Abstract
Kaempferol (KF), a flavonoid compound isolated from herbal medicines, has been reported to play a significant role in inhibiting certain types of cancer. Although recent studies reported that KF exerted inhibitive activity on liver cancer, they failed to elucidate the signaling pathways and synergistic effects in combination with chemotherapeutic drugs currently in use in the clinical setting. In the present study, the signaling pathways and synergistic effects of KF in liver cancer cells were investigated. Nine liver cancer cell lines were used to assess the inhibitive activity and synergistic effects of KF. Cellular behavioral experiments, such as viability, colony formation, cell cycle arrest, apoptotic, wound healing, and Transwell assays were used to assess the effects of KF on the proliferation, apoptosis, migration, and invasion of liver cancer cells. Western blotting was performed to validate the key signaling pathway elements underlying those cellular behaviors. KF exhibited inhibitory effects on nine liver cancer cell lines in time- and dose-dependent manners and was mostly nontoxic to the normal hepatocyte cells. The combination of KF and doxorubicin revealed a stronger inhibitive effect on the viability of liver cancer cells. Combination therapy also revealed higher suppressive effects on colony formation, cell cycle progression, survival, DNA damage response, and mitochondrial function. By western blotting assay, mitochondrial and caspase signaling pathways were determined to be involved in proliferation inhibition. In wound healing and Transwell invasion assays, combination therapy also exhibited more robust inhibitory activity in blocking the migration and invasion of liver cancer cells. PI3K/mTOR/MMP protein pathways were also revealed to be related to cell migration inhibition. KF alone exhibited an inhibitory effect on proliferation, migration, and invasion of liver cancer cells, and its synergistic effects revealed stronger inhibitory activities. The present data indicated that KF is a promising candidate as a complementary medicine to conventional chemotherapeutic drugs.
Keywords: liver cancer, kaempferol, doxorubicin, combination medication, apoptosis, migration, invasion
Introduction
Liver cancer is the most common type of primary liver cancer. It usually occurs in people with chronic liver diseases, such as cirrhosis caused by hepatitis B or hepatitis C infection (1). In 2012, over 70,000 cases were diagnosed worldwide, and the mortality rate due to liver cancer and its incidence are increasing annually. Among these cases, over 80% occurred in East Asia and sub-Saharan Africa (1). Although chemotherapy is an important therapeutic method for liver cancer in combination with surgery and radiotherapy, its side effects are obvious, such as drug resistance and toxicity (2). Traditional Chinese medicine (TCM) is a system of medical care that has been developed in China over thousands of years (3–7). It has been used for treating cancers in clinical application due to advantages such as low toxicity, high efficacy, moderate costs, and high acceptability (8–10). In recent years, TCM has been widely accepted as a type of complementary and alternative medicine (CAM) in numerous countries around the world, such as the United States, Canada, and the European Union (11).
Kaempferol (KF), or 3,4′,5,7-tetrahydroxyflavone, is a natural flavonoid found in numerous herbal medicines and fruits (12). A few studies reported that the application of KF may reduce the risk of various diseases, such as cancers, diabetes, cardiovascular disorders, bacterial infection, and viral infection (13,14). In vitro studies along with some animal testing have demonstrated the wide range of potential antitumor properties of KF (15,16). Antitumor effects have been identified for certain malignant cancer cells (including breast cancer, ovarian cancer, leukemia, as well as bladder, gastric, colorectal, pancreatic and lung cancer), revealing an ability to interrupt cell growth, limit angiogenesis, induce apoptosis, and reduce the ability to metastasize (17,18).
Doxorubicin (DOX) is a chemotherapy medication used to treat numerous cancers. It works through interaction with DNA by the intercalation and inhibition of macromolecular biosynthesis, leading to cellular apoptosis. However, the therapeutic effects of DOX are hampered by its significant side effects and acquired drug resistance (19). In addition to hair loss, bone marrow suppression, vomiting, rash, and other slight side effects, DOX can induce serious allergic reactions, heart damage, radiation recall, treatment-related leukemia, and hand-foot syndrome (20–22). Numerous studies have revealed that the combination of chemotherapy with various types of TCMs can reduce the side effects of DOX and enhance its drug sensitivity. For example, Lin et al revealed that Astragaloside IV could significantly reduce DOX-induced cardiotoxicity (23). Li et al revealed that oridonin could assist DOX against aggressive breast cancer by promoting apoptosis and suppressing angiogenesis (24). Tanshinone IIA, a compound found in the plant Danshen, exhibited the capacity to overcome DOX resistance in gastric cancer cells (25). As a result, a question arises regarding whether the combination of doxorubicin with KF could lead to a better therapeutic outcome than monotherapies.
Materials and methods
Reagents
KF was purchased from Sigma-Aldrich; Merck KGaA (cat. no. K0133) and was initially dissolved in dimethyl sulfoxide (DMSO) into a 50 mg/ml solution according to the manufacturer's instructions, and then diluted in Dulbecco's modified of Eagle's medium (DMEM) (Gibco/BRL; Thermo Fisher Scientific, Inc.) to the desired working concentrations. DOX was purchased from Sigma-Aldrich; Merck KGaA (cat. no. D1515) and directly dissolved in DMEM into the desired concentration. The structure of KF is presented in Fig. 1.
Figure 1.
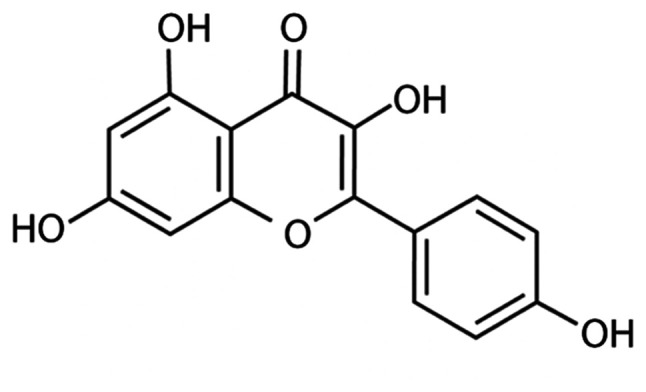
Chemical structure of Kaempferol.
Cell lines and culture
Liver cancer cell lines Huh-7, Huh-1, HepG2, HepG2.2.15, SK-Hep-1, PLC/PRF/5, HLE, HLF, and Hep3B were purchased from the Chinese Academy of Sciences affiliated cell bank. The HepG2 and SK-Hep-1 cell lines were authenticated via STR profiling method. The normal hepatocyte cell line was purchased from Lonza Group, Ltd. Hepatocyte cells were cultured in the growth medium supplied by the manufacturer and liver cancer cells were maintained in DMEM medium (cat. no. 11965084) supplemented with 10% fetal bovine serum (FBS; cat. no. 12483020; both from Gibco/BRL; Thermo Fisher Scientific, Inc.), 100 U/ml penicillin, and 100 mg/ml streptomycin at 37°C in a humidified incubator with 5% CO2.
Cell viability
Cells were seeded in cell culture medium at a concentration of 3×104 cells/100 µl/well in microtiter plates (tissue culture grade, 96 wells, flat bottom) and incubated for 24 h at 37°C and 5% CO2. After the incubation period, 10 µl of MTT-labeling reagent was then added to each well to a final concentration of 0.5 mg/ml. The cells were then incubated for an additional 4 h at 37°C in a humidified atmosphere. Solubilization solution (Sigma-Aldrich; Merck KGaA) was then added at 100 µl per well and allowed to stand overnight in the incubator in a humidified atmosphere. After overnight incubation, the cells were checked to ensure the complete solubilization of purple formazan crystals, and absorbance was then measured at a wavelength 570 nm using a microplate (ELISA) reader.
Colony formation assay
Cells were separated into a 6-well plate at a density of 500 cells/well and cultured for 10 days until most of the single colonies contained >50 cells per colony. The growth medium was refreshed every three days. After 10 days of growth, the colonies were then stained with 0.25% crystal violet for at least 15 min at room temperature, washed three times with phosphate-buffered solution (PBS), and dried at room temperature. The number of colonies was then counted under a light microscope (magnification, ×100).
Fluorescence-activated cell sorting analysis (FACS)
To validate the cell cycle progression and apoptotic ratio, flow cytometry was performed. Liver cancer cells were seeded at a density of 2×105 cells per dish. After culturing for 48 h (37°C, 5% CO2), cells were harvested and fixed in 75% ethanol overnight at 4°C. The cells were then stained with 50 µg/ml propidium iodine (PI) solution and incubated at 4°C for 1 h in the incubator. PI-stained cells were then analyzed by flow cytometry (FACSCalibur, BD Biosciences). Cell cycle analysis was then performed using the accompanying flow cytometric software (CellQuest Pro, BD Biosciences).
Hoechst 33258 staining
Hoechst 33258 staining kit (Dojindo Molecular Technologies, Inc.) was used to detect changes in cellular apoptosis. Cells were cultured in 12-well plates and incubated at 37°C for 24 h. Then reagents were then added and cells were incubated at 37°C for another 24 h. The cells were then fixed in 4% methanol and permeabilized for 20 min at 4°C and stained with Hoechst 33258 reagent at room temperature for 15 min. After washing three times with PBS, the morphological changes of liver cancer cells and nuclear chromatin agglutination were validated with a fluorescence microscope (magnification, ×400). The images were then acquired and documented.
Mitochondrial apoptosis detection
A fluorometric mitochondrial apoptosis detection kit was purchased from BioVision, Inc. (cat. no. K250). Cells (2×105 cells/ml) were seeded in 12-well plates and incubated at 37°C for 24 h. After incubation with reagents at 37°C for another 24 h, the MitoCapture reagent diluted at 1:1,000 in pre-warmed incubation buffer was then added. Cells were incubated with the MitoCapture working solution in the incubator at 37°C for 20 min and washed with pre-warmed incubation buffer. After washing, cells were visualized and documented with fluorescence microscopy (magnification, ×40).
Wound healing assay
Liver cancer cells were seeded in 6-well plates and cultured until confluent, generally after 24 h. A 100-µl pipette tip was used to produce a straight scratch to simulate a wound. Then, the scratch was washed with pre-warmed PBS to remove cell debris. After washing, cells were cultured in DMEM which contained 1% FBS in order to inhibit cell proliferation. Images were captured under a light microscope (magnification, ×40) at 24, 48, and 72 h after wounding.
Transwell assay
A 6.5-mm Transwell with an 8-µm pore polyester membrane insert was used to perform this experiment. Before being applied to the Transwell insert, the Matrigel was pre-cooled on ice, and then 100 µl was used for one chamber. Then, the Transwell inserts were placed in the incubator for 30 min at 37°C to complete precoating. Next, 5×104 cells were seeded in the upper chamber and cultured in DMEM without serum. DMEM with 10% FBS was added to the lower chamber. Cells were administered with chemotherapeutic agents at 37°C for 48 h and then fixed in 4% methanol for 15 min and stained with 0.25% crystal violet for 15 min at room temperature. Subsequently, invasive cells in the lower chamber were visualized and documented with light microscopy (magnification, ×100).
Western blotting
After being treated with drugs for 24 h, the cells were lysed in an ice bath for 30 min in RIPA Lysis Buffer (Solarbio Life Sciences). The lysates were centrifuged at 10,000 × g for 10 min at 4°C and the supernatant was harvested. Protein concentrations of the supernatants were estimated using a DC Protein assay kit (Bio-Rad Laboratories, Inc.). Then, 30 µg protein sample per lane was loaded into the 10% SDS-PAGE which was performed to separate the various proteins by their molecular weight. The separated proteins were then transferred onto Bio-Rad polyvinylidene difluoride membranes in a semi-dry transmembrane device. The transferred membranes were firstly soaked in the blocking solution (with 3% BSA) for 1 h at room temperature and then incubated with corresponding primary antibodies at 4°C overnight. These antibodies which were purchased from Cell Signaling Technology, Inc. included: Bax (1:1,000; product no. 5023), Bcl-2 (1:1,000; product no. 3498), cytochrome c (1:1,000; product no. 4280), Bid (1:1,000; product no. 2002), pro-caspase-3 (1:1,000; product no. 9662), cleaved caspase-3 (1:1,000; product no. 9664), cleaved caspase-8 (1:1,000; product no. 8592), cleaved caspase-9 (1:1,000; product no. 9509), MMP-2 (1:1,000; product no. 40994), MMP-9 (1:1,000; product no. 13667), PI3K (1:1,000; product no. 4249), Akt (1:2,000; product no. 4685), mTOR (1:1,000; product no. 2983), S6K (1:1,000; product no. 9202), and β-actin (1:1,000; product no. 4970). After they were washed with TBST solution three times, the membranes were incubated with HRP-labeled secondary antibody (1:3,000; product no. 7074; Cell Signaling Technology, Inc.) at 37°C for 1 h and then washed with TBST three more times. Finally, protein bands were then detected by an ECL detection kit (cat. no. RPN2209; Cytiva). The protein expression level was analyzed by ImageJ 1.53 (National Institutes of Health).
Statistical analysis
Data are expressed as the mean ± standard deviation (SD). Differences between the two groups were compared using unpaired Student's t-test, while differences between multiple groups were compared by Tukey's post hoc tests following ANOVA, using SPSS 19.0 (IBM Corp.). Results with a P-value <0.05 were considered to indicate a statistically significant difference.
Results
Inhibitive effect on proliferation by KF and DOX in liver cancer cells and toxicity assessment in normal cells
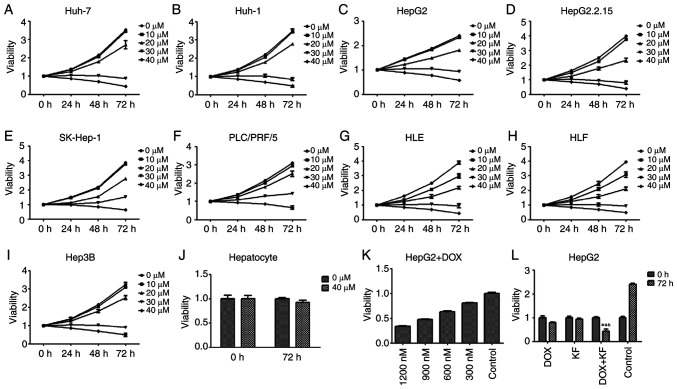
The inhibitive effect of KF on the proliferation of liver cancer cells was assessed by MTT assay. The viability of Huh-7, Huh-1, HepG2, HepG2.2.15, SK-Hep-1, PLC/PRF/5, HLE, HLF, and Hep3B cells treated with increasing concentrations of KF was detected at 24, 48, and 72 h post-treatment. As revealed (Fig. 2A-I), KF had an inhibitive effect on the nine cell lines in time- and dose-dependent manners. After treatment with 40 µM KF for 72 h, the growth inhibition rate of HepG2 cells was 47.15% (Fig. 2C) while in SK-Hep-1 cells it was 52.05% (Fig. 2E). Due to it being close to the IC50 of both cell lines, 40 µM KF was used for future experiments. After treatment with KF for 72 h, the growth inhibitory effect on hepatocytes (Fig. 2J) was not noticeable compared with the control group.
Figure 2.
Growth inhibitory effect of KF and doxorubicin. (A-I) Cells were treated with various concentrations of KF for 24, 48, and 72 h, and cell viability was determined by MTT assay. (J) Normal hepatocytes were treated with 0 and 40 µM of KF and the cell viability was determined by MTT assay. (K) Cells were treated with various concentrations of DOX for 24 h, and cell viability was determined by MTT assay. (L) Four treatment groups (DOX, KF, DOX + KF, and Control) were assessed in cells for 72 h, and cell viability was determined by MTT assay. The data represent the mean ± SD. (n=3). *P<0.05 compared with the DOX group; #P<0.05 compared with the KF group; &P<0.05 compared with the Control group. KF, Kaempferol; DOX, doxorubicin.
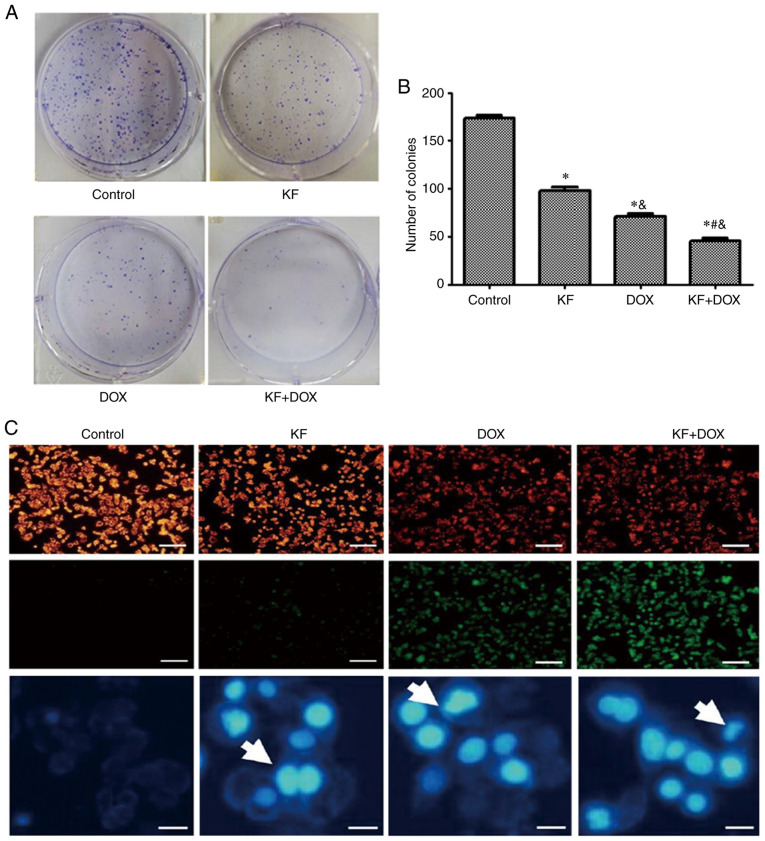
HepG2, which is one of the most commonly used liver cancer cell lines, was used to determine the effect of KF and the combined therapy on cell proliferation and apoptosis in the next experiment. As a positive control, the inhibitory effect of DOX at 24 h was detected in HepG2 cells (Fig. 2K). The effect was also revealed to be in a dose-dependent manner. At a concentration of 900 nM, doxorubicin had a similar inhibitory effect as KF at 40 µM. Four treatment groups (the DOX group, KF group, DOX+KF combination group and control group) were then detected for changes in cellular viability. The result revealed that the combination group had a more substantial inhibitive effect in HepG2 cells (Fig. 2L). The examination of the four treatment groups in HepG2 cells revealed that a combination treatment could significantly inhibit the colony formation of the liver cancer cells (Fig. 3A and B).
Figure 3.
Inhibition of HepG2 cell colony formation, mitochondrial potential disruption, and DNA damage by KF. (A and B) The results revealed that the combination treatment group led to stronger colony formation inhibition. (C) KF and DOX both triggered mitochondrial potential disruption and DNA damage in liver cancer cells (scale bar, 1,000 and 200 µm, respectively). The data represent the mean ± SD (n=3). *P<0.05 compared with the Control group; #P<0.05 compared with the DOX group; &P<0.05 compared with the KF group. KF, Kaempferol; DOX, doxorubicin.
KF and DOX induce apoptosis in HepG2 cells
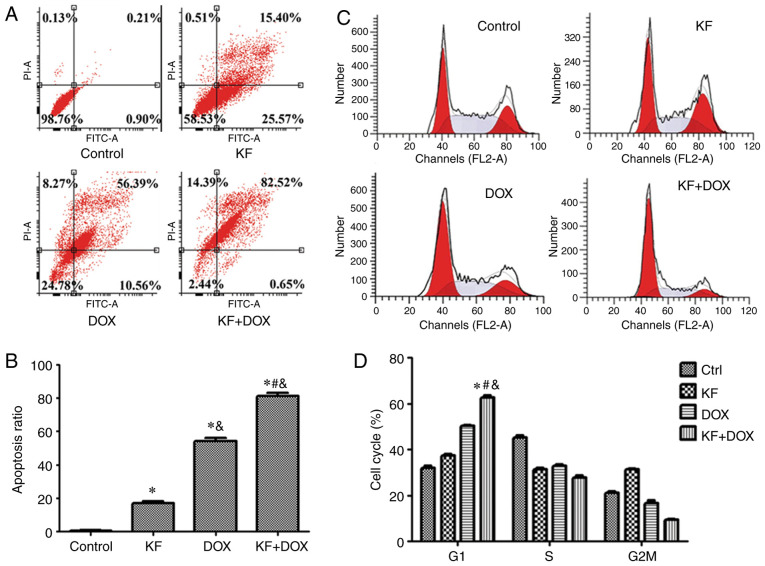
To ascertain whether KF and DOX in combination induce apoptosis in HepG2 cells, mitochondrial potential staining was performed. Marked changes in mitochondrial potential were observed in the combined treatment group as compared with slight changes in the KF group (Fig. 3C). Furthermore, DAPI staining also revealed an increased number of highlighted nuclei, indicative of apoptosis (Fig. 3C). The cellular apoptotic ratio was then measured by Annexin V-FITC/PI double-staining and analyzed by flow cytometry. The percentage of HepG2 cells was significantly increased in the combination group, and in the two monotherapy groups the apoptotic ratio was also observed to be increased compared with the control group (Fig. 4A and B).
Figure 4.
Effect of KF and DOX on cell cycle arrest and apoptosis based on flow cytometry. (A and B) KF, DOX, and combination KF and DOX groups all induced apoptosis in HepG2 cells, as revealed by flow cytometry. The apoptotic ratio of the combination group was markedly higher than the two monotherapy groups. (C and D) Following 48 h of treatment, the four groups of cells were collected to assess the cell cycle distribution. The combination group exhibited higher G1-phase cells than the two monotherapy groups. The data represent the mean ± SD (n=3). *P<0.05 compared to the Control group; #P<0.05 compared to the DOX group; &P<0.05 compared to the KF group. KF, Kaempferol; DOX, doxorubicin; Ctrl, control.
KF and DOX induce cell cycle arrest in HepG2 cells
To investigate whether cell cycle arrest contributed to cell proliferation and colony formation inhibition, the cell cycle of HepG2 cells was analyzed using flow cytometry. Administration of KF and DOX arrested liver cancer cells in the G1 phase and accordingly decreased the cell ratio in the G2/M and S phases (Fig. 4C and D).
Effect of KF on the production of apoptotic proteins in HepG2 cells
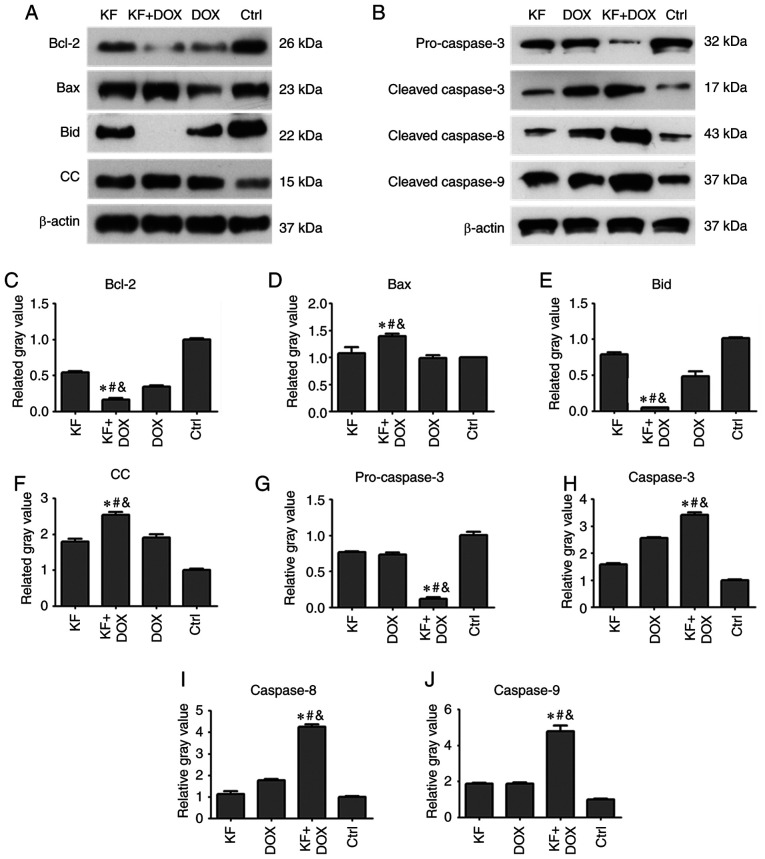
To determine changes in the level of apoptotic proteins impacted by drug treatments in HepG2 cells, the expression levels of apoptotic proteins, such as caspase-3, caspase-8, caspase-9, Bcl-2, Bax, Bid, and cytochrome c were detected by western blotting assay. The cells were separated into four groups: The KF, DOX, combination of DOX and KF group, and control group (Fig. 5A and B). Following a 72-h treatment, protein expression levels of Bcl-2, Bid, and pro-caspase-3 were clearly reduced in the combined treatment group compared with KF and DOX single-treatment groups (Fig. 5C, E and G). The protein expression levels of Bax, cytochrome c (CC), caspase-3, caspase-8, and caspase-9 were upregulated in the combined treatment group compared to KF and DOX single-treatment groups (Fig. 5D, F and H-J).
Figure 5.
Expression level of apoptosis-related proteins following treatment with a combination of drugs in liver cancer cells. The HepG2 cells were treated for 72 h, and then the expression levels of (A and C-F) Bcl-2, Bax, Bid, and cytochrome c and (B and G-J) pro-caspase-3, cleaved caspase-3/8/9 were analyzed by western blotting. The data represent the mean ± SD (n=3). *P<0.05 compared to the KF group; #P<0.05 compared to the DOX group; &P<0.05 compared to the Control group. KF, Kaempferol; DOX, doxorubicin; Ctrl, control; CC, cytochrome c.
KF inhibits migration of SK-Hep-1 cells
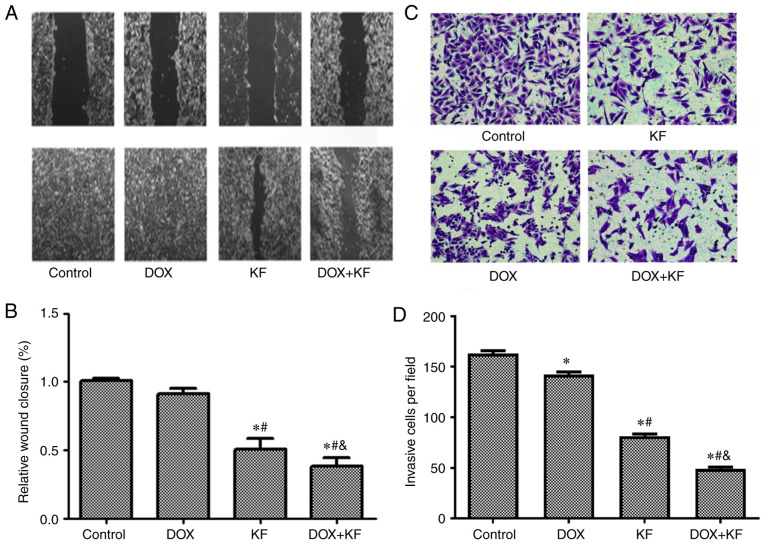
To study the functions of KF on cell migration and invasion, the liver adenocarcinoma cell line, SK-Hep-1, which had a higher level of malignancy was used. The migration was assessed by monolayer scratch wound healing assay. The results revealed impaired wound closure of cells treated with KF and/or DOX. The combination group exhibited the largest opening area from the scratch, while KF treatment alone had a weaker effect than the combined treatment (Fig. 6A and B). This finding indicates that KF alone or in combination with DOX can inhibit the motility of liver cancer cells.
Figure 6.
Inhibitive effect of DOX and KF on the migration and invasion of liver cancer cells. (A and B) In the wound healing experiment, the SK-Hep-1 cells were administered with chemotherapeutic agents for 72 h, and then the changes in the scratched wound width were observed and recorded under a microscope (scale bar, 1,000 µm). Changes in the wound widths are presented in the histograms of B. (C and D) For the Transwell invasion experiment, the SK-Hep-1 cells were administered with chemotherapeutic agents for 48 h, and then the cells were removed and fixed in methanol, stained with cell labeling dye, and recorded under a microscope (Scale bar, 400 µm). The number of invasive cells were presented in the histograms of D. The data represent the mean ± SD (n=3). *P<0.05 compared to the Control group; #P<0.05 compared to the DOX group; &P<0.05 compared to the KF group. DOX, doxorubicin; KF, Kaempferol.
KF inhibits invasion of SK-Hep-1 cells
To further evaluate the function of KF on the invasion capability, Transwell chamber assay was performed using SK-Hep-1 cells. The results from the Transwell invasion experiments indicated that KF alone and combined treatment could restrict the invasive ability of liver cancer cells (Fig. 6C and D) and significantly decrease the number of invasive cells.
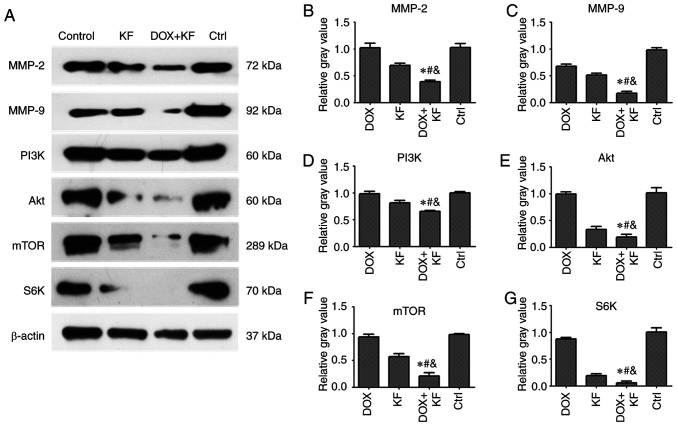
Effect of KF on the expression of migration and invasion-related proteins in SK-Hep-1 cells
To validate the expression of migration and invasion-related proteins affected by KF and the combination group in liver cancer cells, the expression levels of MMP-2, MMP-9, PI3K, Akt, mTOR, and S6K were measured by western blotting assay. There were four groups: The DOX group, KF group, combination of DOX and KF group, and control group. Following treatment for 72 h, downregulated levels of MMP-2, MMP-9, PI3K, Akt, mTOR, and S6K were observed in the combined treatment group compared with the group treated with either KF or DOX alone in SK-Hep-1 cells (Fig. 7A-G).
Figure 7.
Expression level of migration- and invasion-related proteins after being administered with combination therapy in liver cancer cells. (A-G) The SK-Hep-1 cells were administered with chemotherapeutic agents for 72 h, and then the expression levels of MMP-2, MMP-9, PI3K, Akt, mTOR, and S6K were detected by western blotting. The data represent the mean ± SD (n=3). *P<0.05 compared to the DOX group; #P<0.05 compared to the KF group; &P<0.05 compared to the Control group. DOX, doxorubicin; KF, Kaempferol.
Discussion
Liver cancer is one of the most lethal gastrointestinal malignancies worldwide. It is typically characterized by late-stage presentation, which limits treatment options and results in poor prognoses (26). At present, surgery and chemotherapy are the main preferred therapeutic strategies for advanced liver cancer. However, with metastasis, and given the drug-resistant nature of terminal liver cancer, patients with advanced liver cancer are generally not suitable for surgery. In addition, there have been few effective chemotherapeutic agents available for liver cancer treatment (27). To date, sorafenib was the only approved targeted drug for advanced liver cancer, and numerous attempts using other monoclonal antibodies or small-molecule tyrosine kinase inhibitors failed to demonstrate their efficacy (28). Thus, combination chemotherapeutic strategies were investigated to contribute to the development of a more effective treatment for liver cancer. Some studies have reported that traditional Chinese medicine has the ability to inhibit liver cancer growth by inducing apoptosis and suppressing invasion through the blocking of crucial cellular signaling pathways (29,30). KF was reported to be able to inhibit numerous cancers both in vitro and in vivo (31). Hepatocellular carcinoma, as a major cancer worldwide, has also been reported to be inhibited by KF by several research groups. Seydi et al reported that KF exhibited selective cytotoxicity toward hepatocellular carcinoma cells in a rat model (32). Han et al revealed that KF could induce autophagic cell death in hepatocellular carcinoma cells by activating adenosine 5′-monophosphate (AMP)-activated protein kinase (AMPK) signaling (33). Another study indicated that KF induced apoptosis in HepG2 cells via activation of the endoplasmic reticulum stress pathway (34). However, these studies were only performed using monotherapy in liver cancer cells, and no data showing the effects of combination therapy of KF with conventional chemotherapeutic agents were available. Thus, in the present study it was examined whether KF or a combination of KF and DOX could achieve more robust antitumor effects.
In our initial studies, liver cancer cells treated with KF alone exhibited inhibitive effects in time- and dose-dependent manners. KF was then used in combination with DOX. The results indicated that a combination of KF and DOX exhibited a significantly stronger growth inhibition than either KF or DOX alone (P<0.05).
Previous studies have revealed that flavonoids can induce cell death by activating apoptosis and mitochondrial signaling pathways, and inhibit cell migration and invasion by suppressing mTOR signaling pathways (35,36). The results of the present study revealed that KF could induce apoptosis in liver cancer cells by inducing changes in the protein expression levels of critical factors involved in the mitochondrial apoptotic signaling pathways. Furthermore, combination treatment exhibited a more significant effect on apoptotic activity. However, through wound healing and Transwell invasion assays, it was observed that KF exhibited obvious inhibitive activities on liver cancer cells. Combination treatment also revealed higher inhibitive activity on migration and invasion-related proteins (including MMP-2, MMP-9, PI3K, Akt, mTOR, and S6K) than in the case of KF or doxorubicin treatment alone.
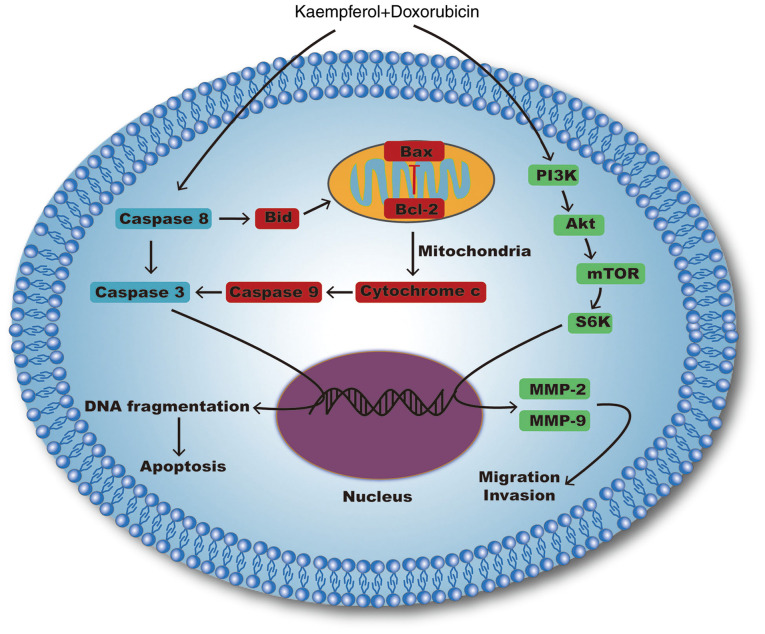
Notably, our research indicated that KF could inhibit the proliferation of liver cancer cells by activating mitochondrial signaling pathways and suppressing migration and invasion by inhibiting the PI3K/mTOR/MMP signaling pathway (Fig. 8). By contrast, KF treatment on normal cells revealed markedly low toxicity, which suggests that KF may be a safer complementary medicine for clinical application. Furthermore, combined treatment with DOX and KF induced a higher inhibitive effect than either of the monotherapies. In a future study, the safety and efficacy of this combined therapy in vivo will be confirmed, in order to determine the theoretical basis on its clinical application. Additionally, the effects of KF combined with other chemotherapeutics will be explored as well. It is theorized that KF as a hypotoxic compound could affect carcinoma cells by interrupting cell survival signaling pathways and could compensate for the shortcomings of the present monotherapies. Moreover, the merit of the multi-targeting effect of KF could lead to a less drug-resistant response. Therefore, KF, as an adjuvant medicine, combined with other chemotherapeutics, even targeted drugs could potentially be a more effective approach to advance the therapeutic outcomes and quality of life of patients with liver cancer.
Figure 8.
Apoptosis, migration, and invasion-related signaling pathways affected by Kaempferol combined with doxorubicin.
Acknowledgements
Not applicable.
Funding Statement
The present study was supported by the Fund Program for the Scientific Activities of Selected Returned Overseas Professionals in Shanxi Province.
Funding
The present study was supported by the Fund Program for the Scientific Activities of Selected Returned Overseas Professionals in Shanxi Province.
Availability of data and materials
The datasets used during the present study are available from the corresponding author upon reasonable request.
Authors' contributions
GY, JS and MZ conceived and designed the experiments. GY and JX performed the experiments. GY and BA analyzed the data. BA, GY, JX and JS contributed to the statistical analysis. GY and JS wrote the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet. 2012;391:1301–1314. doi: 10.1016/S0140-6736(18)30010-2. [DOI] [PubMed] [Google Scholar]
- 2.Anwanwan D, Singh SK, Singh S, Saikam V, Singh R. Challenges in liver cancer and possible treatment approaches. Biochim Biophys Acta Rev Cancer. 2020;1873:188314. doi: 10.1016/j.bbcan.2019.188314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jiao B, Gao JJ. Intensive research on the prospective use of complementary and alternative medicine to treat systemic lupus erythematosus. Drug Discov Ther. 2013;7:167–171. [PubMed] [Google Scholar]
- 4.Huang H, Peng X, Zhong C. Idiopathic pulmonary fibrosis: The current status of its epidemiology, diagnosis, and treatment in China. Intractable Rare Dis Res. 2013;2:88–93. doi: 10.5582/irdr.2013.v2.3.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gao J, Inagaki Y, Li X, Kokudo N, Tang W. Research progress on natural products from traditional Chinese medicine in treatment of Alzheimer's disease. Drug Discov Ther. 2013;7:46–57. [PubMed] [Google Scholar]
- 6.Melendez-Martinez AJ, Nascimento AF, Wang Y, Liu C, Mao Y, Wang XD. Effect of tomato extract supplementation against high-fat diet-induced hepatic lesions. Hepatobiliary Surg Nutr. 2013;2:198–208. doi: 10.3978/j.issn.2304-3881.2013.07.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tao Z, Gao J, Zhang G, Xue M, Yang W, Tong C, Yuan Y. Shufeng Jiedu Capsule protect against acute lung injury by suppressing the MAPK/NF-κB pathway. BioSci Trends. 2014;8:45–51. doi: 10.5582/bst.8.45. [DOI] [PubMed] [Google Scholar]
- 8.Qi FH, Wang ZX, Cai PP, Zhao L, Gao JJ, Okudo N, Li AY, Han JQ, Tang W. Traditional Chinese medicine and related active compounds: A review of their role on hepatitis B virus infection. Drug Discov Ther. 2013;7:212–24. doi: 10.5582/ddt.2013.v7.6.212. [DOI] [PubMed] [Google Scholar]
- 9.Xia J, Inagaki Y, Gao J, Qi F, Song P, Han G, Sawakami T, Gao B, Luo C, Kokudo N, et al. Combination of Cinobufacini and Doxorubicin increases apoptosis of hepatocellular carcinoma cells through the Fas- and mitochondria-mediated pathways. Am J Chin Med. 2017;45:1537–1556. doi: 10.1142/S0192415X17500835. [DOI] [PubMed] [Google Scholar]
- 10.Xia J, Rong L, Sawakami T, Inagaki Y, Song P, Hasegawa K, Sakamoto Y, Tang W. Shufeng Jiedu Capsule and its active ingredients induce apoptosis, inhibit migration and invasion, and enhances doxorubicin therapeutic efficacy in hepatocellular carcinoma. Biomed Pharmacother. 2018;99:921–930. doi: 10.1016/j.biopha.2018.01.163. [DOI] [PubMed] [Google Scholar]
- 11.Wong R, Sagar CM, Sagar SM. Integration of Chinese medicine into supportive cancer care: A modern role for an ancient tradition. Cancer Treat Rev. 2001;27:235–246. doi: 10.1053/ctrv.2001.0227. [DOI] [PubMed] [Google Scholar]
- 12.Holland TM, Agarwal P, Wang Y, Leurgans SE, Bennett DA, Booth SL, Morris MC. Dietary flavonols and risk of Alzheimer dementia. Neurology. 2020;29:e1749–e1756. doi: 10.1212/WNL.0000000000008981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Veeresham C, Rama Rao A, Asres K. Aldose reductase inhibitors of plant origin. Phytotherapy Research. 2014;28:317–333. doi: 10.1002/ptr.5000. [DOI] [PubMed] [Google Scholar]
- 14.Khalil MI, Sulaiman SA. The potential role of honey and its polyphenols in preventing heart diseases: A review. Afr J Tradit Complement Altern Med. 2010;7:315–321. doi: 10.4314/ajtcam.v7i4.56693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen AY, Chen YC. A review of the dietary flavonoid, kaempferol on human health and cancer chemoprevention. Food Chem. 2013;138:2099–2107. doi: 10.1016/j.foodchem.2012.11.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Niestroy J, Barbara A, Herbst K, Rode S, van Liempt M, Roos PH. Single and concerted effects of benzo[a]pyrene and flavonoids on the AhR and Nrf2-pathway in the human colon carcinoma cell line Caco-2. Toxicol In Vitro. 2011;25:671–683. doi: 10.1016/j.tiv.2011.01.008. [DOI] [PubMed] [Google Scholar]
- 17.Calderón-Montaño JM, Burgos-Morón E, Pérez-Guerrero C, López-Lázaro M. A review on the dietary flavonoid kaempferol. Mini Rev Med Chem. 2011;11:298–344. doi: 10.2174/138955711795305335. [DOI] [PubMed] [Google Scholar]
- 18.Kim SH, Choi KC. Anti-cancer effect and underlying mechanism(s) of kaempferol, a phytoestrogen, on the regulation of apoptosis in diverse cancer cell models. Toxicol Res. 2013;29:229–234. doi: 10.5487/TR.2013.29.4.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Speth PA, van Hoesel QG, Haanen C. Clinical pharmacokinetics of doxorubicin. Clin Pharmacokinet. 1988;15:15–31. doi: 10.2165/00003088-198815010-00002. [DOI] [PubMed] [Google Scholar]
- 20.Chatterjee K, Zhang J, Honbo N, Karliner JS. Doxorubicin cardiomyopathy. Cardiology. 2010;115:155–162. doi: 10.1159/000265166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kaczmarek A, Brinkman BM, Heyndrickx L, Vandenabeele P, Krysko DV. Severity of doxorubicin-induced small intestinal mucositis is regulated by the TLR-2 and TLR-9 pathways. J Pathol. 2012;226:598–608. doi: 10.1002/path.3009. [DOI] [PubMed] [Google Scholar]
- 22.Bun S, Yunokawa M, Tamaki Y, Shimomura A, Shimoi T, Kodaira M, Shimizu C, Yonemori K, Fujiwara Y, Makino Y, et al. Symptom management: The utility of regional cooling for hand-foot syndrome induced by pegylated liposomal doxorubicin in ovarian cancer. Support Care Cancer. 2018;26:2161–2166. doi: 10.1007/s00520-018-4054-z. [DOI] [PubMed] [Google Scholar]
- 23.Lin J, Fang L, Li H, Li Z, Lyu L, Wang H, Xiao J. Astragaloside IV alleviates doxorubicin induced cardiomyopathy by inhibiting NADPH oxidase derived oxidative stress. Eur J Pharmacol. 2019;859:172490. doi: 10.1016/j.ejphar.2019.172490. [DOI] [PubMed] [Google Scholar]
- 24.Li J, Wu Y, Wang D, Zou L, Fu C, Zhang J, Leung GP. Oridonin synergistically enhances the anti-tumor efficacy of doxorubicin against aggressive breast cancer via pro-apoptotic and anti-angiogenic effects. Pharmacol Res. 2019;146:104313. doi: 10.1016/j.phrs.2019.104313. [DOI] [PubMed] [Google Scholar]
- 25.Xu Z, Chen L, Xiao Z, Zhu Y, Jiang H, Jin Y, Gu C, Wu Y, Wang L, Zhang W, et al. Potentiation of the anticancer effect of doxorubicinin drug-resistant gastric cancer cells by tanshinone IIA. Phytomedicine. 2018;51:58–67. doi: 10.1016/j.phymed.2018.05.012. [DOI] [PubMed] [Google Scholar]
- 26.Caldwell S, Park SH. The epidemiology of hepatocellular cancer: From the perspectives of public health problem to tumor biology. Journal of Gastroenterology. 2009;44(Suppl 19):S96–S101. doi: 10.1007/s00535-008-2258-6. [DOI] [PubMed] [Google Scholar]
- 27.El-Serag HB, Marrero JA, Rudolph L, Reddy KR. Diagnosis and treatment of hepatocellular carcinoma. Gastroenterology. 2008;134:1752–1763. doi: 10.1053/j.gastro.2008.02.090. [DOI] [PubMed] [Google Scholar]
- 28.Gnoni A, Santini D, Scartozzi M, Russo A, Licchetta A, Palmieri V, Lupo L, Faloppi L, Palasciano G, Memeo V, et al. Hepatocellular carcinoma treatment over sorafenib: Epigenetics, microRNAs and microenvironment. Is there a light at the end of the tunnel? Expert Opin Ther Targets. 2015;19:1623–1635. doi: 10.1517/14728222.2015.1071354. [DOI] [PubMed] [Google Scholar]
- 29.Han G, Xia J, Gao J, Inagaki Y, Tang W, Kokudo N. Anti-tumor effects and cellular mechanisms of resveratrol. Drug Discov Ther. 2015;9:1–12. doi: 10.5582/ddt.2015.01007. [DOI] [PubMed] [Google Scholar]
- 30.Anand David AV, Arulmoli R, Parasuraman S. Overviews of biological importance of quercetin: A bioactive flavonoid. Pharmacogn Rev. 2016;10:84–89. doi: 10.4103/0973-7847.194044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ren J, Lu Y, Qian Y, Chen B, Wu T, Ji G. Recent progress regarding kaempferol for the treatment of various diseases. Exp Ther Med. 2019;18:2759–2776. doi: 10.3892/etm.2019.7886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Seydi E, Salimi A, Rasekh HR, Mohsenifar Z, Pourahmad J. Selective cytotoxicity of luteolin and kaempferol on cancerous hepatocytes obtained from Rat model of hepatocellular carcinoma: Involvement of ROS-mediated mitochondrial targeting. Nutr Cancer. 2018;70:594–604. doi: 10.1080/01635581.2018.1460679. [DOI] [PubMed] [Google Scholar]
- 33.Han B, Yu YQ, Yang QL, Shen CY, Wang XJ. Kaempferol induces autophagic cell death of hepatocellular carcinoma cells via activating AMPK signaling. Oncotarget. 2017;8:86227–86239. doi: 10.18632/oncotarget.21043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Guo H, Ren F, Zhang L, Zhang X, Yang R, Xie B, Li Z, Hu Z, Duan Z, Zhang J. Kaempferol induces apoptosis in HepG2 cells via activation of the endoplasmic reticulum stress pathway. Mol Med Rep. 2016;13:2791–2800. doi: 10.3892/mmr.2016.4845. [DOI] [PubMed] [Google Scholar]
- 35.Huang WW, Chiu YJ, Fan MJ, Lu HF, Yeh HF, Li KH, Chen PY, Chung JG, Yang JS. Kaempferol induced apoptosis via endoplasmic reticulum stress and mitochondria-dependent pathway in human osteosarcoma U-2 OS cells. Mol Nutr Food Res. 2010;54:1585–1595. doi: 10.1002/mnfr.201000005. [DOI] [PubMed] [Google Scholar]
- 36.Zhu G, Liu X, Li H, Yan Y, Hong X, Lin Z. Kaempferol inhibits proliferation, migration, and invasion of liver cancer HepG2 cells by down-regulation of microRNA-21. Int J Immunopathol Pharmacol. 2018;32:2058738418814341. doi: 10.1177/2058738418814341. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used during the present study are available from the corresponding author upon reasonable request.