Abstract
More than half of stroke patients live with different levels of motor dysfunction after receiving routine rehabilitation treatments. Therefore, new rehabilitation technologies are urgently needed as auxiliary treatments for motor rehabilitation. Based on routine rehabilitation treatments, a new intelligent rehabilitation platform has been developed for accurate evaluation of function and rehabilitation training. The emerging intelligent rehabilitation techniques can promote the development of motor function rehabilitation in terms of informatization, standardization, and intelligence. Traditional assessment methods are mostly subjective, depending on the experience and expertise of clinicians, and lack standardization and precision. It is therefore difficult to track functional changes during the rehabilitation process. Emerging intelligent rehabilitation techniques provide objective and accurate functional assessment for stroke patients that can promote improvement of clinical guidance for treatment. Artificial intelligence and neural networks play a critical role in intelligent rehabilitation. Multiple novel techniques, such as brain-computer interfaces, virtual reality, neural circuit-magnetic stimulation, and robot-assisted therapy, have been widely used in the clinic. This review summarizes the emerging intelligent rehabilitation techniques for the evaluation and treatment of motor dysfunction caused by nervous system diseases.
Keywords: brain, central nerve, injury, nerve, nerve function, neurogenesis, plasticity, repair, spinal cord
Introduction
Motor dysfunction results from a wide range of neurological diseases, such as stroke and spinal cord injury, and it brings tremendous economic and social burdens to sufferers and the community. Patients with motor dysfunction generally suffer from limited mobility and impaired normal daily activities and they have a high demand for rehabilitation training assistance. Despite substantial improvements in motor rehabilitation, residual disability and persisting neurological deficits remain a major medical concern. Approximately one-third of stroke patients experience a permanent motor deficit that impacts their daily activities (Dimyan and Cohen, 2011).
The understanding of brain plasticity and the development of artificial intelligence technology can play a critical role in functional motor recovery. Brain plasticity is the ability of the human brain to adapt to change and environmental stimuli, such as brain damage, therapeutic treatment and experiences, by reorganizing its structure, function, and connections (Cramer et al., 2011). The basic structural reserve and the anatomical plasticity of the brain is an important parameter for significant motor recovery (Di Pino et al., 2014). Recently, a new intelligent rehabilitation platform has been developed based on conventional rehabilitation treatments for rehabilitation training and accurate evaluation of progress. Emerging intelligent rehabilitation technologies can promote the development of rehabilitation in terms of informatization, standardization and intelligence. Various advanced intelligent technologies, such as brain-computer interfaces (BCI), virtual reality (VR), neural circuit-magnetic stimulation, and robot-assisted therapy, are currently being developed to promote functional assessment and effective rehabilitation treatments for patients with motor dysfunction. Accurate evaluation of the degree of impairment following motor dysfunction is important for assessing the efficacy of rehabilitation treatment, determining the most effective treatment interventions, and predicting functional recovery. In this review, we focus on advances in intelligent rehabilitation techniques and their efficacy in treating motor impairment.
Search Strategy and Selection Criteria
All available articles concerning intelligent rehabilitation, functional assessment, and motor dysfunction were obtained by searching the major literature databases, Google Scholar, Web of Science, PubMed, and the IEE Electronic Library. Keywords for searching: rehabilitation, motor dysfunction, stroke, spinal cord injury (SCI), functional assessment, motor recovery, neurological biomarker, electrophysiological, biomechanical, rehabilitation training, functional electrical stimulation (FES), functional magnetic stimulation, biofeedback, neurofeedback, robot-assisted, brain-computer interface (BCI), virtual reality (VR), intelligent rehabilitation, stem cell, and various combinations of the above terms. The search dates were set for articles published between 2005 and 2020.
Advances in Assessment of Motor Function Rehabilitation
With the growing emphasis on evidence-based clinical practice, it has become increasingly important to develop objective and efficient methods to precisely assess the functional deficits of patients with motor dysfunction. An accurate and quantitative assessment system for evaluating and predicting the functional status of patients is necessary for realistic rehabilitation goal-setting and for efficient allocation of treatment resources following motor dysfunction. At present, different subjective assessment scales are applied by clinicians that mainly involve assessment of neurological deficits (National Institutes of Health Stroke Scale), motor pattern (Brunnstrom recovery states), motor performance (Fugl-Meyer assessment), balance (Berg Balance Scale), and activities of daily living (Barthel Index). The reliability of these scales depends largely on the experience and expertise of clinicians. The inherent subjectivity of these assessment scales makes it difficult to accurately and objectively reflect the functional performance of patients. Thus, it is hard to follow functional changes in the rehabilitation process and to adjust rehabilitation treatment accordingly.
Objective assessment methods, such as biomechanical tests, electrophysiological measurements, and neuroimaging have been gradually developed and clinically applied to supplement and improve traditional assessment. These methods now constitute a new field for functional assessment of motor rehabilitation. Based on biomechanics, muscle strength can be objectively quantified under isometric conditions to clarify the degree of motor deficits in stroke individuals (Rabelo et al., 2016). The electrophysiology techniques of electromyography, mechanomyography, and motor evoked potentials can be used in the clinic to objectively evaluate neuromuscular condition. Generally, the presence or absence of motor evoked potentials in paretic limbs within a few hours or days following motor dysfunction is associated with recovery outcomes (Di Pino et al., 2014). Visual tracking systems with digital recording and image analysis, mainly collect parameters such as surface electromyography signals, trajectory of movement, joint angels, and joint angular velocity to objectively assess the motor function of stroke individuals (Taniguchi et al., 2015).
Neurological biomarkers derived from neuroimaging technologies have more prognostic and predictive value for motor recovery than clinical behavioral biomarkers (Borich et al., 2014; Bernhardt et al., 2016). As a non-invasive probe of brain anatomy, function and physiology, magnetic resonance imaging (MRI) is a highly versatile modality that can provide detailed biophysical information of lesion areas and plays a crucial part in assessing motor dysfunction recovery (Macintosh and Graham, 2013). A study based on functional MRI (fMRI) showed that disruption of inter-hemispheric functional connectivity in the somatomotor network was significantly correlated with upper extremity impairment in stroke patients (Carter et al., 2010). Analysis of inter- or intra-hemispheric connectivity by fMRI has been clinically applied to investigate the effect of stroke on the cerebral network and to assess the health of brain networks, with implications for prognosis and recovery from stroke (Zhao et al., 2016). The structural integrity of white matter pathways can be detected by diffusion-weighted MRI. Greater loss of ipsilesional tract integrity, at the level of the posterior limb of the internal capsule is associated with worse motor outcome for stroke patients (Puig et al., 2017). Accumulating evidence shows that the extent of damage to corticospinal tract integrity measured by diffusion tensor imaging is a key biomarker for predicting the prognosis of motor function after stroke (Puig et al., 2017). In recent years, functional near-infrared spectroscopy (fNIRS), an optical neuroimaging technique, has enabled the noninvasive capture of neural activation based on hemodynamic responses (Ferrari and Quaresima, 2012). Compared with fMRI, the unique advantages of fNIRS (continuity, dynamic monitoring, resistance to motion artifacts) make it possible to detect brain reorganization patterns during functional motor tasks to evaluate the effect of treatment on rehabilitation (Petracca et al., 2015). A fNIRS study showed an altered pattern of activation in the contralesional and ipsilesional motor cortices following stroke (Huo et al., 2019). A greater understanding of fNIRS-based cerebral reorganization will open a new field for motor functional assessment following motor dysfunction.
In summary, the time after the onset of stroke, lesion location, and integrity of corticospinal tracts are all key factors that significantly influence functional outcome after stroke. A combination of neurological biomarkers and clinical outcomes has been proposed to enable accurate clinical evaluation and prognosis after stroke (Stinear et al., 2017). Clinical databases that archive motor function tests, detailed imaging information, and neurophysiological data may benefit the accurate prognosis and treatment decisions for individuals with motor dysfunction (Puig et al., 2017). In the future, based on clinical functional scales, electrophysiological tests, biomechanical tests, and neuroimaging tests, a multimodal assessment method combined the central-peripheral information can be developed to precisely determine functional deficits of patients with motor dysfunction (Figure 1). Such a multimodal functional assessment method will enable accurate prognosis and guide treatment choice for rehabilitation, and predict functional recovery after motor dysfunction.
Figure 1.
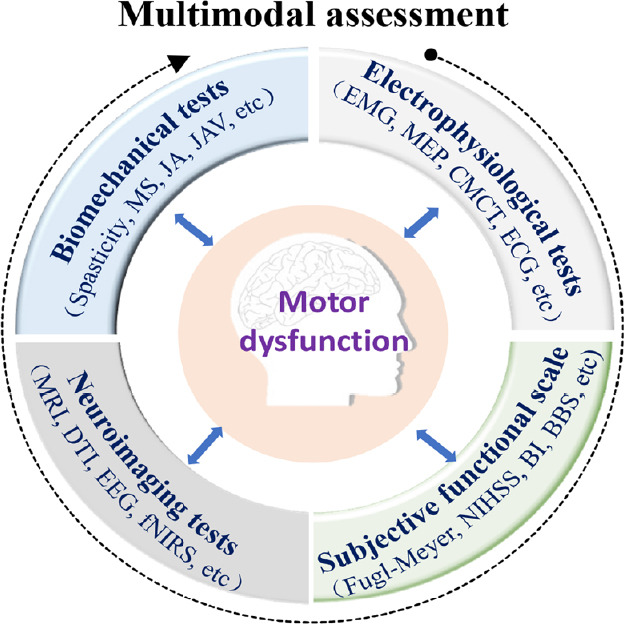
Multimodal assessment combines the clinical functional scale and electrophysiological, biomechanical, and neuroimaging tests.
BBS: Berg Balance Scale; BI: Barthel Index; CMCT: central motor conduction time; DTI: diffusion tensor imaging; ECG: electrocardiogram; EEG: elecroencephalogram; EMG: electromyogram; fNIRS: functional near-infrared spectroscopy; JA: joint angels; JAV: joint angular velocity; MEP: motor evoked potentials; MRI: magnetic resonance imaging; MS: muscle strength; NIHSS: National Institutes of Health Stroke Scale.
Advances in Motor Function Rehabilitation Training
Recovery of motor dysfunction is a complex process, which is not only related to the ability to perform movement and to successfully complete a task to the pre-injury standard, but is also associated with functional restoration in neural tissue (Levin et al., 2009). Neuroplasticity is the basic mechanism underlying improvement in functional outcome after stroke (Pekna et al., 2012). Therefore, the focus of motor dysfunction rehabilitation is the recovery of the affected neuromuscular functions, establishment of neural circuits of sensation-motion and achievement of independent body control (Resquín et al., 2016). At present, constraint-induced therapy (Bang et al., 2018), bilateral training (Coupar et al., 2010), motor imagery (Langhorne et al., 2009), mirror therapy (Bai et al., 2019), and treadmill training plus body-weight support (Huang et al., 2019) are common rehabilitation therapies that improve motor capabilities of patients with motor dysfunction. A combination of task-specific training therapies remains the gold standard treatment for post-stroke motor rehabilitation (Dimyan and Cohen, 2011). However, positive results of motor rehabilitation are limited. Approximately one-third of stroke patients experience a permanent motor deficit that impacts their daily activities (Dimyan and Cohen, 2011). Therefore, developing more effective therapies that target motor function is of great importance for motor dysfunction rehabilitation.
FES
Somatosensory input is necessary for accurate motor performance and effective motor learning (Lai et al., 2016). Recently, FES over peripheral structures showed potential to augment the effect of sensory afferent inputs to the central nervous system and to concurrently induce increased motor cortex excitability (Christiansen and Perez, 2018). FES is gaining popularity as an effective stroke rehabilitation therapy to artificially activate the sensorimotor system (Po-pović, 2014). It has also been proposed as a possible adjuvant strategy capable of improving spasticity and balance in patients with stroke (Cho et al., 2013). Motor training is best performed when the stroke-affected motor cortex is excitable and is in a higher functional connectivity state with the muscles. Given that plastic changes induced by FES can last for 30–110 minutes (Lai et al., 2016), peripheral FES can be integrated within a conventional stroke rehabilitation program to enhance motor recovery in patients with stroke (Salhab et al., 2016). Somatosensory stimulation of the paretic hand immediately prior to physical practice can enhance the training of functional hand tasks in patients with chronic stroke (Conrad et al., 2011). The therapeutic effects of this combination rehabilitation protocol are promising and contribute to consolidation of rehabilitative treatment effects.
Functional magnetic stimulation
Neuromodulation has been a fast-growing discipline during the past two decades and has radically changed the treat-ment of several nervous system disorders (Staudt et al., 2019). Neuromodulation technology generates therapeutic effects on CNS signal transmission, exciting, inhibiting or regulating neuronal and neural network activities (Blackmore et al., 2019). Transcranial magnetic stimulation (TMS) as a non-invasive brain stimulation technique that has been widely used because of its advantages of no pain, no damage, safety, reliability, and ease of operation. As an exogenous stimulus, TMS can safely affect brain metabolism, nerve conduction (Rossi et al., 2009), cortical neuron action potentials, and can modify the state of tissue excitability. The accumulated effect of repeated stimulation produces artificially induced action potentials to promote axoplasmic transport, thereby improving brain metabolism and stimulating neural plasticity (Valero-Cabré et al., 2007).
The imbalance of interhemispheric inhibition between contralesional and ipsilesional hemispheres after stroke is correlated with motor functional performance (Dodd et al., 2017). Motor improvement may be facilitated by the reduction of excessive inhibition from the contralesional to the ipsilesional hemisphere. Thus, TMS protocols commonly use low-frequency repeated TMS (< 1 Hz) applied to the unaffected side to suppress local neural activities, and high-frequency repeated TMS (> 10 Hz) applied to the affected side to activate them. A combination protocol of TMS and occupational therapy can maximize brain plasticity and produce beneficial functional reorganization in the affected hemisphere after stroke (Yamada et al., 2013). The involvement of multiple descending motor pathways plays a critical role in functional motor recovery following motor dysfunction, the corticospinal tracts being one of the most important pathways. In patients with chronic incomplete spinal cord injury, a targeted TMS protocol based on the principle of spike-timing dependent plasticity, induced the plasticity of residual corticospinal projections and spontaneously increased motor output to enhance motor function (Christiansen and Perez, 2018). Animal experiments showed that magnetic stimulation after spinal cord injury can protect spinal nerve tissue and promote the regeneration of nerve fibers to achieve nerve reinnervation of the damaged limbs. With a TMS protocol of 10 Hz repeated for 8 weeks, the movements of rats with T10–11 damage were significantly improved and were related to a higher density of serotonergic fibers (Poirrier et al., 2004) and increased expression of Nestin in the damaged spinal cord tissue (Cullen and Young, 2016).
Neural circuit reconstruction is the process of nerve repair and regeneration and it is an important way to achieve functional reconstruction after nerve damage. For motor dysfunction resulting from central nervous system injury, the goal of neural circuit reconstruction is to achieve recanalization of sensation-motion circuits, including functional repair or remodeling of autonomous and involuntary movements, shallow sensations and proprioception, and autonomic nerves. Neural circuit reconstruction depends not only on local nerve regeneration, but also on effective stimulation of the remaining nerve fibers in the damaged area and maximization of their function. In this theoretical framework, nerve circuit stimulation should not be limited to cortical stimulation. More multi-target, multimodal progressive repetitive intervention is needed for sensorimotor circuit reconstruction. The establishment of a task-oriented functional magnetic stimulation rehabilitation mode, combining magnetic stimulation technology with rehabilitation tasks, would produce more encouraging results. However, it is necessary for stimulation to be accurate and the precision of rehabilitation can be realized using neurophysiological assessment and neuroimaging techniques. It is noticeable that only when longer duration interventions were given, could long-lasting effects be achieved with possible mechanisms detected at both the spinal cord and cerebral cortex levels.
Biofeedback-based rehabilitation training
Biofeedback-based interventions mainly integrate task-dependent physical therapy and cognitive stimuli within an interactive and multimodal environment, and are gaining interest in motor dysfunction rehabilitation. Generally, bio-feedback is defined as a modality that provides information about subconscious body processes to make patients con-sciously modify the movement process (Wing, 2001). The procedure utilizes electronic equipment to continuously monitor a selected neurophysiological response (e.g., heart rate, breathing rate, muscle tension, or brain activity) and to then convert the measurements of these responses into a visual or auditory representation for the individual. The individual can then easily perceive the information and can be asked to modify their performance accordingly to achieve better predefined results. The rehabilitation system with biofeedback enabled the measurable evaluation of performance and generation of media-based feedback by quantifying active movement intention and muscle status to promote comprehensive improvement of several movements (Lehrer et al., 2011).
Surface electromyography, containing a wealth of physiological information, has been widely applied in biofeedback for stroke rehabilitation (Cesqui et al., 2013). Motor learning together with biofeedback provision can result in better retention of learned skills (Subramanian et al., 2010). Rehabilitation of hand function based on sensing and force-feedback can improve grasping capabilities with several objects (Ben-Tzvi et al., 2016). Kinesthetic haptic bio-feedback can also provide a beneficial therapeutic effect by improving balance in patients with stroke (Afzal et al., 2015). FES can elicit strong sensory input and enhance sensorimotor integration (Lai et al., 2016); however, FES alone for stroke rehabilitation lacks feedback information of the patient’s status. It is difficult to ensure a patient’s active participation during the process, leading to lower rehabilitation efficiency. A closed loop FES system using surface electromyography bias biofeedback from bilateral arms has been proposed for enhancing recovery of upper-limb function and rehabilitation efficiency. The stimulation process can be adjusted according to the neuromuscular states in real time and muscle fatigue can be effectively alleviated compared with constant cycling FES (Quandt and Hummel, 2014). Surface electromyography-triggered FES can induce corticospinal plasticity and shows an advantage over FES alone for motor improvement in the chronic stage of stroke (Fujiwara et al., 2009).
Neurofeedback therapy is a specialized form of biofeedback that targets circumscribed neural activation and provides visual representation about the measurements to patients. Neurofeedback information related to tasks enables patients to achieve self-modulation in real time during a motor process in stroke rehabilitation treatment (Wang et al., 2018). Recent studies have shown that fMRI (blood-oxygenation level-dependent signal), fNIRS (hemodynamics), elecroen-cephalogram (EEG) (brain waves), and magnetoencephalography are effective at reflecting brain information as a neurofeedback tool. Neural plasticity is guided in different ways depending on the feedback modality. The user can receive visual or auditory feedback about the brainwave activity associated with their efforts and can thus voluntarily self-modulate their brain activity to reach training goals. Neurofeedback training of brain rhythms for motor learning is likely to enhance motor recovery following stroke. Neurofeedback interventions based on fNIRS have enhanced motor recovery in post-stroke patients with hemiparesis (Mihara, 2018).
Advanced intelligence rehabilitation techniques
Robot-assisted therapy
Intensive motor rehabilitation can improve the motor function of patients with motor dysfunction, but the clinical demand for rehabilitation is currently greater than the supply. Therefore, there is a need for a service that allows motor dysfunction survivors to be less reliant upon a therapist for motor learning skills and to enable self-management of long-term disability. Over the past decade, several technological systems have been developed to facilitate independent rehabilitation, such as robot-assisted therapy. Commonly used robot-assisted devices for motor training include end-effector and exoskeleton types (Mehrholz and Pohl, 2012). End-effector robots have the advantage of easy setup. However, they suffer from the limited control of proximal limb joints. This disadvantage can result in abnormal movement patterns. In contrast, exoskeleton-type devices have robot axes aligned with the anatomical axes of the user, which provides direct control of individual joints and minimizes abnormal posture or movement. According to clinical needs, a real-time two-axis mirror robot system was developed as a simple add-on module for conventional mirror therapy with a closed feedback mechanism, which controls real-time movement of the hemiplegic arm (Beom et al., 2016). Diverse applications of robotic rehabilitation devices and their effectiveness at recovering arm and hand function have been reviewed (Huang et al., 2017). The most important advantage of robotic rehabilitation therapy is the ability to deliver high-dose and high-intensity training, making it useful for patients with motor disorders to intensify their therapy without placing excessive demands on therapists (Chang and Kim, 2013). However, there is limited evidence showing significant superior effectiveness of robotic therapy over conventional therapy (Huang et al., 2017). Currently, robot-assisted therapy in stroke rehabilitation is considered an adjunct tool rather than a substitute for conventional rehabilitation therapy and the patient-therapist relationship (Huang et al., 2017). Compared with mainly relying on robotic devices, the right combination of robot-assisted therapy and conventional rehabilitation is far more likely to achieve the desired effect of motor function recovery with flexible control of sensation to motion.
BCI
Advances in neuroscience and engineering technology have enabled BCI to become a new technique for neural function rehabilitation and assistive motor control for motor rehabilitation (Cervera et al., 2018). BCI is defined as a system that can measure brain activity and convert it into artificial output that replaces, restores, enhances, supplements, or improves central nervous system output and thereby drives external devices (Buch et al., 2008). BCI can directly decode the motor thinking information in the central nervous system and construct a brain-machine feedback loop to assist patients in rehabilitation training. The application of BCI technology to motor rehabilitation can not only enable motor dysfunction patients to regain the control of limbs, but to also establish an active closed-loop stimulus feedback to achieve motor function reconstruction. More importantly, BCI can monitor and feedback the functional activities of the central nervous system, making the process of active participation in rehabilitation training observable. This technology offers promising strategies to modulate neuroplasticity and has the potential to provide an enhanced rehabilitation treatment for patients who have lost limb movement control after stroke. The prolonged use of BCI training by stroke patients can induce clinical improvement of upper limb function (Mukaino et al., 2014) with plasticity changes in brain activity (Grosse-Wentrup et al., 2011). The volitional control of neuromagnetic activity recorded over central scalp regions can be achieved with BCI training after stroke and can promote motor improvement, muscle reactivation, and structural and functional reorganization of the ipsilesional hemisphere. Results of neurological activation and motor function recovery demonstrate that BCI technology can enhance the effect of stroke rehabilitation training and further verifies the effectiveness of BCI training for motor function recovery (Pichiorri et al., 2015). A BCI system based on EEG has recently been proposed as a therapeutic intervention for environmental interaction and motor rehabilitation in patients with severe hemiplegia (Zhang et al., 2017). Patients using EEG-BCIs volitionally generate patterns of brain activity that are detected by the BCI and translated into movement of the paretic limb by external effectors, thereby bridging the disconnection between intention to move and execution (Várkuti et al., 2013). A BCI-FES training platform has been designed for rehabilitation of motor dysfunction patients to improve the rehabilitation process. In this platform, the FES was driven by users’ motor intention detected from brain activities related to motor tasks (Jure et al., 2016). A recent study presented a platform with a complete sensorimotor closed-loop system that combined a motor imaging-based BCI, FES, and visual feedback technologies for post-stroke rehabilitation (Irimia et al., 2017). This system can be used as a novel tool to improve the rehabilitation process by detecting a patient’s motor intention, performing it by FES and finally receiving appropriate feedback (Jure et al., 2016). The application of effective rehabilitation technology will enhance motor capabilities to progress motor recovery following motor dysfunction.
VR-based rehabilitation
VR systems provide a multidimensional experience within an immersive, semi-immersive or non-immersive perspective, enabling users to interact with virtual simulated environments in stroke rehabilitation settings (Laver et al., 2015). VR-based rehabilitation intervention has been applied extensively to neurological diseases, and has generally achieved positive outcomes (Tak et al., 2015). VR systems capture participants’ movements, which are then visualized on the computer screen in different ways, a process called movement visualization. The major forms of movement visualization have been summarized as indirect, abstract and augmented reality (Ferreira Dos Santos et al., 2016). In stroke patients, movement visualization contributed to the observation of limb movement, which activated the mirror neuron system in the frontoparietal cortex area. Moreover, fMRI showed that mirror neuron activity could be enhanced in healthy volunteers during the movement observation task whether the virtual limb was presented on the screen or not (Modroño et al., 2019). These findings indicated the possible correlation between VR systems and the mirror neuron system. In most cases, the VR system was supplemented or integrated with other stroke rehabilitation approaches. The integration of kinetic-based VR and a cognitive strategy improved motor function and occupational performance in patients with chronic stroke (Faria et al., 2018). Kinetic-based VR and physical therapy was administered to chronic stroke patients to assess upper extremity function and results were compared with those of a group receiving only physical therapy; the motor function and active range of motion of the upper extremities were significantly improved (Aşkın et al., 2018). FES of the wrist and finger extensors was integrated into a VR based-wearable device to stimulate adequate contraction of wrist and digits extensors (Lee et al., 2018). VR has also been integrated within a BCI , which used elecroencephalography to detect the attempt of upper extremity movement in the brain, and synchronized electromyography of peripheral muscle activity (Vourvopoulos et al., 2019). The virtual motion of the upper extremity in VR depending on the patient’s volition benefited patients with severe impairment the most. VR-FES and BCI-VR both linked the central and peripheral nervous systems, forming a nervous loop and might facilitate cortical excitability and neuroplasticity. Analogously, constraint-induced movement therapy was delivered to chronic stroke patients at home via a VR game, which showed promising results without adverse events (Borstad et al., 2018). VR based-devices have been acknowledged to be effective for movement disorders as an adjunct therapy. The effect of rehabilitation approaches could be augmented and enhanced via VR training systems to achieve better outcomes for patients with neurological disease compared with isolated techniques alone. VR in combination with motor imagery could also elicit excitability in the motor cortex and reduced intracortical inhibition to regenerate nerves in patients after stroke (Im et al., 2016). Motor imagery using a VR system significantly enhanced cortical excitability. Proprioceptive neuromuscular facilitation was also combined with VR in stroke patients in randomized controlled trials; however, no significant differences in upper limb function among proprioceptive neuromuscular facilitation, VR, and VR-proprioceptive neuromuscular facilitation groups were seen (Junior et al., 2019). The immersive and amusing nature of VR systems can motivate individuals to devote more time to training. Future research will focus on the various types of VR training systems (customized or commercial) and VR training dosage.
Prospects and Challenges
Performing complex motor tasks requires high levels of attention and sensorimotor processing to integrate visual, proprioceptive, and somatosensory feedback information with motor output. In recovering stroke patients, this process involves the pathophysiological processes of neurological repair, synaptic plasticity, and neural circuit reconstruction. At present, clinical decisions for patients with motor dysfunction mainly rely on the knowledge and judgment of individual therapists. Additionally, the success of rehabilitation programs for chronic patients with severe hemiparesis remains limited. Patients with lack of volitional muscle activity are usually excluded from existing rehabilitation strategies because most of them rely on the existence of residual limb functionality (Daly et al., 2009). Recently, emerging biological technologies, such as stem cell therapy, have been applied to treat stroke. The therapeutic potential of stem cell therapy for stroke recovery, by improving neural regrowth and promoting brain plasticity, has been reviewed (Stem Cell Therapies as an Emerging Paradigm in Stroke Participants, 2009; Pekna et al., 2012). Stroke rehabilitation might be maximally effective by combining stem cell therapy with appropriate physical therapy to promote endogenous neural plasticity and improve behavioral outcomes (Hicks et al., 2007). The application of stem cells to reconstruct neural circuits after stroke is an important goal for motor function recovery. However, at present, substantial advances in understanding the basic physiology of stem cells are required before they can be routinely used to treat patients (Dihné et al., 2011).
With the introduction of intelligent technology in clinical settings, objective functional evaluation and effective treatments can be provided in real-time. Intelligent rehabilitation techniques, such as FES, neural circuit-magnetic stimulation, BCI, VR, and robot-assisted therapy show great potential for augmenting clinical rehabilitation. It should be emphasized that intelligent rehabilitation is currently an adjunct to rather than a substitute for conventional rehabilitation therapy. The combination of traditional rehabilitation therapies and intelligent therapies has the potential to enhance motor dysfunction rehabilitation and improve clinical efficiency. While some findings have highlighted the potential of intelligent rehabilitation treatments for motor dysfunction patients, there is currently no uniform standard or application guide for evaluation of treatment using intelligent rehabilitation technology. Improvements in signal acquisition hardware, validation, dissemination and reliability will promote the clinical application of rehabilitation technology. Well-designed studies with large numbers of participants that demonstrate superior efficacy for motor recovery are necessary to establish intelligence-based therapies as standard, integral therapeutic modalities in motor rehabilitation. Ongoing improvements of intelligent technology may open a door to routine clinical use.
Additional file: Open peer review report 1 (82.5KB, pdf) .
Footnotes
Conflicts of interest: The authors have no actual or potential conflicts of interest.
Financial support: This work was supported by the National Key Research and Development Project of China, No. 2020YFC2004200 (to ZYL), the National Natural Science Foundation of China, Nos. 61761166007 (to ZYL), 81772453 (to DSX), 81974358 (to DSX), 31771071 (to ZYL), and Fundamental Research Funds for Central Public Welfare Research Institutes, No. 118009001000160001 (to ZYL).
Copyright license agreement: The Copyright License Agreement has been signed by all authors before publication.
Plagiarism check: Checked twice by iThenticate.
Peer review: Externally peer reviewed.
Open peer reviewer: Xia Shen, Shanghai Yangzhi Rehabilitation Hospital, China.
P-Reviewer: Shen X; C-Editor: Zhao M; S-Editors: Yu J, Li CH; L-Editors: Yu J, Song LP; T-Editor: Jia Y
Funding: This work was supported by the National Key Research and Development Project of China, No. 2020YFC2004200 (to ZYL), the National Natural Science Foundation of China, Nos. 61761166007 (to ZYL), 81772453 (to DSX), 81974358 (to DSX), 31771071 (to ZYL), and Fundamental Research Funds for Central Public Welfare Research Institutes, No. 118009001000160001 (to ZYL).
References
- 1.Afzal MR, Byun HY, Oh MK, Yoon J. Effects of kinesthetic haptic feedback on standing stability of young healthy subjects and stroke patients. J Neuroeng Rehabil. 2015;12:27. doi: 10.1186/s12984-015-0020-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Aşkın A, Atar E, Koçyiğit H, Tosun A. Effects of Kinect-based virtual reality game training on upper extremity motor recovery in chronic stroke. Somatosens Mot Res. 2018;35:25–32. doi: 10.1080/08990220.2018.1444599. [DOI] [PubMed] [Google Scholar]
- 3.Bai Z, Zhang J, Zhang Z, Shu T, Niu W. Comparison between movement-based and task-based mirror therapies on improving upper limb functions in patients with stroke: a pilot randomized controlled trial. Front Neurol. 2019;10:288. doi: 10.3389/fneur.2019.00288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bang DH, Shin WS, Choi HS. Effects of modified constraint-induced movement therapy with trunk restraint in early stroke patients: A single-blinded, randomized, controlled, pilot trial. NeuroRehabilitation. 2018;42:29–35. doi: 10.3233/NRE-172176. [DOI] [PubMed] [Google Scholar]
- 5.Ben-Tzvi P, Danoff J, Ma Z. The design evolution of a sensing and force-feedback exoskeleton robotic glove for hand rehabilitation application. J Mech Robot. 2016;8:051019. [Google Scholar]
- 6.Beom J, Koh S, Nam HS, Kim W, Kim Y, Seo HG, Oh BM, Chung SG, Kim S. Robotic Mirror Therapy System for Functional Recovery of Hemiplegic Arms. J Vis Exp. 2016:54521. doi: 10.3791/54521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bernhardt J, Borschmann K, Boyd L, Thomas Carmichael S, Corbett D, Cramer SC, Hoffmann T, Kwakkel G, Savitz SI, Saposnik G, Walker M, Ward N. Moving rehabilitation research forward: Developing consensus statements for rehabilitation and recovery research. Int J Stroke. 2016;11:454–458. doi: 10.1177/1747493016643851. [DOI] [PubMed] [Google Scholar]
- 8.Blackmore J, Shrivastava S, Sallet J, Butler CR, Cleveland RO. Ultrasound Neuromodulation: A Review of Results, Mechanisms and Safety. Ultrasound Med Biol. 2019;45:1509–1536. doi: 10.1016/j.ultrasmedbio.2018.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Borich MR, Brown KE, Boyd LA. Motor skill learning is associated with diffusion characteristics of white matter in individuals with chronic stroke. J Neurol Phys Ther. 2014;38:151–160. doi: 10.1097/NPT.0b013e3182a3d353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Borstad AL, Crawfis R, Phillips K, Lowes LP, Maung D, McPherson R, Siles A, Worthen-Chaudhari L, Gauthier LV. In-Home Delivery of Constraint-Induced Movement Therapy via Virtual Reality Gaming. J Patient Cent Res Rev. 2018;5:6–17. doi: 10.17294/2330-0698.1550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Buch E, Weber C, Cohen LG, Braun C, Dimyan MA, Ard T, Mellinger J, Caria A, Soekadar S, Fourkas A, Birbaumer N. Think to move: a neuromagnetic brain-computer interface (BCI) system for chronic stroke. Stroke. 2008;39:910–917. doi: 10.1161/STROKEAHA.107.505313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Carter AR, Astafiev SV, Lang CE, Connor LT, Rengachary J, Strube MJ, Pope DL, Shulman GL, Corbetta M. Resting interhemispheric functional magnetic resonance imaging connectivity predicts performance after stroke. Ann Neurol. 2010;67:365–375. doi: 10.1002/ana.21905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cervera MA, Soekadar SR, Ushiba J, Millán JDR, Liu M, Birbaumer N, Garipelli G. Brain-computer interfaces for post-stroke motor rehabilitation: a meta-analysis. Ann Clin Transl Neurol. 2018;5:651–663. doi: 10.1002/acn3.544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cesqui B, Tropea P, Micera S, Krebs HI. EMG-based pattern recognition approach in post stroke robot-aided rehabilitation: a feasibility study. J Neuroeng Rehabil. 2013;10:75. doi: 10.1186/1743-0003-10-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chang WH, Kim YH. Robot-assisted therapy in stroke rehabilitation. J Stroke. 2013;15:174–181. doi: 10.5853/jos.2013.15.3.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cho HY, In TS, Cho KH, Song CH. A single trial of transcutaneous electrical nerve stimulation (TENS) improves spasticity and balance in patients with chronic stroke. Tohoku J Exp Med. 2013;229:187–193. doi: 10.1620/tjem.229.187. [DOI] [PubMed] [Google Scholar]
- 17.Christiansen L, Perez MA. Targeted-Plasticity in the Corticospinal Tract After Human Spinal Cord Injury. Neurotherapeutics. 2018;15:618–627. doi: 10.1007/s13311-018-0639-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Conrad MO, Scheidt RA, Schmit BD. Effects of wrist tendon vibration on targeted upper-arm movements in poststroke hemiparesis. Neurorehabil Neural Repair. 2011;25:61–70. doi: 10.1177/1545968310378507. [DOI] [PubMed] [Google Scholar]
- 19.Coupar F, Pollock A, van Wijck F, Morris J, Langhorne P. Simultaneous bilateral training for improving arm function after stroke. Cochrane Database Syst Rev. 2010;2010 doi: 10.1002/14651858.CD006432.pub2. CD006432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cramer SC, Sur M, Dobkin BH, O'Brien C, Sanger TD, Trojanowski JQ, Rumsey JM, Hicks R, Cameron J, Chen D, Chen WG, Cohen LG, deCharms C, Duffy CJ, Eden GF, Fetz EE, Filart R, Freund M, Grant SJ, Haber S, et al. Harnessing neuroplasticity for clinical applications. Brain. 2011;134:1591–1609. doi: 10.1093/brain/awr039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cullen CL, Young KM. How does transcranial magnetic stimulation influence glial cells in the central nervous system. Front Neural Circuits. 2016;10:26. doi: 10.3389/fncir.2016.00026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Daly JJ, Cheng R, Rogers J, Litinas K, Hrovat K, Dohring M. Feasibility of a new application of noninvasive Brain Computer Interface (BCI): a case study of training for recovery of volitional motor control after stroke. J Neurol Phys Ther. 2009;33:203–211. doi: 10.1097/NPT.0b013e3181c1fc0b. [DOI] [PubMed] [Google Scholar]
- 23.Di Pino G, Pellegrino G, Assenza G, Capone F, Ferreri F, Formica D, Ranieri F, Tombini M, Ziemann U, Rothwell JC, Di Lazzaro V. Modulation of brain plasticity in stroke: a novel model for neurorehabilitation. Nat Rev Neurol. 2014;10:597–608. doi: 10.1038/nrneurol.2014.162. [DOI] [PubMed] [Google Scholar]
- 24.Dihné M, Hartung HP, Seitz RJ. Restoring neuronal function after stroke by cell replacement: anatomic and functional considerations. Stroke. 2011;42:2342–2350. doi: 10.1161/STROKEAHA.111.613422. [DOI] [PubMed] [Google Scholar]
- 25.Dimyan MA, Cohen LG. Neuroplasticity in the context of motor rehabilitation after stroke. Nat Rev Neurol. 2011;7:76–85. doi: 10.1038/nrneurol.2010.200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dodd KC, Nair VA, Prabhakaran V. Role of the contralesional vs. ipsilesional hemisphere in stroke recovery. Front Hum Neurosci. 2017;11:469. doi: 10.3389/fnhum.2017.00469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Faria AL, Cameirão MS, Couras JF, Aguiar JRO, Costa GM, Bermúdez IBS. Combined cognitive-motor rehabilitation in virtual reality improves motor outcomes in chronic stroke - a pilot study. Front Psychol. 2018;9:854. doi: 10.3389/fpsyg.2018.00854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ferrari M, Quaresima V. A brief review on the history of human functional near-infrared spectroscopy (fNIRS) development and fields of application. Neuroimage. 2012;63:921–935. doi: 10.1016/j.neuroimage.2012.03.049. [DOI] [PubMed] [Google Scholar]
- 29.Ferreira Dos Santos L, Christ O, Mate K, Schmidt H, Krüger J, Dohle C. Movement visualisation in virtual reality rehabilitation of the lower limb: a systematic review. Biomed Eng Online. 2016;15:144. doi: 10.1186/s12938-016-0289-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fujiwara T, Kasashima Y, Honaga K, Muraoka Y, Tsuji T, Osu R, Hase K, Masakado Y, Liu M. Motor improvement and corticospinal modulation induced by hybrid assistive neuromuscular dynamic stimulation (HANDS) therapy in patients with chronic stroke. Neurorehabil Neural Repair. 2009;23:125–132. doi: 10.1177/1545968308321777. [DOI] [PubMed] [Google Scholar]
- 31.Grosse-Wentrup M, Mattia D, Oweiss K. Using brain-computer interfaces to induce neural plasticity and restore function. J Neural Eng. 2011;8:025004. doi: 10.1088/1741-2560/8/2/025004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hicks AU, Hewlett K, Windle V, Chernenko G, Ploughman M, Jolkkonen J, Weiss S, Corbett D. Enriched environment enhances transplanted subventricular zone stem cell migration and functional recovery after stroke. Neuroscience. 2007;146:31–40. doi: 10.1016/j.neuroscience.2007.01.020. [DOI] [PubMed] [Google Scholar]
- 33.Huang S, Yu X, Lu Y, Qiao J, Wang H, Jiang LM, Wu X, Niu W. Body weight support-Tai Chi footwork for balance of stroke survivors with fear of falling: A pilot randomized controlled trial. Complement Ther Clin Pract. 2019;37:140–147. doi: 10.1016/j.ctcp.2019.101061. [DOI] [PubMed] [Google Scholar]
- 34.Huang X, Naghdy F, Naghdy G, Du H, Todd C. Robot-assisted post-stroke motion rehabilitation in upper extremities: a survey. Int J Disabil Hum Dev. 2017;16:233–247. [Google Scholar]
- 35.Huo C, Xu G, Li Z, Lv Z, Liu Q, Li W, Ma H, Wang D, Fan Y. Limb linkage rehabilitation training-related changes in cortical activation and effective connectivity after stroke: A functional near-infrared spectroscopy study. Sci Rep. 2019;9:6226. doi: 10.1038/s41598-019-42674-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Im H, Ku J, Kim HJ, Kang YJ. Virtual reality-guided motor imagery increases corticomotor excitability in healthy volunteers and stroke patients. Ann Rehabil Med. 2016;40:420–431. doi: 10.5535/arm.2016.40.3.420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Irimia DC, Cho W, Ortner R, Allison BZ, Ignat BE, Edlinger G, Guger C. Brain-computer interfaces with multi-sensory feedback for stroke rehabilitation: a case study. Artif Organs. 2017;41:E178–184. doi: 10.1111/aor.13054. [DOI] [PubMed] [Google Scholar]
- 38.Junior V, Santos MS, Ribeiro N, Maldonado IL. Combining proprioceptive neuromuscular facilitation and virtual reality for improving sensorimotor function in stroke survivors: a randomized clinical trial. J Cent Nerv Syst Dis. 2019;11:1179573519863826. doi: 10.1177/1179573519863826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jure FA, Carrere LC, Gentiletti GG, Tabernig CB. BCI-FES system for neuro-rehabilitation of stroke patients. J Phys Conf Ser. 2016;705:012058. [Google Scholar]
- 40.Lai MI, Pan LL, Tsai MW, Shih YF, Wei SH, Chou LW. Investigating the effects of peripheral electrical stimulation on corticomuscular functional connectivity stroke survivors. Top Stroke Rehabil. 2016;23:154–162. doi: 10.1080/10749357.2015.1122264. [DOI] [PubMed] [Google Scholar]
- 41.Langhorne P, Coupar F, Pollock A. Motor recovery after stroke: a systematic review. Lancet Neurol. 2009;8:741–754. doi: 10.1016/S1474-4422(09)70150-4. [DOI] [PubMed] [Google Scholar]
- 42.Laver KE, George S, Thomas S, Deutsch JE, Crotty M. Virtual reality for stroke rehabilitation. Cochrane Database Syst Rev. 2015 doi: 10.1002/14651858.CD008349.pub3. CD008349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lee SH, Lee JY, Kim MY, Jeon YJ, Kim S, Shin JH. Virtual reality rehabilitation with functional electrical stimulation improves upper extremity function in patients with chronic stroke: a pilot randomized controlled study. Arch Phys Med Rehabil. 2018;99:1447–1453e1. doi: 10.1016/j.apmr.2018.01.030. [DOI] [PubMed] [Google Scholar]
- 44.Lehrer N, Chen Y, Duff M, S LW, Rikakis T. Exploring the bases for a mixed reality stroke rehabilitation system, Part II: design of interactive feedback for upper limb rehabilitation. J Neuroeng Rehabil. 2011;8:54. doi: 10.1186/1743-0003-8-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Levin MF, Kleim JA, Wolf SL. What do motor “recovery” and “compensation” mean in patients following stroke. Neurorehabil Neural Repair. 2009;23:313–319. doi: 10.1177/1545968308328727. [DOI] [PubMed] [Google Scholar]
- 46.Macintosh BJ, Graham SJ. Magnetic resonance imaging to visualize stroke and characterize stroke recovery: a review. Front Neurol. 2013;4:60. doi: 10.3389/fneur.2013.00060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mehrholz J, Pohl M. Electromechanical-assisted gait training after stroke: a systematic review comparing end-effector and exoskeleton devices. J Rehabil Med. 2012;44:193–199. doi: 10.2340/16501977-0943. [DOI] [PubMed] [Google Scholar]
- 48.Mihara M. S3-1. Therapeutic intervention for neurological patients with neurofeedback technique. Clin Neurophysiol. 2018;129:e23. [Google Scholar]
- 49.Modroño C, Bermúdez S, Cameirão M, Pereira F, Paulino T, Marcano F, Hernández-Martín E, Plata-Bello J, Palenzuela N, Núñez-Pádron D, Pérez-González JM, González-Mora JL. Is it necessary to show virtual limbs in action observation neurorehabilitation systems. J Rehabil Assist Technol Eng 6:2055668319859140.? J Rehabil Assist Technol Eng. 2019;6:2055668319859140. doi: 10.1177/2055668319859140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mukaino M, Ono T, Shindo K, Fujiwara T, Ota T, Kimura A, Liu M, Ushiba J. Efficacy of brain-computer interface-driven neuromuscular electrical stimulation for chronic paresis after stroke. J Rehabil Med. 2014;46:378–382. doi: 10.2340/16501977-1785. [DOI] [PubMed] [Google Scholar]
- 51.Pekna M, Pekny M, Nilsson M. Modulation of neural plasticity as a basis for stroke rehabilitation. Stroke. 2012;43:2819–2828. doi: 10.1161/STROKEAHA.112.654228. [DOI] [PubMed] [Google Scholar]
- 52.Petracca A, Carrieri M, Avola D, Moro SB, Brigadoi S, Lancia S, Spezialetti M, Ferrari M, Quaresima V, Placidi G. 2015 International Conference on Virtual Rehabilitation (ICVR) Valencia, Spain: A virtual ball task driven by forearm movements for neuro-rehabilitation; pp. 162–163. [Google Scholar]
- 53.Pichiorri F, Morone G, Petti M, Toppi J, Pisotta I, Molinari M, Paolucci S, Inghilleri M, Astolfi L, Cincotti F, Mattia D. Brain-computer interface boosts motor imagery practice during stroke recovery. Ann Neurol. 2015;77:851–865. doi: 10.1002/ana.24390. [DOI] [PubMed] [Google Scholar]
- 54.Poirrier AL, Nyssen Y, Scholtes F, Multon S, Rinkin C, Weber G, Bouhy D, Brook G, Franzen R, Schoenen J. Repetitive transcranial magnetic stimulation improves open field locomotor recovery after low but not high thoracic spinal cord compression-injury in adult rats. J Neurosci Res. 2004;75:253–261. [Google Scholar]
- 55.Popović DB. Advances in functional electrical stimulation (FES) J Electromyogr Kinesiol. 2014;24:795–802. doi: 10.1016/j.jelekin.2014.09.008. [DOI] [PubMed] [Google Scholar]
- 56.Puig J, Blasco G, Schlaug G, Stinear CM, Daunis IEP, Biarnes C, Figueras J, Serena J, Hernández-Pérez M, Alberich-Bayarri A, Castellanos M, Liebeskind DS, Demchuk AM, Menon BK, Thomalla G, Nael K, Wintermark M, Pedraza S. Diffusion tensor imaging as a prognostic biomarker for motor recovery and rehabilitation after stroke. Neuroradiology. 2017;59:343–351. doi: 10.1007/s00234-017-1816-0. [DOI] [PubMed] [Google Scholar]
- 57.Quandt F, Hummel FC. The influence of functional electrical stimulation on hand motor recovery in stroke patients: a review. Exp Transl Stroke Med. 2014;6:9. doi: 10.1186/2040-7378-6-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Rabelo M, Nunes GS, da Costa Amante NM, de Noronha M, Fachin-Martins E. Reliability of muscle strength assessment in chronic post-stroke hemiparesis: a systematic review and meta-analysis. Top Stroke Rehabil. 2016;23:26–36. doi: 10.1179/1945511915Y.0000000008. [DOI] [PubMed] [Google Scholar]
- 59.Resquín F, Cuesta Gómez A, Gonzalez-Vargas J, Brunetti F, Torricelli D, Molina Rueda F, Cano de la Cuerda R, Miangolarra JC, Pons JL. Hybrid robotic systems for upper limb rehabilitation after stroke: A review. Med Eng Phys. 2016;38:1279–1288. doi: 10.1016/j.medengphy.2016.09.001. [DOI] [PubMed] [Google Scholar]
- 60.Rossi S, Hallett M, Rossini PM, Pascual-Leone A. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol. 2009;120:2008–2039. doi: 10.1016/j.clinph.2009.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Salhab G, Sarraj AR, Saleh S. 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC) Orlando, FL, USA: Mirror therapy combined with functional electrical stimulation for rehabilitation of stroke survivors' ankle dorsiflexion; pp. 4699–4702. [DOI] [PubMed] [Google Scholar]
- 62.Staudt MD, Herring EZ, Gao K, Miller JP, Sweet JA. Evolution in the Treatment of Psychiatric Disorders: From Psychosurgery to Psychopharmacology to Neuromodulation. Front Neurosci. 2019;13:108. doi: 10.3389/fnins.2019.00108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Stem Cell Therapies as an Emerging Paradigm in Stroke Participants (2009) Stem Cell Therapies as an Emerging Paradigm in Stroke (STEPS): bridging basic and clinical science for cellular and neurogenic factor therapy in treating stroke. Stroke. 40:510–515. doi: 10.1161/STROKEAHA.108.526863. [DOI] [PubMed] [Google Scholar]
- 64.Stinear CM, Byblow WD, Ackerley SJ, Smith MC, Borges VM, Barber PA. PREP2: A biomarker-based algorithm for predicting upper limb function after stroke. Ann Clin Transl Neurol. 2017;4:811–820. doi: 10.1002/acn3.488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Subramanian SK, Massie CL, Malcolm MP, Levin MF. Does provision of extrinsic feedback result in improved motor learning in the upper limb poststroke. A systematic review of the evidence. Neurorehabil Neural Repair. 2010;24:113–124. doi: 10.1177/1545968309349941. [DOI] [PubMed] [Google Scholar]
- 66.Tak S, Choi W, Lee S. Game-based virtual reality training improves sitting balance after spinal cord injury: a single-blinded, randomized controlled trial. Med Sci Tech. 2015;56:53–59. [Google Scholar]
- 67.Taniguchi K, Yu Y, Noma T, Hayash R, Matsumoto S, Shimodozono M, Kawahira K. 2015 IEEE International Conference on Mechatronics (ICM) Nagoya, Japan: Research of training and evaluation aid device with DOF selective constraint mechanism for hemiplegic upper limbs rehabilitation; pp. 448–453. [Google Scholar]
- 68.Valero-Cabré A, Payne BR, Pascual-Leone A. Opposite impact on 14C-2-deoxyglucose brain metabolism following patterns of high and low frequency repetitive transcranial magnetic stimulation in the posterior parietal cortex. Exp Brain Res. 2007;176:603–615. doi: 10.1007/s00221-006-0639-8. [DOI] [PubMed] [Google Scholar]
- 69.Várkuti B, Guan C, Pan Y, Phua KS, Ang KK, Kuah CW, Chua K, Ang BT, Birbaumer N, Sitaram R. Resting state changes in functional connectivity correlate with movement recovery for BCI and robot-assisted upper-extremity training after stroke. Neurorehabil Neural Repair. 2013;27:53–62. doi: 10.1177/1545968312445910. [DOI] [PubMed] [Google Scholar]
- 70.Vourvopoulos A, Pardo OM, Lefebvre S, Neureither M, Saldana D, Jahng E, Liew SL. Effects of a brain-computer interface with virtual reality (VR) neurofeedback: A pilot study in chronic stroke patients. Front Hum Neurosci. 2019;13:210. doi: 10.3389/fnhum.2019.00210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Wang T, Mantini D, Gillebert CR. The potential of real-time fMRI neurofeedback for stroke rehabilitation: A systematic review. Cortex. 2018;107:148–165. doi: 10.1016/j.cortex.2017.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wing K. Effect of neurofeedback on motor recovery of a patient with brain injury: a case study and its implications for stroke rehabilitation. Top Stroke Rehabil. 2001;8:45–53. doi: 10.1310/4G2F-5PLV-RNM9-BGGN. [DOI] [PubMed] [Google Scholar]
- 73.Yamada N, Kakuda W, Senoo A, Kondo T, Mitani S, Shimizu M, Abo M. Functional cortical reorganization after low-frequency repetitive transcranial magnetic stimulation plus intensive occupational therapy for upper limb hemiparesis: evaluation by functional magnetic resonance imaging in poststroke patients. Int J Stroke. 2013;8:422–429. doi: 10.1111/ijs.12056. [DOI] [PubMed] [Google Scholar]
- 74.Zhang W, Sun F, Liu C, Su W, Tan C, Liu S. 2017 IEEE International Conference on Systems, Man, and Cybernetics (SMC) Banff, AB, USA: A hybrid EEG-based BCI for robot grasp controlling; pp. 3278–3283. [Google Scholar]
- 75.Zhao Z, Wang X, Fan M, Yin D, Sun L, Jia J, Tang C, Zheng X, Jiang Y, Wu J, Gong J. Altered effective connectivity of the primary motor cortex in stroke: a resting-state fMRI study with granger causality analysis. PLoS One. 2016;11:e0166210. doi: 10.1371/journal.pone.0166210. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


