Abstract
OBJECTIVES
This study aims at comparing the tone-burst (TB) and narrow-band (NB) CE-chirp stimuli in terms of amplitude, latency, and interaural asymmetry ratio (IAR) in ocular vestibular evoked myogenic potentials (oVEMP).
MATERIALS and METHODS
In this prospective study, we enrolled 60 healthy subjects (27 men, 33 women) with a mean age of 25.83 (range, 18–48) years. Otological examination was normal in all the subjects. The subjects did not have any otological disease. All the subjects underwent oVEMP testing. We used 500 Hz TB stimulus and 500 Hz NB CE-chirp stimulus in random order. oVEMP test was performed at 100 dB normalized hearing level. P1 latency, N1 latency, and P1N1 amplitude were measured for each ear and stimulus, and IAR was calculated.
RESULTS
Ocular VEMPs were obtained from all the subjects for both the stimuli. P1 and N1 latencies were significantly shorter in chirp stimulus than in TB stimulus for both the sides (p<0.0001). P1 and N1 amplitudes were significantly higher for chirp stimulus than for TB stimulus for both the sides (p<0.0001). There was no significant difference between the ears in IAR between the 2 types of stimuli.
CONCLUSION
Narrow-band CE-chirp stimulus is an effective stimulus to evoke oVEMP with higher amplitudes and shortened latencies.
Keywords: Ocular vestibular evoked myogenic potentials, narrow band, chirp, tone-burst
INTRODUCTION
Vestibular stimulated myogenic potentials (VEMPs) are vestibular reflexes thought to originate from otolithic end organs. VEMPs are recorded from the extraocular and cervical muscles. Vestibular organs are stimulated by air or bone pathways to elicit these reflexes. To stimulate these reflexes, short bursts of loud air-conducted sound or bone-conducted skull vibration are used. Surface electrodes are used to record the muscle activity [1].
Ocular VEMPs (oVEMPs) are recorded from the extraocular muscles. Inferior oblique muscles reflect the otolithic function of the contralateral utricle [2, 3]. The upward gaze increases the amplitude of the response recorded in oVEMP by increasing the inferior oblique muscle contraction [4]. The best oVEMP responses are recorded at 400–1,000 Hz range. However, the optimal frequency range is still controversial [5–7].
Chirp signal is used to increase the synchronous activity of the hearing nerve [8]. Especially, CE-chirp stimulation has been increasingly used in audiological tests, such as auditory brain responses (ABRs) and auditory steady-state responses [9].
Knowledge on waveform characteristics of oVEMP in response to chirp stimulus is very limited. This study aims at comparing the tone-burst (TB) and narrow-band (NB) CE-chirp stimuli in terms of amplitude, latency, and interaural asymmetry ratio (IAR).
MATERIALS AND METHODS
Subjects
In this prospective study, we enrolled 60 healthy subjects (27 men, 33 women) with a mean age of 25.83 (range 18–48) years. All the subjects underwent neuro-otological evaluation including audiometric examination. Individuals without neurological or otological illnesses with normal pure tone audiometry and tympanometry were included. All the subjects signed the consent form before the tests.
All of the procedures were performed in accordance with the Declaration of Helsinki and were approved by the Ethics Committee of University of Health Science Turkey (18/269).
Ocular VEMP Recording
oVEMP recording was performed in a quiet room in a sitting position. Surface electrodes were used. Electrodes’ impedance level was set below 5 kΩ. The test was performed with Interacoustics Eclipse EP 15 (Eclipse EP15; Interacoustics, Assens, Denmark). In each eye, the negative electrode was placed 1 cm below the lower eyelid (on inferior oblique muscle), the positive electrode was placed on the chin, and the ground electrode was placed on the forehead (Ambu®Neuroline™ 720; Ambu, Ballerup, Denmark). The subjects were asked to look at a fixation point approximately 60 cm from the eyes and had an upward gaze of approximately 30°.
We used 500 Hz TB, and the stimuli intensity was 100 dB normalized hearing level (nHL). The device was calibrated by licensed technical personnel according to the International Organization for Standardization 389-6 standards. For 500 Hz TB, rise–fall and plateau time were 2 ms (2-2-2). Furthermore, 500 Hz NB CE-chirp (360–720 Hz) stimulus was also delivered at 100 dB nHL intensity; stimulus duration was 9 ms. Stimulus rate was set to 5.1/s, analysis time to 55 ms, and polarity rarefaction. Total of 250 stimuli were averaged.
The electromyography was amplified (10,000×) and was bandpass filtered (10–1,200 Hz). Calibrated insert headphones (Ear Tone ABR 3A; 3M, Minneapolis, USA) were used.
Evaluation of Test Results
For each stimulus, P1-N1 waveform was observed. For each stimulus and each ear, P1 latency, N1 latency, and P1N1 amplitude were recorded. IAR was also calculated (IAR=left ear P1N1 amplitude–right ear P1N1 amplitude/left ear P1N1 amplitude+right ear P1N1 amplitude). When the same waveform with the same latency was obtained in 2 repeated tests, the waveform was accepted to take into account.
Statistical Analysis
Data were analyzed with the software Statistical Package for the Social Sciences version 20.0 for Mac (IBM Corp.; Armonk, NY, USA). Distribution of the results was analyzed using the Shapiro–Wilk test. When normal distribution was observed, groups were compared using the paired t-test. Otherwise, the groups were compared with the Wilcoxon test. p<0.05 was considered statistically significant.
RESULTS
There were 60 volunteers, and their mean age was 25.83 (range, 18–48 years) years. Both the stimuli evoked oVEMPs in all the subjects. P1 and N1 latencies were significantly shorter in chirp stimulus than in TB stimulus for both the sides (p<0.0001). Chirp stimulus evoked more robust oVEMP amplitudes compared with TB stimulus for both the sides (p<0.0001). TB and CE-Chirp responces in right ear are shown in Figure 1 and in left ear are shown in Figure 2. IAR for TB stimulus was 0.13±0.11, and IAR for chirp stimulus was 0.13±0.11. There was no significant difference in IAR between the two types of stimuli (p=0.928). Descriptive statistics of oVEMP by stimuli types are shown in Table 1.
Figure 1.
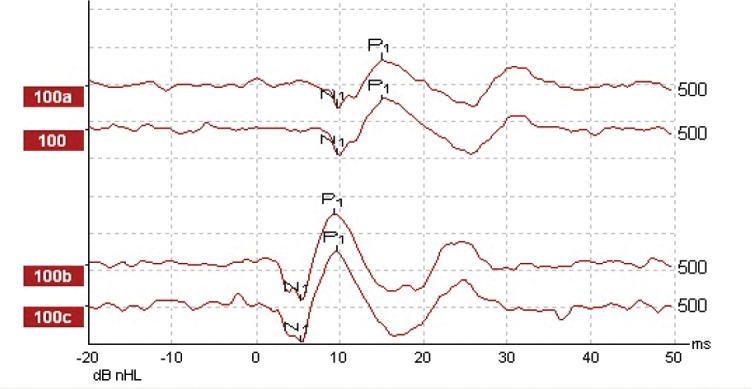
Tone-burst (top) and chirp (bottom) evoked oVEMPs on right side. Note the differences in latencies and amplitudes of the waves and waveform quality of oVEMP in response to tone-bursts and chirps.
Figure 2.

Tone-burst (top) and chirp (bottom) evoked oVEMPs on left side. Note the differences in latencies and amplitudes of the waves and waveform quality of oVEMP in response to tone-bursts and chirps.
Table 1.
oVEMP results by stimuli type (mean±standard deviation, median, minimum-maximum values)
| Right 500 Hz tone-burst | Right 500 Hz narrow-band chirp | p | Left 500 Hz tone-burst | Left 500 Hz narrow-band chirp | p | |
|---|---|---|---|---|---|---|
| P1 latency (ms) | <0.0001 | <0.0001 | ||||
| mean±SD | 15.31±1.64 | 9.77±1.46 | 15.70±1.54 | 9.84±1.47 | ||
| median | 15.33 | 9.67 | 15.67 | 9.67 | ||
| min–max | (12.33–22.67) | (6.67–16.00) | (10.67±21.00) | (6.67±15.67) | ||
| IQR | 1.34 | 1.33 | 1.67 | 1.67 | ||
| N1 latency (ms) | <0.0001 | <0.0001 | ||||
| mean±SD | 10.73±1.57 | 5.12±1.64 | 10.53±1.33 | 5.08±1.45 | ||
| median | 10.16 | 5.00 | 10.16 | 4.83 | ||
| min–max | (9.33±19.00) | (3.33±13.33) | (8.00±16.00) | (3.00±11.33) | ||
| IQR | 2.00 | 1.67 | 1.57 | 1.67 | ||
| P1N1 amplitude (μV) | <0.0001 | <0.0001 | ||||
| mean±SD | 12.66±10.09 | 17.08±13.41 | 11.87±8.56 | 16.25±11.75 | ||
| median | 9.57 | 17.08 | 8.77 | 13.19 | ||
| min–max | (2.83–45.97) | (2.14–56.45) | (1.60–42.13) | (3.03–50.6) | ||
| IQR | 9.58 | 13.04 | 1.25 | 1.67 |
ms: millisecond; SD: standard deviation; IQR: interquartile range; μV: microvolt.
p<0.05 was considered statistically significant
DISCUSSION
Ocular VEMP is a noninvasive electrophysiological measure that shows the function of the contralateral utricle and the superior division of the vestibular nerve [10]. oVEMP plays an important role in the diagnosis of superior canal dehiscence, vestibular neuritis, benign paroxysmal positional vertigo, vestibular schwannoma, Menière’s disease, and central vestibular disorders [1].
There are many factors affecting oVEMP response, such as stimulus frequency, stimulus level, gaze elevation, and electrode location. Response rates, amplitudes, and latency are directly affected by these factors [4].
The main objective of this study is to compare the TB and NB CE-chirp stimuli in terms of amplitude, latency, and IAR. We obtained a very high response rate (100%). However, Özgür et al. [11] preferred TB stimulation rather than chirp stimulus because of a higher response rate for cervical VEMP. On the basis of our findings, one can propose that with chirp stimulus, latencies were shorter, and amplitudes were higher. Waveform morphology was also better. These results clearly show that 500 Hz NB CE-chirp stimulus is effective to evoke oVEMP. This result is consistent with the results of Walther et al.,[12] who compared band-limited chirp stimulation and short TB stimulation in healthy individuals. However, consistent with the findings of Özgür et al. [11], which showed lower amplitudes with chirp stimulus, Zakaria et al. [13] also found lower amplitudes with their custom-made chirp stimulus.
We also checked contralateral wave recordings to make sure that we have evaluated the correct measurements and also to avoid ipsilateral muscle responses. On the contralateral waves, we observed prolonged latencies and decreased amplitudes shown in Figure 3.
Figure 3.
Tone-burst and chirp stimulus wave examples with contralateral waves. C indicates contralateral waves. Note the different morphology and characteristics of the waves in response to each tone-burst and chirp stimuli, consistency of response repetitions, and also latency shift in contralateral waves.
In healthy individuals, stimulus frequencies between 400 and 800 Hz have been shown to be the most effective range for generating oVEMP [6, 7]. These findings made the authors of these studies think that tuning was determined at or before the level of the vestibular nucleus with the greatest contribution of otolith organs [4]. Cebulla et al. [14] published the results of their pilot study in which they used sequential and quasi-simultaneous NB chirps for evoking cervical evoked myogenic potentials and they have found that the amplitudes were highest at 500 Hz as expected. Therefore, we used 500 Hz band-limited chirp stimuli and 500 Hz TB stimuli in this study. To avoid muscle fatigue, NB CE-chirp and TB stimuli were delivered in random order.
Literature was focused on the use of chirp stimulus in ABR testing. In ABR, wave V latency is shorter in chirp stimulus than in any other stimuli [15]. It is believed that it happens because stimulation at all the frequency regions of the cochlea provides a faster cochlear stimulation [11]. We obtained P1 and N1 waves at shorter latencies with NB CE-chirp stimuli, which are in compliance with the literature. Probably, TB stimulus’ rise–fall time also causes this delay.
The upward gaze is one of the main factors that affect oVEMP amplitude and response rate because it is believed that an upward gaze makes the inferior oblique muscle more superficial. Murnane et al. [4] reported an increased response rate of oVEMP testing after 20° gaze elevation. Govender et al. [16] suggested that maximum gaze elevation should be applied before deciding that the response is absent. We used 30° upward gaze and obtained a 100% response rate.
Average threshold of sound level suggested by Wang et al. [17] is 110 dBpeak SPL. In another study, Murnane et al. [4] had an average oVEMP threshold of 125 dB peak SPL at 500 Hz. We used 100 dB nHL and obtained 100% response rates, which can be affected by sound level.
The limitation of this study is the fact that it lacks patient groups.
CONCLUSION
Narrow-band CE-chirp stimulus is an effective stimulus to evoke oVEMP with higher amplitudes and shortened latencies. NB CE-chirp might be a promising stimulus for clinical use.
MAIN POINTS.
oVEMP is a myogenic response which is originated from utricle and activates superior vertibular nerve.
Chirp signal is used to increase the synchronous activity of the hearing nerve.
NB CE-chirp stimulus is an effective stimulus to evoke oVEMP with higher amplitudes and shortened latencies.
Footnotes
Ethics Committee Approval: Ethics committee approval was received from the Ethics Committee of University of Health Science Turkey, Ankara, Turkey.
Informed Consent: Written informed consent was obtained from the patients who participated in this study.
Peer-review: Externally peer-reviewed.
Author Contributions: Concept – C.K., B.S.; Design - C.K., F.C.A.Ö.; Supervision - B.S.; Resource - V.K.C., B.S.; Materials - C.K., F.C.A.Ö.; Data Collection and/or Processing – V.K.C., F.C.A.Ö.; Analysis and/or Interpretation – C.K., F.C.A.Ö.; Literature Search – C.K.; Writing – C.K; Critical Reviews – B.S.
Conflict of Interest: The authors have no conflict of interest to declare.
Financial Disclosure: The authors declared that this study has received no financial support.
REFERENCES
- 1.Weber KP, Rosengren SM. Clinical utility of ocularvestibular-evoked myogenic potentials (oVEMPs) Curr Neurol Neurosci Rep. 2015;15:22. doi: 10.1007/s11910-015-0548-y. [DOI] [PubMed] [Google Scholar]
- 2.Oh SY, Kim JS, Yang TH, Shin BS, Jeong SK. Cervical and ocular vestibular-evoked myogenic potentials in vestibular neuritis: comparison between air- and bone-conducted stimulation. J Neurol. 2013;260:2102–9. doi: 10.1007/s00415-013-6953-8. [DOI] [PubMed] [Google Scholar]
- 3.Murofushi T, Nakahara H, Yoshimura E, Tsuda Y. Association of air-conducted sound oVEMP findings with cVEMP and caloric test findings in patients with unilateral peripheral vestibular disorders. Acta Otolaryngol. 2011;131:945–50. doi: 10.3109/00016489.2011.580003. [DOI] [PubMed] [Google Scholar]
- 4.Murnane OD, Akin FW, Kelly KJ, Byrd S. Effects of stimulus and recording parameters on the air conduction ocular vestibular evoked myogenic potential. J Am Acad Audiol. 2011;22:469–80. doi: 10.3766/jaaa.22.7.7. [DOI] [PubMed] [Google Scholar]
- 5.Todai JK, Congdon SL, Sangi-Haghpeykar H, Cohen HS. Ocular vestibular evoked myogenic potentials in response to three test positions and two frequencies. Laryngoscope. 2014;124:E237–40. doi: 10.1002/lary.24487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Singh NK, Barman A. Characterizing the frequency tuning properties of air-conduction ocular vestibular evoked myogenic potentials in healthy individuals. Int J Audiol. 2013;52:849–54. doi: 10.3109/14992027.2013.822994. [DOI] [PubMed] [Google Scholar]
- 7.Park HJ, Lee IS, Shin JE, Lee YJ, Park MS. Frequency tuning characteristics of cervical and ocular vestibular evoked myogenic potentials induced by air-conducted tone bursts. Clin Neurophysiol. 2010;121:85–9. doi: 10.1016/j.clinph.2009.10.003. [DOI] [PubMed] [Google Scholar]
- 8.Bargen GA. Chirp-Evoked Auditory Brainstem Response in Children: A Review. Am J Audiol. 2015;24:573–83. doi: 10.1044/2015_AJA-15-0016. [DOI] [PubMed] [Google Scholar]
- 9.Walther LE, Cebulla M. Band limited chirp stimulation in vestibular evoked myogenic potentials. Eur Arch Otorhinolaryngol. 2016;273:2983–91. doi: 10.1007/s00405-015-3888-y. [DOI] [PubMed] [Google Scholar]
- 10.Felipe L, Kingma H. Ocular vestibular evoked myogenic potentials. Int Arch Otorhinolaryngol. 2014;18:77–9. doi: 10.1055/s-0034-1388953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Özgür A, Çelebi Erdivanlı Ö, Özergin CZ, Terzi S, Yiğit E, Demirci M, et al. Comparison of Tone Burst, Click and Chirp Stimulation in Vestibular Evoked Myogenic Potential Testing in Healthy People. J Int Adv Otol. 2015;11:33–5. doi: 10.5152/iao.2015.927. [DOI] [PubMed] [Google Scholar]
- 12.Walther LE, Cebulla M. Band limited chirp stimulation in vestibular evoked myogenic potentials. Eur Arch Otorhinolaryngol. 2016;273:2983–91. doi: 10.1007/s00405-015-3888-y. [DOI] [PubMed] [Google Scholar]
- 13.Zakaria MN, Zainun Z, Cheu Lih A. Considerations when Analyzing Vestibular Evoked Myogenic Potential (VEMP) Outcomes Elicited by Chirp Stimulus in Healthy Participants. J Int Adv Otol. 2015;11:271–2. doi: 10.5152/iao.2015.1703. [DOI] [PubMed] [Google Scholar]
- 14.Cebulla M, Walther LE. Cervical vestibular evoked myogenic potentials via air conduction delivered by either sequentially or quasi-simultaneously presented narrow-band chirp stimuli. Int J Audiol. 2019;58:174–9. doi: 10.1080/14992027.2018.1534280. [DOI] [PubMed] [Google Scholar]
- 15.Rodrigues GR, Ramos N, Lewis DR. Comparing auditory brainstem responses (ABRs) to toneburst and narrow band CE-chirp in young infants. Int J Pediatr Otorhinolaryngol. 2013;77:1555–60. doi: 10.1016/j.ijporl.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 16.Govender S1, Rosengren SM, Colebatch JG. The effect of gaze direction on the ocular vestibular evoked myogenic potential produced by air-conducted sound. Clin Neurophysiol. 2009;120:1386–91. doi: 10.1016/j.clinph.2009.04.017. [DOI] [PubMed] [Google Scholar]
- 17.Wang SJ, Jaw FS, Young YH. Ocular vestibular-evoked myogenic potentials elicited from monaural versus binaural acoustic stimulations. Clin Neurophysiol. 2009;120:420–3. doi: 10.1016/j.clinph.2008.10.157. [DOI] [PubMed] [Google Scholar]



