Abstract
Background
Acceptance and commitment therapy (ACT) is a behavioral health intervention with strong empirical support for chronic pain but, to date, widespread dissemination is limited. Digital solutions improve access to care and can be integrated into patients’ everyday lives.
Objective
ACTsmart, a guided smartphone-delivered ACT intervention, was developed to improve the accessibility of an evidence-based behavioral treatment for chronic pain. In the present study, we evaluated the preliminary efficacy of ACTsmart in adults with chronic pain.
Methods
The study was an open-label pilot trial. The treatment lasted for 8 weeks, and participants completed all outcome measures at pretreatment and posttreatment and at 3-, 6-, and 12-month follow-ups, with weekly assessments of selected measures during treatment. The primary outcome was pain interference. The secondary outcomes were psychological flexibility, values, insomnia, anxiety, depressive symptoms, health-related quality of life, and pain intensity. All outcomes were analyzed using linear mixed-effects models.
Results
The sample consisted of 34 adults (88% women) with long-standing chronic pain (M=20.4 years, SD=11.7). Compliance to treatment was high, and at the end of treatment, we observed a significant improvement in the primary outcome of pain interference (d = –1.01). All secondary outcomes significantly improved from pretreatment to posttreatment with small to large effect sizes. Improvements were maintained throughout 12 months of follow-up.
Conclusion
The results of this pilot study provide preliminary support for ACTsmart as an accessible and effective behavioral health treatment for adults with chronic pain and warrant a randomized controlled trial to further evaluate the efficacy of the intervention.
Keywords: Acceptance and Commitment Therapy, Chronic Pain, mHealth, Psychological Inflexibility, Digital Behavioral Health Intervention
Introduction
Chronic pain is common and affects 20% of adults [1], often leading to reduced functioning in daily activities, in social situations, and at work [2]. Adults with chronic pain are at an elevated risk for insomnia, anxiety, depression, suicidality, and reduced quality of life (QoL) [3, 4]. Chronic pain is complex, and neither medical procedures nor pharmacological treatments are sufficiently effective at alleviating symptoms and pain-related functional impairments [4]. Behavioral health interventions are often needed to meaningfully increase function and QoL [5–7].
Acceptance and commitment therapy (ACT) is a behavioral treatment approach developed from cognitive behavioral therapy (CBT). Although ACT was not originally developed to treat chronic pain, considerable evidence supports its use [8–11]. Many chronic pain patients focus on reducing or eliminating their pain and the associated emotional discomfort (e.g., by avoiding situations that might elicit pain). This is usually an effective strategy in the short term, as it is followed by immediate reductions in pain or discomfort. In the long term, however, these avoidance behaviors commonly lead to a way of life that is governed by attempts to control the level of pain and discomfort at the cost of reducing engagement in important life activities. In ACT, this avoidant behavior pattern is called psychological inflexibility [12], and in the context of chronic pain these behaviors can lead to reduced pain tolerance as well as increased pain intensity [13, 14]. The objective of ACT is not to reduce pain but to increase an individual’s psychological flexibility and hence reduce the dominance of pain and pain-related discomfort on a person’s behavior as well as to increase value-driven behaviors. Multiple systematic and meta-analytic reviews have concluded that ACT for chronic pain is efficacious and effective in comparison with inactive control conditions or treatment as usual, with small to large effect sizes for measures of physical functioning, anxiety, depression, and psychological flexibility; medium to large effect sizes for measures of pain acceptance and psychological flexibility; medium effect sizes for pain interference; and small effect sizes for disability, pain intensity, and QoL [8–11, 15].
Unfortunately, access to ACT and other CBT programs for chronic pain treatment continues to be restricted due to health care system constraints such as long waiting lists and a shortage of trained health care professionals coupled with patient-derived challenges, such as physical and mobility limitations and patients residing far from a clinic. Altogether, limited access results in many patients with chronic pain either failing to receive or waiting very long for evidence-based care [16, 17].
One major breakthrough in improving access to behavioral health treatments is internet-based solutions [18, 19]. A recent systematic review on internet treatment for chronic pain based on CBT or ACT principles presented a number of beneficial effects [20]. For example, positive effects of small to moderate effect sizes were found for disability or pain interference outcomes, pain intensity, catastrophizing, depression, and anxiety. These results are in line with those of a previous systematic review on internet treatments for pain [21]. Beyond treatment effects, additional strengths of internet-delivered behavioral treatment include reduced cost and increased convenience for users, ability to reach isolated groups, continual access to treatment content, and reduced health care system costs [20]. All in all, six randomized controlled trials on digitally delivered ACT for patients with chronic pain have been conducted thus far; one was a noninferiority trial comparing video teleconferencing with in-person delivery [22–27].
One digital solution with increasing traction is delivering content via smartphone. Smartphones are portable, most often on, connected [28], and have exceeded desktop computers in terms of internet use [29]. Smartphones provide opportunities for real-time assessment and monitoring, dynamic adaptations and personalization of interventions, and context awareness to create opportunities for ecological momentary assessments [28]. The use of smartphone-based treatment also increases a therapist’s ability to react in real time to a patient’s need for support during the intervention [30]. Prior research has demonstrated that smartphone applications that use reminders and instant feedback and that bring psychoeducation, assignments, and assessments closer to users’ everyday lives increase the accessibility and reach of effective treatment [31]. The many easily accessible interactions with the treatment may also enhance treatment outcomes.
There are several smartphone applications available that are marketed to improve chronic pain, and attempts have been made to establish the quality of these [32–34]. Overall, the findings show that the majority of available applications do not follow evidence-based guidelines, do not involve health care providers or experts in the development process, and have not undergone any scientific studies of effect and/or validity. Thus, despite the wealth of pain applications, few have undergone efficacy testing.
In light of this, we developed ACTsmart, a stand-alone smartphone-delivered ACT treatment for adult patients with chronic pain developed by clinicians and researchers in collaboration with experienced end-user professionals, programmers, and potential end users. The aim of the present study was to evaluate the preliminary immediate and long-term effects of ACTsmart on pain interference, psychological flexibility, values, insomnia, depression, anxiety, pain intensity, and health-related QoL.
Methods
Design and Procedure
The present study was a pilot trial with one intake conducted at a tertiary pain clinic. Participants were recruited through self-referral via links shared on social media and were considered eligible for participation if their pain had lasted for at least 6 months. For inclusion in the study, it was also required that participants 1) be between 18 and 70 years old; 2) be able to understand and communicate in Swedish; 3) have a smartphone or tablet connected to the internet; 4) not have a psychiatric comorbidity that required immediate attention (based on severity rather than specific psychiatric disorders); 5) not be acutely suicidal; 6) be stable on their pain medication for at least 2 months, with no planned changes in medication or other pain treatment; and 7) not have received treatment with CBT or ACT during the previous 3 months.
Individuals interested in participating completed an online registration, provided informed consent, and were thereafter contacted via telephone for a structured clinical interview. The interview consisted of assessments of eligibility, pain-related dysfunction, and psychiatric comorbidities assessed with sections A (Depressive Disorder), B (Suicidal Ideation), D (Panic Disorder), E (Agoraphobia), F (Social Anxiety), G (Obsessive Compulsive Disorder), H (Posttraumatic Stress Disorder), I (Alcoholism), J (Substance-Related Disorders), L (Anorexia), M (Bulimia), and N (Generalized Anxiety Disorder) of version 7 of the Mini-International Neuropsychiatric Interview (MINI) [35, 36] corresponding to the Diagnostic and Statistical Manual of Mental Disorders (DSM–5) [37]. The interviews were conducted by clinical psychologists. If diagnoses or other criteria were ambiguous, participant eligibility was discussed by a multiprofessional team of psychologists, a physician, and a physiotherapist who all specialized in pain. If a participant was considered to be in need of medical care, in need of other psychological treatment that was more relevant, or acutely suicidal, they were contacted by a psychologist or physician for immediate consultation.
Ethics
This study was approved by the regional ethics review board in Stockholm, Sweden, on November 3, 2015 (2015/1638–31/2) and registered at ClinicalTrials.gov on November 17, 2017, under registration number NCT03344926. Participants provided written informed consent prior to enrollment in the study.
ACTsmart Intervention
The treatment was an ACT-based intervention derived from a clinical model used at a tertiary pain clinic [38–41] that was then converted into a smartphone-delivered treatment using a process that followed the guidelines of the mHealth Agile Development & Evaluation Lifecycle [42]. Prior to the present study, ACTsmart underwent rigorous alpha testing, and the present efficacy testing was done using a beta testing sample, who also tested the solution with regard to feasibility (acceptability, usage, practicality), a procedure described in detail elsewhere [43].
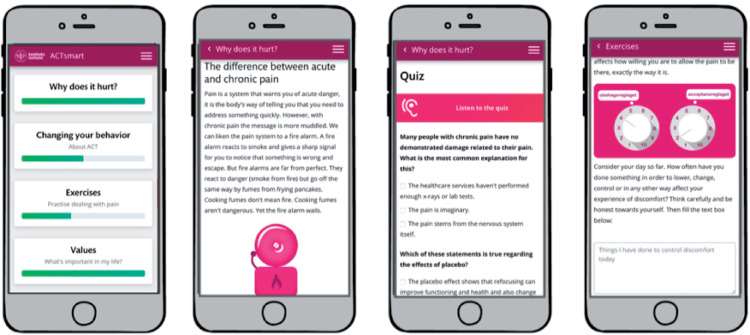
Participants underwent the digital treatment program for 8 weeks and were prompted (in the eligibility interviews and by their therapist during treatment) to work with the treatment content for 20 minutes every weekday. The intervention consisted of four different sections—pain education, self-help texts, exercises, and values—and was delivered as text, audio, pictures, and animations. The treatment target was to decrease avoidant behaviors and to increase value-based behaviors and other psychologically flexible behaviors. Content was divided into six different themes intended to reflect the core processes of psychological flexibility—acceptance, distance to thoughts, distance to emotional and bodily experiences, noticing and changing behaviors, self-observation, and values [44]. All sections except the values section were accessible to the participants at all times, but participants were recommended to start with the educational sections, then move on to exercises and finally work with value formulation and exposure guided by their own values. To obtain access to the values work section, participants had to receive psychoeducation on values and values formulation. All text-based content could be either read or listened to as a means to adapt to participants’ different needs. After completion of an exercise, patients were asked to provide a written reflection that was visible from the therapist interface. An overview of the intervention sections and content is presented in Table 1. Screenshots of the patient interface are shown in Figure 1.
Table 1.
Treatment content
| Section | Content | Primary Skill Taught |
|---|---|---|
| Why does it hurt? | Introduction to pain education, six educational chapters, one knowledge quiz | Knowledge about pain, how the pain system works, categories of pain, potential explanations for chronic pain, how and why the context affects pain experience, information about medication and why they do not always work, proposed focus shift from pain reduction to increased function and quality of life |
| Changing your behavior | Self-help texts, 15 educational chapters tagged with theme, links to related exercises | Introduction to ACT and own change process; divided into six themes |
| Exercises | Twenty-nine ACT-consistent exercises, graded and tagged with theme and with level of difficulty | Divided into six themes |
| Values | Introduction to values; exercises designed to start a thought process on life values; a short test to grade prioritized areas to work with during treatment and formulation of values, goals, and steps | Clarify and formulate life values, value-based exposure |
| Theme | Content | Primary Skill Taught |
| Acceptance | Education, exercises | Acceptance: Increase willingness and ability to let pain and other inner discomfort be, exactly as they are |
| Distance to thoughts | Education, exercises | Defusion: Taking perspective on thoughts, looking at them skeptically and making active choices when deciding if the thoughts are helpful for the current situation |
| Distance to experiences | Education, exercises | Defusion: Taking perspective on unpleasant inner experiences such as pain but also other internal discomfort such as anxiety, fatigue, or fear |
| Your behaviors | Education, exercises | Behavior analysis: Understanding and analyzing own adaptive or maladaptive behavior patterns, behavior in relation to context, antecedents and direct consequences of the behavior, and long-term consequences of maintaining or changing maladaptive behaviors |
| Self-observation | Education, exercises | Contact with present moment, observe in nonjudgmental way, acceptance, defusion |
| Values | Education, exercises | Clarify life values, valued action |
ACT = acceptance and commitment therapy.
Figure 1.
Screenshots of the patient interface in ACTsmart (translated from Swedish to English).
Therapists were instructed to log in to check on each participant every other day. Participants received feedback at least weekly from their therapist via a secure messaging function within the treatment platform. Feedback was also given automatically using checkboxes (for completed exercises), progress bars, thumbs-up (when participants reported taking a step toward a goal), and collection of achievement stars (when participants reported having reached a set goal). If a participant was inactive for 2 days, the therapist sent a chat message in the treatment platform to encourage the participant to engage with the treatment and offered help. If a participant did not log in for 4 days, the therapist sent a mobile text message notification, and if a participant was absent for a week or more, they received a phone call. If a participant did not respond to phone calls or still did not log in for 2 more weeks after contact attempts, the participant was regarded as a dropout.
Four therapists at the tertiary pain clinic participated in the study: one licensed psychologist with extensive knowledge of ACT and three intern psychologists who had had several months of training in ACT and treatment of adult pain patients. The intern psychologists received weekly supervision by a clinician with extensive training in ACT for chronic pain and ACTsmart. Participants were randomly allocated to a therapist by a randomizing algorithm in the digital platform.
Data Collection and Measures
Background variables (age, gender, education), including pain variables (intensity, type, duration, location, pain-related dysfunction, and additional symptoms [i.e., fatigue, stress sensitivity]) were collected at registration as self-report data. Efficacy data were self-reported and collected at pretreatment, posttreatment, and follow-up at 3, 6, and 12 months. Selected variables (pain interference, avoidance, value progress, and value obstruction) were also assessed weekly during treatment. The assessments were provided on a secure internet platform.
Primary Outcome
Pain interference was measured using the Pain Interference Index (PII) [45]. The PII is a six-item questionnaire measuring pain interference in adults in the last 2 weeks. The 6-point Likert scale ranges from “not at all” to “very much,” with lower scores indicating lower interference. In the original validation study [45], the instrument showed a one-factor structure and satisfactory internal consistency (Cronbach’s α = 0.85).
Secondary Outcomes
Psychological inflexibility was measured using the Psychological Inflexibility in Pain Scale (PIPS) [46]. The PIPS assesses self-rated psychological inflexibility related to pain. It is a 12-item questionnaire that consists of two subscales: Avoidance (eight items) and Cognitive Fusion (four items). It is rated on a 7-point Likert scale that ranges from 1 (“never true”) to 7 (“always true”). Higher scores indicate higher levels of psychological inflexibility. The PIPS has shown satisfactory internal consistency, with Cronbach’s α = 0.87 for the total scale, Cronbach’s α = 0.89 for the Avoidance subscale, and Cronbach’s α = 0.66 for the Cognitive Fusion subscale [47, 48].
Values were assessed using the Valuing Questionnaire (VQ) [49]. The VQ is a 10-item questionnaire with two subscales: Progress (VQ-p) and Obstruction (VQ-o). Items are rated on a 7-point Likert scale ranging from 0 (“not at all true”) to 6 (“completely true”). Higher scores on the Progress subscale indicate greater progress toward values, whereas higher scores on the Obstruction subscale indicate greater obstruction to values. The two-factor solution has shown excellent model fit and satisfactory internal consistency (Cronbach’s α = 0.87) for both the Progress and Obstruction subscales [49]. The VQ has been translated into Swedish and validated in a Swedish chronic pain sample with adequate psychometric properties [50].
Insomnia was assessed using the Insomnia Severity Index (ISI) [51]. The ISI is a seven-item questionnaire that evaluates sleep onset, sleep latency, early awakening, the feeling of being rested, how sleep problems affect daily life, and how concerned the individual is with their sleep pattern. The 5-point scale ranges from 0 (“not at all”) to 4 (“very much”), and a higher score indicates more symptoms of insomnia. The ISI has shown satisfactory internal consistency (Cronbach’s α = 0.74) [52] and has been validated in a Swedish chronic pain sample [53].
Depressive symptoms were assessed using the Nine-Item Patient Health Questionnaire (PHQ-9) [54]. Patients rate the frequency of depressive symptoms on a 4-point Likert scale ranging from 0 (“not at all”) to 3 (“almost every day”), where higher scores indicate more symptoms of depression. The PHQ-9 has been found to be reliable and valid with satisfactory internal consistency (Cronbach’s α = 0.86) [54]. The PHQ-9 has been validated for migraine patients [55] as well as chronically ill patients [56].
Anxiety was assessed using the Seven-Item Generalized Anxiety Disorder Scale (GAD-7) [57]. The GAD-7 assesses the frequency of anxiety symptoms during the last 2 weeks on a 4-point Likert scale ranging from 0 (“not at all”) to 3 (“nearly every day”), with higher scores indicating higher levels of distress. The GAD-7 has shown strong internal consistency (Cronbach’s α = 0.92) [57] and is validated for chronic pain patients with migraine [58].
Health-related QoL was assessed using the European Quality of Life Questionnaire (EQ-5D-3L [59, 60]. The EQ-5D-3L has five dimensions—mobility, self-care, usual activities, pain, and anxiety or depression—and three levels: 1) no problems; 2) moderate problems; and 3) severe problems. In this study, items were summarized to a total score without the use of index scores, a procedure proposed by Parkin et al. [61]. As proposed by the EuroQol Group, the scale was reversed, with higher scores indicating better health, and the total score ranged from 5 to 15. The EQ-5D-3L has been validated in chronic pain samples [62], and the Swedish version of the EQ-5D-3L has been validated in several Swedish samples [63–65].
Pain intensity was assessed using a numeric rating scale from 0 (“no pain”) to 10 (“worst imaginable pain”). At baseline, participants were asked to assess their current pain intensity as well as their average pain intensity during the last 2 weeks. At posttreatment and at follow-up, participants only rated their current pain intensity. Analyses of pain intensity were therefore performed using patients’ current pain intensity.
Data Analysis
Preliminary Comparisons and Descriptive Analyses
Statistical analyses were performed using SPSS version 26.0 (IBM SPSS Statistics for Macintosh, version 26.0, Armonk, NY). Figures 3–6 were created using Stata version 15 (StataCorp., College Station, TX). Descriptive statistics were used to calculate participant characteristics at baseline, attrition, and therapist time, as well as weekly means for pain interference, avoidance, value progress, and value obstruction. Independent-sample t tests were conducted for all treatment variables at posttreatment to determine whether treatment results varied with the therapist’s level of training (intern psychologist or licensed psychologist). Frequency distributions were plotted using histograms and Q-Q plots, and tests of normality, skewness, and kurtosis were performed. Outliers were detected using the outlier labeling test as described by Hoaglin et al. [66].
Analyses of Primary and Secondary Outcomes
All outcomes from the included participants were analyzed with an intent-to-treat approach, with full-information maximum-likelihood estimation for model fit using linear mixed-effects models. Prior to running the analyses, the assumptions of the modeling approach were tested, the amount and pattern of missing data were analyzed, and Little’s missing completely at random (MCAR) test was performed to ensure that data were missing at random. Linear mixed-effects models for repeated measures were fit to model longitudinal change. Random effects were included based on their contribution to the model fit. Pretreatment to posttreatment assessments were analyzed for detection of immediate treatment effects, and posttreatment to 12-month follow-up assessments were analyzed for detection of maintained or prolonged treatment effects. This piecewise approach was used to model change with how likely it is to occur in a trial that includes follow-up assessments [67]. For all outcomes, the same model was used for both piece 1 and piece 2. For the weekly assessed variables (avoidance, pain interference, value progress, and value obstruction), 10 time points (pretreatment, eight weekly time points, and posttreatment) were entered to calculate changes from pretreatment to posttreatment, and four time points (at posttreatment and at the 3-, 6-, and 12-month follow-ups) were entered to calculate changes from posttreatment through follow-up. For all other variables, pretreatment to posttreatment calculations consisted of two time points (pretreatment, posttreatment), and posttreatment to follow-up consisted of four time points (at posttreatment and at the 3-, 6-, and 12-month follow-ups). For all variables with weekly assessments during treatment, the model showing the best model fit included the fixed effect of time, random intercepts, and random slopes, where covariance between random effects was constrained to be zero. For all other variables, best model fit was found using the fixed effect of time and the random intercepts.
Effect sizes for the within-group effect were calculated using model-implied estimates of standard deviations and mean differences [68, 69]. Effect sizes were calculated separately for pretreatment to posttreatment and posttreatment to follow-up.
Deterioration
Negative outcomes were evaluated as a feasibility aspect using a deterioration criterion of ≥30% from the participant’s own baseline [70].
Results
Sample Characteristics
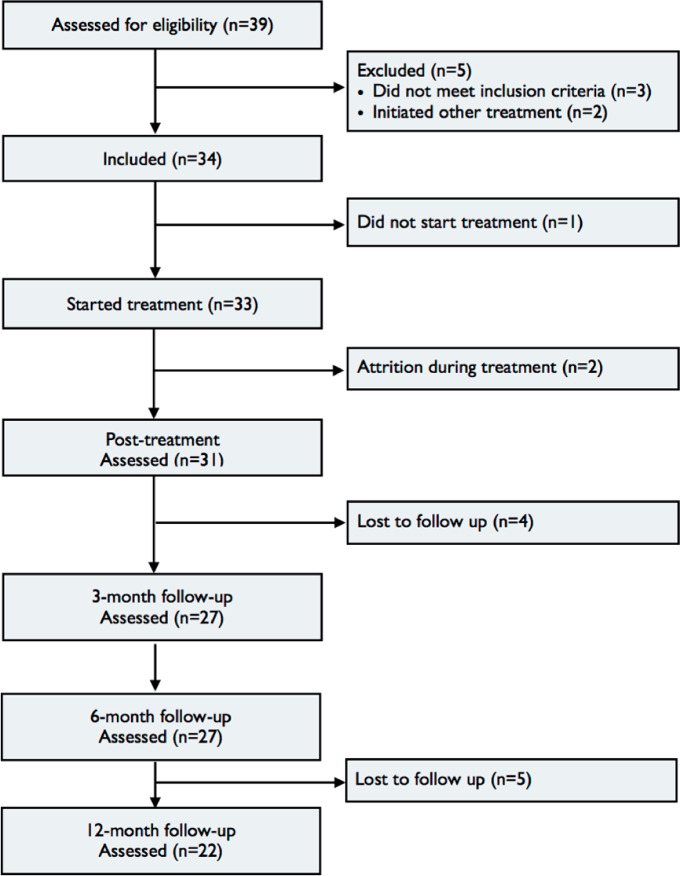
In total, 34 participants were enrolled in the study. One participant (3%) withdrew before the start of treatment, and two participants (6%) never started working with the treatment content and were considered dropouts. Figure 2 presents the flowchart of the study.
Figure 2.
Participant flow throughout treatment.
The sample was predominantly female (88%), with a mean age of 44.4 years (SD = 8.59). The mean pain duration at baseline was 20.4 years (SD = 11.67) with an average of 5.8 pain locations (SD = 2.78). Eighteen participants (53%) fulfilled the criteria for one or more psychiatric diagnoses, with depression being the most common (24%). All participants (100%) suffered one or more additional symptoms in addition to pain; sensitivity to stress (88%) and fatigue (85%) were the most common concerns. Twenty-three (68%) participants were unable to work or study full time due to their pain. Further sample characteristics at baseline are listed in Table 2.
Table 2.
Participant characteristics at baseline
| Participant Characteristic | n=34 |
M (SD) | |
|---|---|---|---|
| Percentage | n | ||
| Sex | |||
| Female | 88% | 30 | — |
| Male | 12% | 4 | — |
| Age | — | — | 44.3 (8.59) |
| Pain duration, y | — | — | 20.4 (11.7) |
| No. of pain locations | — | — | 5.9 (2.8) |
| Education | |||
| Up to 12 y | 41% | 14 | — |
| 12–15 y | 21% | 7 | — |
| 15+y | 38% | 13 | — |
| Occupational status (multiple options possible) | |||
| Working, studying, or on parental leave full time | 32% | 11 | — |
| Working or studying part time | 21% | 7 | — |
| Sick leave, temporary | 29% | 10 | — |
| Sick leave, permanent | 24% | 8 | — |
| Pain diagnosis type | |||
| Nociceptive (spinal disc hernia, rheumatic diseases, endometriosis, whiplash) | 35% | 12 | — |
| Neuropathic (MS, nerve damage) | 3% | 1 | — |
| Nociplastic (fibromyalgia, CRPS) | 29% | 10 | — |
| Headaches (migraine) | 3% | 1 | — |
| Other or unclear | 9% | 3 | — |
| Unspecified diagnosis | 21% | 7 | — |
| Pain medications (multiple options possible) | |||
| Opioids | 47% | 16 | — |
| Antiepileptics | 15% | 5 | — |
| NSAIDs | 29% | 10 | — |
| Antidepressants | 29% | 10 | — |
| Sedatives or relaxants | 9% | 3 | — |
| Other analgesics | 68% | 22 | — |
| Ataractics | 6% | 2 | — |
| Migraine medication | 3% | 1 | — |
| None | 3% | 1 | — |
| No. of medications | — | — | 3.5 (2.5) |
| Additional symptom burden (multiple options possible) | |||
| Recurring fever | 15% | 5 | — |
| Sickness feeling | 50% | 17 | — |
| Fatigue | 85% | 29 | — |
| Concentration difficulties | 77% | 27 | — |
| Memory deficits | 77% | 26 | — |
| Sensitive to stress | 88% | 30 | — |
| Other nonspecified symptom | 21% | 6 | — |
| None | 0% | 0 | — |
| Psychiatric diagnoses (multiple diagnoses possible) | |||
| Depression | 24% | 8 | — |
| Moderately suicidal | 21% | 7 | — |
| Panic disorder | 3% | 1 | — |
| Social phobia | 3% | 1 | — |
| Posttraumatic stress disorder (PTSD) | 6% | 2 | — |
| General anxiety disorder (GAD) | 21% | 7 | — |
| Specific phobia | 3% | 1 | — |
| None | 47% | 16 | — |
SD = standard deviation; MS = multiple sclerosis; CRPS = complex regional pain syndrome; NSAID = nonsteroidal anti-inflammatory drug.
Compliance and Practicality
Of the remaining 31 participants, 28 (90%) completed treatment according to the predefined criteria of completion of at least eight exercises and reported behavior changes toward at least one value. On average, participants completed 84% of the treatment content (SD=17.81; range, 26.5–100), divided over pain education (97%, SD=17.96), self-help texts (93%, SD=21.55), preparations for values work (89%, SD=26.43), and exercises (16.24 of 29; 56%, SD=33.08). Twenty-six participants (84%) formulated values, goals, and steps, and the average number of formulated values was 3.85 (SD=1.99) among those participants who formulated one or more values (3.23 [SD=2.32] in total).
Therapists spent, on average, 16 minutes (range, 2–32) per patient per week and sent, on average, 15.6 (SD = 4.9) chat messages throughout the course of the treatment. Participants sent, on average, 7.1 (SD = 6.4; range, 0–24) chat messages to their therapist during the treatment.
Analysis of Primary and Secondary Outcomes
Missing Data and Assumptions of Linear Mixed-Effects Model
At baseline, there were no missing values. At posttreatment, there was 8.8% missing data in total on all variables. The missing data were due to the three participants who chose to not start or discontinued treatment. Due to how the questionnaire platform worked, it was impossible for participants to refrain from answering single items or questionnaires—either they answered all items on all questionnaires or completely refrained from responding. At the 3- and 6-month follow-ups, 20.6% of the sample did not complete assessments. At the 12-month follow-up, 35.3% of the sample did not complete assessments. Little’s MCAR test [71] showed that data were MCAR (χ2 = 724.170, df = 1,647, P = 1.000). Residuals on all outcome variables were normally distributed.
Treatment Outcome
The means and standard deviations of the pretreatment, posttreatment, and follow-up assessments for all outcome variables are listed in Table 3. The means for the weekly measured variables of pain interference, avoidance, value progress, and value obstruction during treatment are shown in Figures 3–6. The results from linear mixed-effects analyses for primary and secondary outcome measures are listed in Table 4. There were no significant differences on any study variable at posttreatment between participants receiving treatment from an intern psychologist or a licensed psychologist (all P values were greater than 0.05).
Table 3.
Observed means and standard deviations at pretreatment and posttreatment and at 3-, 6-, and 12-month follow-up
| Pretreatment | Posttreatment | Three-Month Follow-Up | Six-Month Follow-Up | Twelve-Month Follow-Up | |
|---|---|---|---|---|---|
| Pain interference | 25.62 (6.03) | 19.03 (8.55) | 17.41 (8.70) | 17.41 (9.56) | 19.18 (9.81) |
| Avoidance | 37.47 (7.84) | 28.87 (9.62) | 28.11 (11.85) | 28.59 (12.66) | 26.82 (10.79) |
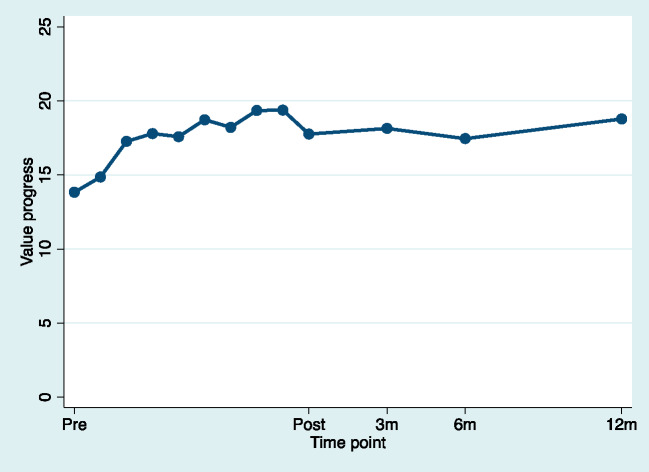
| Value progress | 13.82 (7.80) | 17.71 (6.79) | 18.07 (7.47) | 17.56 (7.39) | 18.86 (7.62) |
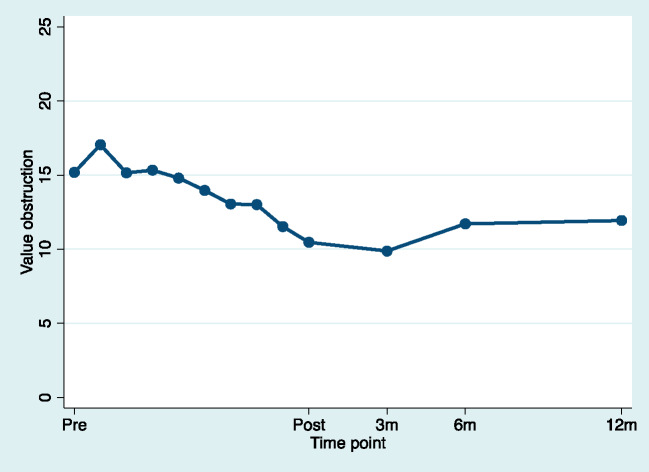
| Value obstruction | 15.18 (7.33) | 10.61 (7.13) | 10.67 (7.81) | 12.44 (8.02) | 12.36 (8.26) |
| Insomnia | 16.42 (6.33) | 14.06 (7.50) | 12.81 (7.73) | 12.35 (7.60) | 13.27 (8.21) |
| Anxiety | 8.68 (5.39) | 6.06 (4.60) | 6.59 (5.43) | 7.54 (6.17) | 7.05 (6.32) |
| Depression | 12.45 (5.56) | 9.55 (6.23) | 9.15 (6.53) | 10.46 (7.62) | 10.64 (7.72) |
| Health-related quality of life | 5.58 (1.77) | 6.10 (1.70) | 6.07 (1.96) | 6.00 (1.86) | 5.82 (1.97) |
| Fusion | 22.09 (4.39) | 18.52 (5.14) | 19.33 (4.91) | 18.15 (5.99) | 17.50 (6.26) |
| Pain intensity | 6.52 (1.55) | 5.35 (2.03) | 4.74 (2.07) | 4.56 (1.91) | 5.18 (1.76) |
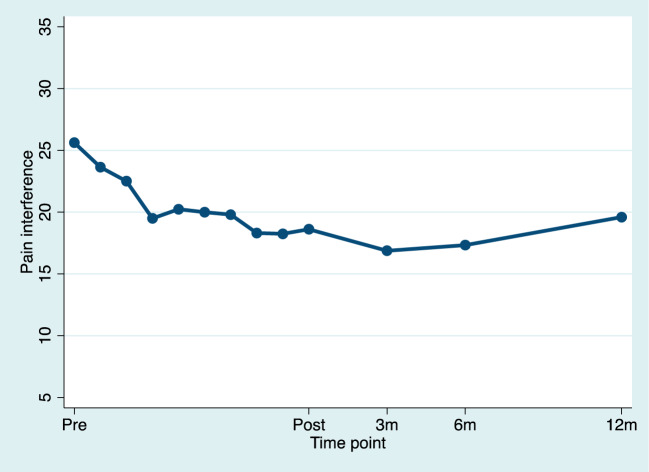
Figure 3.
Estimated means for pain interference for pretreatment, during treatment, and posttreatment (pretreatment to posttreatment d = –1.01). For illustrative purposes, time was used as a discrete variable to model weekly estimates to the mean.
Table 4.
Results from piecewise linear mixed-effects models for primary and secondary outcome variables
| Outcome | Estimate (SE) | 95% CI |
P | d | 95% CI |
||
|---|---|---|---|---|---|---|---|
| Lower Limit | Upper Limit | Lower Limit | Upper Limit | ||||
| Pain interference | |||||||
| Pretreatment to posttreatment | –6.54 (1.18) | –8.94 | –4.15 | 0.000* | –1.01 | 1.38 | –0.64 |
| Posttreatment to 12-mo follow-up | 1.17 (1.36) | –1.62 | 3.97 | 0.395 | 0.19 | –0.26 | 0.63 |
| Avoidance | |||||||
| Pretreatment to posttreatment | –9.81 (1.58) | –13.02 | –6.61 | 0.000* | –1.34 | –1.78 | –0.90 |
| Posttreatment to 12-mo follow-up | 1.56 (1.16) | –3.87 | 0.76 | 0.185 | 0.23 | –0.57 | 0.11 |
| Fusion | |||||||
| Pretreatment to posttreatment | –3.64 (0.70) | –5.06 | –2.22 | 0.000* | –0.67 | –0.93 | –0.41 |
| Posttreatment to 12-mo follow-up | –1.33 (0.69) | –0.05 | 0.04 | 0.057 | –0.27 | –0.01 | 0.01 |
| Value progress | |||||||
| Pretreatment to posttreatment | 4.21 (1.03) | 2.11 | 6.31 | 0.000* | 0.78 | 0.39 | 1.17 |
| Posttreatment to 12-mo follow-up | 0.62 (1.09) | –1.63 | 2.86 | 0.576 | 0.12 | –0.31 | 0.54 |
| Value obstruction | |||||||
| Pretreatment to posttreatment | –5.32 (1.08) | –7.53 | –3.12 | 0.000* | –1.02 | –1.44 | –0.60 |
| Posttreatment to 12-mo follow-up | 2.18 (1.12) | –0.05 | 4.41 | 0.055 | 0.42 | –0.01 | 0.85 |
| Insomnia | |||||||
| Pretreatment to posttreatment | –2.40 (0.77) | –3.97 | –0.82 | 0.004* | –0.47 | –0.78 | –0.16 |
| Posttreatment to 12-mo follow-up | –0.05 (0.82) | –1.69 | 1.59 | 0.955 | –0.01 | –0.36 | 0.34 |
| Anxiety | |||||||
| Pretreatment to posttreatment | –2.57 (0.62) | –3.83 | –1.31 | 0.000* | –0.67 | –1.01 | –0.34 |
| Posttreatment to 12-mo follow-up | 0.85 (0.68) | –0.51 | 2.20 | 0.217 | 0.25 | –0.15 | 0.64 |
| Depressive symptoms | |||||||
| Pretreatment to posttreatment | –2.89 (0.74) | –4.39 | –1.39 | 0.000* | –0.63 | –0.96 | –0.30 |
| Posttreatment to 12-mo follow-up | 1.30 (0.86) | –0.39 | 3.03 | 0.129 | 0.30 | –0.09 | 0.70 |
| Health-related quality of life | |||||||
| Pretreatment to posttreatment | 0.52 (0.19) | 0.90 | 0.13 | 0.010* | 0.16 | 0.28 | 0.04 |
| Posttreatment to 12-mo follow-up | –0.05 (0.07) | –1.57 | –4.55 | 0.444 | –0.02 | –0.51 | –1.48 |
| Pain intensity | |||||||
| Pretreatment to posttreatment | –1.16 (0.30) | –1.76 | –0.56 | 0.000† | –0.44 | –0.66 | –0.21 |
| Posttreatment to 12-mo follow-up | –0.017 (0.31) | –0.63 | 0.60 | 0.955 | –0.01 | –0.25 | 0.24 |
SE = standard error; CI = confidence interval; *= P < 0.05.
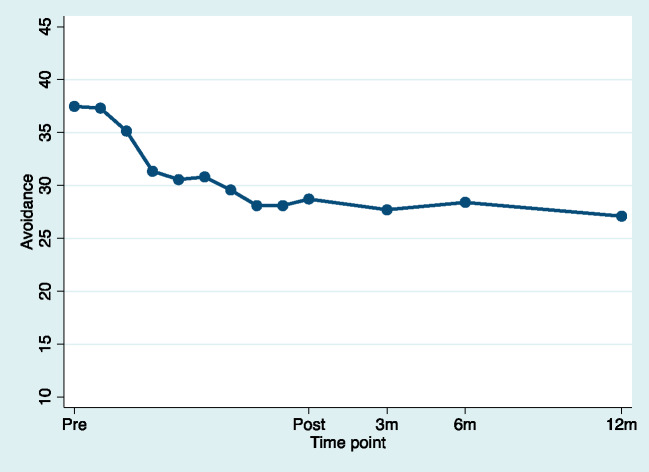
Figure 4.
Estimated means for avoidance for pretreatment, during treatment, and posttreatment (pretreatment to posttreatment d = –1.34). For illustrative purposes, time was used as a discrete variable to model weekly estimates to the mean.
Figure 5.
Estimated means for value progress for pretreatment, during treatment, and posttreatment (pretreatment to posttreatment d = 0.78). For illustrative purposes, time was used as a discrete variable to model weekly estimates to the mean.
Figure 6.
Estimated means for value obstruction for pretreatment, during treatment, and posttreatment (pretreatment to posttreatment d = –1.02). For illustrative purposes, time was used as a discrete variable to model weekly estimates to the mean.
Primary Treatment Outcome
For pain interference, there was a significant linear effect of time from pretreatment to posttreatment, indicating improvement with a large effect size (d = –1.01). There were no significant changes in pain interference in the follow-up phase (posttreatment to 12-month follow-up), illustrating stability in improvement from posttreatment to follow-up.
Secondary Treatment Outcomes
For the secondary treatment outcomes of avoidance, fusion, value progress, value obstruction, insomnia, anxiety, depression, health-related QoL, and pain intensity, improvement was seen in all study variables, illustrated by a significant linear effect of time from pretreatment to posttreatment. There were no changes in any of the variables from posttreatment through the follow-up phase, illustrating stability in improvements. Effect sizes in the secondary outcome variables varied, with health-related QoL presenting the smallest effect size (d = 0.16) and avoidance presenting the largest (d = –1.34) effect size.
Deterioration
No serious adverse events (psychiatric hospitalization, self-harm, suicidal behaviors) were reported during the study. However, a clinically significant deterioration (>30%) was reported by 11 participants (35%) at posttreatment; five participants deteriorated on one variable, four participants deteriorated on two variables, and two participants deteriorated on three variables. Five participants were improved at follow-up and ended up with no change or a minor improvement. For three participants, deterioration took place during treatment and was not improved during follow-up. The variables concerned were value progress, value obstruction, and anxiety. For another three participants, deterioration took place during treatment, and the participants were then lost to follow-up. The variables concerned were pain interference, value progress, value obstruction, and two cases of health-related QoL. Thus, at the end of the follow-up period, deterioration was still present in six participants (19%).
Discussion
This pilot study provides the first evaluation of treatment effects for ACTsmart, a smartphone-delivered ACT treatment for adult patients with chronic pain. There were significant improvements from pretreatment to posttreatment on all variables. Large effect sizes were seen in the primary outcome of pain interference and in the secondary outcomes of avoidance, value obstruction, and anxiety. Significant medium effect sizes were found for the secondary outcomes of value progress, depressive symptoms, fusion, and insomnia; small effect sizes were seen for the secondary outcomes of pain intensity and health-related QoL. All results were maintained for 12 months following the end of treatment.
The preliminary efficacy results of the present study support previous findings on internet-delivered (primarily using a desktop computer) ACT for chronic pain patients [22–26, 72], but our results also extend these findings, with promising effects on insomnia, values, and QoL. Sleep disorders are among the most common concerns related to pain [73]. Sleep-related variables have been shown to predict pain-related outcomes such as disability [74], and sleep disruption is related to increased pain sensitivity and lowered pain thresholds [75]. Randomized controlled studies concerning internet-delivered ACT for chronic pain have so far either failed to show improvements in insomnia [26] or have not used it as a measure of outcome, which implies that the moderate improvement shown in this study is promising and warrants future studies.
The obtained results are also comparable with those of clinical studies of face-to-face interventions of ACT for adults with chronic pain in which significant small to medium effect sizes have been found for pain, depression, anxiety, physical well-being, and QoL [9, 11, 15]. This is promising, as it provides the opportunity to offer a treatment with similar effects as face-to-face treatment with less therapist time spent per patient. This can increase availability, reach, and accessibility, as each therapist can treat more patients and patients can access treatment regardless if they live far away from a treatment facility, have mobility issues, or have difficulty getting away from work. Another interesting research objective would be to examine the combination of face-to-face and smartphone-delivered treatment (e.g., in a blended care approach). The addition of smartphone support reminding patients to practice daily and facilitating therapist support between sessions could potentially increase compliance and enhance treatment effects.
As previously described, the treatment target in the present study was to decrease avoidance behaviors, increase psychological flexibility, increase value-based behavior, and thereby decrease pain interference. Future studies should expand on the theoretical and empirically supported notion that psychological flexibility may function as a vehicle for changes in the other outcomes.
The aim of the intervention in the present study was not explicitly to reduce pain intensity. A small but significant improvement in pain intensity was, however, seen at posttreatment and remained at the 12-month follow-up. Although small, the improvement is encouraging, especially as the study group showed a mean pain duration of >20 years. As encouraging as it is that the present study showed significant effects despite the long pain duration, several studies instead point to the importance of early interventions in order to prevent the pain from becoming chronic [76, 77]. Smartphone-delivered treatment may be a way to enable early intervention, with its promise of increased availability, reach, and accessibility.
In comparison with previous research on internet-delivered (via a desktop computer) ACT treatment for chronic pain patients [20, 23–25], the compliance rate in the present study was high (90%). This might, of course, be due to differences in requirements for completion and adherence, but this might also be due to differences in the design and structure of the intervention. In the present study, the requirements for treatment completion were completion of at least eight exercises or formulation of and reported behavior changes toward at least one value. By fulfilling these requirements, a participant was considered to have undergone a sufficient proportion of active treatment. In the study conducted by Trompetter et al. [23], completion and sufficient dose of active treatment required the participants to complete the first six modules of the intervention, and adherence was defined as both completing the intervention and engaging with the treatment for at least 3 hours per week. As attention, sitting still, and processing large amounts of information are tasks that are known to be difficult for chronic pain patients [3, 4, 78], perhaps the microinteractions possible with smartphone-delivered treatment better suit the needs of the chronic pain population. In addition, the present intervention underwent rigorous alpha testing focused on user friendliness and comprehensibility based on user feedback before this study started.
The therapists in the present study were all trained in ACT and chronic pain, which may have influenced the magnitude of the effects observed. Further efficacy testing is needed with less experienced therapists or with therapists who do not specialize in chronic pain, such as primary care clinicians. The structure and content of ACTsmart (and many other internet-delivered treatments) limit the risk of therapist drift, but the level of training that is required remains an empirical question. Moreover, there is a need to further investigate dose-response relationships (e.g., varying levels of therapist support, varying treatment lengths, and whether the treatment is delivered as a stand-alone treatment or in combination with face-to-face treatment).
Limitations to this pilot study that should be considered when interpreting the results include the lack of a control condition, the small sample size, the predominance of female participants, and the proportion of missing data at the 12-month follow-up. Also, the sample is self-referred, which may reduce the external validity of the findings, as self-referred patients may possibly be more healthy, better educated, and more motivated than clinical patients. However, baseline characteristics in the present study such as pain intensity and pain duration correspond with those of previous research using participants recruited from pain clinics [38, 45, 48]. The sample is also representative of the chronic pain population with regard to psychiatric comorbidities [22] as well as additional symptom burden. Furthermore, a recent direct comparison between clinical and self-referred pain patients showed similar effects and similar degrees of feasibility for both groups when receiving a therapist-guided ACT protocol through the internet [79].
The promising results of the present study warrant further research, primarily randomized controlled trials with larger sample sizes, but also studies on cost-effectiveness and dose-response relationships, as well as implementation studies in various care settings with varying levels of clinical expertise. Also, as some participants deteriorated during treatment, a high-priority research objective is to find models to predict who will likely be a treatment responder or nonresponder.
Conclusion
In conclusion, the present study provides preliminary support for the usefulness of ACTsmart as a digital behavioral intervention to improve functioning in individuals with chronic pain. Larger clinical trials with an active control condition and studies using patients recruited from tertiary care pain units are warranted to validate these findings.
Acknowledgments
We would like to thank psychologists Aleksi Bell, Marie Lilja, and Andreas Fällman for conducting eligibility interviews and for treatment support, as well as Dr. Jan Persson for his medical expertise in eligibility discussions and for categorizing medication and diagnoses. We would also like to thank the team at the Karolinska Institutet University Library: Sofia Samuelsson, Mats Ronquist, Mikael Jergefeldt, Åsa Jenslin, David Hansson, Fredrik Persson, and Erik Svallingson. Their dedication, knowledge, and curiosity made ACTsmart possible. In addition, we are grateful to Brjánn Ljótsson for generously allowing us to use his digital platform for baseline data collection.
Conflicts of interest: The authors have no conflicts of interest to declare.
References
- 1. Reid KJ, Harker J, Bala MM, et al. Epidemiology of chronic non-cancer pain in Europe: Narrative review of prevalence, pain treatments and pain impact. Curr Med Res Opin 2011;27(2):449–62. [DOI] [PubMed] [Google Scholar]
- 2. Breivik H, Eisenberg E, O’Brien T. The individual and societal burden of chronic pain in Europe: The case for strategic prioritisation and action to improve knowledge and availability of appropriate care. BMC Public Health 2013;13(1):1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Breivik H, Collett B, Ventafridda V, Cohen R, Gallacher D. Survey of chronic pain in Europe: Prevalence, impact on daily life, and treatment. Eur J Pain 2006;10(4):287–333. [DOI] [PubMed] [Google Scholar]
- 4. Tunks ER, Crook J, Weir R. Epidemiology of Chronic Pain with Psychological Comorbidity: Prevalence, Risk, Course, and Prognosis. Can J Psychiatry 2008; 53(4):224–234. [DOI] [PubMed] [Google Scholar]
- 5. Vlaeyen WSJ, Morley WSS. Cognitive-behavioral treatments for chronic pain: What works for whom? Clin J Pain 2005;21(1):1–8. [DOI] [PubMed] [Google Scholar]
- 6. Gatchel RJ, Peng YB, Peters ML, Fuchs PN, Turk DC. The biopsychosocial approach to chronic pain: Scientific advances and future directions. Psychol Bull 2007;133(4):581–624. [DOI] [PubMed] [Google Scholar]
- 7. Turk DC, Wilson HD, Cahana A. Treatment of chronic non-cancer pain. Lancet 2011;377(9784):2226–35. [DOI] [PubMed] [Google Scholar]
- 8. Feliu-Soler A, Montesinos F, Gutiérrez-Martínez O, Scott W, McCracken LM, Luciano JV. Current status of acceptance and commitment therapy for chronic pain: A narrative review. J Pain Res 2018;11:2145–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Hann KEJ, McCracken LM. A systematic review of randomized controlled trials of Acceptance and Commitment Therapy for adults with chronic pain: Outcome domains, design quality, and efficacy. J Context Behav Sci 2014;3(4):217–27. [Google Scholar]
- 10. Hughes SL, Clark AJ, Colclough AJ, Dale AE, McMillan AD. Acceptance and Commitment Therapy (ACT) for chronic pain: A systematic review and meta-analyses. Clin J Pain 2017;33(6):552–68. [DOI] [PubMed] [Google Scholar]
- 11. Veehof MM, Trompetter HR, Bohlmeijer ET, Schreurs KMG. Acceptance- and mindfulness-based interventions for the treatment of chronic pain: A meta-analytic review. Cogn Behav Ther 2016;45(1):5–31. [DOI] [PubMed] [Google Scholar]
- 12. Hayes SC, Wilson KG, Gifford EV, Follette VM, Strosahl K. Experiential avoidance and behavioral disorders: A functional dimensional approach to diagnosis and treatment. J Consult Clin Psychol 1996;64(6):1152–68. [DOI] [PubMed] [Google Scholar]
- 13. Goubert L, Crombez G, Eccleston C, Devulder J. Distraction from chronic pain during a pain-inducing activity is associated with greater post-activity pain. Pain (Amsterdam) 2004;110(1–2):220–7. [DOI] [PubMed] [Google Scholar]
- 14. Masedo AI, Rosa Esteve M. Effects of suppression, acceptance and spontaneous coping on pain tolerance, pain intensity and distress. Behav Res Ther 2007;45(2):199–209. [DOI] [PubMed] [Google Scholar]
- 15. Veehof MM, Oskam M-J, Schreurs KMG, Bohlmeijer ET. Acceptance-based interventions for the treatment of chronic pain: A systematic review and meta-analysis. Pain 2011;152(3):533–42. [DOI] [PubMed] [Google Scholar]
- 16. Hogg MN, Gibson S, Helou A, DeGabriele J, Farrell MJ. Waiting in pain: A systematic investigation into the provision of persistent pain services in Australia. Med J Aust 2012;196(6):386–90. [DOI] [PubMed] [Google Scholar]
- 17. Keogh E, Rosser BA, Eccleston C. e-Health and chronic pain management: Current status and developments. Pain 2010;151(1):18–21. [DOI] [PubMed] [Google Scholar]
- 18. Macea DD, Gajos K, Daglia Calil YA, Fregni F. The efficacy of web-based cognitive behavioral interventions for chronic pain: A systematic review and meta-analysis. J Pain 2010;11(10):917–29. [DOI] [PubMed] [Google Scholar]
- 19. Bender JL, Radhakrishnan A, Diorio C, Englesakis M, Jadad AR. Can pain be managed through the Internet? A systematic review of randomized controlled trials. Pain 2011;152(8):1740–50. [DOI] [PubMed] [Google Scholar]
- 20. Buhrman M, Gordh T, Andersson G. Internet interventions for chronic pain including headache: A systematic review. Internet Interv 2016;4:17–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Eccleston C, Fisher E, Craig L, Duggan GB, Rosser BA, Keogh E. Psychological therapies (Internet-delivered) for the management of chronic pain in adults. Cochrane Database Syst Rev 2014;2020(10):CD010152–CD010152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Buhrman M, Skoglund A, Husell J, et al. Guided internet-delivered acceptance and commitment therapy for chronic pain patients: A randomized controlled trial. Behav Res Ther 2013;51(6):307–15. [DOI] [PubMed] [Google Scholar]
- 23. Trompetter H, Bohlmeijer E, Veehof M, Schreurs K. Internet-based guided self-help intervention for chronic pain based on Acceptance and Commitment Therapy: A randomized controlled trial. J Behav Med 2015;38(1):66–80. [DOI] [PubMed] [Google Scholar]
- 24. Lin J, Paganini S, Sander L, et al. An internet-based intervention for chronic pain. Dtsch Arztebl Int 2017;114(41):681–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Scott W, Chilcot J, Guildford B, Daly-Eichenhardt A, McCracken LM. Feasibility randomized-controlled trial of online Acceptance and Commitment Therapy for patients with complex chronic pain in the United Kingdom. Eur J Pain 2018;22(8):1473–84. [DOI] [PubMed] [Google Scholar]
- 26. Simister HD, Tkachuk GA, Shay BL, Vincent N, Pear JJ, Skrabek RQ. Randomized controlled trial of online acceptance and commitment therapy for fibromyalgia. J Pain 2018;19(7):741–53. [DOI] [PubMed] [Google Scholar]
- 27. Herbert MS, Afari N, Liu L, et al. Telehealth versus in-person acceptance and commitment therapy for chronic pain: A randomized noninferiority trial . J Pain 2017;18(2):200–11. [DOI] [PubMed] [Google Scholar]
- 28. Boschen MJ, Casey LM. The use of mobile telephones as adjuncts to cognitive behavioral psychotherapy. Prof Psychol Res Pract 2008;39(5):546–52. [Google Scholar]
- 29. Clement J. Mobile internet usage worldwide - Statistics & Facts. 2019. Available at: https://www.statista.com/topics/779/mobile-internet/. accessed February 2020.
- 30. Andersson G, Titov N. Advantages and limitations of Internet‐based interventions for common mental disorders. World Psychiatry 2014;13(1):4–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Lindner P, Ivanova E, Ly KH, Andersson G, Carlbring P. Guided and unguided CBT for social anxiety disorder and/or panic disorder via the Internet and a smartphone application: Study protocol for a randomised controlled trial. Trials 2013;14(1):437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Lalloo AC, Jibb NL, Rivera NJ, Agarwal NA, Stinson NJ. “There’s a pain app for that”: Review of patient-targeted smartphone applications for pain management. Clin J Pain 2015;31(6):557–63. [DOI] [PubMed] [Google Scholar]
- 33. Rosser BA, Eccleston C. Smartphone applications for pain management. J Telemed Telecare 2011;17(6):308–12. [DOI] [PubMed] [Google Scholar]
- 34. Reynoldson C, Stones C, Allsop M, et al. Assessing the quality and usability of smartphone apps for pain self-management. Pain Med 2014;15(6):898–909. [DOI] [PubMed] [Google Scholar]
- 35. Sheehan DV, Lecrubier Y, Harnett Sheehan K, et al. The validity of the Mini International Neuropsychiatric Interview (MINI) according to the SCID-P and its reliability. Eur Psychiatry 1997;12(5):232–41. [Google Scholar]
- 36. Sheehan DV, Lecrubier Y, Sheehan KH, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I): The development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 1998;59(suppl 20):22–33;quiz 34–57. [PubMed] [Google Scholar]
- 37.American Psychiatric Association. Diagnostic and statistical manual of mental disorders (5th ed.). Washington, DC: Author; 2013. [Google Scholar]
- 38. Wicksell RK, Ahlqvist J, Bring A, Melin L, Olsson GL. Can exposure and acceptance strategies improve functioning and life satisfaction in people with chronic pain and whiplash-associated disorders (WAD)? A randomized controlled trial. Cogn Behav Ther 2008;37(3):169–82. [DOI] [PubMed] [Google Scholar]
- 39. Wicksell RK, Melin L, Lekander M, Olsson GL. Evaluating the effectiveness of exposure and acceptance strategies to improve functioning and quality of life in longstanding pediatric pain—A randomized controlled trial. Pain 2009;141(3):248–57. [DOI] [PubMed] [Google Scholar]
- 40. Kemani KM, Olsson LG, Lekander KM, Hesser KH, Andersson KE, Wicksell KR. Efficacy and cost-effectiveness of acceptance and commitment therapy and applied relaxation for longstanding pain: A randomized controlled trial. Clin J Pain 2015;31(11):1004–16. [DOI] [PubMed] [Google Scholar]
- 41. Wicksell RK, Kemani M, Jensen K, et al. Acceptance and commitment therapy for fibromyalgia: A randomized controlled trial. Eur J Pain 2013;17(4):599–611. [DOI] [PubMed] [Google Scholar]
- 42. Wilson K, Bell C, Wilson L, Witteman H. Agile research to complement agile development: A proposal for an mHealth research lifecycle. NPJ Digit Med 2018;1(1 ):46–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Gentili C, Zetterqvist V, Rickardsson J, Holmström L, Simons LE, Wicksell RK. ACTsmart – Development and feasibility of digital Acceptance and Commitment Therapy for adults with chronic pain. NPJ Digit Med 2020;3(1):20–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Hayes SC, Luoma JB, Bond FW, Masuda A, Lillis J. Acceptance and commitment therapy: Model, processes and outcomes. Behav Res Ther 2006;44(1):1–25. [DOI] [PubMed] [Google Scholar]
- 45. Kemani MK, Zetterqvist V, Kanstrup M, Holmström L, Wicksell RK. A validation of the pain interference index in adults with long‐standing pain. Acta Anaesthesiol Scand 2016;60(2):250–8. [DOI] [PubMed] [Google Scholar]
- 46. Wicksell RK, Renöfält J, Olsson GL, Bond FW, Melin L. Avoidance and cognitive fusion—Central components in pain related disability? Development and preliminary validation of the Psychological Inflexibility in Pain Scale (PIPS). Eur J Pain 2008;12(4):491–500. [DOI] [PubMed] [Google Scholar]
- 47. Wicksell RK, Lekander M, Sorjonen K, Olsson GL. The Psychological Inflexibility in Pain Scale (PIPS)—Statistical properties and model fit of an instrument to assess change processes in pain related disability. Eur J Pain 2010;14(7):771.e1–14. [DOI] [PubMed] [Google Scholar]
- 48. Wicksell RK, Olsson GL, Hayes SC. Psychological flexibility as a mediator of improvement in Acceptance and Commitment Therapy for patients with chronic pain following whiplash. Eur J Pain 2010;14(10):1059.e1–11. [DOI] [PubMed] [Google Scholar]
- 49. Smout M, Davies M, Burns N, Christie A. Development of the Valuing Questionnaire (VQ). J Context Behav Sci 2014;3(3):164–72. [Google Scholar]
- 50. Rickardsson J, Zetterqvist V, Kemani M, Holmström L, Andersson E, Wicksell KR. Assessing values – Psychometric properties of the Swedish version of the Valuing Questionnaire in adults with chronic pain. J Contextual Behav Sci 2019;14:40–49. [Google Scholar]
- 51. Morin CM. Insomnia: Psychological Assessment and Management. New York, NY: Guilford Press; 1993. [Google Scholar]
- 52. Bastien CH, Vallières A, Morin CM. Validation of the Insomnia Severity Index as an outcome measure for insomnia research. Sleep Med 2001;2(4):297–307. [DOI] [PubMed] [Google Scholar]
- 53. Dragioti E, Wiklund T, Alföldi P, Gerdle B. The Swedish version of the Insomnia Severity Index: Factor structure analysis and psychometric properties in chronic pain patients. Scand J Pain 2015;9(1):22–7. [DOI] [PubMed] [Google Scholar]
- 54. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: Validity of a brief depression severity measure. J Gen Intern Med 2001;16(9):606–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Seo J-G, Park S-P. Validation of the Patient Health Questionnaire-9 (PHQ-9) and PHQ-2 in patients with migraine. J Headache Pain 2015;16(1):1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Wu SF Rapid screening of psychological well-being of patients with chronic illness: Reliability and validity test on WHO-5 and PHQ-9 scales. Depress Res Treat 2014;2014:239490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Spitzer RL, Kroenke K, Williams JBW, Löwe B. A brief measure for assessing generalized anxiety disorder: The GAD-7. Arch Intern Med 2006;166(10):1092–7. [DOI] [PubMed] [Google Scholar]
- 58. Seo J-G, Park S-P. Validation of the Generalized Anxiety Disorder-7 (GAD-7) and GAD-2 in patients with migraine. J Headache Pain 2015;16(1):1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.EuroQol Group. EuroQol—A new facility for the measurement of health-related quality of life. Health Policy 1990;16(3):199–208. [DOI] [PubMed] [Google Scholar]
- 60. Rabin R, Charro F. EQ-5D: A measure of health status from the EuroQol Group. Ann Med 2001;33(5):337–43. [DOI] [PubMed] [Google Scholar]
- 61. Parkin D, Rice N, Devlin N. Statistical analysis of EQ-5D profiles: Does the use of value sets bias inference? Med Decis Making 2010;30(5):556–65. [DOI] [PubMed] [Google Scholar]
- 62. Obradovic M, Lal A, Liedgens H. Validity and responsiveness of EuroQol-5 dimension (EQ-5D) versus Short Form-6 dimension (SF-6D) questionnaire in chronic pain. Health Qual Life Outcomes 2013;11(1):110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Brooks R. EuroQol: The current state of play. Health Policy 1996;37(1):53–72. [DOI] [PubMed] [Google Scholar]
- 64. Burström K, Johannesson M, Diderichsen F. Health-related quality of life by disease and socio-economic group in the general population in Sweden. Health Policy 2001;55(1):51–69. [DOI] [PubMed] [Google Scholar]
- 65. Burström K, Johannesson M, Diderichsen F. Swedish population health-related quality of life results using the EQ-5D. Qual Life Res 2001;10(7):621–35. [DOI] [PubMed] [Google Scholar]
- 66. Hoaglin DC, Iglewicz B, Tukey JW. Performance of some resistant rules for outlier labeling. J Am Stat Assoc 1986;81(396):991–9. [Google Scholar]
- 67. Hesser H. Modeling individual differences in randomized experiments using growth models: Recommendations for design, statistical analysis and reporting of results of internet interventions. Internet Interv 2015;2(2):110–20. [Google Scholar]
- 68. Feingold A. Effect sizes for growth-modeling analysis for controlled clinical trials in the same metric as for classical analysis. Psychol Methods 2009;14(1):43–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Feingold A. Confidence interval estimation for standardized effect sizes in multilevel and latent growth modeling. J Consult Clin Psychol 2015;83(1):157–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Ostelo WJGR, Deyo AR, Stratford MP, et al. Interpreting change scores for pain and functional status in low back pain: Towards international consensus regarding minimal important change. Spine 2008;33(1):90–4. [DOI] [PubMed] [Google Scholar]
- 71. Little RJA. A test of missing completely at random for multivariate data with missing values. J Am Stat Assoc 1988;83(404):1198–202. [Google Scholar]
- 72. Kristjánsdóttir OB, Fors EA, Eide E, et al. A smartphone-based intervention with diaries and therapist-feedback to reduce catastrophizing and increase functioning in women with chronic widespread pain: Randomized controlled trial. J Med Internet Res 2013;15(1):e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Smith MT, Haythornthwaite JA. How do sleep disturbance and chronic pain inter-relate? Insights from the longitudinal and cognitive-behavioral clinical trials literature. Sleep Med Rev 2004;8(2):119–32. [DOI] [PubMed] [Google Scholar]
- 74. McCracken LM, Iverson GL. Disrupted sleep patterns and daily functioning in patients with chronic pain. Pain Res Manag 2002;7(2):75–9. [DOI] [PubMed] [Google Scholar]
- 75. Staffe AT, Bech MW, Clemmensen SLK, Nielsen HT, Larsen DB, Petersen KK. Total sleep deprivation increases pain sensitivity, impairs conditioned pain modulation and facilitates temporal summation of pain in healthy participants. PLoS One 2019;14(12):e0225849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Kosek E, Rosen A, Carville S, et al. Lower placebo responses after long-term exposure to fibromyalgia pain. J Pain 2017;18(7):835–43. [DOI] [PubMed] [Google Scholar]
- 77. Martín J, Torre F, Aguirre U, Padierna A, Matellanes B, Quintana JM. Assessment of predictors of the impact of fibromyalgia on health-related quality of life 12 months after the end of an interdisciplinary treatment. J Affect Disord 2017;208:76–81. [DOI] [PubMed] [Google Scholar]
- 78. Eccleston C, Crombez G. Pain demands attention: A cognitive-affective model of the interruptive function of pain. Psychol Bull 1999;125(3):356–66. [DOI] [PubMed] [Google Scholar]
- 79. Rickardsson J, Zetterqvist V, Gentili C, et al. Internet-delivered acceptance and commitment therapy (iACT) for chronic pain—Feasibility and preliminary effects in clinical and self-referred patients. mHealth 2020;6:27. [DOI] [PMC free article] [PubMed] [Google Scholar]