Abstract
Background
The posterior cruciate ligament (PCL) is one of the essential stabilizers of the knee joint and it was demonstrated that its degenerative change related to the knee osteoarthritis (OA). We aimed to evaluate signal of the PCL in OA patients in comparison with healthy young and elderly volunteers using the ultra-short echo timeenhanced (UTE)-T2∗ mapping, and to validate these findings with histology.
Methods
Thirty asymptomatic volunteers, 13 young people (younger group) and 17 elderly people (elder group), and 27 patients who had undergone total knee arthroplasty (OA group) were enrolled in this study. UTE-T2∗ maps of PCL were obtained from all participants. The PCL was divided into proximal, middle, and distal parts and the UTET2∗ values obtained from each part were compared among the groups. In OA group, the sacrificed PCLs were evaluated histologically in each part corresponding to the part of UTE-T2∗ maps and compared.
Results
The UTE-T2∗ values in OA group were significantly higher than those in other groups except in distal part. In elder group, the UTE-T2∗ values were significantly higher than those in younger group only in the proximal part. Moreover, in OA group, the UTE-T2∗ values in proximal and middle parts were significantly higher than those in distal part. There was a moderate correlation between the UTE-T2∗ values and histological scores.
Conclusions
The specific signal intensity pattern of the PCL in patients with OA was demonstrated using UTE-T2∗ mapping, and these findings were related to histological degenerated status of the PCL.
Keywords: Posterior cruciate ligament (PCL), Osteoarthritis (OA), Magnetic resonance image (MRI), Ultra-short echo time-enhanced (UTE)
1. Introduction
Osteoarthritis (OA) is a progressive joint disorder which devastates daily living due to pain and dysfunction of the joint, especially in elderly people.1,2 OA is recognized as whole joint disease, and the degenerative changes are found not only in cartilage and meniscus but in ligaments.3 Although the posterior cruciate ligament (PCL) is recognized as an essential stabilizer of the knee4, histopathological degenerative changes in PCL early begin even before degeneration in articular cartilage was observed.5 According to Kleinbart et al., 85% of the PCL from OA knees have degenerative changes and contained marked loose, stiff, cystic, mucinous, or myxoid degenerative changes and an abnormal, disorganized parallel collagen structure compared to normal cruciate ligaments.6 Although such degenerative changes would be helpful to predict the progression of OA, those features are difficult to be detected even from its gross appearance, especially in early stage.7,8
Magnetic resonance imaging (MRI) is widely used to detect abnormal signals non-invasively with relatively high sensitivity and specificity.9 Since T2 values are usually measured using long echo times (TE -10 ms), it is difficult to detect abnormal signals from tendons, ligaments, and menisci, which are organized with rich collagenous fibrils with extremely rapid signal decay. 10, 11, 12, 13, 14 Therefore, a novel standardized diagnostic tool for early detection of such subtle degenerative changes in the PCL is necessary to predict development of OA.
Currently, the Ultra-short echo time-enhanced (UTE) MRI techniques is being developed to visualize these short-T2 tissues. Besides, the UTE T2∗ mapping has been developed as a novel quantitative technique. 10,15, 16, 17, 18 UTE T2∗ mapping would enable to analyze the degenerative pattern of the PCL in OA patients, and evaluate the degree of the degeneration quantitively. However, no previous in vivo study has compared the UTE T2∗ values of the PCL and their corresponding histological features. We hypothesized that the quantitative UTE-T2∗ mapping technique enables noninvasive and indirect evaluation of histological degeneration of the PCL in vivo.
The purpose of this study is to evaluate signal pattern of the PCL in OA patients in comparison with healthy young and elderly volunteers using the quantitative UTE-T2∗ mapping, and to validate these findings with histology of the PCL in OA patients.
2. Materials and method
2.1. Patients
This study was approved by the Institutional Review Board and the Ethics Committee of our institute and was in accordance with the Helsinki Declaration. Informed consent was obtained from all individual participants included in the study. From July 2017 to January 2019, thirty asymptomatic healthy volunteers were publicly recruited and enrolled in this study. These 30 volunteers comprised 13 young people aged 13–44 years old (younger group) and 17 elderly people aged 60–82 years old (elder group), and a further 27 patients aged 66–89 years old who had undergone total knee arthroplasty (TKA) under the diagnosis of osteoarthritis (OA). In the current study, with the knowledge of experienced surgeon, PCL sacrificing TKA (PS-TKA) were selected for 9 patients, and PCL retaining TKA (CR-TKA) were selected for the other 18 patients. Healthy volunteers were defined as those who had no prior history of either knee pain or injury. Exclusion criteria included subjects who showed contra-indications to MRI, who suffered from a systemic disease such as rheumatoid arthritis and/or had a past medical history of knee joint injury, infection, or neurovascular disease. The demographic data of each group including sex, age, and range of motion (ROM) are summarized in Table 1. For OA group, the Kellgren-Lawrence (K/L) classification 19 and the femoro-tibial angle (FTA), measured on a standing weight-bearing anteroposterior radiograph, were evaluated. However, the data for younger and elder groups were not evaluated due to radiation exposure problems.
Table 1.
Demographic data of each group (K/L: Kellgren-Lawrence classification; FTA: Femorotibial angle; a: significant difference vs elder group and OA group; b: significant difference vs younger and elder group).
| Younger (n = 13) | Elder (n = 17) | OA (n = 27) | ||
|---|---|---|---|---|
| Sex (Male/Female) | 9/4 | 0/17 | 7/20 | |
| Age (year-old) | 27.7 ± 10.9a | 71.8 ± 7.2 | 74.5 ± 5.8 | |
| ROM(°) |
Extension Flexion |
0.0 ± 0.0 | −0.9 ± 2.0 | −7.7 ± 6.5 b |
| 147.3 ± 2.6 | 147.3 ± 4.7 | 124.2 ± 14.8 b | ||
| K/L (Ⅲ/Ⅳ) | – | – | 12/15 | |
| FTA (°) | – | – | 182.8 ± 8.2 | |
2.2. MRI
All images were obtained from a commercially available 3.0-T scanner (Vantage Titan 3T v3.5, Canon Medical Systems, Otawara, Japan) using 16ch SPEEDER Knee coil apparatus. All volunteers and patients were laid in a supine position, with the knee joint firmly fixed and centered in the coil, to minimize subject motion during the examination. MRI scan in OA group were obtained preoperatively. A Gradient echo pulse sequence with radial kappa-space acquisition was implemented, using the manufacturer-delivered sequence development software environment. Data acquisitions were implemented along non-Cartesian trajectories to achieve ultra-short TE. A total of 200 slices of UTE T2∗ mapping sequences were captured in sagittal plane with following acquisition parameters: 5 different echo times (0.5, 0.7, 2.5, 3.0, 5.0 ms), 20.0 mm field of view, 1.0 mm slice thickness, 5.0°flip angle, 9.0 ms repetition time, scan time of 3 min 6 s.
2.3. Image analysis
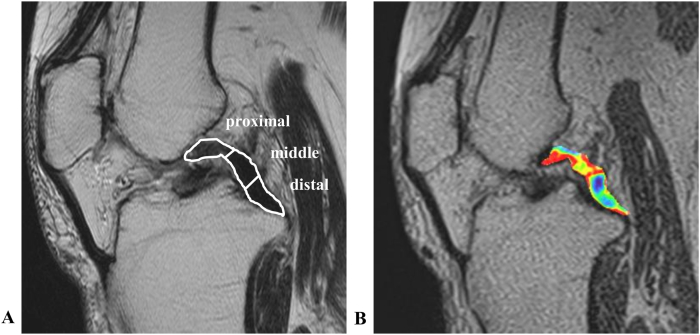
The slice of MRI image was randomly selected which showed the whole length of PCL in sagittal slice of image. T2∗ maps were automatically obtained by post-processing using image analysis software (calculation mode) built into the scanner and analyzed as a color-coded image. Quantitative UTE T2∗ mapping values were colored in the range of 0 (exhibited with deep blue) to 20 (exhibited with red) msec, and the rainbow scale was used to make it easy to visually distinguish from highly degenerate areas to normal areas. The colored image of the PCL was manually extracted and superimposed onto the image of the acquired 3D T2 weighted image, to produce a fusion image (Fig. 1).
Fig. 1.
(A) Manually demarcated ROIs dividing the whole length of the PCL into three parts: proximal; middle; and distal part. (B) Image of UTE-T2∗ color mapping. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
The regions of interest (ROIs) were manually drawn by an expert radiologist with over 10 years of experience. As shown in Fig. 1, the whole length of the PCL was evenly divided into three parts: proximal; middle; and distal part. The mean UTE T2∗ mapping of all pixels included in each part was used as the standard parameter. Moreover, the mean UTE T2∗ mapping values in each part were evaluated by examining at least three different slices of acquired MRI images, and their average value was used for statistical analysis.
2.4. Histological evaluation
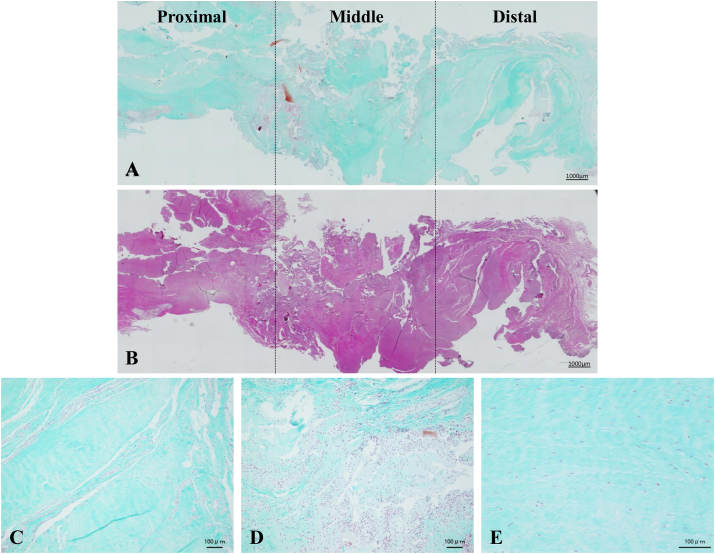
The PCL was obtained from 27 patients out of OA group at the time of TKA and used for histological evaluation. Whole length of the PCL was obtained from 8 patients who had undergone PS-TKA. The proximal and distal parts of PCL were partially obtained at the size of 3 mm × 3 mm × 3 mm from 13 patients who had undergone CR-TKA. Then, it was fixed in 4% paraformaldehyde phosphate-buffered saline (Wako Pure Chemical Industries Ltd., Osaka, Japan) for 24 h, after which it was embedded in paraffin and cut into 5 μm thick sections. Each specimen was stained with Safranin-O fast-green and Haematoxylin-eosin for histological evaluation (Fig. 2). As shown in Fig. 2, the PCL was divided into three parts: proximal, middle, and distal part. At least three randomized regions from each part were histologically evaluated with a high-power field ( × 100), and the average values of the histological scores were used for statistical analysis.
Fig. 2.
(A, B) Whole length of PCL ( × 40) was divided into three parts (Left to right indicate proximal to distal); (C) proximal, (D) middle, and (E) distal ( × 100).
A modified semiquantitative scale (Table 2) was used for histological evaluation. 20 For each subscale, classifications of normal (0 point), minimal changes (0.5 point), mild degeneration (1 point), moderate degeneration (2 points), and severe degeneration (3 points) were used. A total score of 15 represented severe degeneration, and a total score of 0 represented normal. Two blinded investigators assessed each sample individually.
Table 2.
Modified semiquantitative histological scoring22; For each subscale, classifications of normal (0 point), minimal changes (0.5 point), mild degeneration (1 point), moderate degeneration (2 points), and severe degeneration (3 points) were used. A total score of 15 represented severe degeneration, and a total score of 0 represented normal.
| Inflammation in the ligament substance |
|---|
| Mucoid degeneration |
| Chondroid metaplasia |
| Cystic changes |
| Orientation of collagen fibers |
| Total (0–15) |
2.5. Statistical analysis
The results are expressed as mean ± standard deviation for each parameter examined. One-way analysis of variance (ANOVA) tests, followed by individual post hoc Fisher’s PLSD tests, were used to compare (1) the UTE T2∗ values among each group at each part; (2) the UTE T2∗ values among each part within each group; (3) each UTE subcategory or total amount of histological scoring among each part in OA group. Pearson’s correlation test was used to analyze the correlation between the UTE T2∗ values and the histological scores. The reliability of the histological evaluation was assessed by examining the intra- and inter-observer reliabilities using the kappa (κ) coefficient. For all analysis, P-values of less than 0.05 were considered statistically significant.
3. Results
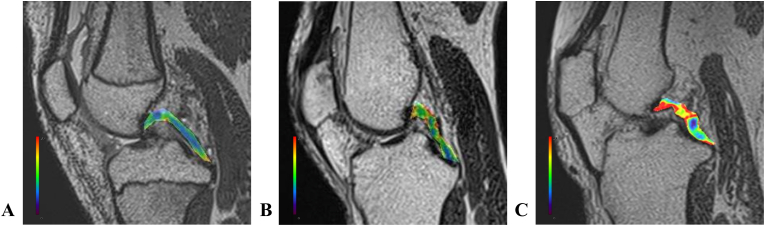
Acquired UTE T2∗ mapping images revealed that the PCL in healthy and elder groups showed blue ∼ green color through the full length of PCL. In elder group, a slight red area was observed on the synovium covering PCL (Fig. 3A, B). On the other hand, blue ∼ green area decreased in the substance of PCL and yellow ∼ red area increased in the proximal and middle part of PCL in the OA group (Fig. 3C).
Fig. 3.
Color overlaid UTE T2∗ maps of PCL from (A) younger, (B) elder, and (C) OA group. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
The UTE T2∗ values in the proximal and middle part of OA group were significantly higher than those in younger and elder groups, and the UTE T2∗ values in the proximal part of elder group were significantly higher than those in younger group (P < 0.01). There was no significant difference of the UTE T2∗ values of the distal part among 3 groups. Moreover, the UTE T2∗ values in distal part were significantly lower than those in proximal and middle parts in OA group (P < 0.01) (Table 3).
Table 3.
Mean values of UTE T2∗ mapping (ms) (significant difference, a: vs younger group, b: vs elder group, A: vs proximal part; B: vs middle part).
| Group ∖ Part | Proximal | Middle | Distal |
|---|---|---|---|
| Younger | 9.5 ± 2.0 | 9.9 ± 1.8 | 9.2 ± 1.3 |
| Elder | 12.6 ± 2.6a | 12.2 ± 2.9 | 10.9 ± 3.0 |
| OA | 15.5 ± 4.6a,b | 16.3 ± 0.2a,b | 11.3 ± 3.8A,B |
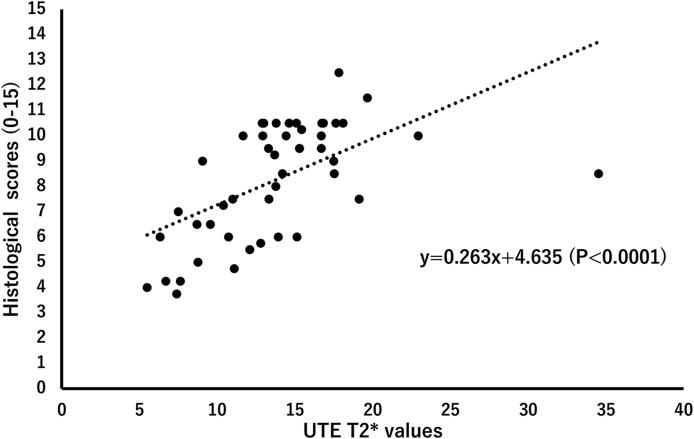
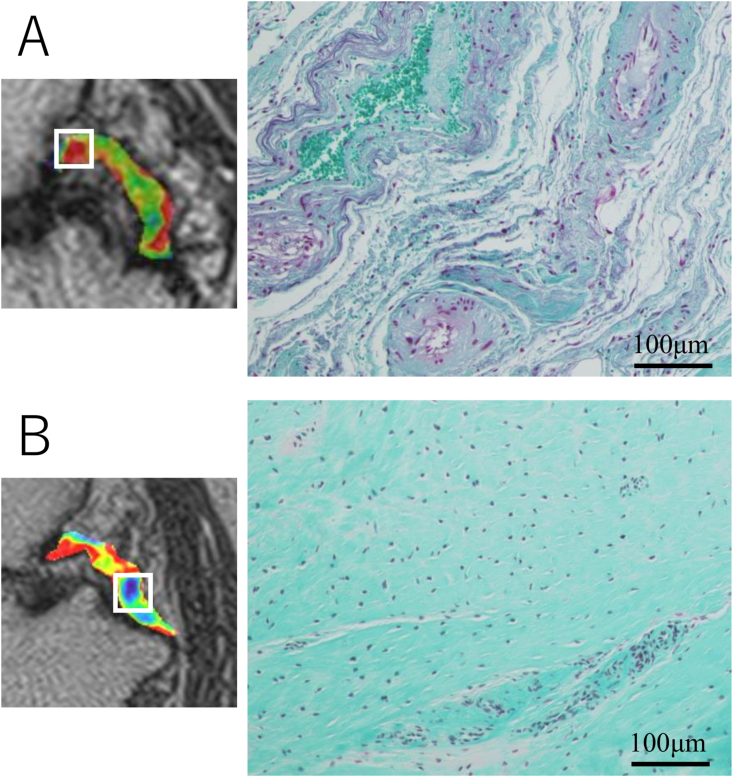
The mean histological scores were summarized in Table 4. The histological scores in distal part were significantly lower than those in the proximal and middle parts (P < 0.01). In detail, there were significant differences in the subcategories of inflammation in the ligament substance, cystic changes, and orientation of collagen fibers (P < 0.01). There was significant moderate correlation (r = 0.572, P < 0.01) between the UTE T2∗ values and histological scores (Fig. 4). In the high UTE T2∗ value region, severe inflammatory changes, cystic changes, and collapse of collagen fiber orientation were observed. On the other hand, in the low UTE T2∗ value region, collagen fiber orientation was normal with minimal inflammatory changes (Fig. 5).
Table 4.
Mean values of modified semiquantitative histological scoring at each part in OA group (A: significant difference vs distal part).
| Histological scoring∖Part | Proximal | Middle | Distal |
|---|---|---|---|
| Inflammation in the ligament substance | 2.1 ± 0.9A | 2.6 ± 0.4A | 0.9 ± 0.7 |
| Mucoid degeneration | 1.4 ± 0.5 | 1.3 ± 0.6 | 1.1 ± 0.7 |
| Chondroid metaplasia | 1.5 ± 0.9 | 1.6 ± 0.7 | 1.9 ± 1.1 |
| Cystic changes | 2.1 ± 0.7A | 2.3 ± 0.7A | 1.1 ± 0.4 |
| Orientation of collagen fibers | 2.4 ± 0.6A | 2.6 ± 0.4A | 1.3 ± 0.6 |
| Total (0–15) | 9.4 ± 1.3A | 10.5 ± 1.6A | 6.3 ± 1.7 |
Fig. 4.
Relationship between the UTE T2∗ values and the histological scores.
Fig. 5.
Representative case of the histological findings. (A) Severe inflammatory changes, cystic changes, and collapse of collagen fiber orientation were observed. The actual UTE T2∗ value was16.7 and the histological score of 10.5. (B) Collagen fiber orientation was normal with minimal inflammatory changes. The actual UTE T2∗ value of 10.7 and the histological score of 6.
The κ coefficients for intra- and inter-observer reliabilities for histological evaluation were 0.91 and 0.76, respectively.
4. Discussion
The current study revealed that the UTE-T2∗ mapping technique of MRI could describe the specific signal intensity pattern of the PCL in young, elderly, and patients with knee OA. Especially, in the knee OA patients, the value of UTE-T2∗ and histological degenerative status of the PCL was well correlated, i.e. higher value in more degenerative status.
In OA patients, the signal intensity change in the PCL was observed in the proximal two-third on the UTE-T2∗ mapping and histological degeneration was confirmed from corresponding PCL specimens. Also this change was observed at the proximal one-third in elder asymptomatic group. From our study, UTE-T2∗ mapping enables us to describe the specific signal intensity pattern of the PCL with quantitative manner.
A minimal number of studies has reported the usefulness of UTE-T2∗ mapping analysis for detecting early-stage degenerative changes in joint structures. 15,17,18 Although changes in UTE-T2∗ relaxation time are nonspecific, there is thought to be a correlation between multiple factors associated with degenerative change, including inflammation, changes in hydration, macromolecular content, and disruption of the collagen fiber network. 16,19 Generally, the low UTE-T2∗ values represent zero or minimal histological degeneration, and the high UTE-T2∗ values represent severe degeneration. 18 These findings collaborate our UTE-T2∗ results in OA group, which showed significantly higher values than those in younger group. Moreover, Kleinbart et al. reported that the histologic degenerative changes they identified in the PCL from OA knees were different from age-related changes.6 Levy YD et al. also reported that over the age of 50 years old, PCL degeneration is more associated with the presence of cartilage damage rather than with age. 5 These reports support our results that the UTE-T2∗ values in the proximal two-thirds of the PCL in OA group is significantly higher than those in elder group, which means that the degenerative change in OA group is more severe than that in elder group. Besides, even in asymptomatic healthy elderly group, signal change has already occurred from proximal part of the PCL without OA.
The significantly higher UTE T2∗ values in proximal part in elder and OA group compared to those in younger group would indicate that the degeneration of PCL begins in the proximal part with aging. In addition, the significantly higher UTE T2∗ values in OA group in proximal and middle parts compared to the values in distal part, also suggested that signal intensity change of PCL would progress from the proximal to distal part as the OA progresses. From our observation, the histological scores in proximal and middle parts were significantly higher, more degenerative change, than that in distal part. Considering that there was significant correlation between the exact UTE-T2∗ values and the histological score, the degenerative PCL changes seemed to proceed through the proximal part as the OA progressed. However, Wilson et al. evaluated the PCL in an asymptomatic population and reported that the T2 values in the distal one-third were significantly higher than those in the proximal one-third, but there was no significant difference between their T2∗ values. 21Although they considered that higher T2 values could be related to degenerative changes, they did not evaluate the histological findings. Basically, they did not evaluate the degenerative change on PCL using UTE-T2∗.
Several limitations of the current study should be noted. First, histological evaluation was not performed in younger and elder groups. A cadaveric study will be needed to understand the correlation between the UTE-T2∗ values and their histological findings. Second, gender was not considered in this study. Previous reports have shown that meniscal T2 values in women are greater than in men. 22However, in the current study, it was difficult to recruit those volunteers within recruitment period. In future, an age and gender comparative study should be performed. Third, the UTE-T2∗ mapping demonstrates highly heterogenous distributions of UTE-T2∗ values, which means that the averaged values of the zonal ROI in the current study do not truly reflect the region where focal histological findings and scoring were evaluated. Although the histological evaluations were performed with several randomized regions from each part, and the average values were used for evaluation in the current study to minimize technical errors. Fourth, it is difficult to determine whether the degenerative findings of PCL are caused by OA or the cause of OA. However, degenerative changes in PCL early begin even before degeneration in articular cartilage was observed5, the higher UTE-T2∗ value observed in elderly people without any OA symptoms might mean the degenerative PCL would be one of a cause of early OA. Finally, functional evaluation of the PCL was not performed in this study. The potential correlation between UTE-T2∗ values and the mechanical function of the PCL should be elucidated in future consecutive study. Degenerative changes such as mucin deposition, cyst formation and fragmentation of the collagen bundles correlate to structural weakness in the PCL.7 With our results, quantitative UTE T2∗ values were well related to the histological aspects of the PCL, which would also mean that the biological strength of PCL might be indirectly described.
5. Conclusion
The current study showed that the specific signal intensity pattern of the PCL in patients with OA using UTE-T2∗ mapping technique of MRI, and these findings were related to histological degenerated status of PCL. Since degenerative changes in PCL early begin even before degeneration in articular cartilage is observed, detection of early degenerative change using UTE-T2∗ mapping would be helpful for understanding of OA pathology to establish the appropriate preventive intervention for OA progression.
Funding
Dr Hayashi receives research grant of The Japanese Orthopaedic Society of Knee, Arthroscopy and Sports Medicine, 2017.
Dr. Awai receives research grants from Canon Medical Systems.
Any other competing interests, such as employment, consultancy, patents, products in development, or marketed products, do not exist regarding this manuscript.
Acknowledgement
We thank K. Ushio and T. Omoto for their grateful support for collecting data and its analysis.
References
- 1.Creamer P., Hochberg M.C. Osteoarthritis. Lancet. 1997;350(9076):503–508. doi: 10.1016/S0140-6736(97)07226-7. [DOI] [PubMed] [Google Scholar]
- 2.Dieppe P.A., Lohmander L.S. Pathogenesis and management of pain in osteoarthritis. Lancet. 2005;365(9463):965–973. doi: 10.1016/S0140-6736(05)71086-2. [DOI] [PubMed] [Google Scholar]
- 3.Harkey M.S., Davis J.E., Lu B. Early pre-radiographic structural pathology precedes the onset of accelerated knee osteoarthritis. BMC Muscoskel Disord. 2019;22(1):241. doi: 10.1186/s12891-019-2624-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.LaPrade C.M., Civitarese D.M., Rasmussen M.T., LaPrade R.F. Emerging updates on the posterior cruciate ligament: a review of the current literature. Am J Sports Med. 2015;43(12):3077–3092. doi: 10.1177/0363546515572770. [DOI] [PubMed] [Google Scholar]
- 5.Levy Y.D., Hasegawa A., Patil S., Koziol J.A., Lotz M.K., D’Lima D.D. Histopathological changes in the human posterior cruciate ligament during aging and osteoarthritis: correlations with anterior cruciate ligament and cartilage changes. Ann Rheum Dis. 2013;72(2):271–277. doi: 10.1136/annrheumdis-2012-201730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kleinbart F.A., Bryk E., Evangelista J., Scott W.N., Vigorita V.J. Histologic comparison of posterior cruciate ligaments from arthritic and age-matched knee specimens. J Arthroplasty. 1996;11(6):726–731. doi: 10.1016/s0883-5403(96)80012-x. [DOI] [PubMed] [Google Scholar]
- 7.Nelissen R.G., Hogendoorn P.C. Retain or sacrifice the posterior cruciate ligament in total knee arthroplasty? A histopathological study of the cruciate ligament in osteoarthritic and rheumatoid disease. J Clin Pathol. 2001;54(5):381–384. doi: 10.1136/jcp.54.5.381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Allain J., Goutallier D., Voisin M.C. Macroscopic and histological assessments of the cruciate ligaments in arthrosis of the knee. Acta Orthop Scand. 2001;72(3):266–269. doi: 10.1080/00016470152846592. [DOI] [PubMed] [Google Scholar]
- 9.Phelan N., Rowland P., Galvin R., O’Byrne J.M. A systematic review and meta-analysis of the diagnostic accuracy of MRI for suspected ACL and meniscal tears of the knee. Knee Surg Sports Traumatol Arthrosc. 2016;24(5):1525–1539. doi: 10.1007/s00167-015-3861-8. [DOI] [PubMed] [Google Scholar]
- 10.Chang E.Y., Du J., Chung C.B. UTE imaging in the musculoskeletal system. J Magn Reson Imag. 2015;41(4):870–883. doi: 10.1002/jmri.24713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hodler J., Haghighi P., Trudell D., Resnick D. The cruciate ligaments of the knee: correlation between MR appearance and gross and histologic findings in cadaveric specimens. AJR Am J Roentgenol. 1992;159(2):357–360. doi: 10.2214/ajr.159.2.1632355. [DOI] [PubMed] [Google Scholar]
- 12.Kannus P. Structure of the tendon connective tissue. Scand J Med Sci Sports. 2000;10(6):312–320. doi: 10.1034/j.1600-0838.2000.010006312.x. [DOI] [PubMed] [Google Scholar]
- 13.Rath E., Richmond J.C. The menisci: basic science and advances in treatment. Br J Sports Med. 2000;34(4):252–257. doi: 10.1136/bjsm.34.4.252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Servant C.T., Ramos J.P., Thomas N.P. The accuracy of magnetic resonance imaging in diagnosing chronic posterior cruciate ligament injury. Knee. 2004;11(4):265–270. doi: 10.1016/j.knee.2003.11.005. [DOI] [PubMed] [Google Scholar]
- 15.Chu C.R., Williams A.A., West R.V. Quantitative magnetic resonance imaging UTE-T2∗ mapping of cartilage and meniscus healing after anatomic anterior cruciate ligament reconstruction. Am J Sports Med. 2014;42(8):1847–1856. doi: 10.1177/0363546514532227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kijowski R., Wilson J.J., Liu F. Bicomponent ultrashort echo time T2∗ analysis for assessment of patients with patellar tendinopathy. J Magn Reson Imag. 2017;46(5):1441–1447. doi: 10.1002/jmri.25689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Williams A., Qian Y., Bear D., Chu C.R. Assessing degeneration of human articular cartilage with ultra-short echo time (UTE) T2∗ mapping. Osteoarthritis Cartilage. 2010;18(4):539–546. doi: 10.1016/j.joca.2010.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Williams A., Qian Y., Golla S., Chu C.R. UTE-T2∗ mapping detects sub-clinical meniscus injury after anterior cruciate ligament tear. Osteoarthritis Cartilage. 2012;20(6):486–494. doi: 10.1016/j.joca.2012.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kellgren J.H., Lawrence J.S. Radiological assessment of osteo-arthrosis. Ann Rheum Dis. 1957;16(4):494–502. doi: 10.1136/ard.16.4.494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee G.C., Cushner F.D., Vigoritta V., Scuderi G.R., Insall J.N., Scott W.N. Evaluation of the anterior cruciate ligament integrity and degenerative arthritic patterns in patients undergoing total knee arthroplasty. J Arthroplasty. 2005;20(1):59–65. doi: 10.1016/j.arth.2004.10.010. [DOI] [PubMed] [Google Scholar]
- 21.Wilson K.J., Surowiec R.K., Ho C.P. Quantifiable imaging biomarkers for evaluation of the posterior cruciate ligament using 3-T magnetic resonance imaging: a feasibility study. Orthop J Sports Med. 2016;4(4) doi: 10.1177/2325967116639044. 2325967116639044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tsai P.H., Chou M.C., Lee H.S. MR T2 values of the knee menisci in the healthy young population: zonal and sex differences. Osteoarthritis Cartilage. 2009;17(8):988–994. doi: 10.1016/j.joca.2009.03.009. [DOI] [PubMed] [Google Scholar]