Abstract
Non-sebaceous lymphadenoma of the salivary glands is a rare benign lesion, first described in 1991. We present the case of a 54-year-old woman, with a right parotid mass. She underwent right superficial parotidectomy, and histopathology reported a non-sebaceous lymphadenoma due to an encapsulated lesion and multiple non-atypical epithelial inclusions without sebaceous differentiation. The etiology of non-sebaceous lymphadenoma is not yet understood, but it can arise predominantly from the parotid gland. Surgical excision is the treatment of choice.
Keywords: Parotid tumor, non-sebaceous lymphadenoma, parotid
Introduction
Non-sebaceous lymphadenoma (NSL) of the salivary glands is a rare benign lesion characterized by predominantly lymphoid background, with embedded solid nondescript or squamous epithelial nests without sebaceous differentiation.1 Few cases of NSL have been reported in the literature.1-4 Herein, we report a case of a 54-year-old woman with a parotid mass treated with superficial parotidectomy, with a final diagnosis of NSL. A review of the literature is also performed.
Presentation of Case
A 54-year-old woman with no medical history presented to the clinic with a 2-month history of pain and mild swelling of the right cheek. On physical examination, a right-sided soft non-mobile parotid gland tumor of 3 cm × 3 cm was palpated, with no neurological affection. An ultrasound (US) and computed tomographic (CT) scan revealed a 23 mm × 19 mm × 17 mm parotid tumor with well-circumscribed and isoechoic texture without any associated lymphadenopathy (Figure 1). With initial clinical and imaging diagnosis as pleomorphic adenoma, she underwent right superficial parotidectomy, with facial nerve preservation (Figure 2). The patient was discharged 2 days after her surgical procedure without any complications. Histopathology reported a well-circumscribed tumor of 3.6 cm × 2.2 cm × 1.6 cm, surrounded by normal parotid tissue, white yellowish color, and some cystic spaces (Figure 3A). The tumor was concordant with an NSL due to an encapsulated lesion and, on a lymphoid background, multiple non-atypical epithelial inclusions, without sebaceous differentiation (Figure 3B). The epithelial inclusions had 2 types of disposition; the most frequent was in the form of cysts with 1 to 3 cells in thickness and eosinophilic fluid with histiocytes in its interior (Figure 4A). Few solid nests of epithelial cells were also identified; in addition, some areas with keratin were found (Figure 4B). The rest of the parenchyma of the gland showed no lesion (Figure 5). Absence of nodal capsule was reported.
Figure 1.
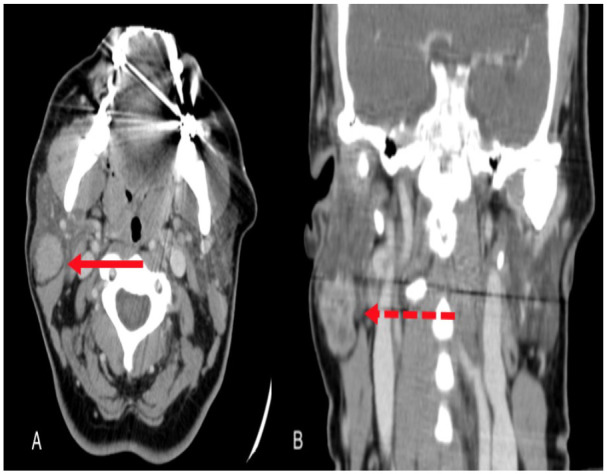
Arrow showing the parotid tumor on computed tomographic scan with contrast: (A) axial view and (B) coronal view.
Figure 2.
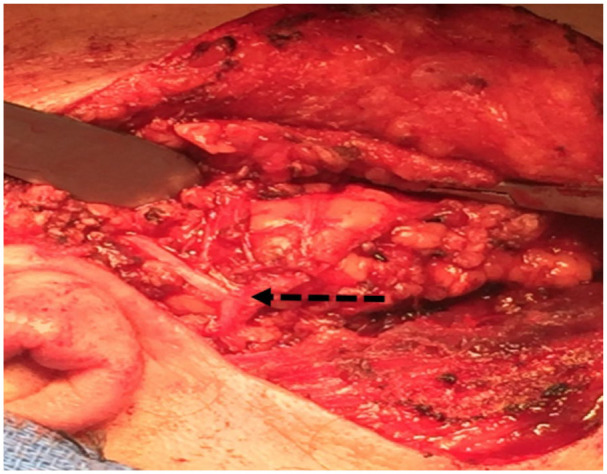
Superficial parotidectomy with facial nerve preservation (dashed arrow).
Figure 3.
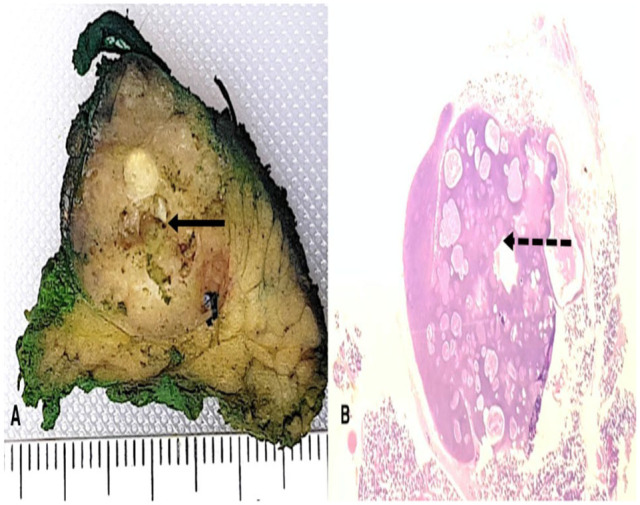
(A) Well-circumscribed tumor (arrow) surrounded by normal parotid tissue. (B) Encapsulated lesion, lymphoid background, non-atypical epithelial inclusions, and without sebaceous differentiation (dashed arrow).
Figure 4.
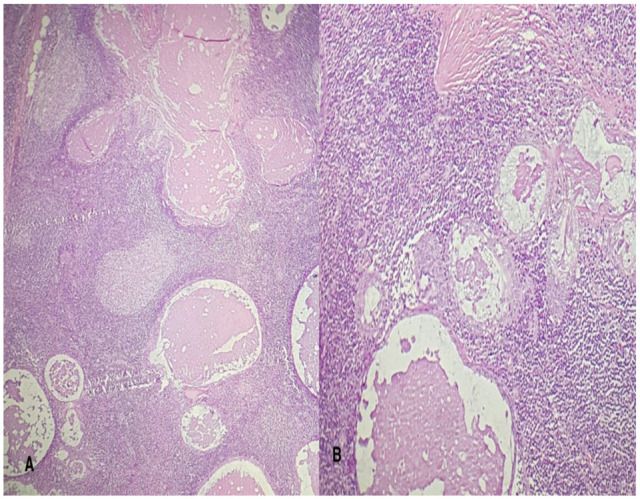
(A) Cellular background composed of lymphoid tissue and cyst with eosinophilic fluid. (B) Cyst with eosinophilic fluid surrounded by 1 to 3 epithelial cells.
Figure 5.
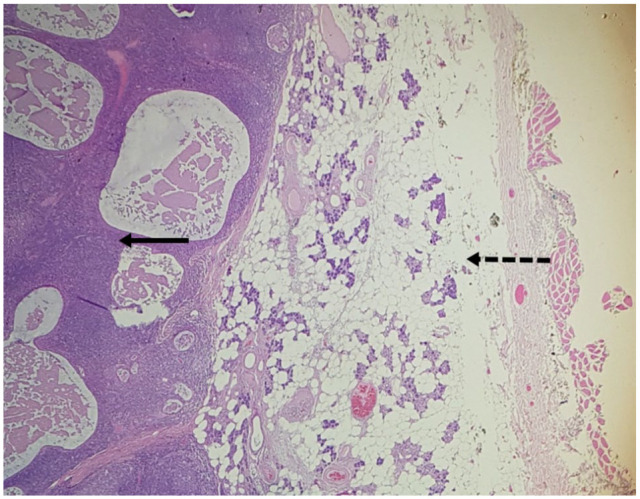
Tumor (arrow) and normal parotid tissue (dashed arrow).
Discussion
Non-sebaceous lymphadenomas are described as benign non-Warthin lymphoid stroma-rich tumors with inconstant mixtures of ductal, squamous, and myoepithelial components, and the central morphologic distinction is the lack of sebaceous parts. The mean age of presentation is 48 years, with a rare malignant transformation.4-6 The etiology of this clinical entity is not well known, but there are 2 theories about its development: the first has been suggested to be embryonic salivary gland inclusions in the intraparotid or periparotid lymph nodes;3 the second theory addresses the lymphoid element as reactive tumor–associated lymphoid tissue.7 In our patient, due to its well-circumscribed tumor surrounded by normal parotid tissue, we believe that the embryonic inclusions theory fits best to our case.
This tumor can arise from any salivary gland. In a series of 33 patients with lymphadenoma tumor, most of these tumors arose in the parotid gland (n = 26, 79%), followed by the neck (n = 3, 9%) and the submandibular gland and the oral cavity, 2 each (6%). Of these tumors, 30% were NSLs, with a remarkable female preponderance on this group, making it comparable to our patient who was a woman in her mid-50s.8
The clinical findings include a painless, well-defined, slow-growing mass, with symptoms ranging from months to several years.2,8 Approximately 80% of salivary gland tumors are benign and appear in the parotid gland; if a rapid growth is encountered, along with pain or cranial nerve involvement, a malignant tumor should be suspected.9 The primary diagnostic test in an adult with persistent neck mass should be contrast-enhanced CT, which offers information regarding the size, extent, location, content, and consistency of the mass; US is beneficial to distinguish cystic from solid lesions and helpful in guiding fine needle aspiration (FNA) biopsy/cytology.9
Fine needle aspiration biopsy/cytology is a frequently performed diagnostic test in the assessment of a parotid mass, with a specificity of 98% and sensitivity of 78% in differentiating benign from malignant parotid tumors and with an overall accuracy of 95%. The benefit of this technique is that can be performed in the ambulatory setting; furthermore, it is a safe procedure, with a low cost and minimum risk.10-12
The cytopathological findings of FNA included hypercellular smears with tight fragments of epithelial cells with basaloid phenotype, showing unchanging round to oval and naked nuclei, missing nucleoli, and association with metachromatic myxoid stroma or basement membrane-like material; numerous lymphocytes, cords, and strands of stromal matrix often formed a branching pattern with no myoepithelial cells or cells with oncocytic or squamous diversity present.4 On our patient, we did not perform an FNA because the original diagnosis on imaging was a pleomorphic adenoma of the parotid gland.
The histological findings include a well-confined, encapsulated tumor, with proliferated epithelium and noticeable lymphoid component; the epithelial element took the form of anastomosing trabeculae, solid tubules, glands, or solid basaloid islands with or without cyst formation, presenting solid nests. Squamous differentiation with keratinization is an infrequent finding. The lymphocytic infiltrate compromise mixed populations of B and T lymphocytes. The growth pattern of the epithelial element generally lacks resemblance to other benign salivary gland tumors; lymphadenoma is thus a form of adenoma with an absence of sebaceous differentiation but an obvious lymphocytic infiltrate.6,8,10,12-14
The diagnostic criteria for NSL were first established by Dardick and Thomas6; they proposed 5 characteristic findings, including (1) non-sebaceous differentiation; (2) non-oncocytic epithelium; (3) prevalent lymphocytic component with or without germinal centers; (4) solid, glandular, or cystic epithelial nests; and (5) well-defined tumor with an absence of nodal capsule or subcapsular sinusoids. All of these criteria were fulfilled by our patient. Also due to academic purposes, we performed BerEP4 staining to all tissue blocks, and it showed no sebaceous differentiation. The differential diagnoses of NSL include Warthin tumor, lymphoepithelial sialadenitis, chronic sialadenitis, benign lymphoepithelial cysts, lymphoepithelial carcinoma, low-grade mucoepidermoid carcinoma, acinic cell carcinoma, metastatic neoplasms, and malignant lymphoma.4
The immunohistochemical findings are p63 highlighting the nuclei of the cells in the outer portion of the glands, trabeculae, and islands; S100 protein and 34ßE12 immunostaining showed an alike pattern of positive cells. Some tumors are positive for HMWK, CK 5/6, calponin, SMA, and SMMS and negative for Ki-67. The lymphoid cells included a combination of CD20+ B cells and CD3+ T cells.1,3,14
Although there is no propensity for NSL to invade adjacent structures, complete surgical excision is the treatment of choice, with superficial parotidectomy and facial nerve preservation as the standard diagnostic-therapeutic procedure. The prognosis is excellent with a low recurrence rate.3,13,15 To our knowledge, only 1 case has been reported with a transformation to malignant NSL.16
In our patient, complete excision was accomplished without any vascular or nerve injury, and final diagnosis was achieved upon pathology report. Most of these cases are first reported by radiology as tumors with characteristics of pleomorphic adenomas, which are the most common tumors from the parotid gland. Therefore, surgeons often do not require an FNA to perform a superficial parotidectomy because most of these tumors are benign. Similar to our case, NSL is quite often a definitive histopathology diagnosis. It is a rare finding with important relevance to literature and recognition within the parotid tumor classification.
In conclusion, NSL of the salivary gland is a rare benign lesion. The primary morphologic difference is the lack of sebaceous components, and malignant transformation rarely occurs. Diagnosis could be done by clinical findings, CT, US, and FNA biopsy/cytology, and the treatment of choice is surgical excision. We believed that these rare cases should continue to be reported because their clinical and radiological presentation varies. In addition, its contribution to academic and scientific environment allows knowledge of these pathologies, allowing a precise definitive diagnosis in pathology reports.
Footnotes
Funding:The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions: CAMM—conception of the work, drafting of manuscript, final approval and agreement for the accountability of work; MG-U—data interpretation, revising the manuscript, final approval and agreement for the accountability of work; AERM—data interpretation, revising the manuscript, final approval and agreement for the accountability of work; ABQ and EAGH—data interpretation, revising the manuscript, final approval and agreement for the accountability of work.
Ethical Approval: All procedures performed in studies involving human participants were in accordance with the ethical standards of Tecnologico de Monterrey Ethics Committee and Institutional Review Board number 122 and has therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed Consent: Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
ORCID iDs: Mauricio Gonzalez-Urquijo  https://orcid.org/0000-0001-5101-1541
https://orcid.org/0000-0001-5101-1541
Eduardo Alberto Guzmán Huerta  https://orcid.org/0000-0001-6750-2548
https://orcid.org/0000-0001-6750-2548
References
- 1. Gallego L, Junquera L, Fresno MF. Non-sebaceous lymphadenoma of the parotid gland: immunohistochemical study and DNA ploidy analysis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107:555-558. doi: 10.1016/j.tripleo.2008.11.018. [DOI] [PubMed] [Google Scholar]
- 2. Yang S, Chen X, Wang L, Zhang J. Non-sebaceous lymphadenoma of the salivary gland: case report with immunohistochemical investigation. Virchows Arch. 2007;450:595-599. [DOI] [PubMed] [Google Scholar]
- 3. Weiler C, Agaimy A, Zengel P, Zenk J, Kirchner T, Ihrler S. Nonsebaceous lymphadenoma of salivary glands: proposed development from intraparotid lymph nodes and risk of misdiagnosis. Virchows Arch. 2012;460:467-472. [DOI] [PubMed] [Google Scholar]
- 4. Castelino-Prabhu S, Li QK, Ali SZ. Nonsebaceous lymphadenoma of the parotid gland: cytopathologic findings and differential diagnosis. Diagn Cytopathol. 2010;38:137-140. [DOI] [PubMed] [Google Scholar]
- 5. Seethala RR, Stenman G. Update from the 4th edition of the World Health Organization classification of head and neck tumours: tumors of the salivary gland. Head Neck Pathol. 2017;11:55-67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Dardick I, Thomas MJ. Lymphadenoma of parotid gland: two additional cases and a literature review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105:491-494. [DOI] [PubMed] [Google Scholar]
- 7. Ishii A, Kawano H, Tanaka S, et al. Non-sebaceous lymphadenoma of the salivary gland with serous acinic cell differentiation, a first case report in the literature. Pathol Int. 2013;63:272-276. [DOI] [PubMed] [Google Scholar]
- 8. Seethala RR, Thompson LDR, Gnepp DR, et al. Lymphadenoma of the salivary gland: clinicopathological and immunohistochemical analysis of 33 tumors. Mod Pathol. 2012;25:26-35. doi: 10.1038/modpathol.2011.135. [DOI] [PubMed] [Google Scholar]
- 9. Haynes J, Arnold KR, Aguirre-Oskins C, Chandra S. Evaluation of neck masses in adults. Am Fam Physician. 2015;91:698-706. [PubMed] [Google Scholar]
- 10. Liu CC, Jethwa AR, Khariwala SS, Johnson J, Shin JJ. Sensitivity, specificity, and posttest probability of parotid fine-needle aspiration: a systematic review and meta-analysis. Otolaryngol Head Neck Surg. 2016;154:9-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Salgarelli AC, Cappare P, Bellini P, Collini M. Usefulness of fine-needle aspiration in parotid diagnostics. Oral Maxillofac Surg. 2009;13:185-190. [DOI] [PubMed] [Google Scholar]
- 12. Gudmundsson JK, Ajan A, Abtahi J. The accuracy of fine-needle aspiration cytology for diagnosis of parotid gland masses: a clinicopathological study of 114 patients. 2016;24:561-567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Gnepp DR. My journey into the world of salivary gland sebaceous neoplasms. Head Neck Pathol. 2012;6:101-110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Ma J, Chan JKC, Chow CW, Orell SR. Lymphadenoma: a report of three cases of an uncommon salivary gland neoplasm. Histopathology. 2002;41:342-350. [DOI] [PubMed] [Google Scholar]
- 15. To VSH, Chan JYW, Tsang RKY, Wei WI. Review of salivary gland neoplasms. ISRN Otolaryngol. 2012;2012:1-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Kara H, Sönmez S, Bağbudar S, Güllüoğlu M, Başaran B. Malignant transformation of parotid gland non-sebaceous lymphadenoma: case report and review of literature. Head Neck Pathol. 2020;14:1123-1128. doi: 10.1007/s12105-020-01133-3. [DOI] [PMC free article] [PubMed] [Google Scholar]


