Abstract
Background
Ventral hernia repair (VHR) is a common procedure associated with significant postoperative morbidity and prolonged hospital length of stay (LOS). The use of epidural analgesia in VHR has not been widely evaluated.
Purpose
To compare the outcomes of general anesthesia plus epidural analgesia (GA + EA) versus general anesthesia alone (GA) in patients undergoing ventral hernia repair.
Methods
The American College of Surgeons National Surgical Quality Improvement Program database was used to identify elective cases of VHR. Propensity score-matched analysis was used to compare outcomes in GA vs GA + EA groups. Cases receiving transverse abdominus plane blocks were excluded.
Results
A total of 9697 VHR cases were identified, resulting in two matched cohorts of 521 cases each. LOS was significantly longer in the GA + EA group (5.58 days) vs the GA group (5.20 days, p = 0.008). No other statistically significant differences in 30-day outcomes were observed between the matched cohorts.
Conclusion
Epidural analgesia in VHR is associated with statistically significant, but not clinically significant increase in LOS and may not yield any additional benefit in cases of isolated, elective VHR. Epidural analgesia may not be beneficial in this surgical population. Future studies should focus on alternative modes of analgesia to optimize pain control and outcomes for this procedure.
Keywords: ventral, hernia, epidural, outcomes, anesthesia, regional, complications
Introduction
Ventral hernias are increasingly common, with repair rates projected to rise 1% per year.1 Repair techniques have evolved to address more complex ventral hernias requiring elaborate abdominal wall reconstruction and occasionally concurrent intraabdominal procedures.2 This increasing complexity has been associated with higher risks of postoperative complications, and prolonged hospital stays.3
Alongside the changing landscape of the United States healthcare system, emerging efforts to optimize resource utilization and outcomes by minimizing complications and hospital length of stay (LOS) continue to evolve, such as multimodal enhanced recovery after surgery pathways.1,4 It is well established that both intraoperative and postoperative pain management have been shown to significantly impact such measures, and many studies have found favorable outcomes associated with epidural analgesia (EA) for abdominal procedures.5 In addition to shorter hospital length of stay (LOS) and lower costs,6 purported advantages include opioid-sparing effects,7 expeditious recovery of both bowel function8 and functional status,5 lower mortality rates,9 improved pulmonary function10 and deceased risk of complications.11 Lower complication rates associated with EA are hypothesized to result from lower sympathetic tone and therefore systemic vascular resistance, which allows for lower abdominal pressures and improved lung mechanics.6,10 Other proposed mechanisms include reducing neuroendocrine responses, increasing oxygen tension within the wound, and decreasing protein catabolism resulting in preservation of lean body mass.12
Recent years have seen several studies examine the use of EA in ventral hernia repair (VHR),13 but the above mentioned benefits have yet to be confirmed in this population. In addition, the studies that do exist on this topic have produced conflicting results. For instance, Prabhu et al. found increased LOS and higher post-operative complication rates in a retrospective study comparing patients who received EA for VHR to those who did not.14 Similarly, Karamanos et al. also found an increased LOS and higher incidence of complications among patients who received EA following elective ventral hernia repair.15 However, it is important to note that the study by Prabhu et al. may have had patients with more complex hernia disease receiving EA, as acknowledged by the authors.
In opposition to the aforementioned data, Fischer et al. demonstrated decreased medical morbidities and unplanned reoperations with epidural use in VHR.6 Given the conflicting existing data, it is unclear whether EA improves outcomes and affects LOS for patients undergoing VHR. The purpose of this study is to compare the outcomes of EA plus general anesthesia (GA) versus GA alone in patients undergoing ventral hernia repair. We hypothesized that the combination of EA + GA would result in fewer complications on an individual patient level and a shorter hospital length of stay.
Methods
The American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) database contains de-identified information collected from more than 600 hospitals, amassing over 1,000,000 surgical cases per year. ACS-NSQIP describes patient demographics, preoperative risk factors, procedure characteristics, and 30-day outcomes. This study was approved by the institutional review board (protocol #2015P002706), and this analysis was exempt from the patient consent requirement because of the deidentified nature of the data. Diagnoses and procedures are coded with the Current Procedural Terminology (CPT) codes.16 This study included cases from the January 1, 2014 through December 31, 2016.
Cases of open VHR were identified using Current Procedural Terminology (CPT) code 49560 (initial repair of reducible incisional or ventral hernia). Patients included in the study cohort underwent the surgery on an elective, non-emergent basis and were admitted as inpatients post-operatively. All patients received general anesthesia (GA) as the primary anesthetic. All VHR identified as outpatient or emergency case were excluded. Cases with the principal anesthesia technique identified as local anesthesia, monitored anesthesia care (MAC) or regional anesthesia were excluded.
Use of adjunctive epidural anesthesia (EA) was identified using the coded secondary anesthetics. The secondary anesthetic variable is a non-hierarchical variable where all additional anesthetic types are listed. Cases were defined by the presence of an additional epidural anesthetic (GA + EA) or no coded secondary anesthetic (GA).
Variables included for analysis were demographic, (age, race, sex), baseline health characteristics (body mass index (BMI), functional status, American Society of Anesthesiologists (ASA) Physical Status (PS) classification, preoperative hematocrit, and comorbidities), and 30-day outcomes (mortality, readmission, reoperation, and complications). Analyses were limited to available data.
Statistical Analysis
Continuous demographic variables were reported as mean ± standard deviation (SD). Continuous variables were compared with Wilcoxon’s rank sum test; categorical variables were compared with Pearson’s Chi-squared test.
To assess the impact of epidural anesthesia on outcomes after VHR, a propensity score matched analysis was performed. First, to identify whether preoperative or operative characteristics were independent risk factors associated with anesthetic type, a univariable logistic regression model was fitted for all variables. Risk factors were defined as statistically significant when the odds ratios (OR) did not include 1.00 in their 95% confidence interval (CI), representing an alpha of < 0.05.
The matched cohort was developed using a propensity scoring method. All statistically significant variables (defined as p < 0.05) from the unmatched analysis were incorporated into a propensity score model. A 1:1 greedy, nearest neighbor matching strategy with a caliper set at 0.1 was employed utilizing the MatchIt library, successfully matching 521 cases performed under GA + EA to 521 cases under GA. Success of the matching process was evaluated using univariable logistic regression and calculation of the absolute standardized differences between variables and revealed adequate matching on all variables except Hispanic ethnicity. This category was therefore included in subsequent analysis of post-operative outcomes. Analysis of the primary and secondary endpoints between anesthesia types for the matched cohort was done using multivariable logistic regression for all outcomes. Length of stay was analyzed using a Poisson regression. Survival after surgery was assessed using the Kaplan-Meier method.
For the logistic regression, odds ratios (OR) were reported with associated 95% confidence intervals (CI). OR not including 1.00 in the 95% CI were considered statistically significant. To account for multiple comparisons and maintain a Family-Wise Error Rate of 0.05, a Bonferroni adjustment was performed, yielding an adjusted p-value for significance of 0.0025. Results reported as statistically significant in the manuscript were those that were significant after the Bonferroni adjustment. Sample size was defined by the availability of cases using the inclusion and exclusion criteria as outlined above. Power calculations were performed on our data using G*Power, version 3.1.9.5 (Dusseldorf, Germany) assuming a linear multiple regression model with a single regression coefficient and a fixed model with a total sample size of 1,042, alpha error of 0.0028, and 2 total predictors. Using the assumptions above and setting a desired power of 0.8 and a two-tailed model, the analysis had sufficient power to detect an effect size difference of means of 0.24. Post-hoc analysis of our results demonstrated an achieved power of 0.9. All analyses were conducted using R Studio Version 1.2.5033 (Boston, MA) and R Project for Statistical Computing, v3.6.2 (Vienna, Austria).
Results
From January 1, 2014 through December 31, 2016, a total of 9697 elective open VHR cases were identified; the majority of cases (8877) received only general anesthesia (GA), while fewer than 10% (820) received both general anesthesia and epidural analgesia (GA + EA) (Table 1). The majority of patients were female (54%), and mean age was 58 years. In the unmatched cohorts, the epidural group was comprised of a significantly smaller proportion of females (51.1% vs. 54.9%, p = 0.037) as well as a smaller proportion of white (71.8% vs. 76.5%, p < 0.001) and Hispanic (4.4% vs. 8.0%, p = 0.001) patients. GA group had a significantly higher rate of bleeding disorders (3.38%) vs. GA + EA group (1.46%, p = 0.003). Baseline characteristics including functional status, ASA PS classification, preoperative labs, and comorbidities between the two unmatched groups were not otherwise significantly different. Both groups had the same relative value units (RVUs) as only one CPT code was utilized; however, operative time was significantly longer in GA + EA (190.06 minutes vs GA 137.21 minutes, [odds ratio 1.01 (95% confidence interval 1–1.01)], p < 0.001).
Table 1. Demographics of GA versus GA + EA Groups.
| GA + EA | GA Only | |||||||||||
| Category | N (820) | % | N (8877) | % | OR (95% CI) | p-value | ||||||
| Age | ||||||||||||
| < 50 | 212 | 25.85 | 2275 | 25.63 | Reference | 0.048 | ||||||
| 50–65 | 326 | 39.76 | 3489 | 39.3 | 1 (0.84–1.2) | |||||||
| 65–80 | 263 | 32.07 | 2727 | 30.72 | 1.03 (0.86–1.25) | |||||||
| > 80 | 19 | 2.32 | 386 | 4.35 | 0.53 (0.33–0.85) | |||||||
| Sex | ||||||||||||
| Male | 401 | 48.9 | 4004 | 45.11 | Reference | 0.037 | ||||||
| Female | 419 | 51.1 | 4873 | 54.89 | 0.86 (0.74–0.99) | |||||||
| Demographics | ||||||||||||
| White | 589 | 71.83 | 6789 | 76.48 | Reference | < 0.001 | ||||||
| Black | 60 | 7.32 | 909 | 10.24 | 0.76 (0.58–1) | |||||||
| Asian | 5 | 0.61 | 104 | 1.17 | 0.55 (0.23–1.36) | |||||||
| Other | 7 | 0.85 | 46 | 0.52 | 1.75 (0.79–3.9) | |||||||
| Not reported | 159 | 19.39 | 1029 | 11.59 | 1.78 (1.48–2.15) | |||||||
| Hispanic | 30 | 4.41 | 631 | 7.97 | 0.53 (0.37–0.77) | 0.001 | ||||||
| BMI | ||||||||||||
| < 18.5 | 3 | 0.37 | 61 | 0.69 | 0.67 (0.21–2.19) | 0.062 | ||||||
| 18.5–25 | 90 | 11 | 1231 | 13.95 | Reference | |||||||
| 25–30 | 234 | 28.61 | 2526 | 28.62 | 1.27 (0.98–1.63) | |||||||
| > 30 | 491 | 60.02 | 5008 | 56.74 | 1.34 (1.06–1.69) | |||||||
| Functional status | ||||||||||||
| No dyspnea | 763 | 93.05 | 8307 | 93.58 | Reference | 0.519 | ||||||
| Dyspnea with moderate exertion | 56 | 6.83 | 545 | 6.14 | 1.12 (0.84–1.49) | |||||||
| Dyspnea at rest | 1 | 0.12 | 25 | 0.28 | 0.44 (0.06–3.22) | |||||||
| Independent in ADLs | 805 | 99.26 | 8712 | 98.78 | Reference | 0.222 | ||||||
| Partially/Totally dependent in ADLs | 6 | 0.74 | 108 | 1.22 | 0.6 (0.26–1.37) | |||||||
| ASA physical status | ||||||||||||
| 1 | 15 | 1.88 | 228 | 2.66 | Reference | 0.274 | ||||||
| 2 | 332 | 41.71 | 3689 | 43.05 | 1.37 (0.8–2.33) | |||||||
| 3/4 | 449 | 56.41 | 4653 | 54.29 | 1.47 (0.86–2.5) | |||||||
| Comorbidities | ||||||||||||
| Smoking | 152 | 18.54 | 1699 | 19.14 | 0.96 (0.8–1.16) | 0.674 | ||||||
| Hypertension | 405 | 49.39 | 4593 | 51.74 | 0.91 (0.79–1.05) | 0.198 | ||||||
| Diabetes | 136 | 16.59 | 1635 | 18.42 | 0.88 (0.73–1.07) | 0.194 | ||||||
| COPD | 40 | 4.88 | 518 | 5.84 | 0.83 (0.59–1.15) | 0.26 | ||||||
| CHF | 5 | 0.61 | 57 | 0.64 | 0.95 (0.38–2.37) | 0.911 | ||||||
| CKD | 7 | 0.85 | 118 | 1.33 | 0.64 (0.3–1.37) | 0.248 | ||||||
| Steroid use | 71 | 8.66 | 643 | 7.24 | 1.21 (0.94–1.57) | 0.138 | ||||||
| Weight loss | 8 | 0.98 | 48 | 0.54 | 1.81 (0.85–3.84) | 0.116 | ||||||
| Wound infection | 16 | 1.95 | 142 | 1.6 | 1.22 (0.73–2.06) | 0.447 | ||||||
| Bleeding disorder | 12 | 1.46 | 300 | 3.38 | 0.42 (0.24–0.76) | 0.003 | ||||||
| Labs | ||||||||||||
| Hematocrit | 40.7 | 1.42 | 40.4 | 1.52 | 1.14 (0.96–1.35) | 0.118 | ||||||
| Wound Class | ||||||||||||
| 1-Clean | 689 | 84.02 | 7584 | 85.43 | Reference | 0.443 | ||||||
| 2-Clean/Contaminated | 98 | 11.95 | 998 | 11.24 | 1.08 (0.87–1.35) | |||||||
| 3-Contaminated | 22 | 2.68 | 171 | 1.93 | 1.42 (0.9–2.22) | |||||||
| 4-Dirty/Infected | 11 | 1.34 | 124 | 1.4 | 0.98 (0.52–1.82) | |||||||
| Surgical complexity | ||||||||||||
| Operative Time | 190.06 | 104.49 | 137.21 | 82.03 | 1.01 (1–1.01) | < 0.001 | ||||||
GA = general anesthesia; EA = epidural analgesia; ASA = American Society of Anesthesiologists; BMI = body mass index: ADLs = activities of daily living.
Propensity score matching resulted in successful matching of 521 cases under GA + EAA to 521 cases under GA. There were no statistically significant differences in baseline health characteristics between the two cohorts after matching (Table 2).
Table 2. Comparison of Baseline Health Characteristics in GA versus GA + EA Groups.
| GA + EA | GA Only | |||||||||||
| Category | n | % | n | % | OR (95% CI) | p-value | ||||||
| Age | ||||||||||||
| < 50 | 141 | 27.06 | 125 | 23.99 | 0.353 | |||||||
| 50–65 | 205 | 39.35 | 199 | 38.2 | 0.91 (0.67–1.24) | |||||||
| 65–80 | 163 | 31.29 | 188 | 36.08 | 0.77 (0.56–1.06) | |||||||
| > 80 | 12 | 2.3 | 9 | 1.73 | 1.18 (0.48–2.9) | |||||||
| Sex | ||||||||||||
| Male | 259 | 49.71 | 283 | 54.32 | Reference | 0.137 | ||||||
| Female | 262 | 50.29 | 238 | 45.68 | 1.2 (0.94–1.53) | |||||||
| Demographics | ||||||||||||
| White | 447 | 85.8 | 464 | 89.06 | Reference | 0.574 | ||||||
| Black | 40 | 7.68 | 33 | 6.33 | 1.26 (0.78–2.03) | |||||||
| Asian | 5 | 0.96 | 4 | 0.77 | 1.3 (0.35–4.86) | |||||||
| Other | 4 | 0.77 | 2 | 0.38 | 2.08 (0.38–11.38) | |||||||
| Not reported | 25 | 4.8 | 18 | 3.45 | 1.44 (0.78–2.68) | |||||||
| Hispanic | 28 | 5.37 | 15 | 2.88 | 1.92 (1.01–3.63) | 0.043 | ||||||
| BMI | ||||||||||||
| < 18.5 | 1 | 0.19 | 0 | 0 | 252065.85 (0–∞) | 0.635 | ||||||
| 18.5–25 | 58 | 11.13 | 51 | 9.79 | Reference | |||||||
| 25–30 | 153 | 29.37 | 162 | 31.09 | 0.83 (0.54–1.28) | |||||||
| > 30 | 309 | 59.31 | 308 | 59.12 | 0.88 (0.59–1.33) | |||||||
| Functional status | ||||||||||||
| No dyspnea | 489 | 93.86 | 493 | 94.63 | Reference | 0.674 | ||||||
| Dyspnea with moderate exertion | 31 | 5.95 | 26 | 4.99 | 1.2 (0.7–2.05) | |||||||
| Dyspnea at rest | 1 | 0.19 | 2 | 0.38 | 0.5 (0.05–5.57) | |||||||
| Independent in ADLs | 518 | 99.42 | 515 | 98.85 | Reference | 0.315 | ||||||
| Partially/Totally dependent in ADLs | 3 | 0.58 | 6 | 1.15 | 0.5 (0.12–2) | |||||||
| ASA physical status | ||||||||||||
| 1 | 9 | 1.73 | 9 | 1.73 | Reference | 0.875 | ||||||
| 2 | 195 | 37.43 | 187 | 35.89 | 1.04 (0.41–2.68) | |||||||
| 3/4 | 317 | 60.84 | 325 | 62.38 | 0.98 (0.38–2.49) | |||||||
| Comorbidities | ||||||||||||
| Smoking | 97 | 18.62 | 98 | 18.81 | 0.99 (0.72–1.35) | 0.937 | ||||||
| Hypertension | 264 | 50.67 | 276 | 52.98 | 0.91 (0.72–1.16) | 0.457 | ||||||
| Diabetes | 81 | 15.55 | 104 | 19.96 | 0.74 (0.54–1.02) | 0.062 | ||||||
| COPD | 23 | 4.41 | 32 | 6.14 | 0.71 (0.41–1.22) | 0.212 | ||||||
| CHF | 4 | 0.77 | 3 | 0.58 | 1.34 (0.3–6) | 0.705 | ||||||
| CKD | 6 | 1.15 | 6 | 1.15 | 1 (0.32–3.12) | 1 | ||||||
| Steroid use | 62 | 11.9 | 48 | 9.21 | 1.33 (0.89–1.98) | 0.158 | ||||||
| Weight loss | 5 | 0.96 | 3 | 0.58 | 1.67 (0.4–7.04) | 0.478 | ||||||
| Wound infection | 13 | 2.5 | 13 | 2.5 | 1 (0.46–2.18) | 1 | ||||||
| Bleeding disorder | 10 | 1.92 | 9 | 1.73 | 1.11 (0.45–2.76) | 0.817 | ||||||
| Labs | ||||||||||||
| Hematocrit | 40.7 | 1.42 | 41.1 | 1.45 | 0.81 (0.62–1.05) | 0.113 | ||||||
| Wound Class | ||||||||||||
| 1-Clean | 431 | 82.73 | 423 | 81.19 | Reference | 0.556 | ||||||
| 2-Clean/Contaminated | 70 | 13.44 | 76 | 14.59 | 0.9 (0.64–1.28) | |||||||
| 3-Contaminated | 13 | 2.5 | 10 | 1.92 | 1.28 (0.55–2.94) | |||||||
| 4-Dirty/Infected | 7 | 1.34 | 12 | 2.3 | 0.57 (0.22–1.47) | |||||||
| Surgical complexity | ||||||||||||
| Operative Time | 204.23 | 98.63 | 198.29 | 101.66 | 1 (1–1) | 0.339 | ||||||
GA = general anesthesia; EA = epidural analgesia; ASA = American Society of Anesthesiologists; BMI = body mass index: ADLs = activities of daily living; COPD = chronic obstructive pulmonary disease; CHF = chronic heart failure; CKD = chronic kidney disease.
Figure 1 demonstrates survival probability (from 0 to 1) as a function of days following surgery (up to 30 days). The matched GA + EA group had a 0% thirty-day mortality rate vs 0.77% in GA (p = 0.99, Figure 1). The most common complication observed in the matched cohorts was surgical site infection (8.06% GA + EA vs 8.06% GA, [1.01 (0.65–1.58)], p = 0.958) (Table 3). There was a reoperation rate of 4.2% and 4.9% for GA + EA and GA, respectively ([0.86 (0.48–1.54)], p = 0.62). Readmission rates were 10.4% in GA + EA vs 10.6% in GA ([0.99 (0.66–1.47)], p = 0.96). Propensity score matched analysis found a statistically significant longer LOS in the GA + EA group (5.58 ± 4.00 days) vs the GA group (5.16 ± 4.22 days, [1.08 (1.03–1.14)], p = 0.003). No other significant differences in 30-day outcomes were observed between the matched cohorts.
Figure 1.
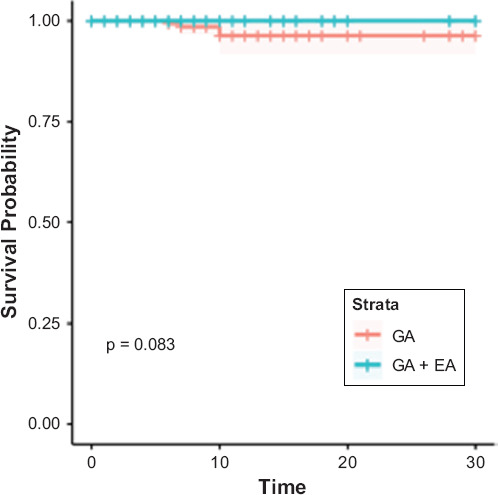
Survival Probability (From 0 to 1) As a Function of Days Following Surgery (Up to 30 Days)
Table 3. Outcomes in General Anesthesia versus General Anesthesia and Epidural.
| GA + EA | GA Only | |||||||||||
| Category | n | % | n | % | OR (95% CI) | p-value | ||||||
| Outcomes | ||||||||||||
| Death | 0 | 0 | 4 | 0.77 | 0 (0–Inf) | 0.993 | ||||||
| Return to OR | 22 | 4.22 | 26 | 4.99 | 0.86 (0.48–1.54) | 0.618 | ||||||
| Failure to wean | 3 | 0.58 | 6 | 1.15 | 0.51 (0.13–2.05) | 0.343 | ||||||
| Reintubation | 6 | 1.15 | 11 | 2.11 | 0.55 (0.2–1.51) | 0.249 | ||||||
| Surgical site infection | 42 | 8.06 | 42 | 8.06 | 1.01 (0.65–1.58) | 0.958 | ||||||
| Dehiscence | 8 | 1.54 | 7 | 1.34 | 1.08 (0.39–3.02) | 0.882 | ||||||
| Pneumonia | 9 | 1.73 | 16 | 3.07 | 0.57 (0.25–1.3) | 0.182 | ||||||
| Renal insufficiency | 2 | 0.38 | 4 | 0.77 | 0.51 (0.09–2.8) | 0.440 | ||||||
| Renal failure | 0 | 0 | 1 | 0.19 | 0 (0–Inf) | 0.996 | ||||||
| Stroke | 0 | 0 | 4 | 0.77 | 0 (0–Inf) | 0.993 | ||||||
| Cardiac arrest | 1 | 0.19 | 5 | 0.96 | 0.2 (0.02–1.75) | 0.147 | ||||||
| Acute MI | 4 | 0.77 | 3 | 0.58 | 1.37 (0.31–6.16) | 0.680 | ||||||
| Transfusion | 14 | 2.69 | 17 | 3.26 | 0.84 (0.41–1.72) | 0.636 | ||||||
| VTE | 7 | 1.34 | 18 | 3.45 | 0.39 (0.16–0.94) | 0.037 | ||||||
| UTI | 9 | 1.73 | 10 | 1.92 | 0.89 (0.36–2.22) | 0.804 | ||||||
| Sepsis | 14 | 2.69 | 14 | 2.69 | 1 (0.47–2.13) | 0.992 | ||||||
| Readmission | 54 | 10.36 | 55 | 10.56 | 0.99 (0.66–1.47) | 0.957 | ||||||
| Reoperation | 22 | 4.22 | 26 | 4.99 | 0.86 (0.48–1.54) | 0.618 | ||||||
| Length of Stay | 5.58 | 4 | 5.16 | 4.22 | 1.08 (1.03–1.14) | 0.003 | ||||||
| Number of complications | 0.22 | 0.51 | 0.28 | 0.54 | 0.82 (0.64–1.05) | 0.110 | ||||||
GA = general anesthesia; EA = epidural analgesia; OR = operating room; MI = myocardial ischemia; VTE = venous thromboembolism; UTI = urinary tract infection.
Discussion
Epidural analgesia is a common adjunctive technique for VHR. Recent studies have called into question the benefit of this technique, and our study aims to compare outcomes in VHR after epidural analgesia plus general anesthesia to general anesthesia alone. Our analysis demonstrates no difference in rates of post-operative complications with GA + EA, but does report a small, but statistically significant increase in hospital LOS with GA + EA.
This study builds on three prior conflicting studies evaluating the outcomes of EA in VHR. Our findings are consistent with results from two prior multicenter database studies in VHR. Prabhu et al. in their analysis of the Americas Hernia Society Quality Collaborative data, found that the EA group actually experienced an increased LOS, an increased risk of having any postoperative complication associated with having EA, and higher (not lower) pain scores.17 Karamanos et al. retrospectively analyzed 4983 patients who underwent elective abdominal wall reconstruction.15 The authors surprisingly found no impact of EA on pain control and actually longer hospital LOS and complication rates.
Although the single institution study by Fischer et al. demonstrated a reduced LOS and decreased medical morbidities with epidural use in VHR in those receiving epidurals, this reduction disappeared upon subgroup analysis where only the patients undergoing concurrent procedures in addition to VHR continued to demonstrate this benefit.6 This may suggest the majority of the morbidity benefit was derived from the effects of EA in the concurrent procedure rather than in VHR itself. Additionally, the epidural subgroup in Fischer et al. who underwent isolated hernia repair had lower ASA physical status scores than their GA counterparts. The better overall health of the EA group likely contributed to the reductions in complication rate, postoperative renal failure and overall better outcomes.
However, our study found no difference in complication rates after VHR using EA versus EA + GA, in contrast to some of the outcomes data from the aforementioned studies. In addition to comparable rates of complication between groups in our study, our analysis failed to demonstrate any statistically significant mortality benefits. This is particularly relevant in the era of enhanced recovery after surgery (ERAS) protocols.13 ERAS protocols are evidence-based quality improvement pathways reportedly associated with improved outcomes, shortened LOS, and decreased postoperative complications; in part, these protocols rely on multimodal analgesia to optimize pain control, accelerate recovery, and ultimately decrease resource utilization.18 Interestingly, in evaluating ERAS in VHR, two recent studies have reported conflicting results regarding LOS. While Majumder et al.4 found ERAS pathways to decrease LOS and improve recovery of bowel function, Stearns et al.13 found no difference in LOS. The reason, according to Stearns et al. was due to institutional preference to leave epidurals in until postoperative day three, upon which they were removed only if patients were not experiencing nausea, vomiting, or other signs of ileus. The authors thereby advocate for earlier removal of EA in patients who undergo less extensive dissection.4 In an ERAS study by Jensen et al. all 32 patients who underwent abdominal wall reconstruction for giant ventral hernia received EA which was discontinued on the second postoperative day.19 The authors found that pain scores, nausea and fatigue were decreased, although there was no difference in readmissions, postoperative complications or reoperation in the ERAS group versus controls. However, since EA was just one of many components of the ERAS pathway in the study group, it is not possible to draw any definite conclusions about its impact on postoperative outcomes.
In the present study, it is possible that the implementation of ERAS in VHR may partially account for the lack of benefit from EA, as the protocols appear able to achieve results similar to those originally intended with EA. However, no definitive data are yet available to support this conclusion. Regardless, our results are consistent with the growing body of literature arguing against EA for all VHRs.
The analgesic benefit derived from EA has classically been considered the gold standard for major abdominal surgery, as it achieves adequate pain control while avoiding the risks inherent with opioid-based regimes such as respiratory depression and gastrointestinal dysmotility. While this is true, the epidurals themselves are not devoid of risk. Complications of epidurals, although rare, can be serious and include epidural hematomas, infection, dural puncture, cerebrospinal fluid leak and spinal headache. More practically speaking, epidurals may be associated with certain everyday issues that complicate healthcare resource utilization and patient care. The time required to place epidurals is generally negligible but in difficult patients can delay operative start times. Institutional variability in the management of epidurals can also determine their clinical impact; patients may not be allowed out of bed with epidurals in place to theoretically reduce falls,20 thus delaying ambulation, which is well known to improve outcomes after abdominal surgery;21 urinary retention caused by epidurals leads some hospitals to require concomitant urinary catheterization, itself associated with side effects such as urinary tract infections.22 Many institutions allow only physicians or advanced practice nurses to manage epidurals, which may delay therapeutic intervention, especially on a busy surgical or acute pain management service.17 These factors must be considered when evaluating the role of epidurals in any surgical subtype.
Advances in regional anesthesia also call into question the role of EA. The presumptive analgesic benefit of epidurals in abdominal surgery may be achieved with alternative techniques such as transversus abdominus plane (TAP) or rectus sheath blocks. These techniques avoid the serious complications of epidurals, may be easier to perform and less costly to manage, and can achieve analgesia over a similar dermatomal pattern. Additionally, catheters may be placed with these techniques or liposomal bupivacaine may be used if longer-duration analgesia is desired. Indeed, recent studies have confirmed the analgesic potential of these blocks. Felling et al. reported that liposomal bupivacaine TAP blocks achieved equivalent analgesia with less adjunctive opioid use than epidurals in patients undergoing elective colorectal surgery.23 Other studies have similarly reported no benefit in pain reduction, outcomes, or LOS from epidurals versus peripheral regional anesthesia in transplant and orthopedic surgery.24 Further, while EA has shown improvements in outcomes in certain cardiac,25 vascular,26 and orthopedic27 surgeries, a growing body of literature suggests that there may be no benefit or even deleterious effects of EA in pancreaticoduodenal28 and colorectal surgery.29 As ERAS and multimodal analgesic protocols continue to be refined, the parameters and characteristics of patients, severity of, and type of surgery will allow better allocation of the appropriate analgesic technique to the correct patient.30,31,32
It is important to note that, in our study, the difference in the mean LOS between both groups was a fraction of a day. This difference is of questionable clinical significance and dependent on resource utilization, most prominently bed utilization. If patient rooms are used for additional patients promptly after discharge, then this may be considered a clinically significant improvement in resource utilization. However, if the bed remains empty for a prolonged period, then this observed difference in LOS is of no clinical or economic benefit. Other factors such as staffing costs are considered fixed costs that are less influenced by small differences in LOS. Therefore, discharging patients earlier in the same day does not necessarily decrease resource utilization.33
The limitations of our study stem from the nature of the ACS NSQIP database and its retrospective design. As with any multicenter dataset, ACS NSQIP is subject to potential reporting and sampling biases in addition to human error. However, quality control measures are in place to minimize such instances.16 Additionally, important variables which may have allowed for further interpretation of our results such as timing of epidural placement, specific drugs used in the epidural, and pain scores/measures of epidural efficacy were not included in this registry. Future multicenter prospective studies should consider these variables to allow for more robust analysis. Although the ACS NSQIP database enabled compilation of a large VHR population, propensity-score matching diminished our population size whereby increasing potential risk for type II error. However, propensity score matching also decreased effects of confounding by accounting for baseline health characteristics, and our results were similar to those found by two other large VHR studies.14,15
Conclusion
In the present investigation, epidural analgesia was associated with prolonged hospital LOS, and did not decrease the incidence of adverse 30-day outcomes in VHR. Epidural analgesia may therefore not be beneficial in this surgical population. Future studies should focus on alternative modes of analgesia to optimize pain control and outcomes for this common procedure.
Author Disclosure Statement
The authors have no financial affiliations to disclose.
Author Contributions
RDU, EYB, and AEW contributed to study conception and design. RDU, EYB, EEW, and AEW contributed to acquisition of data. RDU, EYB, EEW, and AEW contributed to analysis and interpretation of data. RDU, MRJ, EYB, EEW, AEW, ADK, and SPA contributed to drafting of manuscript. All authors contributed to critical revision of the manuscript.
Orcid ID
Mark R. Jones: 0000-0002-4781-2167; Ethan Y. Brovman: 0000-0003-3361-4351; Samuel P. Ang: 0000-0003-0913-3419; Alan D. Kaye: 0000-0003-2464-0187; Richard D. Urman: 0000-0002-0516-5977.
Abbreviations
VHR, ventral hernia repair; LOS, length of stay; GA + EA, general anesthesia plus epidural analgesia; GA, general anesthesia alone; ACS NSQIP, American College of Surgeons National Surgical Quality Improvement Program; ICD-9-CM, International Classification of Diseases, Ninth Revision, Clinical Modification; ICD-10-CM, International Classification of Diseases, Tenth Revision, Clinical Modification; CPT, Current Procedural Terminology; MAC, monitored anesthesia care; BMI, body mass index; ASA, American Society of Anesthesiologists; SD, standard deviation; RVU, relative value unit; ERAS, enhanced recovery after surgery.
References
- 1.Poulose BK, Shelton J, Phillips S, Moore D, Nealon W, Penson D, Beck W, Holzman MD. Epidemiology and cost of ventral hernia repair: making the case for hernia research. Hernia. 2012;16:179–183. doi: 10.1007/s10029-011-0879-9. [DOI] [PubMed] [Google Scholar]
- 2.Faylona JM. Evolution of ventral hernia repair. Asian J Endosc Surg. 2017;10:252–258. doi: 10.1111/ases.12392. [DOI] [PubMed] [Google Scholar]
- 3.Bisgaard T, Kehlet H, Bay-Nielsen MB, Iversen MG, Wara P, Rosenberg J, Friis-Andersen HF, Jorgensen LN. Nationwide study of early outcomes after incisional hernia repair. Br J Surg. 2009;96:1452–1457. doi: 10.1002/bjs.6728. [DOI] [PubMed] [Google Scholar]
- 4.Majumder A, Fayezizadeh M, Neupane R, Elliott HL, Novitsky YW. Benefits of multimodal enhanced recovery pathway in patients undergoing open ventral hernia repair. J Am Coll Surg. 2016;222:1106–1115. doi: 10.1016/j.jamcollsurg.2016.02.015. [DOI] [PubMed] [Google Scholar]
- 5.Carli F, Mayo N, Klubien K, Schricker T, Trudel J, Belliveau P. Epidural analgesia enhances functional exercise capacity and health-related quality of life after colonic surgery: results of a randomized trial. Anesthesiology. 2002;97:540–549. doi: 10.1097/00000542-200209000-00005. [DOI] [PubMed] [Google Scholar]
- 6.Fischer JP, Nelson JA, Wes AM, Wink JD, Yan C, Braslow BM, Chen L, Kovach SJ. The use of epidurals in abdominal wall reconstruction: an analysis of outcomes and cost. Plast Reconstr Surg. 2014;133:687–699. doi: 10.1097/01.prs.0000438048.91139.31. [DOI] [PubMed] [Google Scholar]
- 7.Jorgensen H, Wetterslev J, Moiniche S, Dahl JB. Epidural local anaesthetics versus opioid-based analgesic regimens on postoperative gastrointestinal paralysis, PONV and pain after abdominal surgery. Cochrane Database Syst Rev. 2000:CD001893. doi: 10.1002/14651858.CD001893. [DOI] [PubMed] [Google Scholar]
- 8.Guay J, Nishimori M, Kopp S. Epidural local anaesthetics versus opioid-based analgesic regimens for postoperative gastrointestinal paralysis, vomiting and pain after abdominal surgery. Cochrane Database Syst Rev. 2016;7:CD001893. doi: 10.1002/14651858.CD001893.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rigg JR, Jamrozik K, Myles PS, Silbert BS, Peyton PJ, Parsons RW, Collins KS, Group MATS Epidural anaesthesia and analgesia and outcome of major surgery: a randomised trial. Lancet. 2002;359:1276–1282. doi: 10.1016/S0140-6736(02)08266-1. [DOI] [PubMed] [Google Scholar]
- 10.Groeben H. Epidural anesthesia and pulmonary function. J Anesth. 2006;20:290–299. doi: 10.1007/s00540-006-0425-6. [DOI] [PubMed] [Google Scholar]
- 11.Rodgers A, Walker N, Schug S, McKee A, Kehlet H, van Zundert A, Sage D, Futter M, Saville G, Clark T, MacMahon S. Reduction of postoperative mortality and morbidity with epidural or spinal anaesthesia: results from overview of randomised trials. BMJ. 2000;321:1493. doi: 10.1136/bmj.321.7275.1493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lattermann R, Wykes L, Eberhart L, Carli F, Meterissian S, Schricker T. A randomized controlled trial of the anticatabolic effect of epidural analgesia and hypocaloric glucose. Reg Anesth Pain Med. 2007;32:227–232. doi: 10.1016/j.rapm.2007.02.009. [DOI] [PubMed] [Google Scholar]
- 13.Stearns E, Plymale MA, Davenport DL, Totten C, Carmichael SP, Tancula CS, Roth JS. Early outcomes of an enhanced recovery protocol for open repair of ventral hernia. Surg Endosc. 2018;32:2914–2922. doi: 10.1007/s00464-017-6004-0. [DOI] [PubMed] [Google Scholar]
- 14.Prabhu AS, Krpata DM, Perez A, Phillips S, Huang LC, Haskins IN, Rosenblatt S, Poulose BK, Rosen MJ. Is it time to reconsider postoperative epidural analgesia in patients undergoing elective ventral hernia repair?: an AHSQC analysis. Ann Surg. 2018;267:971–976. doi: 10.1097/SLA.0000000000002214. [DOI] [PubMed] [Google Scholar]
- 15.Karamanos E, Dream S, Falvo A, Schmoekel N, Siddiqui A. Use of epidural analgesia as an adjunct in elective abdominal wall reconstruction: a review of 4983. Cases. Perm J. 2017;21 doi: 10.7812/TPP/16-115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. American College of Surgeons. American College of Surgeons-National surgery Quality Improvement Program. ACS-NSQIP Participant Use File User’s Guide [Online] [Google Scholar]
- 17.Prabhu AS, Krpata DM, Perez A, Phillips S, Huang LC, Haskins IN, Rosenblatt S, Poulose BK, Rosen MJ. Is it time to reconsider postoperative epidural analgesia in patients undergoing elective ventral hernia repair?: an AHSQC analysis. Ann Surg. 2018;267:971–976. doi: 10.1097/SLA.0000000000002214. [DOI] [PubMed] [Google Scholar]
- 18.Lau CS, Chamberlain RS. Enhanced Recovery after surgery programs improve patient outcomes and recovery: a meta-analysis. World J Surg. 2017;41:899–913. doi: 10.1007/s00268-016-3807-4. [DOI] [PubMed] [Google Scholar]
- 19.Jensen KK, Brondum TL, Harling H, Kehlet H, Jorgensen LN. Enhanced recovery after giant ventral hernia repair. Hernia. 2016;20:249–256. doi: 10.1007/s10029-016-1471-0. [DOI] [PubMed] [Google Scholar]
- 20.Elsharydah A, Williams TM, Rosero EB, Joshi GP. Epidural analgesia does not increase the rate of inpatient falls after major upper abdominal and thoracic surgery: a retrospective case-control study. Can J Anaesth. 2016;63:544–551. doi: 10.1007/s12630-016-0602-5. [DOI] [PubMed] [Google Scholar]
- 21.Asada J, Ida M, Sato M, Okamoto N, Kawaguchi M. Associated factors with delayed ambulation after abdominal surgery. J Anesth. 2019;33:680–684. doi: 10.1007/s00540-019-02696-4. [DOI] [PubMed] [Google Scholar]
- 22.Haskins IN, Ilie RN, Krpata DM, Perez AJ, Butler RS, Prabhu AS, Rosenblatt S, Rosen MJ. Association of thoracic epidural pain management with urinary retention after complex abdominal wall reconstruction. Am Surg. 2018;84:1808–1813. [PubMed] [Google Scholar]
- 23.Felling DR, Jackson MW, Ferraro J, Battaglia MA, Albright JJ, Wu J, Genord CK, Brockhaus KK, Bhave RA, McClure AM, Shanker BA, Cleary RK. Liposomal bupivacaine transversus abdominis plane block versus epidural analgesia in a colon and rectal surgery enhanced recovery pathway: a randomized clinical trial. Dis Colon Rectum. 2018;61:1196–1204. doi: 10.1097/DCR.0000000000001211. [DOI] [PubMed] [Google Scholar]
- 24.Khan J, Katz J, Montbriand J, Ladak S, McCluskey S, Srinivas C, Ko R, Grant D, Bradbury A, LeManach Y, Clarke H. Surgically placed abdominal wall catheters on postoperative analgesia and outcomes after living liver donation. Liver Transpl. 2015;21:478–486. doi: 10.1002/lt.24073. [DOI] [PubMed] [Google Scholar]
- 25.Caputo M, Alwair H, Rogers CA, Pike K, Cohen A, Monk C, Tomkins S, Ryder I, Moscariello C, Lucchetti V, Angelini GD. Thoracic epidural anesthesia improves early outcomes in patients undergoing off-pump coronary artery bypass surgery: a prospective, randomized, controlled trial. Anesthesiology. 2011;114:380–390. doi: 10.1097/ALN.0b013e318201f571. [DOI] [PubMed] [Google Scholar]
- 26.Park WY, Thompson JS, Lee KK. Effect of epidural anesthesia and analgesia on perioperative outcome: a randomized, controlled Veterans Affairs cooperative study. Ann Surg. 2001;234:560–569. doi: 10.1097/00000658-200110000-00015. discussion 9–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Farag E, Dilger J, Brooks P, Tetzlaff JE. Epidural analgesia improves early rehabilitation after total knee replacement. J Clin Anesth. 2005;17:281–285. doi: 10.1016/j.jclinane.2004.08.008. [DOI] [PubMed] [Google Scholar]
- 28.Pratt WB, Steinbrook RA, Maithel SK, Vanounou T, Callery MP, Vollmer CM., Jr. Epidural analgesia for pancreatoduodenectomy: a critical appraisal. J Gastrointest Surg. 2008;12:1207–1220. doi: 10.1007/s11605-008-0467-1. [DOI] [PubMed] [Google Scholar]
- 29.Zutshi M, Delaney CP, Senagore AJ, Mekhail N, Lewis B, Connor JT, Fazio VW. Randomized controlled trial comparing the controlled rehabilitation with early ambulation and diet pathway versus the controlled rehabilitation with early ambulation and diet with preemptive epidural anesthesia/analgesia after laparotomy and intestinal resection. Am J Surg. 2005;189:268–272. doi: 10.1016/j.amjsurg.2004.11.012. [DOI] [PubMed] [Google Scholar]
- 30.Stone A, Roberts A, de Boer HD, Kaye AD, Fawcett WJ, Urman RD. Recommendations for Managing Opioid-Tolerant Surgical Patients within Enhanced Recovery Pathways. Curr Pain Headache Rep. 2020 May 8;24(6):28. doi: 10.1007/s11916-020-00856-w. doi. [DOI] [PubMed] [Google Scholar]
- 31.Elhassan A, Elhassan I, Elhassan A, Sekar KD, Rubin RE, Urman RD, Cornett EM, Kaye AD. Essential Elements for Enhanced Recovery After Intra-Abdominal Surgery. Curr Pain Headache Rep. 2019 Apr 30;23(5):35. doi: 10.1007/s11916-019-0772-2. doi. [DOI] [PubMed] [Google Scholar]
- 32.Kim AJ, Yong RJ, Urman RD. The Role of Transversus Abdominis Plane Blocks in Enhanced Recovery After Surgery Pathways for Open and Laparoscopic Colorectal Surgery. Laparoendosc Adv Surg Tech A. 2017 Sep;27(9):909–914. doi: 10.1089/lap.2017.0337. doi. Epub 2017 Jul 25. [DOI] [PubMed] [Google Scholar]
- 33.Dexter F, Epstein RH, Traub RD, Xiao Y. Making management decisions on the day of surgery based on operating room efficiency and patient waiting times. Anesthesiology. 2004;101:1444–1453. doi: 10.1097/00000542-200412000-00027. [DOI] [PubMed] [Google Scholar]


