence of cataract surgery was 40% after 5 years, and 52% after 10 years (Fig. 3B). In people of 50 years and older, the 5‐year cumulative incidence of cataract surgery was 83%. The maximum follow‐up duration in this group was 8 years, resulting in an 8‐year cumulative incidence of 91% (Fig. 3B).
Long‐term complications
We then investigated the complications that occurred in the study eye (Table 2). Most complications occurred within 1 year after the primary vitrectomy. Elevated IOP was the most frequent complication and occurred in 81 out of 217 (37%) patients. A recurrent vitreous haemorrhage was observed in a total of 31 patients (of which 20 (65%) required a re‐vitrectomy. Tractional retinal detachment after the primary vitrectomy occurred in 16 patients (7%), and a combined tractional and rhegmatogenous retinal detachment in 9 patients (4%). There was no statistical difference in complication rate between 20‐ and 23‐gauge vitrectomy (data not shown).
Table 2.
Incidence of complications in the study eye after the initial vitrectomy.
| <1 year | 1–5 years | >5 years | Total | |
|---|---|---|---|---|
| Recurrent vitreous haemorrhage* | 24 (11%) | 5 (2%) | 2 (1%) | 31 (14%) |
| Retinal detachment | 22 (10%) | 3 (1%) | 0 (0%) | 25 (12%) |
| Tractional | 13 (6%) | 3 (1%) | 0 (0%) | 16 (7%) |
| Rhegmatogenous + tractional | 9 (4%) | 0 | 0 (0%) | 9 (4%) |
| Macular oedema | 39 (18%) | 19 (9%) | 5 (4%) | 63 (29%) |
| Elevated intraocular pressure | 71 (33%) | 6 (3%) | 4 (3%) | 81 (37%) |
| Neovascular glaucoma | 7 (3%) | 1 (0.5%) | 0 (0%) | 8 (4%) |
| Valid number | 217 | 207 | 140 | 217 |
Only comprises delayed recurrent vitreous haemorrhage (≥1 month after primary vitrectomy).
Discussion
In the current study, we investigated the long‐term outcome in patients undergoing vitrectomy for PDR. In this relatively young patient population, besides the outcome of the operated eye, important outcome measures are the risk of visual loss of the fellow eye, the prognosis for the bilateral visual function and the need for additional surgery, because they have a large impact on everyday functioning. In the consultation room, knowledge of the long‐term prognosis in these patients is essential for management of expectations and long‐term planning.
In the majority of patients (86% after 5 years), relatively good (>0.3) visual function was maintained in at least one eye, and blindness in both eyes was observed in only 6 persons (3%). Moreover, no new cases of low vision in both eyes occurred after 5 years, suggesting that visual function in the long term can be stabilized with proper medical care. Higher age, lower BCVA in the fellow eye and the presence of tractional retinal detachment at baseline were significantly associated with an increased risk of developing low vision (0.05–0.3) in both eyes. These findings corroborate the results in previous reports, although these studies focused solely on visual outcome in the study eye (Yorston et al. 2008). Systemic measurements and the presence of other diabetic complications were also entered in the multivariate regression model, but none of these variables were associated with low vision in both eyes.
Re‐vitrectomy was indicated in a total of 42 patients (21%) and was performed within the first 3 years in the vast majority (98%) of the patients. The re‐vitrectomy rate in our study is comparable or slightly higher than the rates reported earlier, ranging between 14 and 24% (Table S2). This might be explained by the relatively short follow‐up in these studies (mean or median < 18 months) and differences in the application of photocoagulation (Smiddy et al. 1995; Mason et al. 2005; Zaninetti et al. 2005; Jackson et al. 2016). In the fellow eye, a total of 81 patients (40%) required a vitrectomy. This shows that patients undergoing a primary vitrectomy for PDR are highly likely to have significant pathology in their fellow eye, potentially requiring vitreoretinal surgery, especially when PDR in the fellow eye is already present. Patients with no or mild DR in their fellow eye at the time of the primary vitrectomy are at a much lower risk of a fellow eye vitrectomy, although this risk is not negligible. Our findings are in correspondence with Vote et al., who described a 38% intervention rate for the fellow eye (Vote et al. 2004). In our study, high HbA1c level, low preoperative BCVA in the fellow eye, a more severe DR in the fellow eye and short DM duration at the time of the primary vitrectomy were associated with an increased risk of fellow eye vitrectomy.
The presence and severity of DR have repeatedly been associated with increased mortality, irrespective of diabetes type (van Hecke et al. 2005; Kramer et al. 2011; Fisher et al. 2016). The 5‐year survival rate we found in our study was 81%. This is comparable to previous studies where 5‐year survival rates ranging between 68% and 86% were reported (Gollamudi et al. 1991; Helbig et al. 1996; Lux et al. 2012; Banerjee et al. 2016; Kim et al. 2017). The 10‐year survival rate in our study was 67%. Blankenship et al. provided a 10‐year survival rate of 50% in patients vitrectomized between 1970 and 1973; however, diabetes care has improved significantly in the last fifty years (Blankenship & Machemer 1985). Our data indicate that patients undergoing a diabetic vitrectomy have a higher mortality risk than patients in the general population. Mortality in patients undergoing a diabetic vitrectomy is often due to other micro‐ and macrovascular complications, such as renal insufficiency and ischaemic heart disease (Gollamudi et al. 1991; Helbig et al. 1996; Banerjee et al. 2016; Kim et al. 2017).
Vitrectomy may accelerate the development of cataract. The underlying mechanism of this complication is unclear, but may relate to an increased oxygen tension after vitrectomy, iatrogenic damage and the type of tamponade (Thompson 2004; Holekamp et al. 2005). In addition, age is a significant risk factor (Smiddy & Feuer 2004; Feng & Adelman 2014). In patients below the age of 50, we observed that 40% needed cataract surgery in the first 5 years of follow‐up. This figure more than doubled to 83% in individuals of 50 years and older. Prior to vitrectomy, patients should be informed about this differentiated risk of cataract extraction. Given the high rate of postvitrectomy cataract extraction in patients over 50, a combined vitrectomy and cataract extraction with intraocular lens implantation can have significant benefits, including faster recovery of visual function. However, a more conservative strategy is warranted in younger persons, as the implant of a non‐multifocal intraocular lens will not compensate the loss of accommodation.
Strengths of this study include the long duration of follow‐up, the relatively large sample size and the collection of initial vitrectomy data at a single tertiary centre, reducing external causes of variability. In addition, we studied multiple endpoints, providing a comprehensive evaluation of the long‐term outcomes of a vitrectomy for PDR. We furthermore employed broad inclusion criteria, studying all patients that underwent vitrectomy for the complications of PDR. Although not all patients met with any of the predefined indications for diabetic vitrectomy according to the AAO, we argue that exclusion of these patients would have led to an incorrect reflection of real‐life clinical practice. We should also acknowledge some limitations to the current study. A major limitation is the loss to follow‐up, which may result in under‐ or overestimation of the reported outcomes. Additionally, the retrospective nature of the study did not allow standardization of the measurements. A limitation that inherently applies to the study of long‐term outcomes is that vitrectomies were performed 6–13 years ago. Retinal surgery as it is performed today has inevitably advanced in relation to the studied period, for example regarding preoperative administration of anti‐VEGF of the use of smaller instruments. Although smaller instruments certainly have advantages over larger 20‐gauge instruments in terms of duration of surgery, patient comfort and recovery, we did not find a difference in any of the long‐term outcomes between 20‐ and 23‐gauge vitrectomy (Thompson 2011).
In conclusion, this study provides insight into the long‐term outcomes after vitrectomy for PDR and its consequences. In the majority of patients, visual function of >0.3 in at least one eye is maintained over the course of 5 to 10 years, allowing patients to perform daily activities without far‐reaching visual restrictions. We furthermore identified vision loss in the fellow eye to be predictive for a poor prognosis, with respect to both low vision and re‐vitrectomy. These results can be used for counselling patients with PDR by providing insight into the prognosis following vitrectomy including visual function of the fellow eye.
Supporting information
Fig. S1. Course of visual acuity before and after vitrectomy in decimals for both eyes.
Table S1. Predictive variables for low vision in both eyes, re‐PPV in the study eye, and PPV in the fellow eye, using Cox regression survival analysis.
Table S2. Comparison of study results in this study with previous diabetic vitrectomy studies.
The corresponding author is a member of the Dutch Ophthalmological Societies.
References
- Antonetti DA, Klein R & Gardner TW (2012): Diabetic retinopathy. N Engl J Med 366: 1227–1239. [DOI] [PubMed] [Google Scholar]
- Banerjee PJ, Moya R, Bunce C, Charteris DG, Yorston D & Wickham L (2016): Long‐term survival rates of patients undergoing vitrectomy for proliferative diabetic retinopathy. Ophthalmic Epidemiol 23: 94–98. [DOI] [PubMed] [Google Scholar]
- Blankenship GW & Machemer R (1985): Long‐term diabetic vitrectomy results. Report of 10 year follow‐up. Ophthalmology 92: 503–506. [DOI] [PubMed] [Google Scholar]
- Feng H & Adelman RA (2014): Cataract formation following vitreoretinal procedures. Clin Ophthalmol 8: 1957–1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher DE, Jonasson F, Klein R, Jonsson PV, Eiriksdottir G, Launer LJ, Gudnason V & Cotch MF (2016): Mortality in older persons with retinopathy and concomitant health conditions: the age, gene/environment susceptibility‐Reykjavik study. Ophthalmology 123: 1570–1580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gollamudi SR, Smiddy WE, Schachat AP, Michels RG & Vitale S (1991): Long‐term survival rate after vitreous surgery for complications of diabetic retinopathy. Ophthalmology 98: 18–22. [DOI] [PubMed] [Google Scholar]
- van Hecke MV, Dekker JM, Stehouwer CD et al. (2005): Diabetic retinopathy is associated with mortality and cardiovascular disease incidence: the EURODIAB prospective complications study. Diabetes Care 28: 1383–1389. [DOI] [PubMed] [Google Scholar]
- Helbig H, Kellner U, Bornfeld N & Foerster MH (1996): Life expectancy of diabetic patients undergoing vitreous surgery. Br J Ophthalmol 80: 640–643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holekamp NM, Shui YB & Beebe DC (2005): Vitrectomy surgery increases oxygen exposure to the lens: a possible mechanism for nuclear cataract formation. Am J Ophthalmol 139: 302–310. [DOI] [PubMed] [Google Scholar]
- Holladay JT (1997): Proper method for calculating average visual acuity. J Refract Surg 13: 388–391. [DOI] [PubMed] [Google Scholar]
- Jackson TL, Johnston RL, Donachie PH, Williamson TH, Sparrow JM & Steel DH (2016): The Royal College of Ophthalmologists' National Ophthalmology Database Study of Vitreoretinal Surgery: Report 6, Diabetic Vitrectomy. JAMA Ophthalmol 134: 79–85; quiz 120. [DOI] [PubMed] [Google Scholar]
- Kim BZ, Lee KL, Guest SJ & Worsley D (2017): Long‐term survival following diabetic vitrectomy. N Z Med J 130: 69–77. [PubMed] [Google Scholar]
- Kramer CK, Rodrigues TC, Canani LH, Gross JL & Azevedo MJ (2011): Diabetic retinopathy predicts all‐cause mortality and cardiovascular events in both type 1 and 2 diabetes: meta‐analysis of observational studies. Diabetes Care 34: 1238–1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lux A, Ostri C, Dyrberg E, Sander B, Lund‐Andersen H & la Cour M (2012): Survival rates after diabetic vitrectomy compared with standard diabetes and general populations. Acta Ophthalmol 90: e650–652. [DOI] [PubMed] [Google Scholar]
- Mason JO 3rd, Colagross CT, Haleman T, Fuller JJ, White MF, Feist RM, Emond TL & McGwin G Jr (2005): Visual outcome and risk factors for light perception and no light perception vision after vitrectomy for diabetic retinopathy. Am J Ophthalmol 140: 231–235. [DOI] [PubMed] [Google Scholar]
- McCannel CA, Berrocal AM, Holder GE, Kim SJ, Leonard BC, Rosen RB, Spaide RF & Sun JK (2018): Retina and vitreous basic and clinical science course. San Francisco, USA.
- Ostri C, Lux A, Lund‐Andersen H & la Cour M (2014): Long‐term results, prognostic factors and cataract surgery after diabetic vitrectomy: a 10‐year follow‐up study. Acta Ophthalmol 92: 571–576. [DOI] [PubMed] [Google Scholar]
- Schulze‐Bonsel K, Feltgen N, Burau H, Hansen L & Bach M (2006): Visual acuities "hand motion" and "counting fingers" can be quantified with the freiburg visual acuity test. Invest Ophthalmol Vis Sci 47: 1236–1240. [DOI] [PubMed] [Google Scholar]
- Smiddy WE & Feuer W (2004): Incidence of cataract extraction after diabetic vitrectomy. Retina 24: 574–581. [DOI] [PubMed] [Google Scholar]
- Smiddy WE, Feuer W, Irvine WD, Flynn HW Jr & Blankenship GW (1995): Vitrectomy for complications of proliferative diabetic retinopathy. Functional outcomes. Ophthalmology 102: 1688–1695. [DOI] [PubMed] [Google Scholar]
- Thompson JT (2004): The role of patient age and intraocular gas use in cataract progression after vitrectomy for macular holes and epiretinal membranes. Am J Ophthalmol 137: 250–257. [DOI] [PubMed] [Google Scholar]
- Thompson JT (2011): Advantages and limitations of small gauge vitrectomy. Surv Ophthalmol 56: 162–172. [DOI] [PubMed] [Google Scholar]
- Vaideanu D, Sandhu SS, Ling J, Richardson J & Steel DH (2014): Rate of diabetic vitrectomy in a defined geographical part of North East England. Ophthalmic Epidemiol 21: 178–183. [DOI] [PubMed] [Google Scholar]
- Vote BJ, Gamble GD & Polkinghorne PJ (2004): Auckland proliferative diabetic vitrectomy fellow eye study. Clin Exp Ophthalmol 32: 397–403. [DOI] [PubMed] [Google Scholar]
- WHO (2015): International classification of diseases and health related problems 10th revision (ICD‐10).
- Yau JW, Rogers SL, Kawasaki R et al. (2012): Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 35: 556–564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yorston D, Wickham L, Benson S, Bunce C, Sheard R & Charteris D (2008): Predictive clinical features and outcomes of vitrectomy for proliferative diabetic retinopathy. Br J Ophthalmol 92: 365–368. [DOI] [PubMed] [Google Scholar]
- Zaninetti M, Petropoulos IK & Pournaras CJ (2005): Proliferative diabetic retinopathy: vitreo‐retinal complications are often related to insufficient retinal photocoagulation. J Fr Ophtalmol 28: 381–384. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Fig. S1. Course of visual acuity before and after vitrectomy in decimals for both eyes.
Table S1. Predictive variables for low vision in both eyes, re‐PPV in the study eye, and PPV in the fellow eye, using Cox regression survival analysis.
Table S2. Comparison of study results in this study with previous diabetic vitrectomy studies.


