Abstract
Background:
Silver nanoparticles (AgNP) are commonly used metallic nanoparticles in health care systems. Colon cancer incidence is increasing worldwide. In this study, AgNP was synthesized using β-sitosterol and its cytotoxic potential was evaluated in human colon cancer (HT-29) cells.
Methods:
Characterization of AgNP was analyzed by TEM and spectrophotometry analysis. HT-29 cells were treated with different concentrations (2, 4, 6, 8 and 10 ng/ml) of AgNPs and cytotoxicity was evaluated by MTT assay. The apoptosis was analyzed by the flow cytometry. The expression of p53 protein was analyzed by western blotting.
Results:
β-sitosterol mediated AgNP are spherical in shape and induced concentration-dependent cytotoxicity in HT-29 cells. AgNP caused apoptosis related morphological changes as evidenced by annexin positive staining. AgNP treatments also induced the p53 expression in HT-29 cells.
Conclusion:
Our present result suggests that β-sitosterol mediated AgNP induce apoptosis in colon cancer cells and this finding may pave the way for further experimental analysis in vivo.
Keywords: Apoptosis, Colon neoplasms, HT29 cells, Silver nanoparticles, β-sitosterol
Introduction
Conventional anticancer therapies with the standard chemotherapeutic drugs have several limitations in terms of adverse and off-target effects against healthy cells 1. Nanotechnology has made significant contributions to cancer treatment over the past decades 2. Silver nanoparticles (AgNP) are commonly employed metallic nanoparticles in health care systems due to their unique physiochemical and biological properties and are used as antibacterial, antifungal, antiviral and anti-cancer agents. β-sitosterol is a phytosterol commonly isolated from traditional Chinese herbs, such as Polygonum hydropiper, Trifolium repens, Houttuynia cordata and Lasia spinosa 3. β-sitosterol has been used in the treatment of several diseases due to its antioxidant, anti-inflammatory, anti-proliferative and anticancer properties 4. Despite significant clinical advancements, colon cancer remains one of the most common causes of cancer-related deaths. Therefore, there is an urgent need for the development of therapeutic drug candidate for the treatment of colon cancer. Also, studies regarding the anticancer potential of β-sitosterol-mediated AgNP are not available. Therefore, this study was conducted to explore the above lacunae.
Materials and Methods
Synthesis and characterization of β-sitosterol mediated AgNP
First, 100 mg of β-sitosterol was (used as a reducing agent) dissolved in 10 ml of distilled water and mixed thoroughly. To this, 1 mM of 90 ml silver nitrate solution was added and mixed well and kept in a shaker for nanoparticles synthesis at 300 RPM. The color change of β-sitosterol with silver nitrate solution was observed carefully under conditions of surface plasmon resonance using UV-Vis spectrophotometer. The synthesized nanoparticle was dried for the characterization of morphology using transmission electron microscopy. ImageJ software was used to measure different sizes of nanoparticles and the graph was plotted.
Cell culture and maintenance
The human colon cancer (HT-29) was obtained from National Centre for Cell Science (Pune, Maharashtra, India). HT-29 cells were cultured using low glucose DMEM containing penicillin (100 units/ml) and streptomycin (100 μg/ml) and maintained under standard culture conditions with 5% CO2 at 37°C. AgNP were dissolved in distilled water and the treatments were given for 24 hr.
MTT assay
MTT assay was done to evaluate AgNP induced cytotoxic effect in HT-29 cells 5. Briefly, 1×104 cells/well were seeded in 96 well plates. After 24 hr, existing medium was replaced with fresh expansion culture medium containing different concentrations of AgNP, i.e. 2, 4, 6, 8, 10 ng/ml and incubated for 24 hr. Then, cells were incubated with MTT (0.5 mg/ml) for 4 hr and after incubation, intensity of purple-blue formazan was quantified using Perkin Elmer multimode Reader (USA) at 570 nm.
Calcein-AM staining assay
Cell viability was analyzed by calcein-AM for live cell staining according to manufacturer’s instruction (BD Biosciences, San Jose, CA). Briefly, after treatments, cells were washed twice with PBS and were stained with calcein-AM (0.2 μM) and incubated for 30 min at 37°C and then images were captured using Nikon Eclipse Ti Fluorescence Microscope (Nikon Instruments Inc., NY, USA)
Morphological analysis of apoptosis using annexin V staining
Cells were seeded at a density of 1×104 cells/well in 96 well plates for adherence. At the end of the treatment period, cells were stained with FITC (fluorescein isothiocyanate)-annexin V kit (BD Biosciences, San Jose, CA) for 15 min. Then, cells were washed thrice with PBS and representative fields were captured immediately using Nikon Eclipse Ti Fluorescence Microscope (Nikon Instruments Inc., NY, USA).
Apoptosis detection by flow cytometry
The apoptotic cells were quantified by annexin VFITC/PI (Propidium iodide) co-staining assay. Briefly, at the end of the 24 hr incubation, the cells were centrifuged at 1800 rpm for 8 min. The pellet was resuspended in 50 μl of binding buffer containing 0.5 μl of annexin V-FITC and then incubated at 4°C for 30 min in the dark. PI (50 μg/ml) in 200 μl binding buffer was added to each of the tubes and incubated for 5 min. Finally, the cells were analyzed in flow cytometer (Cy-An ADP Analyzer, Beckman Coulter, USA).
Western blot analysis
Total protein estimation was done using Bovine Serum Albumin (BSA). Electrophoresis was done with protein extracts and electroblotted onto polyvinylidene difluoride membrane. Then, the membrane was blocked with 5% BSA and incubated overnight with monoclonal p53 primary antibody at 4°C for 2 hr with corresponding secondary antibodies at room temperature. The enhanced chemiluminescence with protein A-horseradish peroxidase was used to detect the immunoreactive bands.
Statistical analysis
Data were expressed as mean±SEM and analyzed by one way ANOVA followed by Dunnett’s multiple comparison and p<0.05 was considered statistically significant (Graph Pad prism 7.0, CA, USA).
Results
Cytotoxic potential of AgNP in HT-29 cells
The color transition confirmed the formation of β-sitosterol assisted AgNP (Supplementary file). The UV-visible spectrophotometric analysis of AgNPs shows the absorbance peak between 410 to 440 nm which indicates the formation of AgNP (Figure 1A). TEM analysis illustrates that AgNP are spherical with a diameter of 4–21 nm (Figures 1B and C). The control and AgNP treated cells showed typical cancer cell morphology (Figure 2A). Initially, to evaluate the cytotoxic potential of AgNP, treatment was given at different concentrations from 2, 4, 6, 8 to 10 ng/ml. The AgNP induced a concentration dependent cytotoxicity in HT-29 cells (p<0.001) (Figure 2B). The IC50 of AgNP in HT-29 cells was calculated at concentration of 7 ng/ml. Therefore, further studies were carried out with two different concentrations of AgNP, i.e., 7 and 14 ng/ml.
Supplementary file.

β-sitosterol Mediated Silver Nanoparticles synthesis and colour transition.
Figure 1.
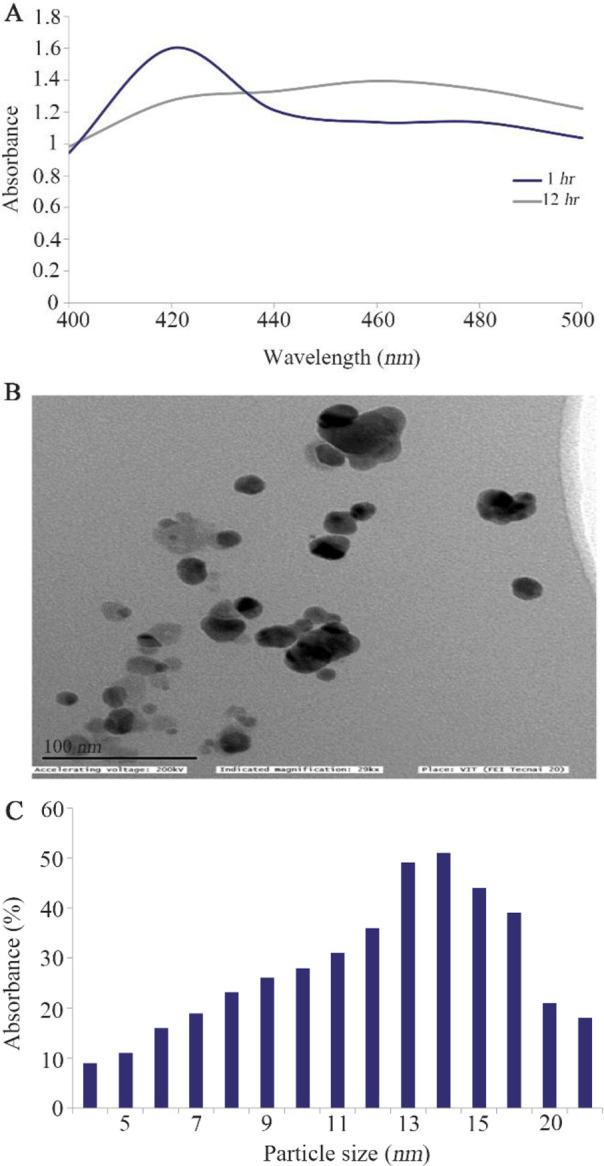
Synthesis and characterization of AgNP using sitosterol. A) UV-vis spectroscopic spectrum of AgNP. B) TEM image of AgNP. C) Histogram shows the size distribution of nanoparticles. ImageJ software was used to measure different sizes of nanoparticles.
Figure 2.
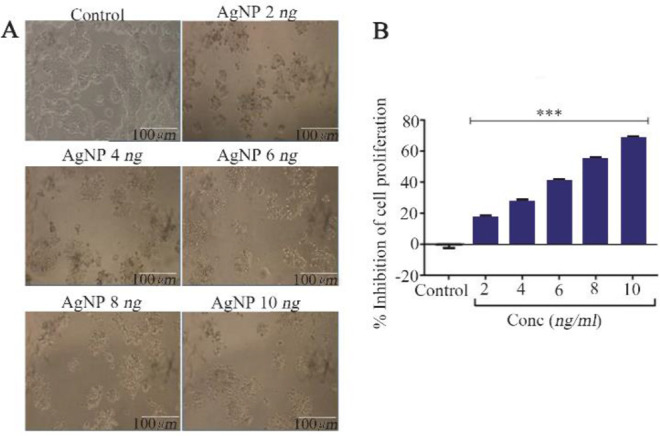
β-sitosterol mediated silver nanoparticles (AgNP) induced changes in the proliferation of HT-29 cells. A) Morphology of control and AgNP treated HT-29 cells, scale bar of 100 μm. B) Cytotoxi-city analysis by MTT assay, n=3, *** p<0.001 vs. control.
Morphological analysis of apoptosis
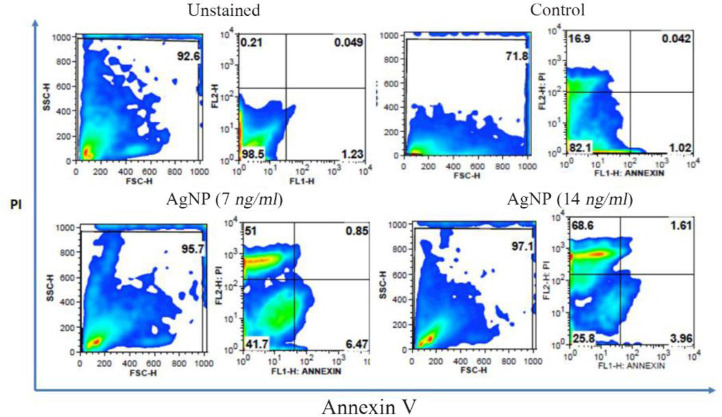
Increased calcein release was noticed in control cells which indicates that cells have high proliferation index. AgNP treatments at two different concentrations (7 and 14 ng/ml) caused prominent decrease in calcein staining and cell viability when compared to control cells (Figure 3). The control cells remained unstained by annexin v as their cell membrane was intact. AgNP treatments in HT-29 cells caused a concentration dependent expression of annexin v (Figure 4A). The percentage of annexin v expressed cells increased in a concentration dependent manner as compared to control (p<0.001) (Figure 4B). The unstained (without annexin v and PI) control cells (With annexin v and PI without AgNP treatments) did not show any significant change in their viability and their viability was 98.5 and 82.1%, respectively. AgNP treated cells at concentrations of 7 and 14 ng/ml showed prominent apoptotic changes, i.e., 51 and 68.6%, respectively and decreased viability of 41.7 and 25.8%, respectively (Figure 5).
Figure 3.
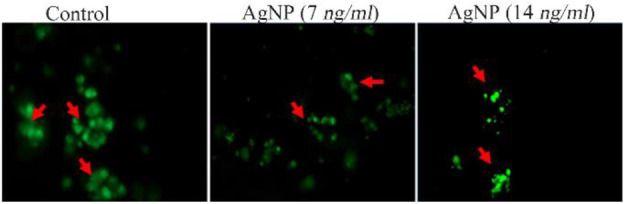
Cell viability analysis by calcein AM staining. Red arrow showing green fluorescence indicates the viable cells.
Figure 4.
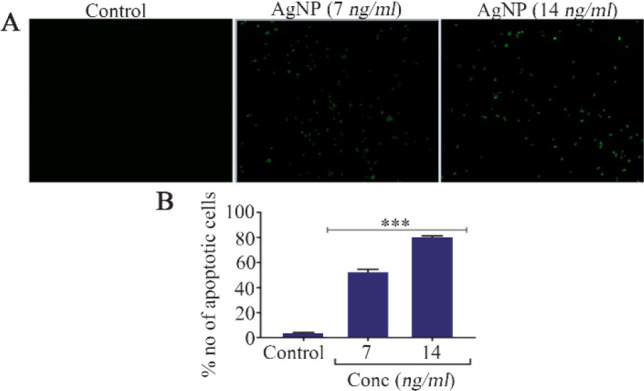
A) Morphological analysis of apoptosis by annexin V staining, B) Quantification of apoptotic cells, n=3, *** p<0.001 vs. control.
Figure 5.
Flow cytometry analysis of apoptosis using annexin v and propidium iodide staining. Left upper quadrant percentage indicates the presence of apoptotic cells.
AgNP treatments and increased p53 protein expression
The HT-29 colon cancer cells as control cells showed reduced p53 protein expression. AgNP treatments caused a significant and concentration dependent increase in the p53 protein expression (Figure 6A). The quantification of p53 expression also confirmed the significant increase (7 ng/ml vs. p<0.05, and 14 ng/ml vs. p<0.01) upon AgNP treatments (Figure 6B).
Figure 6.
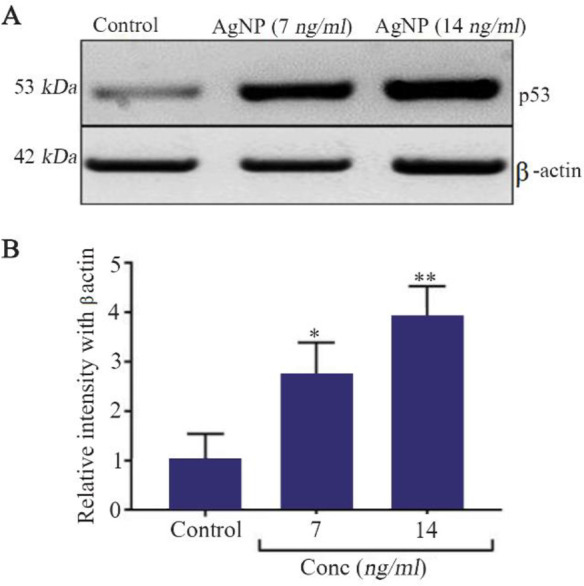
A) Western blot analysis of p53 expression. B) Quantification of p53 protein expression by densitometry analysis, n=3, *p< 0.05 and ** p<0.01 vs. control.
Discussion
In previous studies, AgNP were biosynthesized using medicinal plants, and their derived phyto-compounds and microbes were demonstrated to induce cytotoxicity in a variety of cancer cells including HT-29 cells 6. It is likely that regardless of mode synthesis, AgNP are cytotoxic to HT-29 cancer cells.
Viable cells convert calcein-AM into calcein by esterase enzyme and emit a strong green fluorescence 7. In this study, the reduced green fluorescence was observed in cells treated with AgNP indicating the fall in cell viability. In early apoptotic stage, phosphatidylserine residues migrate from the inner to outer leaflet of the plasma membrane and annexin V precisely binds to the exposed phosphatidylserine residues and therefore, it has been used as a specific probe for morphological investigation of apoptosis 5,8. In view of the above reports, our current result also confirms the presence of phosphatidylserine exposed on the surface of early apoptotic cells only in the experimental groups.
p53 is a tumor suppressor protein and its functional loss was observed frequently in a variety of human cancers 9. Studies have shown that AgNP has the potential to induce p53 protein expression in human cancer cells 10. However, in this study, it was shown that AgNP induced an increase in p53 protein expression. The AgNP induced p53 expression could be the possible cause for the apoptosis observed in this study.
Conclusion
The present study demonstrates that β-sitosterol derived AgNP are <21 nm in size and they can effectively induce cytotoxicity in human colon cancer cells. AgNP induced early apoptosis via enhanced p53 protein expression. The present study provides the support to possible application of AgNP in colon cancer therapy and further in vivo studies are warranted to substantiate this notion.
Acknowledgement
Authors wish to thank Dr. R. Parameshwari, the researcher of Sri Ramachandra Institute of Higher Education and Research, Chennai, Tamil Nadu, India for her scientific contribution.
Footnotes
Conflict of Interest
The authors declare they have no conflict of interest.
References
- 1.Jahangirian H, Kalantari K, Izadiyan Z, Rafiee-Moghaddam R, Shameli K, Webster TJ. A review of small molecules and drug delivery applications using gold and iron nanoparticles. Int J Nanomedicine 2019;14:1633–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Shi J, Kantoff PW, Wooster R, Farokhzad OC. Cancer nanomedicine: progress, challenges and opportunities. Nat Rev Cancer 2017;17(1):20–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Vijaya Yadav AK. In vitro anthelmintic assessment of selected phytochemicals against Hymenolepis diminuta, a zoonotic tapeworm. J Parasit Dis 2016;40(3):1082–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shi C, Wu F, Zhu XC, Xu J. Incorporation of beta-sitosterol into the membrane increases resistance to oxidative stress and lipid peroxidation via estrogen receptor-mediated PI3K/GSK3beta signaling. Biochim Biophys Acta 2013;1830(3):2538–44. [DOI] [PubMed] [Google Scholar]
- 5.PonselviInduja M, Ezhilarasan D, Ashok Vardhan N. Evolvulus alsinoides methanolic extract triggers apoptosis in HepG2 cells. Avicenna J Phytomed 2018;8(6):504–12. [PMC free article] [PubMed] [Google Scholar]
- 6.Gurunathan S, Kang MH, Kim JH. Combination effect of silver nanoparticles and histone deacetylases inhibitor in human alveolar basal epithelial cells. Molecules 2018;23 (8):E2046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kummrow A, Frankowski M, Bock N, Werner C, Dziekan T, Neukammer J. Quantitative assessment of cell viability based on flow cytometry and microscopy. Cytometry A 2013;83(2):197–204. [DOI] [PubMed] [Google Scholar]
- 8.Akther T, Mathipi V, Kumar NS, Davoodbasha MA, Srinivasan H. Fungal-mediated synthesis of pharmaceutically active silver nanoparticles and anticancer property against A549 cells through apoptosis. Environ Sci Pollut Res Int 2019;26(13):13649–57. [DOI] [PubMed] [Google Scholar]
- 9.Rohit Singh T, Ezhilarasan D. Ethanolic extract of Lagerstroemia speciosa (L.) Pers., induces apoptosis and cell cycle arrest in HepG2 cells. Nutr Cancer 2020;72(1): 146–56. [DOI] [PubMed] [Google Scholar]
- 10.Kovács D, Igaz N, Keskeny C, Bélteky P, Tóth T, Gáspár R, et al. Silver nanoparticles defeat p53-positive and p53-negative osteosarcoma cells by triggering mitochondrial stress and apoptosis. Sci Rep 2016;6:27902. [DOI] [PMC free article] [PubMed] [Google Scholar]