Dear Editor,
The most recognised renal manifestations of the current COVID-19 pandemic are multifactorial acute renal failure, usually prerenal, associated with the treatments used in severe disease or complications such as sepsis.1, 2 Furthermore, as the infection progresses, a procoagulant state develops that increases the risk of thromboembolic events. The most commonly reported are pulmonary and cerebral embolisms. We present the case of a patient with renal infarction.
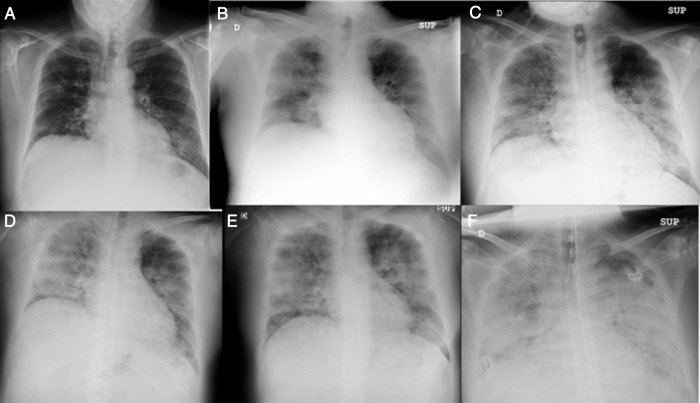
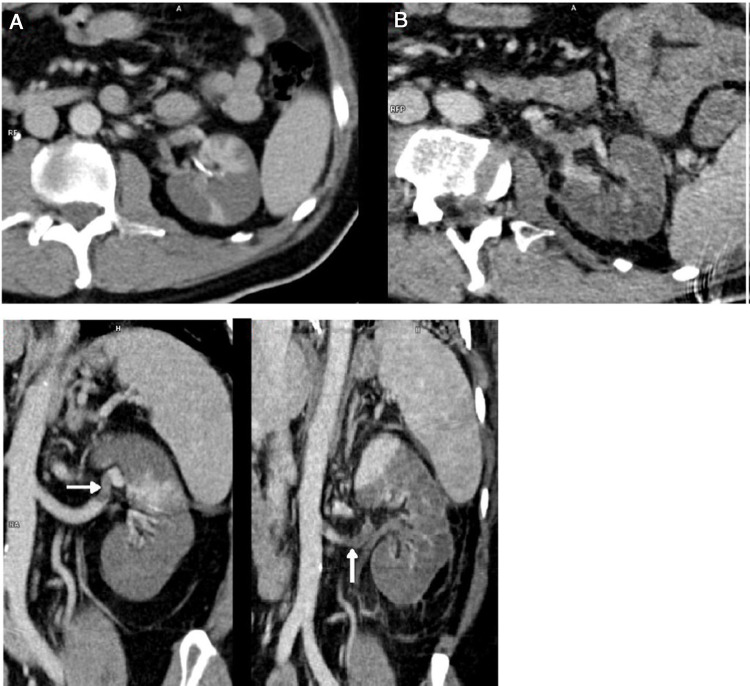
56-year-old male of Peruvian origin who was admitted from the emergency department on 31/03/2020 due to a five-day history of intense asthenia with general malaise, fever: 38.5 °C, cough and diarrhoea. He had well-controlled type 2 diabetes mellitus, a one-year history of mixed dyslipidaemia, and uncomplicated diverticulosis. He was receiving treatment with dapagliflozin/metformin and plantago ovata and adhering to a low-fat diet. On examination, he had a fever of 37.5 °C, was normotensive, and had moderate tachypnoea and sparse bibasal crackles. Height 174 cm, weight 94 kg (BMI 31). The main blood test results are summarised in Table 1 . Chest X-ray with patchy bilateral interstitial alveolar infiltrates. Initiated treatment with hydroxychloroquine, azithromycin, ceftriaxone, enoxaparin 0.5 mg/kg/day and ventilatory support with nasal cannula at 2 bpm. At 24 h, the positive result for SARS-CoV-2 was received following a RT-PCR test on a nasopharyngeal swab. The patient's clinical course was unfavourable, with a greater oxygen requirement, continuous fever and an increase in acute phase reactants, so on the third day IV methylprednisolone boluses were started at 250 mg/day for 3 days, and 40 mg/day thereafter, and on the fourth day a single IV dose of tocilizumab 600 mg was administered. The patient’s condition continued to deteriorate, with clinical, laboratory and radiological progression (Fig. 1 ). On the sixth day, the enoxaparin dose was increased to 0.8 mg/kg/day. The patient remained stable until the ninth day, when his condition further deteriorated. The tenth day began with intense, continuous, non-radiating pain in the left iliac fossa, without accompanying bladder dysfunction. Intestinal perforation was suspected in relation to anti-interleukin treatment and an abdominal CT scan with intravenous contrast was performed (Fig. 2 ). Acute diverticulitis was identified and in the left kidney, hypodense, wedge-shaped areas were observed, findings consistent with extensive renal infarction, as well as associated thrombosis of the left renal artery (arrow). Renal function only showed a slight deterioration (sCr: 0.9–1.1 mg/dl and estimated GFR CKD-EPI from greater than 90–67 ml/min/1.73 m2) and the patient, who six months earlier had urine without proteinuria or haematuria and negative microalbuminuria, developed stage A3 microalbuminuria, microhaematuria and subnephrotic proteinuria. He was admitted to the intensive care unit, where he remains at the time of writing. Invasive mechanical ventilation, treatment with norepinephrine and anticoagulation with low molecular weight heparin were required. Spontaneous diuresis greater than 0.5 ml/kg/h and anticoagulation with enoxaparin. The patient’s respiratory status is improving slowly.
Table 1.
Blood test results and their progression.
| Values/day | Admission | 2 | 3 | 4 | 5 | 6 | 8 | 10 | 11 | 12 | 13 | 18 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Leukocytes | 6.13 | 5.77 | 6.71 | 8.97 | 9.28 | 11.95 | 13.52 | 12.27 | 13.27 | 24.89 | 22.14 | 11.9 |
| Lymphocytes | 400 | 600 | 400 | 500 | 300 | 400 | 300 | 300 | 200 | 600 | 100 | 700 |
| Hb | 16.5 | 14.6 | 15.9 | 14.9 | 14.9 | 14.4 | 16.5 | 15.5 | 15.2 | 16.5 | 15.6 | 12.4 |
| Platelets | 181 | 196 | 218 | 249 | 287 | 356 | 400 | 310 | 281 | 321 | 173 | 212 |
| Activ. PT | 82.3 | 78 | 76 | 73 | 81.9 | 79.9 | 71.2 | 79.4 | 75 | 65.8 | 78 | 99.6 |
| APTT | 27.4 | 28.7 | 30 | 28.8 | 24.4 | 21 | 22 | 21.9 | 21.5 | 23.4 | 27.4 | 27.7 |
| Fibrinogen | >500 | >500 | >500 | >500 | >500 | 675 | 487 | 423 | 317 | 414 | 446 | >500 |
| Creatinine | 1.17 | 1.01 | 1.14 | 1.01 | 1.04 | 0.94 | 0.91 | 0.90 | 1.17 | 0.95 | 1.08 | 0.97 |
| eGFR CKD-EPI | 69 | 83 | 72 | 83 | 80 | >90 | >90 | >90 | 69 | 89 | 76 | 87 |
| Lactate | 2.47 | 2.06 | 3.49 | 2.44 | 3.09 | 1.62 | 1.68 | 0.99 | ||||
| CPK | 99 | 160 | 82 | 61 | 104 | 33 | 21 | |||||
| LDH | 245 | 248 | 311 | 405 | 418 | 450 | 552 | 698 | 1718 | 461 | 1975 | 773 |
| GPT (ALT) | 35 | 31 | 31 | 33 | 43 | 51 | 67 | 47 | 99 | 167 | 131 | 44 |
| GOT (AST) | 40 | 36 | 44 | 50 | 49 | 49 | 57 | 56 | 202 | 236 | 91 | 50 |
| CRP | 107.5 | 105 | 183 | 191 | 190 | 69 | 17 | 15 | 15 | 28 | 44 | 94 |
| d-dimer | 2710 | 940 | 1470 | 5580 | 1080 | 1211 | 1886 | 8790 | 3350 | |||
| Ferritin | 546 | 475 | 1053 | |||||||||
| IL-6 | 99 | >1000 | ||||||||||
| aPL Ab | Negative | |||||||||||
| Urinalysis | Microhaem, PRT 100 mg/dl, isolated leukocyturia | |||||||||||
| Spontaneous urine: | MAu/Cru: 278 | |||||||||||
| PCR: 1870 mg/g Cr | ||||||||||||
| TT | AZT | MPRN 250 × 3 | TOC | ENOX1 mg/kg/12 h | MPRN 40 | MEROP | ||||||
| HCQ | ANAK | LINEZ | ||||||||||
| CEFT | ||||||||||||
| ENOX 0.5 mg/kg/d |
ANAK: anakinra; AZT: azithromycin; CEFT: ceftriaxone; ENOX: enoxaparin; HCQ: hydroxychloroquine; LINEZ: linezolid; Mau/Cru: microalbuminuria/creatinine in urine; MEROP: meropenem; MICROHAEM: microhaematuria; MPRN: metal prednisolone; PCR: urine protein creatinine ratio; PRT: proteinuria; TOC: tocilizumab.
Units: leukocytes *10E3/µl; lymphocytes *10E3/µl; haemoglobin g/dl; platelets 10E3/µl; APTT sec; activ. prothrombin (Activ. PT) sec; derived fibrinogen mg/dl; creatinine mg/dl; lactate mmol/l; CPK U/l; LDH U/l; GPT (ALT) U/l; GOT (AST) U/l; estimated glomerular filtration rate [CKD-EPI] ml/min/1.73 m2; C-reactive protein (CRP) mg/l; d-dimer mg/dl; ferritin mg/dl; interleukin 6 (IL-6) ng/ml; SO: urinalysis, aPL Ab: antiphospholipid antibodies (beta-2 glycoprotein 1 IgG IU/ml, beta-2 glycoprotein 1 IgM IU/ml, anticardiolipin IgG GPL/ml and anticardiolipin IgM MPL/ml).
Fig. 1.
Chest X-ray, progression on days 1 (A), 2 (B), 3 (C), 6 (D), 9 (E) and 12 (F).
Fig. 2.
Abdominal CT scan on days 10 (A) and 18 (B).
Renal infarctions are uncommon lesions that can be seen in various situations: after intrarenal haematoma,3 renal artery dissection in instrumental procedures, dyslipidaemia,4 PR3-ANCA positive vasculitis,5 fibromuscular dysplasia, atherosclerotic disease, connective tissue diseases, embolic heart disease (atrial fibrillation, valvular heart disease, ventricular aneurysms, heart attacks, dilated cardiomyopathies), idiopathic heart disease6 and thrombophilias such as dysfibrinogenemia.7
Various series8, 9 have identified the following as the main risk factors: hypertension, smoking, atrial fibrillation, obesity, peripheral vascular disease, previous thromboembolic event, diabetes mellitus and oestroprogestin therapy. There is little consensus on whether the conservative approach with antiplatelet therapy and anticoagulation, or the interventionist approach with fibrinolysis and angioplasty, is more effective.
Coronavirus can directly infect endothelial cells, platelets and megakaryocytes by combining with its receptor CD13,10, 11 inducing platelet damage, endotheliitis and apoptosis, which triggers the recruitment of macrophages and granulocytes that will synthesise pro-inflammatory cytokines. If the infection is not controlled, the inflammation progresses, exacerbates tissue and microvascular damage, stimulates the extrinsic pathway of coagulation and inhibits fibrinolysis, which could trigger a hypoxia-aggravated consumption coagulopathy that would induce platelet aggregation, synthesis of thrombopoietin, fibrinogen, VEFG and thrombin, and lead to decreased anticoagulant factors such as antithrombin III and plasminogen activator inhibitor.12, 13, 14 This mechanism would be confirmed by increased d-dimer and thrombocytopenia levels. The resulting thromboinflammatory and procoagulant environment12 would trigger thrombotic events in the different affected areas, 4.5% cerebral, 11.4% pulmonary and 20% deep vein thrombosis. Our patient maintained glomerular filtration stability at all times and was treated with anticoagulant doses of enoxaparin. To our knowledge, this is the second reported case of renal artery thrombosis in this context.15
Footnotes
Please cite this article as: Cuevas Tascón G, Salazar Chiriboga DE, Lorente Ramos R, Díaz Díazc D, Rodríguez Ruiz C, Procaccini FL, et al. Infarto renal en paciente con COVID-19. Nefrologia. 2021;41:84–87.
References
- 1.Fanelli V., Fiorentino M., Cantaluppi V., Gesualdo L., Stallone J., Ronco C. Acute kidney injury in SARS-CoV-2 infected patients. Crit Care. 2020;24:155. doi: 10.1186/s13054-020-02872-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Richardson S., Hirsch J.S., Narasimhan M., Crawford J.M., McGinn T., Davidson K.W., The Northwell COVID-19 Research Consortium Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area. JAMA. 2020 doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Park S., Lee G.H., Jin K., Park K.M., Kim Y.W., Park B.S. Renal infarction caused by isolated spontaneous renal artery intramural hematoma. Am J Case Rep. 2015;16:832–836. doi: 10.12659/AJCR.895285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.El Barzouhi A., van Buren M., van Nieuwkoop C. Renal and Splenic infarction in a patient with familial hypercholesterolemia and previous cerebral infarction. Am J Case Rep. 2018;19:1463–1466. doi: 10.12659/AJCR.911990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bottomley M.J., Gibson M., Alchi B. PR3 vasculitis presenting with symptomatic splenic and renal infsrction: a case report and literature review. BMC Nephrol. 2019;20:84. doi: 10.1186/s12882-019-1266-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Eren Z., Koyuncu H. Renal Infarction in a Young Man. Balkan Med J. 2019;36:290–291. doi: 10.4274/balkanmedj.galenos.2019.2019.1.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Keinath K., Church T., Fogarty, Sadowski B., Perkins J. Acute renal artery infarction secondary to dysfibrinogenemia. BMJ Case Rep. 2017;2017 doi: 10.1136/bcr-2017-221375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mesiano P., Rollino C., Beltrame G., Ferro M., Quattrocchio G., Fenoglio R. Acute renal infarction: a single center experience. J Nephrol. 2017;30:103–107. doi: 10.1007/s40620-015-0259-0. [DOI] [PubMed] [Google Scholar]
- 9.Caravaca-Fontán F., Pampa Saico S., Elías Triviño S., Galeano Álvarez C., Gomis Couto A., Pecharromán de las Heras I. Acute renal infarction: clinical characteristics and prognostic factors. Nefrologia. 2016;36:141–148. doi: 10.1016/j.nefro.2015.09.015. [DOI] [PubMed] [Google Scholar]
- 10.Han H., Yang L., Liu R., Liu F., Wu K.L., Li J. Prominent changes in blood coagulation of patients with SARS-CoV-2 infection. Clin Chem Lab Med. 2020 doi: 10.1515/cclm-2020-0188. [DOI] [PubMed] [Google Scholar]
- 11.Varga Z., Flammer A.J., Steiger P., Haberecker M., Andermatt R., Zinkernagel A.S. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Connors J.M., Levy J.H. Thromboinflammation and the hypercoagulability of COVID-19. J Thromb Haemost. 2020 doi: 10.1111/jth.14849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mei H., Hu Y. Characteristics, causes, diagnosis and treatment of coagulation dysfunction in patients with COVID-19. Zhonghua Xue Ye Xue Za Zhi. 2020;41:E002. doi: 10.3760/cma.j.issn.0253-2727.2020.0002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yin S., Huang M., Li D., Tang N. Difference of coagulation features between severe pneumonia induced by SARS-COV2 and non- SARS-CoV2. J Thromb Thrombolysis. 2020 doi: 10.1007/s11239-020-02105-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lushina N., Kuo J.S., Shaikh H.A. 2020. Pulmonary, cerebral, and renal thromboembolic disease associated with COVID-19 infection radiology. [DOI] [PMC free article] [PubMed] [Google Scholar]