Abstract
Background
There is still no clinical evidence available to support or to oppose corticosteroid treatment for coronavirus disease 2019 (COVID-19) pneumonia.
Objective
To investigate the efficacy and safety of corticosteroid given to the hospitalized patients with COVID-19 pneumonia.
Methods
This was a prospective, multicenter, single-blind, randomized control trial. Adult patients with COVID-19 pneumonia who were admitted to the general ward were randomly assigned to either receive methylprednisolone or not for 7 days. The primary end point was the incidence of clinical deterioration 14 days after randomization.
Results
We terminated this trial early because the number of patients with COVID-19 pneumonia in all the centers decreased in late March. Finally, a total of 86 COVID-19 patients underwent randomization. There was no difference of the incidence of clinical deterioration between the methylprednisolone group and control group (4.8 vs. 4.8%, p = 1.000). The duration of throat viral RNA detectability in the methylprednisolone group was 11 days (interquartile range, 6–16 days), which was significantly longer than that in the control group (8 days [2–12 days], p = 0.030). There were no significant differences between the 2 groups in other secondary outcomes. Mass cytometry discovered CD3+ T cells, CD8+ T cells, and NK cells in the methylprednisolone group which were significantly lower than those in the control group after randomization (p < 0.05).
Conclusions
From this prematurely closed trial, we found that the short-term early use of corticosteroid could suppress the immune cells, which may prolong severe acute respiratory syndrome coronavirus 2 shedding in patients with COVID-19 pneumonia.
Trial Registration
ClinicalTrials.gov, NCT04273321.
Keywords: Coronavirus disease 2019, Pneumonia, Corticosteroid, Outcome, Virus shedding
Introduction
The rapid spread of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) that emerged in late 2019 [1] has been labeled a public health emergency of international concern by the World Health Organization (WHO). Globally, as of September 22, 2020, a total of 31,132,906 confirmed cases, including 962,008 deaths, have been reported to the WHO [2]. In a cohort of 179 cases with COVID-19, we have demonstrated that 73 patients underwent acute respiratory distress syndrome (ARDS), and 21 (28.8%) of them died [3, 4]. For the severe COVID-19 patients, neither lopinavir-ritonavir therapy nor remdesivir seemed to not accelerate clinical benefits from some randomized controlled trials [5, 6]. However, there was a double-blind, randomized, placebo-controlled trial of intravenous remdesivir in adults hospitalized with COVID-19, which showed it could shorten the time to recovery compared with placebo but without information about the influence on virus shedding and mortality [7].
Although the WHO does not recommend, systemic corticosteroids are routinely given to patients with COVID-19 outside of clinical trials unless they are indicated for another reason [8]. These drugs were used widely during the outbreak in China [3, 9, 10, 11, 12]. In a retrospective cohort study, Wu et al. [13] have shown that the administration of methylprednisolone appears to have reduced the risk of death in COVID-19 patients with ARDS. However, it still lacks evidence about the details of corticosteroid application in the treatment of COVID-19 pneumonia, such as virus shedding and immunological function. There is still no clinical evidence available to support or to oppose corticosteroid treatment for COVID-19 pneumonia. We conducted a prospective, multicenter, single-blind, randomized control trial to investigate the efficacy and safety of corticosteroid given to the hospitalized patients with COVID-19 pneumonia.
Materials and Methods
Patients
Patients who were laboratory confirmed of SARS-CoV-2 infection and had pneumonia confirmed by chest computed tomography were diagnosed with COVID-19 pneumonia [8]. Patients with COVID-19 pneumonia aged 18 years or older, admitted to the general wards for less than 72 h, and able to sign informed consent were eligible for this trial. Exclusion criteria included severe immunosuppression (human immunodeficiency virus infection and long-term use of immunosuppressive agents), pregnant or breastfeeding women, corticosteroid needed for other diseases, refractory hypertension, epilepsy or delirium, glaucoma, active gastrointestinal bleeding within 3 months, refractory hypokalemia, secondary bacterial or fungal infection, unwilling or unable to participate or complete the study, and participation in other studies.
Trial Design and Oversight
This was a prospective, multicenter, single-blind, randomized, clinical control trial (ClinicalTrials.gov number, NCT04273321). Patients were recruited from the respiratory departments or infectious disease department of 7 tertiary hospitals in Beijing and Hubei province of China. After screening, eligible participants were randomly assigned (in a 1:1 ratio) to either the methylprednisolone group or control group for treatment. Randomization was stratified by the statistician of the leading site, who produced computer-generated block randomization lists with a block size of 4 patients. This was a single-blind trial; the physicians were aware of the treatment assignment, but the participants were blinded. And during the whole study period, the data collection and end point judgement were blinded, and the statisticians were also blinded during the statistical analysis.
Procedures
After informed consent was obtained, baseline data including medical history, relevant comorbidities, symptoms, vital signs, variables required for the calculation of pneumonia severity index (PSI) [14], CURB-65 [15], and sequential organ failure assessment (SOFA) [16] were collected, and baseline blood samples were also drawn.
All the participants received standard therapy of COVID-19 according to the Chinese Diagnosis and Treatment Plan for COVID-19 (trial version 6) [17]. Patients started receiving study medication 1 h after randomization. In the methylprednisolone group, 1 mg/kg per day of methylprednisolone (produced by Pfizer Manufacturing Belgium NV) dissolved in 100 mL 0.9% normal saline was administered intravenously for 7 days. In the control group, 100 mL 0.9% normal saline was administered intravenously. Clinical data were recorded on paper case record forms and then double entered in an electronic database and validated by trial staff.
End Points
The primary end point was the incidence of clinical deterioration 14 days after randomization. The secondary end points included the incidence of clinical cure 14 days after randomization, the incidence of intensive care unit (ICU) admission, in-hospital mortality, the time from randomization to clinical cure, the time from the onset to virus shedding of SARS-CoV-2 in respiratory tract samples, hospitalization duration, and complications including blood glucose abnormal, stress ulcer, and secondary infections.
The clinical deterioration fulfilled at least one of the following criteria: the clinical symptoms and signs continue to deteriorate, new pulmonary or extrapulmonary lesions appear, the chest computed tomography indicates the progress, or the patient is transferred to the ICU or is dead. The clinical cure fulfilled all of the following criteria: the clinical symptoms and signs of COVID-19 improved or alleviated (body temperature for 3 consecutive days, respiratory symptoms improved significantly, and computed tomographic images showed obvious absorption of bilateral extensive ground-glass opacification and/or consolidation), and no additional or alternative treatments were needed. The criteria of virus shedding was SARS-CoV-2-negative result of the nucleic acid tests from throat swabs for 2 consecutive times (sampling interval of at least 1 day) [17]. RT-PCR was used to test SARS-CoV-2.
Mass Cytometry
To investigate the impact of methylprednisolone on the immune cell profile, we collected peripheral blood mononuclear cells of 5 patients from the methylprednisolone group and 5 from the control group, who were hospitalized to Beijing You-an Hospital, for conducting mass cytometry analysis (see online suppl. materials; see www.karger.com/doi/10.1159/000512063 for all online suppl. material).
Statistical Analysis
According to the findings reported in the first Wuhan cohort [11], 30% of the overall population with COVID-19 pneumonia might deteriorate to a severe or critical stage. Due to a lack of corticosteroid data in COVID-19 pneumonia, we speculated corticosteroid could reduce the incidence of being severely ill from 30 to 15% based on our experience (α = 5%; β = 20%). By using the superiority test, we calculated a sample size of 121 patients in each of the 2 parallel groups. Considering that 10% of the participants may be dropped, a total of 270 cases were required for this trial.
Data analysis was performed using SPSS 23.0 software. Categorical variables were summarized using frequencies and percentages, and continuous data are presented as means and standard deviations or medians and interquartile ranges. Comparisons of clinical characteristics between the 2 groups were performed by using Student's t test or Wilcoxon's rank sum test for continuous variables and by using χ2 test or Fisher's exact test for categorical variables. Repeated measures analysis of the two-way ANOVA followed by Bonferroni's test or repeated measures analysis of the one-way ANOVA followed by Dunnett's test was performed for the data obtained at multiple time points. The primary and secondary outcomes were assessed using the Kaplan-Meier approach with the log-rank test. As data from all participants entered in this trial were available for the final statistical analysis, an analysis on an intention-to-treat basis was not necessary. Univariate analysis for stratification variables used logistic regression analysis, and the time variable, estimated by the Cox proportional risk model. We deemed a two-tailed p value <0.05 to be significant.
Results
Patients
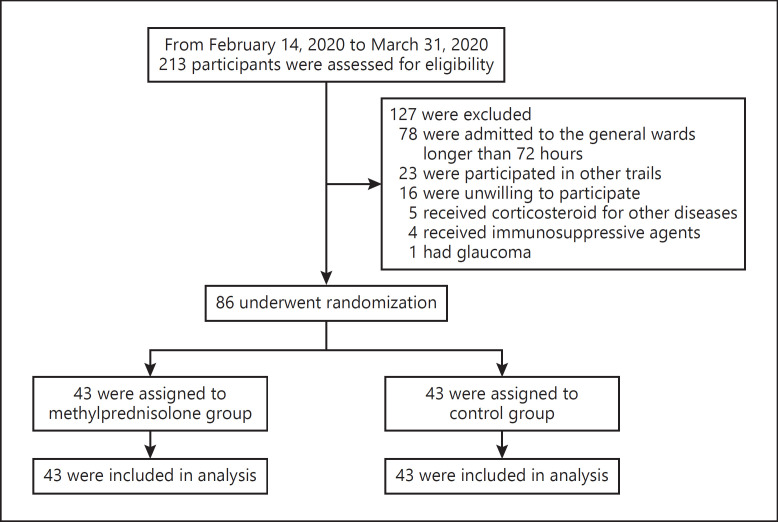
The numbers of confirmed cases in China reached the peak on February 1, 2020, and gradually decreased thereafter [18]. The first patient was enrolled in this trial on February 19, 2020. Because the number of patients with COVID-19 pneumonia in all the centers decreased in late March, we enrolled the last patient on March 31, 2020 and terminated the trial on April 15, 2020. A total of 213 potential eligible patients were screened, and 127 of them were excluded because they did not meet the inclusion criteria (Fig. 1). Eventually, 86 patients from 7 centers (online suppl. Table 2) were randomized to either the methylprednisolone group (n = 43) or control group (n = 43) (Fig. 1). None of the patients withdrew from the trial or were excluded from the analyses because of missing or incomplete data.
Fig. 1.
Randomization and treatment assignment of patients with COVID-19. COVID-19, coronavirus disease 2019.
As shown in Table 1, the median age of the whole cohort was 56 years (interquartile range, 39–66 years) and 41 (47.7%) patients were male. The median time from the symptom onset to randomization was 8 days (6–16 days). At randomization, 61 (70.9%) patients received oxygen therapy via a nasal cannula, while 41 (47.7%) underwent hypoxemia respiratory failure. The median arterial partial pressure of oxygen/fractional inspired oxygen was 311.5 mm Hg (236.0–354.0 mm Hg), and 41 of 86 (47.7%) patients had their arterial partial pressure of oxygen/fractional inspired oxygen below 300 mm Hg. At randomization, there was no difference in all the demographic, clinical, and laboratory characteristics between the 2 groups except that more sputum production was seen in the control group (Table 1; online suppl. Table 3).
Table.
Clinical characteristics of patients at randomization
| Characteristic | Total (N = 86) | Methylprednisolone (N = 43) | Control (N = 43) | p value |
|---|---|---|---|---|
| Age, median (IQR), years | 56 (39–66) | 57 (49–67) | 55 (38–65) | 0.594 |
| Weight, median (IQR), kg | 64 (58–73) | 62 (60–72) | 65 (56–71) | 0.593 |
| Body mass index, median (IQR), kg/m2 | 23.4 (21.5–25.3) | 23.9 (20.5–25.4) | 23.0 (21.6–25.1) | 0.792 |
| Gender (male), n (%) | 41 (47.7) | 21 (48.8) | 20 (46.5) | 0.829 |
| Smoke, n (%) | 11 (12.9) | 6 (14.3) | 5 (11.6) | 0.909 |
| Time from the symptom onset to diagnosis, median (IQR), days | 6 (4–10) | 5 (3–10) | 6 (4–10) | 0.565 |
| Time from the symptom onset to admission, median (IQR), days | 7 (4–15) | 6 (4–11) | 10 (4–18) | 0.149 |
| Time from the symptom onset to randomization, median (IQR), days | 8 (6–16) | 8 (6–13) | 11 (6–21) | 0.131 |
| Symptom, n (%) | ||||
| Fever | 66 (76.7) | 36 (83.7) | 30 (69.8) | 0.126 |
| Cough | 58 (67.4) | 33 (76.7) | 25 (58.1) | 0.066 |
| Sputum | 23 (26.7) | 17 (39.5) | 6 (14.0) | 0.007 |
| Chest tightness | 23 (26.7) | 15 (34.9) | 8 (18.6) | 0.088 |
| Chest pain | 5 (5.8) | 3 (7.0) | 2 (4.7) | 0.645 |
| Short of breath | 20 (23.3) | 11 (25.6) | 9 (20.9) | 0.610 |
| Dyspnea | 13 (15.1) | 7 (16.3) | 6 (14.0) | 0.763 |
| Fatigue | 22 (25.6) | 12 (27.9) | 10 (23.3) | 0.621 |
| Myalgia | 13 (15.1) | 6 (14.0) | 7 (16.3) | 0.763 |
| Snot | 5 (5.8) | 3 (7.0) | 2 (4.7) | 0.645 |
| Diarrhea | 2 (2.3) | 1 (2.3) | 1 (2.3) | 0.999 |
| Nausea | 2 (2.3) | 2 (4.7) | 0 (0) | 0.152 |
| Comorbidities, n (%) | ||||
| Chronic obstructive pulmonary disease | 3 (3.5) | 2 (4.7) | 1 (2.3) | 0.543 |
| Asthma | 2 (2.4) | 1 (2.3) | 1 (2.3) | 0.987 |
| Hypertension | 31 (36.0) | 16 (37.3) | 15 (34.9) | 0.758 |
| Coronary heart disease | 6 (7.0) | 3 (7.0) | 3 (7.0) | 0.976 |
| Diabetes | 8 (9.3) | 3 (7.0) | 5 (11.6) | 0.479 |
| Chronic renal failure | 1 (1.2) | 0 (0) | 1 (2.3) | 0.320 |
| Vital sign, median (IQR) | ||||
| Heart rate, beats per min | 92 (84–100) | 92 (90–101) | 91 (80–100) | 0.979 |
| Systolic pressure, mm Hg | 135 (120–146) | 137 (122–143) | 135 (119–147) | 0.509 |
| Diastolic pressure, mm Hg | 80 (71–92) | 80 (70–91) | 81 (74–92) | 0.194 |
| Respiratory rate, times per min | 20 (20–24) | 21 (20–30) | 20 (19–29) | 0.481 |
| Temperature | 38.2 (37.8–38.6) | 38.1 (37.8–38.5) | 38.3 (37.9–38.6) | 0.557 |
| PSI score, n (%) | ||||
| Class I | 3 (3.5) | 1 (2.3) | 2 (4.7) | 0.780 |
| Class II | 18 (20.9) | 8 (18.6) | 10 (23.3) | |
| Class III | 40 (46.5) | 19 (44.2) | 21 (48.8) | |
| Class IV | 11 (12.8) | 7 (16.3) | 4 (9.3) | |
| Class V | 14 (16.3) | 8 (18.6) | 6 (14.0) | |
| CURB-65, median (IQR) | 0 (0–1) | 1 (0–1) | 0 (0–1) | 0.193 |
| SOFA score, median (IQR) | 1 (0–2) | 2 (1–2) | 1 (0–2) | 0.083 |
| Complications, n (%) | ||||
| Hypoxemia respiratory failure | 41 (47.7) | 22 (51.2) | 19 (44.2) | 0.450 |
| Abnormal liver function | 13 (15.1) | 6 (14.0) | 7 (16.3) | 0.799 |
CURB-65 denotes a six-point score, one point for each of confusion: urea >7 mmol/L, respiratory rate >30/min, low systolic (<90 mm Hg) or diastolic (<60 mm Hg) blood pressure, age >65 years. PSI, pneumonia severe index; SOFA, sequential organ failure assessment; IQR, interquartile range.
During hospitalization, 67 (77.9%) patients in both groups were administered antivirus drugs, and 61 (70.9%) were given antibiotics. Seventy (88.1%) patients needed nasal cannula oxygen therapy, 3 (3.5%) used high-flow nasal cannula oxygen therapy, 4 (4.6%) received mechanical ventilation because of disease deterioration, and 2 of whom underwent extracorporeal membrane oxygenation. Overall, there was no difference between the 2 groups in the medicine application and respiratory support (online suppl. Table 4).
Primary Outcome
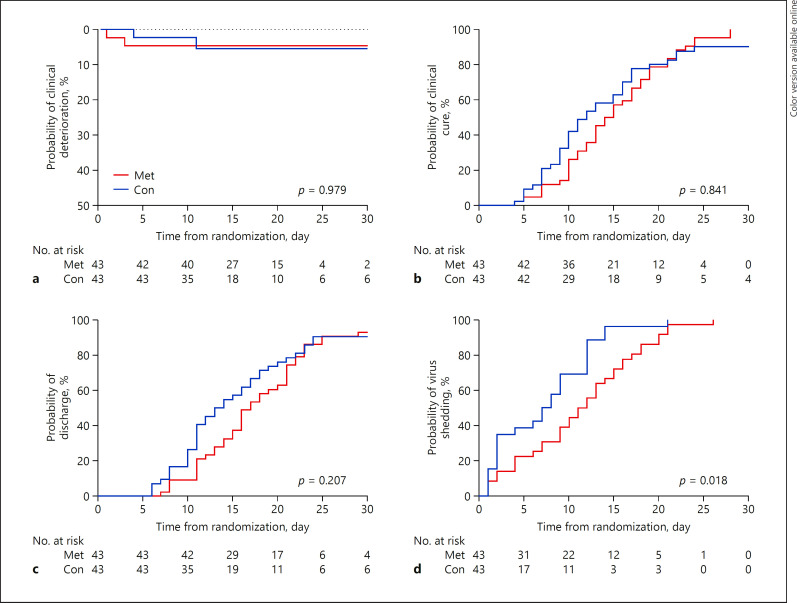
Totally, 4 (4.8%) patients showed clinical deterioration in 14 days after randomization, and there was no difference in the incidence of clinical deterioration between the methylprednisolone group and control group (4.8 vs. 4.8%; odds ratio, 1.000 [95% confidence interval, 0.134–7.442]; p = 1.000) (Table 2; Fig. 2a).
Table 2.
Primary and secondary outcomes
| Characteristic | Total (N = 86) | Methylprednisolone (N = 43) | Control (N = 43) | Regression analysis |
||
|---|---|---|---|---|---|---|
| OR, or HR* (95% CI) | p value | |||||
| Primary outcome, n (%) | ||||||
| Clinical deterioration 14 days after randomization | 4(4.8) | 2 (4.8) | 2 (4.8) | OR 1.000 (0.134–7.442) | 1.000 | |
| Secondary outcome | ||||||
| Clinical cure 14 days after randomization, n (%) | 47 (54.7) | 22 (51.2) | 25 (58.1) | OR 1.326 (0.566–3.106) | 0.516 | |
| Time from randomization to clinical cure, median (IQR), days | 13(10-19) | 14 (10–19) | 12 (9–17) | HR 1.043 (0.673–1.617) | 0.850 | |
| ICU admission, n (%) | 4 (4.8) | 2 (4.8) | 2 (4.8) | OR 1.000 (0.134–7.442) | 1.000 | |
| Hospitalization duration, median (IQR), days | 16 (11–21) | 17 (13–22) | 13 (10–20) | HR 1.300 (0.844–2.002) | 0.235 | |
| In-hospital mortality, n (%) | 1 (1.2) | 0 (0) | 1 (2.3) | OR 0.977 (0.933–1.023) | 0.314 | |
| Time from virus shedding of SARS-CoV-2, median (IQR), days | 9 (4–14) | 11 (6–16) | 8 (2–12) | HR 1.782 (1.057–3.003) | 0.030 | |
CI, confidence interval; HR, hazard ratio; ICU, intensive care unit; IQR, interquartile range; OR, odds ratio; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
Differences were expressed as ORs (95% CIs) analyzed by logistic regression analysis adjusted for the stratification variables, and HRs (95% CIs), estimated by Cox proportional risk model for time variable.
Fig. 2.
Kaplan-Meier curves of the primary outcome and secondary outcomes between the Met group and Con group. Shown are the probability of clinical deterioration (a), clinical cure (b), discharge (c), and SARS-CoV-2 shedding (d) over time. The Kaplan-Meier approach with the log-rank test was used to assess the primary and secondary outcomes. SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; Met, methylprednisolone; Con, control.
Secondary Outcome
As shown in Table 2, Figure 2b and c, there was no difference in the percentage of clinical cure 14 days after randomization, time from randomization to clinical cure, ICU admission, hospitalization duration, and in-hospital mortality between the methylprednisolone group and control group (all p > 0.05). The median time from randomization to SARS-CoV-2 shedding in the methylprednisolone group was significantly longer than that in the control group (median, 11 days vs. 8 days; hazard ratio, 1.782 [1.057–3.003]; p = 0.030) (Table 2; Fig. 2d).
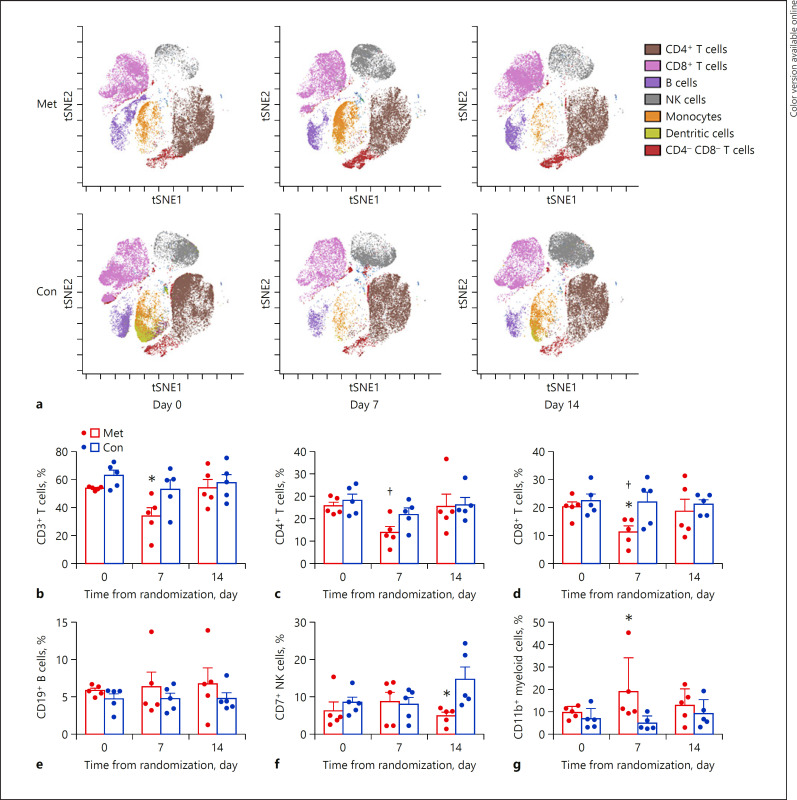
Effect of Methylprednisolone on Immune Cell Profile
Mass cytometry analysis of blood CD45+ immune cells revealed that the profile of the main cell subsets gated from the total CD45+ cells have changed after treatment with methylprednisolone (Fig. 3a). Further analysis discovered that 7 days after randomization and methylprednisolone treatment, CD3+ T cells and CD8+ T cells in the methylprednisolone group were significantly lower than those in the control group (both p < 0.05) (Fig. 3b, d); 14 days after randomization, NK cells in the methylprednisolone group were significantly lower than those in the control group (p < 0.05) (Fig. 3f). We also found that methylprednisolone treatment led to an increase in CD11b+ myeloid cells on day 7 (Fig. 3g). In addition, CD4+ and CD8+ T cells were reduced on day 7 after methylprednisolone treatment as compared to day 0 (both p < 0.05) (Fig. 3c, d). In addition, methylprednisolone treatment did not exert effect on CD19+ B cells and the other immune cells (Table 3).
Fig. 3.
Mass cytometric analysis of CD45+ immune cells from peripheral blood. a Representative t-SNE plots of CD45+ immune cells on day 0 (at randomization), 7 and 14 days after randomization derived from one COVID-19 patients from the Met group (upper panels) and one from the Con group (bottom panels) are shown. One point represents one cell, and the dark and light colors shows high expression and low expression. The comparisons of the percentages of blood CD3+ T cells (b), CD4+ T cells (c), CD8+ T cells (d), CD19+ T cells (e), CD7+ NK cells (f), and CD11b+ myeloid cells (g) between the Met group and Con group. Data are presented as mean ± SD. *p < 0.05 compared with the Con group on the same time point by repeated measures analysis of the two-way ANOVA followed by Bonferroni's test. †p < 0.05 compared with day 0 in the Met group by repeated measures analysis of the one-way ANOVA followed by Dunnett's test. COVID-19, coronavirus disease 2019; Met, methylprednisolone group; Con, control group.
Table 3.
Effect of methylprednisolone treatment of immune cell profile determined by mass cytometry
| Methylprednisolone |
Control |
|||||
|---|---|---|---|---|---|---|
| day 0 | day 7 | day 14 | day 0 | day 7 | day 14 | |
| Of CD45+ cells, % | ||||||
| CD3+ T cells | 53.6±0.6 | 33.6±6.2* | 54.0±5.8 | 62.8±3.8 | 52.9±6.7 | 57.8±5.7 |
| CD4+ T cells | 30.7±2.0 | 16.4±3.3† | 30.4±6.7 | 33.7±3.4 | 26.3±3.3 | 31.3±3.9 |
| CD8+ T cells | 20.3±1.7 | 11.±2.1 *,† | 18.7±4.3 | 23.4±3.2 | 21.9±3.6 | 21.1±1.7 |
| CD19+ B cells | 5.8±0.3 | 6.3±1.9 | 6.8±2.0 | 4.7±0.7 | 4.8±0.7 | 4.8±0.8 |
| CD7+ NK cells | 6.3±2.3 | 8.6±2.6 | 4.9±1.0* | 8.4±1.5 | 8.0±1.9 | 14.6±3.4 |
| CD11b+ myeloid cells | 9.7±1.2 | 18.9±6.8 | 12.8±3.3 | 6.8±2.1 | 4.9±1.4 | 9.1±2.8 |
| CD16+ neutrophils | 0.06±0.02 | 0.85±0.71 | 0.35±0.21 | 0.23±0.013 | 0.08±0.05 | 0.32±0.14 |
| CD11c+ DCs | 0.4±0.1 | 0.2±0.1 | 0.3±0.1 | 0.3±0.2 | 0.1±0.0 | 0.6±0.2 |
| CD14+ monocytes | 9.1±0.9 | 8.4±0.6 | 9.2±3.0 | 4.8±1.8 | 3.4±1.1 | 6.8±2.3 |
| Of CD4+ cells, % | ||||||
| CD4+CD45RA+ naïve CD4+ cells | 49.1±6.3 | 50.9±6.2 | 37.4±2.8 | 43.3±4.1 | 44.9±4.6 | 45.5±4.3 |
| CD4+CD45RO+ memory CD4+ cells | 24.3±6.6 | 20.1±4.5 | 32.6±4.8 | 38.8±7.1 | 25.7±4.0 | 32.8±5.5 |
| CD4+CD25++ regulatory T cells | 3.5±0.6 | 3.6±0.9 | 3.5±0.4 | 2.6±0.1 | 3.4±0.3 | 2.9±0.4 |
| CD4+CCR4+ cells | 41.0±1.3 | 44.9±1.5 | 42.9±4.7 | 39.6±3.5 | 38.8±2.7 | 44.7±4.1 |
| CD4+CCR5+ cells | 2.1±0.4 | 2.8±0.4 | 2.3±0.3 | 3.7±1.2 | 4.3±0.6 | 4.7±1.1 |
| CD4+CCR6+ cells | 54.7±3.0 | 61.6±2.6 | 56.6±4.0 | 55.3±2.6 | 53.4±3.3 | 57.5±2.2 |
| CD4+CXCR3+ cells | 51.8±2.4 | 55.9±3.8 | 49.3±4.1 | 49.5±3.5 | 47.8±3.0 | 54.0±3.4 |
| CD4+CXCR5+ cells | 6.1±1.2 | 4.4±1.4 | 5.9±1.3 | 5.5±0.6 | 5.2±0.9 | 7.2±1.0 |
| CD4+CD161+ CCR6+ cells | 2.2±0.6 | 2.3±0.6 | 3.4±0.6 | 4.6±1.8 | 3.9±0.9 | 4.3±1.0 |
| Of CD8+ cells, % | ||||||
| CD8+CD45RA+ naïve CD8+ cells | 38.2±3.8 | 45.7±8.3 | 35.6±5.5 | 47.0±5.9 | 45.2±6.5 | 50.2±3.6 |
| CD8+CD45RO+ memory CD8+ cells | 37.6±3.6 | 28.6±5.5 | 36.6±6.7 | 28.3±4.9 | 28.7±5.6 | 31.5±5.5 |
| CD8+CCR4+ cells | 28.1±2.8 | 34.0±2.5 | 32.3±6.8 | 27.2±4.0 | 27.1±2.1 | 35.5±4.1 |
| CD8+CCR5+ cells | 11.2±1.8 | 18.6±4.0 | 13.3±3.7 | 13.2±4.0 | 15.4±2.7 | 17.5±2.7 |
| CD8+CCR6+ cells | 37.9±2.5 | 49.3±2.5 | 44.0±6.8 | 41.9±4.9 | 45.4±2.7 | 50.1±1.9 |
| CD8+CXCR3+ cells | 47.5±5.3 | 55.5±3.1 | 50.1±7.2 | 50.8±6.3 | 52.1±4.4 | 59.9±2.8 |
| CD8+CXCR5+ cells | 2.0±0.3 | 2.0±0.3 | 1.4±0.1 | 2.1±0.3 | 1.9±0.3 | 2.4±0.8 |
| Of CD19+ B cells, % | ||||||
| CD5+ regulatory B cells | 6.5±3.5 | 5.2±3.4 | 1.6±0.3 | 3.2±1.4 | 5.8±3.2 | 3.2±0.5 |
| CD27–IgD+ naive B cells | 50.9±9.3 | 42.3±7.1 | 51.2±4.8 | 57.8±3.9 | 50.1±7.6 | 61.7±1.7 |
| CD19+CD27+ memory B cells | 19.1±4.9 | 21.5±4.1 | 19.7±1.9 | 15.5±1.8 | 19.2±3.5 | 12.3±1.4 |
| CD27+CD38+ plasma B cells | 0.7±0.2 | 0.4±0.1 | 0.6±0.3 | 0.4±0.1 | 0.8±0.2 | 0.7±0.2 |
Data are presented as mean ± SEM.
p < 0.05 compared with the control group at the same time points after randomization by repeated measures analysis of the two-way ANOVA followed by Bonferroni's test.
p < 0.05 compared with day 0 after randomization in the methylprednisolone group by repeated measures analysis of the one-way ANOVA followed by Dunnett's test.
Stratified by Acute Hypoxemic Respiratory Failure
We stratified patients by whether they underwent acute hypoxemic respiratory failure at randomization (online suppl. Table 5). All 4 patients showing clinical deterioration were those with respiratory failure. There was no difference in the incidence and time from randomization to clinical deterioration between the 2 groups (online suppl. Table 6). Among patients without respiratory failure, the median time from randomization to the median time from randomization to virus shedding in the methylprednisolone group was 10 days (6–17 days), which was significantly longer than that in the control group (6 days [2–9 days]; hazard ratio, 2.730 [1.247–5.974]; p = 0.012). In contrast, the above effects of methylprednisolone did not exist among patients with acute hypoxemic respiratory failure (online suppl. Table 6).
Side Effects
Hyperglycemia with the need for additional therapy after randomization occurred in 3 (7.0%) patients in the methylprednisolone group and in 7 (16.3%) in the control group (p = 0.313). Two (4.7%) patients in the methylprednisolone group and one (2.3%) in the control group acquired ventilator-associated pneumonia after they had been transferred to the ICU and received invasive mechanical ventilation (p = 0.557). None of the patient in both groups had stress ulcer, gastrointestinal hemorrhage, or delirium (online suppl. Table 7). Corticosteroid-associated long-term complications were not estimated because of the short observation duration.
Discussion
This was a randomized control trial to investigate the application of corticosteroids in patients with COVID-19 pneumonia. Although with less statistical power as it was prematurely closed, our trial found that methylprednisolone added to standard therapy may not make the patients with COVID-19 pneumonia benefit from the outcomes of clinical deterioration, clinical cure, ICU admission, or mortality. However, based on the existing data and mass cytometry, we discovered that the viral shedding time in the methylprednisolone group was much longer than that in the control group. It may be associated with immune cells suppressed by the application of corticosteroid.
Because of very limited availability of evidence for harm or benefit from corticosteroids in treatment of viral pneumonia including COVID-19, the use of corticosteroids remains controversial [19, 20]. A retrospective study suggested that use of corticosteroid in critically ill SARS patients resulted in lowered mortality and a shorter hospitalization stay [21]. Another retrospective study suggested that treatment with methylprednisolone decreased the risk of death among COVID-19 patients with ARDS [13]. In contrast, several retrospective studies found no evidence of a beneficial effect of corticosteroids in severe patients with influenza A/H1N1 [22, 23] or A/H7N9 infection [24] since corticosteroids were independently associated with higher mortality. In a retrospective observational study reporting on 309 patients with critically ill MERS, corticosteroid therapy was not associated with a difference in mortality after adjustment for time varying confounders but was associated with delayed clearance of viral RNA from respiratory tract secretions [25]. These data suggest that corticosteroids may be hazardous when administered early in the context of severe viral pneumonia.
There have already been some studies about corticosteroid therapy application in critically ill patients of COVID-19, including different dosages of dexamethasone [26, 27] and hydrocortisone [28, 29]. Most results of these research studies concluded a significant clinical benefit from the application of corticosteroid in the patients of COVID-19-induced acute respiratory failure. And the meta-analysis from the WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group showed administration of systemic corticosteroids was associated with lower 28-day all-cause mortality in critically ill patients with COVID-19 [30]. However, the present study acquired an opposite result that early use of low-dose methylprednisolone may not obtain any clinical benefits from early methylprednisolone treatment in the patients of COVID-19, even prolonged the virus shedding. Considering that the patients in this trial were relatively less critical, so based on the above studies and our result, corticosteroids administered in the selected patients might help acquire better prognosis.
A pathological study of a patient who died from COVID-19 pneumonia reveals bilateral diffuse alveolar damage with cellular fibromyxoid exudates, interstitial lymphocyte infiltrates, and multinucleated syncytial cells in the intra-alveolar spaces [31]. Clinical studies suggested that dysregulation of immune response, especially T cells, might be highly involved in the pathological process of COVID-19 [32, 33, 34]. Our previous studies further demonstrated that a remarkable reduction in CD8+ T cells is one of the predictors for mortality of COVID-19 patients [3]. A small prospective, randomized double-blinded, placebo-controlled trial has revealed that early steroid treatment results in a higher subsequent plasma viral load in SARS patients [35]. Similar results have also been observed in a retrospective study that corticosteroid was associated with delayed MERS coronavirus RNA clearance [25]. In the present study, our trial found CD4+ cells, CD8+ T, and NK cells were reduced on day 7 after methylprednisolone treatment as compared to randomization through mass cytometry. These immune cells all play an important role in the immune response of antivirus [36]. Therefore, we provided the evidence to show that methylprednisolone could retard virus shedding by suppressing immune cells in patients with COVID-19 pneumonia, such as CD4+ T cells, CD8+ T cells, and NK cells.
Usually, corticosteroids are prescribed for viral pneumonia patients with severe disease. Our inclusion criterion was set to recruit all hospitalized patients with confirmed diagnosis of COVID-19 pneumonia. About half of the participants demonstrated acute hypoxemia respiratory failure during randomization. In the subgroup analysis, we did not find that methylprednisolone affected clinical deterioration either in patients with or without respiratory failure. However, methylprednisolone did extend the virus shedding time and hospital days in patients without acute respiratory failure. In China, patients with COVID-19 were not allowed to be discharged until their SARS-CoV-2 tests turned out to be negative. These data suggested that COVID-19 pneumonia patients could not gain benefits in clinical improvement from the addition of methylprednisolone, while patients even without acute respiratory failure could only gain harm from such a treatment.
Corticosteroid application in treating viral pneumonia has many potential risks, such as secondary infections and other long-term complications. One retrospective study found that corticosteroid-treated patients are more likely to suffer from secondary bacterial pneumonia or invasive fungal infection than those who were not administered with corticosteroids [23]. However, another retrospective study showed that the use of corticosteroids in SARS patients was not associated with secondary lower respiratory infection and other complications [21]. In this study, we applied a short-term low-dose of methylprednisolone in the early stage among patients with COVID-19 pneumonia and noted that neither hyperglycemia nor secondary lower respiratory infection was associated with methylprednisolone treatment. It is worth noting that such incidence of methylprednisolone-related complications might be underestimated because of the small sample size.
Our trial has 2 limitations. First, the study was stopped prior to reaching the goal size due to the decreased number of cases of COVID-19 in China, and this may have blunted benefit/harm and meaningful differences in the clinical course if any. Second, we did not detect quantitative viral load measurement whereas just detected the presence or absence of SARS-CoV-2 RNA in throat swabs but not in the blood or the other specimens. Third, due to the short observation duration, it was difficult to follow up the long-term complications.
Conclusions
Due to early termination of this trial, most outcomes were difficult to be estimated because of the low statistical power. However, we found that the short-term early use of corticosteroid could suppress the immune cells, which may prolong SARS-CoV-2 shedding in patients with COVID-19 pneumonia, especially for the patients without acute respiratory failure. It was suggested that corticosteroids should not be added to standard therapy as a general treatment for the patients of COVID-19 patients; moreover, it should be evaluated according to the severity and necessity. The interpretation still needs to be further verified by a large sample size and randomized clinical trials.
Statement of Ethics
The study protocol was approved by the Ethics Committee of Beijing Chao-Yang Hospital (2020-KE-22) and all the other participating centers, and informed consent was obtained from the patients themselves or their legal guardians.
Conflict of Interest Statement
The authors declare that they have no competing interests.
Funding Sources
This work was supported by the Beijing Municipal Administration of Hospitals' Mission Plan, China (SML20150301), Excellence Program of Beijing Clinical Key Specialty (2018) and Novel Coronavirus Pneumonia Key Technology Research and Development Funding of Beijing Municipal Administration of Hospitals (COVID-19-BHA03).
Author Contributions
H.Z.S. and B.S. conceived the idea, designed and supervised the study, drafted the manuscript, had full access to all of the data, and took responsibility for the integrity of the data. J.X.N., Y.M.F., L.M.L., K.H., J.X.Z., J.W.C., J.C.Z., Y.L.L., Y.Z., J.S., X.L.H., J.Y.Z., Z.D., and J.X. collected data. X.T., W.W., and X.Z.W. analyzed data and performed statistical analysis. R.H.J., Y.M.F. and X.Z.W. did mass cytometry analysis. All of the authors reviewed and approved the final version of the manuscript.
Xiao Tang, Ying-Mei Feng, Ji-Xiang Ni, Jia-Ying Zhang, Li-Min Liu, Ke Hu and Xiu-Zhi Wu contributed equally.
References
- 1.Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med. 2020 Mar 26;382((13)):1199–207. doi: 10.1056/NEJMoa2001316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.World Health Organization Coronavirus disease (COVID-19) situation dashboard. 2020. Available from: https://covid19.who.int/ Accessed 2020 Sep 22.
- 3.Du RH, Liang LR, Yang CQ, Wang W, Cao TZ, Li M, et al. Predictors of mortality for patients with COVID-19 pneumonia caused by SARS-CoV-2: a prospective cohort study. Eur Respir J. 2020 May 7;55((5)):2000524. doi: 10.1183/13993003.00524-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tang X, Du RH, Wang R, Cao TZ, Guan LL, Yang CQ, et al. Comparison of hospitalized patients with ARDS caused by COVID-19 and H1N1. Chest. 2020 Jul;158((1)):195–205. doi: 10.1016/j.chest.2020.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, et al. A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020 May 7;382((19)):1787–99. doi: 10.1056/NEJMoa2001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020 May 16;395((10236)):1569–78. doi: 10.1016/S0140-6736(20)31022-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, et al. Remdesivir for the treatment of Covid-19: preliminary report. N Engl J Med. 2020 Sep 3;383((10)):993–4. doi: 10.1056/NEJMc2022236. [DOI] [PubMed] [Google Scholar]
- 8.World Health Organization Clinical management of severe acute respiratory infection when novel coronavirus (nCov) infection is suspected: interim guidance. 2020. Jan 28. Available from: https://www.Who.Int/publications-detail/clinical-management-of-severe-acute-respiratory-infection-when-novel-coronavirus-(ncov)-infection-is-suspected Accessed 2020 May 6.
- 9.Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020 Feb 15;395((10223)):507–13. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020 Apr 30;382((18)):1708–20. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020 Feb 15;395((10223)):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020 Mar 17;323((11)):1061–9. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020 Jul 1;180((7)):934–43. doi: 10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fine MJ, Auble TE, Yealy DM, Hanusa BH, Weissfeld LA, Singer DE, et al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med. 1997 Jan 23;336((4)):243–50. doi: 10.1056/NEJM199701233360402. [DOI] [PubMed] [Google Scholar]
- 15.Lim WS, van der Eerden MM, Laing R, Boersma WG, Karalus N, Town GI, et al. Defining community acquired pneumonia severity on presentation to hospital: an international derivation and validation study. Thorax. 2003 May;58((5)):377–82. doi: 10.1136/thorax.58.5.377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vincent JL, Moreno R, Takala J, Willatts S, De Mendonça A, Bruining H, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 1996 Jul;22((7)):707–10. doi: 10.1007/BF01709751. [DOI] [PubMed] [Google Scholar]
- 17.Diagnosis and Treatment Plan for COVID-19 (Trial Version 6) Chin Med J. 2020 Mar 17 [Google Scholar]
- 18.Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the chinese center for disease control and prevention. JAMA. 2020 Apr 7;323((13)):1239–42. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 19.Russell CD, Millar JE, Baillie JK. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet. 2020;395((10223)):473–5. doi: 10.1016/S0140-6736(20)30317-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shang L, Zhao J, Hu Y, Du R, Cao B. On the use of corticosteroids for 2019-nCoV pneumonia. Lancet. 2020 Feb 29;395((10225)):683–4. doi: 10.1016/S0140-6736(20)30361-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chen RC, Tang XP, Tan SY, Liang BL, Wan ZY, Fang JQ, et al. Treatment of severe acute respiratory syndrome with glucosteroids: the Guangzhou experience. Chest. 2006 Jun;129((6)):1441–52. doi: 10.1378/chest.129.6.1441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Brun-Buisson C, Richard JC, Mercat A, Thiebaut AC, Brochard L. Group R-SAHNvR. Early corticosteroids in severe influenza A/H1N1 pneumonia and acute respiratory distress syndrome. Am J Respir Crit Care Med. 2011 May 1;183((9)):1200–6. doi: 10.1164/rccm.201101-0135OC. [DOI] [PubMed] [Google Scholar]
- 23.Kim SH, Hong SB, Yun SC, Choi WI, Ahn JJ, Lee YJ, et al. Corticosteroid treatment in critically ill patients with pandemic influenza A/H1N1 2009 infection: analytic strategy using propensity scores. Am J Respir Crit Care Med. 2011 May 1;183((9)):1207–14. doi: 10.1164/rccm.201101-0110OC. [DOI] [PubMed] [Google Scholar]
- 24.Cao B, Gao H, Zhou B, Deng X, Hu C, Deng C, et al. Adjuvant corticosteroid treatment in adults with influenza A (H7N9) viral pneumonia. Crit Care Med. 2016 Jun;44((6)):e318–28. doi: 10.1097/CCM.0000000000001616. [DOI] [PubMed] [Google Scholar]
- 25.Arabi YM, Mandourah Y, Al-Hameed F, Sindi AA, Almekhlafi GA, Hussein MA, et al. Corticosteroid therapy for critically ill patients with Middle East respiratory syndrome. Am J Respir Crit Care Med. 2018 Mar 15;197((6)):757–67. doi: 10.1164/rccm.201706-1172OC. [DOI] [PubMed] [Google Scholar]
- 26.Group RC, Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, et al. Dexamethasone in hospitalized patients with Covid-19: preliminary report. N Engl J Med. 2020 Jul 17;:NEJMoa2021436. [Google Scholar]
- 27.Tomazini BM, Maia IS, Cavalcanti AB, Berwanger O, Rosa RG, Veiga VC, et al. Effect of dexamethasone on days alive and ventilator-free in patients with moderate or severe acute respiratory distress syndrome and Covid-19: the CoDEX randomized clinical trial. JAMA. 2020 Oct 6;324((13)):1307–16. doi: 10.1001/jama.2020.17021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dequin PF, Heming N, Meziani F, Plantefeve G, Voiriot G, Badie J, et al. Effect of hydrocortisone on 21-day mortality or respiratory support among critically ill patients with COVID-19: a randomized clinical trial. JAMA. 2020 Oct 6;324((13)):1298–306. doi: 10.1001/jama.2020.16761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Writing Committee for the R-CAPI. Angus DC, Derde L, Al-Beidh F, Annane D, Arabi Y, et al. Effect of hydrocortisone on mortality and organ support in patients with severe COVID-19: the REMAP-CAP COVID-19 corticosteroid domain randomized clinical trial. JAMA. 2020 Oct 6;324((13)):1317–29. doi: 10.1001/jama.2020.17022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.WHOREAfC-TW Group. Sterne JAC, Murthy S, Diaz JV, Slutsky AS, Villar J, et al. Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19: a meta-analysis. JAMA. 2020 Oct 6;324((13)):1330–41. doi: 10.1001/jama.2020.17023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020 Apr;8((4)):420–2. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cao X. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol. 2020 May;20((5)):269–70. doi: 10.1038/s41577-020-0308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Li H, Liu L, Zhang D, Xu J, Dai H, Tang N, et al. SARS-CoV-2 and viral sepsis: observations and hypotheses. Lancet. 2020 May 9;395((10235)):1517–20. doi: 10.1016/S0140-6736(20)30920-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin Infect Dis. 2020 Jul 28;71((15)):762–68. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lee N, Allen Chan KC, Hui DS, Ng EK, Wu A, Chiu RW, et al. Effects of early corticosteroid treatment on plasma SARS-associated coronavirus RNA concentrations in adult patients. J Clin Virol. 2004 Dec;31((4)):304–9. doi: 10.1016/j.jcv.2004.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.National Research Project for SARS, Beijing Group The involvement of natural killer cells in the pathogenesis of severe acute respiratory syndrome. Am J Clin Pathol. 2004 Apr;121((4)):507–11. doi: 10.1309/WPK7Y2XKNF4CBF3R. [DOI] [PMC free article] [PubMed] [Google Scholar]