Abstract
Enlargements of distal airspaces can indicate pathological changes in the lung, but accessible and precise techniques able to measure these regions are lacking. Airspace Dimension Assessment with inhaled nanoparticles (AiDA) is a new method developed for in vivo measurement of distal airspace dimensions. The aim of this study was to benchmark the AiDA method against quantitative measurements of distal airspaces from hyperpolarised 129Xe diffusion-weighted (DW)-lung magnetic resonance imaging (MRI). AiDA and 129Xe DW-MRI measurements were performed in 23 healthy volunteers who spanned an age range of 23–70 years. The relationship between the 129Xe DW-MRI and AiDA metrics was tested using Spearman’s rank correlation coefficient. Significant correlations were observed between AiDA distal airspace radius (rAiDA) and mean 129Xe apparent diffusion coefficient (ADC) (p < 0.005), distributed diffusivity coefficient (DDC) (p < 0.001) and distal airspace dimension (LmD) (p < 0.001). A mean bias of − 1.2 µm towards rAiDA was observed between 129Xe LmD and rAiDA, indicating that rAiDA is a measure of distal airspace dimension. The AiDA R0 intercept correlated with MRI 129Xe α (p = 0.02), a marker of distal airspace heterogeneity. This study demonstrates that AiDA has potential to characterize the distal airspace microstructures and may serve as an alternative method for clinical examination of the lungs.
Subject terms: Diagnostic markers, Chronic obstructive pulmonary disease, Diagnostic devices, Imaging techniques and agents
Introduction
Detection of microstructural changes in the distal airspaces can be crucial for clinical evaluation of early stage lung disease and longitudinal monitoring of pulmonary diseases. The standard procedures for detection of disease in the distal airspaces include spirometry, test of diffusing capacity of the lung for carbon monoxide (DLCO), and lung density analysis from computed tomography (CT). However, all the mentioned techniques can fail to reveal early indications of pathological changes1,2. Therefore, considerable disease can be present with minimal effect on standard pulmonary function tests (PFTs).
Diffusion-weighted (DW) magnetic resonance imaging (MRI) with inhaled hyperpolarised noble gases helium-3 (3He) or xenon-129 (129Xe) is an in vivo imaging method that is sensitive to changes in the distal airspaces3–5. The method is based on measurement of the Brownian diffusional restriction of the inhaled hyperpolarised gas atoms within the distal airspace walls. This property is used to derive the apparent diffusion coefficient (ADC), which provides 3D in vivo information on the distal airspace microstructure. In addition to ADC, theoretical models of hyperpolarised gas diffusion within the lungs, such as the stretched exponential model (SEM)6,7 and the cylinder airway model (CM)8,9, can be used to derive distal airspace dimensions, analogous to those obtained through histological analysis. These in vivo distal airspace measurements from hyperpolarised gas DW-MRI have shown good agreement with direct morphometric measurements in validation studies with lung specimens8,10,11. Numerous studies have used hyperpolarised gas DW-MRI to elucidate changes in distal airspaces related to smoking12–14, ageing15, lung inflation16, and diseases such as COPD17,18, asthma19, and idiopathic pulmonary fibrosis (IPF)20. However, due to the high cost and complex infrastructure required for hyperpolarised gas MRI, the technique is not typically employed in standard pulmonary function testing or screening for pulmonary disease.
Airspace Dimension Assessment (AiDA) with inhaled nanoparticles, could potentially be more cost efficient and more widely accessible than MRI with hyperpolarised gases. The method is based on measurement of the exhaled recovery of inhaled nanoparticles which deposit in the distal airspaces due to Brownian diffusion21. The fraction of deposited particles is directly related to the size of the airspaces, and analysis of the nanoparticle recovery yields two metrics. The first metric is an effective airspace radius (rAiDA), which is a root mean square measure of a collection of airspaces, and the second metric is the recovery at an imaginary zero-seconds breath-hold (R0). rAiDA has been found to correlate with the extent of emphysema22 and proton lung tissue density as quantified by standard pulmonary structural MRI23. R0 significantly correlates with the carbon monoxide transfer coefficient (KCO) and age24. Further benchmarking of the technique with established methods of in vivo distal airspace assessment is required to evaluate the clinical potential of the AiDA technique.
The aim of this study was to benchmark inhaled nanoparticle measurements with AiDA against 129Xe DW-MRI derived ADC and distal airspace dimensions. Since both AiDA21 and 129Xe DW-MRI 6 use diffusion of nanoparticles and gas molecules, we hypothesise that measures from the two different techniques will correlate.
Results
Volunteer demographics, pulmonary function test data, DW-MRI metrics and AiDA variables
Table 1 summarises volunteer demographics, pulmonary function test results, hyperpolarised 129Xe DW-MRI metrics and AiDA values for the 23 volunteers.
Table 1.
Summary of volunteer demographics and global measurements.
| Characteristic | Mean ± SD | Median (IQR) | Min | Max |
|---|---|---|---|---|
| Age (years) | 48 ± 17 | 54 (34) | 23 | 70 |
| Height (cm) | 173.7 ± 9.8 | 175 (15) | 155 | 191 |
| Weight (kg) | 75.5 ± 14.4 | 77 (24) | 55 | 103 |
| FEV1 (% pred) | 96.0 ± 9.7 | 95.8 (10.9) | 80.1 | 115.4 |
| VC (% pred) | 96.5 ± 12.4 | 94.8 (10.3) | 74.7 | 132.4 |
| TLC (% pred) | 97.9 ± 11.6 | 96.8 (14.5) | 80.3 | 128.2 |
| RV (% pred) | 102.5 ± 24.8 | 102.2 (27.6) | 57 | 182 |
| FRC (% pred) | 93.3 ± 16.8 | 93.4 (25.0) | 57.6 | 116.5 |
| DLCO (% pred) | 102.8 ± 18.8 | 101.3 (23.7) | 67.5 | 153.7 |
| KCO (% pred) | 113.2 ± 16.2 | 112 (21.4) | 85 | 150.3 |
| ADC (cm2/s) | 0.034 ± 0.004 | 0.034 (0.0041) | 0.0263 | 0.0434 |
| α (a.u.) | 0.862 ± 0.015 | 0.863 (0.023) | 0.826 | 0.889 |
| DDC (cm2/s) | 0.030 ± 0.004 | 0.030 (0.005) | 0.022 | 0.030 |
| LmD (µm) | 281 ± 19 | 277 (20) | 244 | 322 |
| rAiDA (µm) | 279 ± 25 | 284 (42) | 240 | 325 |
| R0 (a.u.) | 0.484 ± 0.132 | 0.51 (0.21) | 0.183 | 0.675 |
Data are presented as mean ± SD, median (IQR), minimum values and maximum values.
IQR interquartile range, FEV1 forced expiratory volume in 1 s, VC vital capacity, TLC total lung capacity, RV residual volume, FRC forced respiratory capacity; DLCO diffusing capacity of lung for carbon monoxide, KCO carbon monoxide transfer coefficient, ADC apparent diffusion coefficient, α heterogeneity index, DDC distributed diffusivity coefficient, LmD mean diffusive length scale, rAiDA distal airspace radius, R0 zero-second recovery.
Benchmarking of AiDA using the DW-MRI measurements
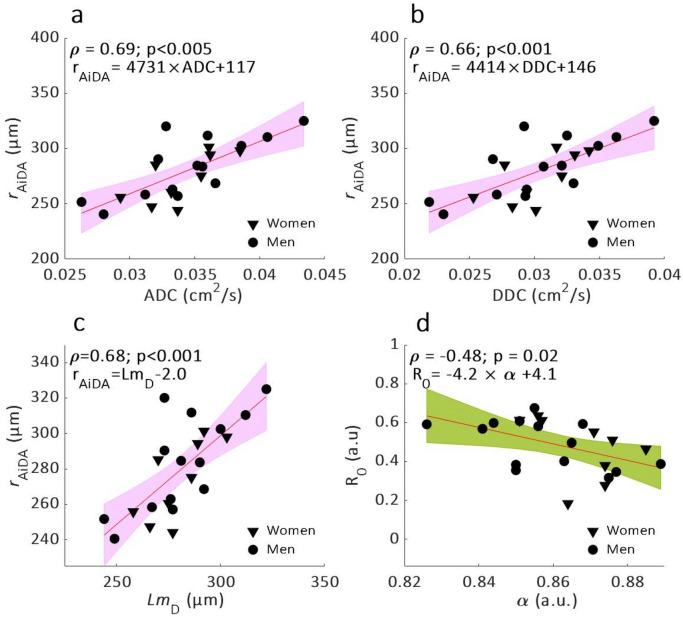
Statistically significant linear correlations were observed between rAiDA and 129Xe DW-MRI metrics ADC (p < 0.005), DDC (p < 0.001) and LmD (p < 0.001) (Fig. 1a–c). The R0 intercept measurement from AiDA was significantly correlated with 129Xe α heterogeneity index (p = 0.02) (Fig. 1d), but not with any of the other 129Xe DW-MRI metrics. Bland–Altman analysis of rAiDA and 129Xe LmD showed a mean bias of − 1.2 µm (95% agreement limits − 36.0 to 33.6 µm) towards rAiDA (Fig. 2). rAiDA and LmD deviated less than 0.05% for the whole group and on average 8% between subjects.
Figure 1.
Linear regressions (with 95% confidence intervals in color) and Spearman’s correlation (ρ) of rAiDA as a function of 129Xe DW-MRI metrics: (a) ADC, (b) DDC, (c) LmD and of R0 as a function of α heterogeneity index (d).
Figure 2.
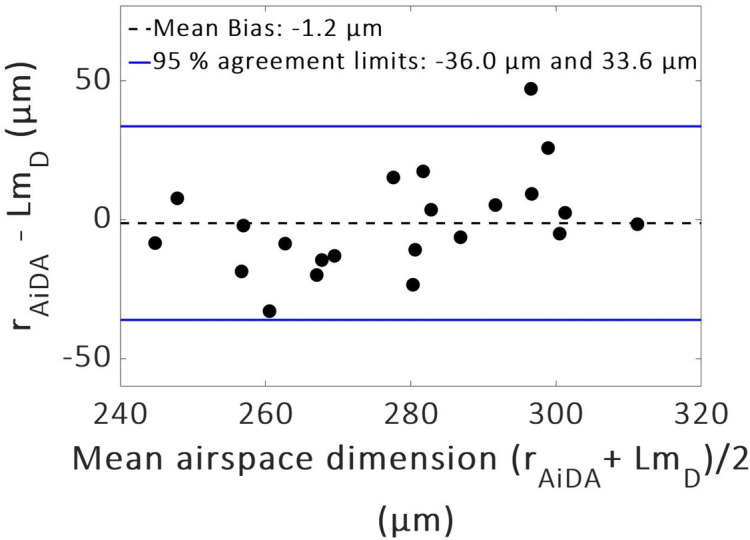
Bland–Altman plot of distal airspace radius rAiDA and mean diffusive length scale LmD. A mean bias of 1.2 µm towards rAiDA and 95% agreement limits − 36.0 to 33.6 µm were found.
For the comparison in the Bland–Altman plot, a linear relationship of strength ρ = 0.48 was observed (p = 0.02) suggesting rAiDA increased more than LmD with increasing airspace size. No difference was seen between men and women for the AiDA and 129Xe DW-MRI metric distributions.
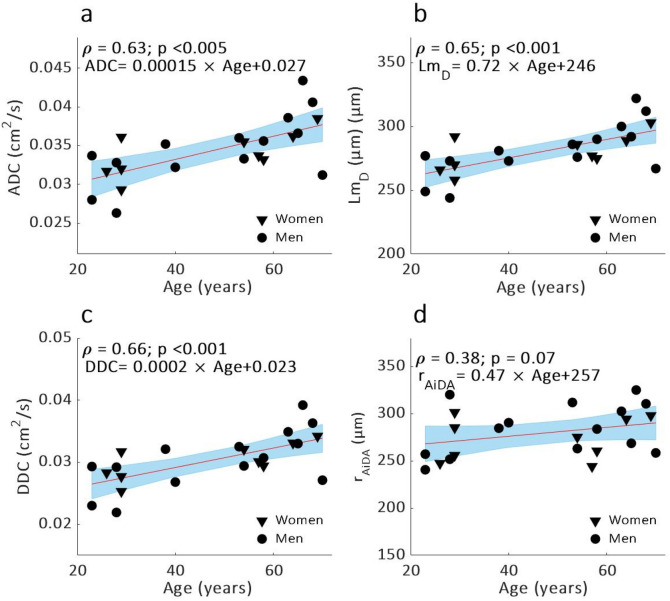
Significant correlations were observed between volunteer age 129Xe DW-MRI metrics ADC (p < 0.005), DDC (p < 0.001) and LmD (p < 0.001) (Fig. 3a–c). A trend towards increased rAiDA with age was observed (p = 0.07) (Fig. 3d). A positive trend between R0 and FEV1 was observed (p = 0.07). AiDA and 129Xe DW-MRI variables did not correlate significantly with any of the PFT measurements.
Figure 3.
Linear regressions (with 95% confidence intervals in blue) and Spearman’s correlation (ρ) of (a) ADC, (b) LmD, (c) DDC and (d) rAiDA as a function of age.
Discussion
Measurements of distal airspace dimensions were acquired with AiDA and 129Xe DW-MRI in 23 healthy volunteers. AiDA metrics, rAiDA and R0, corresponded to previously reported values from healthy volunteers22–24. 129Xe DW-MRI metrics of ADC, DDC and distal airspace dimensions (LmD) were larger than the values previously reported for younger healthy volunteers (29 ± 4 years), but smaller than values in ex-smoker volunteers6.
Distal airspace radius rAiDA, significantly correlated with 129Xe DW-MRI metrics ADC, DDC and LmD, which confirm to us that rAiDA is a measure of distal airspace dimensions. Agreement between rAiDA and 129Xe DW-MRI LmD was confirmed with Bland–Altman analysis where a mean bias of − 1.2 µm towards rAiDA, corresponding to 0.43% deviation from the mean measured rAiDA, was observed. Both AiDA and 129Xe DW-MRI measure the Brownian motion, presumably within the same distal airspace. The breath-hold times and diffusion times were optimised specifically for diffusion across small distances but assume that the particles stay within the same airway duct. The statistically significant correlation between R0 and α heterogeneity index (p = 0.02) could be indicating that R0 represents heterogeneity in the acini, which both metrics are hypothesized to measure6,24.
The ADC, DDC and LmD dependency on age suggests a prevalence of age-related distal airspace changes in the older healthy volunteers25. The significant correlations between volunteer age and 129Xe ADC and LmD further demonstrate the sensitivity of DW-MRI to age-related airspace changes, and matches trends previously observed with 3He DW-MRI15,26. A trend towards increasing rAiDA with age was observed. α heterogeneity correlated with volunteer height (p < 0.001). AiDA and 129Xe DW-MRI metrics did not correlate with any other volunteer demographic or PFT data.
The bias and relatively wide 95% agreement limits (− 36.0 to 33.6 µm) observed in the Bland–Altman plot can be explained by the difference in acquisition method of the two techniques. AiDA is acquired over multiple breath-holds with diffusion times ranging between 5 and 15 s. In contrast, 129Xe DW-MRI is a single breath-hold acquisition with an 8.5 ms diffusion time. The linear trend in bias visible in the Bland Altman plot (Fig. 2) suggests that the agreement between rAiDA and LmD changes with increasing mean airspace dimension size. This implies that the measurements from the two methods might diverge for increasing distal airspaces with an increasing bias between rAiDA and LmD in larger airspaces. The increasing bias may be attributed to the difference between the diffusion coefficients for xenon (0.14 cm2/s when mixed with air in the lungs27 and the 50 nm nanoparticles (2 × 10–5 cm2/s28). Due to the much smaller mass and size of 129Xe-atoms they diffuse much faster than the nanoparticles. Hence, the exponential decay, from which the diffusion distance in the lung is calculated, is different for the two techniques. For 129Xe DW-MRI the theoretical 1D free diffusion length () is approximately 500 µm ( 8.5 ms, cm2/s) while for 50 nm nanoparticles the corresponding displacement in one direction is approximately 200 µm ( 10 s, 2 × 10−5 cm2/s).
While the 129Xe DW-MRI method provides voxel-wise regional information about the lung structure the AiDA method provides one measure of the distal airspace dimensions. Therefore, the 129Xe DW-MRI method can give more detailed and regional information about the structure of the distal airspaces compared to AiDA. However, as indicated by this study, the AiDA method has the potential to give a faster and more accessible, but still precise, measurement of the distal airspace dimensions, which can be of great importance when hyperpolarised lung MRI is not available. To further compare the relative sensitivity between the two methods more measurements, in particular including a large variation in distal space sizes, are needed.
Although the two methods obtained approximately similar airspace dimensions (average rAiDA and LmD deviated less than 0.5%), there were also several significant differences in the measurement procedures for the subjects. 129Xe DW-MRI measurements were made at FRC + 1 L while AiDA measurements were made at TLC. Hence, the lung inflation was larger for all AiDA measurements as compared to 129Xe DW-MRI measurements. Previous studies have shown that measured ADC in hyperpolarised 3He DW-MRI increases with increasing lung inflation volumes29. Which means that, AiDA could be expected to have measured larger dimensions rAiDA when compared to LmD.
Distal airspace size also depends on posture and ADC decreases from the non-dependent region of the lung down to the dependent region. For hyperpolarised 3He DW-MRI measurements, ADC has been found to vary significantly depending on posture, and this was attributed to the compression of parenchyma, due to the lungs own weight, and the mass of the heart30. In this study AiDA measurements were performed in upright sitting position while the 129Xe DW-MRI measurements were performed in supine position. Therefore, a postural variation between whole lungs AiDA measurement and the regionally averaged 129Xe DW-MRI metrics is expected. For a more elaborate comparison of the two methods the breath-hold volumes could be set to be equal and breath-hold times set to correspond to the same diffusion distances. In addition, AiDA could potentially be measured with subjects in a supine posture enabling an even more efficient comparison with minimized systematic errors.
AiDA measurements have been shown to be repeatable to approximately < 2.4% when measured at different times over a period of 18 months31. Previous studies have shown that 3He and 129Xe ADC is highly repeatable in COPD patients with a coefficient of variation of 2.98% and 2.77% respectively, over 5 visits32. Similarly it has been showed that 3He LmD in patients with idiopathic pulmonary fibrosis is highly repeatable with a 0.6% difference between same-day visits20.
This is the first study that compares AiDA with an independent and validated non-invasive method for assessment of distal airspaces. The study includes a limited number of healthy subjects, which mainly was due to the logistics related to travel between Sweden and the UK. The subject group was thus homogenous with small variations in PFT results. However, even with the small group of volunteers, significant correlations were found between AiDA and 129Xe DW-MRI metrics, and interestingly these increased with age indicating age dependent changes in alveolar size or ‘aging emphysema’25. Ideally, a future extension of this study would include subjects with a greater range of distal airspace sizes, such as patients with emphysematous lung disease.
In conclusion, this work has compared estimates of airspace radii from inhaled nanoparticles by the AiDA method with 129Xe DW-MRI in a healthy volunteer cohort. The significant correlations show that the distal airspace radius of the lungs measured by AiDA (rAiDA) can be related to distal airspace microstructure dimensions as quantified by 129Xe DW-MRI. Quantitavely the two methods are in close agreement, with mean rAiDA and LmD deviating < 0.5% for the whole group and on average 8% on an individual level. Further benchmarking in selected groups of patients could be used to evaluate the relative sensitivity of AiDA and 129Xe DW-MRI in detecting early emphysematous changes to the distal airspace microstructure.
Methods
Study subjects and study design
The study enrolled 23 healthy adult volunteers (14M, 9F) in the age range 23–70 years with no history of pulmonary disease, in Sweden in the spring 2019. The study was approved by the Regional Ethical Review Board in Lund, Sweden (application number 2018/659), and performed in accordance with the Declaration of Helsinki, including obtaining informed written consent from all volunteers. All volunteers underwent spirometry, body plethysmography, carbon monoxide gas transfer, and AiDA measurements at Skåne University Hospital in Malmö, Sweden. Pulmonary function tests were performed according to the European Respiratory Society guidelines33. All 129Xe DW-MRI measurements were performed at University of Sheffield, Sheffield, UK.
AiDA measurements
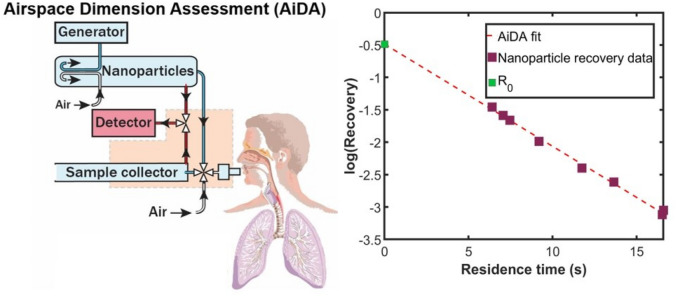
The AiDA method and instrumentation has been described in detail elsewhere31. Figure 4 displays a schematic illustration of the AiDA instrument23 and example data from one subject.
Figure 4.
(Left) Schematic illustration of the AiDA method. The aerosol particles from the distal airspaces were sampled to obtain the distal airspace radius rAiDA and the zero-second recovery R0. (Right) Representative recovery data as a function of residence time in the lungs and AiDA fit obtained for one volunteer.
In short, each subject initially inhaled particle-free air to remove background particles from their lungs. The subject was then instructed to exhale to residual volume (RV) prior to inhaling 50 nm polystyrene latex (PSL) aerosol particles to total lung capacity (TLC), hold their breath for a predefined time and finally exhale. For each measurement, the subject sat upright and a nose clip prevented them from breathing through the nose. Inhaled and exhaled nanoparticle number concentrations were registered for 8 consecutive measurements, with breath-hold times between 5 and 15 s. The inhaled nanoparticle concentration employed for AiDA was less than 10,000 cm−3, which is lower than the concentration of ambient nanoparticles in an urban environment34.
The probability of particle deposition in the distal airspaces depends on residence time and airspace size. Enlarged airspaces yield a lower deposited fraction, corresponding to a higher particle recovery () in exhaled gas. The estimation of airspace dimensions using AiDA is based on the solution of the diffusion equation in circular tubes randomly distributed with axisymmetric boundary conditions21. The solution shows that the recovery R decays exponentially with residence time in the lung35 according to:
where is the recovery at zero-second breath-hold and and t1/2 is the deposition half-life time. Residence time t was established and linear least-squares regression was fitted to the data. From the fit, R0 and were determined. R0 is presumably related to the dynamic phase of breathing and small conducting airways (generation 10–1536), but remains to be evaluated further21,24. The airspace radius rAiDA, was calculated from and the diffusion coefficient () for 50 nm particles according to:
129Xe DW-MRI measurements
Hyperpolarised 129Xe DW-MRI was performed on a GE HDx 1.5T scanner with a flexible transmit/receive quadrature vest coil (Clinical MR Solutions, Brookfield, WI, USA) after the inhalation of a 1 L gas mixture containing 550 mL 129Xe (> 25% polarization37) and 450 mL N2 from the level of function residual capacity. A 3D multiple b-value spoiled gradient echo (SPGR) sequence with compressed sensing was used with a 16 s breath-hold6. Specific DW-MRI acquisition parameters were: TE/TR = 14.0/17.3 ms, 129Xe diffusion time = 8.5 ms, b = [0, 12, 20, 30 s/cm2]. 129Xe ADC was calculated using a mono-exponential fit between the signal of the b = 0 (S0) and 12 (Sb=12) s/cm2 interleaves:
The mean diffusive length scale (LmD), a measure of mean distal airspace dimension from the SEM was derived by fitting the 129Xe diffusion signal from all four b-values to a stretched exponential function6:
where is the probability distribution of different apparent diffusivities within each image voxel, DDC is the distributed diffusivity coefficient, and α is the heterogeneity index that describes the deviation from a mono-exponential decay (α = 1). α is thought to be a measure of the underlying complexity or heterogeneity of the geometry of the restricting distal boundaries38. From α and DDC a numerical expression for is estimated39. Further, is related to diffusion length scales associated with different apparent diffusivities (D) by the characteristic diffusion length ( diffusion time) which represent a measure of the distribution of microscopic dimensions. LmD, a measure of mean distal airspace dimensions, is defined as the expectation value of the probability distribution of diffusion length and is related to by:
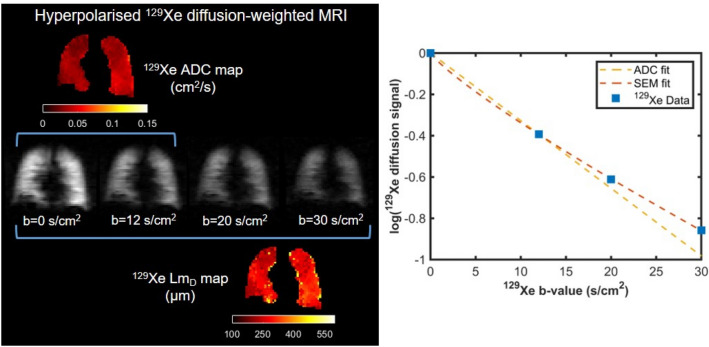
Both ADC and SEM metrics were calculated on a voxel-by-voxel basis, and averaged across the entire lung volume to derive global means. Figure 5 displays examples of ADC and SEM-derived LmD maps from the hyperpolarised 129Xe DW-MRI.
Figure 5.
Example hyperpolarised 129Xe diffusion-weighted (DW) MRI data from one volunteer. (Left) Maps of ADC and LmD values calculated from 129Xe DW-MRI. (Right) Representative global 129Xe signal as a function of b-value, ADC- and SEM-fits obtained for the same volunteer.
Benchmarking of AiDA versus DW-MRI measurements
All of the AiDA analysis, 129Xe DW-MRI lung morphometry calculations and comparisons of metrics were implemented using MATLAB Version 2020a (The MathWorks, Inc., Natick, Massachusetts, United States). Correlations between AiDA variables, 129Xe DW-MRI metrics and standard PFT measurements were assessed using Spearman’s rank correlation test. The significance threshold was set at 0.05. Bland–Altman analysis was used to assess the agreement between the rAiDA and LmD.
Acknowledgements
The authors would like to acknowledge Haris Zilic, for performing the clinical lung function tests and AiDA measurements at Skåne University Hospital. The authors would also like to thank all members of the POLARIS research group at the University of Sheffield for their support. In particular, the author would like to thank Paul Hughes for assisting with MRI scanning, and Oliver Rodgers for polarisation of 129Xe during MRI.
Author contributions
The study was conceived, planned and designed by M.P.S., H.F.C., J.L., P.W., L.E.O. and J.M.W. Experiments were planned, designed and performed by M.P.S., H.F.C., G.J.C. and G.N. with substantial support from J.L., P.W. and J.M.W. Data analysis was performed by M.P.S. and H.F.C. M.P.S., H.F.C., G.J.C., G.N., J.L., P.W., L.E.O. and J.M.W. interpreted and analysed the results. M.P.S. and H.F.C. prepared the figures and wrote the manuscript with substantial input from all other authors. All authors were involved in several rounds of critically revising the manuscript and approved the submitted version.
Funding
Open access funding provided by Lund University. This work was supported by the Swedish Heart and Lung foundation (Grant No 2017-0644, Grant No 2018-0483 and Grant No 2020-0855) and the Swedish Research Council for Health, Working Life and Welfare (Grant No 2017-00690) and NanoLund. This work was supported by National Institute for Health Research grant (NIHR-RP-R3-12-027) and Medical Research Council Grant (MR/M008894/1). The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research or the Department of Health.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
Prof. P. Wollmer received personal fees from Chiesi Pharmaceuticals during the conduct of the study. In addition, Prof. P Wollmer and Assoc. Prof. J. Löndahl have a patent for “Device and Method for pulmonary function measurements” issued. The other authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Madeleine Petersson-Sjögren and Ho-Fung Chan.
These authors jointly supervised this work: Jakob Löndahl and Jim M. Wild.
References
- 1.Fain SB, et al. Early emphysematous changes in asymptomatic smokers: Detection with 3He MR imaging. Radiology. 2006;239:875–883. doi: 10.1148/radiol.2393050111. [DOI] [PubMed] [Google Scholar]
- 2.Shaker SB, et al. The extent of emphysema in patients with COPD. Clin. Respir. J. 2009;3:15–21. doi: 10.1111/j.1752-699X.2008.00102.x. [DOI] [PubMed] [Google Scholar]
- 3.Saam BT, et al. MR imaging of diffusion of (3)He gas in healthy and diseased lungs. Magn. Reson. Med. 2000;44:174–179. doi: 10.1002/1522-2594(200008)44:2<174::aid-mrm2>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- 4.Salerno M, Altes TA, Brookeman JR, de Lange EE, Mugler JP., 3rd Dynamic spiral MRI of pulmonary gas flow using hyperpolarized (3)He: Preliminary studies in healthy and diseased lungs. Magn. Reson. Med. 2001;46:667–677. doi: 10.1002/mrm.1244. [DOI] [PubMed] [Google Scholar]
- 5.Kaushik SS, et al. Diffusion-weighted hyperpolarized 129Xe MRI in healthy volunteers and subjects with chronic obstructive pulmonary disease. Magn. Reson. Med. 2011;65:1154–1165. doi: 10.1002/mrm.22697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chan HF, Stewart NJ, Norquay G, Collier GJ, Wild JM. 3D diffusion-weighted Xe-129 MRI for whole lung morphometry. Magn. Reson. Med. 2018;79:2986–2995. doi: 10.1002/mrm.26960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chan HF, Stewart NJ, Parra-Robles J, Collier GJ, Wild JM. Whole lung morphometry with 3D multiple b-value hyperpolarized gas MRI and compressed sensing. Magn. Reson. Med. 2017;77:1916–1925. doi: 10.1002/mrm.26279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yablonskiy DA, et al. Quantification of lung microstructure with hyperpolarized 3He diffusion MRI. J. Appl. Physiol. 2009;107:1258–1265. doi: 10.1152/japplphysiol.00386.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sukstanskii AL, Yablonskiy DA. Lung morphometry with hyperpolarized 129Xe: theoretical background. Magn. Reson. Med. 2012;67:856–866. doi: 10.1002/mrm.23056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chan, H. F., Collier, G. J. & Wild, J. M. Validation of theoretical models of hyperpolarized gas diffusion MRI with finite element simulation in geometrical and realistic models of lung acinar airways from micro-CT. In: Proceedings of the 27th Annual Meeting of ISMRM, Montréal, Canada; 2019. Abstract 1878.
- 11.Woods JC, et al. Hyperpolarized 3He diffusion MRI and histology in pulmonary emphysema. Magn. Reson. Med. 2006;56:1293–1300. doi: 10.1002/mrm.21076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Quirk JD, et al. In vivo detection of acinar microstructural changes in early emphysema with (3)He lung morphometry. Radiology. 2011;260:866–874. doi: 10.1148/radiol.11102226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Paulin GA, et al. Noninvasive quantification of alveolar morphometry in elderly never- and ex-smokers. Physiol. Rep. 2015 doi: 10.14814/phy2.12583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Swift AJ, et al. Emphysematous changes and normal variation in smokers and COPD patients using diffusion 3He MRI. Eur. J. Radiol. 2005;54:352–358. doi: 10.1016/j.ejrad.2004.08.002. [DOI] [PubMed] [Google Scholar]
- 15.Quirk JD, et al. Experimental evidence of age-related adaptive changes in human acinar airways. J. Appl. Physiol. 2016;1985(120):159–165. doi: 10.1152/japplphysiol.00541.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hajari AJ, et al. Morphometric changes in the human pulmonary acinus during inflation. J. Appl. Physiol. 2012;1985(112):937–943. doi: 10.1152/japplphysiol.00768.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ouriadov A, et al. Lung morphometry using hyperpolarized (129) Xe apparent diffusion coefficient anisotropy in chronic obstructive pulmonary disease. Magn. Reson. Med. 2013;70:1699–1706. doi: 10.1002/mrm.24595. [DOI] [PubMed] [Google Scholar]
- 18.Ouriadov A, Lessard E, Sheikh K, Parraga G. Pulmonary MRI morphometry modeling of airspace enlargement in chronic obstructive pulmonary disease and alpha-1 antitrypsin deficiency. Magn. Reson. Med. 2018;79:439–448. doi: 10.1002/mrm.26642. [DOI] [PubMed] [Google Scholar]
- 19.Wang C, et al. Assessment of the lung microstructure in patients with asthma using hyperpolarized 3He diffusion MRI at two time scales: Comparison with healthy subjects and patients with COPD. J. Magn. Reson. Imaging. 2008;28:80–88. doi: 10.1002/jmri.21408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chan HF, et al. Airway microstructure in idiopathic pulmonary fibrosis: Assessment at hyperpolarized (3)He diffusion-weighted MRI. Radiology. 2019;291:223–229. doi: 10.1148/radiol.2019181714. [DOI] [PubMed] [Google Scholar]
- 21.Londahl J, Jakobsson JKF, Broday DM, Aaltonen HL, Wollmer P. Do nanoparticles provide a new opportunity for diagnosis of distal airspace disease? Int. J. Nanomed. 2017;12:41–51. doi: 10.2147/Ijn.S121369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Aaltonen H, et al. Deposition of inhaled nanoparticles is reduced in subjects with COPD and correlates with the extent of emphysema: Proof of concept for a novel diagnostic technique. Clin. Physiol. Funct. Imaging. 2018;38:1008–1014. doi: 10.1111/cpf.12517. [DOI] [PubMed] [Google Scholar]
- 23.Aaltonen HL, et al. Airspace dimension assessment with nanoparticles reflects lung density as quantified by MRI. Int. J. Nanomed. 2018;13:2989–2995. doi: 10.2147/IJN.S160331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jakobsson J, Wollmer P, Löndahl J. Charting the human respiratory tract with airborne nanoparticles: Evaluation of the airspace dimension assessment technique. BMC J. Appl. Physiol. 2018;125:1832–1840. doi: 10.1152/japplphysiol.00410.2018. [DOI] [PubMed] [Google Scholar]
- 25.Gillooly M, Lamb D. Airspace size in lungs of lifelong non-smokers: Effect of age and sex. Thorax. 1993;48:39–43. doi: 10.1136/thx.48.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fain SB, et al. Detection of age-dependent changes in healthy adult lungs with diffusion-weighted 3He MRI. Acad. Radiol. 2005;12:1385–1393. doi: 10.1016/j.acra.2005.08.005. [DOI] [PubMed] [Google Scholar]
- 27.Chen XJ, et al. Spatially resolved measurements of hyperpolarized gas properties in the lung in vivo. Part I: Diffusion coefficient. Magn. Reson. Med. 1999;42:721–728. doi: 10.1002/(SICI)1522-2594(199910)42:4<721::AID-MRM14>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 28.Hinds WC. Aerosol Technology: Properties, Behavior, and Measurement of Airborne Particles. 2. Hoboken: Wiley; 1999. [Google Scholar]
- 29.Halaweish AF, et al. Effect of lung inflation level on hyperpolarized 3He apparent diffusion coefficient measurements in never-smokers. Radiology. 2013;268:572–580. doi: 10.1148/radiol.13120005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fichele S, et al. MRI of helium-3 gas in healthy lungs: Posture related variations of alveolar size. J. Magn. Reson. Imaging. 2004;20:331–335. doi: 10.1002/jmri.20104. [DOI] [PubMed] [Google Scholar]
- 31.Jakobsson JKF, Hedlund J, Kumlin J, Wollmer P, Löndahl J. A new method for measuring lung deposition efficiency of airborne nanoparticles in a single breath. Sci. Rep. 2016;6:36147. doi: 10.1038/srep36147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Stewart NJ, et al. Comparison of (3) He and (129) Xe MRI for evaluation of lung microstructure and ventilation at 1.5T. J. Magn. Reson. Imaging. 2018;48:632–642. doi: 10.1002/jmri.25992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Quanjer PH, et al. Lung volumes and forced ventilatory flows. Report working party standardization of lung function tests, european community for steel and coal. Official statement of the European Respiratory Society. Eur. Respir. J. Suppl. 1993;16:5–40. doi: 10.1183/09041950.005s1693. [DOI] [PubMed] [Google Scholar]
- 34.Kumar P, Robins A, Vardoulakis S, Britter R. A review of the characteristics of nanoparticles in the urban atmosphere and the prospects for developing regulatory controls. Atmos. Environ. 2010;44:5035–5052. doi: 10.1016/j.atmosenv.2010.08.016. [DOI] [Google Scholar]
- 35.Goldberg IS, Smith RB. Settling and diffusion of aerosol particles in small airways during breath holding. Ann. Biomed. Eng. 1981;9:557–575. doi: 10.1007/BF02364771. [DOI] [PubMed] [Google Scholar]
- 36.Weibel ER. Morphometry of the Human Lung. Berlin: Springer-Verlag; 1963. [Google Scholar]
- 37.Norquay G, Collier GJ, Rao M, Stewart NJ, Wild JM. 129Rb spin-exchange optical pumping with high photon efficiency. Phys. Rev. Lett. 2018;121:153201. doi: 10.1103/PhysRevLett.121.153201. [DOI] [PubMed] [Google Scholar]
- 38.Parra-Robles, J., Marshall, H. & Wild, J. M. Characterization of 3He diffusion in lungs using a stretched exponential model. In: Proceedings of the 21st Annual Meeting of ISMRM, Salt Lake City, UT; 2013. Abstract 820.
- 39.Berberan-Santos MN, Bodunov EN, Valeur B. Mathematical functions for the analysis of luminescence decays with underlying distributions 1. Kohlrausch decay function (stretched exponential) Chem. Phys. 2005;315:171–182. doi: 10.1016/j.chemphys.2005.04.006. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.