Abstract
Background/Setting A subject presenting with a unilateral sensorineural hearing loss and with vertigo/imbalance and a lesion of internal acoustic meatus (IAM) most often represents a vestibular schwannoma. Several alternative pathologies involving the region, with clinical and neuroradiological similarities, could lead to an error in judgement and management. Rare tumors of the IAM pose unique diagnostic difficulty. A rare case that we present here had a typical history and imaging findings suggestive of vestibular schwannoma. A primary central nervous system (CNS) lymphoma was diagnosed in later stages of brain involvement warranting a retrospective analysis of the entity.
Case Summary An 80-year-old male presented with unilateral sensorineural hearing loss, vertigo, and imbalance. On imaging, he was found to have a lesion in the left internal auditory meatus, reported as a vestibular schwannoma and operated upon. Subject's condition worsened with time and a repeat imaging was suggestive of a CNS lymphoma with lesions involving bilateral cerebellum and subcortical white matrix.
Conclusion To conclude, primary CNS lymphoma presenting an isolated lesion in the IAM with no other parenchymal lesions at presentation is a rare incidence; to our knowledge this is the first case of such unique presentation.
Keywords: vestibular schwannoma, internal acoustic meatus, CNS lymphoma, imaging of internal acoustic meatus lesions
Introduction
A subject presenting to the outpatient department with unilateral hearing loss, disequilibrium/giddiness, tinnitus, and a space-occupying lesion) in the internal acoustic meatus (IAM) on imaging usually diverts the clinicians toward suspecting a vestibular schwannoma (VS). Vestibular schwannoma represents 6 to 7% of all intracranial tumor entity with incidence of 1 in 100000 1 2 3 4 5 with nonschwannoma tumors being seldom diagnosed (accounting only 10% of lesions). 6 7 8
We, through this write up, aim at discussing pathologies that mimic VS clinically and neuroradiologically leading to an error in judgement and management. Rare tumors of the IAC include lipomas, hemangiomas, meningiomas, neurofibromas, hamartomas, and arachnoidal cysts with each posing unique diagnostic difficulties. 7 A rare case that we present here had a classical history and imaging, convincing us of a VS, with a primary central nervous system (CNS) lymphoma being diagnosed in later stages of brain involvement warranting a retrospective analysis of the entity.
Case Report
An 80-year-old male presented to our outpatient department with a history of progressively worsening hearing loss on the left ear, with vertigo and imbalance during walking, of 2 months duration. The symptoms were unnerving and hampering his daily activities. Pure tone audiometry was performed and indicated a severe-to-profound left-sided sensorineural hearing loss with preserved hearing on the right ear. Magnetic resonance imaging (MRI) scan with gadolinium was performed that showed a 1.5 × 0.8 × 0.5 cm lesion located in the intracanalicular portion of the left internal auditory meatus, isointense on T1 and hypointense on T2, with heterogeneity suggestive of a VS ( Fig. 1A and B ). High-resolution computed tomography (HRCT) temporal bone done preoperatively demonstrated a mild widening of the left IAM. In the same scan, there were small discoid enhancing lesions in both cerebellar hemispheres that were hyperintense on T2 and isointense on T1 sequences. They were considered as ischemic foci; a MR spectroscopy and perfusion was done that appeared hypoperfused on 3D arterial spin labeling (noncontrast perfusion) and dynamic susceptibility contrast perfusion and showed normal spectral pattern, suggestive of ischemic foci ( Fig. 1C ).
Fig. 1.
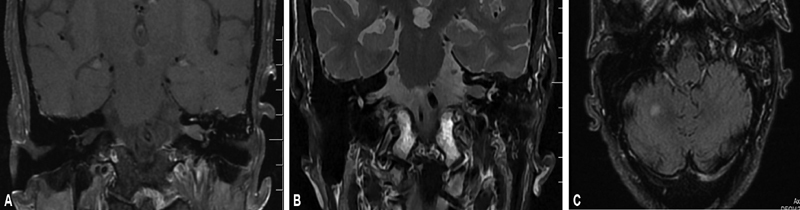
( A , B ) Coronal T1 postcontrast and coronal T2 images showing moderately enhancing lesion in the left internal auditory canal. ( C ) Small enhancing lesion in the right cerebral hemisphere. The lesion is fluid-attenuated inversion recovery hyperintense and hypoperfused.
Having, thus, obtained radiological confirmation of VS and a characteristic history supporting the diagnosis led us to suggest surgery to the subject due to his incapacitating symptomatology. A translabyrinthine approach was performed since there was profound hearing loss on the left ear. On opening the IAM, surprisingly, no lesion was detected within the meatus. The dura of the IAM was incised open and sent for histopathological examination that subsequently was reported as inflammatory. Meanwhile, the patient's complaints of vertigo and imbalance worsened with repeated falling that prompted a repeat MRI scanning of the brain within 3 weeks wherein new findings of fairly large, ill-defined lesions with altered signal intensity were identified in bilateral cerebellar hemispheres and left anterior subcortical white matter, later involving the left thalamus ( Fig. 2A and B ). A navigation biopsy of the lesion, later, suggested a non-Hodgkin's lymphoma. Serial scans of the subject through 1-year follow-up are as depicted in Fig. 3A–D .
Fig. 2.
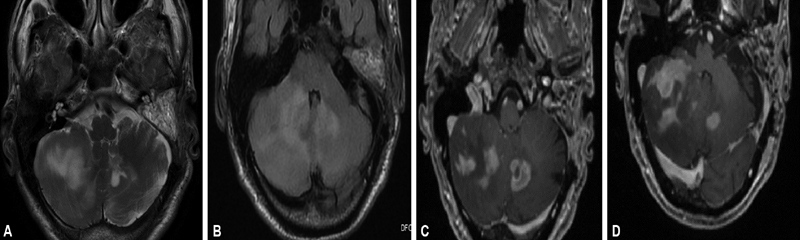
( A ) Axial T2-weighted imaging showing patchy area of altered signal intensity scattered in bilateral cerebellar hemisphere. ( B ) Axial fluid-attenuated inversion recovery images showing area of hyperintensities in cerebellar hemisphere. ( C , D ) On axial T1-weighted images, these areas of altered signal intensity show moderate heterogenous enhancement.
Fig. 3.
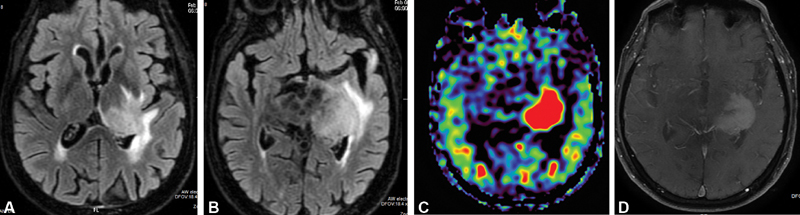
( A , B ) Axial fluid-attenuated inversion recovery images showing area of altered signal intensity in left thalamus and basal ganglia region. ( C ) Axial arterial spin labeling image showing hyperperfusion in these regions of altered signal intensity. ( D ) On axial T1-weighted images, these areas of altered signal intensity show moderate heterogenous enhancement.
A positron emission tomography scan was done that negated any other system involvement and a diagnosis of primary CNS lymphoma was made ( Fig. 4 ). Patient underwent chemotherapy for the same. Warranting a retrospective analysis was the fact that the patient presented with a history and imaging mimicking VS, adding much to clinician's dilemma.
Fig. 4.
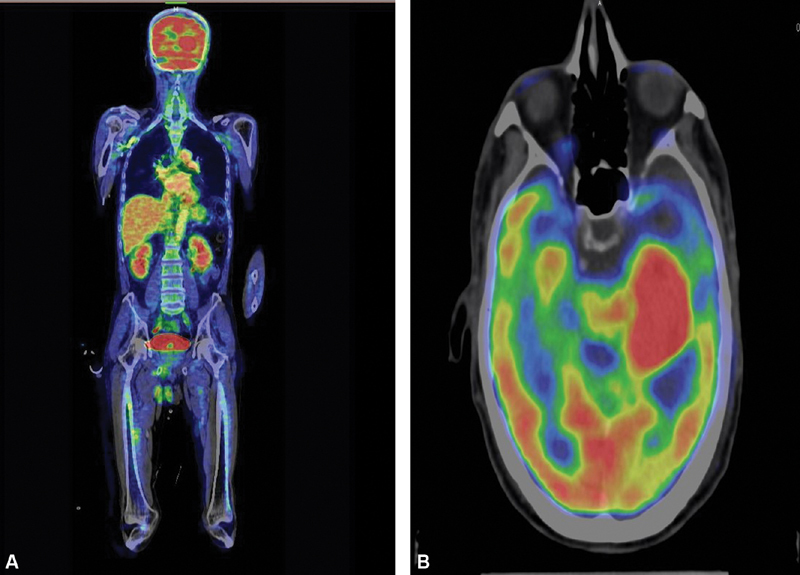
( A , B ) Whole body positron emission tomography computed tomography image showing no area of abnormal metabolic activity apart from focal hyperperfusion seen in the thalamocapsular region.
Diffusion-weighted imaging (DWI) sequence is a part of all our standard MRI protocols and was performed in this case as well. No area of altered diffusivity was identified on DWI images in this particular patient ( Fig. 5A, B ).
Fig. 5.
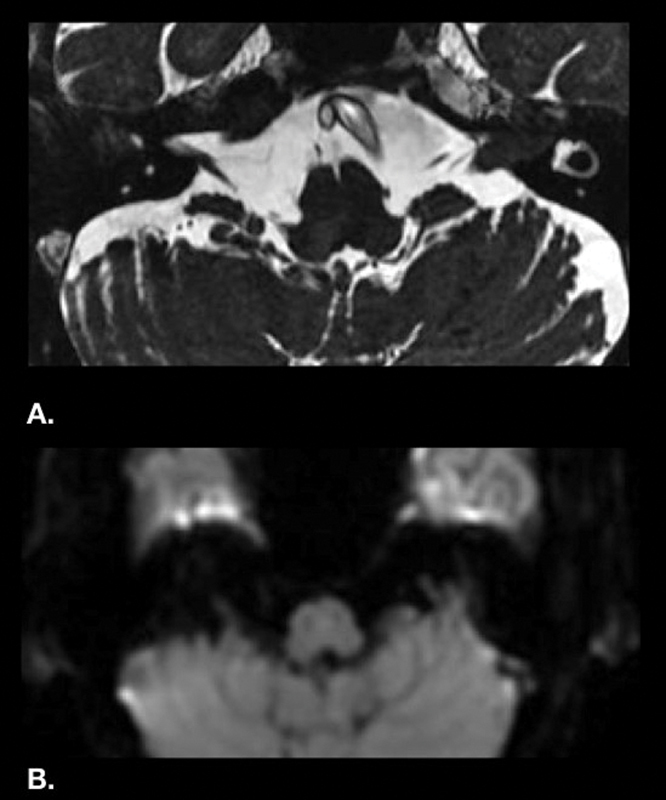
( A ) Axial FIESTA magnetic resonance imaging showing nodular T2 hypointense lesion in left cerebellopontine angle cistern extending into the internal acoustic meatus. ( B ) Diffusion-weighted imaging (B = 1,000) does not show any altered diffusivity or restricted diffusion.
Discussion
In spite of a thorough evaluation by our renowned neuroradiological team and an elaborate discussion of the nature of lesion, a misdiagnosis could not be averted in our case necessitating consideration of fallacies of standard approach.
Primary CNS lymphoma is reported to be having an age standardized incidence of 0·4 to 0·5 per 1,00,000 population per year, accounting for less than 1% of all non-Hodgkin's lymphomas and 3% of all brain malignancies. 9 10 11 It has been documented to present with focal neurological deficits (70–73%), neuropsychiatric symptoms (28–43%), seizures (9–14%), ocular symptoms (4%), and increased intracranial pressure (3–33%). 12 13 Most lymphomas are supratentorial in an intra-axial location. The majority of supratentorial lesions occurs either in the cerebral white matter or within the corpus callosum, few in the deep central gray matter, in the posterior fossa, and rarely in the spinal cord. 13 The majority of infratentorial tumors is found in the cerebellum. Seldom have there been any incidence of disease presenting both infra- and supratentorial. 14 With respect to the IAM, it can present as an extra-axial mass, intra-axial mass extending into the cerebellopontine angle (CPA) or a leptomeningeal lymphoma presenting as a CPA mass. 13
The first case report of a lymphoma presenting in bilateral IAC with symptoms of hearing loss and imbalance was only in 1993. 15 Since then there have been a few publications similar to it. 16 17 As per our knowledge, ours is the first report of a lymphoma that is unilateral and with uniqueness of involving only IAM and not brain parenchyma at presentation.
Various tumors of the IAM include VS, lipoma, facial neuroma, hemangioma, meningioma, neurofibroma, hamartoma, and arachnoidal cyst. MRI with contrast has time and again proved its efficacy as the investigation of choice for lesions of the CPA/IAM. It has the advantage of being noninvasive, recognizing the site, extent, and peculiar signal intensities aiding in an apt diagnosis. 18
VS —The majority of patients with a VS presents with asymmetrical hearing loss (slowly progressing) disequilibrium with or without asymmetrical tinnitus. 19 20 21 In relation to the pons, schwannomas are usually isointense or mildly hypointense on T1-weighted images and mildly hyperintense on T2-weighted images. With relation to cerebrospinal fluid, it is noted to be hyperintense on T1 and isointense-hypointense on T2. 22 23 24 25 26
Lipomas —They are thought to be congenital malformations arising from the primitive meninx that forms the subarachnoid space and meninges. 27 The most common signs of CPA lipomas are hearing loss, tinnitus, vertigo, facial symptoms (facial and trigeminal nerve signs), and headaches. 28 When lipomas are viewed using CT, they appear as hypodense masses (−40 to −100 Hounsfield units [HU]) and do not enhance with contrast administration. 29 On a T1-weighted MRI, lipomas are hyperintense, whereas on T2-weighted sequences, the appearance of a lipoma can vary from hypointense to hyperintense. Unique to lipomas is when fat suppression is applied, there is a decrease in or a complete loss of signal intensity from lipomas. 29 30
Meningiomas —Meningiomas involving the IAM present either as tumors primarily originating in the IAM or those extending from adjacent regions. 31 32 CT scan shows characteristic calcification with or without adjacent bone hyperostosis or enostotic spurs. 32 On MRI, they are isointense on T1 and T2, with homogeneous avid enhancement, similar to schwannomas. In 60 to 70% of cases, a “Dural tail” sign may be seen that represents enhancement of non-neoplastic thickening of the peritumoral dura. 32
Cavernous hemangiomas of the IAM originate from the capillary bed of the epineurium surrounding Scarpa's ganglion and can either compress or infiltrate the nerve. 11 33 Most common presentation is that of progressive hearing loss followed by facial nerve involvement ranging from facial spasm to facial paralysis of various grades. 34 On MRI, the characteristic feature of cavernous hemangioma of the IAM is heterogeneous enhancement with gadolinium that distinguishes it from VS, while HRCT reveals irregular bony erosion of the IAM and characteristic intralesional calcification. 35
Facial neuroma —Facial neuroma involving the IAM is a rare entity with patients presenting with facial palsy, paresis, or twitching that is unlikely with a VS. On MRI, lesion is mildly hypointense or isointense relative to brain on noncontrasted T1-weighted images and is enhanced following gadolinium administration. Heterogeneity or cystic change, well appreciated on T2-weighted images, may also be seen. 36
In our case, we encountered IAM to be the primary site of presentation of a primary CNS lymphoma as the patient characteristically presented with unilateral sensorineural hearing loss and subjective giddiness. Cases of primary CNS lymphoma with tendency to spread perineurally have been known and reported in literature. 11
Conclusion
To conclude, an intracanalicular lesion of IAM represents a VS in most instances. In the presence of facial paresis, this could possibly represent a facial neuroma, although they involve the labyrinthine/tympanic or mastoid segment of facial nerve. The above rare but possible differential diagnosis of lesions needs to be considered when dealing with the region reviewed carefully before concluding the lesion as VS only.
With improved imaging modalities, attention must be given to a detailed MRI. A thorough discussion with the neuroradiological team should be a routine protocol that could aid in avoiding potential misdiagnosis of the pathology.
Conflict of Interest • The authors have no ethical conflicts to disclose.
• The authors have no conflicts of interest to declare.
Author Contributions
1. Operating surgeon, rafting the work or revising it critically for important intellectual content. Involved in final approval.
2. Substantial contributions to the conception or design of the work.
3. Radiology team involved in the process.
4. Radiology team involved in the process.
References
- 1.Howitz M F, Johansen C, Tos M, Charabi S, Olsen J H. Incidence of vestibular schwannoma in Denmark, 1977-1995. Am J Otol. 2000;21(05):690–694. [PubMed] [Google Scholar]
- 2.Lin D, Hegarty J L, Fischbein N J, Jackler R K. The prevalence of “incidental” acoustic neuroma. Arch Otolaryngol Head Neck Surg. 2005;131(03):241–244. doi: 10.1001/archotol.131.3.241. [DOI] [PubMed] [Google Scholar]
- 3.Propp J M, McCarthy B J, Davis F G, Preston-Martin S. Descriptive epidemiology of vestibular schwannomas. Neuro-oncol. 2006;8(01):1–11. doi: 10.1215/S1522851704001097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stangerup S E, Tos M, Thomsen J, Caye-Thomasen P.True incidence of vestibular schwannoma? Neurosurgery 201067051335–1340., discussion 1340 [DOI] [PubMed] [Google Scholar]
- 5.Parker G D, Harnsberger H R. Clinical-radiologic issues in perineural tumor spread of malignant diseases of the extracranial head and neck. Radiographics. 1991;11(03):383–399. doi: 10.1148/radiographics.11.3.1852933. [DOI] [PubMed] [Google Scholar]
- 6.Brackmann D E, Bartels L J. Rare tumors of the cerebellopontine angle. Otolaryngol Head Neck Surg (1979) 1980;88(05):555–559. doi: 10.1177/019459988008800508. [DOI] [PubMed] [Google Scholar]
- 7.Van der Meulen M, Dinmohamed A G, Visser O, Doorduijn J K, Bromberg J E. Improved survival in primary central nervous system lymphoma up to age 70 only: a population-based study on incidence, primary treatment and survival in the Netherlands, 1989–2015. Leukemia. 2017 Aug;31(8):1822.treatment and survival in the Netherlands, 1989–2015. Leukemia. 2017;31:1822–1825. doi: 10.1038/leu.2017.128. [DOI] [PubMed] [Google Scholar]
- 8.Villano J L, Koshy M, Shaikh H, Dolecek T A, McCarthy B J. Age, gender, and racial differences in incidence and survival in primary CNS lymphoma. Br J Cancer. 2011;105(09):1414–1418. doi: 10.1038/bjc.2011.357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.European Association for Neuro-Oncology Task Force on Primary CNS Lymphoma . Hoang-Xuan K, Bessell E, Bromberg J. Diagnosis and treatment of primary CNS lymphoma in immunocompetent patients: guidelines from the European Association for Neuro-Oncology. Lancet Oncol. 2015;16(07):e322–e332. doi: 10.1016/S1470-2045(15)00076-5. [DOI] [PubMed] [Google Scholar]
- 10.Lenarz M, Durisin M, Becker H, Lenarz T, Nejadkazem M. Primary central nervous system lymphoma presenting as bilateral tumors of the internal auditory canal. Skull Base. 2007;17(06):409–412. doi: 10.1055/s-2007-991114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Danchaivijitr N, Hesselink J R, Aryan H E, Herndier B. Cerebello-Pontine angle (CPA) lymphoma with perineural extension into the middle fossa: case report. Surg Neurol. 2004;62(01):80–85. doi: 10.1016/j.surneu.2003.07.013. [DOI] [PubMed] [Google Scholar]
- 12.Coulon A, Lafitte F, Hoang-Xuan K. Radiographic findings in 37 cases of primary CNS lymphoma in immunocompetent patients. Eur Radiol. 2002;12(02):329–340. doi: 10.1007/s003300101037. [DOI] [PubMed] [Google Scholar]
- 13.Garth R JN, Coddington R, Brightwell A P. Primary cerebral lymphoma presenting with bilateral cerebellopontine angle lesions. J Laryngol Otol. 1993;107(10):937–939. doi: 10.1017/s0022215100124843. [DOI] [PubMed] [Google Scholar]
- 14.Jaiswal A K, Mahapatra A K, Sharma M C. Primary central nervous lymphoma presenting as bilateral cerebellopontine angle lesions: a rare case report. J Clin Neurosci. 2004;11(03):328–331. doi: 10.1016/S0967-5868(03)00110-3. [DOI] [PubMed] [Google Scholar]
- 15.Larner A J, D'Arrigo C, Scaravilli F, Howard R S. Bilateral symmetrical enhancing brainstem lesions: an unusual presentation of primary CNS lymphoma. Eur J Neurol. 1999;6(06):721–723. doi: 10.1046/j.1468-1331.1999.660721.x. [DOI] [PubMed] [Google Scholar]
- 16.Nishimura T, Uchida Y, Fukuoka M, Ono Y, Kurisaka M, Mori K.Cerebellopontine angle lymphoma: a case report and review of the literature Surg Neurol 19985005480–485., discussion 485–486 [DOI] [PubMed] [Google Scholar]
- 17.Wang Y T, Su H H, Hou Y. Diffuse large B-cell lymphoma of the cerebellopontine angle in a patient with sudden hearing loss and facial palsy. Journal of the Chinese Medical Association. 2007 Jul 1;70(7):294–7. … Wen CY, Huang PH, Chang CJ, Hsieh CT. Primary cerebellopontine angle lymphoma. Kaohsiung J Med Sci. 2015;31(01):55–56. doi: 10.1016/S1726-4901(07)70008-4. [DOI] [PubMed] [Google Scholar]
- 18.Fortnum H, O'Neill C, Taylor R.The role of magnetic resonance imaging in the identification of suspected acoustic neuroma: a systematic review of clinical and cost effectiveness and natural history Health Technol Assess 20091318iii–iv., ix–xi, 1–154 [DOI] [PubMed] [Google Scholar]
- 19.Godefroy W P, Kaptein A A, Vogel J J, van der Mey A G. Conservative treatment of vestibular schwannoma: a follow-up study on clinical and quality-of-life outcome. Otol Neurotol. 2009;30(07):968–974. doi: 10.1097/MAO.0b013e3181b4e3c9. [DOI] [PubMed] [Google Scholar]
- 20.Press G A, Hesselink J R. MR imaging of cerebellopontine angle and internal auditory canal lesions at 1.5 T. AJR. 1988;150(06):1371–1381. doi: 10.2214/ajr.150.6.1371. [DOI] [PubMed] [Google Scholar]
- 21.Tali E T, Yuh W T, Nguyen H D. Cystic acoustic schwannomas: MR characteristics. AJNR Am J Neuroradiol. 1993;14(05):1241–1247. [PMC free article] [PubMed] [Google Scholar]
- 22.Valvassori G E, Garcia Morales F, Palacios E, Dobben G E. MR of the normal and abnormal internal auditory canal. AJNR Am J Neuroradiol. 1988;9(01):115–119. [PMC free article] [PubMed] [Google Scholar]
- 23.Daniels D L, Millen S J, Meyer G A. MR detection of tumor in the internal auditory canal. AJR Am J Roentgenol. 1987;148(06):1219–1222. doi: 10.2214/ajr.148.6.1219. [DOI] [PubMed] [Google Scholar]
- 24.Singh K, Singh M P, Thukral C, Rao K, Singh K, Singh A. Role of magnetic resonance imaging in evaluation of cerebellopontine angle schwannomas. Indian J Otolaryngol Head Neck Surg. 2015;67(01):21–27. doi: 10.1007/s12070-014-0736-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Truwit C L, Barkovich A J. Pathogenesis of intracranial lipoma: an MR study in 42 patients. AJNR Am J Neuroradiol. 1990;11(04):665–674. [PMC free article] [PubMed] [Google Scholar]
- 26.Lagman C, Voth B L, Chung L K. Evaluating the utility of a scoring system for lipomas of the cerebellopontine angle. Acta Neurochir (Wien) 2017;159(04):739–750. doi: 10.1007/s00701-017-3076-5. [DOI] [PubMed] [Google Scholar]
- 27.Scuotto A, Cappabianca S, D'Errico C. Lipomas of the cerebellopontine angle: neuroradiological and surgical considerations. Review of the literature and report of our experience. Clin Neurol Neurosurg. 2013;115(10):2280–2283. doi: 10.1016/j.clineuro.2013.07.029. [DOI] [PubMed] [Google Scholar]
- 28.Bertot B, Steele W J, Boghani Z, Britz G. Diagnostic dilemma: cerebellopontine angle lipoma versus dermoid cyst. Cureus. 2017;9(11):e1894. doi: 10.7759/cureus.1894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.von Eckardstein K L, Driscoll C L, Link M J. Outcome after microsurgery for meningiomas involving the internal auditory canal. Neurosurgery. 2010;67(05):1236–1242. doi: 10.1227/NEU.0b013e3181efe412. [DOI] [PubMed] [Google Scholar]
- 30.Pollock B E, Link M J, Foote R L, Stafford S L, Brown P D, Schomberg P J.Radiosurgery as primary management for meningiomas extending into the internal auditory canal Stereotact Funct Neurosurg 200482(2-3):98–103. [DOI] [PubMed] [Google Scholar]
- 31.Helie O, Soulie D, Sarrazin J L, Derosier C, Cordoliani Y S, Cosnard G. [Magnetic resonance imaging and meningiomas of the posterior cerebral fossa. 31 cases] J Neuroradiol. 1995;22(04):252–270. [PubMed] [Google Scholar]
- 32.Wallace E W. The dural tail sign. Radiology. 2004;233(01):56–57. doi: 10.1148/radiol.2331021332. [DOI] [PubMed] [Google Scholar]
- 33.Barrera J E, Jenkins H, Said S. Cavernous hemangioma of the internal auditory canal: a case report and review of the literature. Am J Otolaryngol. 2004;25(03):199–203. doi: 10.1016/j.amjoto.2003.11.002. [DOI] [PubMed] [Google Scholar]
- 34.Omojola M F, al Hawashim N S, Zuwayed M A, al Ferayan A. CT and MRI features of cavernous haemangioma of internal auditory canal. Br J Radiol. 1997;70(839):1184–1187. doi: 10.1259/bjr.70.839.9536913. [DOI] [PubMed] [Google Scholar]
- 35.Shaida A M, McFerran D J, da Cruz M, Hardy D G, Moffat D A. Cavernous haemangioma of the internal auditory canal. J Laryngol Otol. 2000;114(06):453–455. doi: 10.1258/0022215001905823. [DOI] [PubMed] [Google Scholar]
- 36.Kertesz T R, Shelton C, Wiggins R H, Salzman K L, Glastonbury C M, Harnsberger R. Intratemporal facial nerve neuroma: anatomical location and radiological features. Laryngoscope. 2001;111(07):1250–1256. doi: 10.1097/00005537-200107000-00020. [DOI] [PubMed] [Google Scholar]


