Abstract
In recent years, metal peroxide (MO2) such as CaO2 has received more and more attention in cancer treatment. MO2 is readily decompose to release metal ions and hydrogen peroxide in the acidic tumor microenvironment (TME), resulting metal ions overloading, decreased acidity and elevated oxidative stress in TME. All of these changes making MO2 an excellent tumor therapeutic agent. Moreover, by combining MO2 with photosensitizers, enzymes or Fenton reagents, MO2 can assist and promote various tumor therapies such as photodynamic therapy and chemodynamic therapy. In this review, the synthesis and modification methods of MO2 are introduced, and the representative studies of MO2-based tumor monotherapy and combination therapy are discussed in detail. Finally, the current challenges and prospects of MO2 in the field of tumor therapy are emphasized to promote the development of MO2-based cancer treatment.
Keywords: Metal peroxide, Metal ions overloading, Oxidative stress, Combination therapy, Cancer treatment
Graphical abstract
In this review, the synthesis and modification methods of metal peroxide (MO2) are introduced, and the representative studies of MO2-based tumor monotherapy and combination therapy are discussed in detail. In addition, the current challenges and prospects of MO2 in the field of tumor therapy are also emphasized to promote the development of MO2-based cancer treatment.
Highlights
-
•
Metal peroxide (MO2) has becoming a potential therapeutic agent for tumor treatment.
-
•
MO2 can alleviate hypoxia and remodel tumor microenvironment by self-supplying O2.
-
•
MO2 can assist and promote various tumor therapies to enhance therapeutic efficacy.
-
•
The metal ions in MO2 endow MO2 with special functions and extend its applications.
1. Introduction
Cancer has always been one of the most threatening diseases for human survival. In the past ten years, the incidence of malignant tumors in China has maintained an annual increase of about 3.9%, even worse, estimated 9.6 million died of cancer worldwide in 2018 [1,2]. Therefore, the development of cancer therapeutic methods to fight against cancer is urgely needed.
With the progress of science and technology, in addition to traditional chemotherapy and radiotherapy, scientists have already developed various novel cancer therapies, including photodynamic therapy (PDT), chemodynamic therapy (CDT), photothermal therapy and so on [[3], [4], [5]]. However, the rapid proliferation of cancer cells resulted the supply and consumption of oxygen (O2) unbalanced, combined with the abnormal structure and function of tumor blood vessels, made hypoxia the most prominent feature of tumor microenvironment (TME) [6,7]. As a result, many therapeutic resistance effects will occur. For example, insufficient O2 sources lead to low reactive oxygen species (ROS) production and decrease cell sensitivity to ROS, which greatly reduce the effectiveness of O2-dependent therapies such as PDT and radiation therapy [8,9]. Moreover, some chemotherapeutic drugs also failed to be activated in a hypoxic TME. Therefore, it is necessary to regulate the hypoxic TME to improve the antitumor effect.
To cope with the challenge of tumor hypoxia, the hyperbaric oxygen (HBO) therapy in which patients breathe pure O2 or high-concentration O2 in a hyperbaric chamber can used as an adjuvant therapy. HBO therapy is beneficial to increase the partial pressure of O2 in the plasma and promote the O2 transport to hypoxic tumor tissue [10]. However, HBO therapy has many side effects such as O2 poisoning, barotrauma and decompression sickness [11]. The method by employing catalase (CAT) or MnO2 to convert the intracellular hydrogen peroxide (H2O2) to O2 has also been applied to overcome the hypoxia [[12], [13], [14]]. However, the O2 supplied by MnO2 degradation and CAT catalysis also face to be exhausted due to the limited intratumoral H2O2. Fortunately, metal peroxides (MO2) has been explored better to alleviate hypoxia by a disproportionation reaction with H2O in tumor tissue.
MO2, including CaO2, CuO2, MgO2, BaO2, ZnO2 etc., can be considered as the product of two hydrogen atoms in H2O2 replaced by metal ions. MO2 can cause strong oxidation effect by its decomposition products (such as H2O2) under acidic condition, while it can also slowly release O2 in water or under heating conditions [15]. These properties making it widely used in antibacterial [16], agricultural production [17], environmental protection [18], and other aspects [19]. Many of its other properties, if exploited, can also provide new ideas for cancer therapy: (I) Under acidic conditions, MO2 can react with H2O to produce H2O2, which causes increased oxidative stress; (II) The generated H2O2 reacts with Fenton or Fenton-like reagents (such as Fe2+, Mn2+, Cu+, Co2+, etc.) to produce hydroxyl radical (⋅OH) and realize CDT; (III) The produced H2O2 can be decomposed by CAT or MnO2 to produce O2 and increase the efficacy of O2-dependent cancer therapies such as PDT and radiation therapy; (IV) The released metal ions have some unexpected effects, for instance, calcium overloading caused by Ca2+ ions that released from CaO2 makes mitochondria damage [20]; while the Ba2+ ions generated by the disproportionation reaction of BaO2 act as a potassium ion channel inhibitor to inhibit tumor cell proliferation [21]. The application of MO2 in cancer treatment is developed fastly, and there is no doubt that MO2 is a promising candidate to regulate TME for various treatments.
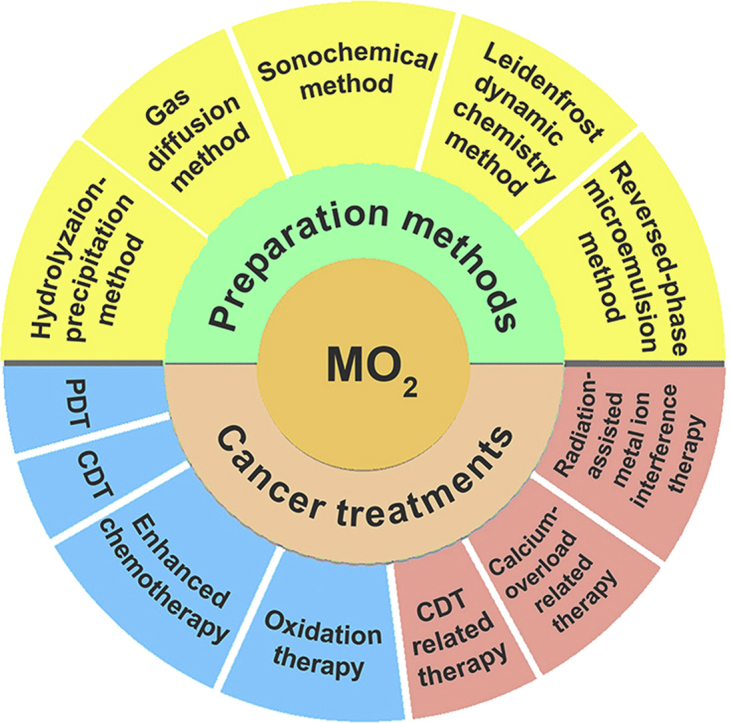
In this review, recent advances in the preparation and application of MO2 in cancer therapy will be discussed. As shown in Scheme 1, we starting from the preparation and surface modification of MO2, and then elaborate the design ideas and application examples of MO2 in CDT, PDT, ion interference therapy and various types of synergistic therapy. Finally, the current challenges and prospects of MO2-based cancer treatment will be presented.
Scheme 1.
Preparations and applications of MO2 in tumor therapy.
2. Synthesis and surface modification of MO2
To date, several preparation methods of MO2 are developed, including hydrolyzation-precipitation method [22,23], underwater Leidenfrost dynamic chemistry method [24], reversed-phase microemulsion method [25], gas diffusion method [26], and sonochemical method [27]. Due to the instability of MO2, some surface modification is necessary to achieve better applications in biomedical fields, frequently used surface modifiers are polyethylene glycol (PEG), polyvinyl pyrrolidone (PVP), CO-520, and hyaluronic acid (HA) [23,25,28,29]. Proper surface modification not only increases the stability of MO2, but also improves the dispersion of nanoparticles (NPs), even be possible to target the tumor. The preparation and surface modification of MO2 are summarized in Table 1, and will be discussed in detail in this section.
Table 1.
The synthesis, surface modification and applications of MO2-based biomaterials.
| MO2 | Preparation | Morphology and size | Surface modifier | Applications | Ref. |
|---|---|---|---|---|---|
| CaO2 | Hydrolyzation-precipitation method | Particle, <5 nm | SH | Calcium-overload/Oxidation therapy | [20] |
| CaO2 | Hydrolyzation-precipitation method | Particle, 15 ± 10 nm | PEG-200 | PDT | [22] |
| CaO2 | Hydrolyzation-precipitation method | Spherical structure, 5–15 nm | PEG-200 | PDT | [23] |
| CaO2 | Hydrolyzation-precipitation method | Particle | PVP | PDT | [28] |
| CaO2 | Hydrolyzation-precipitation method | Particle, 107 ± 11 nm | HA | CDT | [29] |
| CaO2 | Hydrolyzation-precipitation method | Clusters, 300 ± 20 nm | PEG-200 | PDT | [30] |
| CaO2 | Hydrolyzation-precipitation method | Spherical structure, 116.0 ± 7.6 nm | PEG-200 | PDT | [31] |
| CaO2 | Hydrolyzation-precipitation method | Particle, <200 nm | PEG-200 | Chemotherapy/CDT | [15] |
| CaO2 | Hydrolyzation-precipitation method | Particle, 20–30 nm | PEG-200 | Magnetic hyperthermia therapy/CDT | [32] |
| CaO2 | Hydrolyzation-precipitation method | Particle, ~20 nm | PEG-400 | CDT/PDT | [33] |
| CaO2 | Hydrolyzation-precipitation method | Particle, ~18 nm | PEG-200 | PTT/CDT | [34] |
| CaO2 | Hydrolyzation-precipitation method | Particle, ~8 nm | PVP | PTT/CDT | [35] |
| CaO2 | Hydrolyzation-precipitation method | Particle, 200–240 nm | TA | – | [36] |
| CaO2 | Gas diffusion method | Spherical structure, 90 nm | SiO2 | Immunochemotherapy | [26] |
| CaO2 | Reversed-phase microemulsion method | Particle, 273.4 ± 7.8 nm | CO-520 | Chemotherapy | [25] |
| CuO2 | Hydrolyzation-precipitation method | Nanodots, ~16.3 nm | PVP | CDT | [37] |
| ZnO2 | Hydrolyzation-precipitation method | Particle, 66.1 nm | PVP | Oxidation therapy | [38] |
| ZnO2 | Leidenfrost dynamic chemistry method | Particle, size-tailored | – | Oxidation therapy | [24] |
| ZnO2 | Sonochemical method | Particle | – | – | [27] |
| MgO2 | Reversed-phase microemulsion method | Nanosheets, 100–200 nm | CO-520 | Molecular dynamic therapy | [39] |
| BaO2 | Hydrolyzation-precipitation method | Bamboo-structure, <15 nm | GLDA | Radiation-assisted metal ion interference therapy | [21] |
Notes: PEG, polyethylene glycol; HA, hyaluronic acid; SH, sodium hyaluronate; PVP, polyvinyl pyrrolidone; TA, tannic acid; GLDA, N, N-bis(carboxymethyl)-l-glutamic acid tetrasodium salt.
2.1. Hydrolyzation-precipitation method
Currently, the hydrolyzation-precipitation method is the most widely used method to prepare MO2. This method usually used metal chloride, metal acetate or metal carbonate as precursors, through adding H2O2 to the alkaline aqueous solution of metal salt to precipitating the water-insoluble MO2. This method has the advantages of simple, mild conditions, low cost, and the size of NPs can be adjusted to several nanometers for the further construction of nanostructures.
Take the synthesis of CaO2 for example, the CaO2 hydrate was obtained by Eq. (1), and then the reaction was promoted to precipitate in favor of the metal peroxide by adding ammonia to neutralize the HCl, Eq. (2) [40].
| CaCl2+ H2O2→ CaO2 (hydrate) + 2HCl | (1) |
| 2HCl+2NH3→ 2NH4Cl | (2) |
Based on the above theory, Xia et al. reported an approach to synthesize CaO2 nanocrystals and their spherical aggregates [41]. They chose ethanol as the solvent to reduce the hydrolysis of CaO2 NPs. Typically, 2–15 nm primary nanocrystals were obtained through adding H2O2 into an ethanol solution with CaCl2 and PVP, in which PVP cooperated with Ca2+ to affect the growth and assembly of the nanocrystalline, resulting in spherical aggregates of polycrystalline structure with uniform size (Fig. 1). Therefore, the size of NPs can be easily controlled by adjusting the concentration of CaCl2 and/or PVP. The prepared CaO2 could greatly improve the antibacterial activity by releasing H2O2 and O2 at a faster rate after contact with H2O, which provided a new way for CaO2 to be used as a nano-drug. It is worth mentioning that PVP not only regulates particle size as mentioned above, but also acts as a stabilizer in hydrolyzed precipitation. Similarly, PEG can also be used as a stabilizer to modify the surface of MO2 [22,33,37,38].
Fig. 1.
(A) Schematic illustration of the formation of spherical aggregates through PVP-directed aggregation of CaO2 primary nanocrystals. (B) TEM images of CaO2 spherical aggregates synthesized in the presence of CaCl2 at different concentrations: a) 2.1, b) 4.2, c) 8.4, d) 25.2, e) 42, and f) 168 mM, respectively. (C) TEM images of CaO2 spherical aggregates synthesized in the presence of PVP at different concentrations: a) 43.2, b) 30.9, c) 21.6, d) 6.17, e) 1.23, and f) 0 mg mL−1, respectively. Reproduced with permission from Ref. [41]. Copyright 2019, Wiley-VCH.
Bu et al. reported the synthesis of BaO2 by hydrolysis and precipitation reaction [21]. In short, sodium formate and BaCl2 aqueous solution were mixed with ultrasonic and then added to anhydrous methanol. Excessive amounts of H2O2 were dropped into the mixture after vigorously stirring, choline hydroxide aqueous solution was followed to precipitate the BaO2 NPs (Fig. 2A). They pointed out that the selection of organic ligands has a great influence on the growth rate and orientation of BaO2 nanocrystal, so products of different particle sizes and morphology will be obtained. In their study, BaO2 particle with sub-micron scale can be obtained by using free Ba2+ as the precursor, while BaO2 nanocrystal with bamboo-structure will be formed by using formate coordinated Ba2+ as the precursor. The authors made a variety of attempts and found that the stronger the coordination ability with Ba2+, the smaller products will be obtained (Fig. 2B). Therefore, the introduction of a ligand with a certain coordination ability with Ba2+ into the reaction can effectively limit the growth of the crystal to get nanosized BaO2 particles. Shape controllability gives nanomaterials different specific surface areas and functions, which is an important link in the construction of nanomaterials theranostics platform.
Fig. 2.
(A) Schematic diagram of the preparation procedure of GL-BaO2 NPs. (B) TEM images of the formed BaO2 particles with different precursors, including Ba2+, formate + Ba2+, and citric acid + Ba2+. Reproduced with permission from Ref. [21]. Copyright 2019, Royal Society of Chemistry.
In the example above, the authors modified BaO2 with a biodegradable strong chelating agent l-glutamic acid (N-diacetic acid) to reduce the damage of free Ba2+ to normal tissues. When drugs entered the tumor, X-ray stimulation could separate Ba2+ and chelating agent, promoting the treatment of tumor. Which proved that MO2 can be modified by chelating method and enhanced the performance of MO2 performance.
In addition, hyaluronic acid (HA), sodium hyaluronate (SH), and tannic acid (TA) are also used for surface modification of MO2. For example, after stabilization by HA, CaO2 can remain stable in the humoral environment, and can only be degraded in the acidic TME to achieve material stabilization and tumor targeting [20,29,36]. To sum up, proper surface modification not only increases the stability of MO2, but also improves the dispersion of NPs, even be possible to target tumor.
2.2. Leidenfrost dynamic chemistry method
As we all known, a drop of water on an iron plate at 100 °C will boil and evaporate quickly. But if the temperature of the iron continues to rise, the water droplets will roll around the iron plate and evaporate at a slower rate. Leidenfrost first found and put forward the phenomenon in 1756, and pointed out that when the iron temperature reached the Leidenfrost point, water droplets in contact with the iron part will quickly form a steam, other part will remain liquid, due to the heat transfer of vapor is much slower than liquid water. Vapor layer can form a protective layer to avoid liquid water contact with the iron plate directly, so as to reduce the evaporation rate of the water. This phenomenon is well known Leidenfrost phenomenon [42,43].
Inspired by the volcano-induced dynamic chemistry of the deep sea [44], Moheb Abdelaziz and co-authors found that Leidenfrost can occur underwater. By virtue of the research of underwater Leidenfrost phenomenon, they designed a new tool for customized creation of nanoclusters of ZnO2 (Fig. 3A) [24]. In this method, the nucleation and growth of NPs are separated into two parts: firstly, at the overheated zone, nanochemistry occurs and the formed NPs assembled as nanoclusters; secondly, these nanoclusters will erupt into colder regions for further growth. Such a tendency could be harnessed to regulate the size of NPs. Specifically, zinc acetate solution was mixed with H2O2 and placed in a Petri dish, and then suddenly introduced to a superheated plate (300 °C), the solution can be observed to change from colorless to milky white, ZnO2 NPs are thus formed.
Fig. 3.
(A) Scheme of Leidenfrost dynamic chemistry approach. (B) SEM images of the ZnO2 NPs with different sizes synthesized by regulating the concentration of zinc acetate: a) 70 mM, ~70 nm; b) 50 mM, ~126 nm; c) 20 mM, ~220 nm; d) 5 mM, ~680 nm. Reproduced with permission from Ref. [24]. Copyright 2017, Nature Publishing Group.
All in all, the size of ZnO2 NPs prepared by this method could be adjusted by changing the concentration of zinc acetate (Fig. 3B). Its synthesis path is very simple and takes a short time, and the NPs prepared are very uniform and excellent monodispersity, which is suitable for mass production. On the basis of ZnO2, more MO2 nanomaterials can be prepared.
2.3. Reversed-phase microemulsion method
As a new preparation method, reversed-phase microemulsion method has simple equipment and technology. It can provide a nano-scale microreactor and control the appearance of NPs more precisely. The prepared NPs have the advantages of small particle size, good dispersion, and no impurities, etc. Which is a promising preparation method and MO2 can also be prepared by reversed microemulsion method.
CO-520 is always used as a nonionic surfactant in reversed-phase microemulsion. There had a report to synthesize MgO2 nanosheets [39]. Cyclohexane and CO-520 were added into MgCl2 solution to create a microemulsion system. After 30 min stirring, ammonium hydroxide was rapidly injected to forming the Mg(OH)2 and keep stirring for 30 min. Then H2O2 was added to control the reaction process and anhydrous ethanol was used to destroy the reverse microemulsion system to obtain the MgO2 nanosheets. Similarly, Xiang et al. fabricated CaO2 nanoparticle and simultaneous incorporation of cisplatin and coating with negatively charged phospholipid through reverse microemulsion method [25].
In the reaction of microemulsion, the size of NPs can be adjusted by adjusting the water content and pH of reversed micelles. The organic solvent phase and surfactant membrane in the microemulsion system effectively isolated the precipitation particles and improved the dispersibility of the particles. Some chemotherapeutic drugs can also be directly added into the microemulsion system to prepare NPs and carry out drug loading at the same time. Thus, reversed-phase microemulsion is a convenient and rapid method for the preparation of MO2.
2.4. Gas diffusion method
The gas diffusion method is often used as the preferred method for studying biomimetic synthesis of calcium carbonate (CaCO3) minerals, which has the advantages of simple operation and easy observation. Deng and his colleagues designed a new synthesis approach of CaO2 based on the gas diffusion synthesis of CaCO3 [26]. Typically, the beaker containing the ethanol solution with CaCl2 and H2O2 was covered by parafilm with a few pores, afterwards another beaker containing ammonia was placed in the same desiccator. The gas diffusion reaction will lasts for 2 h at 35 °C, the preparation of CaO2 was completed. In fact, it is also an extension of the hydrolytic precipitation process described above.
This reaction is mild and simple, moreover, the prepared NPs presented monodispersed spherical morphology, the size was controlled below 100 nm and uniformed, it layed a foundation for the further construction of nanoplatform with it as the core.
2.5. Sonochemical method
Sonochemical method is a kind of “green” chemical synthesis method, the cavitation collapse of ultrasonic will produce chemical and physical effects so as to drive some reactions, the process of sonochemical synthesis is mild with improved yields and selectivities, can even replace some dangerous reagents, which gradually become a new method in the field of nanochemical synthesis [45].
Mahtab Pirouzmand and co-workers prepared ZnO2 through sonochemical approach innovatively [27]. Their approach is so simple that ZnSO4·7H2O was dissolved in distilled water, and NaOH was added dropwise to adjust pH to 8.0. Followed by adding H2O2 and the mixture was irradiated under ultra-sound for half an hour, and then the ZnO2 particles with great uniform size distribution and spherical shape were obtained. However, agglomeration of particles was observed. It remains to be explored how to adjust the particle size and improve the dispersion of NPs.
3. MO2 based monotherapy
In recent years, MO2 often be introduced as an O2-generating material applied to the construction of tumor theranostics nanoplatform, which can regulate the TME to create a new work environment for those therapy whose efficacy is limited by the original TME. In addition, free metal ions can be involved in biological applications such as imaging and bone regeneration, made MO2 a potential biological materials. This section will focus on MO2-based monotherapy, including photodynamic therapy (PDT), chemodynamic therapy (CDT), and chemotherapy.
3.1. Oxidation therapy
Reactive oxygen species (ROS) including superoxide anion radical (⋅O2−), 1O2, H2O2, and ⋅OH can damage lipids, proteins, and DNA, resulting in cell apoptosis and death [46]. The process of ROS levels exceeding the antioxidant capacity of cells and leading to cell death is called oxidative stress [47,48]. Oxidative stress has been widely used in tumor treatment in recent years, MO2 is such a good material can lead oxidative stress in cells.
In the work of Chen, a PVP-modified ZnO2 NPs was developed and doped with paramagnetic Mn2+ ions through cation exchange method [38]. In this system, ZnO2 will be decomposed into Zn2+ and H2O2 in the weakly acidic TME. It is worth mentioning that Zn2+ has been reported to increase the mitochondria production of ROS by inhibiting the electron transfer chain [[49], [50], [51]], and the release of H2O2 increases the exogenous H2O2 to the cell. The endogenous generation combined with the exogenous release of ROS resulting in better tumor killing effect.
Bu et al. constructed transferrin-modified MgO2 nanosheets (TMNSs), which have a corresponding response to the neutral acidity and low CAT activity of TME, MgO2 reacts with H+ to generate H2O2 rapidly and damages the structure of transferrin on the surface of the nanosheets [39]. Then, transferrin releases the trapped Fe3+ and generates cytotoxic ⋅OH through the Fenton reaction. The high concentration of H2O2 and the generated ⋅OH destroyed tumor cells together, while TMNSs in weakly alkaline normal cells only generate a small amount of H2O2 that is enough to be decomposed by CAT. Thus, this nanosystem showed excellent tumor selectivity.
3.2. Photodynamic therapy
PDT utilizes photosensitizers (PS) to convert the local molecular oxygen into cytotoxic ROS, ROS can damage biological macromolecules and induce cell apoptosis [52,53]. However, the efficacy of PDT is extremely dependent on the O2 concentration, thus the hypoxia of solid tumors has limited the efficacy of PDT [54], and the further O2 consumption of PDT will aggravate the tumor's hypoxia and form a vicious circle [55]. In order to solve the above problems, MO2 has been developed as an O2 self-sufficient material to enhance the effect of PDT.
For instance, Zhang et al. designed a liposome-based nanoplatform for dual-stage light-driven PDT [22]. They encapsulated the hydrophilic PS (methylene blue, MB) and CaO2 NPs into the aqueous cavity and hydrophobic layer, respectively. When LipoMB/CaO2 reach the tumor tissue, CaO2 inside liposomes would react with H2O to generate O2 in the mild acidic TME, which can alleviate tumor hypoxia. Then, a short time irradiation is applied in the first stage, the singlet oxygen (1O2) would activated by MB and break the liposome by oxidized the phospholipid bilayer, CaO2 is further exposed to H2O and generate more O2. At last, a long time irradiation is given in the second stage, the PDT effect will improved a lot in such an O2 sufficient TME. In a deeper analysis, after the alleviation of tumor hypoxia, the down-regulation of hypoxia-inducible factor-1α and vascular endothelial growth factor expression may also reduce the rate of tumor metastasis [56]. This two-stage light strategy based on CaO2 is ingeniously designed to maximize the O2 supply capacity of CaO2. It is a nano-platform worth learning for alleviating tumor hypoxia and anti-tumor metastasis.
Similarly, hydrophobic aza-BODIPY dye (B1), oxygen-generating CaO2 and hydrophilic ammonium bicarbonate (NH4HCO3) encapsulated in PEG shelled liposome to realize self-supplying O2 PDT therapy also be reported [23]. In this study, NH4HCO3 acts as a thermoresponsive molecule. When the liposome system irradiated by near-infrared (NIR), B1 will cause the temperature to rise. Once the temperature reached 40 °C, NH4CO3 will be thermally decomposed to produce CO2 [57], which will expand and destroy the liposomes, causing CaO2 and CO2 fully react to produce O2 to improve the PDT effect of B1 (Fig. 4) [58]. Finally, they conducted tumor treatment experiments in vivo and find that enough O2 generation from CaO2/B1/NH4HCO3 liposome was favorable to produce 1O2 in the presence of photosensitizer B1 and inhibit tumor growth even induced tumor disappearance. In addition to the above examples of light-triggered O2-generation, pH seems another feasible trigger. In the work of Callan et al., CaO2 particles were coated with a pH-responsive methacrylate-based co-polymer. The tertiary amine unit of the copolymer will be ionized in an acidic environment, and CaO2 will therefore exposed and producing O2. The TME creates a favorable conditions for the dissolution of copolymers [59]. Finally, with the help of CaO2 NPs, PDT from rose bengal (a kind of PS) achieved the best effect [31].
Fig. 4.
(A) Scheme illustration of O2 self-supplying enhanced PDT of CaO2/B1/NH4HCO3 lipo: a) NIR-regulated generation of O2 and enhancement of 1O2; b) the mechanism of O2 generation; c) O2 self-sufficient CaO2/B1/NH4HCO3 lipo enhanced PDT. (B) Photograph of tumors obtained from the mice after 14 days. (C) Tumor volume ratio of mice during the treatments. Reproduced with permission from Ref. [23]. Copyright 2019, Royal Society of Chemistry.
CaO2 not only generates O2 itself but also provides a reaction substrate for other O2-generating materials. For example, CaO2 reacts with H2O to generate H2O2 (Eq (3)) which provide a raw material for manganese dioxide (MnO2) for the generation of O2 in a mild acid environment, Eq (4) [60]. By this way, Shi and his colleagues prepared a CaO2/MnO2@PDA-MB, system through MnO2 nanosheet coated on the surface of CaO2, in acidic TME, polydopamin (PDA) dissolution enables Mn2+ to fully react with H2O2 produced by CaO2 and generate O2, further promoting the PDT effects of MB that the singlet oxygen quantum yield reached 0.18 and realized switch-control fluorescence imaging [28].
| CaO2 +2H2O → H2O2 + Ca(OH)2 | (3) |
| MnO2 + H2O2 + 2H+ → Mn2+ +2H2O + O2 | (4) |
3.3. Chemodynamic therapy
CDT is an emerging nanotheranostic technology which catalyzes the conversion of H2O2 into ⋅OH through an elaborately designed Fenton nano-catalyst [61,62]. However, limited by the concentration of endogenous H2O2 in the tumor, the effect of CDT is often unsatisfactory. Since MO2 can generate H2O2 in the mild acidic TME, it can be designed to enhance CDT efficacy. Moreover, the metal ions like Cu2+, Co2+, Mn2+ that consisted in MO2 possesses excellent Fenton catalytic activity, making MO2 become a promising H2O2 self-supply CDT nanoagent [[63], [64], [65], [66]].
Based on above, Jiang et al. controlled the self-assembly of Fe3O4 on the surface of HA-stabled CaO2 to form CaO2–Fe3O4@HA NPs. It also realizes H2O2 self-supplying CDT treatment and demonstrated a desirable performance in tumor growth inhibition rate of 69.1%, moreover, by loading Cy7 the fluorescence imaging can be combined with therapy [29].
In another case, Chen and co-authors developed a Fenton-type copper peroxide (CP) nanodots that anchored by PVP with the aid of hydroxide ion [37]. The prepared CP nanodots could reversely decompose into Cu2+ and H2O2 in an acidic environment, thereby realizing the H2O2 self-supplying CDT (Fig. 5A). The pH-sensitive CP nanodots were internalized by tumor by enhanced permeability and retention effects [67], and generate large amounts of ⋅OH through Fenton-like reaction in the acidic endo/lysosomal compartments, which can induced lysosomal membrane permeabilization-mediated tumor cell killing by lysosomal lipid peroxidation [68,69]. Finally, the authors evaluated the biological distribution of CP nanopods in U87MG tumor-bearing mice by inductively coupled plasma optical emission spectrometry found that the uptake of CP nanopods by tumors reached 5.96 ± 0.79% and showed excellent CDT anti-tumor effect with negligible weight loss (Fig. 5B–D).
Fig. 5.
(A) Formation of CP nanodots for H2O2 self-supplying CDT. (B) Biodistribution of Cu in major organs and tumor of U87MG tumor-bearing mice at 24 h post i.v. injection with CP nanodots. (C) Relative tumor volume and (D) variation of body weight of the mice after different treatments. Reproduced with permission from Ref. [37]. Copyright 2019, American Chemical Society.
3.4. Enhanced chemotherapy
Tumor cells have very strict mechanisms to deal with hypoxia and resist oxidation. These mechanisms are closely combined which made abnormal factors such as hypoxia, acidosis, and high glutathione (GSH) levels are simultaneously present in the TME, which promotes the drug resistance of tumor cells especially ROS-dependent drugs [[70], [71], [72], [73]]. Fortunately, MO2 can generates O2 or by acting as a reaction substrate to reverse tumor hypoxia and providing more O2 for chemo-drugs to receive enhanced chemotherapy.
Xiang et al. reported a lipid-coated CaO2/cisplatin NPs which used the O2 production and oxidation capabilities of CaO2 at the same time to overcome tumor hypoxia and reduce GSH levels [25]. More importantly, CaO2 can significantly elevate the local pH and further accelerate GSH oxidation. After the TME was reversed, the binding of cisplatin to GSH was reduced, and the production of O2 downregulated the hypoxia inducible factor 1 and resistance-associated protein 2, then the efflux pathway of cisplatin is blocked (Fig. 6A). Combined with the above process, an enhanced chemotherapy effect was achieved. As shown in Fig. 6B, fluorescent imaging showed the lipid-coated CaO2/cisplatin NPs has a signal in the tumor for more than 48 h, indicating that it can achieve long circulation and efficient tumor accumulation, and the size of the tumor also showed that CaO2 significantly enhanced the antitumor effect of cisplatin in vivo anti-tumor experiments intuitively (Fig. 6C and D). The work of Sung and his colleagues also proved that the O2 production of CaO2 also has an effect on the improvement of the efficacy of the chemotherapy drug doxorubicin (DOX) [74]. Deng et al. used CaO2, MnO2 and DOX to construct CaO2/DOX@SiO2/DOX-MnO2 nanoreactor, and proved that the drug can effectively relieve hypoxia and reverse immunosuppressive TME to enhances anti-tumor immune responses from an immunological point of view [26]. What is most worth mentioning is the drug-loading method of this nano-platform. DOX is directly added when CaO2 NPs were synthesized, which can form Ca-DOX complex so that DOX can directly combine with CaO2 NPs to form the CaO2/DOX core and greatly improved the drug-loading efficiency.
Fig. 6.
(A) Schematic illustration of LipoCaO2/DPP for comprehensive TME modulation and cisplatin efflux pathway blockade: 1) produce Ca(OH)2 to raise the local pH; 2) oxidize GSH under alkaline conditions and reduce cisplatin/GSH binding; 3) generate O2 for inhibition of MRP2 by HIF-1 degradation, preventing the cisplatin–GSH adduct from pumping out of cells. (B) Real-time fluorescence imaging of free fluorescein (DIR) and LipoCaO2/DDP loaded DIR treated mice. (C) Tumor photographs and (D) tumor volume after different treatments. Reproduced with permission from Ref. [25]. Copyright 2019, Royal Society of Chemistry.
4. MO2 based combined therapy
The effect of monotherapy is limited, for most tumors, the treatment effect is unsatisfactory. Therefore, the combination of two or more therapeutic methods is particularly important. Each individual therapeutic agent has anti-tumor activity, and they can be built on a platform to achieve the effect of combined therapy. In addition, if the tumor killing mechanism of each therapeutic agent can complement each other, it can also realize the “1 + 1>2″ synergistic therapeutic effect [3]. After the introduction on the application of MO2 to monotherapy above, combination therapies based on MO2 will be discussed in this section.
4.1. CDT-based combination therapy
CDT can exert better curative effect by combining with other therapies [[75], [76], [77]]. Utilized the characteristics of CaO2 that it can react with H2O to generate H2O2 and O2, Dong et el. constructed a H2O2/O2 self-supplied thermoresponsive nanosystem (MSNs@CaO2-ICG)@LA by using manganese silicate (MSNs) to load CaO2 NPs and indocyanine green (ICG) and then modifying a layer of thermally dissolved lauric acid (LA, melting point: 44–46 °C). This nanosystem realized PDT/CDT synergistic cancer therapy and the tumors of MCF-7 bearing mice were completely eliminated [33].
In the work of Zhang and co-authors, they built a cobalt-based metal-organic framework (ZIF-67) on the surface of CaO2@DOX [15]. The slightly acid in tumor decomposed ZIF-67 and quickly released Co2+ and DOX. The H2O2 produced by CaO2 will be catalyzed by Co2+ and produced highly toxic ⋅OH through the Fenton-like reaction, while the produced O2 can improve the efficacy of DOX to enhance combined efficacy of CDT/chemotherapy. In another work, the Fe-GA/CaO2@PCM NPs developed by Dong et al. used an organic phase change material (PCM) with a melting point of 46 °C as a protective layer, and co-encapsulate the hydrophilic iron-gallic acid (Fe-GA) and CaO2 NPs [34]. After 808 nm laser irradiation, the temperature increase of Fe-GA causes the PCM to melt and exert the effect of photothermal therapy (PTT). H2O2 and Ca2+ produced by CaO2 participate in the Fe-based Fenton reaction and induce mitochondrial damage, respectively. PTT can futher accelerate the generation of ⋅OH. This nanoplatform can realized on-demand H2O2 self-supply for enhanced CDT/PTT treatment.
Moreover, MO2-based materials can also be used in three dimensional (3D) printing technology. Among a multifunctional “all-in-one” biomaterial platform, CaO2 and Fe3O4 NPs were co-loaded into a 3D printing akermanite (AKT) scaffold, named AKT-Fe3O4–CaO2 (Fig. 7) [32]. To put it simply, CaO2 reacts with H+ to produce H2O2 and Ca2+, H2O2 was catalyzed by Fe3O4 to produce ⋅OH, while Ca2+ can also be used for bone regeneration. In addition, Fe3O4 will produce hyperthermia under the action of alternating magnetic field (AMF). Thus, the scaffold with magnetic hyperthermia-synergistic H2O2 self-sufficient CDT and bone-regeneration function can be used effectively in the treatment of osteosarcoma.
Fig. 7.
(A) Schematic of the cancer-therapeutic performance and bone-regeneration bioactivity of AKT-Fe3O4–CaO2. (B) In vivo anti-tumor effect. (C) In vivo osteogenesis capability of AKT-Fe3O4–CaO2 scaffolds: I) 3D reconstruction of micro-CT images of the cranium and scaffolds (red, newborn bone tissues; white, residual scaffolds). II) CLSM images of slices from the bone defect area (yellow, newborn bone stained by tetracycline hydrochloride injected at week 2; red, newborn bone stained by Alizarin Red S injected at week 4; green, newborn bone stained by calcein injected at week 6. III) Microscopy image of VG stained slices of cranium with two defects implanted with AKT or AKT-Fe3O4–CaO2 scaffolds. Scale bar, 500 μm. Reproduced with permission from Ref. [32]. Copyright 2019, Wiley-VCH. F
4.2. Calcium-overload based combination therapy
Some harmful factors can cause dysfunction of calcium balance system and disorder of calcium distribution, leading to abnormal increase of intracellular calcium concentration, called calcium overload. Calcium overload can affect mitochondrial oxidative phosphorylation process, decrease the mitochondrial membrane potential, resulting in decrease of tissue ATP and activation of phospholipases and proteases in the cytoplasm, which cause irreversible cell damage [20,78]. In clinical treatment, internal calcification is frequently observed in certain tumors after radiotherapy or chemotherapy [79,80], so calcification is usually considered as a by-product of tumor treatment and it was found that calcified tumors often showed better treatment response. Given the importance role of Ca2+ in cell proliferation, metabolism, and death, the overloading process may be a destructive factor against tumor cells, and provide alternative drug-free method for cancer therapy.
Based on the above, Bu et al. developed a calcium-based nanomedicine, the sodium hyaluronate (SH)-modified CaO2 (SH–CaO2), to induce intracellular calcium overloading for cancer treatment (Fig. 8A) [20]. Results revealed that Ca2+ and H2O2 produced by CaO2 caused intracellular calcium overload and oxidative stress, respectively. For the CAT down-regulated tumor cells, oxidative stress will change the function of the protein and hinder the accurate transmission of the calcium signal, thereby causing uncontrollable accumulation of Ca2+ and inducing cell death [81]. Computer tomography (CT) imaging and von Kossa staining further showed that SH-CaO2 NPs can accelerate the process of tumor calcification (Fig. 8B and C). In addition, the in vivo experiments also proved that SH-CaO2 NPs has significant anti-tumor effect whether intratumoral injection or intravenous injection, and the anti-tumor effect of intratumoral injection is better, which caused the tumor almost disappeared during the observation period of 14 days after the injection (Fig. 8D). Similarly, the nanosystem constructed by Yin et al. with CaO2 as O2 source and hematoporphyrin monomethyl either as photosensitizer also combined PDT and calcium overload [82].
Fig. 8.
(A) Schematic illustration of the functional pattern of SH-CaO2 NPs. (B) CT images of mice after the following treatments: i) control, ii) 3 h after intratumor injection of SH-CaO2 NPs, iii) 3 days after the injection of a single dose of SH-CaO2 NPs for a small tumor, and iv) 12 days after the injection of multiple dose (4 times, injected every 2 days) for a larger tumor. (C) von Kossa staining of tumor tissue sections after multiple injections with SH-CaO2 NPs. (D) Relative tumor volume changes during the treatments. Reproduced with permission from Ref. [20]. Copyright 2019, Elsevier.
4.3. Radiation-assisted metal ion interference therapy
High Z elements have a significant effect on radiosensitization [83], but most metal-based nanomedicines are hindered in clinical application due to the biological toxicity of heavy metals. Bu and his co-authors developed a BaO2-based N,N-bis(carboxymethyl)-l-glutamic acid tetrasodium salt (GLDA) modified nanoplatform [21]. The chelation of Ba2+ with GLDA can reduce the toxic and side effects of the drug in normal tissues, then the ⋅OH produced after X-ray radiation will destroy the chemical structure of GLDA and release Ba2+. Benefit from this point, they proposed an ion interference therapy. The free Ba2+ not only enhanced the radiosensitization effect but also targeted and competitively binded to potassium channels after entering the cell, then the potassium conducting pores would be blocked and the outflow of potassium ions (K+) would be prevented [84,85], which further affect cell membrane potential and the osmotic pressure in the cell, ultimately inhibit cell proliferation and induce cell death [86,87].
5. Conclusions
MO2 has been developed in terms of self-supplying O2 and self-supplying H2O2, and has become a very potential therapeutic agent for tumor. Under acidic conditions, the generated H2O2 through MO2 reacting with H2O not only leads to oxidative stress, but also produce more O2 by acting as a reaction substrate for substances such as CAT or MnO2, so as to alleviate tumor hypoxia and reverse TME. More importantly, the characteristics of MO2 can be perfectly combined with photosensitizer, enzyme, metal NPs, Fenton reagent or chemotherapy drugs, etc., which can assist and promote various treatments such as PDT, CDT and chemotherapy. If combined multiple treatments, MO2-based combination therapy will achieve more excellent anti-tumor effects.
Here, we have introduced the preparation and surface modification methods of MO2-based nanomaterials and their application in tumor therapy in detail, including monotherapy and combination therapy, with emphasis on MO2-based PDT, CDT, chemotherapy, oxidation therapy and ion therapy (Table 1). However, the application of MO2-based nanomaterials in tumor therapy is still in the preliminary stage of research, and there are still many problems and challenges to be solved.
Firstly, morphology and size are key factors affecting the efficacy of nanomaterials, from previous studies, large NPs are more likely to retention in tumor tissue than small ones, while permeability is opposite, the smaller the size, the greater the ability to penetrate tumor tissue [88]. The size design of MO2 should in accordance with the functional requirements to regulate. If MO2 is used as the cargo, it is better design small size, which can not only implement efficient load but can accelerate the reaction rate of MO2 in the TME. On the other side, if MO2 is used as the carrier, large size may increase the rate of drug loading. In addition to particle size, morphology also plays an important role in tumor penetration. Most studies have shown that the tumor penetration ability of spherical NPs is lower than that of other shaped NPs [89]. At present, the morphology of MO2 is basically spherical, it is necessary to design MO2 with other morphology to increase the tumor permeability.
Secondly, there are few types of MO2 reported for tumor therapy so far, and most of the reported researches are focused on CaO2-based nanomaterials. Other MO2-based nanomaterials such as MgO2, BaO2, ZnO2, CuO2-based materials have not been fully developed and their biological applications are also limited, developing them by modifying them appropriately or combining them with other therapeutic agents may be a trend of future research. In the abovementioned MO2, we believe that CaO2 has the most clinical translation value by far. Ca2+ is widely distributed in the body, endowing CaO2 with good biocompatibility. In addition, Ca2+ is also distributed in tumor cells, so treatment strategies such as calcium overload have universality, and Ca2+ has the effect of accelerating osteogenesis, which can be well applied in the treatment of bone tissue related cancer such as osteosarcoma. However, the preparation and storage of CaO2 even MO2 are facing challenges, because of the instability, the morphology, size and dispersion of MO2 are difficult to precise control, made it difficult to realize the mass production. Therefore, exploring some new methods for preparing MO2 batch production is imperative, the Leidenfrost dynamic chemistry method is a good exploration [24]. For the clinical translation of MO2, improve the stability of MO2, extend drug life and ensure drug function is an urgent problem to be solved.
Thirdly, MO2 has the potential to be used in a variety of therapeutic modalities, and its biological applications remained to be explored. In the reported studies, MO2-based nanomaterials were mainly used in CDT, PDT and oxidation therapy, etc., but their application in magnetic heat, radiation, gas treatment and so on is rarely reported. Therefore, exploring the applications of MO2 in a variety of therapies should be more innovative.
Fourthly, as summarized in this paper, different ions in MO2 have additional functions. For example, Ba2+ not only plays the role of radiotherapy sensitization, but also leads to cell death and inhibit proliferation through K+ outflow from tissues [21]. Ca2+ not only induces calcium overload but also enhances CT imaging [20]. When CaO2 is used for the treatment of osteosarcoma, it had the ability to accelerate bone regeneration [32]. In addition to Ba2+ and Ca2+ ions, the functions of other ions have yet to be developed. Hence, mining the special function of ions in MO2 may futher broaden its application in the field of tumor therapy, even the imaging effects attached to some ions can be used for tumor theranostics [90].
Next, hypoxia TME will greatly weaken the effect of immunotherapy, hypoxia-A2-adenosinergic tumor biology is a barrier to be overcome in immunotherapy [78]. MO2 can reverse TME by self-supplying O2 and suppressing hypoxia-adenosinergic signaling, further reducing the expression of hypoxia-inducible factor 1 and CD39/CD73 in T cells to reduce the immunosuppression effect of tumor TME [91]. Therefore, the mechanism of MO2 in immunotherapy and its combination with other therapeutic methods remains to be further studied and developed.
Finally, the application of MO2 needs to pay attention to its biosafety and long-term toxicity. Although the reported MO2-based nanodrugs caused no damage to the normal tissues and organs even at a high dose of 50 mg kg−1, the biosafety should be systematically evaluated for a longer time and on larger animal models. Moreover, cytotoxicity induced by other parts except MO2 of nanodrug should also be considered. Furthermore, how to modify MO2 materials to ensure its anti-tumor effect and biosafety remains to be solved. Although there are still many problems to be solved, MO2 has brought new approaches to tumor therapy and its application in biological fields is worth developing and expanding. We hope that MO2-based nanodrugs can be applied in more anti-tumor methods and bring good news to patients.
Declaration of competing interest
The authors declare no conflict of interest.
Acknowledgments
This work is financially supported by the National Key R&D Program of China (2018YFA0704000), the National Natural Science Foundation of China (21807074, 51802202, 31900945, 82071985), Shenzhen Science and Technology Program (KQTD20190929172538530, JCYJ20180507182413022, JCYJ20170412111100742), the Guangdong Province Natural Science Foundation of PhD Start-up Fund (2018A030310574 and 2018A030310566), the Guangdong Province Natural Science Foundation of Major Basic Research and Cultivation Project (2018B030308003), and the Fundamental Research Funds for the Central Universities (2020CDJQY-A060).
Footnotes
Peer review under responsibility of KeAi Communications Co., Ltd.
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bioactmat.2021.01.026.
Appendix A. Supplementary data
The following is the supplementary data to this article:
References
- 1.Chen W., Zheng R., Baade P.D., Zhang S., Zeng H., Bray F. Cancer statistics in China, 2015. CA A Cancer J. Clin. 2016;66:115–132. doi: 10.3322/caac.21338. [DOI] [PubMed] [Google Scholar]
- 2.Bray F., Ferlay J., Soerjomataram I., Siegel R.L., Torre L.A., Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 3.Fan W., Yung B., Huang P., Chen X. Nanotechnology for multimodal synergistic cancer therapy. Chem. Rev. 2017;117:13566–13638. doi: 10.1021/acs.chemrev.7b00258. [DOI] [PubMed] [Google Scholar]
- 4.Fu L.H., Qi C., Lin J., Huang P. Catalytic chemistry of glucose oxidase in cancer diagnosis and treatment. Chem. Soc. Rev. 2018;47:6454–6472. doi: 10.1039/c7cs00891k. [DOI] [PubMed] [Google Scholar]
- 5.Fu L.H., Qi C., Hu Y.R., Lin J., Huang P. Glucose oxidase-instructed multimodal synergistic cancer therapy. Adv. Mater. 2019;31 doi: 10.1002/adma.201808325. [DOI] [PubMed] [Google Scholar]
- 6.Gilkes D.M., Semenza G.L., Wirtz D. Hypoxia and the extracellular matrix: drivers of tumour metastasis. Nat. Rev. Canc. 2014;14:430–439. doi: 10.1038/nrc3726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu J.N., Bu W., Shi J. Chemical design and synthesis of functionalized probes for imaging and treating tumor hypoxia. Chem. Rev. 2017;117:6160–6224. doi: 10.1021/acs.chemrev.6b00525. [DOI] [PubMed] [Google Scholar]
- 8.Meijer T.W.H., Kaanders J.H.A.M., Span P.N., Bussink J. Targeting hypoxia, HIF-1, and tumor glucose metabolism to improve radiotherapy efficacy. Clin. Canc. Res. 2012;18:5585–5594. doi: 10.1158/1078-0432.CCR-12-0858. [DOI] [PubMed] [Google Scholar]
- 9.Turan I.S., Yildiz D., Turksoy A., Gunaydin G., Akkaya E.U. A bifunctional photosensitizer for enhanced fractional photodynamic therapy: singlet oxygen generation in the presence and absence of light. Angew. Chem. Int. Ed. 2015;55:2875–2878. doi: 10.1002/anie.201511345. [DOI] [PubMed] [Google Scholar]
- 10.Petre P.M., Jr F.A.B., Tigan S., Spears J.R. Hyperbaric oxygen as a chemotherapy adjuvant in the treatment of metastatic lung tumors in a rat model. J. Thorac. Cardiovasc. Surg. 2003;125:85–95. doi: 10.1067/mtc.2003.90. [DOI] [PubMed] [Google Scholar]
- 11.Plafki C., Peters P., Almeling M., Welslau W., Busch R. Complications and side effects of hyperbaric oxygen therapy. Aviat Space Environ. Med. 2000;71:119. [PubMed] [Google Scholar]
- 12.Wang H., Chao Y., Liu J., Zhu W., Wang G., Xu L. Photosensitizer-crosslinked in-situ polymerization on catalase for tumor hypoxia modulation & enhanced photodynamic therapy. Biomaterials. 2018;181:310–317. doi: 10.1016/j.biomaterials.2018.08.011. [DOI] [PubMed] [Google Scholar]
- 13.Shi C., Li M., Zhang Z., Yao Q., Peng X. Catalase-based liposomal for reversing immunosuppressive tumor microenvironment and enhanced cancer chemo-photodynamic therapy. Biomaterials. 2020;233:119755. doi: 10.1016/j.biomaterials.2020.119755. [DOI] [PubMed] [Google Scholar]
- 14.Lin T., Zhao X., Zhao S., Yu H., Guo H. O2-generating MnO2 nanoparticles for enhanced photodynamic therapy of bladder cancer by ameliorating hypoxia. Theranostics. 2018;8:990–1004. doi: 10.7150/thno.22465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gao S., Jin Y., Ge K., Li Z., Liu H., Dai X. Self-supply of O2 and H2O2 by a nanocatalytic medicine to enhance combined chemo/chemodynamic therapy. Adv. Sci. 2019;6:1902137. doi: 10.1002/advs.201902137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Navik R., Thirugnanasampanthan L., Venkatesan H., Kamruzzaman M., Shafiq F., Cai Y. Synthesis and application of magnesium peroxide on cotton fabric for antibacterial properties. Cellulose. 2017;24:3573–3587. [Google Scholar]
- 17.Chou M.L., Jean J.S., Yang C.M., Hseu Z.Y., Chen Y.H., Wang H.L. Inhibition of ethylenediaminetetraacetic acid ferric sodium salt (EDTA-Fe) and calcium peroxide (CaO2) on arsenic uptake by vegetables in arsenic-rich agricultural soil. J. Geochem. Explor. 2016;163:19–27. [Google Scholar]
- 18.Cassidy D.P., Irvine R.L. Use of calcium peroxide to provide oxygen for contaminant biodegradation in a saturated soil. J. Hazard Mater. 1999;69:25–39. doi: 10.1016/s0304-3894(99)00051-5. [DOI] [PubMed] [Google Scholar]
- 19.Bae H.J., Cho D.C., Kwon S.H. Enviro-chemical changes in shoreline sediment by MgO2 for enhancement of indigenous microbial activity. J. Environ. Sci. Int. 2010;19:617–625. [Google Scholar]
- 20.Zhang M., Song R., Liu Y., Yi Z., Meng X., Zhang J. Calcium-overload-mediated tumor therapy by calcium peroxide nanoparticles. Inside Chem. 2019;5:2171–2182. [Google Scholar]
- 21.Zhang M., Shen B., Song R., Wang H., Lv B., Meng X. Radiation-assisted metal ion interference tumor therapy by barium peroxide-based nanoparticles. Mater. Horiz. 2019;6:1034–1040. [Google Scholar]
- 22.Liu L.H., Zhang Y.H., Qiu W.X., Zhang L., Gao F., Li B. Dual-stage light amplified photodynamic therapy against hypoxic tumor based on an O2 self-sufficient nanoplatform. Small. 2017;13:1701621. doi: 10.1002/smll.201701621. [DOI] [PubMed] [Google Scholar]
- 23.Yu Q., Huang T., Liu C., Zhao M., Xie M., Li G. Oxygen self-sufficient NIR-activatable liposomes for tumor hypoxia regulation and photodynamic therapy. Chem. Sci. 2019;10:9091–9098. doi: 10.1039/c9sc03161h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Elbahri M., Abdelaziz R., Disci-Zayed D., Homaeigohar S., Sosna J., Adam D. Underwater Leidenfrost nanochemistry for creation of size-tailored zinc peroxide cancer nanotherapeutics. Nat. Commun. 2017;8:15319. doi: 10.1038/ncomms15319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.He C., Zhang X., Yan R., Zhao P., Chen Y., Li M. Enhancement of cisplatin efficacy by lipid-CaO2 nanocarrier-mediated comprehensive modulation of the tumor microenvironment. Biomater. Sci. 2019;7:4260–4272. doi: 10.1039/c9bm00797k. [DOI] [PubMed] [Google Scholar]
- 26.Wang J., Fang L., Li P., Ma L., Na W., Cheng C. Inorganic nanozyme with combined self-oxygenation/degradable capabilities for sensitized cancer immunochemotherapy. Nano-Micro Lett. 2019;11:74. doi: 10.1007/s40820-019-0305-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pirouzmand M., Sani P.S., Ghasemi Z., Azizi S. Citric acid-crosslinked beta-cyclodextrin supported zinc peroxide as a biocompatible H2O2 scavenger. J. Biol. Inorg. Chem. 2020;25:411–417. doi: 10.1007/s00775-020-01771-6. [DOI] [PubMed] [Google Scholar]
- 28.Ji C., Lu Z., Xu Y., Shen B., Yu S., Shi D. Self-production of oxygen system CaO2/MnO2@PDA-MB for the photodynamic therapy research and switch-control tumor cell imaging. J. Biomed. Mater. Res. B. 2018;106:2544–2552. doi: 10.1002/jbm.b.34071. [DOI] [PubMed] [Google Scholar]
- 29.Han Y., Ouyang J., Li Y., Wang F., Jiang J.H. Engineering H2O2 self-supplying nanotheranostic platform for targeted and imaging-guided chemodynamic therapy. ACS Appl. Mater. Interfaces. 2020;12:288–297. doi: 10.1021/acsami.9b18676. [DOI] [PubMed] [Google Scholar]
- 30.Hu Y., Wang X., Zhao P., Wang H., Gu W., Ye L. Nanozyme-catalyzed oxygen release from calcium peroxide nanoparticles for accelerated hypoxia relief and image-guided super-efficient photodynamic therapy. Biomater. Sci. 2020;8:2931–2938. doi: 10.1039/d0bm00187b. [DOI] [PubMed] [Google Scholar]
- 31.Sheng Y., Nesbitt H., Callan B., Taylor M.A., Love M., Mchale A.P. Oxygen generating nanoparticles for improved photodynamic therapy of hypoxic tumours. J. Contr. Release. 2017;264:333–340. doi: 10.1016/j.jconrel.2017.09.004. [DOI] [PubMed] [Google Scholar]
- 32.Dong S., Chen Y., Yu L., Lin K., Wang X. Magnetic hyperthermia–synergistic H2O2 self‐sufficient catalytic suppression of osteosarcoma with enhanced bone‐regeneration bioactivity by 3D‐printing composite scaffolds. Adv. Funct. Mater. 2019;30:1907071. [Google Scholar]
- 33.Liu C., Cao Y., Cheng Y., Wang D., Xu T., Su L. An open source and reduce expenditure ROS generation strategy for chemodynamic/photodynamic synergistic therapy. Nat. Commun. 2020;11:1735. doi: 10.1038/s41467-020-15591-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhang S.C., Cao C.Y., Lv X.Y., Dai H.M., Zhong Z.H., Liang C. A H2O2 self-sufficient nanoplatform with domino effects for thermal-responsive enhanced chemodynamic therapy. Chem. Sci. 2020;11:1926–1934. doi: 10.1039/c9sc05506a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gao S., Lu X., Zhu P., Lin H., Yu L., Yao H. Self-evolved hydrogen peroxide boosts photothermal-promoted tumor-specific nanocatalytic therapy. J. Mater. Chem. B. 2019;7:3599–3609. [Google Scholar]
- 36.Park J.S., Song Y.J., Lim Y.G., Park K. Facile fabrication of oxygen-releasing tannylated calcium peroxide nanoparticles. Materials. 2020;13:3864. doi: 10.3390/ma13173864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lin L.S., Huang T., Song J., Ou X.Y., Wang Z., Deng H. Synthesis of copper peroxide nanodots for H2O2 self-supplying chemodynamic therapy. J. Am. Chem. Soc. 2019;141:9937–9945. doi: 10.1021/jacs.9b03457. [DOI] [PubMed] [Google Scholar]
- 38.Lin L.S., Wang J.F., Song J., Liu Y., Zhu G., Dai Y. Cooperation of endogenous and exogenous reactive oxygen species induced by zinc peroxide nanoparticles to enhance oxidative stress-based cancer therapy. Theranostics. 2019;9:7200–7209. doi: 10.7150/thno.39831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tang Z.M., Liu Y.Y., Ni D.L., Zhou J.J., Zhang M., Zhao P.R. Biodegradable nanoprodrugs: "delivering" ROS to cancer cells for molecular dynamic therapy. Adv. Mater. 2020;32 doi: 10.1002/adma.201904011. [DOI] [PubMed] [Google Scholar]
- 40.Khodaveisi J., Banejad H., Afkhami A., Olyaie E., Lashgari S., Dashti R. Synthesis of calcium peroxide nanoparticles as an innovative reagent for in situ chemical oxidation. J. Hazard Mater. 2011;192:1437–1440. doi: 10.1016/j.jhazmat.2011.06.060. [DOI] [PubMed] [Google Scholar]
- 41.Shen S., Mamat M., Zhang S., Cao J., Hood Z.D., Figueroa-Cosme L. Synthesis of CaO2 nanocrystals and their spherical aggregates with uniform sizes for use as a biodegradable bacteriostatic agent. Small. 2019;15 doi: 10.1002/smll.201902118. [DOI] [PubMed] [Google Scholar]
- 42.Garmett W.M. Leidenfrost' phenomenon. Nature. 1878;17:466. [Google Scholar]
- 43.Gottfried B.S., Bell K.J. Film boiling of spheroidal droplets. Leidenfrost phenomenon. Ind. Eng. Chem. Fund. 1966;5:561–568. [Google Scholar]
- 44.Henley R.W., Berger B.R. Self-ordering and complexity in epizonal mineral deposits. Annu. Rev. Earth Planet Sci. 2000;28:669–719. [Google Scholar]
- 45.Cintas P., Luche J.L. Green chemistry. The sonochemical approach. Green Chem. 1999;1:115–125. [Google Scholar]
- 46.Yang B., Chen Y., Shi J. Reactive oxygen species (ROS)-Based nanomedicine. Chem. Rev. 2019;119:4881–4985. doi: 10.1021/acs.chemrev.8b00626. [DOI] [PubMed] [Google Scholar]
- 47.Murphy M.P. How mitochondria produce reactive oxygen species. Biochem. J. 2009;417:1–13. doi: 10.1042/BJ20081386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Apel K., Hirt H. Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004;55:373–399. doi: 10.1146/annurev.arplant.55.031903.141701. [DOI] [PubMed] [Google Scholar]
- 49.Balaban R.S., Nemoto S., Finkel T. Mitochondria, oxidants, and aging. Cell. 2005;120:483–495. doi: 10.1016/j.cell.2005.02.001. [DOI] [PubMed] [Google Scholar]
- 50.Kolenko V., Teper E., Kutikov A., Uzzo R. Zinc and zinc transporters in prostate carcinogenesis. Nat. Rev. Urol. 2013;10:219–226. doi: 10.1038/nrurol.2013.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Franklin R.B., Costello L.C. Zinc as an anti-tumor agent in prostate cancer and in other cancers. Arch. Biochem. Biophys. 2007;463:211–217. doi: 10.1016/j.abb.2007.02.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fan W., Huang P., Chen X. Overcoming the Achilles' heel of photodynamic therapy. Chem. Soc. Rev. 2016;45:6488–6519. doi: 10.1039/c6cs00616g. [DOI] [PubMed] [Google Scholar]
- 53.Zhou Z., Song J., Nie L., Chen X. Reactive oxygen species generating systems meeting challenges of photodynamic cancer therapy. Chem. Soc. Rev. 2016;45:6597–6626. doi: 10.1039/c6cs00271d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Harris A.L. Hypoxia - a key regulatory factor in tumour growth. Nat. Rev. Canc. 2002;2:38–47. doi: 10.1038/nrc704. [DOI] [PubMed] [Google Scholar]
- 55.Fingar V.H. The role of microvascular damage in photodynamic therapy: the effect of treatment on vessel constriction, permeability, and leukocyte adhesion. Canc. Res. 1992;52:4914–4921. [PubMed] [Google Scholar]
- 56.Shannon A.M., Bouchier-Hayes D.J., Condron C.M., Toomey D. Tumour hypoxia, chemotherapeutic resistance and hypoxia-related therapies. Canc. Treat Rev. 2003;29:297–307. doi: 10.1016/s0305-7372(03)00003-3. [DOI] [PubMed] [Google Scholar]
- 57.Boddien A., Gartner F., Federsel C., Sponholz P., Mellmann D., Jackstell R. CO2 -"neutral" hydrogen storage based on bicarbonates and formates. Angew. Chem. Int. Ed. 2011;50:6411–6414. doi: 10.1002/anie.201101995. [DOI] [PubMed] [Google Scholar]
- 58.Zhao M., Xu Y., Xie M., Zou L., Wang Z., Liu S. Halogenated Aza‐BODIPY for imaging‐guided synergistic photodynamic and photothermal tumor therapy. Adv. Healthc. Mater. 2018;7:1800606. doi: 10.1002/adhm.201800606. [DOI] [PubMed] [Google Scholar]
- 59.Fang J.S., Gillies R.D., Gatenby R.A. Adaptation to hypoxia and acidosis in carcinogenesis and tumor progression. Semin. Canc. Biol. 2008;18:330–337. doi: 10.1016/j.semcancer.2008.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yuan J., Cen Y., Kong X.J., Wu S., Chu X. MnO2-nanosheet-modified upconversion nanosystem for sensitive turn-on fluorescence detection of H2O2 and glucose in blood. ACS Appl. Mater. Interfaces. 2015;7:10548. doi: 10.1021/acsami.5b02188. [DOI] [PubMed] [Google Scholar]
- 61.Zhongmin T., Yanyan L., Mingyuan H., Wenbo B. Chemodynamic therapy : tumour microenvironment‐mediated Fenton and Fenton‐like reactions. Angew. Chem. 2019;131 doi: 10.1002/anie.201805664. 985-968. [DOI] [PubMed] [Google Scholar]
- 62.Ranji-Burachaloo H., Gurr P.A., Dunstan D.E., Qiao G.G. Cancer treatment through nanoparticle facilitated fenton reaction. ACS Nano. 2018;12:11819–11837. doi: 10.1021/acsnano.8b07635. [DOI] [PubMed] [Google Scholar]
- 63.Lin L.S., Song J., Song L., Ke K., Liu Y. Simultaneous Fenton‐like ion delivery and glutathione depletion by MnO2‐based nanoagent to enhance chemodynamic therapy. Angew. Chem. 2017;57:4902–4906. doi: 10.1002/anie.201712027. [DOI] [PubMed] [Google Scholar]
- 64.Yuanlin W., Zhenglin L., Ying H., Jing L., Mengyu G., Hengxiang W. Photothermal conversion-coordinated Fenton-like and photocatalytic reactions of Cu2-xSe-Au Janus nanoparticles for tri-combination antitumor therapy. Biomaterials. 2020;255:120167. doi: 10.1016/j.biomaterials.2020.120167. [DOI] [PubMed] [Google Scholar]
- 65.Fernandez J., Maruthamuthu P., Renken A., Kiwi J. Bleaching and photobleaching of orange II within seconds by the oxone/Co2+ reagent in Fenton-like processes. Appl. Catal. B Environ. 2004;49:207–215. [Google Scholar]
- 66.Lingling Z., Jiulong Z., Yongkang C., Yuting Z., Jinfeng L., Jiayan Z. MoS2-ALG-Fe/GOx hydrogel with Fenton catalytic activity for combined cancer photothermal, starvation, and chemodynamic therapy. Colloids Surf., B. 2020;195:111243. doi: 10.1016/j.colsurfb.2020.111243. [DOI] [PubMed] [Google Scholar]
- 67.Maruyama K. Intracellular targeting delivery of liposomal drugs to solid tumors based on EPR effects. Adv. Drug Deliv. Rev. 2011;63:161–169. doi: 10.1016/j.addr.2010.09.003. [DOI] [PubMed] [Google Scholar]
- 68.Fehrenbacher N., Jäättelä M. Lysosomes as targets for cancer therapy. Canc. Res. 2005;65:2993–2995. doi: 10.1158/0008-5472.CAN-05-0476. [DOI] [PubMed] [Google Scholar]
- 69.Boya Kroemer. Lysosomal membrane permeabilization in cell death. Oncogene. 2008;27:6434–6451. doi: 10.1038/onc.2008.310. [DOI] [PubMed] [Google Scholar]
- 70.Tan Q., Saggar J.K., Yu M., Wang M., Tannock I.F. Mechanisms of drug resistance related to the microenvironment of solid tumors and possible strategies to inhibit them. Canc. J. 2015;21:254–262. doi: 10.1097/PPO.0000000000000131. [DOI] [PubMed] [Google Scholar]
- 71.Tredan O., Galmarini C.M., Patel K., Tannock I.F. Drug resistance and the solid tumor microenvironment. J. Natl. Cancer Inst. (Bethesda) 2007;99:1441–1454. doi: 10.1093/jnci/djm135. [DOI] [PubMed] [Google Scholar]
- 72.Kietzmann T., Gorlach A. Reactive oxygen species in the control of hypoxia-inducible factor-mediated gene expression. Semin. Cell Dev. Biol. 2005;16:474–486. doi: 10.1016/j.semcdb.2005.03.010. [DOI] [PubMed] [Google Scholar]
- 73.Li W., Kong A.N. Molecular mechanisms of Nrf2-mediated antioxidant response. Mol. Carcinog. 2010;48:91–104. doi: 10.1002/mc.20465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Huang C.C., Chia W.T., Chung M.F., Lin K.J., Hsiao C.W., Jin C. An implantable depot that can generate oxygen in situ for overcoming hypoxia-induced resistance to anticancer drugs in chemotherapy. J. Am. Chem. Soc. 2016;138:5222–5225. doi: 10.1021/jacs.6b01784. [DOI] [PubMed] [Google Scholar]
- 75.Fu L.H., Hu Y.R., Qi C., He T., Jiang S., Jiang C. Biodegradable manganese-doped calcium phosphate nanotheranostics for traceable cascade reaction-enhanced anti-tumor therapy. ACS Nano. 2019;13:13985–13994. doi: 10.1021/acsnano.9b05836. [DOI] [PubMed] [Google Scholar]
- 76.Zhang L., Wan S.S., Li C.X., Xu L., Cheng H., Zhang X.Z. An adenosine triphosphate-responsive autocatalytic fenton nanoparticle for tumor ablation with self-supplied H2O2 and acceleration of Fe(III)/Fe(II) conversion. Nano Lett. 2018;18:7609–7618. doi: 10.1021/acs.nanolett.8b03178. [DOI] [PubMed] [Google Scholar]
- 77.Qi C., He J., Fu L.-H., He T., Blum N.T., Yao X. Tumor-specific activatable nanocarriers with gas-generation and signal amplification capabilities for tumor theranostics. ACS Nano. 2020 doi: 10.1021/acsnano.0c09223. [DOI] [PubMed] [Google Scholar]
- 78.Sitkovsky M.V., Hatfield S., Abbott R., Belikoff B., Lukashev D., Ohta A. Hostile, hypoxia-A2-adenosinergic tumor biology as the next barrier to overcome for tumor immunologists. Canc. Immunol. Res. 2014;2:598–605. doi: 10.1158/2326-6066.CIR-14-0075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Pantngrag-Brown L. Calcification and fibrosis in mesenteric carcinoid tumor: CT findings and pathologic correlation. Am. J. Roentgenol. 1995;164:387–391. doi: 10.2214/ajr.164.2.7839976. [DOI] [PubMed] [Google Scholar]
- 80.Dershaw D.D., Giess C.S., Mccormick B., Borgen P., Liberman L., Abramson A.F. Patterns of mammographically detected calcifications after breast-conserving therapy associated with tumor recurrence. Cancer. 2015;79:1355–1361. doi: 10.1002/(sici)1097-0142(19970401)79:7<1355::aid-cncr13>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- 81.Orrenius S., Zhivotovsky B., Nicotera P. Regulation of cell death: the calcium-apoptosis link. Nat. Rev. Mol. Cell Biol. 2003;4:552–565. doi: 10.1038/nrm1150. [DOI] [PubMed] [Google Scholar]
- 82.Rui X., Yang Y., Wu J., Chen J., Chen Q., Ren R. Multi-path tumor inhibition via the interactive effects between tumor microenvironment and an oxygen self-supplying delivery system for a photosensitizer. Photodiagn. Photodyn. 2020;29:101642. doi: 10.1016/j.pdpdt.2019.101642. [DOI] [PubMed] [Google Scholar]
- 83.Lux F., Sancey L., Bianchi A., Crémillieux Y., Roux S., Tillement O. Gadolinium-based nanoparticles for theranostic MRI-radiosensitization. Nanomedicine. 2015;10:1801–1815. doi: 10.2217/nnm.15.30. [DOI] [PubMed] [Google Scholar]
- 84.Quayle J.M., Standen N.B., Stanfield P.R. The voltage-dependent block of ATP-sensitive potassium channels of frog skeletal muscle by caesium and barium ions. J. Physiol. (Lond.) 1988;405:677–697. doi: 10.1113/jphysiol.1988.sp017355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bhoelan B.S., Stevering C.H., d B Van A.T.J., d H Van M.A.G. Barium toxicity and the role of the potassium inward rectifier current. Clin. Toxicol. 2014;52:584–593. doi: 10.3109/15563650.2014.923903. [DOI] [PubMed] [Google Scholar]
- 86.Prevarskaya N., Skryma R., Shuba Y. Ion channels and the hallmarks of cancer. Trends Mol. Med. 2010;16:107–121. doi: 10.1016/j.molmed.2010.01.005. [DOI] [PubMed] [Google Scholar]
- 87.Sinicropi M.S., Amantea D., Caruso A., Saturnino C. Chemical and biological properties of toxic metals and use of chelating agents for the pharmacological treatment of metal poisoning. Arch. Toxicol. 2010;84:501–520. doi: 10.1007/s00204-010-0544-6. [DOI] [PubMed] [Google Scholar]
- 88.Yu W., Liu R., Zhou Y., Gao H. Size-tunable strategies for a tumor targeted drug delivery system. ACS Cent. Sci. 2020;6:100–116. doi: 10.1021/acscentsci.9b01139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Sun Q., Ojha T., Kiessling F., Lammers T., Shi Y. Enhancing tumor penetration of nanomedicines. Biomacromolecules. 2017;18:1449–1459. doi: 10.1021/acs.biomac.7b00068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Chen H., Zhang W., Zhu G., Xie J., Chen X. Rethinking cancer nanotheranostics. Nat. Rev. Mater. 2017;2:17024. doi: 10.1038/natrevmats.2017.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Perrot I., Michaud H.A., Giraudon-Paoli M., Augier S., Docquier A., Gros L. Blocking antibodies targeting the CD39/CD73 immunosuppressive pathway unleash immune responses in combination cancer therapies, Cell. For. Rep. 2019;27:2411–2425. doi: 10.1016/j.celrep.2019.04.091. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.











