Abstract
The coronavirus disease 2019 (COVID-19) triggered by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) erupted in Hubei Province of China in December 2019 and has become a pandemic. Severe COVID-19 patients who suffer from acute respiratory distress syndrome (ARDS) and multi-organ dysfunction have high mortality. Several studies have shown that this is closely related to the cytokine release syndrome (CRS), often loosely referred to as cytokine storm. IL-6 is one of the key factors and its level is positively correlated with the severity of the disease. The molecular mechanisms for CRS in COVID-19 are related to the effects of the S-protein and N-protein of the virus and its ability to trigger NF-κB activation by disabling the inhibitory component IκB. This leads to activation of immune cells and the secretion of proinflammatory cytokines such as IL-6 and TNF-α. Other mechanisms related to IL-6 include its interaction with GM-CSF and interferon responses. The pivotal role of IL-6 makes it a target for therapeutic agents and studies on tocilizumab are already ongoing. Other possible targets of treating CRS in COVID-19 include IL-1β and TNF-α. Recently, reports of a CRS like illness called multisystem inflammatory syndrome in children (MIS-C) in children have surfaced, with a variable presentation which in some cases resembles Kawasaki disease. It is likely that the immunological derangement and cytokine release occurring in COVID-19 cases is variable, or on a spectrum, that can potentially be governed by genetic factors. Currently, there are no approved biological modulators for the treatment of COVID-19, but the urgency of the pandemic has led to numerous clinical trials worldwide. Ultimately, there is great promise that an anti-inflammatory modulator targeting a cytokine storm effect may prove to be very beneficial in reducing morbidity and mortality in COVID-19 patients.
Keywords: COVID-19, cytokine release syndrome, IL-6, hemophagocytic lymphohistiocytosis (HLH), Kawasaki disease, multisystem inflammatory syndrome in children (MIS-C), NF-κB, SARS-CoV-2
Introduction
COVID-19 is a pandemic and as of October 30, 2020, the SARS-CoV-2 virus has infected over 45,218,361 people and caused over 1184,072 deaths worldwide. SARS-CoV-2 primarily affects the lung and, in some individuals, can progress to acute respiratory distress syndrome (ARDS). Once this occurs, the disease can rapidly progress to multi-organ dysfunction syndrome (MODS) and death. The pathogenesis of lung injury and MODS in COVID-19 has not been fully elucidated, but it is suspected that a hyper-inflammatory state may occur in the form of cytokine release syndrome (CRS).
The term "cytokine release syndrome” generally refers to a severely over-reactive immune system that progresses in an unregulated manner. We now know that unfettered cytokine release can occur in many conditions, including infections, but the first introduction of the term was in a paper by Ferrera et al describing the effects of cytokine dysregulation in graft-versus-host disease (GVHD) in 1993 [1]. Since the early 2000s, this term has also been used to describe abnormal release of large quantities of inflammatory mediators in infectious diseases caused by cytomegalovirus, Epstein-Barr virus, SARS-CoV and H5N1 influenza virus [2]. CRS may be a feature of infection related secondary hemophagocytic lymphohistiocytosis (HLH) and is thought to be associated with acute lung injury (ALI) and ARDS occurring in SARS and the Middle East Respiratory Syndrome (MERS). Although this term has gained recognition and acceptance in the medical community, the triggers and mechanisms that lead to CRS remain elusive.
In this review, we attempt to describe how structural proteins of the SARS-CoV-2 induce production of abundant IL-6, the effect of the IL-6 receptor on downstream inflammatory pathways, and possible interactions with different inflammatory cytokines at the molecular level. The involvement of other pro-inflammatory cytokines including tumor necrosis factor-alpha (TNFα) and interleukin-1beta (IL-1β) are also discussed. A better understanding of the mechanisms will help us to identify future targets for the treatment of COVID-19.
Pathogenesis of the CRS in COVID-19
Clinical features of COVID-19
A study of a cohort of COVID-19 patients showed that excessive amounts of proinflammatory cytokines were detected in the blood of those who died [3]. It was found that levels of cytokines IL-1β, IFN-γ, IL-10, and MCP-1 were higher in COVID-19 patients than healthy controls, and levels of IP-10, MCP1, MIP1a, and TNF-α were higher in ICU patients than in non-ICU patients, suggesting that excessive cytokine production may be associated with severity of the disease [1, 4]. In another study, more than half of the 99 patients (51%) with COVID-19 exhibited elevated IL-6 levels [5]. The effects of CRS may lead to pulmonary consolidation and edema, which may proceed to ARDS and fatal multi-organ failure [6]. Collectively, serum concentrations of cytokines and the resulting CRS are positively correlated with the pathogenesis and severity of COVID-19.
The relationship between CRS and comorbid factors of COVID-19 is also noteworthy. It has been reported that hypertension, diabetes and cardiovascular disease are the most common comorbidities [7, 8], which may result from chronic and systemic metabolic alterations caused by inflammation in adipose tissue and increase the infection risk of SARS-CoV-2 [9]. Studies have indicated that diabetes may activate the differentiation of CD4+ cells by Th1 and Th2 cells and lead to dysfunction of Th17 and Treg cells, which breaks the balance of proinflammation and anti-inflammation and induces inflammatory cytokines [10]. The concentrations of IL-2, IL-6 and TNFα were significantly higher in a diabetes group than that of a non-diabetes group, also with a higher mortality rate [10], which may be due to decreased pulmonary function, excessive immune inflammation and aggressive glycosylation. Aggressive glycosylation resulting from diabetes is thought to be associated with immunoglobulin dysfunction and lead to susceptibility to COVID-19 and impaired viral clearance [11]. Angiotensin II (Ang II), closely related to hypertension, is increased in patients with COVID-19 compared to healthy controls [12]. Ang II activates p44/42MAPK, p38MAPK, NF-κB and c-Jun pathways by up-regulating the expression of LOX-1 (a lectin-like ox-LDL receptor), Ang II type 1 and type 2 receptors, which further increases the expression of proinflammatory genes, such as IL-6, IL-10 and TNF-α [13], and contributes to the severity of CRS.
Features of CRS
SARS, MERS and COVID-19 may possess certain common features that lead to CRS and greater morbidity and even mortality. Monocytes/macrophages and dendritic cells are part of the first line of defense after initial exposure to antigens, and they accumulate at sites of early infection. However, immune cells containing viral particles may migrate to other organs and tissues, a concept known as homing. The widespread infiltration of innate immune cells during early viral exposure is followed by increased T cell expression of proinflammatory cytokines and chemokines. CRS, a result of excessive cytokines and chemokines, is characterized by leakage of plasma, increased vascular permeability, diffuse intravascular coagulation (DIC) and immunodeficiency, facilitating spread of the virus and an even further inflammatory response in the manner of vicious cycle [14]. Multiple in vivo studies have detected an increased level of lymphocyte apoptosis in these diseases [2, 3]. The hyperactivity of T lymphocytes may also result in T-cell exhaustion, which is consistent with the observation of lymphopenia in many patients with severe COVID-19.
Possible mechanisms for CRS in COVID-19
NF-κB plays an important role in the mediation of the inflammatory process by promoting maintenance, activation, differentiation and proliferation of naive T cells. In mammals, the NF-κB family is composed of five transcription factors: p50, p52, p65, c-Rel and RelB. These transcription factors form homo- and heterodimers through their shared N-terminal DNA-binding/dimerization domains. NF-κB complexes are inactivated after combination with inhibitory IκB proteins in a complex that exists mainly in the cytoplasm. When signaling pathways are activated, the IκB protein degrades and NF-κB enters into the nucleus, activating the expression of target genes such as TNF-α, IL-1 and IL-6 [15].
Plasma cytokine analysis showed that levels of IL-6 were found to be higher in both mild and severe groups of COVID-19 patients and was also positively correlated with disease severity. In a study of 123 patients, elevated IL-6 levels were found in 30.39% of patients with mild and 76.19% of patients with severe COVID-19 [16]. This suggests that IL-6 generation during infection may be the key cytokine triggering severe inflammation and acute injury to multiple organ systems [17]. By stimulating vascular endothelial cells to produce VEGF and up-regulating the expression of C5a receptor on vascular endothelial cells, IL-6 decomposes cadherin and stimulates mast cells to secrete histamine to increase vascular permeability, resulting in hypotension, hypoxia and peripheral edema. In addition, weakened contraction of the papillary muscle in response to IL-6 leads to cardiac dysfunction and aggravates diseases [17].
IL-6 also induces the differentiation of megakaryocytes into platelets and is considered a platelet augmentation factor. Tissue factor III, or platelet tissue factor, encoded by the F3 gene and expressed in subendothelial tissue and leukocytes, initiates thrombin formation from prothrombin. Excessive thrombin generation not counterbalanced by anti-coagulation by anti-thrombin and protein C is a critical factor in the pathogenesis of disseminated intravascular coagulation in infected individuals. Indeed, clotting abnormalities are now being increasingly associated with COVID-19 infection [18].
Structural proteins of SARS-CoV-2 and the NF-κB pathway
Coronaviruses are large single-stranded positive-sense RNA viruses that consist morphologically of four major structural proteins, including the Spike (S) protein, Envelope (E) protein, Matrix Protein (M) and Nucleocapsid Protein (N) [19].
The S protein is divided into two functional subunits: the S1 subunit that promotes viral infection by binding to receptors of the host, and the S2 subunit that contributes to membrane fusion [19]. Compared with SARS, the higher binding affinity of SARS-CoV-2 S protein to the human ACE2 receptor explains why SARS-CoV-2 is more infectious than SARS [20].
Previous studies suggest that coronavirus is involved in metabolic dysregulation and immunoregulatory processes by regulating NF-κB signaling [21]. The S protein of SARS-CoV-2, which binds to angiotensin-converting enzyme 2 (ACE2) in alveolar epithelial cells and can induce neutralizing antibodies against SARS-CoV-2, plays a significant role in COVID-19 pathogenesis. In a study by Wang et al. [22], it was discovered that purified recombinant S protein can stimulate IL-6 and TNF-α release, with no activation of IL-8, by inducing NF-κB activation in RAW264 (a murine macrophage cell line). However, Dosch et al. [23] found that the S protein can stimulate IL-8 production in a dose dependent manner and this increased production of IL-8 can be suppressed by a NF-κB inhibitor in human monocyte macrophages in vitro. A recent study also revealed after binding to ACE2 receptors, the S protein downregulates anti-viral mediators and upregulates NF-κB pathway and reactive oxygen species (ROS), which lead to the production of IFN-β [24, 25]. The NF-κB pathway is also activated by down-regulation of ACE2 and moderated by ROS in a positive feedback [24].
Multiple studies have also demonstrated a role of the N protein in the NF-κB pathway. Based on research by Zhang [26], the N protein activates the IL-6 promoter and up-regulates IL-6 protein production in A549 cells. Moreover, the N protein induces IL-6 expression by facilitating the translocation of p65 (a subunit of NF-κB). Similarly, Lai et al [27] also reported that the N protein of the human coronavirus OC43, a strain which is associated with the common cold, activates NF-κB.
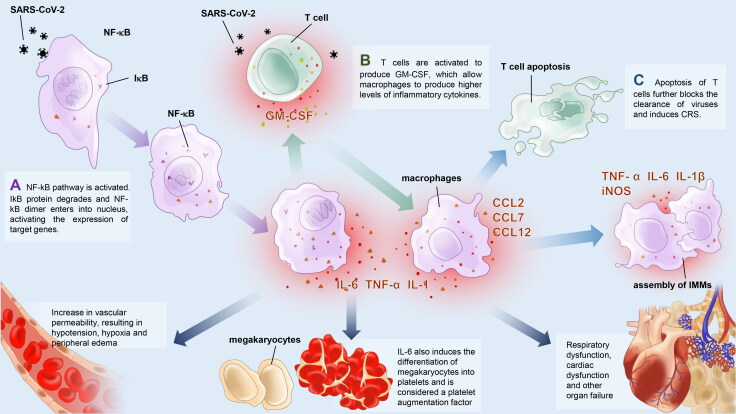
The subsequent production of IL-6 is thought to be key to the development of CRS. In Wei’s study of immunological indices, T cells are rapidly activated to produce Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF) and IL-6 soon after SARS-CoV-2 infection and the NF-κB pathway is thought to contribute to this process [28] (Figure 1). GM-CSF further activates CD14+CD16+ monocytes, producing [25] higher levels of IL-6 and other inflammatory cytokines, resulting in severe immune injury to the lungs and other organs. A study by Zhou points out that CD4+ T lymphocytes are activated by the SARS-CoV-2 and differentiate into pathogenic T helper 1 cells, which generate GM-CSF. The monocytes become macrophages in the pulmonary circulation, and together with T cells and dendritic cells, induce the development of CRS [29]. IL-6 signaling is positively correlated with the production of other proinflammatory cytokines and chemokines. Moreover, IL-6 further activates the differentiation of monocytes and macrophages, inhibits Treg cells and attracts other immune cells, which can potentially amplify CRS.
Figure 1.
IL-6 and GM-CSF in NF-?B pathways. A. The N and S protein of SARS-CoV-2 interact with the NF-?B complex, leading to phosphorylation and degradation of I?B, and to the release of NF-?B dimers and translocation from the cytoplasm to the nucleus. The NF-?B dimers further bind to DNA binding sites and upregulate the expression of target genes such as IL-6, TNF-? and GM-CSF. B. GM-CSF further stimulates CD14 + CD16+ monocytes to produce higher levels of IL-6 and other inflammatory cytokines. C. By promoting the production of other cytokines and chemokines, differentiation of monocyte and macrophage, attraction of other immune cells and inhibiting Treg cells, IL-6 may play an important role in apoptosis of T cells and development of CRS.
Anomalies in the IL-6R receptors
IL-6 receptors are categorized into two types: soluble IL-6 receptors (sIL-6R) and membrane-bound receptors (mIL-6R). The latter can be transformed into the former primarily by shedding but also, to a lesser extent, by splicing. IL-6 activates a proinflammatory response through sIL-6R, but triggers an anti-inflammatory response through mIL-6R. IL-6, upon binding to IL-6R, triggers the receptor to form a hexametric signal transducing complex with gp130, and thereby activates the downstream JAK/STAT3 signaling pathway [14]. Notably, these downstream effects may vary among different cell types or states, which are usually pre-determined by distinct chromatin states [30].
The binding of IL-6 to the receptor depends on the concentration of the receptor. During the inflammatory process, mIL-6R may increase 100,000-fold, while sIL-6R only increases by 2-5-fold. Upon stimulation by antigens, IL-6 first binds to sIL-6R and triggers a proinflammatory response. When the concentration of mIL-6R exceeds that of sIL-6R, an anti-inflammatory response predominates. Consistently, it has been observed that a selective sIL-6 pathway blocker is more effective for controlling inflammation in mouse models when compared to the nonspecific IL-6R blockers [31]. Therefore, IL-6R may be considered to be an immunoregulatory master receptor in the pathogenesis of CRS. Accordingly, further research is required to understand the molecular mechanism underlying the sustained trans-signaling activation that leads to CRS-mediated disease progression.
Delayed IFN response
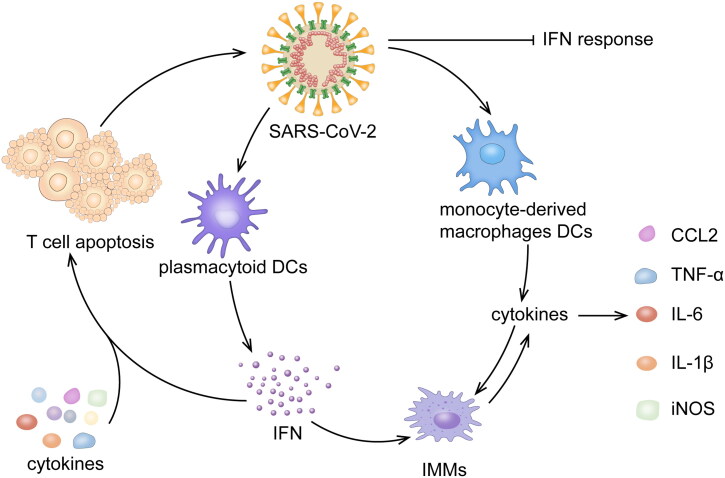
A delayed type 1 IFN response has been proposed as a mechanism for the development of CRS. This is believed to be similar to the case of SARS and MERS [32–34] (Figure 2). RIG-I-like receptors (RLRs) or toll-like receptors (TLRs) recognize viral RNA and the caspase activation recruitment domains of RLRs combined with adapter mitochondrial antiviral signaling protein (MAVS), which recruits TANK-binding kinase 1 (TBK1) and inducible IκB kinase (IKKi). Interferon regulatory factor 3/7(IRF3/7) is phosphorylated by TBK1 and IKKi and further activates the expression of IFN-α/β, which induces the production of IFN-stimulated genes (ISGs) via the JAK-STAT pathway to counter the virus [35]. Coronavirus has been reported to antagonize IFN production and signaling by evading recognition by PRRs, inhibiting RIG-1 or TLRs signaling or blocking IRF3 activation. A recent study showed that expression of open reading frame 6 (ORF6) inhibited RLRs, MAVS and IRF3 through its C-terminus to limit IFN activation and the subsequent interferon-stimulated response element, ISG56 and STAT signal to delay IFN response in the early stages [35]. Low levels of IFN and high levels of proinflammatory cytokines and chemokines are significant characteristics of a delayed IFN response. Monocyte-derived macrophages and DCs respond to viral infection early and release inflammatory cytokines, while the production of IFN-by plasmacytoid dendritic cells is delayed [34].
Figure 2.
IMMs are induced to accumulate and produce large quantities of cytokines and chemokines after infection, resulting in the apoptosis of T cells together with a delayed IFN-?/? response. The T cell apoptosis further blocks the clearance of viruses and induces proinflammatory cytokine release, which may further aggravate a cytokine storm.
During the late phases of infection, overproduction of IFN-α/β activates extensive IFN-stimulated genes and induces an overproduction of pathogenic inflammatory monocyte-macrophages (IMMs) in the lung [32]. The accumulation of IMMs produces elevated levels of monocyte chemokines (CCL2, CCL7 and CCL12), leading to further assembly of IMMs, which aggravate the disease further. Enhanced levels of proinflammatory cytokines (TNF-α, IL-6, IL-1β and iNOS) are also produced by IMMs. Subsequently, T cells are activated by IFN-α/β or proinflammatory cytokines to undergo apoptosis, further blocking viral clearance [6, 32].
Recently, IFNλs were found to maintain antiviral responses in respiratory tract at a lower viral load before type 1 IFN responses. IFNλs are tissue-protective and anti-inflammatory, and suppress viral replication to alleviate CRS by stimulating Th1 and cytotoxic T cells [36]. Thus, impaired IFNλs production and secondary type I IFN responses may both contribute to CRS. IFNλs also induce the expression of ACE2, which is insufficient to enhance SARS-CoV-2 replication [37]. Although there are still concerns regarding the potential for IFNλs to lead to bacterial superinfection and whether the resultant anti-inflammatory effect is beneficial [38], some studies have proposed the use of IFNλs to treat CRS in COVID-19 patients [37, 39].
Antibody dependent enhancement
Excessive cytokine release can also result from a phenomenon called antibody dependent enhancement (ADE) [40]. This phenomenon was first observed with arboviruses in 1964 and has been observed in multiple viral illnesses including flaviviruses such as Dengue. ADE results from the presence of cross-reactive or low levels of non-neutralizing antibodies to the virus, whereby the antibody-virus interactions facilitate various viral functions including viral entry into the cell or viral replication, as well as the development of an increased inflammatory response. Previous studies have shown that a neutralizing monoclonal antibody against the receptor-binding domain of the S protein of the MERS virus facilitated viral entry [41]. In patients with COVID-19, an increased IgG response and higher levels of total antibodies have also been encountered, and were associated with worse outcomes [42], especially liver injury [43]. Antibodies against S protein were found to promote the accumulation of IMMs in lung [44].
The mechanism of ADE involves the binding of antibody to the virus, and the resultant viral complex leads to enhanced viral replication and complement induced exaggerated inflammatory reactions. The source of these antibodies can be variable; thus, ADE can be a problem in a variety of scenarios. They can be derived from non-neutralizing antibodies against SARS-CoV-2 or can be cross reacting antibodies that exist in a patient by virtue of a previous infection by another serotype or a totally different coronavirus, such as one that causes the common cold. However, cross reacting antibodies against different serotypes are short-lived. In Dengue virus, patients tend to develop a more severe course when reinfected by a different serotype as the protective antibody titer attenuates. It has also been shown that patients with a prior infection to other viruses, including influenza virus and SARS-CoV, are more likely to become infected by SARS-CoV-2 and progress to a more severe illness [45].
There have been several mechanisms to explain ADE in Dengue viruses. Studies have found that [46] spleen tyrosine kinase (Syk) can mediate IL-1β induction by primary human monocytes during antibody-enhanced Dengue virus infection. Dengue virus immune complexes activate Syk soon after inoculation, which activates ERK1/2, leading to the elevated IL-1β secretion. Caspase-1 and NLRP3 are required in the maturation of pro-IL-1β and caspase-1 can be enhanced by ADE and ATP, a common inflammasome agonist. IL-6 and TNF are also induced by Syk, though the precise signaling pathway needs further investigation. Antibody dependent enhancement clearly also plays a significant role in the development of a safe vaccine, as the development of the wrong type of antibody may actually lead to worsening illness.
Other hypotheses
There are also other thought-provoking hypotheses. Ferritin is an inflammatory marker that is thought to be relevant in CRS-related diseases and in autoimmune diseases. Hyperferritinemia is a common feature of SLE and RA. Macrophages activated by proinflammatory cytokines are the main producers of ferritin. Ferritin, as a proinflammatory molecule, induces the production of cytokines and chemokines, which aggravates the positive feedback loop [33]. There is also a theory that heme metabolism and iron release becomes disorganized when the virus combines with the beta chain of porphyrins in erythrocytes [44].
Covid-19, Kawasaki Disease, Multisystem Inflammatory Syndrome of Children (MIS-C) and HLH
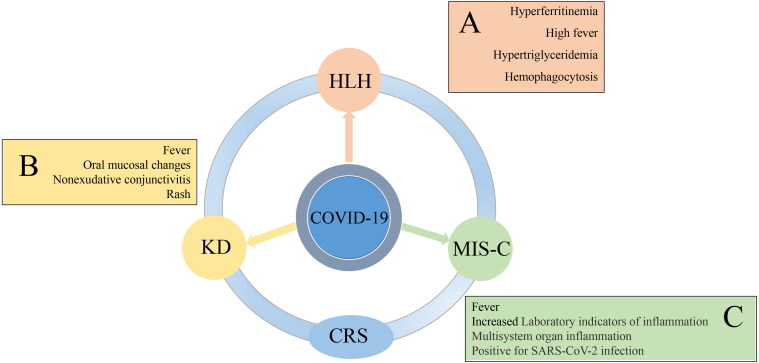
A multisystem inflammatory syndrome has been increasingly recognized as a manifestation of COVID-19 in children (Figure 3). In some ways, it is similar to Kawasaki disease. Patients present with fever, rash, shock, conjunctivitis, generalized edema and gastrointestinal symptoms. Molecular and/or serological testing for SARS-CoV-2 are positive [47]. This name of this condition has undergone several iterations in a short time. Referred to early on as pediatric multisystem inflammatory syndrome (PMIS) or pediatric inflammatory multisystem syndrome (PIMS), it was eventually re-named multisystem inflammatory syndrome in children (MIS-C) [48]. Increased interferon signaling, as well as elevated IL-6 and IL-10 in plasma were observed many of the patients with MIS-C [49].
Figure 3.
An illustration showing that cytokine release syndrome, cytokine storm, MIS-C, and a Kawasaki disease like syndrome seen in COVID-19 patients and hemophagocytic lymphohistiocytosis are all variations of a hyperactive inflammatory disorder associated with infection. The manifestation of these syndromes or lack thereof is highly dependent on the immune system responses specific for each individual infected host and there may be both genetic (inherent) and environmental (external) factors that lead to the variability in the response. CRS = Cytokine release syndrome, MIS-C = multisystem inflammatory syndrome in children, HLH = Hemophagocytic lymphohistiocytosis, KD (COVID-19) = Kawasaki disease seen in COVID-19 patients. A, B and C show symptoms in COVID-19 patients that may also be present in related inflammatory conditions.
MIS-C and Kawasaki disease share many features, but MIS-C can be distinguished from Kawasaki disease by significant cardiac dysfunction, severe enteropathy and relative thrombocytopenia rather than thrombocytosis [48]. The reason why some children develop MIS-C while others do not is unknown, but genetic and environmental factors may play variable roles in the pathogenesis [50].
HLH is a hyperinflammatory condition induced by T cells leading to persistent and uncontrolled activation of IFN-γ-dependent TLRs, antigen-presenting cells and T cells, resulting in a form of CRS [51]. It has been suggested that features of secondary hemophagocytic lymphohistiocytosis (sHLH) may be present in COVID-19 patients. Some of the criteria for sHLH in COVID-19 patients include hyperferritinemia, cytopenias, splenomegaly and high fever.
Although COVID-19 patients may develop different immunological syndromes, the common feature involves a hyperinflammatory microenvironment as the basic pathological process leading to a cytokine release syndrome that causes significant mortality and morbidity [51]. This finding suggests that the use of immunomodulating agents may be of benefit in the treatment of COVID-19 patients who develop these syndromes.
Potential therapies
Highly pathogenic viral infections and subsequent CRS are positively correlated with high mortality. From studies on SARS and MERS, therapeutic strategies aimed at controlling viral load and attenuating the inflammatory response have been generally successful, though the response to specific therapies has been variable or uncertain. Therapies targeting IL-6 and other forms of immunosuppression have been attempted in the treatment of COVID-19. (Table 1)
Table 1.
Summary of potential therapies for COVID-19.
| Therapies | Mode of action | Trial/Phase (as of 09/20/2020) | ||
|---|---|---|---|---|
| Tocilizumab | IL-6 receptor inhibitor | NCT04445272, Phase 2 | ||
| Hydroxychloroquine and Chloroquine | Regulation of pH values and immunity, interference of glycosylation | NCT04261517, Phase 3 | ||
| Baricitinib | JAK-STAT pathway inhibitor | NCT04358614, Phase 2/3 | ||
| Anakinra | IL-1 receptor antagonist | NCT04443881, Phase 2/3 | ||
| Glucocorticoids | Immunoregulator | NCT04244591, Phase 2/3 | ||
| Intravenous immunoglobulin | Pooled IgG to block inflammatory | NCT04354831, Phase 2 | ||
| Zinc | Inhibition of RNA polymerase and regulation of inflammation | NCT04370782, Phase 4 | ||
| ACEI/ARB | RAAS inhibitor | NCT04353596, Phase 4 | ||
| Vitamin D | Immunoregulator and RAAS regulator | NCT04482673, Phase 4 | ||
| IFNβ | Anti-viral medicine and immunoregulator | NCT04343768, Phase 2 | ||
| IFNλ | Anti-viral medicine and immunoregulator | NCT04343976, Phase 2 | ||
| Atorvastatin | Lipid-lowering medicine | NCT04486508, Phase 3 | ||
| Ulinastatin | Protease inhibitor | NCT04393311, Phase 1/2 | ||
| Mesenchymal stem cells | Immunoregulator | NCT04288102, Phase 2 |
Targeted therapies
Tocilizumab
Tocilizumab is a humanized monoclonal antibody directed against the IL-6 receptor. It was first used in the treatment of hematologic tumors with Chimeric Antigen Receptor T-cell (CAR-T) immunotherapy to suppress CRS generated during the treatment process without affecting the anti-tumor therapeutic efficacy [52]. It has since been recommended as the first-line treatment for severe CRS associated with CAR-T therapy.
The beneficial effects of using tocilizumab to counteract the CRS arising from infection by SARS-CoV-2 were unknown at the beginning of the pandemic. Normally, active infection is a contraindication to tocilizumab, which would preclude its use in COVID-19. However, a small sample clinical trial including 21 patients showed that tocilizumab may be an effective treatment in patients with severe COVID-19 infections [53]. Because most studies on tocilizumab are done on patients who are already critical ill or have severe disease, it is not known whether there would be greater benefit if used earlier in the course, before the patients reach a severe or critical state. In the newly released seventh edition of the Chinese CDC guidelines, tocilizumab has been included as trial therapy. Luckily, a recent original research in Italy reported that the application of tocilizumab could decrease the serum levels of CRP, ferritin and fibrinogen to normal levels and thus was highly associated with clinical improvement [54].
Hydroxychloroquine and chloroquine
Chloroquine and Hydroxychloroquine are widely used to prevent and treat malaria and are also an important aspect of the management of autoimmune diseases. These two medications have well-recognized anti-inflammatory and immunomodulatory actions. Hydroxychloroquine and chloroquine can inhibit virus invasion by regulating pH values, affecting the activity of glycosyltransferase or glycosyl modifying enzyme in host cells, interfering with the correct glycosylation of ACE2 and blocking the binding of virus to ACE2, by which SARS-CoV-2 enters cells [55]. Other research has proposed that hydroxychloroquine may additionally prevent SARS-CoV-2 from binding with gangliosides, which in turn may inhibit virion contact with the ACE-2 receptor [56]. Previous studies have shown that effect of chloroquine on pH also interferes with intracellular acidic organelle functions such as endocytosis, golgi apparatus and endoplasmic reticulum, and affects intracellular transport of viruses [57, 58]. Overall, hydroxychloroquine/chloroquine are capable of affecting several cellular pathways and therefore may have several mechanisms of action against SARS-CoV-2.
Unfortunately, the effectiveness of these drugs in treating COVID-19 has never been very conclusive, and the potential risk of heart disease induced by these drugs led the Food and Drug Administration of the United States to withdraw their emergency use authorization (EUA) for the treatment of COVID-19 on June 15, 2020. On July 4th, 2020, the WHO also discontinued its trials of hydroxychloroquine and in combination with lopinavir/ritonavir because of the failure of these pharmacologic treatments to reduce mortality.
Baricitinib
Clathrin-mediated endocytosis (CME) is crucial for viral invasion of pneumocytes, and numb-associated kinase (NAK) participates in this process [49]. Baricitinib is a tyrosine protein kinase inhibitor, which can inhibit NAK, thus limiting the systemic inflammatory response and cytokine production through the inhibition of the canonical JAK–STAT pathway. It can also prevent intracellular viral particle assembly by inhibiting protein kinases [59, 60]. Since interferon transcription is mediated by the JAK-STAT pathway, baricitinib has been suggested as a possibly treatment for patients with moderate COVID-19 who suffer from CRS and require hospital care [61]. There is preliminary clinical evidence that oral administration of baricitinib may be potentially beneficial in the treatment of COVID-19 [62, 63], especially on respiratory function when utilized at a dose of 4 mg in combination with high-dose corticosteroids [64]. The long-term effects of baricitinib in COVID-19 will need to be confirmed by future studies.
Anakinra
Anakinra is an interleukin-1 receptor antagonist that interdicts activity of IL-1α and IL-1β and is used for autoinflammatory disorders. Recent studies have shown that coronavirus regulates the activation of the NLRP3 inflammasome by inducing the maturation and secretion of IL-1β [65]. As is reported, anakinra has been shown to be effective in patients with cytokine storm syndromes [66], and further studies have suggested that high dose IV anakinra is a safe alternative for the treatment of cytokine release syndrome in patients with COVID-19 [67]. Another study suggested that ventilation function improved and D-dimers, ferritin, CRP and PCT reduced after anakinra treatment, but there were no controlled trials to confirm this [68]. It is also worth noting that previous studies have shown that IL-1 inhibition may also induce endothelial dysfunction and affect blood coagulation. A case report recently described reduction in serum inflammatory markers and clinical improvement in a patient with COVID-19 myocarditis after treatment with anakinra [69]. Further clinical studies are undergoing to confirm the efficacy and safety of anakinra and other interleukin-1 inhibitors in the treatment of COVID-19.
Glucocorticoids
Glucocorticoids, widely used in the treatment of SARS and MERS, inhibit excessive immune cell activation and cytokine production. However, glucocorticoids also suppress the immune response and the immune system’s ability to clear viruses. An early study [70] evaluated corticosteroid use in COVID-19 and other infectious diseases, and found no evidence showing corticosteroids were beneficial. It is noteworthy that glucocorticoids were widely used in large quantities for emergency treatment during the SARS epidemic in 2003, leading to the development of femoral head necrosis and pulmonary dysfunction [71]. There are at least two ongoing clinical trials on the use of dexamethasone in the United States, and it has been widely reported that a large scale trial of 6,500 patients in the United Kingdom associated dexamethasone with a 30% decrease in mortality
Intravenous immunoglobulin
Intravenous immunoglobulin (IVIG) uses pooled IgG to perform its immunomodulatory function. IVIG modulates immunoreactivity through several mechanisms, including blocking the production of proinflammatory cytokines and inhibiting the differentiation of pathogenic Th1 and Th17 subsets. IVIG also acts by overwhelming the Fc receptors, thereby inhibiting complement activation, and by neutralizing pathogenic autoantibodies [72, 73]. IVIG has been widely used to treat autoimmune and chronic inflammatory diseases, and has been used as an immune modulator in cases of infection induced hyper-inflammatory states. Interestingly, although most currently available preparations of IVIG were collected from patients long before the COVID-19 pandemic ensued, it has been found that these preparations may contain antibodies against SARS-CoV-2 antigens, possibly because of cross-reactivity with other coronaviruses [73]. A study by Yun Xie showed that IVIG at 20 g/day as adjuvant therapy within 48 h of admission to the ICU to treat COVID-19 patients may reduce the use of mechanical ventilation and improve 28-day survival [74]. Other studies also reported that high-dose IVIG (2 gr/kg or 0.3-0.5g/kg) may play an important role in the recovery [75, 76]. However, Aljaberi reported that a group of COVID-19 patients treated with high-dose IVIG and hydroxychloroquine experienced a higher mortality rate, though the reasons for this are unclear [77]. Other studies have not shown a benefit of IVIG in mortality resulting from COVID-19, and it is not clear if it is because IVIG was only used in the more severe cases. Thrombosis is thought to be a potential complication of IVIG, which is common in critically ill COVID-19 patients [72]. Further investigations are still needed. In the case of Kawasaki disease associated with COVID-19, IVIG is a part of the standard regimen for treatment of Kawasaki disease and is therefore clinically indicated. This usage appears to have been commonly extended to the treatment of MIS-C after weighing potential benefits against the risk of administering IVIG [78].
Other medicines
Zinc
As an important component of numerous enzymes, zinc is essential to the development and function of the immune system. Low zinc status was found to be a risk factor for pneumonia and correlated with increased serum levels of TNFα, IL-6 and IL-8 through a Th17 response in aged people [79, 80]. Increased concentration of intracellular zinc has been shown to inhibit the activity of virus RNA polymerase and viral replication [81]. Zinc also helps to downregulate proinflammatory signaling pathways, such as activation of STAT3 mediated by IL-6, and enhance the antiviral effect of interferon [82]. There is also evidence that zinc supplementation enhances the clinical effect of chloroquine and hydroxychloroquine, with the advantage of broad availability and affordability [83]. While zinc has not been directly shown to be beneficial in COVID-19, zinc supplement has generally been considered to be safe.
Renin-Angiotensin-Aldosterone system inhibitors and vitamin D
ACE2, one of the receptors that SARS-CoV-2 utilizes for cellular entry, is a part of renin-angiotensin-aldosterone system (RAAS), which cleaves the Ang II into angiotensin 1-7 [Ang-(1-7)], which has vasodilatory activity. SARS-CoV-2 binds ACE2 with S protein and reduces ACE2 expression, leading to the generation of Ang II. Ang II mediates the production of reactive oxygen species and inflammatory cytokines and induced apoptosis of alveolar epithelial cells. Induced type 1a Ang II receptors increased pulmonary vascular permeability, which may deteriorate lung inflammation and lead to pneumonia and ALI [84].
Vitamin D, as a natural immunoregulator, has been hypothesized to enhance antimicrobial activity to prevent acute respiratory infections by upregulating antimicrobial peptides and inducing antiviral cytokines, which interfere with the cycle of viral replication. Vitamin D also strengthens cellular immunity and alleviates cytokine storm by reducing the production of inflammatory Th1 cytokines, especially TNFα and IFN-γ [85]. According to recent reviews, vitamin D is a negative RAAS modulator and inhibits expression and generation of renin, ACE and Ang II, while increasing ACE2 concentration [84, 86]. Vitamin D deficiency was considered to be a risk factor for COVID-19 and highly associated with lung inflammation and ARDS. However, opposing views argue that there is no significant difference in Vitamin D levels between COVID-19 positive and negative groups [87]. Although there are no randomized clinical trials on the efficacy of Vitamin D in COVID-19, intake of 40 μg D3/day has been recommended to prevent aggressive inflammation and avoid hypercalcemia [80].
RAAS inhibitors have also been shown to increase ACE2 expression in heart and kidney (though unclear in lung), and there was a concern that increased ACE2 can enhance the infection risk of SARS-CoV-2 [88]. There has been some evidence that ACEI decreases viral load by reducing Th1/Th2 ratios and inflammatory cytokine production, but studies have had very small sample size [12]. It seems prudent for patients to continue taking these medications, given their beneficial effects on cardiovascular diseases [89].
Statins
Toll-like receptors (TLR) recognize pathogens and trigger innate immunity by activating NF-κB pathway. Myeloid differentiation primary response 88(MyD88) is an adapter protein for most TLRs, while statins, known as antagonists to TLR-MyD88 pathway, can stabilize MyD88 levels and attenuate NF-κB activation [90]. NF-κB, cytokines and chemokines are down-regulated correspondingly. Statins inhibit HMG-CoA reductase and thus lower the concentration of LDL cholesterol, which is a strong inflammation promotor. Statins also protect vascular endothelium from reactive oxygen species and may relieve lung injury [90]. In addition, membrane/lipid rafts (MLR), cholesterol-rich areas, are essential for membrane fusion and endocytosis after S protein attaches with ACE2. MLR may be disrupted with lower cholesterol in plasma membrane induced by statins, which interrupts the internalization of viruses [91]. Although lower serum cholesterol levels were associated with lower ability of microbe inactivation [92], Zhang’s study suggested that the use of statins contributed to lower mortality and favorable prognosis [93].
Ulinastatin
Ulinastatin is a naturally occurring protease inhibitor. By inhibiting the generation and release of inflammatory mediators, ulinastatin protects vascular endothelium, improves capillary permeability and effectively reduces 28-day mortality in patients with acute infection. Ulinastatin also reduces the generations of proinflammatory factors such as TNF-α, IL-6, IFN-γ and enhances the level of the anti-inflammatory factor IL-10 to restore the balance between proinflammatory and anti-inflammatory responses [94]. Ulinastatin therefore may play a role in attenuating CRS and blocking the progression of SIRS to MODS. However, ulinastatin will not suppress immune function and may cause femoral head necrosis and other sequelae. A tolerance and dosing study of ulinastatin in healthy adult subjects in China showed good safety and tolerance [95]. A prospective, observational clinical study of ulinastatin injection in the treatment of severe COVID-19 cases has been launched in Guangdong Province in China.
Traditional Chinese medicine
Different clinical symptoms can be divided into many types, such as deficiency of lung and spleen, or Qi and Yin deficiency syndromes. Research on the mechanisms of Chinese herbal medicine suggests that traditional Chinese medicine may have preventive or protective effects on sepsis caused by viruses or bacteria. A study by Chen on Chinese herbal medicine and sepsis revealed that serum concentrations of inflammatory cytokines (TNF-α and IL-6) was remarkably reduced in septic mice after being treated with Xuebijing injection (XBJ), a Chinese herbal medicine. XBJ improves survival rate in septic shock models partially by suppressing immune inflammation and regulating the balance of Treg and Th17 cells [96]. A study by Zhu on Qiangzhi Decotion (QZD) indicates that the survival rate of mice with influenza A pneumonia in the QZD groups is significantly higher than that in the virus control group. The production of inflammatory cytokines including IFN-γ, IL-6, TNF-α, and ICAM-1 were suppressed (P < 0.05) in the QZD group [97].
Supportive treatment
Continuous renal replacement therapy (CRRT) is recommended for the treatment of severe and critically ill patients. The proposed rationale is based on the elimination of inflammatory cytokines which may be responsible for inflammatory tissue injury. CRRT has other benefits, including maintenance of fluid, acid-base and electrolyte balance.
Extracorporeal membrane oxygenation (ECMO) has been used in patients when 1. oxygenation index is less than 80 mmHg for more than 3 to 4 hours and when the FiO2 is greater than 90%; and 2. airway platform pressure is greater than 35cmH2O. However, a study of the ECMO in ARDS showed that there was no significant difference in mortality between the ECMO and non-ECMO groups [98]. Therefore, the beneficial effects of ECMO in COVID-19 require further investigation.
Mesenchymal stem cells
Mesenchymal stem cells (MSC) have a dual role in the regulation of immune function in patients with viral pneumonias. Stem cells may inhibit an excessive immune response and CRS by secreting anti-inflammatory factors. Stem cells may also home to damaged tissues by chemotaxis and activate the function of regulatory immune cells to improve the specificity of immune response [99]. Notably, stem cells themselves are highly resistant to the virus [100], and have been used in patients with severe COVID-19 disease in China and been shown to be effective. In addition, the Italian College of Anesthesia, Analgesia, Resuscitation and Intensive Care have also issued guidelines to treat COVID-19 patients with stem cells [101]. Further studies are needed to confirm this potential treatment modality.
Summary and outlook
Inflammation is an integral part of effective immune responses to clear infections. However, certain highly pathogenic viruses cause an excessive and prolonged inflammatory response of cytokines/chemokines, referred to as CRS, leading to high morbidity and mortality. In COVID-19, it has been observed that there are high levels of proinflammatory cytokines, particularly IL-6, in the blood of patients, especially patients with severe disease. Several reports of the use of tocilizumab, a monoclonal antibody directed against IL-6R, have appeared in the literature. Other anti-inflammatory modulators that decrease the levels of pro-inflammatory cytokines may prove to be beneficial in the treatment of COVID-19 induced CRS.
Author contributions
Guogang Xu and Christopher Chang contributed to the conception of the review. Yifan Que, Yingzhen Du, Kun Wan, Peng Hu, Runsheng Wang and Chao Hu wrote the manuscript and drew the figures. Jiang Luo, Tianzhi Li, Rongyu Ping, Qinyong Hu, Yu Sun, Xudong Wu and Lei Tu helped improve this review and put forward many ideas. Authors affirm that they have approved this work and that this paper is not currently submitted to any other journal. Authors affirm that this work including all figures is original.
Funding Statement
This work was supported by Beijing Municipal Natural Science Foundation General Program (7192197), National Key Research and Development Program (2018YFC2002400) and National Key Research and Development Program (2020YFC2002700).
Declaration of interest
No potential conflict of interest was reported by the author(s).
References
- 1.Ferrara JL, Abhyankar S, Gilliland DG.. Cytokine storm of graft-versus-host disease: a critical effector role for interleukin-1. Transplant Proc. 1993; 25(1 Pt 2):1216–1217. [PubMed] [Google Scholar]
- 2.Tisoncik JR, Korth MJ, Simmons CP, et al. . Into the eye of the cytokine storm. Microbiol Mol Biol Rev. 2012;76(1):16–32. doi: 10.1128/MMBR.05015-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Huang C, Wang Y, Li X, et al. . Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (London, England). 2020;395(10223) :497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bassetti M, Vena A, Giacobbe DR.. The novel Chinese coronavirus (2019-nCoV) infections: Challenges for fighting the storm. Eur J Clin Invest. 2020;50(3):e13209. doi: 10.1111/eci.13209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen N, Zhou M, Dong X, et al. . Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet (London). 2020;395(10223):507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Channappanavar R, Perlman S.. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin Immunopathol. 2017;39(5):529–539. doi: 10.1007/s00281-017-0629-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fang L, Karakiulakis G, Roth M.. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med. 2020;8(4):e21. doi: 10.1016/S2213-2600(20)30116-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schiffrin EL, Flack JM, Ito S, et al. . Hypertension and COVID-19. Am J Hypertens. 2020;33(5):373–374. doi: 10.1093/ajh/hpaa057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.de Lucena TMC, da Silva Santos AF, de Lima BR, et al. . Mechanism of inflammatory response in associated comorbidities in COVID-19. Diabetes Metab Syndr. 2020;14(4):597–600. doi: 10.1016/j.dsx.2020.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yan Y, Yang Y, Wang F, et al. . Clinical characteristics and outcomes of patients with severe covid-19 with diabetes. BMJ Open Diabetes Res Care. 2020;8(1):e001343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Erickson JR, Pereira L, Wang L, et al. . Diabetic hyperglycaemia activates CaMKII and arrhythmias by O-linked glycosylation. Nature. 2013;502(7471):372–376. doi: 10.1038/nature12537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Meng J, Xiao G, Zhang J, et al. . Renin-angiotensin system inhibitors improve the clinical outcomes of COVID-19 patients with hypertension. Emerg Microbes Infect. 2020;9(1):757–760. doi: 10.1080/22221751.2020.1746200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang X, Khaidakov M, Ding Z, et al. . Cross-talk between inflammation and angiotensin II: studies based on direct transfection of cardiomyocytes with AT1R and AT2R cDNA. Exp Biol Med (Maywood). 2012; 237(12):1394–1401. doi: 10.1258/ebm.2012.012212. [DOI] [PubMed] [Google Scholar]
- 14.Mahmud-Al-Rafat A, Majumder A, Taufiqur Rahman KM, et al. . Decoding the enigma of antiviral crisis: Does one target molecule regulate all? Cytokine. 2019;115:13–23. doi: 10.1016/j.cyto.2018.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hayden MS, Ghosh S.. NF-κB in immunobiology. Cell Res. 2011;21(2):223–244. doi: 10.1038/cr.2011.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wan S, Yi Q, Fan S, et al. Characteristics of lymphocyte subsets and cytokines in peripheral blood of 123 hospitalized patients with 2019 novel coronavirus pneumonia (NCP). medRxiv, 2020: p. 2020.02.10.20021832. doi:.
- 17.Tanaka T, Narazaki M, Kishimoto T.. Immunotherapeutic implications of IL-6 blockade for cytokine storm. Immunotherapy. 2016;8(8):959–970. doi: 10.2217/imt-2016-0020. [DOI] [PubMed] [Google Scholar]
- 18.Ji H-L, Zhao R, Matalon S, et al. . Elevated Plasmin(ogen) as a Common Risk Factor for COVID-19 Susceptibility. Physiol Rev. 2020;100(3):1065–1075. doi: 10.1152/physrev.00013.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rabaan AA, Al-Ahmed SH, Haque S, et al. . SARS-CoV-2, SARS-CoV, and MERS-COV: A comparative overview. Infez Med. 2020;28(2):174–184. [PubMed] [Google Scholar]
- 20.Zhou P, Yang X-L, Wang X-G, et al. . A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270–273 doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Poppe M, Wittig S, Jurida L, et al. . The NF-κB-dependent and -independent transcriptome and chromatin landscapes of human coronavirus 229E-infected cells. PLoS Pathog. 2017;13(3):e1006286. doi: 10.1371/journal.ppat.1006286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang W, Ye L, Ye L, et al. . Up-regulation of IL-6 and TNF-alpha induced by SARS-coronavirus spike protein in murine macrophages via NF-kappaB pathway. Virus Res. 2007;128(1–2):1–8. doi: 10.1016/j.virusres.2007.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dosch SF, Mahajan SD, Collins AR.. SARS coronavirus spike protein-induced innate immune response occurs via activation of the NF-kappaB pathway in human monocyte macrophages in vitro. Virus Res. 2009;142(1–2):19–27. doi: 10.1016/j.virusres.2009.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hassan SM, Jawad MJ, Ahjel SW, et al. . The Nrf2 Activator (DMF) and Covid-19: Is there a possible role? Med Arch. 2020;74(2):134–138. doi: 10.5455/medarh.2020.74.134-138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schneider WM, Chevillotte MD, Rice CM.. Interferon-stimulated genes: a complex web of host defenses. Annu Rev Immunol. 2014;32:513–545. doi: 10.1146/annurev-immunol-032713-120231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang X, Wu K, Wang D, et al. . Nucleocapsid protein of SARS-CoV activates interleukin-6 expression through cellular transcription factor NF-kappaB. Virology. 2007;365(2):324–335. doi: 10.1016/j.virol.2007.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lai FW, Stephenson KB, Mahony J, et al. . Human coronavirus OC43 nucleocapsid protein binds microRNA 9 and potentiates NF-κB activation. J Virol. 2014;88(1):54–65. doi: 10.1128/JVI.02678-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liao H-H, Wang Y-C, Chen MC-M, et al. . Down-regulation of granulocyte-macrophage colony-stimulating factor by 3C-like proteinase in transfected A549 human lung carcinoma cells. BMC Immunol. 2011;12:16–16. doi: 10.1186/1471-2172-12-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhou G, Chen S, Chen Z.. Advances in COVID-19: the virus, the pathogenesis, and evidence-based control and therapeutic strategies. Front Med. 2020;14(2):117–125. doi: 10.1007/s11684-020-0773-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Carson WFt, Kunkel SL.. Regulation of cellular immune responses in sepsis by histone modifications. Adv Protein Chem Struct Biol. 2017;106:191–225. doi: 10.1016/bs.apcsb.2016.08.004. [DOI] [PubMed] [Google Scholar]
- 31.Barkhausen T, Tschernig T, Rosenstiel P, et al. . Selective blockade of interleukin-6 trans-signaling improves survival in a murine polymicrobial sepsis model. Crit Care Med. 2011;39(6):1407–1413. [DOI] [PubMed] [Google Scholar]
- 32.Liu B, Li M, Zhou Z, et al. . Can we use interleukin-6 (IL-6) blockade for coronavirus disease 2019 (COVID-19)-induced cytokine release syndrome (CRS)? J Autoimmun. 2020;111:102452. doi: 10.1016/j.jaut.2020.102452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Picchianti Diamanti A, Manuela Rosado M, Pioli C, et al. . Cytokine release syndrome in COVID-19 patients, A new scenario for an old concern: The fragile balance between infections and autoimmunity. Int J Mol Sci. 2020;21(9):3330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ye Q, Wang B, Mao J.. The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19. J Infect. 2020;80(6):607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lei X, Dong X, Ma R, et al. . Activation and evasion of type I interferon responses by SARS-CoV-2. Nat Commun. 2020;11(1):3810. doi: 10.1038/s41467-020-17665-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Andreakos E, Tsiodras S.. COVID-19: lambda interferon against viral load and hyperinflammation. EMBO Mol Med. 2020;12(6):e12465. doi: 10.15252/emmm.202012465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Busnadiego I, Fernbach S, Pohl MO, et al. . Antiviral activity of Type I, II, and III interferons counterbalances ACE2 inducibility and restricts SARS-CoV-2. mBio. 2020;11(5):e01928-20. doi: . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Prokunina-Olsson L, Alphonse N, Dickenson R E, et al. COVID-19 and emerging viral infections: The case for interferon lambda. J Exp Med, 2020;217(5):e20200653. doi:. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Felgenhauer U, Schoen A, Gad H H, et al. Inhibition of SARS-CoV-2 by type I and type III interferons. J Biol Chem, 2020;295(41):13958–13964. doi:. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tetro JA. Is COVID-19 receiving ADE from other coronaviruses? Microbes Infect. 2020;22(2):72–73. doi: 10.1016/j.micinf.2020.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cao X. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol. 2020;20(5):269–270. doi: 10.1038/s41577-020-0308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhao J, Yuan Q, Wang H, et al. Antibody Responses to SARS-CoV-2 in Patients With Novel Coronavirus Disease 2019. Clin Infect Dis, 2020;71(16):2027–2034. doi: . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tian D, Ye Q.. Hepatic complications of COVID-19 and its treatment. J Med Virol. 2020;92(10):1818–1824. doi: 10.1002/jmv.26036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Soy M, Keser G, Atagündüz P, et al. . Cytokine storm in COVID-19: pathogenesis and overview of anti-inflammatory agents used in treatment. Clin Rheumatol. 2020;39(7):2085. doi: 10.1007/s10067-020-05190-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Negro F. Is antibody-dependent enhancement playing a role in COVID-19 pathogenesis? Swiss Med Wkly. 2020;150:w20249. doi: 10.4414/smw.2020.20249. [DOI] [PubMed] [Google Scholar]
- 46.Callaway JB, Smith SA, McKinnon KP, et al. . Spleen Tyrosine Kinase (Syk) mediates IL-1β induction by primary human monocytes during antibody-enhanced dengue virus infection. J Biol Chem. 2015;290(28):17306–17320. doi: 10.1074/jbc.M115.664136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Whittaker E, Bamford A, Kenny J, et al. Clinical Characteristics of 58 Children With a Pediatric Inflammatory Multisystem Syndrome Temporally Associated With SARS-CoV-2. JAMA. 2020;324(3):259–269. doi:. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chiotos K, Bassiri H, Behrens E M, et al. Multisystem inflammatory syndrome in children during the Coronavirus 2019 Pandemic: a Case Series. J Pediatric Infect Dis Soc. 2020;9(3):393–398. doi: . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cheung EW, Zachariah P, Gorelik M, et al. . Multisystem inflammatory syndrome related to COVID-19 in previously healthy children and adolescents in New York City. JAMA. 2020;324(3):294. doi: 10.1001/jama.2020.10374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Deza Leon M P, Redzepi A, McGrath E, et al. COVID-19-Associated Pediatric Multisystem Inflammatory Syndrome. J Pediatric Infect Dis Soc. 2020;9(3):407–408. doi:. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Alunno A, Carubbi F, Rodríguez-Carrio J.. Storm, typhoon, cyclone or hurricane in patients with COVID-19? Beware of the same storm that has a different origin. RMD Open. 2020;6(1):e001295. doi: 10.1136/rmdopen-2020-001295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lee DW, Gardner R, Porter DL, et al. . Current concepts in the diagnosis and management of cytokine release syndrome. Blood. 2014;124(2):188–195. doi: 10.1182/blood-2014-05-552729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Xu X, Han M, Li T, et al. Effective treatment of severe covid-19 patients with Tocilizumab. http://chinaxiv.org/abs/202003.00026. Accessed March 11, 2020. [DOI] [PMC free article] [PubMed]
- 54.Toniati P, Piva S, Cattalini M, et al. . Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: A single center study of 100 patients in Brescia, Italy. Autoimmun Rev. 2020;19(7):102568. doi: 10.1016/j.autrev.2020.102568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Vincent MJ, Bergeron E, Benjannet S, et al. . Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. 2005;2:69. doi: 10.1186/1743-422X-2-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Fantini J, Di Scala C, Chahinian H, et al. . Structural and molecular modelling studies reveal a new mechanism of action of chloroquine and hydroxychloroquine against SARS-CoV-2 infection. Int J Antimicrob Agents. 2020;55(5):105960. doi: 10.1016/j.ijantimicag.2020.105960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Burkard C, Verheije MH, Wicht O, et al. . Coronavirus cell entry occurs through the endo-/lysosomal pathway in a proteolysis-dependent manner. PLoS Pathog. 2014;10(11):e1004502. doi: 10.1371/journal.ppat.1004502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Al-Bari MA. Chloroquine analogues in drug discovery: new directions of uses, mechanisms of actions and toxic manifestations from malaria to multifarious diseases. J Antimicrob Chemother. 2015;70(6):1608–1621. doi: 10.1093/jac/dkv018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Pu S-Y, Xiao F, Schor S, et al. . Feasibility and biological rationale of repurposing sunitinib and erlotinib for dengue treatment. Antiviral Res. 2018;155:67–75. doi: 10.1016/j.antiviral.2018.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Richardson P, Griffin I, Tucker C, et al. . Baricitinib as potential treatment for 2019-nCoV acute respiratory disease. Lancet. 2020;395(10223):e30–e31. doi: 10.1016/S0140-6736(20)30304-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Richardson PJ, Corbellino M, Stebbing J.. Baricitinib for COVID-19: a suitable treatment? - Authors’ reply. Lancet Infect Dis. 2020;20(9):1013–1014. doi: 10.1016/S1473-3099(20)30270-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Stebbing J, Krishnan V, Bono S, et al. Mechanism of baricitinib supports artificial intelligence-predicted testing in COVID-19 patients. EMBO Mol Med, 2020;12(8):e12697. doi: . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cantini F, Niccoli L, Matarrese D, et al. . Baricitinib therapy in COVID-19: A pilot study on safety and clinical impact. J Infect. 2020;81(2):318–356. doi: 10.1016/j.jinf.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Rodriguez-Garcia JL, Sanchez-Nievas G, Arevalo-Serrano J, et al. . Baricitinib improves respiratory function in patients treated with corticosteroids for SARS-CoV-2 pneumonia: an observational cohort study. Rheumatology (Oxford). 2021;60(1):399–407. doi: 10.1093/rheumatology/keaa587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Shakoory B, Carcillo JA, Chatham WW, et al. . Interleukin-1 receptor blockade is associated with reduced mortality in sepsis patients with features of macrophage activation syndrome: Reanalysis of a prior phase III trial. Crit Care Med. 2016;44(2):275–281. doi: 10.1097/CCM.0000000000001402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Dinarello CA, Simon A, van der Meer JW.. Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat Rev Drug Discov. 2012;11(8):633–652. doi: 10.1038/nrd3800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Pontali E, Volpi S, Antonucci G, et al. Safety and efficacy of early high-dose IV anakinra in severe COVID-19 lung disease. J Allergy Clin Immunol, 2020;146(1):213-215. doi: . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Dimopoulos G, de Mast Q, Markou N, et al. . Favorable anakinra responses in severe COVID-19 patients with secondary hemophagocytic lymphohistiocytosis. Cell Host Microbe. 2020;28(1):117–123.e1. doi: 10.1016/j.chom.2020.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Trpkov C, MacMullan P, Feuchter P, et al. Rapid response to cytokine storm inhibition using anakinra in a patient with COVID-19 Myocarditis. CJC Open. 2020; doi:. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Russell CD, Millar JE, Baillie JK.. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet (London, England). 2020;395(10223):473–475. doi: 10.1016/S0140-6736(20)30317-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Griffith JF, Antonio GE, Kumta SM, et al. . Osteonecrosis of hip and knee in patients with severe acute respiratory syndrome treated with steroids. Radiology. 2005;235(1):168–175. doi: 10.1148/radiol.2351040100. [DOI] [PubMed] [Google Scholar]
- 72.Delly F, Syed MJ, Lisak RP, et al. . Myasthenic crisis in COVID-19. J Neurol Sci. 2020;414:116888. doi: 10.1016/j.jns.2020.116888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Jawhara S. Could intravenous immunoglobulin collected from recovered coronavirus patients protect against COVID-19 and strengthen the immune system of new patients? Int J Mol Sci. 2020;21(7):2272. doi: . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Xie Y, Cao S, Dong H, et al. Effect of regular intravenous immunoglobulin therapy on prognosis of severe pneumonia in patients with COVID-19. J Infect, 2020;81(2):318–356. doi: . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Prete M, Favoino E, Catacchio G, et al. . SARS-CoV-2 infection complicated by inflammatory syndrome. Could high-dose human immunoglobulin for intravenous use (IVIG) be beneficial? Autoimmun Rev. 2020;19(7):102559. doi: 10.1016/j.autrev.2020.102559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Cao W, Liu X, Bai T, et al. . High-dose intravenous immunoglobulin as a therapeutic option for deteriorating patients with coronavirus disease 2019. Open Forum Infect Dis. 2020;7(3):ofaa102. doi: 10.1093/ofid/ofaa102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Aljaberi R, Wishah K.. Positive outcome in a patient with coronavirus disease 2019 and common variable immunodeficiency after intravenous immunoglobulin. Ann Allergy Asthma Immunol. 2020;125(3):349–350. doi: . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Feldstein LR, Rose EB, Horwitz SM, et al. . Multisystem inflammatory syndrome in U.S. children and adolescents. N Engl J Med. 2020;383(4):334–346. doi: 10.1056/NEJMoa2021680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Mariani E, Cattini L, Neri S, et al. . Simultaneous evaluation of circulating chemokine and cytokine profiles in elderly subjects by multiplex technology: relationship with zinc status. Biogerontology. 2006;7(5-6):449–459. doi: 10.1007/s10522-006-9060-8. [DOI] [PubMed] [Google Scholar]
- 80.Alexander J, Tinkov A, Strand TA, et al. . Early nutritional interventions with zinc, selenium and vitamin D for raising anti-viral resistance against progressive COVID-19. Nutrients. 2020;12(8):2358. doi: 10.3390/nu12082358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Bauer SR, Kapoor A, Rath M, et al. What is the role of supplementation with ascorbic acid, zinc, vitamin D, or N-acetylcysteine for prevention or treatment of COVID-19? Cleve Clin J Med, 2020. doi:. [DOI] [PubMed] [Google Scholar]
- 82.Mayor-Ibarguren A, Busca-Arenzana C, Robles-Marhuenda Á.. A hypothesis for the possible role of zinc in the immunological pathways related to COVID-19 infection. Front Immunol. 2020;11:1736. doi: 10.3389/fimmu.2020.01736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Derwand R, Scholz M.. Does zinc supplementation enhance the clinical efficacy of chloroquine/hydroxychloroquine to win today’s battle against COVID-19? Med Hypotheses. 2020;142:109815. doi: 10.1016/j.mehy.2020.109815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Malek Mahdavi A. A brief review of interplay between vitamin D and angiotensin-converting enzyme 2: Implications for a potential treatment for COVID-19. Rev Med Virol. 2020;30(5):e2119. doi: 10.1002/rmv.2119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Grant WB, Lahore H, McDonnell SL, et al. . Evidence that Vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients. 2020;12(4):988. doi: 10.3390/nu12040988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Xu J, Yang J, Chen J, et al. . Vitamin D alleviates lipopolysaccharide‑induced acute lung injury via regulation of the renin‑angiotensin system. Mol Med Rep. 2017;16(5):7432–7438. doi: 10.3892/mmr.2017.7546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ferrari D, Locatelli M. No significant association between vitamin D and COVID-19. A retrospective study from a northern Italian hospital. Int J Vitam Nutr Res, 2020; 1–4. doi: . [DOI] [PubMed] [Google Scholar]
- 88.South AM, Brady TM, Flynn JT.. ACE2 (Angiotensin-Converting Enzyme 2), COVID-19, and ACE Inhibitor and Ang II (Angiotensin II) receptor blocker use during the pandemic: The pediatric perspective. Hypertension. 2020;76(1):16–22. doi: 10.1161/HYPERTENSIONAHA.120.15291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Khashkhusha TR, Chan JSK, Harky A.. ACE inhibitors and COVID-19: We don’t know yet. J Card Surg. 2020;35(6):1172–1173. doi: 10.1111/jocs.14582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Subir R, Jagat JM, Kalyan KG.. Pros and cons for use of statins in people with coronavirus disease-19 (COVID-19). Diabetes Metab Syndr. 2020;14(5):1225–1229. doi: 10.1016/j.dsx.2020.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Glende J, Schwegmann-Wessels C, Al-Falah M, et al. . Importance of cholesterol-rich membrane microdomains in the interaction of the S protein of SARS-coronavirus with the cellular receptor angiotensin-converting enzyme 2. Virology. 2008;381(2):215–221. doi: 10.1016/j.virol.2008.08.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Iribarren C, Jacobs DR, Sidney S, et al. . Cohort study of serum total cholesterol and in-hospital incidence of infectious diseases. Epidemiol Infect. 1998;121(2):335–347. doi: 10.1017/s0950268898001435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Zhang X-J, Qin J-J, Cheng X, et al. . In-hospital use of statins is associated with a reduced risk of mortality among individuals with COVID-19. Cell Metab. 2020;32(2):176–187.e4. doi: 10.1016/j.cmet.2020.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Li D, Ji H, Zhao B, et al. . Therapeutic effect of ulinastatin on pulmonary fibrosis via downregulation of TGF‑β1, TNF‑α and NF‑κB. Mol Med Rep. 2018;17(1):1717–1723. doi: 10.3892/mmr.2017.8056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Chen Q, Hu C, Liu Y, et al. . Safety and tolerability of high-dose ulinastatin after 2-hour intravenous infusion in adult healthy Chinese volunteers: A randomized, double-blind, placebo-controlled, ascending-dose study. PloS One. 2017;12(5):e0177425. doi: 10.1371/journal.pone.0177425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Chen X, Feng Y, Shen X, et al. . Anti-sepsis protection of Xuebijing injection is mediated by differential regulation of pro- and anti-inflammatory Th17 and T regulatory cells in a murine model of polymicrobial sepsis. J Ethnopharmacol. 2018;211:358–365. doi: 10.1016/j.jep.2017.10.001. [DOI] [PubMed] [Google Scholar]
- 97.Zhu HY, Huang H, Shi XL, et al. . Qiangzhi decoction protects mice from influenza A pneumonia through inhibition of inflammatory cytokine storm. Chin J Integr Med. 2015;21(5):376–383. doi: 10.1007/s11655-014-2020-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Hardin CC, Hibbert K.. ECMO for Severe ARDS. N Engl J Med. 2018;378(21):2032–2034. doi: 10.1056/NEJMe1802676. [DOI] [PubMed] [Google Scholar]
- 99.Antebi B, Mohammadipoor A, Batchinsky AI, et al. . The promise of mesenchymal stem cell therapy for acute respiratory distress syndrome. J Trauma Acute Care Surg. 2018;84(1):183–191. doi: 10.1097/TA.0000000000001713. [DOI] [PubMed] [Google Scholar]
- 100.Wu X, Dao Thi VL, Huang Y, et al. . Intrinsic immunity shapes viral resistance of stem cells. Cell. 2018;172(3):423–438.e25. doi: 10.1016/j.cell.2017.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Atluri S, Manchikanti L, Hirsch JA.. Expanded Umbilical Cord Mesenchymal Stem Cells (UC-MSCs) as a therapeutic strategy in managing critically ill COVID-19 Patients: The case for compassionate use. Pain Physician. 2020;23(2):E71–e83. [PubMed] [Google Scholar]