Abstract
Osteoporosis is a disease characterized by the degeneration of bone structure and decreased bone mass. Induced pluripotent stem cell-derived mesenchymal stem cells (iPSC-MSCs) have multiple advantages that make them ideal seed cells for bone regeneration, including high-level proliferation, multi-differentiation potential and favorable immune compatibility. Distal-less homeobox (DLX)3, an important member of the DLX family, serves a crucial role in osteogenic differentiation and bone formation. The present study aimed to evaluate the effects of DLX3 on the proliferation and osteogenic differentiation of human iPSC-MSCs. iPSC-MSCs were induced from iPSCs, and identified via flow cytometry. Alkaline phosphatase (ALP), Von Kossa, Oil Red O and Alcian blue staining methods were used to evaluate the osteogenic, adipogenic and chondrogenic differentiation of iPSC-MSCs. DLX3 overexpression plasmids were constructed and transfected into iPSC-MSCs to generate iPSC-MSC-DLX3. iPSC-MSC-GFP was used as the control. Reverse transcription-quantitative PCR (RT-qPCR) and western blotting were performed to measure the expression of DLX3 2 days after transfection. Subsequently, cell proliferation was assessed using a Cell Counting Kit-8 assay on days 1, 3, 5 and 7. RT-qPCR and western blotting were used to analyze osteogenic-related gene and protein expression levels on day 7. ALP activity and mineralized nodules were assessed via ALP staining on day 14. Statistical analysis was performed using an unpaired Student's t-test. Flow cytometry results demonstrated that iPSC-MSCs were positive for CD73, CD90 and CD105, but negative for CD34 and CD45. iPSC-MSC-DLX3 had significantly lower proliferation compared with iPSC-MSC-GFP on days 5 and 7 (P<0.05). mRNA expression levels of osteogenic markers, such as ALP, osteopenia (OPN), osteocalcin (OCN) and Collagen Type I (COL-1), were significantly increased in iPSC-MSC-DLX3 compared with iPSC-MSC-GFP on day 7 (P<0.05). Similarly, the protein expression levels of ALP, OCN, OPN and COL-1 were significantly increased in iPSC-MSC-DLX3 compared with iPSC-MSC-GFP on day 7 (P<0.05). The number of mineralized nodules in iPSC-MSC-DLX3 was increased compared with that in iPSC-MSC-GFP on day 14 (P<0.05). Thus, the present study demonstrated that DLX3 serves a negative role in proliferation, but a positive role in the osteogenic differentiation of iPSC-MSCs. This may provide novel insight for treating osteoporosis.
Keywords: distal-less homeobox 3, proliferation, osteogenic differentiation, induced pluripotent stem cell-derived mesenchymal stem cells
Introduction
Bone is in a dynamic balance between bone formation and bone resorption (1). With aging and after the menopause, an imbalance in bone resorption relative to formation results in osteoporosis (2). Osteoporosis is a common skeletal disease characterized by decreased bone mass, deterioration of bone microarchitecture and increased susceptibility to fractures. With the gradual aging of the population in many countries, osteoporosis has become a major global health concern (3). In the United States, osteoporosis is projected to affect nearly 14 million adults over the age of 50 by the year 2020 (4). Worldwide, ~200 million women have osteoporosis (5).
Skeletal development is regulated by numerous homeodomain proteins, including the distal-less (DLX) family, which play an important role in the development of bone tissue (6). DLX family has six transcription factor members, known as DLX1-6 (7). DLX3 is mapped to 17q21.33 and has a notable impact on the development of organs, including glands, teeth and hair follicles (8,9). In humans, mutation of DLX3 can cause tricho-dento-osseous syndrome, which is characterized by hypoplasia of the hair, enamel and dentin, as well as a high bone density (8).
Bone marrow mesenchymal stem cells (BMSCs) are a type of post-natal stem cell with the potential to differentiate into different cells, such as osteoblasts, chondrocytes and adipocytes (10). Bone formation depends on the osteogenic potential and proliferative capability of BMSCs, but the proliferative ability of BMSCs is limited, which has restricted their application in clinical settings (10,11). Therefore, there is a need for an improved cell source to overcome the limitations of BMSCs (11). Induced pluripotent stem cell-derived mesenchymal stem cells (iPSC-MSCs), which are an unlimited source of MSCs, exhibit favorable proliferation, cell viability, and osteo- and chondrogenic differentiation potential, and thus could be used to meet the requirement of bone regeneration (11–15). Previously, several cell culture supplements have been utilized to derive MSCs from iPSC (11). The most commonly used supplements are synthetic coatings, such as PMEDSAH (15), fibrillar collagen (16), extracellular matrix Geltrex (17) and fibronectin (17). Moreover, a few small molecules, such as SB431542 (TGF-β pathway inhibitor) (18) and CHIR99021 (GSK inhibitor) (19) have been applied to obtain iPSC-MSCs. In the current study, iPSC-MSCs were derived using Geltrex, which retains the complete osteogenesis function of the cells (12–14,17).
A previous study revealed that overexpression of DLX3 enhances the osteogenic differentiation of human BMSCs (20). Similar results have also been reported in human dental pulp cells (DPCs), in which overexpressed DLX3 decreases cell proliferation and increases mRNA expression levels of osteo- and chondrogenic markers, including alkaline phosphatase (ALP), dentin sialophosphoprotein (DSPP) and dentin matrix acidic phosphoprotein 1 (DMP1) (21). Thus, the DLX3 gene is crucial for osteogenic differentiation. However, the role of DLX3 gene in regulating iPSC-MSCs is yet to be fully elucidated.
In the present study, DLX3-overexpression iPSC-MSCs (iPSC-MSC-DLX3) were constructed, the expression of DLX3 in iPSC-MSCs and iPSC-MSC-DLX3 was examined by reverse transcription-quantitative PCR (RT-qPCR) and western blotting, and then the effects of the DLX3 gene on osteogenic differentiation of these cells was evaluated.
Materials and methods
Derivation of iPSC-MSCs
The experiment protocol of the present study was approved by the Ethical Review Committee of Jinan University (approval no. 2015-045). Human iPSC line was obtained from the South China Institute for Stem Cell Biology and Regenerative Medicine Group of the Chinese Academy of Sciences. iPSCs were cultured in mTeSR1 medium (Stemcell Technologies, Inc.) on Geltrex™ LDEV-Free Reduced Growth Factor Basement Membrane Matrix (Gibco; Thermo Fisher Scientific, Inc.) coated dishes for 5 days. Then, the medium was replaced with DMEM (Gibco; Thermo Fisher Scientific, Inc.) containing 10% FBS (Gibco; Thermo Fisher Scientific, Inc.), 2 mM L-glutamine, 1% penicillin/streptomycin and 0.1 mM non-essential amino acids (Gibco; Thermo Fisher Scientific, Inc.). When these cells reached 80% confluence, they were passaged. When most of the cells presented spindle-like morphology, they were collected and applied for subsequent experiments.
Flow cytometry analysis
Flow cytometry was used to evaluate surface markers of iPSC-MSCs. Briefly, the cells were harvested, and then 3% BSA (Gibco; Thermo Fisher Scientific, Inc.) was used to block non-specific antigens on the cell surface at 37°C for 30 min. Subsequently, the cells were incubated with monoclonal antibodies (all purchased from BioLegend, Inc.) against CD73 (cat. no. 344005), CD90 (cat. no. 328113), CD105 (cat. no. 323207), CD34 (cat. no. 343607) and CD45 (cat. no. 368511). Then, the cells were washed three times with BSA to remove non-specific antibodies. A Guava® easyCyte™ flow cytometer and Guava® Suite Software 3.4 (both EMD Millipore) were used to analyze surface antigens.
Differentiation capability analysis
The osteogenic, adipogenic and chondrogenic differentiation capabilities of iPSC-MSCs were analyzed. To study osteogenic differentiation, iPSC-MSCs at 1×104/well density were seeded on 6-well plates (Corning, Inc.) and incubated with 2 ml StemPro™ Osteogenesis Differentiation medium (Gibco; Thermo Fisher Scientific, Inc.) at 37°C for 3 weeks. The Osteogenesis Differentiation medium was replaced every 3 days. The cells were fixed with 4% paraformaldehyde for 30 min at room temperature. The ALP activity of iPSC-MSCs was assayed using an ALP staining kit (Sigma-Aldrich; Merck KGaA) according to the manufacturer's instructions, and the calcified matrix deposition was detected using a Von Kossa staining kit (Nanjing Jiancheng Bioengineering Institute) according to the manufacturer's instructions and observed under a light microscope (Zeiss Axio Observer.Z1; Carl Zeiss AG).
In order to study adipogenic differentiation, iPSC-MSCs at 1×104/well density were seeded on 6-well plates and incubated with 2 ml StemPro™ Adipogenesis Differentiation medium (Gibco; Thermo Fisher Scientific, Inc.) at 37°C for 3 weeks. Adipogenesis Differentiation medium was replaced every 3 days, and subsequently the cells were assayed with an Oil Red O kit (Nanjing Jiancheng Bioengineering Institute) for 15 min at room temperature.
To study chondrogenic differentiation, iPSC-MSCs at 1×104/well density were seeded on 6-well plates and incubated with 2 ml StemPro™ Chondrogenesis Differentiation medium (Gibco; Thermo Fisher Scientific, Inc.) at 37°C for 3 weeks. The Chondrogenesis Differentiation medium was replaced every 3 days, and subsequently the cells were assayed with an Alcian blue kit (Nanjing Jiancheng Bioengineering Institute) for 30 min at room temperature.
Lentiviral plasmid transfection
Human DLX3 gene primers (PrimerBank ID:38327640c1, Table I) were designed according to PrimerBank online (The Massachusetts General Hospital) (22). Subsequently, PCR amplification was performed according to our previous study (23). The amplified DLX3 primers were treated with EcoRI and BamHI, and then combined into the lentivirus vector pCDH-CMV-MCS-EF1-copGFP (pCDH; System Biosciences, LLC) to construct the recombinant plasmid pCDH-DLX3. The target gene was transduced into 4×105 293FT cells per well in 6 well plate using a combination of enveloping plasmid, packaging plasmid and recombinant lentiviral plasmid (3rd generation system; Cyagen Biosciences, Inc.) at 37°C. Then, 48 h later, 293FT cells were centrifuged at 10,000 × g at 4°C for 4 h and the supernatant was collected to infect the 3rd passage of iPSC-MSCs (iPSC-MSC-DLX3) at 37°C. The MOI was 50, and concentration of Polybrene was 5 µg/ml. Cells were cultured at 37°C in DMEM containing 10% FBS with 1 µg/ml puromycin for 3 days in order to select puromycin-resistant cells and 0.25 µg/ml puromycin was used for maintenance. Similarly, iPSC-MSCs transfected with blank lentivirus vector (iPSC-MSC-GFP) were generated and used as the control. After 2 days, reverse transcription-quantitative PCR (RT-qPCR) and western blot analysis were performed to evaluate the expression of DLX3 in the two groups.
Table I.
Primers used for reverse transcription-quantitative PCR.
| Gene | Primer sequences (5→3) | Size (bp) |
|---|---|---|
| DLX3 | F: TACCCTGCCCGAGTCTTCTG | 111 |
| R: TGGTGGTAGGTGTAGGGGTTC | ||
| ALP | F: ACCACCACGAGAGTGAACCA | 79 |
| R: CGTTGTCTGAGTACCAGTCCC | ||
| OPN | F: CTCCATTGACTCGAACGACTC | 230 |
| R: CAGGTCTGCGAAACTTCTTAGAT | ||
| OCN | F: CACTCCTCGCCCTATTGGC | 112 |
| R: CCCTCCTGCTTGGACACAAAG | ||
| COL-1 | F: GAGGGCCAAGACGAAGACATC | 140 |
| R: CAGATCACGTCATCGCACAAC | ||
| GAPDH | F: GGAGCGAGATCCCTCCAAAAT | 197 |
| R: GGCTGTTGTCATACTTCTCATGG |
DLX3, distal-less homeobox 3; ALP, alkaline phosphatase; OPN, osteopontin; OCN, osteocalcin; COL-1, Collagen Type I; F, forward; R, reverse.
Cell proliferation assay
Proliferation of iPSC-MSC-GFP and iPSC-MSC-DLX3 was assessed using Cell Counting Kit-8 (CCK-8; Dojindo Molecular Technologies, Inc.). On the first day, cells were seeded on 96-well plates at 2×103 cells/well, and then cultured with DMEM plus 10% FBS. Cell viability was evaluated on day 1, 3, 5 and 7 post-transfection. Optical density (OD) at 450 nm was recorded using an enzyme immunoassay reader (Bio-Rad Laboratories, Inc.).
RT-qPCR analysis
Total RNA was isolated from iPSC-MSC-GFP and iPSC-MSC-DLX3 using TRIzol® reagent (Invitrogen; Thermo Fisher Scientific, Inc.) on day 7. NanoDrop™ 2000 system (Thermo Fisher Scientific, Inc.) was used to test RNA concentrations. The extracted RNA was reverse-transcribed into cDNA using iScript™ gDNA Clear cDNA Synthesis kit (Invitrogen; Thermo Fisher Scientific, Inc.) following the manufacturer's instructions. qPCR analysis was operated using PowerUp SYBR-Green Master Mix (Invitrogen; Thermo Fisher Scientific, Inc.) and measured by spectrofluorimetric iQ5 Thermal iCycler (Bio-Rad Laboratories, Inc.) according to the manufacturer's instructions. The primer sequences of DLX3, ALP, osteopontin (OPN), osteocalcin (OCN), Collagen Type I (COL-1) and GAPDH are presented in Table I. The relative expression of target genes was determined using the 2−ΔΔCq method (24) and normalized to GAPDH. Each sample was tested three times.
Western blot analysis
A total of 7 days after transfection, iPSC-MSC-GFP and iPSC-MSC-DLX3 were rinsed with PBS and lysed in 0.1 ml RIPA buffer containing 10 mg/ml proteinase inhibitor PMSF (Invitrogen; Thermo Fisher Scientific, Inc.) on ice for 30 min. The lysed cells were centrifuged at 10,000 × g for 10 min at 4°C, and the supernatant was collected. During electrophoresis, 20 µg target total protein/lane was separated via SDS-PAGE on a 10% gel (Beyotime Institute of Biotechnology), which were subsequently transferred to PVDF membranes (Thermo Fisher Scientific, Inc.) at 200 mA for 2 h. PVDF membranes were blocked using 5% non-fat milk with TBS with 0.1% Tween-20 at room temperature for 2 h, and then incubated with the primary antibodies at 4°C overnight. The primary antibodies used were as follows: DLX3 (1:500; cat. no. ab64953; Abcam), ALP (1:500; cat. no. ab83259; Abcam), OPN (1:500; cat. no. ab8448; Abcam), OCN (1:500; cat. no. ab93876; Abcam), COL-1 (1:500; cat. no. ab34710; Abcam) and GAPDH (1:2,500; cat. no. ab9485; Abcam). Then, PVDF membranes were incubated with a secondary antibody (1:2,500; cat. no. ab97051; Abcam) at 37°C for 2 h. GAPDH was used as the control. The blotting results were visualized with chemiluminescent western blotting detection reagents (Pierce; Thermo Fisher Scientific, Inc.) and measured by Image-Pro Plus version 6.0 (Media Cybernetics, Inc.).
ALP staining and mineralized nodule counting
The transfected cells were seeded on 6-well plates at an initial density of 3×104 cells/well and cultured for 14 days to 80% confluence in DMEM containing 10% FBS. The cells were fixed with 4% paraformaldehyde for 30 min at room temperature. The ALP Staining kit (Nanjing Jiancheng Bioengineering Institute) was used to stain cells for 30 min at room temperature on day 14 after transfection. In total, three randomized observation views were selected, and the number of mineralized nodules was counted under a microscope (Zeiss Axio Observer.Z1; Carl Zeiss AG).
Statistical analysis
SPSS 20.0 software (IBM Corp.) was used to analyze the data, which are presented as the mean ± SD. Statistical significance was calculated using an unpaired Student's t-test. P<0.05 was considered to indicate a statistically significant difference.
Results
Characterization of iPSC-MSCs
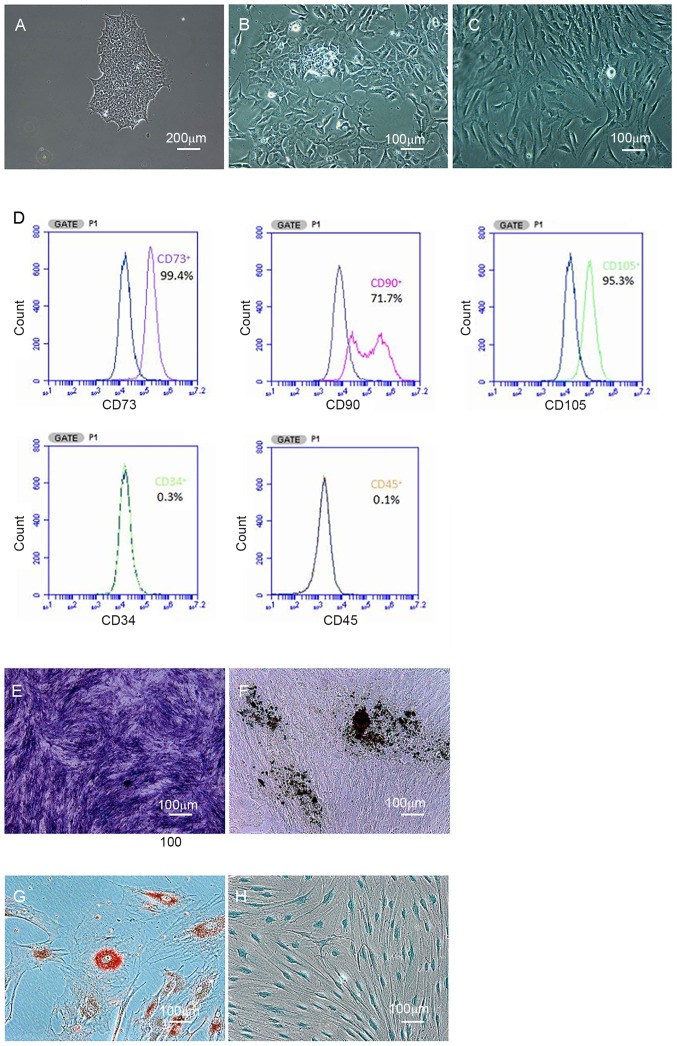
As illustrated in Fig. 1A, human iPSCs presented as packed clones with clear margins. After culturing in MSC medium for 14 days, iPSCs were gradually induced into MSCs. Under the microscope, borders of the colonies were eliminated (Fig. 1B), and cells exhibited homogeneous spindle-like morphology (Fig. 1C). The iPSC-derived cells were positive for CD73 (99%), CD90 (71%) and CD105 (95%), and negative for CD34 and CD45, as determined by flow cytometric analysis (Fig. 1D).
Figure 1.
Generation and identification of iPSC-MSCs. (A-C) Generation of iPSC-MSCs. (A) Scale bar, 200 µm; (B and C) scale bar, 100 µm. (D) Flow cytometric analysis of iPSC-MSCs. (E) Alkaline phosphatase staining. (F) Osteogenesis was detected using Von Kossa staining. (G) Adipogenesis was detected using Oil Red O staining. (H) Chondrogenesis was detected using Alcian blue staining (scale bar, 100 µm). iPSC-MSC, induced pluripotent stem cell-derived mesenchymal stem cell.
After osteogenic induction, iPSC-derived cells displayed positive ALP staining (Fig. 1E) and Von Kossa staining (Fig. 1F). Additionally, iPSC-derived cells had positive Oil Red O and Alcian blue staining after adipogenic (Fig. 1G) and chondrogenic induction (Fig. 1H). Thus, iPSCs were successfully induced into iPSC-MSCs.
Expression of DLX3 in iPSC-MSCs
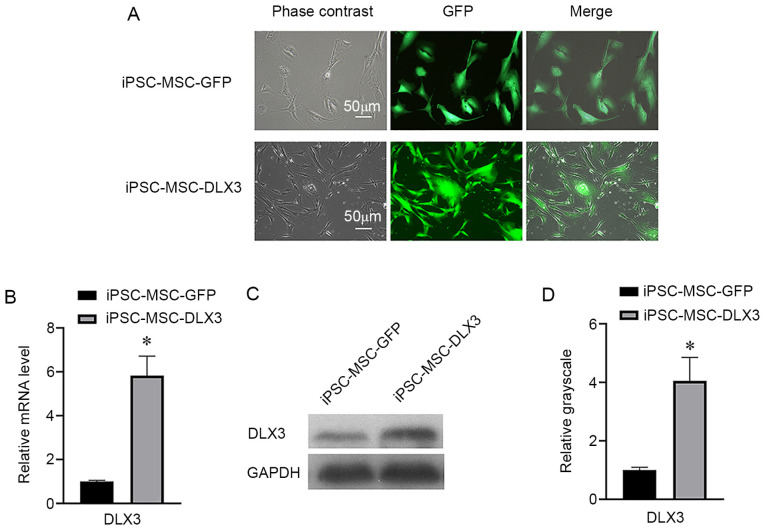
Both iPSC-MSC-GFP and iPSC-MSC-DLX3 groups had a GFP expression percentage of ~100% (Fig. 2A). RT-qPCR results indicated that the relative mRNA expression of DLX3 in iPSC-MSC-GFP and iPSC-MSC-DLX3 was 1.00±0.05 and 5.84±0.89, respectively (Fig. 2B). Moreover, the western blotting results demonstrated that the relative DLX3 expression in iPSC-MSC-GFP and iPSC-MSC-DLX3 was 1.00±0.10 and 4.05±0.81, respectively (Fig. 2C and D). Based on these results, it was suggested that the DLX3 gene was successfully transfected into iPSC-MSCs.
Figure 2.
Lentiviral transfection efficiency. (A) GFP expression percentage in the iPSC-MSC-GFP and iPSC-MSC-DLX3 groups (scale bar, 50 µm). (B) Reverse transcription-quantitative PCR analysis of DLX3 gene expression. (C and D) Western blot analysis of DLX3 protein expression. *P<0.05 vs. iPSC-MSC-GFP. iPSC-MSC, induced pluripotent stem cell-derived mesenchymal stem cell; DLX3, distal-less homeobox 3.
DLX3 regulates proliferation of iPSC-MSCs
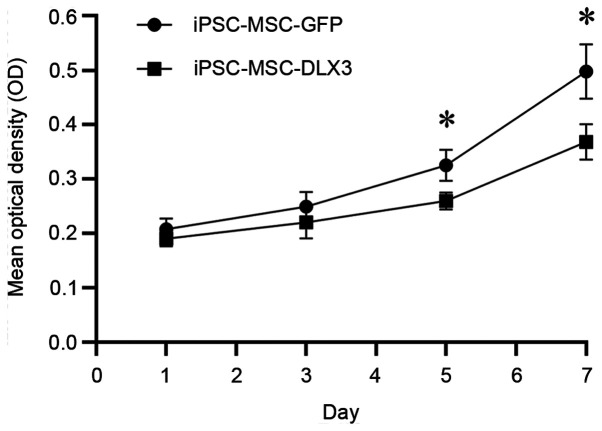
The OD values of iPSC-MSC-GFP on day 1, 3, 5 and 7 were 0.21±0.02, 0.25±0.03, 0.33±0.03 and 0.50±0.05, while the OD values of iPSC-MSC-DLX3 on day 1, 3, 5 and 7 were 0.19±0.01, 0.22±0.03, 0.26±0.02 and 0.37±0.03, respectively (Fig. 3). There was no significant difference in cell numbers between the two groups on days 1 and 3 (P>0.05). However, iPSC-MSC-DLX3 demonstrated significantly lower proliferative activity compared with the iPSC-MSC-GFP group on days 5 and 7 (P<0.05).
Figure 3.
Cell proliferation. Cell proliferation rate was significantly reduced in the iPSC-MSC-DLX3 group compared with the rate observed intheiPSC-MSC-GFP group on days 5 and 7. *P<0.05 vs. iPSC-MSC-DLX3. iPSC-MSC, induced pluripotent stem cell-derived mesenchymal stem cell; DLX3, distal-less homeobox 3.
Expression of osteogenesis related genes and proteins
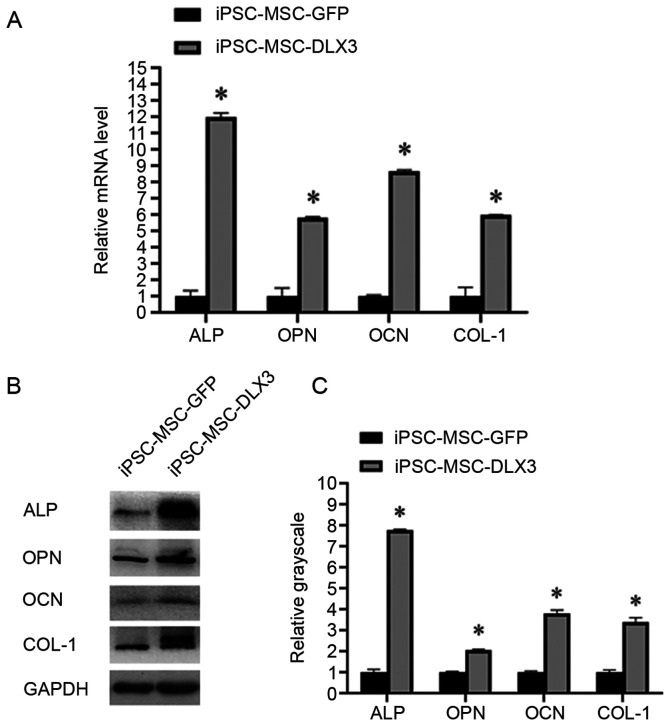
After 7 days of transfection of iPSC-MSC-GFP and iPSC-MSC-DLX3, the relative expression levels of ALP were 1.00±0.33 and 11.99±0.24, those of OPN were 1.00±0.49 and 5.80±0.07, those of OCN were 1.00±0.06 and 8.64±0.11, those of COL-1 were 1.00±0.53 and 5.98±0.02. The results indicated that the mRNA expression levels of osteogenic markers in iPSC-MSC-DLX3 were significantly higher compared with those in iPSC-MSC-GFP (P<0.05; Fig. 4A).
Figure 4.
mRNA and protein expression of osteogenic differentiation-related genes on day 7. (A) Reverse transcription-quantitative PCR analysis of osteogenic-related gene expression. (B and C) Western blot analysis of osteogenic-related protein expression. *P<0.05 vs. iPSC-MSC-GFP. iPSC-MSC, induced pluripotent stem cell-derived mesenchymal stem cell; DLX3, distal-less homeobox 3; ALP, alkaline phosphatase; OPN, osteopenia; OCN, osteocalcin; COL-1, Collagen Type I.
Similarly, the relative expression levels of ALP were 1.00±0.13 and 7.77±0.05, those of OPN were 1.00±0.04 and 2.05±0.04, those of OCN were 1.00±0.05 and 3.79±0.17, and those of COL-1 were 1.00±0.11 and 3.38±0.22. The expression levels of the osteogenic proteins in iPSC-MSC-DLX3 were significantly increased compared with those in iPSC-MSC-GFP (P<0.05; Fig. 4B and C).
ALP activity and mineralization
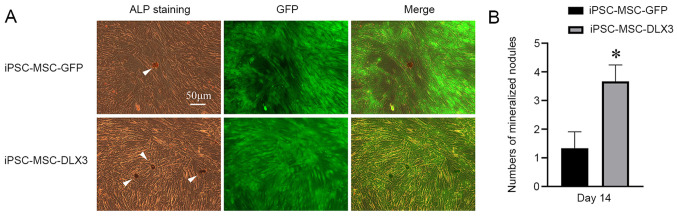
As presented in Fig. 5A, ALP was stained as a golden color. ALP staining in the iPSC-MSC-DLX3 group was increased and brighter compared with that in the iPSC-MSC-GFP group. Mineralized nodules were stained as black/brown in color. The number of mineralized nodules in iPSC-MSC-DLX3 was significantly higher compared with that in iPSC-MSC-GFP (P<0.05; Fig. 5B).
Figure 5.
ALP activity and mineralized nodules. (A) ALP was stained as golden, and mineralized nodules were stained as black/brown (marked with arrow). Scale bar, 50 µm. (B) The number of mineralized nodules in the iPSC-MSC-DLX3 group was higher than that in the iPSC-MSC-GFP group. *P<0.05 vs. iPSC-MSC-GFP. iPSC-MSC, induced pluripotent stem cell-derived mesenchymal stem cell; DLX3, distal-less homeobox 3; ALP, alkaline phosphatase.
Discussion
iPSC-MSCs have been considered as a novel cell resource for bone tissue engineering (11–14,17). The present study used Geltrex as a cell culture coating to generate MSCs from iPSCs, as Geltrex can enhance cell attachment and improve the efficacy to generate iPSC-MSCs (17). The current results suggested that iPSC-MSCs presented a spindle-like morphology, which was consistent with a previous study on BMSCs (17). Moreover, the current results demonstrated that iPSC-MSC attained 99% positive for CD73, 71% positive for CD90 and 95% positive for CD105, which were typical cell markers comparable to BMSCs (25).
While iPSC-MSCs have similar surface markers as BMSCs, their gene expression profile differs (26), and iPSC-MSCs are genetically related to vascular progenitor cells. Additionally, iPSC-MSCs have similar osteogenic and chondrogenic differentiation properties as BMSCs, but their adipogenic differentiation potential is significantly lower compared with that of BMSCs (12,26). Therefore, iPSC-MSCs are not entirely equivalent to BMSCs. Consistent with the aforementioned findings, the present results indicated that iPSC-MSCs had a satisfactory osteogenic and chondrogenic differentiation potential, which suggested that iPSC-MSCs are appropriate for bone tissue engineering.
At present, the role of DLX3 on cell proliferation has not been sufficiently investigated. A previous research group pointed out that stable overexpression of DLX3 gene inhibited the proliferation of human DPCs by inactivating the canonical Wnt pathway (21,27). In the current study, a CCK-8 assay was used to assess the proliferation of iPSC-MSCs after transfection of DLX3 gene on days 1, 3, 5 and 7. Compared with the control group, the iPSC-MSC-DLX3 group demonstrated a significant decreased proliferative rate on days 5 and 7. These results were consistent with the reported studies regarding human DPCs.
DLX3, expressed in osteo-/odontogenic lineages, is a crucial transcription factor for osteo-/odontogenic differentiation, mineralization and skeletal development (21,28–30). In BMSCs, transfection of DLX3 gene promotes the expression of ALP, Runt-related transcription factor 2 (RUNX2), OSX and OCN, as well as the formation of calcified matrix (20). Overexpression of DLX3 stimulates osteoprogenitor cells to express bone matrix proteins, such as COL-1, bone sialoprotein, OCN and ALP (9). Moreover, DLX3 is strongly expressed in differentiating and differentiated osteoblasts, and particularly upregulates OCN and COL-1 (31). An in vitro study also revealed that DLX3 could promote odontoblastic differentiation of human DPCs, and could increase the expression levels of ALP, DSPP, DMP1 and Nestin (21). However, one study provided the opposite evidence in vivo, and reported that neural crest deletion of DLX3 increased bone formation and mineralization in craniofacial bones, which suggested an inhibitory role for DLX3 in osteoblastic differentiation (32). RNA sequencing and chromatin immunoprecipitation-Seq analyses have further demonstrated that DLX3 regulates transcription factors crucial for bone formation, such as DLX5, DLX6, RUNX2 and Sp7, as well as genes important for mineral deposition (bone sialoprotein 2, ectonucleotide pyrophosphatase/phosphodiesterase family member 1 and matrix extracellular phosphoglycoprotein) and bone turnover (tumor necrosis factor receptor superfamily member 11B) (33). Furthermore, with the knockdown of DLX3, researchers have observed increased occupancy of DLX5, as well as enhanced and earlier occupancy of RUNX2 on the bone-specific osteocalcin promoter (33). Taken together, these findings provide evidence that DLX3 attenuates bone mass accrual to support bone homeostasis via osteogenic gene pathway regulation. In the present study, it was identified that DLX3 overexpression significantly upregulated osteogenic differentiation of iPSC-MSC by activating ALP, OCN, OPN and COL-1 in vitro. In addition, iPSC-MSC-DLX3 formed significantly more mineralized nodules compared with the control group. These results were consistent with a number of previous studies (9,20,21,31).
Osteoporosis (OP) is a bone metabolic disease that is characterized by the degeneration of bone structure and decreased bone mass (34). OP occurs in >1/3 of women and 1/5 of men >50 years old, and affects the health and lives of these individuals (35). The primary mechanism of OP is the dysregulation of the dynamic balance between bone formation and resorption, resulting in higher bone resorption than bone formation, which may lead to bone metabolism disorder (36). The present results suggested that DLX3 was a positive transcription factor in regulating osteogenic differentiation. Previous studies have reported that some osteogenic stimulators and transcription factors can induce the protein expression of DLX3. It has been revealed that DLX3 is a novel target of Estrogen receptor α (ER-α), and ER-α regulates osteoblast differentiation via the modulation of DLX3 expression and/or interaction with DLX3 (30). DLX3 is also a novel target of protein kinase A (PKA), and PKA mediates bone morphogenetic protein signaling during osteoblast differentiation, at least in part, by phosphorylating DLX3 and modulating the protein stability and function of DLX3 (37). The present results may facilitate the development of novel strategies for the targeted therapy of OP.
However, the present study has some limitations. It was assumed that multilineage induction experiments were enough to demonstrate the successful induction of human iPSCs into iPSC-MSCs, but the relative marker genes were not analyzed pre/post-osteogenic and chondrogenic induction. Therefore, it is hoped that these limitations can be improved in future studies.
In conclusion, the present study demonstrated that DLX3 exerted a positive role in regulating the osteogenesis of iPSC-MSCs. However, the specific underlying mechanism of DLX3 affecting osteogenic differentiation in iPSC-MSCs is yet to be fully elucidated. Further investigations, both in vitro and in vivo, are required to confirm its effect on osteogenic differentiation. Therefore, future studies will focus on the specific mechanism of DLX3 affecting osteogenic differentiation of iPSC-MSCs. Moreover, in vivo assays of transgenic DLX3 should be performed to further confirm its effect on osteogenesis.
Acknowledgements
Not applicable.
Funding Statement
The present study was supported by the Guangdong Basic and Applied Basic Research Foundation (grant no. 2020A1515010239), National Natural Science Foundation of China (grant no. 81500825), Medical Scientific Research Foundation of Guangdong Province (grant no. A2015423), Special Fund for Public Welfare Research and Capacity Building of Guangdong Province (grant no. 2014A020212211), and Science and Technology Program of Guangzhou (grant no. 201607010205).
Funding
The present study was supported by the Guangdong Basic and Applied Basic Research Foundation (grant no. 2020A1515010239), National Natural Science Foundation of China (grant no. 81500825), Medical Scientific Research Foundation of Guangdong Province (grant no. A2015423), Special Fund for Public Welfare Research and Capacity Building of Guangdong Province (grant no. 2014A020212211), and Science and Technology Program of Guangzhou (grant no. 201607010205).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
WZ and RL designed the study, prepared the figures and drafted the manuscript. JL and QL performed the experiments. YL conceptualized the study and drafted and revised the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The present study was approved by the Ethical Review Committee of Jinan University (approval no. 2015-045).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Feng X, McDonald JM. Disorders of bone remodeling. Annu Rev Pathol. 2011;6:121–145. doi: 10.1146/annurev-pathol-011110-130203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Raisz LG. Pathogenesis of osteoporosis: Concepts, conflicts, and prospects. J Clin Invest. 2005;115:3318–3325. doi: 10.1172/JCI27071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cummings SR, Melton LJ. Epidemiology and outcomes of osteoporotic fractures. Lancet. 2002;359:1761–1767. doi: 10.1016/S0140-6736(02)08657-9. [DOI] [PubMed] [Google Scholar]
- 4.Genant HK, Cooper C, Poor G, Reid I, Ehrlich G, Kanis J, Nordin BE, Barrett-Connor E, Black D, Bonjour JP, et al. Interim report and recommendations of the World Health Organization Task-Force for Osteoporosis. Osteoporos Int. 1999;10:259–264. doi: 10.1007/s001980050224. [DOI] [PubMed] [Google Scholar]
- 5.Aspray TJ, Hill TR. Osteoporosis and the Ageing Skeleton. Subcell Biochem. 2019;91:453–476. doi: 10.1007/978-981-13-3681-2_16. [DOI] [PubMed] [Google Scholar]
- 6.Hassan MQ, Javed A, Morasso MI, Karlin J, Montecino M, van Wijnen AJ, Stein GS, Stein JL, Lian JB. Dlx3 transcriptional regulation of osteoblast differentiation: Temporal recruitment of Msx2, Dlx3, and Dlx5 homeodomain proteins to chromatin of the osteocalcin gene. Mol Cell Biol. 2004;24:9248–9261. doi: 10.1128/MCB.24.20.9248-9261.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhao N, Zeng L, Liu Y, Han D, Liu H, Xu J, Jiang Y, Li C, Cai T, Feng H, et al. DLX3 promotes bone marrow mesenchymal stem cell proliferation through H19/miR-675 axis. Clin Sci (Lond) 2017;131:2721–2735. doi: 10.1042/CS20171231. [DOI] [PubMed] [Google Scholar]
- 8.Li Y, Han D, Zhang H, Liu H, Wong S, Zhao N, Qiu L, Feng H. Morphological analyses and a novel de novo DLX3 mutation associated with tricho-dento-osseous syndrome in a Chinese family. Eur J Oral Sci. 2015;123:228–234. doi: 10.1111/eos.12197. [DOI] [PubMed] [Google Scholar]
- 9.Choi SJ, Song IS, Ryu OH, Choi SW, Hart PS, Wu WW, Shen RF, Hart TC. A 4 bp deletion mutation in DLX3 enhances osteoblastic differentiation and bone formation in vitro. Bone. 2008;42:162–171. doi: 10.1016/j.bone.2007.08.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lin H, Sohn J, Shen H, Langhans MT, Tuan RS. Bone marrow mesenchymal stem cells: Aging and tissue engineering applications to enhance bone healing. Biomaterials. 2019;203:96–110. doi: 10.1016/j.biomaterials.2018.06.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sabapathy V, Kumar S. hiPSC-derived iMSCs: NextGen MSCs as an advanced therapeutically active cell resource for regenerative medicine. J Cell Mol Med. 2016;20:1571–1588. doi: 10.1111/jcmm.12839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kang R, Zhou Y, Tan S, Zhou G, Aagaard L, Xie L, Bünger C, Bolund L, Luo Y. Mesenchymal stem cells derived from human induced pluripotent stem cells retain adequate osteogenicity and chondrogenicity but less adipogenicity. Stem Cell Res Ther. 2015;6:144. doi: 10.1186/s13287-015-0137-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Xie J, Peng C, Zhao Q, Wang X, Yuan H, Yang L, Li K, Lou X, Zhang Y. Osteogenic differentiation and bone regeneration of iPSC-MSCs supported by a biomimetic nanofibrous scaffold. Acta Biomater. 2016;29:365–379. doi: 10.1016/j.actbio.2015.10.007. [DOI] [PubMed] [Google Scholar]
- 14.Jungbluth P, Spitzhorn LS, Grassmann J, Tanner S, Latz D, Rahman MS, Bohndorf M, Wruck W, Sager M, Grotheer V, et al. Human iPSC-derived iMSCs improve bone regeneration in mini-pigs. Bone Res. 2019;7:32. doi: 10.1038/s41413-019-0069-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Villa-Diaz LG, Brown SE, Liu Y, Ross AM, Lahann J, Parent JM, Krebsbach PH. Derivation of mesenchymal stem cells from human induced pluripotent stem cells cultured on synthetic substrates. Stem Cells. 2012;30:1174–1181. doi: 10.1002/stem.1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Liu Y, Goldberg AJ, Dennis JE, Gronowicz GA, Kuhn LT. One-step derivation of mesenchymal stem cell (MSC)-like cells from human pluripotent stem cells on a fibrillar collagen coating. PLoS One. 2012;7:e33225. doi: 10.1371/journal.pone.0033225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.TheinHan WW, Liu J, Tang M, Chen W, Cheng L, Xu HH. Induced pluripotent stem cell-derived mesenchymal stem cell seeding on biofunctionalized calcium phosphate cements. Bone Res. 2013;1:371–384. doi: 10.4248/BR201304008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chen YS, Pelekanos RA, Ellis RL, Horne R, Wolvetang EJ, Fisk NM. Small molecule mesengenic induction of human induced pluripotent stem cells to generate mesenchymal stem/stromal cells. Stem Cells Transl Med. 2012;1:83–95. doi: 10.5966/sctm.2011-0022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Meng X, Su RJ, Baylink DJ, Neises A, Kiroyan JB, Lee WY, Payne KJ, Gridley DS, Wang J, Lau KH, et al. Rapid and efficient reprogramming of human fetal and adult blood CD34+ cells into mesenchymal stem cells with a single factor. Cell Res. 2013;23:658–672. doi: 10.1038/cr.2013.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sun S, Yu M, Fan Z, Yeh IT, Feng H, Liu H, Han D. DLX3 regulates osteogenic differentiation of bone marrow mesenchymal stem cells via Wnt/β-catenin pathway mediated histone methylation of DKK4. Biochem Biophys Res Commun. 2019;516:171–176. doi: 10.1016/j.bbrc.2019.06.029. [DOI] [PubMed] [Google Scholar]
- 21.Li X, Yang G, Fan M. Effects of homeobox gene distal-less 3 on proliferation and odontoblastic differentiation of human dental pulp cells. J Endod. 2012;38:1504–1510. doi: 10.1016/j.joen.2012.07.009. [DOI] [PubMed] [Google Scholar]
- 22.Spandidos A, Wang X, Wang H, Seed B. PrimerBank: A resource of human and mouse PCR primer pairs for gene expression detection and quantification. Nucleic Acids Res. 2010;38(Suppl 1):D792–D799. doi: 10.1093/nar/gkp1005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhang W, Zhang X, Li J, Zheng J, Hu X, Xu M, Mao X, Ling J. Foxc2 and BMP2 induce osteogenic/odontogenic differentiation and mineralization of human stem cells from Apical Papilla. Stem Cells Int. 2018;2018:2363917. doi: 10.1155/2018/2363917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 25.Zou L, Luo Y, Chen M, Wang G, Ding M, Petersen CC, Kang R, Dagnaes-Hansen F, Zeng Y, Lv N, et al. A simple method for deriving functional MSCs and applied for osteogenesis in 3D scaffolds. Sci Rep. 2013;3:2243. doi: 10.1038/srep02243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xu M, Shaw G, Murphy M, Barry F. Induced pluripotent stem cell-derived mesenchymal stromal cells are functionally and genetically different from bone marrow-derived mesenchymal stromal cells. Stem Cells. 2019;37:754–765. doi: 10.1002/stem.2993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhan Y, Li X, Gou X, Yuan G, Fan M, Yang G. Dlx3 inhibits the proliferation of human dental pulp cells through inactivation of canonical wnt/beta-catenin signaling pathway. Front Physiol. 2018;9:1637. doi: 10.3389/fphys.2018.01637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Beanan MJ, Sargent TD. Regulation and function of Dlx3 in vertebrate development. Dev Dyn. 2000;218:545–553. doi: 10.1002/1097-0177(2000)9999:9999<::AID-DVDY1026>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 29.Ghoul-Mazgar S, Hotton D, Lézot F, Blin-Wakkach C, Asselin A, Sautier JM, Berdal A. Expression pattern of Dlx3 during cell differentiation in mineralized tissues. Bone. 2005;37:799–809. doi: 10.1016/j.bone.2005.03.020. [DOI] [PubMed] [Google Scholar]
- 30.Lee SH, Oh KN, Han Y, Choi YH, Lee KY. Estrogen Receptor α Regulates Dlx3-Mediated Osteoblast Differentiation. Mol Cells. 2016;39:156–162. doi: 10.14348/molcells.2016.2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Li H, Marijanovic I, Kronenberg MS, Erceg I, Stover ML, Velonis D, Mina M, Heinrich JG, Harris SE, Upholt WB, et al. Expression and function of Dlx genes in the osteoblast lineage. Dev Biol. 2008;316:458–470. doi: 10.1016/j.ydbio.2008.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Duverger O, Isaac J, Zah A, Hwang J, Berdal A, Lian JB, Morasso MI. In vivo impact of Dlx3 conditional inactivation in neural crest-derived craniofacial bones. J Cell Physiol. 2013;228:654–664. doi: 10.1002/jcp.24175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Isaac J, Erthal J, Gordon J, Duverger O, Sun HW, Lichtler AC, Stein GS, Lian JB, Morasso MI. DLX3 regulates bone mass by targeting genes supporting osteoblast differentiation and mineral homeostasis in vivo. Cell Death Differ. 2014;21:1365–1376. doi: 10.1038/cdd.2014.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sözen T, Özışık L, Başaran NC. An overview and management of osteoporosis. Eur J Rheumatol. 2017;4:46–56. doi: 10.5152/eurjrheum.2016.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zou Z, Liu W, Cao L, Liu Y, He T, Peng S, Shuai C. Advances in the occurrence and biotherapy of osteoporosis. Biochem Soc Trans. 2020;48:1623–1636. doi: 10.1042/BST20200005. [DOI] [PubMed] [Google Scholar]
- 36.Yin X, Zhou C, Li J, Liu R, Shi B, Yuan Q, Zou S. Autophagy in bone homeostasis and the onset of osteoporosis. Bone Res. 2019;7:28. doi: 10.1038/s41413-019-0058-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li H, Jeong HM, Choi YH, Kim JH, Choi JK, Yeo CY, Jeong HG, Jeong TC, Chun C, Lee KY. Protein kinase a phosphorylates Dlx3 and regulates the function of Dlx3 during osteoblast differentiation. J Cell Biochem. 2014;115:2004–2011. doi: 10.1002/jcb.24872. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.