Abstract
Child abuse is linked to lifetime psychopathology including abnormal self-processing. Given self-processing maturation in adolescence, we tested duration, presence, and abuse accumulation's impact upon self-processing neurobiology among depressed youth with (N = 54) and without an abuse history (N = 40). Youth evaluated positive and negative self-descriptors across four points of view in the scanner. Regression analyses showed that longer abuse duration (in days) was associated with lower activity in inferior temporal (e.g. insula, fusiform & parahippocampus), striatal, cerebellar and midbrain structures when processing negative self-descriptors with the least activity in youth exposed to 6+ abuse years. Abuse presence vs. absence was linked to higher neural activity. However, youth exposed to a single abuse instance to 3 years of abuse might drive that relative neural hyperactivity. Results support: 1) the toxic stress model of blunted overall neuro-reactivity underpinning emotion, sensorimotor gating, and social cognition during negative stimuli as an adaptation to pervasively toxic environments and 2) the differential impact of acute versus chronic stress upon neurophysiological indices. Finally, child abuse duration might impact these ancillary and higher socioemotional processes differently among depressed youth primarily for negative but not positive self-processing.
Keywords: Self-processing, Child abuse, Adolescence, Hippocampus, Neuroimaging
1. Introduction
1.1. Self-processing and abuse
Each year, more than 6 million children in the United States are referred to Child Protective Services for abuse or neglect (Fang et al., 2012). A history of child abuse results in more severe and chronic depression (Beth S. Brodsky et al., 2001). Child abuse is longitudinally linked to manifold negative outcomes including treatment-resistant depression, obesity, teen motherhood, substance abuse, blunted stress response, dissociation, and lifetime psychopathology including PTSD (Russo and Nestler, 2013; Trickett et al., 2011). It is linked to abnormal self-processing manifested as intractable negative and/or incoherent self-representations, including severe dissociative symptoms (Bennett et al., 2010), as well as impaired cognitive functions (Hart and Rubia, 2012). As such, abuse likely alters the neural circuitry of self-processing, which recruits regions that support both cognitive and emotional functions, including cortical midline structures and limbic areas (Alarcón et al., 2019; Nejad et al., 2013). This represents a candidate mechanism of treatment-resistant depression pursuant to child abuse from the perspective of cognitive models of depression (Iacoviello et al., 2006).
Negative self-processing is defined as exaggerated encoding, attention, and recall of negative self-related information and neglect of positively valenced self-related information (Harkness, 2008; Quevedo et al., 2017). Both depression and childhood maltreatment are associated with rigidly negatively biased self-processing (Feiring et al., 2010; Tanaka et al., 2011). Although negative self-processing characterizes depression, it is unknown what neural substrates underpin negative self-processes among depressed maltreated individuals or how abuse characteristics such as duration or cumulative adversity impact those neural substrates. Identifying such neural substrates is important because negative self-processing is a potent risk factor for onset and endurance of depression (Iacoviello et al., 2006) and might underpin treatment-resistant depression experienced by adolescents with history of maltreatment.
Adolescence is a neuroplastic period for self-processing maturation (Mills et al., 2014) due to the emergence of abstract thinking enabled by prefrontal cortex maturation, and neurodevelopment during this period culminates in the emergence of stable global cognitive self-representations (Harter, 1999). Understanding how abuse impacts adolescents’ self-processing circuitry may help explain divergent mental health and behavioral outcomes associated with child abuse, such as poor executive function (Gould et al., 2012), instrumental learning and cognitive flexibility (Harms et al., 2017), and abnormal stress response and self-processing (Quevedo et al., 2017). To date, however, we have little information about how neural circuits of self-processing are impacted by maltreatment characteristics.
The goal of the present study was to identify neural functions associated with abuse characteristics such as cumulative adversity (including abuse), abuse presence, and duration of abuse during negatively or positively valenced self-appraisals in depressed youth with varying abuse histories. This knowledge is important because current empirically supported treatment consistently underperforms in depressed abused adolescents (Asarnow et al., 2009). As a result, the sequelae of child abuse ripples across the lifetime of affected individuals (Jaschek et al., 2016; Tanaka et al., 2011; Turanovic and Pratt, 2015). Understanding how abuse characteristics (presence, duration, and cumulative adversity) affects self-processing circuitry might help us personalize new treatments, such as neuromodulation of self-processing circuitry, to treat abused adolescents at risk for treatment-resistant depression. Understanding how abuse characteristics (e.g. duration) affect neural function might lead to a different emphasis in cognitive behavioral therapy (CBT) strategies. For example, to increase behavioral activation (which entails increasing physical activity) versus cognitive re-structuring (which entails identifying and challenging logical fallacies and biased cognition that perpetuate depressed mood).
1.2. Neurobiological sequelae of abuse
Abuse results in a variety of emotional abnormalities (Alink et al., 2009; Orobio de Castro, Veerman, Koops, Bosch and Monshouwer, 2002; Pollak and Sinha, 2002) that may contribute to negative self-representation's stability and dysregulated negative affect. Notably, generalized and highly negative or intrusive autobiographical memories may link childhood maltreatment to negative self-representations (Valentino et al., 2009), and has been directly linked to depression (Burnside et al., 2004; Williams and Moulds, 2007). Perhaps a combination of dysregulated emotions and intractable negative self-processing leads to chronic, treatment-resistant depression among child abuse victims.
Despite adolescence's significance for self-processing development, no studies have focused on the impact of presence, duration, or accumulation of adversity, including abuse, upon the neural substrates of adolescent self-processing. However, research suggests that longer lasting compared to less chronic maltreatment is linked to poorer executive function in childhood (Cowell et al., 2015), adolescent suicide attempts and suicidality (Jonson-Reid et al., 2012; Taussig et al., 2014), borderline personality disorder symptoms (Hecht et al., 2014), alcohol abuse (Shin et al., 2016), heightened threat sensitivity, and emotion dysregulation (Thompson et al., 2014). These studies suggest that longer maltreatment duration and cumulative adversity is likely to affect the circuitry of self-processing beyond the effects of a depressive episode.
Child abuse generally is associated with a host of neurobiological abnormalities. However, the direction and neural localization is varied and difficult to synthesize. Dissimilar results reflect the complex nature of abuse. Abnormal anterior cingulate cortex (ACC) function, which enables emotion regulation, error monitoring and social evaluation (Dedovic et al., 2016) as well as associative learning (Bush et al., 2002), has been observed in abused individuals (Lim et al., 2015). For example, 13-20 year-old participants abused during childhood exhibited hyperactivity in error processing regions (i.e. the dorsomedial prefrontal cortex and ACC) relative to controls during a go-no go inhibition task (Lim et al., 2015), which might indicate heightened monitoring to avoid mistakes that would elicit harsh punishment in abusive settings. Chronic early life stress is also associated with dendritic changes in the prefrontal cortex persisting into adulthood (Arnsten, 2009) and abused adults show medial prefrontal cortex (mPFC) hypoactivity during encoding and recognition of words of any valence compared to controls (van Harmelen et al., 2014). Furthermore, abnormal reward processing, specifically globus pallidus hypoactivity, has been reported among adults abused as children (Dillon et al., 2009). Overall, abuse exposure seems to compromise mPFC and ACC function and elicit striatal hypoactivity in adults abused as children.
Functional neuroimaging of abused adolescents and children yielded similar neural localization of dysfunction as adults abused as children. Reduced cortical thickness in abused versus non-abused children has been observed in the ACC, superior frontal gyrus, and orbitofrontal cortex (Kelly et al., 2013). In addition, reduced cortical surface area was observed within the left middle temporal and lingual gyrus. Local gyrification deficits have been noted in the lingual gyrus and the insula among abused children (Kelly et al., 2013). Smaller prefrontal and orbitofrontal cortex volumes have been reported in abused versus non-abused adolescents and those orbitofrontal abnormalities in physically abused youth were related to social difficulties and school related stress (Hanson et al., 2010). Our research also showed that abuse was associated with prefrontal cortex (specifically dorsal ACC) hypoactivity during positive versus negative self-related information among depressed abused adolescents (Quevedo et al., 2017), but our past work left unexamined the impact of presence of abuse, cumulative adversity across modalities of trauma, or abuse duration upon the circuitry of positive or negative self-processing. As in adults, abnormal reward and punishment processing (i.e., anticipation and experience of reinforcements) is also present among abused adolescents. Specifically, striatal hypoactivity during reward tasks has been reported in adolescents abused as children (Goff et al., 2013; Hanson et al., 2015; Mehta et al., 2010). Yet, behaviorally, abused children take excessive risks and are insensitive to changes primarily when the outcome magnitude of risky options change (Weller and Fisher, 2013). However, not all results are congruent with the adult literature. Unlike adults, abused children show hypoactivation to social rejection cues in the left anterior insula, extending into ventrolateral prefrontal and orbitofrontal cortex, amygdala, inferior parietal, and bilateral visual association cortices (Puetz et al., 2016).
Because the mPFC, insula, and inferior parietal cortex play key roles in self-processing (Molnar-Szakacs and Uddin, 2013) and, as reviewed, are impacted by childhood abuse, we expected that hypoactive and/or altered function in those cortical areas might underpin abnormal self-referential processes in depressed abused youth. Yet, no prior study has examined the effects of duration or cumulative adversity (across traumatic events including abuse) upon the neurobiology of emotionally valenced self-processing. Depression and PTSD pursuant to traumatic stress and abuse are all associated with preferential perception and encoding of negative self-relevant information. Thus, altered cortical substrates of self-processing might partially explain the association between adversity [maltreatment (e.g. neglect), abuse by acts of commission (e.g. physical or sexual abuse), and trauma such as accidents or witnessing violence] and treatment-resistant depression given the well-known role of negative cognitions and intractable negative self-perception in the onset and persistence of depression symptoms (Iacoviello et al., 2006).
1.3. The current study
We examined the impact of abuse presence, duration, and adversity accumulation upon the neurobiology of positive and negative self-processing among depressed adolescents with and without a history of abuse. Given negative self-attribution biases and blunted neural activity during positive information processing in both depression and maltreatment/abuse, we hypothesized that longer abuse duration and/or higher adversity accumulation would be linked to: 1) increased limbic, insular, inferior parietal, and/or mPFC activity during negative self-appraisals, and 2) decreased limbic and/or mPFC activity, insular, inferior parietal, and striatal activity during positive self-appraisals. We further predicted that 3) areas noted in hypotheses 1) and 2) would be unique to abuse or adversity characteristics and would not overlap with areas affected by depression severity and therefore remain after controlling for abuse presence vs. absence in a depressed sample. Finally, exploratory analyses sought to localize where in the brain the various abuse/adversity characteristics (presence, duration, cumulation) would converge in their impact upon the neural basis of self-processing. This research might facilitate developing novel targeted interventions for those most at risk for treatment-resistant depression by illuminating the neural substrates of persistent self-processing abnormalities linked to a maltreatment history.
2. Methods
2.1. Participants
Ninety-four participants, 54 depressed abuse (DEP-A) and 40 depressed (DEP) were recruited from the metropolitan areas of Minneapolis/St. Paul, MN and Pittsburgh, PA (Table 1). University of Minnesota and Pittsburgh IRBs approved all procedures. Participants were recruited from crisis intervention inpatient units, outpatient clinics, and among families who responded to flyers and radio ads. Participants completed a psychological evaluation using the Schedule for Affective Disorders and Schizophrenia for School-Aged Children (KSADS-PL, Kaufman et al., 1997), and Child Depression Rating Scale (CDRS, Poznanski et al., 1979), and were deemed to have a primary depressive disorder. Puberty was measured with the Pubertal Development Scale (PDS, Petersen et al., 1988). IQ was measured using the Wechsler Abbreviated Scale of Intelligence (WASI, Wechsler, 2011). Dissociation symptoms were measured using the Dissociation Experiences Scale (DES, Bernstein and Putnam, 1986). Scanning was completed 1–2 weeks after the initial visit.
Table 1.
Demographic and clinical indexes of depressed youth with maltreatment histories.
| Depressed without Abused |
Depressed Abused |
Comparison Statistic | |
|---|---|---|---|
| n = 40 | n = 54 | ||
| Scanning Site | χ2 (1) = 0.731 | ||
| Minneapolis | 12 (30.0%) | 12 (22.2%) | |
| Pittsburgh | 28 (70.0%) | 42 (77.8%) | |
| Age of Abuse Onset (Months) M (SD) | 0 (0) | 89.33 (52.99) | F (1, 92) = 113.38*** |
| IQ M (SD) | 107.63 (12.73) | 107.50 (18.80) | F (1, 92) = 0.001 |
| Sex N (%) | χ2 (1) = 2.856 | ||
| Male | 17 (42.5%) | 14 (25.9%) | |
| Female | 23 (57.5%) | 40 (74.1%) | |
| Puberty Status | 3.10 (0.52) | 3.16 (0.48) | F (1, 92) = 0.424 |
| Ethnicity N (%) | χ2 (6) = 2.872 | ||
| White | 23 (57.5%) | 26 (48.1%) | |
| African American | 5 (12.5%) | 6 (11.1%) | |
| Hispanic | 4 (10.0%) | 8 (14.8%) | |
| Asian/Asian American | 1 (2.5%) | 1 (1.9%) | |
| Native American | 0 | 2 (3.7%) | |
| Mixed | 6 (15.0%) | 2 (14.8%) | |
| Other | 1 (2.5%) | 3 (5.6%) | |
| χ2 (4) = 12.99 | |||
| Family Structure N (%) | |||
| Married | 30 (75%) | 21 (40.4%) | |
| Cohabiting | 1 (2.5%) | 7 (13.5%) | |
| Separated/Divorced | 4 (10.0%) | 14 (26.9%) | |
| Single | 5 (12.5%) | 8 (15.4%) | |
| Widowed | 0 | 2 (3.8%) | |
| Family Income N (%) | x2 (9) = 12.90 | ||
| < $35,000 | 8 (20%) | 27 (50%) | |
| $35,000 - $75,000 | 14 (35%) | 17 (31.5%) | |
| > $75,000 | 18 (45%) | 10 (28.6%) | |
| Medication N (%) | |||
| Anti-depressants | 18 (45.0%) | 22 (40.7%) | χ2 (1) = 0.17 |
| Antipsychotics | 1 (2.5%) | 5 (9.3%) | χ2 (1) = 1.76 |
| Mood Stabilizers | 0 | 1 (1.9%) | χ2 (1) = 0.75 |
| Stimulants | 7 (17.5%) | 4 (7.4%) | χ2 (1) = 2.27 |
| Anxiolytics | 3 (7.5%) | 4 (7.4%) | χ2 (1) = 0.00 |
| Abuse Types N (%) | |||
| Domestic violence witness | 0 | 38 (70.4%) | X2 (1) = 47.25*** |
| Neglect | 0 | 47 (87.0%) | X2 (1) = 69.63*** |
| Sexual Abuse | 0 | 18 (33.3%) | X2 (1) = 16.49*** |
| Physical Abuse | 0 | 24 (44.4%) | X2 (1) = 23.87*** |
| Abuse Duration | X2 (4) = 94.0*** | ||
| Single event(s) | 0 | 18 (33.3%) | |
| 1–3 years | 0 | 12 (22.2%) | |
| 3–6 years | 0 | 12 (22.2%) | |
| 6+ years | 0 | 12 (22.2%) | |
| Current Depression Severity (CDRS-R Total Score) M (SD) | 59.80 (14.17) | 65.33 (14.29) | F (1, 92) = 3.47 |
| Self-Esteem measured via SPPA Scores M (SD) | 109.78 (21.272) | 108.54 (17.007) | F (1, 92) = 0.098 |
Note: CDRS-R = Children's Depression Rating Scale Revised; Self-Perception.
Profile for Adolescents (SPPA); CASQ = Children's Attributional Style Questionnaire.
*p < .05. **p < .01. ***p < .001.
An earlier paper within this same study examined a more restricted sample size (N = 40) of abused adolescents and positive versus negative-self appraisals only from the self-point of view, in comparison with depressed not abused and healthy controls to test a specific hypothesis with regards to abuse by omission versus commission (Quevedo et al., 2017). The present study differs from that publication in its larger sample size of abused youth, due to more careful coding and added participants, as well as because positive and negative-self appraisal neural substrates were examined across all point of views (Quevedo et al., 2017 examined only the direct point of view), and since coding of the transcripts and tapes yielded additional variables measuring duration of abuse.
2.2. Coding of abuse characteristics
To collect abuse variables (abuse presence, duration, and adversity accumulation), we examined and coded the trauma section in the KSADS-PL. During the interview, participants that reported a history of abuse or traumatic adversity were asked about the nature of the abuse, the age of onset, and when the abuse ended. They were also asked to describe the nature of traumatic events such as accidents and witnessing shocking violence. Participants’ responses were annotated in the protocols and the interviews were videotaped. Experimenters confirmed abuse-related information by examining both written protocols and videotapes. Videos were double coded by two coders: diagnostic agreement for presence of depressive disorder was 98%; agreement on the presence of abuse was 100%. Participants were classified into two groups: depressed without abuse, and depressed with a history of abuse (neglect, witnessing domestic violence, physical abuse and/or sexual abuse). Cumulative adversity was assigned a number (1–4) indicating how many modalities of abuse were experienced with an additional point added for traumatic events experienced that were unrelated to abuse (e.g. being in a car accident or witnessing a murder). All 54 depressed abused adolescent participants included here experienced one or more of the four abuse modalities (See Supplemental Table 1). These classifications were made according to definitions of child maltreatment used by the CDC and other authors (Valentino et al., 2008). Abuse duration was expressed in number of days from the reported year of onset of maltreatment to its end. All participants were able to remember and report the age of onset of maltreatment or the specific time of single instances of maltreatment. See Table 1 for frequencies of participants per type of abuse.
The current paper focuses on depressed adolescents, first, because no healthy youth reported past abuse. Second, the effects of depression and abused types (commission vs. omission) upon the neurobiology of valenced self-processing with the whole sample and from the self-point of view have been reported in a separate publication that tested group-driven hypotheses different from the present inquiry (Quevedo et al., 2017).
2.3. Self-appraisals task
In the scanner, participants heard and saw the following statement: “What do I think about myself? I think …” and then heard positive or negative descriptors reflecting their behavior in social (e.g., “I make friends easily”) and academic (e.g., “It's hard for me to write papers”) domains. An equal number of phrases for the behavioral domains (social or academic) consisted of positive- and negative-valenced content. Each phrase lasted approximately 1 s, after which participants were instructed to indicate whether the phrase was self-descriptive or not via a button box. In total, 20 self-descriptors—10 academic (5 positive and 5 negative) and 10 social (5 positive and 5 negative)—were used. Participants evaluated the descriptors from their mother's, best friend's, or classmates' points of view. Given reviewed biases in self-processing among depressed and abused individuals, we analyzed brain function across all positive or negative statements, regardless of point of view or behavioral domain. The task was administered in a mixed block and event-related design that permitted separate analyses of positive and negative self-descriptors. The presentation of individual events within blocks was algorithmically optimized (Wager and Nichols, 2003) for detection of the difference between positive and negative items. The inter-stimulus interval (ISI) which included time to respond and time between aural delivery of descriptors was varied (M = 6600 ms, SD = 1248.2 ms). This temporal “jittering” allowed us to aggregate positive or negative self-descriptors across points of view and behavioral domains.
Behavioral responses (endorsement or denial) and stimulus presentation were collected using E-prime software. They were coded as 1 if participants endorsed positive self-descriptors or denied negative ones and as 0 for the reverse direction. Accordingly, higher values represented more positive self-representations.
2.4. Neuroimaging data acquisition
Neuroimaging data were collected using 3.0 T S Trio MRI scanners. Structural 3D axial MPRAGE images were acquired in the same session, TR/TE = 2100/3.31 ms; TI: 1050; Flip Angle 8°; Field of View: 256 × 200 mm; Slice-Thickness: 1 mm; Matrix: 256 × 200; 176 continuous slices). Mean BOLD images were then acquired with a gradient echo EPI sequence during 17:02 min covering 60 oblique axial slices (2.0 mm thick; TR/TE = 3340/30 ms; FOV = 200 × 200 mm; matrix 80 × 80; Flip Angle 90°). Movement parameters did not differ between the two sites as reported in Quevedo et al., (2016). Temporal signal to noise ratios were calculated using 3dTstats in AFNI by dividing mean baseline estimates (signal) by standard deviations of the residual time series (noise); these were then extracted and were statistically similar between sites.
2.5. Analyses
To test hypothesis 1 and 2 (i.e. the impact of abuse characteristics on the neurobiology of emotionally valenced self-processing) mean-centered abuse duration (number of days), cumulative adversity (1–4), depression severity (CDRS total scores), gender, medication dosage, and puberty were entered as predictors of brain activity during 1) positive self-descriptors and 2) negative self-descriptors in regression analyses. As would be expected, cumulative adversity and duration of abuse were moderately correlated, r (92) = 0.44, p < .001.
Whole-brain magnitude and cluster-extent thresholds were calculated using Monte Carlo simulations with AFNI18 (Cox, 1996), with the voxel-level threshold of puncorr < 0.005 and cluster-extent threshold of pFWE < 0.01 for all the multiple regression models conducted. Anatomical labels were determined using the xjView GUI in SPM12 and confirmed by visual inspection. Direction of results were also confirmed using SPSS-25 (Corp, 2017) to visualize extracted significant clusters of activity. Parameter estimates were extracted from all significant clusters using the “eigenvariate” toolbox in SPM12 centered on the coordinate of highest peak activity for each significant cluster and exported to SPSS-25 for graphical depictions and additional analysis.
We conducted regression analyses with the listed variables for all the depressed adolescent participants (DEP, N = 94) and for abused depressed adolescents’ sub-sample only (DEP-A, N = 54). These separate models for the DEP (N = 94) or the DEP-A (N = 54), were conducted to test our 3rd hypothesis. Specifically, to test if the impact of abuse upon brain function would remain when controlling for depression severity and presence vs. absence of abuse. If that were the case, this would begin to suggest that neural effects were linked to abuse characteristics and not to depression severity, [which was higher in the DEP-A (N = 54) sub-sample, Table 1]. Running these models would also allow us to identify via conjunction analyses the convergent areas impacted by all abuse characteristics (presence, duration, or cumulative adversity) within and across DEP or DEP-A models, and confirm whether areas impacted by abuse characteristic differed from those affected by other confounding variables. For the DEP (N = 94) model, a dichotomous (1, 0) variable indicated presence vs. absence of abuse which was entered in addition to the above-listed variables.
We also ran regressions with the full sample, including healthy controls (N = 131). Such models included the same variables plus IQ, which differed between depressed and healthy youth. Overlapping or identical areas of activity and direction of results were found for models examining depressed, (N = 94) or in smaller overlapping clusters for depressed abused youth, (N = 54). Results of the larger model (N = 131) are reported in the supplements.
We first visually inspected for possible overlaps and then used conjunction analyses to determine whether the same regions overlapped across models. We found no overlapping regions for abuse characteristics and those linked to depression severity or other confounding variables (puberty, medication dosage or gender). However, there seemed to be overlap for the neural areas affected by abuse presence and duration. Conjunction analyses tested for the conjunction null (Friston et al., 2005). Maps for all contrasts were at a cluster forming threshold of puncorr <0.005 and multiplied. Clusters surviving a cluster-level threshold pFWE < 0.01 were reported.
To aid in the visualization of results, the abuse duration mean centered variable originally entered as days, was split into 4 groups: single occurrence (n = 19), 1–3 years (n = 11), 4–6 years (n = 12) and 7+ years (n = 12).
3. Results
3.1. Neuroimaging results
3.1.1. Positive self-appraisals
There were no associations between brain activity during positive self-descriptors and abuse characteristics for either depressed abused adolescent sub-sample, N = 54, or for the entire depressed adolescent sample, N = 94.
3.1.2. Negative self-appraisals and duration of abuse
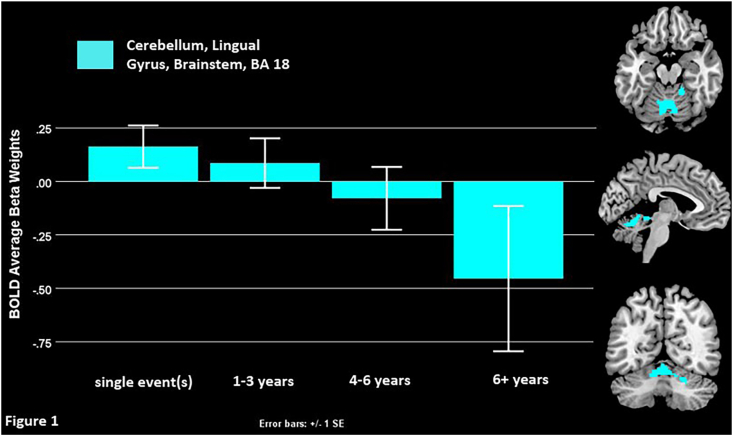
Among just abused depressed adolescents, (DEP-A, N = 54), those who experienced longer durations of abuse showed hypoactivity when evaluating negative self-descriptors in the bilateral cerebellum, lingual gyrus and midbrain (Fig. 1 and Table 2).
Fig. 1.
The effect of abuse duration in the abused depressed population (DEP-A, N = 54).
Table 2.
Abuse variables associated with neural activity during negative self-descriptors for abused adolescents (N = 54) and for all depressed youth (N = 94).
| Table 2 | Cluster Size (K) | Hemisphere | MNI |
T | ||
|---|---|---|---|---|---|---|
| X | Y | Z | ||||
| Abused Participants Only (N = 54) | ||||||
| Effects of abuse duration (negative direction of prediction) | ||||||
| Cerebellum, Lingual Gyrus, Midbrain, BA 18 | 833 | Left & Right | 0 | −58 | −16 | 4.22 |
| All Depressed Participants (N = 94) | ||||||
| Effects of abuse duration (negative direction of prediction) | ||||||
| Lingual Gyrus, Cerebellum, Fusiform, Parahippocampus, Midbrain, Inferior Temporal Gyrus, Thalamus, BA 18, 19, 37 | 2833 | Left & Right | −8 | −68 | −20 | 4.58 |
| Insula, Superior Temporal Gyrus, Putamen, Lentiform Nucleus, BA 13, 21, 22 | 482 | Left | −44 | −10 | −14 | 4.32 |
| Effects of abuse presence in depressed adolescents (positive direction of prediction) | ||||||
| Insula, Superior Temporal Gyrus, Putamen, Lentiform Nucleus, BA 13, 21, 22 | 467 | Left | −44 | −10 | −6 | 4.81 |
| Cerebellum, Occipital Lobe, Lingual Gyrus, Parahippocampus, Fusiform, Thalamus, Precuneus, BA 19, 20, 27, 30 | 532 | Right | 16 | −44 | −14 | 4.72 |
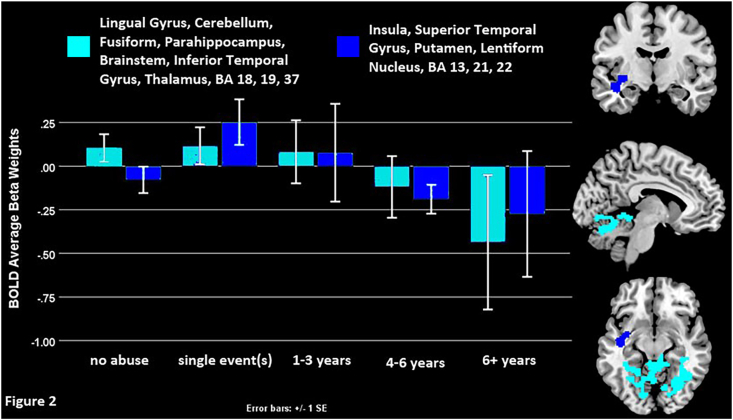
Regression analyses with all the depressed adolescents (DEP, N = 94) showed that youth who had experienced a longer duration of abuse experienced hypoactivity during negative self-appraisals in the left insula, superior and inferior temporal gyri, putamen, lentiform nucleus, as well as bilateral lingual gyrus, cerebellum, fusiform, parahippocampus, midbrain, and thalamus (Fig. 2 and Table 2).
Fig. 2.
The effect of abuse duration on the total depressed adolescent sample (DEP, N = 94).
3.1.3. Negative self-appraisals and presence of abuse
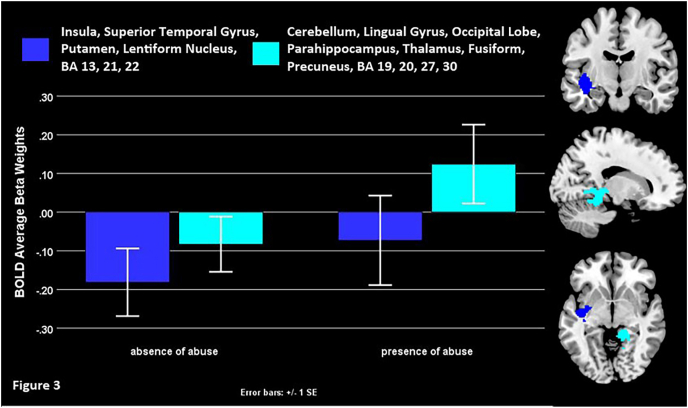
Regression analysis with all the depressed adolescents (DEP, N = 94) showed that presence versus absence of abuse was associated with higher activity in the left insula, putamen, lentiform nucleus, superior temporal gyrus, right cerebellum, occipital lobe, lingual gyrus, parahippocampus, fusiform, precuneus, and thalamus (Fig. 3; Table 2). There was no significant brain activity linked to cumulative adversity in either sample.
Fig. 3.
The effect of presence or absence of abuse among depressed youth (DEP, N = 94).
3.1.4. Conjunction analyses
To answer our exploratory question about the convergent areas impacted by presence or duration of abuse within and across DEP or DEP-A models, conjunction analyses conducted to identify convergence for the effects of abuse across all models showed that abuse duration was associated with hypoactivity in the bilateral cerebellum, lingual gyrus and midbrain in both DEP (N = 94) and DEP-A (N = 54) regression models (Supplementary Figure S1, Table 3).
Table 3.
Conjunction analysis for abuse variables from models for DEP-A (N = 54) and DEP youth (N = 94).
| Table 3. | Cluster Size (K) | Hemisphere | MNI |
||
|---|---|---|---|---|---|
| X | Y | Z | |||
| Conjunction Analysis | |||||
| Areas associated with abuse duration in both DEP-A (N = 54) and DEP (N = 94) | |||||
| Cerebellum, Lingual Gyrus, Midbrain, BA 18 | 818 | Left & Right | 24 | −66 | −30 |
3.1.5. Abuse characteristics and other potentially confounding variables
From visual inspection of the neural correlates of abuse characteristics and other confounding variables, there were no overlap between areas linked to severity of depression, gender, medication dosage or puberty and areas linked to presence and duration of abuse in either the total depressed sample (DEP, N = 94) or in the depressed abused adolescent sub-sample (DEP-A, N = 54).
4. Discussion
To our knowledge, this is the first study that has investigated the specific impact of duration and adversity accumulation on brain function. We investigated the impact of abuse characteristics upon the neural circuitry engaged by positive or negative self-appraisals in depressed adolescents with varying abuse histories. Our hypotheses that longer abuse duration or cumulative adversity would be linked to increased limbic, insular, inferior parietal and/or mPFC activity during negative self-appraisals and to decreased ones during positive self-appraisals were not supported. Instead, positive self-appraisals' circuitry was unrelated to abuse characteristics and chronic abuse was linked to corticolimbic and midbrain hypoactivity during negative self-appraisals (Fig. 1, Fig. 2). Presence versus absence of abuse was linked to higher insula, superior temporal gyrus, lentiform, cerebellar, occipital lobe parahippocampus, fusiform, thalamus, and precuneus activity (Fig. 3), and cumulative adversity was not linked to any changes in activity, possibly because abuse duration accounted for more variance in the same participants who also experienced abuse accumulation. As noted, these variables were moderately correlated. A larger more heterogenous sample of abused youth might have allowed for testing both variables. Finally, our hypothesis that the effects of abuse characteristics (i.e. duration or presence) would not overlap with areas affected by depression severity was confirmed.
4.1. Abuse duration
Longer abuse duration among abused adolescents (DEP-A, N = 54) was associated with hypoactive bilateral cerebellum, lingual gyrus, and midbrain (Fig. 1). Similar regions were hypoactive among adolescents abused for the longest time in the regression with the larger sample (DEP, N = 94). In that model hypoactive regions included the left insula, superior and inferior temporal gyri, putamen, lentiform nucleus, as well as the bilateral fusiform, parahippocampus, midbrain, and thalamus (Fig. 2). To summarize, this networks’ hypoactivity driven by longer duration of abuse (with youth exposed to 4+ years of abuse evidencing significant neural hypoactivation, Fig. 2) might indicate decreased ability to make social evaluations, recall emotional memories and gate, detect, or regulate critical emotional content when processing negative self-relevant information. Prior research on the impact of abuse duration upon neural function is very limited. Vythilingam et al. (2002) found decreased left hippocampal volume in depressed women with a history of severe or recurrent physical or sexual abuse compared to both depressed women without such a history and healthy controls. Here we found hypoactive engagement of areas that support emotional processing, including punishment and reward processing (i.e. the putamen), saliency, arousal, basic reflexes, and social cognition.
Abnormal cerebellar activity is linked to emotion dysregulation and visuomotor control (Hernaez-Goni et al., 2010; Schutter and van Honk, 2009) including fine motor coordination engaged during button presses in this task (Jimsheleishvili and Dididze, 2020). The lentiform nucleus (putamen and globus pallidus) are components of the striatum that orchestrate learning and motor control, language, and reward/punishment processing (Ghandili and Munakomi, 2020). The insula enables empathy, interoceptive awareness and emotional recall (Phan et al., 2002; Singer, 2006) and, along with the putamen, is a key node within the saliency network, which detects and processes arousing emotional stimuli, including emotionally charged faces and facial recognition (Bartels and Zeki, 2000; Menon, 2015). Face processing, including discriminating between familiar vs. non-familiar faces (Rossion et al., 2003) is also associated with the fusiform and inferior temporal lingual gyri (Kitada et al., 2010; Rossion et al., 2003), while the superior temporal gyrus enables social processing through facial movements (Adolphs, 2003). Finally, the parahippocampus enables associative contextual processing and episodic memory recall (Aminoff et al., 2013). The functions of these areas are numerous but it is likely that during the task functions related to social and emotion processing are less called upon during negative self-processing in chronically abused youth. Perhaps, while youth appraise the appropriateness of negative self-descriptors, they are harkening to interactions with the salient individuals and environments in their past and present, whose point of view they are asked to take. Thus, recruiting the parahippocampus and fusiform along with the saliency network. This network's hypoactivity might indicate a decreased ability to recall emotional and social memories related to negative self-relevant information and in integrating all sensory modalities into emotion and memory itself. Speculatively, this might be a protective measure to prevent the re-experience of abuse when recalling negative autobiographical material or/and the past environmental cues linked to abusive experiences elicited by our task.
Alternatively, given that the cerebellum supports emotion regulation (Schutter and van Honk, 2009) along with the superior temporal gyrus, insula and thalamus (Adolphs, 2002), their hypoactivity might underpin blunted emotion processing, and sensory integration and regulation deficits prevalent among victims of child abuse (Gruhn and Compas, 2020). Of note, the thalamic pulvinar nucleus enables fear recognition (Ward et al., 2007) and plays a role in the regulation of defensive reflexive responses such as the inhibition of the startle response (Kumari et al., 2005). It is well described that victims of abuse, particularly those with PTSD, suffer lifetime sleep difficulties and either hyperarousal or hypoarousal (Steine et al., 2012; Wamser-Nanney and Chesher, 2018) as part of the long-lasting consequences of child abuse and chronic trauma.
Given that abuse duration and presence were only associated with negative self-appraisals (not with positive self-appraisals), our results might indicate that chronic abuse blunts the ability to react to and/or evaluate negatively charged self-relevant information in active and/or critical ways. Alternatively, the saliency of negative self-relevant information might be blunted by prolonged abuse exposure due to habituation to hostile and negative personal attacks common in abusive environments. One explanation is that, given hypoactivity in saliency and affect regulation networks, chronically abused youth might dissociate while processing negative self-relevant information. However, dissociation as measured by the DES was uncorrelated with any of the brain areas impacted by abuse characteristics in our sample (though it should be noted that the DES is regarded by some as a relatively insensitive measure). These are speculative explanations, as we did not collect measures of critical or active processing of negative self-relevant information or more developmentally sensitive measures of dissociation.
4.2. Neural convergence of abuse's impact
The areas (bilateral cerebellum extending to lingual gyrus and midbrain) impacted by abuse characteristics in the depressed abused adolescents (DEP-A, N = 54) overlapped with areas impacted in the whole sample (DEP, N = 94).
The bilateral cerebellum, lingual gyrus and midbrain support emotion regulation, spatial memory, arousal and sensorimotor gating (Gomez-Nieto et al., 2020; Hernaez-Goni et al., 2010). Of interest is the association of chronic abuse with blunted midbrain and lingual gyrus activity (Table 3, Supplemental Figure 2). The midbrain supports arousal and sensorimotor gating functions (Gomez-Nieto et al., 2020; Lang and Davis, 2006). Interestingly, we found blunted overall startle magnitude in post-institutionalized adopted adolescents versus non-adopted US born controls (Quevedo et al., 2015). Institutionalized children suffer compounded traumatic experiences including social and cognitive deprivation, neglect and in some cases abuse by commission (i.e. physical and sexual intrusions). We also found that startle potentiation to negative images was negatively related to severity of pre-adoption neglect (Quevedo et al., 2015) suggesting that the more abused children evidenced the less responsive fear/negative startle reactivity. In the same sample, post-institutionalized peri-pubertal youth exhibited blunted cortisol awakening response (an index of hypothalamic pituitary adrenal, HPA, axis) compared to U.S. born and reared controls (Quevedo et al., 2012). In the context of that past research, our findings of hypoactive midbrain (Fig. 3) among other structures linked to more chronic abuse, suggest that prolonged abuse might suppress the activity of early developing ancillary structures that support basic defensive and automatic emotional responses. The midbrain periaqueductal gray, in specific, has been proposed as acting like “a brake” to the sympathetic nervous system during inescapable threats/abuse resulting in dissociative defenses (Terpou et al., 2019), which results in the parasympathetic dominance of the autonomic nervous system, tonic immobility and the inhibition of motor commands.
Potentially, downregulating reactivity in these areas after chronic abuse exposure, might be an adaptive strategy used in toxic negatively arousing environments that might require organistic habituation to conserve energy. This is consistent with the “toxic stress” model, which has been supported by similarly blunted stress reactivity in abused children and adults (Joos et al., 2019; Obasi et al., 2017; Voellmin et al., 2015). Future research ought to explore whether bilateral cerebellum, lingual gyrus and brainstem hypoactivity are part of a broader physiological downregulation that includes the HPA axis and the startle response, and whether these systems’ abnormalities might be reciprocally linked among individuals exposed to more chronic and severe adversity.
4.3. Abuse presence
Presence vs. absence of abuse among depressed youth was associated with higher left insula, putamen, lentiform nucleus, superior temporal gyrus, right cerebellum, occipital lobe, lingual gyrus, parahippocampus, fusiform, precuneus and thalamus activity during negative self-processing (Fig. 3). Because presence of abuse recruited many of the same regions as duration of abuse, the effect presumably still implicates some of the earlier reviewed functions (sensorimotor gating, reward/punishment and learning, emotion regulation and social cognition) enabled by this network. However, this extends to the precuneus. Precuneus engagement was associated with presence of abuse but it was not impacted by duration of abuse. The precuneus underpins self-referential processing, especially from the first person perspective, as well as the retrieval of episodic memory (Cavanna and Trimble, 2006), suggesting that youth who have been abused are integrating negative self-relevant information (generated, perhaps from the first person perspective) into emotion and memory or/and dedicating more cognitive efforts to recovering such memories.
To be clear, there was no significant overlap between the effects of duration of abuse and presence of abuse based on corrected conjunction analysis. However, even though the areas that overlapped failed to meet the threshold of significant cluster sizes, the brain structures implicated are largely the same. Which raises the issue of abuse presence and duration yielding opposite effects on their function. As shown in Fig. 2, there is increased activity in those who had one or several isolated incidents of abuse when examining the effect of duration of abuse in the DEP-A or DEP samples. By contrast, non-abused depressed youth's activity in those areas hovers around 0. Additional visualization might expand this explanation. Supplemental Figure 3 shows that when areas engaged by presence vs. absence are plotted by abuse duration groups, active clusters engaged by abuse presence seem primarily driven by youth who experienced 1 instance or 1–3 years or abuse, while 4 + years of abuse are linked to hypoactivity during the processing of negative self-descriptors. This suggests that the relatively increased activity in presence vs. absence of abuse is driven by youth with acute exposure to abuse experiences. This would tentatively imply that single abusive/traumatic events or/and those reported to last 1–3 years might be associated to higher saliency, memory and vigilance for negatively biased information supported by the engaged structures (insula, STG, striatum, thalamus, parahippocampus, fusiform, and precuneus). By contrast, longer lasting adversity (abuse 4+ years) might be associated with these structures' disengagement during negative self-relevant information.
To expand this tentative explanation, we turned to the PTSD literature. McTeague et al. (2010) found that patients with PTSD who experienced a single, discrete trauma had increased defensive reactivity during aversive imagery. Yet patients with chronic and multiple trauma showed hypoactivity instead. All our results are consistent with the possibility that youth exposed to single or several discrete trauma instances (and perhaps 1–3 years of abuse) might exhibit relatively higher neurophysiological responses during negative stimuli, while those exposed to long-lasting abuse might instead exhibit blunted neurophysiological activity. This suggests that prolonged trauma and short-term trauma might contribute to depressive symptoms via different mechanisms and that blunted saliency, stress, sensorimotor gating and social cognition circuitry might be a marker for treatment-resistant depression. More research is required to confirm whether hypo versus hyper neurophysiologic function predicts treatment-resistant depression pursuant to child maltreatment. Finally, it must be noted that prolonged exposure therapy benefits multiple trauma and single trauma patients equally (Jerud et al., 2017). It is unknown if this extends to treating abuse complicated depression.
Our early research on child abuse yielded that abused participants show dorsal ACC hypoactivity during positive vs. negative self-appraisals, as well as hypoactivity in the dorsal ACC and the caudate during positive self-appraisals compared to depressed not abused and healthy adolescents (Quevedo et al., 2017). In contrast, here we found no significant differences in activity for positive appraisals, nor any implication of the ACC in this sample. This could be due to differences in methodology, namely we explored group differences in the prior study whereas here continuous variables were examined in regressions, additionally we recovered more data for this study, expanding the pool of participants, and we performed these analyses specifically to look at abuse characteristics, variables that were not examined in the previous study.
4.4. Limitations and future directions
There are limitations to this study. Most of the participants were white and female. However, it must be noted that females are more affected by abuse compared to males in the U.S. (Goverment, 2015). All participants in this study were depressed, so the associations between abuse duration and brain activity found here might not generalize to non-depressed maltreated or to more diverse youth samples. It is also not clear if disturbances in self-processing stem from effects of clinical depression, early maltreatment experience, or both. Though it must be noted that the areas linked to depression severity did not overlap with those linked to abuse presence or duration. Furthermore, the areas affected by abuse variables persisted among abused only or when both abused and not abused depressed were examined. An additional limitation of this research pertains to the accuracy of coding cumulative adversity and chronicity of abuse from only the adolescent's report. Youth were not always able to report duration of abuse versus the occurrence of several separate single instances of maltreatment. Though we suspected that for some youth, abuse lasted longer that the single instances reported we were constrained to categorize some experiences as acute exposures. Additional sources should be interviewed to gather more accurate information and a larger sample size would allow for greater flexibility in coding abuse experiences (See Supplemental Table 2).
Future research should examine whether trauma-focused cognitive behavioral therapy or novel interventions (such as self-compassion interventions) can counteract hypoactive networks (that support saliency, sensorimotor gating, arousal, and social cognition), pursuant to chronic abuse. Furthermore, the hypoactive areas in our results ought to be interrogated in terms of their association with symptoms, peripheral stress, and emotion physiology markers, such as the startle response and the HPA axis function, that the current paper was unable to investigate. Furthermore, there may be constructs, such as emotion regulation, that overlap in their networks with self-processing. Intrapsychic factors such as preserved emotion regulation or higher self-compassion might protect certain abused individuals from developing depression; alternatively, environmental support such as the availability of a caring family member may instill resiliency in abused youth. Future research should explore their mechanisms and possible protective effects. Another issue is that while childhood abuse is linked to treatment-resistant depression and while negative self-processing is a robust factor of chronic depression, this study does not demonstrate that hypoactive networks of negative self-processing are the intervening mechanism. Longitudinal research must test that hypothesis. Furthermore, we cannot conclude that hypoactive networks characterize only abnormal self-processing due to chronic abuse. It is possible that neural hypoactivity might affect multiple dimensions pursuant to abuse or that other dimensions may exhibit hyperactivity. Psychological dimensions that might exhibit abnormal neural patterns due to childhood abuse, include emotion regulation, decision making, and reward processing which can also extend to cognitive performance such as attention, executive function and cognitive control. Sampling multiple dimensions would better characterize the impact of abuse duration upon those constructs. Finally, recent evidence suggests that epigenetic factors may play a mediating role in the relationship between early adversity and the development of stress-related disorders, including depression (Klengel and Binder, 2015). Future studies examining these relationships may profit from including epigenetic markers in their models.
5. Conclusion
We examined how abuse variables were associated with brain activity during self-processing in depressed adolescents. Contrary to our hypotheses, we found hypoactivity in limbic, mid-brain and temporal areas during negative self-appraisals in adolescents with a longer duration of abuse, but no effects of cumulative adversity. Neural activity during positive self-appraisals was unrelated to abuse variables. The hypoactive areas associated with duration of abuse are implicated in emotion and its regulation, arousal/sensorimotor gating, memory, and social processing. The hyperactive areas associated with presence of abuse underpin similar processes, possibly driven by those with acute, single traumatic/abusive events (Supplemental Figure 3). This might indicate initial enhanced saliency of negative self-relevant information among acutely abused youth which decreases among those chronically abused. This network's hypoactivity in chronically abused youth may index blunted physiological reactivity during any negative stimuli (including negatively self-relevant information) as an adaptation to chronically noxious environments, as suggested by the toxic stress model. These findings might help us adjust treatment for abuse-complicated depression, particularly in response to singly vs. chronically abused adolescents. For instance, it might be useful to enhance critical processing of negative self-relevant information or increase temporary emotional arousal to such information during clinical sessions, so that chronically abused adolescents might learn to be reasonably critical and/or temporarily be more reactive, with the goal of promoting active rejection of undeserved negative information and discern what should be incorporated into their self-concept.
Credit author statement
Karina Quevedo: Writing original grant, Formal analysis and manuscript writing. Maggie Engstrom: Formal analysis and manuscript writing. Guanmin Liu: Data analysis and manuscript editing. Carmen Santana-Gonzalez: Manuscript editing. Jia Yuan Teoh: Formal analysis, abuse experience coding. Madeline Harms: Manuscript editing. Kiry Koy: Formal analysis, abuse experience coding.
Funding
This work was supported by grants awarded to KQ from the National Institute of Mental Health (NIMH; MH092601), Brain & Behavior Research Foundation (NARSAD Young Investigator Award), and the U of M Clinical and Translational Science Institute for data collection and analysis and manuscript preparation. Funding sources were not involved in the design of this study, collection, analysis and interpretation of data or manuscript preparation.
Declaration of competing interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ynstr.2021.100310.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- Adolphs R. Neural systems for recognizing emotion. Curr. Opin. Neurobiol. 2002;12(2):169–177. doi: 10.1016/S0959-4388(02)00301-X. [DOI] [PubMed] [Google Scholar]
- Adolphs R. Cognitive neuroscience of human social behaviour. Nat. Rev. Neurosci. 2003;4(3):165–178. doi: 10.1038/nrn1056. [DOI] [PubMed] [Google Scholar]
- Alarcón G., Sauder M., Teoh J.Y., Forbes E.E., Quevedo K. Amygdala functional connectivity during self-face processing in depressed adolescents with recent suicide attempt. J. Am. Acad. Child Adolesc. Psychiatry. 2019;58(2):221–231. doi: 10.1016/j.jaac.2018.06.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alink L.R., Cicchetti D., Kim J., Rogosch F.A. Mediating and moderating processes in the relation between maltreatment and psychopathology: mother-child relationship quality and emotion regulation. J. Abnorm. Child Psychol. 2009;37(6):831–843. doi: 10.1007/s10802-009-9314-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aminoff E.M., Kveraga K., Bar M. The role of the parahippocampal cortex in cognition. Trends Cognit. Sci. 2013;17(8):379–390. doi: 10.1016/j.tics.2013.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnsten A.F. Stress signalling pathways that impair prefrontal cortex structure and function. Nat. Rev. Neurosci. 2009;10(6):410–422. doi: 10.1038/nrn2648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asarnow J.R., Emslie G., Clarke G., Wagner K.D., Spirito A., Vitiello B., Brent D. Treatment of selective serotonin reuptake inhibitor-resistant depression in adolescents: predictors and moderators of treatment response. J. Am. Acad. Child Adolesc. Psychiatry. 2009;48(3):330–339. doi: 10.1097/chi.0b013e3181977476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartels A., Zeki S. The neural basis of romantic love. Neuroreport. 2000;11(17):3829–3834. doi: 10.1097/00001756-200011270-00046. [DOI] [PubMed] [Google Scholar]
- Bennett D.S., Sullivan M.W., Lewis M. Neglected children, shame-proneness, and depressive symptoms. Child. Maltreat. 2010;15(4):305–314. doi: 10.1177/1077559510379634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernstein E.M., Putnam F.W. Development, reliability, and validity of a dissociation scale. J. Nerv. Ment. Dis. 1986;174(12):727–735. doi: 10.1097/00005053-198612000-00004. [DOI] [PubMed] [Google Scholar]
- Brodsky Beth S., Oquendo Maria, Ellis Steven P., Haas Gretchen L., Malone Kevin M., Mann J. John. The relationship of childhood abuse to impulsivity and suicidal behavior in adults with major depression. Am. J. Psychiatr. 2001;158(11):1871–1877. doi: 10.1176/appi.ajp.158.11.1871. [DOI] [PubMed] [Google Scholar]
- Burnside E., Startup M., Byatt M., Rollinson L., Hill J. The role of overgeneral autobiographical memory in the development of adult depression following childhood trauma. Br. J. Clin. Psychol. 2004;43(Pt 4):365–376. doi: 10.1348/0144665042388991. [DOI] [PubMed] [Google Scholar]
- Bush G., Vogt B.A., Holmes J., Dale A.M., Greve D., Jenike M.A., Rosen B.R. Dorsal anterior cingulate cortex: a role in reward-based decision making. Proc. Natl. Acad. Sci. U. S. A. 2002;99(1):523–528. doi: 10.1073/pnas.012470999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavanna A.E., Trimble M.R. The precuneus: a review of its functional anatomy and behavioural correlates. Brain. 2006;129(3):564–583. doi: 10.1093/brain/awl004. [DOI] [PubMed] [Google Scholar]
- Corp I. IBM Corp; Armonk, NY: 2017. IBM SPSS Statistics for Windows. Version 25.0. [Google Scholar]
- Cowell R.A., Cicchetti D., Rogosch F.A., Toth S.L. Childhood maltreatment and its effect on neurocognitive functioning: timing and chronicity matter. Dev. Psychopathol. 2015;27(2):521–533. doi: 10.1017/S0954579415000139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox R.W. AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Comput. Biomed. Res. 1996;29(3):162–173. doi: 10.1006/cbmr.1996.0014. [DOI] [PubMed] [Google Scholar]
- Dedovic K., Slavich G.M., Muscatell K.A., Irwin M.R., Eisenberger N.I. Dorsal anterior cingulate cortex responses to repeated social evaluative feedback in young women with and without a history of depression. Front. Behav. Neurosci. 2016;10:64. doi: 10.3389/fnbeh.2016.00064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dillon D.G., Holmes A.J., Birk J.L., Brooks N., Lyons-Ruth K., Pizzagalli D.A. Childhood adversity is associated with left basal ganglia dysfunction during reward anticipation in adulthood. Biol. Psychiatr. 2009;66(3):206–213. doi: 10.1016/j.biopsych.2009.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fang X., Brown D.S., Florence C.S., Mercy J.A. The economic burden of child maltreatment in the United States and implications for prevention. Child Abuse Negl. 2012;36(2):156–165. doi: 10.1016/j.chiabu.2011.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feiring C., Cleland C.M., Simon V.A. Abuse-specific self-schemas and self-functioning: a prospective study of sexually abused youth. J. Clin. Child Adolesc. Psychol. 2010;39(1):35–50. doi: 10.1080/15374410903401112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friston K.J., Penny W.D., Glaser D.E. Conjunction revisited. Neuroimage. 2005;25(3):661–667. doi: 10.1016/j.neuroimage.2005.01.013. [DOI] [PubMed] [Google Scholar]
- Ghandili M., Munakomi S. 2020. Neuroanatomy, Putamen StatPearls. Treasure Island (FL) [PubMed] [Google Scholar]
- Goff B., Gee D.G., Telzer E.H., Humphreys K.L., Gabard-Durnam L., Flannery J., Tottenham N. Reduced nucleus accumbens reactivity and adolescent depression following early-life stress. Neuroscience. 2013;249:129–138. doi: 10.1016/j.neuroscience.2012.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez-Nieto R., Hormigo S., Lopez D.E. Prepulse inhibition of the auditory startle reflex assessment as a hallmark of brainstem sensorimotor gating mechanisms. Brain Sci. 2020;10(9) doi: 10.3390/brainsci10090639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gould F., Clarke J., Heim C., Harvey P.D., Majer M., Nemeroff C.B. The effects of child abuse and neglect on cognitive functioning in adulthood. J. Psychiatr. Res. 2012;46(4):500–506. doi: 10.1016/j.jpsychires.2012.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goverment U.S. 2015. Child Maltreatment. [Google Scholar]
- Gruhn M.A., Compas B.E. Effects of maltreatment on coping and emotion regulation in childhood and adolescence: a meta-analytic review. Child Abuse Neglect. 2020;103:104446. doi: 10.1016/j.chiabu.2020.104446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanson J.L., Chung M.K., Avants B.B., Shirtcliff E.A., Gee J.C., Davidson R.J., Pollak S.D. Early stress is associated with alterations in the orbitofrontal cortex: a tensor-based morphometry investigation of brain structure and behavioral risk. J. Neurosci. 2010;30(22):7466–7472. doi: 10.1523/JNEUROSCI.0859-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanson J.L., Hariri A.R., Williamson D.E. Blunted ventral striatum development in adolescence reflects emotional neglect and predicts depressive symptoms. Biol. Psychiatr. 2015;78(9):598–605. doi: 10.1016/j.biopsych.2015.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harkness K.L., Lumley M.N. Child abuse and neglect and the development of depression in children and adolescents. In: Hankin J.R.Z.A.B.L., editor. Handbook of Depression in Children and Adolescents. Guilford Press; New York, NY, US: 2008. pp. 466–488. [Google Scholar]
- Harms M.B., Birn R., Provencal N., Wiechmann T., Binder E.B., Giakas S.W.…Pollak S.D. Early life stress, FK506 binding protein 5 gene (FKBP5) methylation, and inhibition-related prefrontal function: a prospective longitudinal study. Dev. Psychopathol. 2017;29(5):1895–1903. doi: 10.1017/S095457941700147X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hart H., Rubia K. Neuroimaging of child abuse: a critical review. Front. Hum. Neurosci. 2012;6:52. doi: 10.3389/fnhum.2012.00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harter S. Guilford Press; New York, NY, US: 1999. The Construction of the Self: A Developmental Perspective. [Google Scholar]
- Hecht K.F., Cicchetti D., Rogosch F.A., Crick N.R. Borderline personality features in childhood: the role of subtype, developmental timing, and chronicity of child maltreatment. Dev. Psychopathol. 2014;26(3):805–815. doi: 10.1017/S0954579414000406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hernaez-Goni P., Tirapu-Ustarroz J., Iglesias-Fernandez L., Luna-Lario P. [The role of the cerebellum in the regulation of affection, emotion and behaviour] Rev. Neurol. 2010;51(10):597–609. [PubMed] [Google Scholar]
- Iacoviello B.M., Alloy L.B., Abramson L.Y., Whitehouse W.G., Hogan M.E. The course of depression in individuals at high and low cognitive risk for depression: a prospective study. J. Affect. Disord. 2006;93(1–3):61–69. doi: 10.1016/j.jad.2006.02.012. [DOI] [PubMed] [Google Scholar]
- Jaschek G., Carter-Pokras O., He X., Lee S., Canino G. Association of child maltreatment and depressive symptoms among Puerto Rican youth. Child Abuse Negl. 2016;58:63–71. doi: 10.1016/j.chiabu.2016.06.016. [DOI] [PubMed] [Google Scholar]
- Jerud A.B., Farach F.J., Bedard-Gilligan M., Smith H., Zoellner L.A., Feeny N.C. Repeated trauma exposure does not impair distress reduction during imaginal exposure for posttraumatic stress disorder. Depress. Anxiety. 2017;34(8):671–678. doi: 10.1002/da.22582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jimsheleishvili S., Dididze M. 2020. Neuroanatomy, Cerebellum StatPearls. Treasure Island (FL) [PubMed] [Google Scholar]
- Jonson-Reid M., Kohl P.L., Drake B. Child and adult outcomes of chronic child maltreatment. Pediatrics. 2012;129(5):839–845. doi: 10.1542/peds.2011-2529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joos C.M., McDonald A., Wadsworth M.E. Extending the toxic stress model into adolescence: profiles of cortisol reactivity. Psychoneuroendocrinology. 2019;107:46–58. doi: 10.1016/j.psyneuen.2019.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufman J., Birmaher B., Brent D., Rao U., Flynn C., Moreci P.…Ryan N. Schedule for affective disorders and Schizophrenia for school-age children-present and lifetime version (K-SADS-PL): initial reliability and validity data. J. Am. Acad. Child Adolesc. Psychiatry. 1997;36(7):980–988. doi: 10.1097/00004583-199707000-00021. [DOI] [PubMed] [Google Scholar]
- Kelly P.A., Viding E., Wallace G.L., Schaer M., De Brito S.A., Robustelli B., McCrory E.J. Cortical thickness, surface area, and gyrification abnormalities in children exposed to maltreatment: neural markers of vulnerability? Biol. Psychiatr. 2013;74(11):845–852. doi: 10.1016/j.biopsych.2013.06.020. [DOI] [PubMed] [Google Scholar]
- Kitada R., Johnsrude I.S., Kochiyama T., Lederman S.J. Brain networks involved in haptic and visual identification of facial expressions of emotion: an fMRI study. Neuroimage. 2010;49(2):1677–1689. doi: 10.1016/j.neuroimage.2009.09.014. [DOI] [PubMed] [Google Scholar]
- Klengel T., Binder E.B. Epigenetics of stress-related psychiatric disorders and gene x environment interactions. Neuron. 2015;86(6):1343–1357. doi: 10.1016/j.neuron.2015.05.036. [DOI] [PubMed] [Google Scholar]
- Kumari V., Antonova E., Zachariah E., Galea A., Aasen I., Ettinger U.…Sharma T. Structural brain correlates of prepulse inhibition of the acoustic startle response in healthy humans. Neuroimage. 2005;26(4):1052–1058. doi: 10.1016/j.neuroimage.2005.03.002. [DOI] [PubMed] [Google Scholar]
- Lang P.J., Davis M. Emotion, motivation, and the brain: reflex foundations in animal and human research. Prog. Brain Res. 2006;156:3–29. doi: 10.1016/S0079-6123(06)56001-7. [DOI] [PubMed] [Google Scholar]
- Lim L., Hart H., Mehta M.A., Simmons A., Mirza K., Rubia K. Neural correlates of error processing in young people with a history of severe childhood abuse: an fMRI study. Am. J. Psychiatr. 2015;172(9):892–900. doi: 10.1176/appi.ajp.2015.14081042. [DOI] [PubMed] [Google Scholar]
- McTeague L.M., Lang P.J., Laplante M.-C., Cuthbert B.N., Shumen J.R., Bradley M.M. Aversive imagery in posttraumatic stress disorder: trauma recurrence, comorbidity, and physiological reactivity. Biol. Psychiatr. 2010;67(4):346–356. doi: 10.1016/j.biopsych.2009.08.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta M.A., Gore-Langton E., Golembo N., Colvert E., Williams S.C., Sonuga-Barke E. Hyporesponsive reward anticipation in the basal ganglia following severe institutional deprivation early in life. J. Cognit. Neurosci. 2010;22(10):2316–2325. doi: 10.1162/jocn.2009.21394. [DOI] [PubMed] [Google Scholar]
- Menon V. Salience network. Brain mapping. An Encyclopedic Reference. 2015;2:14. [Google Scholar]
- Mills K.L., Goddings A.L., Clasen L.S., Giedd J.N., Blakemore S.J. The developmental mismatch in structural brain maturation during adolescence. Dev. Neurosci. 2014;36(3–4):147–160. doi: 10.1159/000362328. [DOI] [PubMed] [Google Scholar]
- Molnar-Szakacs I., Uddin L. Self-processing and the default mode network: interactions with the mirror neuron system. Front. Hum. Neurosci. 2013;7(571) doi: 10.3389/fnhum.2013.00571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nejad A., Fossati P., Lemogne C. Self-referential processing, rumination, and cortical midline structures in major depression. Front. Hum. Neurosci. 2013;7(666) doi: 10.3389/fnhum.2013.00666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obasi E.M., Shirtcliff E.A., Cavanagh L., Ratliff K.L., Pittman D.M., Brooks J.J. Hypothalamic-pituitary-adrenal reactivity to acute stress: an investigation into the roles of perceived stress and family resources. Prev. Sci. 2017;18(8):923–931. doi: 10.1007/s11121-017-0759-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orobio de Castro B., Veerman J.W., Koops W., Bosch J.D., Monshouwer H.J. Hostile attribution of intent and aggressive behavior: a meta-analysis. Child Dev. 2002;73(3):916–934. doi: 10.1111/1467-8624.00447. [DOI] [PubMed] [Google Scholar]
- Petersen A.C., Crockett L., Richards M., Boxer A. A self-report measure of pubertal status: reliability, validity, and initial norms. J. Youth Adolesc. 1988;17(2):117–133. doi: 10.1007/BF01537962. [DOI] [PubMed] [Google Scholar]
- Phan K.L., Wager T., Taylor S.F., Liberzon I. Functional neuroanatomy of emotion: a meta-analysis of emotion activation studies in pet and fMRI. Neuroimage. 2002;16(2):331–348. doi: 10.1006/nimg.2002.1087. [DOI] [PubMed] [Google Scholar]
- Pollak S.D., Sinha P. Effects of early experience on children's recognition of facial displays of emotion. Dev. Psychol. 2002;38(5):784–791. doi: 10.1037//0012-1649.38.5.784. [DOI] [PubMed] [Google Scholar]
- Poznanski E.O., Cook S.C., Carroll B.J. A depression rating scale for children. Pediatrics. 1979;64(4):442–450. [PubMed] [Google Scholar]
- Puetz V.B., Viding E., Palmer A., Kelly P.A., Lickley R., Koutoufa I.…McCrory E.J. Altered neural response to rejection-related words in children exposed to maltreatment. JCPP (J. Child Psychol. Psychiatry) 2016;57(10):1165–1173. doi: 10.1111/jcpp.12595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quevedo K., Johnson A., Loman M., Lafavor T., Gunnar M. The confluence of adverse early experience and puberty on the cortisol awakening response. Int. J. Behav. Dev. 2012;36(1):19–28. doi: 10.1177/0165025411406860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quevedo K., Johnson A.E., Loman M.M., Lafavor T., Moua B., Gunnar M.R. The impact of early neglect on defensive and appetitive physiology during the pubertal transition: a study of startle and postauricular reflexes. Dev. Psychobiol. 2015;57(3):289–304. doi: 10.1002/dev.21283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quevedo K., Ng R., Scott H., Smyda G., Pfeifer J.H., Malone S. The neurobiology of self-processing in abused depressed adolescents. Dev. Psychopathol. 2017;29(3):1057–1073. doi: 10.1017/S0954579416001024. [DOI] [PubMed] [Google Scholar]
- Rossion B., Schiltz C., Crommelinck M. The functionally defined right occipital and fusiform “face areas” discriminate novel from visually familiar faces. Neuroimage. 2003;19(3):877–883. doi: 10.1016/S1053-8119(03)00105-8. [DOI] [PubMed] [Google Scholar]
- Russo S.J., Nestler E.J. The brain reward circuitry in mood disorders. Nat. Rev. Neurosci. 2013;14(9):609–625. doi: 10.1038/nrn3381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schutter D.J.L.G., van Honk J. The cerebellum in emotion regulation: a repetitive transcranial magnetic stimulation study. Cerebellum. 2009;8(1):28–34. doi: 10.1007/s12311-008-0056-6. [DOI] [PubMed] [Google Scholar]
- Shin S.H., Chung Y., Rosenberg R.D. Identifying sensitive periods for alcohol use: the roles of timing and chronicity of child physical abuse. Alcohol Clin. Exp. Res. 2016;40(5):1020–1029. doi: 10.1111/acer.13038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singer T. The neuronal basis and ontogeny of empathy and mind reading: review of literature and implications for future research. Neurosci. Biobehav. Rev. 2006;30(6):855–863. doi: 10.1016/j.neubiorev.2006.06.011. [DOI] [PubMed] [Google Scholar]
- Steine I.M., Harvey A.G., Krystal J.H., Milde A.M., Grønli J., Bjorvatn B.…Pallesen S. Sleep disturbances in sexual abuse victims: a systematic review. Sleep Med. Rev. 2012;16(1):15–25. doi: 10.1016/j.smrv.2011.01.006. [DOI] [PubMed] [Google Scholar]
- Tanaka M., Wekerle C., Schmuck M.L., Paglia-Boak A., Team M.A.P.R. The linkages among childhood maltreatment, adolescent mental health, and self-compassion in child welfare adolescents. Child Abuse Negl. 2011;35(10):887–898. doi: 10.1016/j.chiabu.2011.07.003. [DOI] [PubMed] [Google Scholar]
- Taussig H.N., Harpin S.B., Maguire S.A. Suicidality among preadolescent maltreated children in foster care. Child. Maltreat. 2014;19(1):17–26. doi: 10.1177/1077559514525503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terpou B.A., Harricharan S., McKinnon M.C., Frewen P., Jetly R., Lanius R.A. The effects of trauma on brain and body: a unifying role for the midbrain periaqueductal gray. J. Neurosci. Res. 2019;97(9):1110–1140. doi: 10.1002/jnr.24447. [DOI] [PubMed] [Google Scholar]
- Thompson K.L., Hannan S.M., Miron L.R. Fight, flight, and freeze: threat sensitivity and emotion dysregulation in survivors of chronic childhood maltreatment. Pers. Indiv. Differ. 2014;69:28–32. doi: 10.1016/j.paid.2014.05.005. [DOI] [Google Scholar]
- Trickett P.K., Noll J.G., Putnam F.W. The impact of sexual abuse on female development: lessons from a multigenerational, longitudinal research study. Dev. Psychopathol. 2011;23(2):453–476. doi: 10.1017/S0954579411000174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turanovic J.J., Pratt T.C. Longitudinal effects of violent victimization during adolescence on adverse outcomes in adulthood: a focus on prosocial attachments. J. Pediatr. 2015;166(4):1062–1069. doi: 10.1016/j.jpeds.2014.12.059. e1061. [DOI] [PubMed] [Google Scholar]
- Valentino K., Cicchetti D., Rogosch F.A., Toth S.L. Memory, maternal representations, and internalizing symptomatology among abused, neglected, and nonmaltreated children. Child Dev. 2008;79(3):705–719. doi: 10.1111/j.1467-8624.2008.01152.x. CDEV1152 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valentino K., Toth S.L., Cicchetti D. Autobiographical memory functioning among abused, neglected, and nonmaltreated children: the overgeneral memory effect. JCPP (J. Child Psychol. Psychiatry) 2009;50(8):1029–1038. doi: 10.1111/j.1469-7610.2009.02072.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Harmelen A.L., van Tol M.J., Dalgleish T., van der Wee N.J., Veltman D.J., Aleman A.…Elzinga B.M. Hypoactive medial prefrontal cortex functioning in adults reporting childhood emotional maltreatment. Soc. Cognit. Affect Neurosci. 2014;9(12):2026–2033. doi: 10.1093/scan/nsu008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voellmin A., Winzeler K., Hug E., Wilhelm F.H., Schaefer V., Gaab J., Bader K. Blunted endocrine and cardiovascular reactivity in young healthy women reporting a history of childhood adversity. Psychoneuroendocrinology. 2015;51:58–67. doi: 10.1016/j.psyneuen.2014.09.008. [DOI] [PubMed] [Google Scholar]
- Vythilingam M., Heim C., Newport J., Miller A.H., Anderson E., Bronen R., Bremner J.D. Childhood trauma associated with smaller hippocampal volume in women with major depression. Am. J. Psychiatr. 2002;159(12):2072–2080. doi: 10.1176/appi.ajp.159.12.2072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wager T.D., Nichols T.E. Optimization of experimental design in fMRI: a general framework using a genetic algorithm. Neuroimage. 2003;18(2):293–309. doi: 10.1016/s1053-8119(02)00046-0. [DOI] [PubMed] [Google Scholar]
- Wamser-Nanney R., Chesher R.E. Trauma characteristics and sleep impairment among trauma-exposed children. Child Abuse Neglect. 2018;76:469–479. doi: 10.1016/j.chiabu.2017.11.020. [DOI] [PubMed] [Google Scholar]
- Ward R., Calder A.J., Parker M., Arend I. Emotion recognition following human pulvinar damage. Neuropsychologia. 2007;45(8):1973–1978. doi: 10.1016/j.neuropsychologia.2006.09.017. [DOI] [PubMed] [Google Scholar]
- Wechsler D. 2011. WASI-II: Wechsler abbreviated Scale of Intelligence: PsychCorp. [Google Scholar]
- Weller J.A., Fisher P.A. Decision-making deficits among maltreated children. Child. Maltreat. 2013;18(3):184–194. doi: 10.1177/1077559512467846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams A.D., Moulds M.L. An investigation of the cognitive and experiential features of intrusive memories in depression. Memory. 2007;15(8):912–920. doi: 10.1080/09658210701508369. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.