Abstract
Parkinsonism developed owing to viruses is one of the important causes of secondary parkinsonism. After the Spanish flu pandemic, the increase in the number of parkinsonian cases in the long term has drawn attention on the relationship between viruses and parkinsonism. For this reason, the relationship between influenza and parkinsonism has been studied most. Nowadays in which we are experiencing the COVID-19 pandemic, scientists, based on the experiences gained from the Spanish flu pandemic, have drawn attention to the fact that the third wave of the pandemic might be parkinsonism. However, as we have reviewed in the literature, acute parkinsonism due to COVID-19 was not reported during this pandemic. Here, we present a case in which signs of acute parkinsonism developed on the 3rd day of the illness and neurological symptoms regressed with convalescent plasma treatment.
Keywords: Covid 19, Parkinsonism, Neurologic symptoms
1. Introduction
Coronaviruses are an enveloped single-stranded RNA virus belonging to the coronaviridea family. They bind to Angiotensin-converting enzyme2 (ACE 2). ACE 2 is common in alveoli, trachea bronchi and airways. The virus multiplies in these cells and enters new target cells. COVİD-19 causes the most common cold symptoms [1]. As the disease progresses, more complex and severe processes such as acute respiratory distress syndrome and cytokine storm may develop. This is the tip of the iceberg. Our knowledge about this disease is increasing day by day. It has been reported that a hypercoagulable state, heart muscle damage, renal tubule cells damage due to COVID 19 develops. This infection can cause a wide spectrum of illnesses, ranging from a simple cold to multicystemic diseases [1].
As the COVID-19 pandemic progresses, different system involvements and complications of the SARSCoV-2 virus occur. The nervous system makes up an important group of them. In the study involving 40,469 COVID-19 positive patients, 22.5% of patients were shown to have neuropsychiatric symptoms associated with COVID-19 [2]. These findings are thought to be the direct consequences of the virus on the nervous system or para-infectious/post-infectious immune-mediated or neurological complications of the systemic effects of COVID-19 [3]. Neurological involvements and complications such as stroke, encephalitis, headache, seizure, impaired consciousness, hyposmia and hypogeusia, Guillain Barré syndrome (GBS) and Miller Fisher syndrome have been reported to date [4].
Parkinsonism is a progressive disease with resting tremor, cog wheel rigidity, and bradykinesia. It is more common in men.The loss of dopaminergic neurons in the substantia nigra pars compact region in the brain plays a role in Parkinson's pathophysiology. Parkinsonism has 6 cardinal signs: Resting tremor, bradykinesia, rigidity, postural instability, antiflexion posture and motor blocks. To be diagnosed clinically, there should be two of these 6 symptoms, one of which is tremor or bradykinesia [5].
Here, we present the COVID-19 case presented with acute parkinsonism.
2. Case
A 72-year-old male patient was brought to the emergency room with complaints of altered consciousness, retardation in movements, and tremors. It was learned that the patient, who was previously known to have diabetes mellitus, hypertension and peripheral artery disease, was diagnosed with COVID-19 confirmed by Real-Time-Polymerase-Chain-Reaction in the hospital where he applied with sore throat and fever 2 days ago. Paracetamol and favipiravir were started on the patient, and his fever subsided the next day. On the 3rd day of the diagnosis of COVID-19, it was learned that the patient was observed to have dull gaze not answering the questions asked, impaired walking and tremors and that the emergency call system was notified. The patient's blood pressure was 130/80 mmHg, fever was 36.5 degrees, respiratory rate was 22 and saturation was 92%. In the neurological examination, he was conscious, but he did not sense the deixis referring to place, time and person. Asymmetry was not detected in the examination of muscle strength, sensory, cerebellar and head pair. In the tonus examination, it was observed that he had rigidity and cogwheel rigidity. His movements were bradykinetic. His gait was in an antiflexion position and was slowed down. He had a rest tremor. He had rales in the right lung basal. He had no history of drug intake that caused parkinsonism. No acute pathology was detected in his brain tomography and diffusion-weighted magnetic resonance imaging. Increases, due to Covid pneumonia, in bilateral ground glass density of the patient, whose thoracic tomography taken on the day of diagnosis was normal, were observed in the thoracic CT taken at the time of admission to the hospital (Fig. 1 ). Routine biochemical tests were normal in laboratory tests, CRP 13.2 m / dl, ferritin 67μgr/L, D-dimer 0.7 mg/L, Fibrinogen 493 g/L, lymphocyte 1.331033/mm3, platelet 1021033/mm3. On the day of hospitalization, 1 unit of convalescent plasma treatment was administered. Approximately 4 h after the administration of the conventional treatment, it was noticed that his orientation and cooperation improved and his rigidity decreased. In the following day evaluation, the patient was consciously oriented and cooperative, and his rigidity, tremors and bradykinesia disappeared. However, the lymphocyte count fell to 0.721033/mm3. Other tests were normal. As respiratory distress increased, 1more unit convalescent plasma treatment was given, high-flow nasal cannula and noninvasive mechanical ventilation were initiated. On the 5th day saturation decreased (Fig. 2 ), lymphocyte decreased to 0.411033/mm3, platelet to 701033/mm3, Ferritin increased to 309.7 μgr/L, Fibrinogen 918 g/L, CRP 271 mg/L. Plasmapheresis treatment was decided to be applied to the patient who developed ARDS. After 3 days of plasmapheresis, the patient's oxygen saturation improved, respiratory distress decreased, and he was transferred to the service. The patient, who was followed up in the intensive care unit for 14 days and in the ward for 8 days, was discharged with full recovery. His neurological examination in the follow-up 2 months was found normal. Symptoms and signs of parkinsonism were not observed.
Fig. 1.
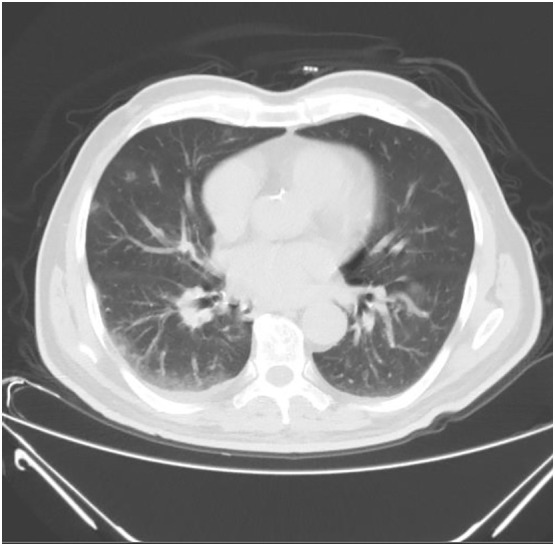
Tomography image of the patient at admission (3th day).
Fig. 2.

Posteroanterior chest radiography of the patient on the 5th day.
3. Discussion
It has been previously reported that acute parkinsonism might be associated with neurotropic viruses [6]. The first link between viruses and parkinsonism was established between the increased cases of parkinsonism after the Spanish flu broke out in 1918. In the acute form, the clinical presentation called encephalitis lethargica appeared [7]. 28 types have been defined so far, and one of them is the form occurring with parkinsonism [7]. Viruses can cause parkinsonism directly or indirectly. The direct effect occurs with the development of virus tropism, replication and subsequent neuronal lysis for the basal ganglia. On the other way, microglia activation, release of proinflammatory factors and T cell response, hyperstokinemia with vascular damage and hypoxic brain are possible indirect pathophysiological mechanisms [4].
The range of neurological symptoms reported in patients is wide, and this suggests the potential for neurotropism of SARS-CoV-2 [8]. Recent findings revealing that coronaviruses are neurotrophic suggest that dopaminergic neurons enable SARS-CoV-2 infection [9]. ACE2 is the co-receptor of virus spike proteins that play an important role in COVID-19. After the spike protein binds to the ACE2 receptor in the target cell, it combines with the cell membrane of the viral envelope and transfers the viral genome to the target cell [10]. ACE2 has been detected in the substantia nigra, ventricles, middle temporal gyrus, posterior cingulate cortex, and olfactory bulb [10]. Especially the possibility of the substantia nigra to be infected might contribute to the acute parkinsonism situation. Beauchamp et al. suggested that the acute effect of the virus on the nervous system was largely overshadowed by respiratory effects and there is need to develop long-term monitoring strategies for individuals in recovery including neurodegenerative sequelae such as virus-associated parkinsonism [8]. To date, one case of parkinsonism associated with Covid-19 has been reported [11]. It occurred in the later stages of the disease.In our case, without respiratory symptoms at the beginning of the disease, parkinsonism developed, and interestingly, the neurological symptoms regressed with convalescent plasma treatment. Convalescent plasma is widely used in epidemics where vaccine and drug therapy are not yet available [12]. Convalescent plasma not only neutralizes the pathogen, but it also provides passive immunomodulatory effects that allow control of the excessive inflammatory response induced by the infectious agent [13]. We are of the opinion that this situation which occurred at the acute phase of the disease should stem from the neurotropic effect of the virus. It should be kept in mind that the option for the convalescent plasma treatment be given not only for respiratory system findings but also for other system involvements. Humanity has been acquainted with the SARS-CoV-2 virus for only a year and its new features are discovered every day. Additional studies are needed to demonstrate the effect of SARS-CoV-2 leading to the development of parkinsonian symptoms and to expand the treatment indications for which convalescent plasma therapy can be applied.
4. Conclusion
Clinicians, especially in emergency rooms, should not focus on the typical symptoms of the epidemic such as fever, cough, muscle pain, and its effects on the respiratory system, but be alert to the many clinical manifestations this mysterious virus might cause.
Fundingandsupport
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Permission was obtained from the patient for the use of materials belonging to the patient.
References
- 1.Abd El-Aziz T.M., Stockand J.D. Recent progress and challenges in drug development against COVID-19 coronavirus (SARS-CoV-2) - an update on the status. Infect. Genet. Evol. 2020;83(104327) doi: 10.1016/j.meegid.2020.104327. 11–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nalleballe K., Onteddu S.R., Sharma R., et al. Spectrum of neuropsychiatric manifestations in COVID-19. Brain Behav. Immun. 2020;88:71–74. doi: 10.1016/j.bbi.2020.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ellul M.A., Benjamin L., Singh B., et al. Neurological associations of COVID-19. Lancet Neurol. 2020;19:767–783. doi: 10.1016/S1474-4422(20)30221-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sulzer D., Antonini A., Leta V., et al. COVID-19 and possible links with Parkinson’s disease and parkinsonism: from bench to bedside. Npj Parkinson’s Dis. 2020;6(18):1–10. doi: 10.1038/s41531-020-00123-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cander B. In: Nörolojik Aciller, Istanbul, Istanbul Medikal Sağlık ve Yayıncılık. Aslan S., Demir R., editors. 2016. Acil Tıp Temel Başvuru Kitabı; pp. 1757–1767. editors. [Google Scholar]
- 6.Jang H., Boltz D.A., Webster R.G., Smeyne R.J. Viral parkinsonism. Biochim. Biophys. Acta. 2009;1792:714–721. doi: 10.1016/j.bbadis.2008.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vilensky J.A., Gilman S. Encephalitis lethargica: could this disease be recognised if the epidemic recurred? Pract. Neurol. 2006;6:360–367. [Google Scholar]
- 8.Beauchamp L.C., Finkelstein D.I., Bush A.I., Evans A.H., Barnham K.J. Parkinsonism as a third wave of the COVID-19 pandemic? J. Parkinsons Dis. 2020;10(4):1343–1353. doi: 10.3233/JPD-202211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yang L., Han Y., Nilsson-Payant B.E., et al. A human pluripotent stem cell-based platform to study SARS-CoV-2 tropism and model virus infection in human cells and organoids. Cell Stem Cell. 2020;27 doi: 10.1016/j.stem.2020.06.015. 125–136.e127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Losy J. SARS-CoV-2 infection: symptoms of the nervous system and implications for therapy in neurological disorders. Neurol. Ther. 2020;23:1–12. doi: 10.1007/s40120-020-00225-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Guerrero A.M., Laespada-García M.I., Grande A.G., et al. Acute hypokinetic-rigid syndrome following SARS-CoV-2 infection. Neurology. 2020;95:e2109–e2118. doi: 10.1212/WNL.0000000000010282. [DOI] [PubMed] [Google Scholar]
- 12.Bloch E.M., Shoham S., Casadevall A., et al. Deployment of convalescent plasma for the prevention and treatment of COVID-19. J. Clin. Invest. 2020;130(6):2757–2765. doi: 10.1172/JCI138745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rojas M., Rodríguez Y., Monsalve D.M., et al. Convalescent plasma in Covid19: possible mechanisms of action. Autoimmun. Rev. 2020;19 doi: 10.1016/j.autrev.2020.102554. [DOI] [PMC free article] [PubMed] [Google Scholar]


