Abstract
Herpesviruses are DNA viruses that infect humans and animals with the ability to induce latent and lytic infections in their hosts, causing critical health complications. The enrolment of nutraceutical anti-herpesvirus drugs in clinical investigations with promising levels of reduced resistance, free or minimal cellular toxicity, and diverse mechanisms of action might be an effective way to defeat challenges that hurdle the progress of anti-herpesvirus drug development, including the problems with drug resistance and recurrent infections. Therefore, in this review, we aim to hunt down all investigations that feature the curative properties of curcumin, a principal bioactive phenolic compound of the spice turmeric, in regard to various human and animal herpesvirus infections and inflammation connected with these diseases. Curcumin was explored with potent antiherpetic actions against herpes simplex virus type 1 and type 2, human cytomegalovirus, Kaposi’s sarcoma-associated herpesvirus, Epstein–Barr virus, bovine herpesvirus 1, and pseudorabies virus. The mechanisms and pathways by which curcumin inhibits anti-herpesvirus activities by targeting multiple steps in herpesvirus life/infectious cycle are emphasized. Improved strategies to overcome bioavailability challenges that limit its use in clinical practice, along with approaches and new directions to enhance the anti-herpesvirus efficacy of this compound, are also reviewed. According to the reviewed studies, this paper presents curcumin as a promising natural drug for the prevention and treatment of herpesvirus infections and their associated inflammatory diseases.
Keywords: curcumin, herpesviruses, phenolics, inflammation, mechanisms and pathways, viral infections, Curcuma longa L.
1. Introduction
Infection with herpesviruses is frequently observed in humans and animals, inducing a range of diseases from moderate uncomplicated mucocutaneous infection to those that are life-threatening [1,2]. Herpesviruses belong to the family of Herpesviridae, which is a large family of DNA viruses with severely contagious properties that use a strategy of infection known as travel and hide [3,4]. These viruses share the feature of forming lifelong infections in a latent phase with the potential of periodic reactivation. During the life cycle, herpesviruses typically infect different cell types in various tissues, and therefore the sub-classification of these pathogens is partially based on their cell and tissue tropism [5,6,7]. So far, the herpesvirus family is divided into three subfamilies, the Alpha-, Beta-, and Gammaherpesvirinae, which include diverse types of herpesvirus known to infect humans. The morbidity and mortality of these viruses are mainly linked with the immunocompromised hosts. However, herpes simplex virus (HSV), varicella-zoster virus (VZV), and Epstein–Barr virus (EBV) were reported with a significant disease burden in people with normal immune function [8,9]. For decades, integrated management of herpesvirus infections remains the main challenge in virology research, where these infections are still incurable due to the viral latency, the problem of recurrent infections, and the resistance to antiherpetic drugs [10,11,12]. Antiviral natural products have gained immense consideration in herpesvirus research after the Nobel Prize in Physiology or Medicine (in 2015) was awarded for the discovery of the natural anti-malarial drug artemisinin. Thus, natural anti-infective agents might pave the road for defeating the problem of drug resistance as well as provide effective and safe treatment of infections caused by herpesviruses [13,14,15,16]. Curcumin, a natural phenolic compound found in the Curcuma longa L. (Zingiberaceae) (turmeric), could help eliminate certain viruses by various mechanisms of action (Figure 1). Therefore, in this review, we aim to collect all in vitro and in vivo studies of curcumin that show its promising inhibitory properties against various types of herpesvirus infections and inflammation linked with these diseases. Moreover, the mechanisms and pathways by which this compound causes anti-herpesvirus effects are discussed. Challenges with its bioavailability and strategies to improve its anti-herpesvirus properties are also reviewed.
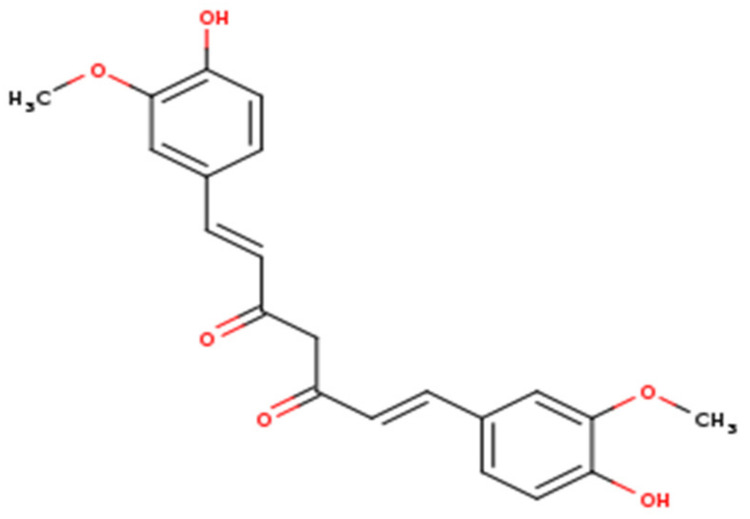
Figure 1.
Chemical structure of curcumin.
Databases such as Web of Science, PubMed, Scopus, SciFinder, Google Scholar, and ScienceDirect were used to perform the literature search, using terms that define curcumin, herpesvirus infections (human and animal herpesviruses), and their associated inflammation. The literature search has covered studies (in vitro and in vivo investigations) published in the years from 1999 to 2020.
2. Curcumin as an Antiviral Agent with Proven Health Benefits
Curcumin is the key component of the yellow pigment and the main bioactive molecule of turmeric [17]. Chemically, this compound belongs to the class of natural phenolic compounds and has been broadly identified in diverse Curcuma spp. In 1910, curcumin was characterized as a symmetrical molecule of two 4-hydroxy-3-methoxyphenyl rings fastened by α,β-unsaturated carbonyl groups [18,19], while its synthesis was defined in 1913 [20]. Curcumin has been employed widely in the traditional medicine systems of various countries and regions in the world [21,22]. Since the complete information about chemical structure and synthesis is acquired, curcumin has been extensively studied in various biological assays and has proven to induce numerous pharmacological and beneficial impacts on human health, including but not limited to the potential treatment of various viral infections such as human immunodeficiency virus, hepatitis B virus, hepatitis C virus, influenza A virus, human papillomavirus, respiratory syncytial virus, arboviruses, and noroviruses [23,24,25,26,27]. Unlike the notable antimicrobial actions, this biomolecule induces several biological effects including but not limited to antioxidant, anti-inflammatory, and anticancer properties [28,29].
3. Anti-Herpesvirus Drugs Used in Clinics
To date, acyclovir and other antiviral nucleoside analogs are used in the first-line treatment strategy for herpesvirus infections. The potent antiherpetic properties of these drugs are related to their ability to block viral replication [30,31]. However, these drugs do not heal the disease while reducing the duration of symptoms and accelerating the recovery of lesions and epithelial damage leading to a return to normal condition. The extensive use of these drugs in the therapy has formed the problem of drug resistance, leading to noticeable treatment failure [32,33,34]. While various ongoing studies have attempted to produce herpesvirus vaccination, no vaccines or antiviral drugs have been approved for the prevention of herpesvirus infections so far [35,36]. Therefore, there is an urgent demand for developing novel antiherpetic drugs that could effectively cure or prevent herpesvirus infections with less resistance, diminished adverse effects, low toxicity, and improved stability [37,38].
4. Role of Curcumin in Inhibition of Herpes Simplex Virus Infections
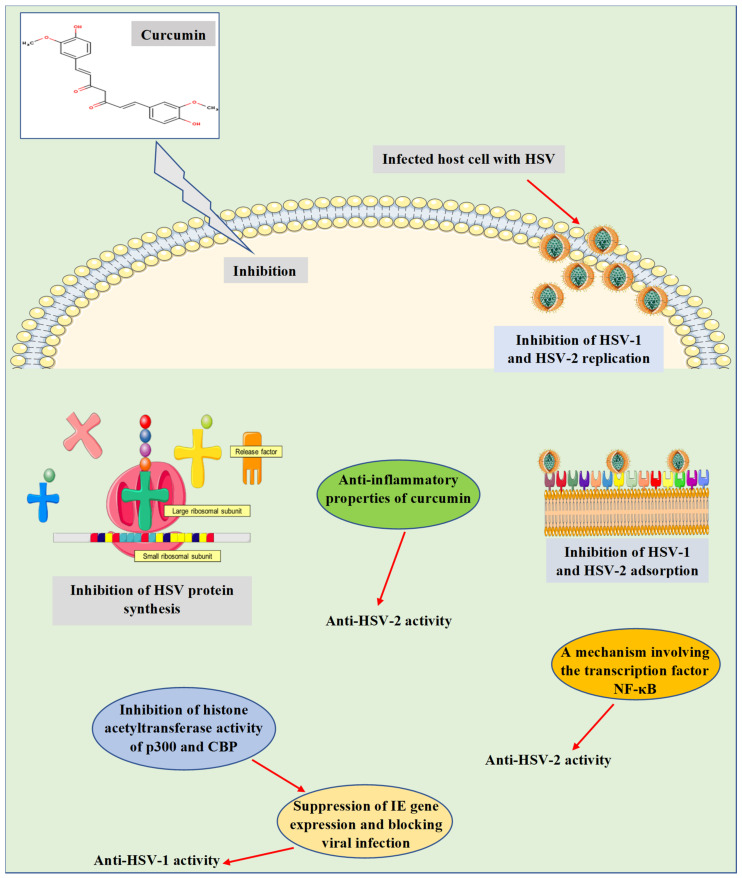
It is known that nonclinical virology studies can assist in dose selection and study design to offer verification of concept and data validating an antiviral claim [39,40]. Consequently, in this section, we aim to embody all available data obtained from nonclinical investigations (in vitro and in vivo studies) that highlight the antiviral properties of curcumin, curcumin metal complexes, and curcumin formulations against the pathogenic herpes simplex virus (HSV) with an emphasis on the effective concentrations or doses and mechanisms of action (Table 1). The alpha-herpesvirus HSV is one of the most prevalent infections in humans, and therefore studies on this virus are intensively performed [41,42]. HSV is classified into two types, HSV-1 and HSV-2. HSV-1 is widely transmitted by oral-to-oral contact to induce oral herpes and in some cases, can also generate genital herpes. Genital herpes is a sexually transmitted disease triggered by HSV-2 [43,44]. Moreover, infection with HSV-2 enhances the risk of transmitting infection with the human immunodeficiency virus (HIV). Most oral and genital herpes infections are asymptomatic and persist lifelong in the host [45,46]. Considering the antiviral capacity of curcumin, defeating the viral infections is one of the main bio-functions reported for this compound via targeting of the viral entry and viral components that are crucial for viral replication as well as other cellular or molecular processes that are involved in the viral life/infectious cycle [47,48,49].
Table 1.
Antiviral activities of curcumin, curcumin metal complexes, and curcumin formulations against herpes simplex virus 1 (HSV-1) and herpes simplex virus 2 (HSV-2) infections.
| Type of Study, Test Performed, Virus, and Cells/Animal Model | Results | Mechanism of Action and Pathway | Reference |
|---|---|---|---|
| In vitro. Plaque assays, ChIP assay, and western blot analysis. HSV-1. HeLa and Vero cells. |
Inhibition of HSV-1 replication and suppression of IE gene expression. | Curcumin was observed to utilize the mechanism independent of the transcriptional coactivator proteins p300/CBP histone acetyltransferase activity to affect the viral transactivator protein VP16-mediated enlistment of RNA polymerase II to IE gene promoters, leading to suppressing gene expression and blocking viral infection. | [50] |
| In vitro and in vivo. Plaque reduction assay. HSV-2. Primary rabbit kidney cells (in vitro). Guinea pig model (in vivo). |
In an in vitro plaque reduction assay, curcumin suppressed the replication of HSV-2 with an ED50 value of 0.32 mg/mL, while at a concentration of 100 mg/mL, the in vivo inhibitory activity was confirmed using a mouse model of genital HSV-2 infection. | The mechanism is unknown. | [51] |
| In vitro. Plaque assay. HSV-2. Human genital epithelial cells. |
In primary human genital epithelial cells, pre-treatment of cells with curcumin (5 µM) decreased HSV-2 shedding by 1000-fold and at a concentration of 50 µM, entirely blocked HSV-2 production. | Investigation of the cellular pathways known to be regulated by curcumin involving the transcription factor NF-κB. | [52] |
| In vitro. Plaque assay and virus adsorption assay. HSV-1 and HSV-2. Vero cells. |
At a concentration of 30 µM, curcumin inhibited the replication of HSV-1 and HSV-2. | Inhibition of adsorption and replication of HSV-1 and HSV-2. | [53] |
| In vivo. Plaque assay. HSV-2. Genital epithelial cells of female C57BL/6 mice. |
Nanoparticle-containing curcumin (0.5 mg) reduced tissue inflammation and the severity of HSV-2 infection in an animal model. | The mechanism of action was detected to be correlated with the anti-inflammatory properties of curcumin. | [54] |
| In vitro. Cytopathic inhibition assay. HSV-1. Vero cells. |
Curcumin, gallium-curcumin, and copper-curcumin inhibited the replication of HSV-1 with IC50 values of 33.0, 13.9, and 23.1 µg/mL, respectively. | The mechanisms of action of both gallium-curcumin and copper-curcumin have been suggested to be investigated in further studies. | [55] |
CBP, CREB-binding protein; ChIP, chromatin immunoprecipitation; ED50, the concentration of drug that decreased the plaque number by 50%; HSV-1, herpes simplex virus 1; HSV-2, herpes simplex virus 2; IC50, 50% inhibitory concentration; IE gene, immediate early gene; NF-κB, nuclear factor kappa B.
Consequently, the mechanisms by which curcumin induces anti-HSV actions were investigated in depth in various in vitro and in vivo studies (Figure 2). These mechanisms were found to relate to the ability of curcumin to hinder a range of cellular and molecular processes, which are essential for viral gene expression, replication, and pathogenesis. For instance, curcumin was detected to inhibit IE gene expression and hindering HSV-1 infection by employing a mechanism independent of the transcriptional coactivator proteins p300/CBP histone acetyltransferase activity to impact the viral transactivator protein VP16-mediated enlistment of RNA polymerase II to IE gene promoters [50]. In preclinical studies (in vitro and in vivo), Bourne et al. [51] stated that curcumin has effectively inhibited the replication of HSV-2. Besides, curcumin was reported to inhibit the replication of HSV-2 through a mechanism involving the transcription factor NF-κB [52]. Additional mechanisms were also verified in an in vitro experiment via inhibiting HSV-1 and HSV-2 adsorption [53]. In an in vivo investigation, the anti-inflammatory properties of curcumin nanoparticles were claimed to be one of the mechanisms responsible for inhibiting the infectivity and replication of HSV-2 [54]. Curcumin metal complexes such as gallium-curcumin and copper-curcumin were studied by Zandi et al. [55], where both complexes were identified to inhibit the replication of HSV-1. However, the mechanism of action has not been revealed, and further investigation needs to be performed. Recently, Treml and colleagues [13] have stated that the hydroxyl groups and phenyl rings of phenolic compounds, including curcumin, are responsible for the induced anti-HSV properties reported for these molecules. This has been probed in various structure-activity relationship analyses.
Figure 2.
Antiviral properties and mechanisms of action of curcumin against herpes simplex virus infections. The presented mechanisms have been explored by in vitro and in vivo studies. CBP, CREB-binding protein; HSV-1, herpes simplex virus 1; HSV-2, herpes simplex virus 2; IE gene, immediate early gene; NF-κB, nuclear factor kappa B.
5. Curcumin Targets Thymidine Kinase Encoded by Herpes Simplex Virus
HSV encodes a set of enzymes that are essential for viral replication along with numerous enzymes involved in nucleotide metabolism that are not necessary for viral replication [2,13]. Such enzymes have been employed in various therapeutic strategies as valuable drug targets useful for the therapy of HSV diseases [2,40,56]. For instance, thymidine kinase (TK) is an essential enzyme that catalyzes the transfer of the gamma-phospho group of adenosine triphosphate (ATP) to thymidine, leading to generate thymidine monophosphate (dTMP). Thus, TK is a critical enzyme in the salvage pathway of pyrimidine synthesis [57]. This enzyme has been detected in both human and HSV with crucial molecular functions [58]. It has been well recognized that HSV-TK, which plays an imperative function in HSV pathogenesis, has a broad substrate specificity including pyrimidines and pyrimidine analogs (deoxycytidine, thymidine, and zidovudine) as well as purine (guanosine) analogs (acyclovir, ganciclovir, penciclovir, and buciclovir) [59,60]. Antiviral drugs that inhibit HSV DNA synthesis (for instance, acyclovir) require HSV-TK to be phosphorylated to monophosphate form. Cellular kinase enzymes then further phosphorylate the drug to the triphosphate form (an active form of the drug). Therefore, HSV TK plays an important role in the design of antiherpetic drugs as a critical mediator protein [61].
Recently, El-Halim et al. [62] have reported in an in silico assay the ability of curcumin to successfully bind to the active site of HSV-1 TK via forming hydrogen bonding and hydrophobic interactions with the amino acid residues of the enzyme active site. The results proposed hydroxyl and carbonyl groups along with phenyl rings of curcumin as functional groups that are accountable for the anti-HSV-1 TK activity. Indeed, the obtained results revealed the mechanism underlying the anti-HSV-1 activity of curcumin through the inhibition of HSV-1 TK. On the other hand, this study might pave the way towards additional investigations, where curcumin could act synergistically with acyclovir and with other nucleoside analogs, leading to improving the treatment efficacy of HSV infections. Unlike the crucial role of TK in HSV pathogenesis, HSV-TK in combination with ganciclovir has been detected to be a promising therapy for melanoma. This has been revealed in an in vivo study using the xenografted melanoma model, where curcumin combined with HSV-TK/ganciclovir efficiently impeded xenografted melanoma growth, which in turn, provided a potential therapeutic approach for improving gene therapy efficacy against skin cancer [63].
6. Role of Curcumin in Inhibition of Various Herpesviruses Infections
In addition to the remarkable antiviral properties against HSV infections, curcumin has been shown to inhibit other human and animal herpesviruses investigated in various assay systems. This section highlights all studies that have reported the antiviral properties of curcumin and curcumin formulations against human cytomegalovirus (HCMV), Kaposi’s sarcoma-associated herpesvirus (KSHV), Epstein–Barr virus (EBV), bovine herpesvirus 1 (BoHV-1), and pseudorabies virus (PRV) infections with a focus on concentrations or doses used along with the mechanisms of action (Table 2).
Table 2.
Antiviral properties of curcumin and curcumin formulations against human cytomegalovirus (HCMV), Kaposi’s sarcoma-associated herpesvirus (KSHV), Epstein–Barr virus (EBV), bovine herpesvirus 1 (BoHV-1), and pseudorabies virus (PRV) infections.
| Herpesvirus and Type of Study | Results | Mechanism of Action and Pathway | Reference |
|---|---|---|---|
| HCMV (in vitro, in vivo, and in silico). | At various concentrations in micromolar ranges, curcumin was detected with anti-HCMV properties. | Inhibition of IEA and UL83A expressions and downregulation of Hsp90. Determination of anti-inflammatory and antioxidant effects as possible mechanisms underlying the anti-HCMV activity. | [66,67,68] |
| KSHV (in vitro). | At various concentrations (in µM), curcumin efficiently inhibited KSHV replication and virus-associated pathogenic properties. | Blocking APE1-mediated redox function. | [73] |
| EBV (in vitro). | Inhibition of EBV reactivation in Raji DR-CAT cells with curcumin treatment (15 µM). | Inhibition of BZLF1 gene transcription. | [77] |
| BoHV-1 (in vitro). | At a concentration of 10 µM, curcumin reduced BoHV-1 titer, leading to inhibiting viral replication. Co-encapsulation of acyclovir and curcumin into three microparticle formulations noticeably reduced the BoVH-1 plaque formation at a concentration of 75 µg/mL. | Inhibition of virus post-binding entry process by upregulating the lipid raft formation. | [80,81] |
| PRV (in vitro). | Treatment with curcumin (30 µM) blocked PRV infectivity in PK-15 cells by decreasing the viral plaque formation. | No mechanism of action was revealed. | [84] |
APE1, apurinic/apyrimidinic endonuclease 1; BoHV-1, bovine herpesvirus 1; EBV, Epstein–Barr virus; HCMV, human cytomegalovirus; Hsp90, heat shock protein 90; IEA, immediate early antigen; KSHV, Kaposi’s sarcoma-associated herpesvirus; PK, porcine kidney; PRV, pseudorabies virus.
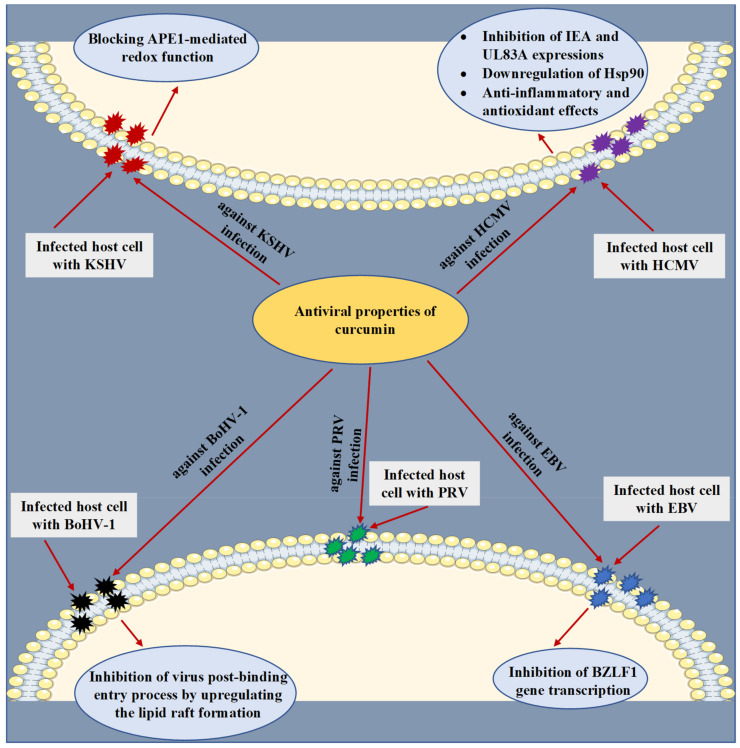
HCMV is a human beta-herpesvirus that causes lifelong infection in the host and has been involved in the development of several diseases in humans, generating severe complications in immunocompromised individuals [64,65]. At various micromolar concentrations, the anti-HCMV activity of curcumin was explored in vitro, in an animal model (Balb/c mice) and in silico experiments. The underlying mechanisms were clarified through the capacity of curcumin to impede HCMV immediate early antigen (IEA) and UL83A expressions [66] and downregulate the heat shock protein 90 (Hsp90) [67] (Figure 3). Through a molecular docking analysis, curcumin was also found to accurately bind to the binding pocket of Hsp90 by establishing crucial contacts such as hydrogen bonds and hydrophobic interactions [67]. Moreover, the anti-inflammatory and antioxidant effects of curcumin were identified as possible mechanisms behind the antiviral action against HCMV [68].
Figure 3.
Antiviral properties and mechanisms of action of curcumin against human cytomegalovirus (HCMV), Kaposi’s sarcoma-associated herpesvirus (KSHV), Epstein–Barr virus (EBV), bovine herpesvirus 1 (BoHV-1), and pseudorabies virus (PRV) infections. The displayed mechanisms have been investigated by in vitro, in vivo, and in silico studies. APE1, apurinic/apyrimidinic endonuclease 1; Hsp90, heat shock protein 90; IEA, immediate early antigen.
KSHV is a gamma-herpesvirus (human herpesvirus 8) known to be one of the DNA tumor viruses involved in the etiology of human cancers [69,70]. It has been reported that the redox function of apurinic/apyrimidinic endonuclease 1 (APE1) plays a crucial role in the replication of KSHV [71,72]. Accordingly, Li et al. [73] examined the antiviral action of curcumin against the replication of KSHV. To reactivate the virus, primary effusive lymphoma (PEL)-BCBL-1 cells were latently infected with the virus and further were treated with 12-O-Tetradecanoyl-phorbol-13-acetate (TPA) and curcumin. The results indicated that treatment with curcumin at a concentration of 30 µM has led to significant blocking of KSHV reactivation by reducing expression of the switch gene replication and transcription activator (RTA) and the delayed-early gene K8. Additionally, with an IC50 value of 8.76 µM and an EC50 value of 6.68 µM, curcumin has notably reduced intracellular and extracellular KSHV genomic DNA levels, respectively. Eventually, the outcome of the study indicated that curcumin diminished the replication of KSHV by blocking APE1-mediated redox function.
EBV is a gamma-herpesvirus known to infect humans that induces mononucleosis and is critically linked with multiple malignancies, including nasopharyngeal carcinoma, Burkitt lymphoma, Hodgkin lymphoma, gastric carcinoma, and posttransplant lymphoproliferative illnesses. Moreover, EBV was detected to be associated with some autoimmune diseases such as systemic sclerosis, multiple sclerosis, and systemic lupus erythematosus [74,75,76]. Hergenhahn and colleagues [77] have proved that curcumin (15 µM) inhibited the EBV reactivation in Raji DR-CAT cells by a mechanism of suppressing BZLF1 gene transcription.
BoHV-1 is an enveloped DNA herpesvirus that infects cattle and causes a severe respiratory infection, leading to significant economic losses for the cattle industry [78,79]. In an in vitro experiment using Madin-Darby Bovine Kidney (MDBK) cells, curcumin (10 µM) substantially reduced viral titer, leading to inhibiting viral replication. The authors suggested that the anti-BoHV-1 activity of curcumin could be related to its capability to upregulate lipid raft formation [80]. On the other hand, co-encapsulation of acyclovir and curcumin into three microparticle formulations composed of the polymers hydroxypropyl methylcellulose, Eudragit® RS100, or both were recently developed by Reolon et al. [81] for improving the antiviral efficacy against BoVH-1 infection. All three microparticle formulations were tested in infected MDBK cells with BoVH-1 and were observed to reduce the viral plaque formation at a concentration of 75 µg/mL.
PRV is an infectious herpesvirus that belongs to the Alphaherpesvirinae subfamily. This virus is an etiological agent of Aujeszky’s disease, a viral infection that primarily affects pigs, causing reproductive and severe neurological complications in affected animals [82,83]. By employing an in vitro assay, the anti-PRV activity of curcumin was studied by infecting the porcine kidney (PK-15) cells with PRV and then treating them with curcumin at a concentration of 30 µM. The research demonstrated that curcumin effectively inhibited the infectivity of PRV by reducing viral plaque formation. On the other hand, the results did not report any mechanism of action [84].
7. Curcumin and Inflammatory Response to Herpesvirus Infections
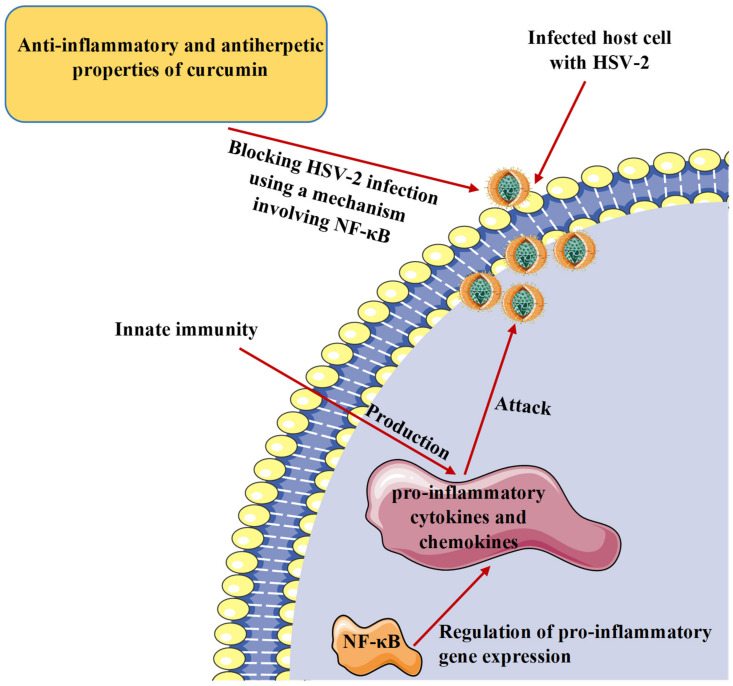
It is recognized that pro-inflammatory cytokines generated by activated innate immune cells are produced as a response to viral infection to stop the pathogen attack and restrict the replication process. However, pro-inflammatory cytokines have a negative effect as they play a vital role in inflammatory diseases of infectious origin [85,86]. The transcription factor nuclear factor kappa B (NF-κB) is known to regulate the expression of various pro-inflammatory genes, including those encoding cytokines and chemokines, and contributes to inflammasome regulation [87,88]. Curcumin has been shown to exhibit anti-inflammatory activities and regulate multiple cell signaling factors such as pro-inflammatory cytokines such as tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6) as well as transcription factors such as NF-κB and activator protein-1 (AP-1) [89,90]. In a study operated on HSV-2, curcumin has successfully blocked the virus infection through a mechanism involving NF-κB [52]. Considering the anti-inflammatory and antiherpetic properties, curcumin could be an effective drug to manage or prevent inflammation associated with herpesvirus infections (Figure 4).
Figure 4.
Protective effect of curcumin against herpes simplex virus and inflammation associated with this infection. HSV-2, herpes simplex virus 2; NF-κB, nuclear factor kappa B.
8. Safety Profile and Reported Undesirable Effects in Clinical Studies
The utilization of curcumin has been confirmed with no toxicity, and the U.S. Food and Drug Administration (FDA) classified it as a ‘’Generally Recognized as Safe’’ (GRAS) compound [91,92]. Based on numerous clinical studies, curcumin has been shown to authenticate an outstanding safety profile and tolerability with no toxicity even at high oral doses of up to 12 g/day [93]. Moreover, the allowable daily intake value (0–3 mg/kg body weight) was recommended and authorized by the Joint Food and Agriculture Organization of the United Nations (FAO)/World Health Organization (WHO) Expert Committee on Food Additives (JECFA) and European Food Safety Authority (EFSA) [94]. Although curcumin has a marked safety record, some clinical observations have documented a few adverse effects on subjects treated with this molecule. For instance, diarrhea, headache, rash, and yellow stool were noted with seven subjects who received 1–12 g of curcumin in a dose-response study and monitored for 72 h [93]. In another clinical investigation, some negative effects such as nausea and diarrhea as well as an accretion in serum alkaline phosphatase and lactate dehydrogenase contents were detected with subjects received 0.45 to 3.6 g/day curcumin for 1–4 months [95].
9. Challenges with Bioavailability and Developed Strategies
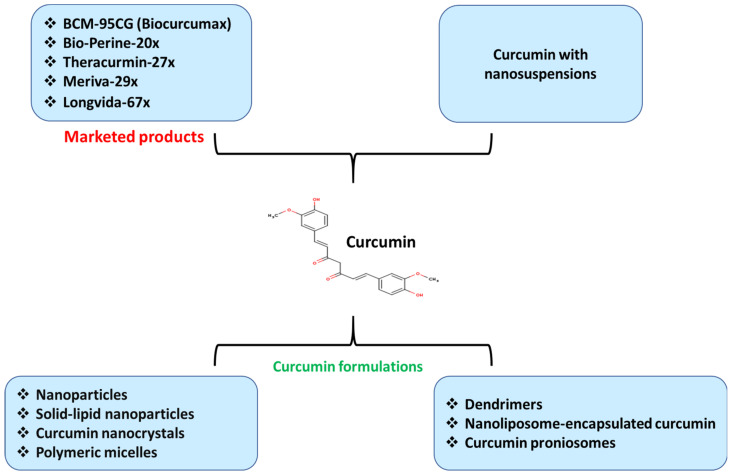
The multipotent biological properties of curcumin against a wide range of diseases, including viral infections, have made this substance a promising drug for the treatment of herpesvirus diseases [96,97]. Nevertheless, curcumin has been reported to have weak bioavailability, which drives to low serum concentrations, diminishing the utilization of its potentially positive therapeutic effects [98]. The low bioavailability has been noted to be correlated with the insolubility in the water (pH = 7) and the potential degradation or crystallization in alkaline and acidic environments, respectively [99]. Over the past 20 years, numerous pharmaceutical approaches were successfully developed to enhance curcumin’s oral bioavailability by producing effective curcumin formulations such as BCM-95CG (Biocurcumax) Bio-Perine-20×, Theracurmin-27×, Meriva-29×, and Longvida-67× [100,101,102,103] (Figure 5). These products are currently accessible in markets with improved absorption and/or bioavailability of curcumin [103].
Figure 5.
Developed drug delivery strategies for enhancing curcumin bioavailability using various types of curcumin formulations assisted by nanotechnology for the treatment of herpesvirus infections and their linked inflammation. Various marketed products with increased absorption and/or bioavailability of curcumin are presented.
It is known that drug delivery is the process of administering a pharmaceutical substance to attain a therapeutic impact on humans or animals, using various approaches, formulations, and technologies, leading to overcoming many obstacles, including low bioavailability and weak absorption. Thus, several other efficient drug delivery systems that combine curcumin with nanosuspensions were generated [104]. Moreover, the recent advances in nanotechnology assisted in solving the challenges that face curcumin drug delivery by exploiting various nano-carriers such as nanoparticles and solid-lipid nanoparticles [105,106,107], curcumin nanocrystals [108], polymeric micelles [109], dendrimers, nanoliposome-encapsulated curcumin, and curcumin proniosomes [110,111]. We highly recommend that readers refer to the cited references to get more detailed information about the mentioned approaches, formulations, and technologies, which were employed to enhance the bioavailability and absorption of curcumin along with the used mechanisms.
10. New Directions for Herpesviruses Treatment
Recently, the CRISPR/Cas9 genome editing technique has been developed as a new strategy that targets viral genetic elements essential for viral fitness. This technique has been developed so that it does not require active replication to perform, and therefore, it is valuable to tackle both productive and latent herpesvirus infections, leading to eradicating viral production from infected cells [112]. This approach has been employed in the genome editing and blocking of latent infections for herpesviruses such as HSV, EBV, KSHV, and HCMV [113,114]. On the other hand, CRISPR/Cas9-mediated genome editing system combined with antiherpetic drugs (natural or synthetic) may significantly change the future of the treatment of herpesviruses and overcome the complications connected with their post-infections.
11. Conclusion and Future Visions
Combating viral diseases, especially those triggered by herpesviruses, has always been a challenge, and the long-term administration of antiherpetic drugs has led to generating various undesirable effects and the problem of drug resistance. Therefore, there is an imperative need for new drug candidates with functional properties to conquer these complications. Plants and their active ingredients are ideal screening libraries for antiviral agents because of their advantages, such as convenient acquisition and low side effects. In recent years, curcumin as a nutraceutical agent has attracted major attention in many research fields due to its great therapeutic potential against various biological targets. Hence, in this paper, we have comprehensively reviewed the curative values of curcumin against numerous animal and human herpesviruses along with the mechanisms by which this compound induces antiherpetic properties, which were examined in vitro and in vivo investigations. Based on several structure-activity relationship studies, hydroxyl groups, carbonyl groups, and phenyl rings of curcumin were observed to be accountable for the induced anti-herpesvirus properties.
Moreover, this paper highlighted several developed strategies to improve curcumin’s bioavailability and antiviral properties against various types of herpesvirus. These approaches made this drug a promising candidate to be involved in clinical studies with enhanced anti-herpesvirus activities. On the other hand, the possible combinatory treatment of curcumin with various antiherpetic nucleoside analogs might be another useful option for the therapy of herpesvirus infections; however, these investigations are still limited or have yet to be proven in vivo.
In conclusion, this review proposes curcumin as a potent and safe drug for the therapy of herpesvirus infections as well as inflammation associated with these infections.
Acknowledgments
The authors would like to thank the Czech University of Life Sciences Prague, Czech Republic for providing the required materials and facilities during the literature search.
Author Contributions
Conceptualization, M.Š. and S.T.S.H.; methodology, S.T.S.H.; validation, M.Š. and S.T.S.H.; formal analysis, M.Š. and S.T.S.H.; investigation, M.Š. and S.T.S.H.; resources, M.Š. and S.T.S.H.; data curation, M.Š. and S.T.S.H.; writing—original draft preparation, M.Š. and S.T.S.H.; writing—review and editing, M.Š. and S.T.S.H.; visualization, M.Š. and S.T.S.H.; supervision, S.T.S.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Brezáni V., Leláková V., Hassan S.T.S., Berchová-Bímová K., Nový P., Klouček P., Maršík P., Dall’Acqua S., Hošek J., Šmejkal K. Anti-Infectivity against Herpes Simplex Virus and Selected Microbes and Anti-Inflammatory Activities of Compounds Isolated from Eucalyptus globulus Labill. Viruses. 2018;10:360. doi: 10.3390/v10070360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hassan S.T.S., Šudomová M., Berchová-Bímová K., Šmejkal K., Echeverría J. Psoromic Acid, a Lichen-Derived Molecule, Inhibits the Replication of HSV-1 and HSV-2, and Inactivates HSV-1 DNA Polymerase: Shedding Light on Antiherpetic Properties. Molecules. 2019;24:2912. doi: 10.3390/molecules24162912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hassan S.T.S., Šudomová M., Masarčíková R. Herpes simplex virus infection: An overview of the problem, pharmacologic therapy and dietary measures. Ceska Slov. Farm. 2017;66:95–102. [PubMed] [Google Scholar]
- 4.Ho D.Y., Enriquez K., Multani A. Herpesvirus Infections Potentiated by Biologics. Infect. Dis. Clin. N. Am. 2020;34:311–339. doi: 10.1016/j.idc.2020.02.006. [DOI] [PubMed] [Google Scholar]
- 5.Roizman B., Pellett P.E. The family Herpesviridae: A brief introduction. In: Knipe D.M., editor. Fields—Virology. 4th ed. Lippincott Williams & Wilkins; Philadelphia, PA, USA: 2001. pp. 2381–2397. [Google Scholar]
- 6.Johnston B.P., McCormick C. Herpesviruses and the Unfolded Protein Response. Viruses. 2019;12:17. doi: 10.3390/v12010017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cohen J.I. Herpesvirus latency. J Clin Investig. 2020;130:3361–3369. doi: 10.1172/JCI136225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pellett P., Roizman B. Herpesviridae. In: Knipe D., Howley P., editors. Fields Virology. Lippincott, Wil-liams and Wilkins; Philadelphia, PA, USA: 2013. pp. 1802–1822. [Google Scholar]
- 9.Savva R. The Essential Co-Option of Uracil-DNA Glycosylases by Herpesviruses Invites Novel Antiviral Design. Microorganisms. 2020;8:461. doi: 10.3390/microorganisms8030461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hassan S.T., Masarčíková R., Berchová K. Bioactive natural products with anti-herpes simplex virus properties. J. Pharm. Pharmacol. 2015;67:1325–1336. doi: 10.1111/jphp.12436. [DOI] [PubMed] [Google Scholar]
- 11.Čulenová M., Sychrová A., Hassan S.T.S., Berchová-Bímová K., Svobodová P., Helclová A., Michnová H., Hošek J., Vasilev H., Suchý P., et al. Multiple In vitro biological effects of phenolic compounds from Morus alba root bark. J. Ethnopharmacol. 2020;248:112296. doi: 10.1016/j.jep.2019.112296. [DOI] [PubMed] [Google Scholar]
- 12.Zheng W., Xu Q., Zhang Y., Xiaofei E., Gao W., Zhang M., Zhai W., Rajkumar R.S., Liu Z. Toll-like receptor-mediated innate immunity against herpesviridae infection: A current perspective on viral infection signaling pathways. Virol. J. 2020;17:192. doi: 10.1186/s12985-020-01463-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Treml J., Gazdová M., Šmejkal K., Šudomová M., Kubatka P., Hassan S.T.S. Natural Products-Derived Chemicals: Breaking Barriers to Novel Anti-HSV Drug Development. Viruses. 2020;12:154. doi: 10.3390/v12020154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hassan S.T.S., Berchová-Bímová K., Petráš J., Hassan K.T.S. Cucurbitacin B interacts synergistically with antibiotics against Staphylococcus aureus clinical isolates and exhibits antiviral activity against HSV-1. S. Afr. J. Bot. 2017;108:90–94. doi: 10.1016/j.sajb.2016.10.001. [DOI] [Google Scholar]
- 15.Andreu S., Ripa I., Bello-Morales R., López-Guerrero J.A. Valproic Acid and Its Amidic Derivatives as New Antivirals against Alphaherpesviruses. Viruses. 2020;12:1356. doi: 10.3390/v12121356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hassan S.T.S. Shedding Light on the Effect of Natural Anti-Herpesvirus Alkaloids on SARS-CoV-2: A Treatment Option for COVID-19. Viruses. 2020;12:476. doi: 10.3390/v12040476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Aggarwal B.B., Kumar A., Bharti A.C. Anticancer potential of curcumin: Preclinical and clinical studies. Anticancer Res. 2003;23:363–398. [PubMed] [Google Scholar]
- 18.Tsuda T. Curcumin as a functional food-derived factor: Degradation products, metabolites, bioactivity, and future perspectives. Food Funct. 2018;9:705–714. doi: 10.1039/C7FO01242J. [DOI] [PubMed] [Google Scholar]
- 19.Milobedeska J., Kostanecki S., Lampe V. Zur kenntnis des curcumins. Ber. Deut. Chem. Ges. 1910;43:2163–2170. doi: 10.1002/cber.191004302168. [DOI] [Google Scholar]
- 20.Lampe V., Milobedeska J. Studien über curcumin. Eur. J. Pharm. Biopharm. 1913;46:2235–2240. doi: 10.1002/cber.191304602149. [DOI] [Google Scholar]
- 21.Zhou Y., Xie M., Song Y., Wang W., Zhao H., Tian Y., Wang Y., Bai S., Zhao Y., Chen X., et al. Two traditional Chinese medicines Curcumae radix and Curcumae Rhizoma: An ethnopharmacology, phytochemistry, and pharmacology review. Evid. Based Complement. Altern. Med. 2016;2016:4973128. doi: 10.1155/2016/4973128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Seidi Damyeh M., Mereddy R., Netzel M.E., Sultanbawa Y. An insight into curcumin-based photosensitization as a promising and green food preservation technology. Compr. Rev. Food Sci. Food Saf. 2020;19:1727–1759. doi: 10.1111/1541-4337.12583. [DOI] [PubMed] [Google Scholar]
- 23.Soleimani V., Sahebkar A., Hosseinzadeh H. Turmeric (Curcuma longa) and its major constituent (curcumin) as nontoxic and safe substances: Review. Phytother. Res. 2018;32:985–995. doi: 10.1002/ptr.6054. [DOI] [PubMed] [Google Scholar]
- 24.Praditya D., Kirchhoff L., Brüning J., Rachmawati H., Steinmann J., Steinmann E. Anti-infective Properties of the Golden Spice Curcumin. Front Microbiol. 2019;10:912. doi: 10.3389/fmicb.2019.00912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hewlings S.J., Kalman D.S. Curcumin: A Review of Its’ Effects on Human Health. Foods. 2017;6:92. doi: 10.3390/foods6100092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kotha R.R., Luthria D.L. Curcumin: Biological, Pharmaceutical, Nutraceutical, and Analytical Aspects. Molecules. 2019;24:2930. doi: 10.3390/molecules24162930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Moghadamtousi S.Z., Kadir H.A., Hassandarvish P., Tajik H., Abubakar S., Zandi K. A review on antibacterial, antiviral, and antifungal activity of curcumin. Biomed Res Int. 2014;2014:186864. doi: 10.1155/2014/186864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jha A., Mohapatra P.P., AlHarbi S.A., Jahan N. Curcumin: Not So Spicy After All. Mini Rev. Med. Chem. 2017;17:1425–1434. doi: 10.2174/1389557517666170228114234. [DOI] [PubMed] [Google Scholar]
- 29.Di Meo F., Margarucci S., Galderisi U., Crispi S., Peluso G. Curcumin, Gut Microbiota, and Neuroprotection. Nutrients. 2019;11:2426. doi: 10.3390/nu11102426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Birkmann A., Zimmermann H. HSV antivirals—Current and future treatment options. Curr. Opin. Virol. 2016;18:9–13. doi: 10.1016/j.coviro.2016.01.013. [DOI] [PubMed] [Google Scholar]
- 31.Kenny K., Leung W., Stephanson K., Ross S. Clinical practice in prevention of neonatal HSV infection: A survey of obstetrical care providers in Alberta. J. Obstet. Gynaecol. Can. 2013;35:131–137. doi: 10.1016/S1701-2163(15)31017-3. [DOI] [PubMed] [Google Scholar]
- 32.Piret J., Boivin G. Antiviral resistance in herpes simplex virus and varicella-zoster virus infections: Diagnosis and management. Curr. Opin. Infect Dis. 2016;29:654–662. doi: 10.1097/QCO.0000000000000288. [DOI] [PubMed] [Google Scholar]
- 33.Rechenchoski D.Z., Faccin-Galhardi L.C., Linhares R.E.C., Nozawa C. Herpesvirus: An underestimated virus. Folia Microbiol. 2017;62:151–156. doi: 10.1007/s12223-016-0482-7. [DOI] [PubMed] [Google Scholar]
- 34.Britt W.J., Prichard M.N. New therapies for human cytomegalovirus infections. Antivir. Res. 2018;159:153–174. doi: 10.1016/j.antiviral.2018.09.003. [DOI] [PubMed] [Google Scholar]
- 35.Maple P.A.C. Cytomegalovirus and Epstein-Barr Virus Associations with Neurological Diseases and the Need for Vaccine Development. Vaccines. 2020;8:35. doi: 10.3390/vaccines8010035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bonizzoli M., Arvia R., di Valvasone S., Liotta F., Zakrzewska K., Azzi A., Peris A. Human herpesviruses respiratory infections in patients with acute respiratory distress (ARDS) Med. Microbiol. Immunol. 2016;205:371–379. doi: 10.1007/s00430-016-0456-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Annunziata G., Maisto M., Schisano C., Ciampaglia R., Narciso V., Tenore G.C., Novellino E. Resveratrol as a Novel Anti-Herpes Simplex Virus Nutraceutical Agent: An Overview. Viruses. 2018;10:473. doi: 10.3390/v10090473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zinser E., Krawczyk A., Mühl-Zürbes P., Aufderhorst U., Draßner C., Stich L., Zaja M., Strobl S., Steinkasserer A., Heilingloh C.S. A new promising candidate to overcome drug resistant herpes simplex virus infections. Antivir. Res. 2018;149:202–210. doi: 10.1016/j.antiviral.2017.11.012. [DOI] [PubMed] [Google Scholar]
- 39.Adler B., Sattler C., Adler H. Herpesviruses and Their Host Cells: A Successful Liaison. Trends Microbiol. 2017;25:229–241. doi: 10.1016/j.tim.2016.11.009. [DOI] [PubMed] [Google Scholar]
- 40.Hassan S.T.S. Brassicasterol with Dual Anti-Infective Properties against HSV-1 and Mycobacterium tuberculosis, and Cardiovascular Protective Effect: Nonclinical in Vitro and In Silico Assessments. Biomedicines. 2020;8:132. doi: 10.3390/biomedicines8050132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Memish Z.A., Almasri M., Chentoufi A.A., Al-Tawfiq J.A., Al-Shangiti A.M., Al-Kabbani K.M., Otaibi B., Assirri A., Yezli S. Seroprevalence of Herpes Simplex Virus Type 1 and Type 2 and Coinfection with HIV and Syphilis: The First National Seroprevalence Survey in Saudi Arabia. Sex. Trans. Dis. 2015;42:526–532. doi: 10.1097/OLQ.0000000000000336. [DOI] [PubMed] [Google Scholar]
- 42.Ma W., He H., Wang H. Oncolytic herpes simplex virus and immunotherapy. BMC Immunol. 2018;19:40. doi: 10.1186/s12865-018-0281-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hassan S.T.S., Švajdlenka E., Berchová-Bímová K. Hibiscus sabdariffa L. and Its Bioactive Constituents Exhibit Antiviral Activity against HSV-2 and Anti-enzymatic Properties against Urease by an ESI-MS Based Assay. Molecules. 2017;22:722. doi: 10.3390/molecules22050722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Johnston C., Corey L. Current Concepts for Genital Herpes Simplex Virus Infection: Diagnostics and Pathogenesis of Genital Tract Shedding. Clin. Microbiol. Rev. 2016;29:149–161. doi: 10.1128/CMR.00043-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Widener R.W., Whitley R.J. Herpes simplex virus. Handb. Clin. Neurol. 2014;123:251–263. doi: 10.1016/B978-0-444-53488-0.00011-0. [DOI] [PubMed] [Google Scholar]
- 46.Goins W.F., Hall B., Cohen J.B., Glorioso J.C. Retargeting of herpes simplex virus (HSV) vectors. Curr. Opin. Virol. 2016;21:93–101. doi: 10.1016/j.coviro.2016.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Joe B., Vijaykumar M., Lokesh B.R. Biological properties of curcumin-cellular and molecular mechanisms of action. Crit. Rev. Food Sci. Nutr. 2004;44:97–111. doi: 10.1080/10408690490424702. [DOI] [PubMed] [Google Scholar]
- 48.Sharma R.A., Gescher A.J., Steward W.P. Curcumin: The story so far. Eur. J. Cancer. 2005;41:1955–1968. doi: 10.1016/j.ejca.2005.05.009. [DOI] [PubMed] [Google Scholar]
- 49.Dai J., Gu L., Su Y., Wang Q., Zhao Y., Chen X., Deng H., Li W., Wang G., Li K. Inhibition of curcumin on influenza A virus infection and influenzal pneumonia via oxidative stress, TLR2/4, p38/JNK MAPK and NF-κB pathways. Int. Immunopharmacol. 2018;54:177–187. doi: 10.1016/j.intimp.2017.11.009. [DOI] [PubMed] [Google Scholar]
- 50.Kutluay S.B., Doroghazi J., Roemer M.E., Triezenberg S.J. Curcumin inhibits herpes simplex virus immediate-early gene expression by a mechanism independent of p300/CBP histone acetyltransferase activity. Virology. 2008;373:239–247. doi: 10.1016/j.virol.2007.11.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bourne K.Z., Bourne N., Reising S.F., Stanberry L.R. Plant products as topical microbicide candidates: Assessment of in vitro and in vivo activity against herpes simplex virus type 2. Antivir. Res. 1999;42:219–226. doi: 10.1016/S0166-3542(99)00020-0. [DOI] [PubMed] [Google Scholar]
- 52.Ferreira V.H., Nazli A., Dizzell S.E., Mueller K., Kaushic C. The anti-inflammatory activity of curcumin protects the genital mucosal epithelial barrier from disruption and blocks replication of HIV-1 and HSV-2. PLoS ONE. 2015;10:e0124903. doi: 10.1371/journal.pone.0124903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Flores D.J., Lee L.H., Adams S.D. Inhibition of Curcumin-Treated Herpes Simplex Virus 1 and 2 in Vero Cells. Adv. Microbiol. 2016;6:276–287. doi: 10.4236/aim.2016.64027. [DOI] [Google Scholar]
- 54.Vitali D., Bagri P., Wessels J.M., Arora M., Ganugula R., Parikh A., Mandur T., Felker A., Garg S., Kumar M.R., et al. Curcumin Can Decrease Tissue Inflammation and the Severity of HSV-2 Infection in the Female Reproductive Mucosa. Int. J. Mol. Sci. 2020;21:337. doi: 10.3390/ijms21010337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zandi K., Ramedani E., Mohammadi K., Tajbakhsh S., Deilami I., Rastian Z., Fouladvand M., Yousefi F., Farshadpour F. Evaluation of antiviral activities of curcumin derivatives against HSV-1 in Vero cell line. Nat. Prod. Commun. 2010;5:1935–1938. doi: 10.1177/1934578X1000501220. [DOI] [PubMed] [Google Scholar]
- 56.Poole C.L., James S.H. Antiviral Therapies for Herpesviruses: Current Agents and New Directions. Clin. Ther. 2018;40:1282–1298. doi: 10.1016/j.clinthera.2018.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Hannigan B.M., Barnett Y.A., Armstrong D.B., McKelvey-Martin V.J., McKenna P.G. Thymidine kinases: The enzymes and their clinical usefulness. Cancer Biother. 1993;8:189–197. doi: 10.1089/cbr.1993.8.189. [DOI] [PubMed] [Google Scholar]
- 58.Fujii H., Harada S., Yoshikawa T., Yamada S., Omura N., Shibamura M., Inagaki T., Kato H., Fukushi S., Saijo M. Differences in the Likelihood of Acyclovir Resistance-Associated Mutations in the Thymidine Kinase Genes of Herpes Simplex Virus 1 and Varicella-Zoster Virus. Antimicrob. Agents Chemother. 2019;63:e00017–e00019. doi: 10.1128/AAC.00017-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Coen N., Duraffour S., Haraguchi K., Balzarini J., van den Oord J.J., Snoeck R., Andrei G. Antiherpesvirus activities of two novel 4′-thiothymidine derivatives, KAY-2-41 and KAH-39-149, are dependent on viral and cellular thymidine kinases. Antimicrob. Agents Chemother. 2014;58:4328–4340. doi: 10.1128/AAC.02825-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Topalis D., Gillemot S., Snoeck R., Andrei G. Thymidine kinase and protein kinase in drug-resistant herpesviruses: Heads of a Lernaean Hydra. Drug Resist. Updat. 2018;37:1–16. doi: 10.1016/j.drup.2018.01.003. [DOI] [PubMed] [Google Scholar]
- 61.Xie Y., Wu L., Wang M., Cheng A., Yang Q., Wu Y., Jia R., Zhu D., Zhao X., Chen S., et al. Alpha-Herpesvirus Thymidine Kinase Genes Mediate Viral Virulence and Are Potential Therapeutic Targets. Front. Microbiol. 2019;10:941. doi: 10.3389/fmicb.2019.00941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.El-Halim S.M.A., Mamdouh M.A., El-Haddad A.E., Soliman S.M. Fabrication of Anti-HSV-1 Curcumin Stabilized Nanostructured Proniosomal Gel: Molecular Docking Studies on Thymidine Kinase Proteins. Sci. Pharm. 2020;88:9. doi: 10.3390/scipharm88010009. [DOI] [Google Scholar]
- 63.Li H., Du H., Zhang G., Wu Y., Qiu P., Liu J., Guo J., Liu X., Sun L., Du B., et al. Curcumin plays a synergistic role in combination with HSV-TK/GCV in inhibiting growth of murine B16 melanoma cells and melanoma xenografts. PeerJ. 2019;7:e7760. doi: 10.7717/peerj.7760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Griffiths P., Baraniak I., Reeves M. The pathogenesis of human cytomegalovirus. J. Pathol. 2015;235:288–297. doi: 10.1002/path.4437. [DOI] [PubMed] [Google Scholar]
- 65.Dooley A.L., O’Connor C.M. Regulation of the MIE Locus during HCMV Latency and Reactivation. Pathogens. 2020;9:869. doi: 10.3390/pathogens9110869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lv Y., An Z., Chen H., Wang Z., and Liu L. Mechanism of curcumin resistance to human cytomegalovirus in HELF cells. BMC Complement. Altern. Med. 2014;14:284. doi: 10.1186/1472-6882-14-284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lv Y., Gong L., Wang Z., Han F., Liu H., Lu X., Liu L. Curcumin inhibits human cytomegalovirus by downregulating heat shock protein 90. Mol. Med. Rep. 2015;12:4789–4793. doi: 10.3892/mmr.2015.3983. [DOI] [PubMed] [Google Scholar]
- 68.Lv Y., Lei N., Wang D., An Z., Li G., Han F., Liu H., Liu L. Protective effect of curcumin against cytomegalovirus infection in Balb/c mice. Environ. Toxicol. Pharmacol. 2014;37:140–147. doi: 10.1016/j.etap.2014.04.017. [DOI] [PubMed] [Google Scholar]
- 69.Guito J., Lukac D.M. KSHV Rta Promoter Specification and Viral Reactivation. Front. Microbiol. 2012;3:30. doi: 10.3389/fmicb.2012.00030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hussein H.A.M., Alfhili M.A., Pakala P., Simon S., Hussain J., McCubrey J.A., Akula S.M. miRNAs and their roles in KSHV pathogenesis. Virus Res. 2019;266:15–24. doi: 10.1016/j.virusres.2019.03.024. [DOI] [PubMed] [Google Scholar]
- 71.Zhong C., Xu M., Wang Y., Xu J., Yuan Y. An APE1 inhibitor reveals critical roles of the redox function of APE1 in KSHV replication and pathogenic phenotypes. PLoS Pathog. 2017;13:e1006289. doi: 10.1371/journal.ppat.1006289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hu J., Wang Y., Yuan Y. Inhibitors of APE1 redox function effectively inhibit γ-herpesvirus replication in vitro and in vivo. Antivir. Res. 2020;185:104985. doi: 10.1016/j.antiviral.2020.104985. [DOI] [PubMed] [Google Scholar]
- 73.Li H., Zhong C., Wang Q., Chen W., Yuan Y. Curcumin is an APE1 redox inhibitor and exhibits an antiviral activity against KSHV replication and pathogenesis. Antivir. Res. 2019;167:98–103. doi: 10.1016/j.antiviral.2019.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Kanda T. EBV-Encoded Latent Genes. Adv. Exp. Med. Biol. 2018;1045:377–394. doi: 10.1007/978-981-10-7230-7_17. [DOI] [PubMed] [Google Scholar]
- 75.Farina A., Cirone M., York M., Lenna S., Padilla C., Mclaughlin S., Faggioni A., Lafyatis R., Trojanowska M., Farina G.A. Epstein-Barr virus infection induces aberrant TLR activation pathway and fibroblast-myofibroblast conversion in scleroderma. J. Investig. Dermatol. 2014;134:954–964. doi: 10.1038/jid.2013.423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ascherio A., Munger K.L. EBV and Autoimmunity. Curr. Top. Microbiol. Immunol. 2015;390:365–385. doi: 10.1007/978-3-319-22822-8_15. [DOI] [PubMed] [Google Scholar]
- 77.Hergenhahn M., Soto U., Weninger A., Polack A., Hsu C.H., Cheng A.L., Rosl F. The chemopreventive compound curcumin is an efficient inhibitor of Epstein-Barr virus BZLF1 transcription in Raji DR-LUC cells. Mol. Carcinog. 2002;33:137–145. doi: 10.1002/mc.10029. [DOI] [PubMed] [Google Scholar]
- 78.Zhu L., Ding X., Tao J., Wang J., Zhao X., Zhu G. Critical role of cholesterol in bovine herpesvirus type 1infection of MDBK cells. Vet. Microbiol. 2010;144:51–57. doi: 10.1016/j.vetmic.2009.12.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Teles A.V., Oliveira T.M.A., Bezerra F.C., Alonso L., Alonso A., Borissevitch I.E., Gonçalves P.J., Souza G.R.L. Photodynamic inactivation of Bovine herpesvirus type 1 (BoHV-1) by porphyrins. J. Gen. Virol. 2018;99:1301–1306. doi: 10.1099/jgv.0.001121. [DOI] [PubMed] [Google Scholar]
- 80.Zhu L., Ding X., Zhang D., Yuan C., Wang J., Ndegwa E., Zhu G. Curcumin inhibits bovine herpesvirus type 1 entry into MDBK cells. Acta Virol. 2015;59:221–227. doi: 10.4149/av_2015_03_221. [DOI] [PubMed] [Google Scholar]
- 81.Reolon J.B., Brustolin M., Accarini T., Viçozzi G.P., Sari M.H.M., Bender E.A., Haas S.E., Brum M.C.S., Gündel A., Colomé L.M. Co-encapsulation of acyclovir and curcumin into microparticles improves the physicochemical characteristics and potentiates in vitro antiviral action: Influence of the polymeric composition. Eur. J. Pharm. Sci. 2019;131:167–176. doi: 10.1016/j.ejps.2019.02.019. [DOI] [PubMed] [Google Scholar]
- 82.Pomeranz L.E., Reynolds A.E., Hengartner C.J. Molecular biology of pseudorabies virus: Impact on neurovirology and veterinary medicine. Microbiol. Mol. Biol. Rev. 2005;69:462–500. doi: 10.1128/MMBR.69.3.462-500.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Freuling C.M., Müller T.F., Mettenleiter T.C. Vaccines against pseudorabies virus (PrV) Vet. Microbiol. 2017;206:3–9. doi: 10.1016/j.vetmic.2016.11.019. [DOI] [PubMed] [Google Scholar]
- 84.Chen T.Y., Chen D.Y., Wen H.W., Ou J.L., Chiou S.S., Chen J.M., Wong M.L., Hsu W.L. Inhibition of enveloped viruses infectivity by curcumin. PLoS ONE. 2013;8:e62482. doi: 10.1371/journal.pone.0062482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Liu T., Zhang L., Joo D., Sun S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017;2:17023. doi: 10.1038/sigtrans.2017.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Carty M., Guy C., Bowie A.G. Detection of viral infections by innate immunity. Biochem. Pharmacol. 2020;183:114316. doi: 10.1016/j.bcp.2020.114316. [DOI] [PubMed] [Google Scholar]
- 87.Prasad S., Gupta S.C., Tyagi A.K., Aggarwal B.B. Curcumin, a component of golden spice: From bedside to bench and back. Biotechnol. Adv. 2014;32:1053–1064. doi: 10.1016/j.biotechadv.2014.04.004. [DOI] [PubMed] [Google Scholar]
- 88.Ghosh S., Banerjee S., Sil P.C. The beneficial role of curcumin on inflammation, diabetes and neurodegenerative disease: A recent update. Food Chem. Toxicol. 2015;83:111–124. doi: 10.1016/j.fct.2015.05.022. [DOI] [PubMed] [Google Scholar]
- 89.Aggarwal B.B., Sung B. Pharmacological basis for the role of curcumin in chronic diseases: An age-old spice with modern targets. Trends Pharmacol. Sci. 2009;30:85–94. doi: 10.1016/j.tips.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 90.Boyanapalli S.S.S., Huang Y., Su Z., Cheng D., Zhang C., Guo Y., Rao R., Androulakis I.P., Kong A.N. Pharmacokinetics and Pharmacodynamics of Curcumin in regulating anti-inflammatory and epigenetic gene expression. Biopharm. Drug Dispos. 2018;39:289–297. doi: 10.1002/bdd.2136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Gupta S.C., Patchva S., Aggarwal B.B. Therapeutic Roles of Curcumin: Lessons Learned from Clinical Trials. AAPS J. 2013;5:195–218. doi: 10.1208/s12248-012-9432-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Yang Z., Liu W., Zhou X., Zhu X., Suo F., Yao S. The effectiveness and safety of curcumin as a complementary therapy in inflammatory bowel disease: A protocol of systematic review and meta-analysis. Medicine. 2020;99:e22916. doi: 10.1097/MD.0000000000022916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lao C.D., Ruffin M.T., Normolle D., Heath D.D., Murray S.I., Bailey J.M., Boggs M.E., Crowell J., Rock C.L., Brenner D.E. Dose escalation of a curcuminoid formulation. BMC Complement. Altern. Med. 2006;6:10. doi: 10.1186/1472-6882-6-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Kocaadam B., Sanlier N. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Crit. Rev. Food Sci. Nutr. 2017;57:2889–2895. doi: 10.1080/10408398.2015.1077195. [DOI] [PubMed] [Google Scholar]
- 95.Sharma R.A., Euden S.A., Platton S.L., Cooke D.N., Shafayat A., Hewitt H.R., Marczylo T.H., Morgan B., Hemingway D., Plummer S.M. Phase I clinical trial of oral curcumin: Biomarkers of systemic activity and compliance. Clin. Cancer Res. 2004;10:6847–6854. doi: 10.1158/1078-0432.CCR-04-0744. [DOI] [PubMed] [Google Scholar]
- 96.Jennings M.R., Parks R.J. Curcumin as an Antiviral Agent. Viruses. 2020;12:1242. doi: 10.3390/v12111242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Yang B., Luo G., Zhang C., Feng L., Luo X., Gan L. Curcumin protects rat hippocampal neurons against pseudorabies virus by regulating the BDNF/TrkB pathway. Sci. Rep. 2020;10:22204. doi: 10.1038/s41598-020-78903-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Liu W., Zhai Y., Heng X., Che F.Y., Chen W., Sun D., Zhai G. Oral bioavailability of curcumin: Problems and advancements. Drug Target. 2016;24:694–702. doi: 10.3109/1061186X.2016.1157883. [DOI] [PubMed] [Google Scholar]
- 99.Lopresti A.L. The Problem of Curcumin and Its Bioavailability: Could Its Gastrointestinal Influence Contribute to Its Overall Health-Enhancing Effects? Adv. Nutr. 2018;9:41–50. doi: 10.1093/advances/nmx011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Antony B., Merina B., Iyer V.S., Judy N., Lennertz K., Joyal S. A pilot crossover study to evaluate human oral bioavailability of BCM-95CG (Biocurcumax), a novel bioenhanced preparation of curcumin. Indian J. Pharm. Sci. 2008;70:445–449. doi: 10.4103/0250-474X.44591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Allegri P., Mastromarino A., Neri P. Management of chronic anterior uveitis relapses: Efficacy of oral phospholipidic curcumin treatment. Long-term follow-up. Clin. Ophthalmol. 2010;4:1201–1206. doi: 10.2147/OPTH.S13271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Kurita T., Makino Y. Novel curcumin oral delivery systems. Anticancer Res. 2013;33:2807–2821. [PubMed] [Google Scholar]
- 103.McFarlin B.K., Venable A.S., Henning A.L., Sampson J.N., Pennel K., Vingren J.L., Hill D.W. Reduced inflammatory and muscle damage biomarkers following oral supplementation with bioavailable curcumin. BBA Clin. 2016;5:72–78. doi: 10.1016/j.bbacli.2016.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Mathew D., Hsu W.L. Antiviral potential of curcumin. J. Funct. Foods. 2018;40:692–699. doi: 10.1016/j.jff.2017.12.017. [DOI] [Google Scholar]
- 105.Li X., Chen S., Zhang B., Li M., Diao K., Zhang Z., Li J., Xu Y., Wang X., Chen H. In situ injectable nano-composite hydrogel composed of curcumin, N,O-carboxymethyl chitosan and oxidized alginate for wound healing application. Int. J. Pharm. 2012;437:110–119. doi: 10.1016/j.ijpharm.2012.08.001. [DOI] [PubMed] [Google Scholar]
- 106.Misra R., Sahoo S.K. Coformulation of doxorubicin and curcumin in poly(D, Llactide- co-glycolide) nanoparticles suppresses the development of multidrug resistance in K562 cells. Mol. Pharm. 2011;8:852–866. doi: 10.1021/mp100455h. [DOI] [PubMed] [Google Scholar]
- 107.Sun M., Gao Y., Guo C.Y., Cao F.L., Song Z.M., Xi Y.W., Yu A., Li A., Zhai G. Enhancement of transport of curcumin to brain in mice by poly(n-butylcyanoacrylate) nanoparticle. J. Nanopart. Res. 2010;12:3111–3122. doi: 10.1007/s11051-010-9907-4. [DOI] [Google Scholar]
- 108.Onoue S., Takahashi H., Kawabata Y., Seto Y., Hatanaka J., Timmermann B., Yamada S. Formulation design and photochemical studies on nanocrystal solid dispersion of curcumin with improved oral bioavailability. J. Pharm. Sci. 2010;99:1871–1881. doi: 10.1002/jps.21964. [DOI] [PubMed] [Google Scholar]
- 109.Gou M., Men K., Shi H., Xiang M., Zhang J., Song J., Long J., Wan Y., Luo F., Zhao X., et al. Curcumin loaded biodegradable polymeric micelles for colon cancer therapy in vitro and in vivo. Nanoscale. 2011;3:1558–1567. doi: 10.1039/c0nr00758g. [DOI] [PubMed] [Google Scholar]
- 110.Thangapazham R.L., Puri A., Tele S., Blumenthal R., Maheshwari R.K. Evaluation of a nanotechnology-based carrier for delivery of curcumin in prostate cancer cells. Int. J. Oncol. 2008;32:1119–1123. doi: 10.3892/ijo.32.5.1119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Aboali F.A., Habib D.A., Elbedaiwy H.M., Farid R.M. Curcumin-loaded proniosomal gel as a biofreindly alternative for treatment of ocular inflammation: In-vitro and in-vivo assessment. Int. J. Pharm. 2020;589:119835. doi: 10.1016/j.ijpharm.2020.119835. [DOI] [PubMed] [Google Scholar]
- 112.Van Diemen F.R., Kruse E.M., Hooykaas M.J., Bruggeling C.E., Schürch A.C., van Ham P.M., Imhof S.M., Nijhuis M., Wiertz E.J., Lebbink R.J. CRISPR/Cas9-Mediated Genome Editing of Herpesviruses Limits Productive and Latent Infections. PLoS Pathog. 2016;12:e1005701. doi: 10.1371/journal.ppat.1005701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Van Diemen F.R., Lebbink R.J. CRISPR/Cas9, a powerful tool to target human herpesviruses. Cell Microbiol. 2017;19:2. doi: 10.1111/cmi.12694. [DOI] [PubMed] [Google Scholar]
- 114.Chen Y.C., Sheng J., Trang P., Liu F. Potential Application of the CRISPR/Cas9 System against Herpesvirus Infections. Viruses. 2018;10:291. doi: 10.3390/v10060291. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.