Abstract
Fat oxidation during exercise is associated with cardio-metabolic benefits, but the extent of which whole-body exercise modality elicits the greatest fat oxidation remains unclear. We investigated the effects of treadmill, elliptical and rowing exercise on fat oxidation in healthy individuals. Nine healthy males participated in three, peak oxygen consumption tests, on a treadmill, elliptical and rowing ergometer. Indirect calorimetry was used to assess maximal oxygen consumption (V̇O2peak), maximal fat oxidation (MFO) rates, and the exercise intensity MFO occurred (Fatmax). Mixed venous blood was collected to assess lactate and blood gases concentrations. While V̇O2peak was similar between exercise modalities, MFO rates were higher on the treadmill (mean ± SD; 0.61 ± 0.06 g·min-1) compared to both the elliptical (0.41 ± 0.08 g·min-1, p = 0.022) and the rower (0.40 ± 0.08 g·min-1, p = 0.017). Fatmax values were also significantly higher on the treadmill (56.0 ± 6.2 %V̇O2peak) compared to both the elliptical (36.8 ± 5.4 %V̇O2peak, p = 0.049) and rower (31.6 ± 5.0 %V̇O2peak, p = 0.021). Post-exercise blood lactate concentrations were also significantly lower following treadmill exercise (p = 0.021). Exercising on a treadmill maximizes fat oxidation to a greater extent than elliptical and rowing exercises, and remains an important exercise modality to improve fat oxidation, and consequently, cardio-metabolic health.
Key points.
The ability to oxidize fat has been associated with improved oxidative enzymes activity and mitochondrial biogenesis.
The present study examined the effects of treadmill, elliptical, and rower exercises on maximal fat oxidation rates (MFO), the intensity were MFO was observed (Fatmax) and on fat oxidation curves in healthy and young participants.
Both MFO and Fatmax were higher during treadmill exercise. Multiple linear mixed-effects regression analyses further revealed an effect of exercise modality on fat oxidation curves.
Adequate selection of exercise modality during training may have a meaningful impact on substrate oxidation. Treadmill exercise should be considered in training design for those looking to maintain or improve metabolic profiling.
Key words: Substrate oxidation, indirect calorimetry, exercise modality, metabolism
Introduction
During aerobic exercise, oxidation of carbohydrates and fat, in exercising muscle, is influenced by fitness level (Holloszy and Coyle, 1984), diet (Flatt, 1987), sex (Venables et al., 2005), and intrinsic biochemical factors (e.g. pH, temperature, substrate availability, etc.) (Gevers, 1979; Robergs et al., 2004; Tarnopolsky et al., 1995). The oxidation of substrates is also known to be further altered extrinsically, via exercise duration (Phillips et al., 1996), and exercise modality (Achten et al., 2003; Capostagno and Bosch, 2010; Egan et al., 2016). Substrate metabolism shifts towards greater fat oxidation and reaches higher rates of maximal fat oxidation (MFO) during walking and running compared to cycling (Achten et al., 2003; Capostagno and Bosch, 2010; Chenevière et al., 2010; Knechtle et al., 2004). These results are believed to stem from differences in muscle recruitment patterns and Type II muscle fiber recruitment in cycling from lower muscle mass contribution to total energy production (Achten et al., 2003; Chenevière et al., 2010). Additionally, Chenevière et al. (2010) found that the exercise intensity where MFO occurs, known as Fatmax (Jeukendrup and Achten, 2001), occurs at higher intensities on the treadmill compared to a cycle ergometer, while Achten et al. (2003) found no significant difference in Fatmax values between these modalities.
Intrinsically, as exercise intensity increases, greater glycolytic activity results in an increased accumulation of lactate and hydrogen ions, decreasing muscle pH. Reductions in fatty acid oxidation are strongly correlated with the accumulation of plasma lactate (Achten and Jeukendrup, 2004). Achten and Jeukendrup (2004) also found that the initial rise in plasma lactate occurred at the same intensity that elicited MFO. It is suggested that decreased muscle pH, due to the accumulation of glycolytic by-products, inhibits the transport of long-chain fatty acid into the mitochondria, reducing fatty acid oxidation (Achten and Jeukendrup, 2004; Sidossis et al., 1997; Starritt et al., 2000).
The elliptical and rower are two modalities increasing in popularity for their ability to offer meaningful aerobic benefits (Brown et al., 2015; Hagerman, 1984; Mercer et al., 2001), as they incorporate whole-body movements. They further provide alternatives to those with limited range of motions and physical disabilities, such as cerebral palsy, multiple sclerosis, and others. Importantly, it has been reported that heart rate and oxygen uptake during elliptical exercise is similar to treadmill when both exercises are performed at self-selected intensities (Porcari et al., 1998). Carey et al. (1974) also demonstrated that maximal oxygen uptake values were not significantly different between treadmill and rowing exercise. Despite some aerobic similarities, MFO and Fatmax was reported higher during rowing compared to cycling (Egan et al., 2016). The crossover point, where energy contribution from CHO and fat to total energy expenditure is equal (Brooks and Mercier, 1994), was also reported to occur at a higher relative exercise intensity during rowing than cycling (Egan et al., 2016). Although the elliptical and rower involve a large level of muscle mass (Egan et al., 2016), they involve more regular activation of muscles in the upper limbs compared to treadmill exercise (Bazzucchi et al., 2013; Hagerman, 1984; Sozen, 2010). The muscles of the upper limbs have a relatively smaller surface area than those of the lower body (Young et al. 1986), and the metabolic demand across these muscle fibers may be larger during elliptical and rowing exercise, leading to a greater reliance on Type II muscle fibers. This could possibly result in not only a greater production of lactate, but also an earlier reliance on carbohydrate oxidation.
Fat oxidation during exercise has been associated to increases in oxidative enzymatic activity and mitochondrial biogenesis in skeletal muscle (Little et al., 2010). Training designed to improve fat oxidation could help to gain or maintain a healthy metabolic profile via an appropriate selection of exercise modality. We consequently investigated the effects of treadmill, elliptical and rowing exercise on MFO, Fatmax, the crossover point, and rates of fat oxidation. It was hypothesized that MFO, Fatmax, the crossover point, and absolute fat oxidation would be greater during the treadmill exercise compared to the elliptical or rowing exercises.
Methods
Participants
Nine healthy males (age: 22 ± 1.1 years, height: 1.78 ± 0.03 m, weight: 78.0 ± 3.7 kg, body fat: 8.70 ± 1.15%) participated in the study. Informed written consent was provided prior to testing. Participants were screened with a Get Active Questionnaire and a health screening form for health conditions or diseases that could be aggravated by exercise. None of the participants were on prescribed medications. All procedures were in accordance with the latest revision of the Declaration of Helsinki and approved by the university’s Research Ethics Board.
Experimental design
Participants reported to the laboratory, following a 12-hour fasted state, for one familiarization session, followed by three experimental exercise sessions, in a balanced design, consisting of an adapted incremental V̇O2peak protocol to exhaustion during either: 1) treadmill exercise (TM), 2) elliptical exercise (EL), or 3) rowing exercise (ROW). Participants performed each exercise session at the same time of day, between 07:00 h and 11:00 h, five to seven days apart, to minimize the influence of circadian variance. Participants were also requested to avoid alcohol consumption, strenuous exercise, caffeine and tobacco 24 hours prior to each experimental exercise session. To control for dietary influences on metabolism during exercise, participants maintained the same diet each day prior to their exercise sessions, with the built-in assumption that this energy standardization method yields significant variability in food intake between participants (Jeacocke and Burke, 2010).
Experimental protocol
Familiarization session. Participants completed one familiarization session to become accustomed to the exercise protocols, instrumentation, and each of the exercise modalities. Additionally, anthropometric measures were collected, and percent body fat was estimated (Jackson and Pollock, 1978).
Treadmill Exercise (TM). An adapted incremental V̇O2peak exercise protocol was used to ensure that there was a sufficient number of stages to build substrate oxidation curves (Achten et al., 2002; Gagnon et al., 2019). Participants began walking at 3.5 km·h-1 with a 1% gradient for the first three-minute stage on the treadmill (BodyGuard 312C Treadmill, BodyGuard Fitness Technology Inc., Saint-Georges, Quebec, CAN). Following the first stage, the speed was increased every three minutes to 4.3 km·h-1, 5.5 km·h-1, 6.4 km·h-1, and finally 7.6 km·h-1 for the next four stages. Following the first five stages, the gradient was increased by 2% at every subsequent stage. The gradient of the treadmill was increased until the participants’ RER values reached 1.0, indicating that only carbohydrates were being used for energy production, at which point the speed was immediately increased to 9.7 km·h-1 with a gradient of 10%. The gradient was then increased by 2% every minute until V̇O2peak was reached.
Elliptical Exercise (EL). An adapted incremental V̇O2peak exercise protocol was used to ensure that there was a sufficient number of stages to build substrate oxidation curves (Dalleck et al., 2004). Participants began exercising at 30 W for the first three-minute stage on the elliptical (S70 Ascent Trainer, Vision Fitness, Cottage Grove, Wisconsin, USA). The placement of hands and feet on the elliptical were standardized for all participants. The gradient was maintained at a midpoint between the low to high ranges, pre-determined on the elliptical, and was maintained throughout the entire test (Dalleck et al., 2004). Every three minutes, the power output was increased by 20 W until participants reached an RER of 1.0. Power output was then increased by 20 W every minute until V̇O2peak was reached. Participants were required to maintain the appropriate exercise intensity by keeping the average power output at the target value.
Rowing Exercise (ROW). An adapted incremental V̇O2peak exercise protocol was used to ensure that there was a sufficient number of stages to build substrate oxidation curves (Egan et al., 2016). Participants began rowing at 30 W for the first three-minute stage on the rower (Model D Rowing Ergometer, Concept2 Inc., Morrisville, Vermont, USA). To standardize the level of drag for all participants, the coefficient of drag was maintained at 130 during all subsequent stages (Egan et al., 2016). The power output was increased by 30 W every three minutes until participants reached an RER of 1.0. Power output was then increased by 30 W every minute until V̇O2peak was reached. Participants were required to maintain the appropriate exercise intensity by keeping the average power output at the target value.
For exercise on all three modalities, V̇O2 was considered to be peak when at least two of the three following conditions occurred, 1) if heart rate did not significantly increase with increasing workload (defined as an increase of no more than 5 bpm), 2) if RER was greater than 1.05, or 3) if V̇O2 did not significantly increase with increasing workload (defined as an increase of no more than 2 ml·kg-1·min-1).
Instrumentation and measures
Heart rate (HR) was recorded continuously using a Polar Heart Rate Monitor (H10). Fingertip blood samples were collected using a contact-activated lancet (BD Microtainer, BD, USA) prior to, and immediately following, each exercise session to assess blood lactate (Lactate Pro Test Strip, Arkray, Kyoto, Japan) and glucose (FreeStyle Precision Neo, Abbott Point of Care, Illinois, USA) concentrations. Venous blood samples, from an antecubital vein, were also collected prior to, and immediately following each exercise session to assess concentrations of pH levels, base excess of extracellular fluid (BEecf), bicarbonate levels (HCO3-), partial pressure of oxygen (pO2), partial pressure of carbon dioxide (pCO2), total carbon dioxide levels (TCO2), and oxygen saturation (SO2) (i-STAT Handheld, Abbott Point of Care, Illinois, USA).
Breath-by-breath cardiorespiratory data were collected continuously during exercise using an open circuit ergospirometer in breath-by-breath mode (CPX, MGC Diagnostics, Saint Paul, MN) to obtain measures of oxygen consumption (V̇O2), carbon dioxide production (V̇CO2), respiratory exchange ratio (RER), and ventilation rate (V̇E). Gas analyzers were calibrated with air tanks containing 12% O2 and 5% CO2 and the gas flow sensor was calibrated using a 3L calibration syringe before each exercise session. Gas flow was measured through a bidirectional pitot tube flow sensor attached to the face-fitting mask worn by all participants during the exercise sessions.
Calculations and Analyses
For each three-minute stage of the V̇O2peak protocols, the last thirty seconds was averaged for the following measures: HR, V̇O2, V̇CO2, and V̇E. The highest mean value over the entire exercise protocol for both HR and V̇E was used to determine peak heart rate (HRpeak), and peak ventilation rate (V̇Epeak) values for each participant.
Calculations of fat and carbohydrate oxidation were completed for each stage using the following stoichiometric equations (Jeukendrup and Wallis, 2005):
|
(<50% V̇O2peak) CHO (g·min-1) = 4.344 • V̇CO2 – 3.061 • V̇O2 – 0.40 • n |
(1) |
|
(>50% V̇O2peak) CHO (g·min-1) = 4.210 • V̇CO2 – 2.962 • V̇O2 – 0.40 • n |
(2) |
| Fat (g·min-1) = 1.695 • V̇O2 – 1.701 • V̇CO2 – 1.77 • n | (3) |
where values of V̇O2 and V̇CO2 (L·min-1) were collected during exercise and “n” represents nitrogen excretion from the oxidation of proteins (135 μg·kg-1·min-1) (Jeukendrup and Wallis, 2005). Protein oxidation was not directly calculated as short-term exercise does not alter its contribution (Vallerand and Jacobs, 1989).
Normal distribution of the data was verified via skewness and kurtosis tests, Kolmogorov-Smirnov and Shapiro-Wilk tests, and visual inspection of histograms. Sphericity of the data was verified through Mauchly’s test. One-way analyses of variance with repeated measures were used to identify differences between exercise modalities for the following variables: MFO, Fatmax, V̇O2peak, HRpeak, V̇Epeak, V̇O2/HR, and the crossover point. Two-way analyses of variance with repeated measures (factors: exercise modality and time) were performed to identify differences in blood variables from pre and post exercise between exercise conditions, and for V̇E/V̇O2, V̇E/V̇CO2 throughout each condition. When a significant F ratio was observed, pairwise comparisons with Bonferroni correction was conducted. To assess whether differences were present among substrate oxidation curves between exercise modalities, multiple linear mixed-effects regression analyses were performed to examine differences among relative and absolute oxidation curves with %V̇O2peak, and exercise modality as variables. To respond to the quadratic (fat oxidation) and exponential (CHO oxidation) natures of the curves, %V̇O2peak squared was added within the model. Statistical significance was set at p < 0.05. It was estimated that a sample of nine participants would be sufficient for detection of large effect sizes (η2 = 0.20, f = 0.5), assuming standard deviations similar to those previously observed (Capostagno and Bosch, 2010) with a statistical power (1-β err. prob.) of 80% (G*power, Düsseldorf, Germany). Statistical analyses were completed using SPSS statistical package (Version 24, IBM, Armonk, NY, 2016).
Results
Cardiorespiratory Variables
There was no significant difference in V̇O2peak values across the three exercise conditions (p ≥ 0.054, TM: 60.1 ± 7.0 mL·kg-1·min-1, EL: 57.9 ± 7.9 mL·kg-1·min-1, ROW: 57.6 ± 7.9 mL·kg-1·min-1). However, there was a significant difference in mean HRpeak between modalities (p = 0.004), where HRpeak was higher on the TM (195 ± 5 bpm) vs. ROW (p = 0.005, 189 ± 7 bpm) but not EL (194 ± 6 bpm). Mean V̇Epeak was also not different between the exercise modalities (p = 0.465, TM: 139.2 ± 15.8 L·min-1, EL: 143.4 ± 10.6 L·min-1, ROW: 144.6 ± 13.3 L·min-1). Mean V̇O2/HR was not significantly different between modalities (p = 0.295). V̇E/V̇O2 and V̇E/V̇CO2 were not different between exercise modalities over the duration of exercise (p ≥ 0.070).
Metabolism
Mean MFO was higher in TM vs. EL (p = 0.022) and ROW (p = 0.017), but there was no significant difference between EL and ROW (p = 1.000, TM: 0.61 ± 0.17 g·min-1, EL: 0.41 ± 0.23 g·min-1, ROW: 0.40 ± 0.23 g·min-1) (Figure 1A). Similarly, Fatmax was higher in TM vs. EL (p = 0.049) and ROW (p = 0.021). However, mean Fatmax was not different between EL and ROW (p = 0.765, TM: 55.9 ± 18.5 %V̇O2peak, EL: 36.8 ± 16.2 %V̇O2peak, ROW: 31.6 ± 14.9 %V̇O2peak) (Figure 1B). There was no difference observed in the mean crossover point between the exercise modalities (p = 0.388, TM: 42.2 ± 13.9 %V̇O2peak, EL: 34.0 ± 16.9 %V̇O2peak, ROW: 35.4 ± 15.9 %V̇O2peak).
Figure 1.
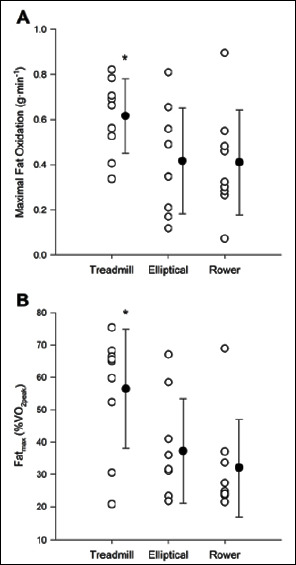
(A) Mean MFO rates between treadmill, elliptical and rowing exercise, (B) Mean Fatmax between treadmill, elliptical and rowing exercise. * significantly different from elliptical and rower (p < 0.05).
Figure 2 presents absolute rates of fat and CHO oxidation across the exercise intensity spectrum during treadmill, elliptical and rower exercise. Regression analyses revealed a strong influence of modality on both fat and CHO oxidation curve during exercise (p < 0.001). Examination of the fat oxidation curves revealed that curves became linear, and the distribution of the data was greater in the elliptical and rower exercises. The CHO oxidation curves were similar in shape between exercise modalities, but a greater distribution of the data was found in the elliptical condition. Results of the regression model for the relative fat and CHO oxidation are presented in Table 1.
Figure 2.
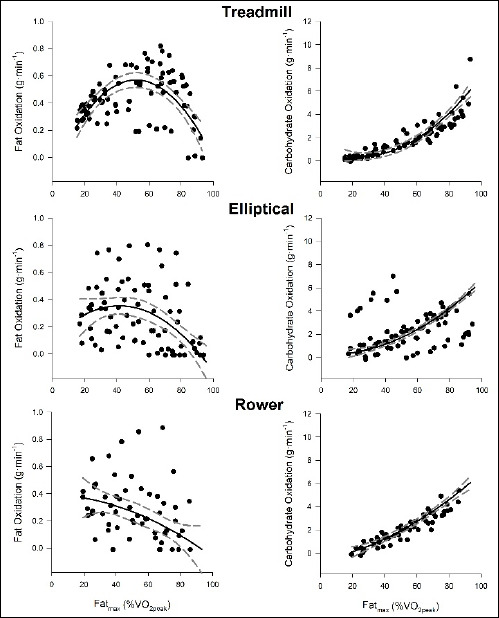
Scatter plot of fat and CHO oxidation over %V̇O2max during treadmill, elliptical and rowing exercise. Trend line represents the nonlinear 2nd order polynomial regression and dotted lines represents the 95% confidence intervals. Regression values are presented in Table 1.
Table 1.
Multiple linear mixed-effect regression analyses for treadmill, elliptical and rowing exercise for relative energy contribution of fat and carbohydrates to total energy expenditure, as plotted in Figure 2.
| Coefficient (β) | Std. Err. | Z | P value | 95% confidence interval | |||
|---|---|---|---|---|---|---|---|
| FAT | %V̇O2peak | 0.0323 | 0.0038 | 8.56 | 0.000 | 0.0249 | 0.397 |
| %V̇O2peak2 | -0.0004 | 0.0000 | -10.79 | 0.000 | -0.0004 | -0.0003 | |
| Modality | -0.1768 | 0.0340 | -5.20 | 0.000 | -0.2435 | -0.1101 | |
| Constant | -0.1992 | 0.1185 | -1.68 | 0.093 | -0.4314 | 0.0330 | |
| CHO | %V̇O2peak | -0.0244 | 0.0094 | -2.60 | 0.009 | -0.0428 | -0.0060 |
| %V̇O2peak2 | 0.0009 | 0.0000 | 10.63 | 0.000 | 0.0007 | 0.0011 | |
| Modality | 0.3696 | 0.0848 | 4.36 | 0.000 | 0.2034 | 0.5358 | |
| Constant | 0.5210 | 0.2742 | 1.90 | 0.057 | -0.0164 | 1.0585 | |
CHO – carbohydrate; %V̇O2peak – percent peak of oxygen consumption; %V̇O2peak2 – percent peak of oxygen consumption squared
Blood Variables
Mean pH was significantly lower post-exercise compared to pre-exercise for all three exercise modalities (p<0.001) (Fig. 3A). However, there was no difference in pH between exercise modalities (p = 0.061). Blood lactate concentrations increased across time (p < 0.001) (Figure 3B), and were also higher post-exercise in ROW compared to TM (p = 0.021). Blood glucose concentrations were not significantly different between exercise modalities (p = 0.801). However, glucose was significantly higher post-exercise compared to pre-exercise (p = 0.004, Pre-Exercise: 4.36 ± 0.16 mmol·L-1, Post-Exercise: 4.83 ± 0.17 mmol·L-1), regardless of exercise modality.
Figure 3.
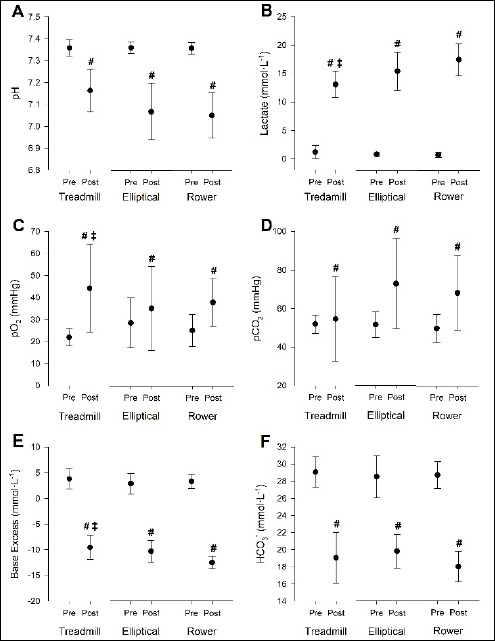
(A) Mean venous blood pH, (B) mean venous blood lactate concentrations, (C) mean partial pressure of oxygen, (D) mean partial pressure of carbon dioxide, (E) mean base excess of extracellular fluid, and (F) mean bicarbonate levels, pre- and post-exercise, between exercise modality. # significantly (p < 0.05) different from pre-exercise values, ‡ significantly (p < 0.05) different from rower post-exercise.
Mean pO2 (Figure 3C) and pCO2 (Figure 3D) were both significantly higher post-exercise compared to pre-exercise across exercise modalities (pO2: p = 0.004; pCO2: p = 0.006). Additionally, pO2 was higher post-exercise in TM vs. ROW (p = 0.021). There was also no significant difference in pCO2 between exercise modalities (p = 0.158).
Mean BEecf was significantly lower post-exercise compared to pre-exercise (p ≤ 0.001) (Figure 3E). BEecf was also significantly lower post-exercise during ROW exercise compared vs. TM (p = 0.045) and EL (p = 0.037). Mean HCO3- was significantly lower post-exercise compared to pre-exercise (p ≤ 0.001) (Figure 3F). There was no significant difference in HCO3- between exercise modalities (p = 0.304).
There was no significant difference in mean sO2% between exercise modalities (p = 0.767, TM: 47.4 ± 24.27 %, EL: 48.4 ± 28.24 %, ROW: 43.5 ± 19.60 %) or between time points (p = 0.067, Pre-Exercise: 40.1 ± 17.75 %, Post-Exercise: 52.81 ± 27.72 %). Mean TCO2 was significantly lower post-exercise (20.9 ± 2.79 mmHg) compared to pre-exercise (p ≤ 0.001, 30.5 ± 2.06 mmHg). There was no significant difference in TCO2 between exercise modalities (p = 0.482, TM: 25.8 ± 5.82 mmHg, EL: 26.0 ± 4.86 mmHg, ROW: 25.28 ± 5.81 mmHg).
Discussion
The present study investigated the influence of whole-body exercises, including treadmill, elliptical, and rower exercise, on fat oxidation parameters in healthy individuals. The main findings of this study were: i) MFO and Fatmax were higher during the treadmill condition compared to both the elliptical and rower conditions, ii) there was no difference in crossover points between the three conditions, and iii) exercise modality influenced absolute rates of CHO and fat oxidation, and thereby modulated their respective curves across the exercise intensity spectrum. The present study supports previous work demonstrating that treadmill exercise results in higher MFO rates than other exercise modalities, and greater metabolic benefits may stem from walking and running exercise (Achten et al., 2003; Chenevière et al., 2010). This work provides critical metabolic insights that may promote further research focused on those suffering from metabolic conditions and physical disabilities.
V̇O2peak values were similar between all three exercise modalities. Maximal V̇O2 values reflect the quantity of active muscle mass during exercise, indicating that all three modalities elicited similar levels of muscle activation. Previous research has identified level of active muscle mass as a mechanism for differences in fat oxidation, particularly between cycling and other whole-body modalities. Achten et al. (2003) and Chenevière et al. (2010) observed higher Fatmax values during treadmill exercise compared to cycling in moderately trained and trained individuals. Walking and running on a treadmill utilizes different muscle contraction regimes and elicits greater mechanical efficiency compared to cycling, which may be extended to include elliptical and rowing exercise. The eccentric component of the landing phase during treadmill exercise allows for muscle ‘preloading’, where mechanical energy is absorbed by the muscle and may improve the efficiency of the next contraction (Cavagna 1977; Chenevière et al. 2010). Additionally, the return of elastic energy may reduce recruitment of larger motor units consisting of greater Type II muscle fibres, influencing CHO metabolism, lactate production, and onset of peripheral fatigue (Carter et al. 2000; Chenevière et al. 2010).
Egan et al. (2016) and Hagerman et al. (1988) both found that MFO was higher on the rower than the cycle ergometer, again attributable to greater muscle mass activation during rowing. However, the mean MFO rates and Fatmax values in the present study were lower on the rowing ergometer than Egan et al. (2016) reported (MFO: 0.71 ± 0.05 g·min-1, Fatmax: 42 ± 1 %V̇O2peak). Modality-specific training status is highly correlated with MFO and metabolic efficiency, promoting greater fat oxidation (Hagerman, 1984). The present study used healthy males with no explicit training in rowing, while Egan et al. (2016) used club standard rowers. However, this study was designed to provide information for, and reflect how recreational individuals respond to different exercise modalities. Despite this, the lack of familiarization and training with alternative whole-body exercises, particularly during rowing, may have led to decreased efficiency and greater reliance on the muscles of the upper body compared to those who utilize these modalities more often. While untrained rowers display similar muscle synergies (spatiotemporal pattern of muscle activation) as trained rowers (Shaharudin and Agrawal, 2016), body posture and stroke technique can vary greatly between trained and untrained rowers (Cerne et al., 2013). Secher (1993) also detailed that untrained individuals will utilize less upper body movement and focus on the drive of the legs during rowing strokes with increased training. Therefore, even though similar levels in activation of muscle mass occurred between exercise modalities, as indicated by similar V̇O2peak values, the specific muscles utilized for each modality may explain differences in fat oxidation.
Moreover, the lower MFO and Fatmax in elliptical and rower vs. treadmill could be associated with an increased recruitment of Type II muscle fibers (Achten et al., 2003; Chenevière et al., 2010). The present study demonstrated higher blood lactate concentrations post-exercise in both the elliptical and rowing conditions. Type II muscle fibres are less efficient than Type I muscle fibers since they rely primarily on carbohydrates as their energy source and produce lactate as a by-product (Knechtle et al., 2004). Treadmill exercise primarily uses muscles of the lower extremities, such as the gastrocnemius (Sozen, 2010), composed of a high percentage of Type I fibres (Costill et al., 1976). The elliptical and rower involve greater activation of many muscles of the upper body compared to the treadmill (Bazzucchi et al., 2013; Hagerman, 1984; Sozen, 2010). Although not all of the muscles of the upper body are mainly composed of Type II muscle fibres, the muscles of the upper limbs (i.e. Biceps Brachii, Triceps Brachii) have been known to have a greater percentage of these fibres (Klein et al., 2003; Miller et al., 1993). Finally, these muscles also generally have a smaller surface area than those of the lower body (Young et al., 1986) and the metabolic stress per muscle fibre is expected to be greater (Achten et al., 2003; Chenevière et al., 2010), thus increasing the production of lactate and CHO oxidation.
The increased reliance on muscles of the upper body, and subsequently Tyle II muscle fibres, may have influenced the overall fat oxidation curves for elliptical and rowing exercises (Figure 2). The regression analysis (Table 1) revealed that both fat and CHO oxidation curves were influenced by changes in %VO2, as well as exercise modality. Visually, curve fitting demonstrated a loss of curvilinearity of the fat oxidation curves in the elliptical and rowing exercise, as the apex of the curve (i.e. the point of maximal fat oxidation) occurred at low exercise intensities. Fatmax values for elliptical and rowing exercises were similar to those observed in overweight (Bogdanis et al., 2008) and inactive obese individuals during treadmill walking (Perez-Martin et al., 2001). The reduced ability of muscles to oxidize fat observed in obesity is suggested to result from increased intracellular lipid accumulation, leading to production of reactive oxygen species and disruption of mitochondiral oxidative enzymes (Wells et al., 2008). Therefore, factors such as exercise modalitiy and body weight, can heavily influence intrinsic cellular processes in skeletal muscle, altering substrate metabolism in these tissues.
The accumulation of lactate, and subsequent decrease in pH, lowers fatty acid oxidation with increasing exercise intensities (Achten and Jeukendrup, 2004) as they are known to inhibit long-chain fatty acid entry into the mitochondria (Sidossis et al., 1997). Lower muscle pH decreases the activity of carnitine palmitoyl transferase 1 (CPT-1), responsible for transporting long-chain fatty acids into the mitochondria (Starritt et al., 2000). Even though we did not observe a difference in plasma pH at the end of exercise between modalities, lactate concentrations were different between treadmill and rower, and it has been suggested that plasma lactate concentrations reflect changes in muscle pH (MacLean et al., 1999; Street et al., 2001). Our results further substantiate this response with higher base excess of extracellular fluid in the treadmill and elliptical compared to the rower condition. Previous research has shown that the lactate threshold occurs at a lower V̇O2 during incremental rowing exercise compared to treadmill, even when the same V̇O2max is attained (Weltman et al., 1994). Blood lactate increases in a curvilinear fashion with increasing exercise intensity, resulting in an exponential increase during work above the lactate threshold (Goodwin et al., 2007). The onset of blood lactate accumulation is also highly correlation with the onset of muscle deoxygenation (Grassi et al., 1999). The potential for increased lactate accumulation at an earlier stage (lower V̇O2) during rowing exercise may have resulted in a prompter shift in metabolism, and therefore a higher concentrations of lactate at the end of exercise compared to the treadmill.
Interestingly, a higher venous partial pressure of O2 was observed in the treadmill condition compared to the rower at the end of exercise. Increases in pO2 are generally indicative of impaired diffusive O2 capacity from the circulatory system to active muscles, as observed in mitochondrial myopathic patients (Taivassalo et al., 2002). Additionally, minute ventilation and oxygen pulse remained unchanged across conditions, thereby eliminating the possibility of a higher O2 uptake at the pulmonary level. While a direct relationship would conclude that higher O2 availability in venous blood would be the by-product of lower O2 extraction in the rower condition, V̇O2peak was similar across conditions. However, without blood flow or arterial blood gases we cannot conclusively state that lower O2 extraction was at play in the present results.
Considerations
Limited use of an elliptical and/or rowing ergometer vs. treadmill in normal exercise routines may have impacted our results, as familiarity and technique with the elliptical and rowing ergometer varied between participants. Even though proper technical instructions and a familiarization session were provided, we cannot ignore possible influence of technique and lower mechanical efficiency, especially during rowing, on substrate oxidation. Therefore, these results may not be generalizable to individuals who use an elliptical or rowing ergometer frequently. Additionally, blood samples were only obtained from the antecubital vein for all three exercise modalities. Blood samples for the rowing and elliptical exercises may have been influenced by the metabolic activity of the arm due to greater upper extremity muscle activation during these exercises. Moreover, the sample used in the present study was composed of young and healthy individuals. Whether clinical or pathological populations would present similar metabolic responses to a variety of exercise modalities remains unknown.
Conclusion
While treadmill, elliptical and rower exercise modalities yield similar V̇O2peak values, the research findings of the present study demonstrated that treadmill exercise elicited higher MFO, Fatmax, and absolute rates of fat oxidation compared to elliptical and rower exercise, and that substrate oxidation curves were clearly influenced by exercise modality. The treadmill remains the most efficient modality to increase fat oxidation during exercise and should be considered in training design for those looking to maintain a healthy metabolic profile.
Acknowledgements
Thank you to all who participated in this study. We also want to thank Robert Jack and Francis Theriault for their technical assistance, and Dr. Bruce Oddson for his support in statistical analyses. The experiments comply with the current laws of the country in which they were performed. The authors report no conflict of interest. The datasets generated during and/or analyzed during the current study are not publicly available, but are available from the corresponding author who was an organizer of the study.
Biographies
Michelle FILIPOVIC
Employment
Medical Student at the Northern Ontario School of Medicine.
Degree
BSc, (MD Cand.)
Research interests
Exercise physiology.
E-mail: mfilipovic@nosm.ca
Stephanie MUNTEN
Employment
Research Assistant in the Laboratory for Environmental Physiology, School of Kinesiology and Health Sciences at Laurentian University.
Degree
MSc
Research interests
Environmental exercise physiology, metabolism.
E-mail: smunten@laurentian.ca
Karl-Heinz HERZIG
Employment
Professor in the Faculty of Medicine at the University of Oulu
Degree
MD, PhD
Research interests
Environmental Physiology, Exercise Physiology, Metabolism, Gastroenterology, Peptides
E-mail: karl-heinz.herzig@oulu.fi
Dominique D. GAGNON
Employment
Assistant Professor in the Laboratory for Environmental Physiology, School of Kinesiology and Health Sciences at Laurentian University.
Degree
PhD
Research interests
Environmental Physiology, Exercise Physiology, Metabolism, oxygen transport.
E-mail: ddgagnon@laurentian.ca
References
- Achten J., Gleeson M., Jeukendrup A. E. (2002) Determination of the exercise intensity that elicits maximal fat oxidation. Medicine and Science in Sports and Exercise 34, 92-97. [DOI] [PubMed] [Google Scholar]
- Achten J., Jeukendrup A. E. (2004) Relation between plasm lactate concentration and fat oxidation rates over a wide range of exercise intensities. International Journal of Sports Medicine 25, 32-37. [DOI] [PubMed] [Google Scholar]
- Achten J., Venables M. C., Jeukendrup A. E. (2003) Fat oxidation rates are higher during running compared with cycling over a wide range of intensities. Metabolism: Clinical and Experimental 52, 747-752. [DOI] [PubMed] [Google Scholar]
- Bazzucchi I., Sbriccoli P., Nicolò A., Passerini A., Quinzi F., Felici F., Sacchetti M. (2013) Cardio-respiratory and electromyographic responses to ergometer and on-water rowing in elite rowers. European Journal of Applied Physiology 113, 1271-1277. [DOI] [PubMed] [Google Scholar]
- Bogdanis G. C., Vangelakoudi A., Maridaki M. (2008) Peak fat oxidation rate during walking in sedentary overweight men and women. Journal of Sports Science and Medicine 7(4), 525-531. [PMC free article] [PubMed] [Google Scholar]
- Brooks G. A., Mercier J. (1994) Balance of carbohydrate and lipid utilization during exercise: The ‘crossover’ concept. Journal of Applied Physiology 76, 2253-2261. [DOI] [PubMed] [Google Scholar]
- Brown A. B., Kueffner T. E., O’Mahony E. C., Lockard M. M. (2015) Validity of arm-leg elliptical ergometer for VO2max analysis. Journal of Strength and Conditioning Research 29, 1551-1555. [DOI] [PubMed] [Google Scholar]
- Capostagno B., Bosch A. (2010) Higher fat oxidation in running than cycling at the same exercise intensities. International Journal of Sport Nutrition and Exercise Metabolism 20, 44-55. [DOI] [PubMed] [Google Scholar]
- Carey P., Stensland M., Harley L. H. (1974) Comparison of oxygen uptake during maximal work on the treadmill and the rowing ergometer. Medicine and Science in Sports 6, 101-103. [PubMed] [Google Scholar]
- Carter H., Jones A. M., Barstow T. J., Burnley M., Williams C. A., Doust J. H. (2000) Oxygen uptake kinetics in treadmill running and cycle ergometry: a comparison. Journal of Applied Physiology 89, 899-907. [DOI] [PubMed] [Google Scholar]
- Cavagna G. A. (1977) Storage and utilization of elastic energy in skeletal muscle. Exercise and Sport Sciences Reviews 5, 89-129. [PubMed] [Google Scholar]
- Cerne T., Kamnik R., Vesnicer B., Zganec Gros J., Munih M. (2013) Differences between elite, junior and non-rowers in kinematic and kinetic parameters during ergometer rowing. Human Movement Science 32, 691-707. [DOI] [PubMed] [Google Scholar]
- Chenevière X., Malatesta D., Gojanovic B., Borrani F. (2010) Differences in whole-body fat oxidation kinetics between cycling and running. European Journal of Applied Physiology 109, 1037-1045. [DOI] [PubMed] [Google Scholar]
- Costill D. L., Daniels J., Evans W., Fink W., Krahenbuhl G., Saltin B. (1976) Skeletal muscle enzymes and fiber composition in male and female track athletes. Journal of Applied Physiology 40, 149-154. [DOI] [PubMed] [Google Scholar]
- Dalleck L. C., Kravitz L., Robergs R. A. (2004) Maximal exercise testing using the elliptical cross-trainer and treadmill. Journal of Exercise Physiology Online 7, 94-101. [Google Scholar]
- Egan B., Ashley D. T., Kennedy E., O’Connor P. L., O’Gorman D. J. (2016) Higher rate of fat oxidation during rowing compared with cycling ergometer exercise across a range of exercise intensities. Scandinavian Journal of Medicine & Science in Sports 26, 630-637. [DOI] [PubMed] [Google Scholar]
- Flatt J. P. (1987) Dietary fat, carbohydrate balance and weight maintenance; effect of exercise. American Journal of Clinical Nutrition 45, 296-306. [DOI] [PubMed] [Google Scholar]
- Gagnon D. D., Perrier L., Dorman S. C., Oddson B., Larivière C., Serresse O. (2019) Ambient temperature influences metabolic substrate oxidation curves during running and cycling in healthy men. European Journal of Sport Science 20(6), 1-10. [DOI] [PubMed] [Google Scholar]
- Gevers W. (1979) Generations of protons by metabolic processes other than glycolysis in muscle cells: a critical review. Journal of Molecular and Cellular Cardiology 11, 325-330. [DOI] [PubMed] [Google Scholar]
- Goodwin M. L., Harris J. E., Hernandez A., Gladden L. B. (2007) Blood lactate measurements and analysis during exercise: A guide for clinicians. Journal of Diabetes Science and Technology 1, 558-569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grassi B., Quaresima V., Marconi C., Ferrari M., Cerretelli P. (1999) Blood lactate accumulation and muscle deoxygenation during incremental exercise. Journal of Applied Physiology 87, 348-355. [DOI] [PubMed] [Google Scholar]
- Hagerman F. C., Lawrence R. A., Mansfield M. C. (1988) A comparison of energy expenditure during rowing and cycling ergometry. Medicine and Science in Sports and Exercise 20, 479-488. [PubMed] [Google Scholar]
- Hagerman F. C. (1984) Applied physiology of rowing. Sports Medicine 1, 303-326. [DOI] [PubMed] [Google Scholar]
- Holloszy J. O., Coyle E. F. (1984) Adaptations of skeletal muscle to endurance exercise and their metabolic consequences. Journal of Applied Physiology 56, 831-838. [DOI] [PubMed] [Google Scholar]
- Jackson A. S., Pollock M. L. (1978) Generalized equations for predicting body density. British Journal of Nutrition 40, 497-504. [DOI] [PubMed] [Google Scholar]
- Jeacocke N. A., Burke L. M. (2010) Methods to standardize dietary intake before performance testing. International Journal of Sport Nutrition and Exercise Metabolism 20, 87-103. [DOI] [PubMed] [Google Scholar]
- Jeukendrup A. E., Achten J. (2001) Fatmax: A new concept to optimize fat oxidation during exercise? European Journal of Sport Science 1, 1-5. [Google Scholar]
- Jeukendrup A. E., Wallis G. A. (2005) Measurement of substrate oxidation during exercise by means of gas exchange measurements. International Journal of Sports Medicine 26, 28-37. [DOI] [PubMed] [Google Scholar]
- Klein C. S., Marsh G. D., Petrella R. J., Rice C. L. (2003) Muscle fiber number in the biceps brachii muscle of young and old men. Muscle and Nerve 28, 62-68. [DOI] [PubMed] [Google Scholar]
- Knechtle B., Müller G., Willmann F., Kotteck K., Eser P., Knecht H. (2004) Fat oxidation in men and women endurance athletes in running and cycling. International Journal of Sports Medicine 25, 38-44. [DOI] [PubMed] [Google Scholar]
- Little J.P., Safdar A., Wilkin G.P., Tarnopolsky M.A, Gibala M.J. (2010) A practical model of low-volume high-intensity interval training induces mitochondrial biogenesis in human skeletal muscle: potential mechanisms. Journal of Physiology 588, 1011-1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacLean D. A., Bangsbo J., Saltin B. (1999) Muscle interstitial glucose and lactate levels during dynamic exercise in humans determined by microdialysis. Journal of Applied Physiology 87, 1483-1490. [DOI] [PubMed] [Google Scholar]
- Mercer J. A., Dufek J. S., Bates B. T. (2001) Analysis of peak oxygen consumption and heart rate during elliptical and treadmill exercise. Journal of Sport Rehabilitation 10, 48-56. [Google Scholar]
- Miller A. E., MacDougall J. D., Tarnopolsky M. A., Sale D. G. (1993) Gender differences in strength and muscle fiber characteristics. European Journal of Applied Physiology and Occupational Physiology 66, 254-262. [DOI] [PubMed] [Google Scholar]
- Perez-Martin A., Dumortier M., Raynaud E., Brun J. F., Fedou C., Bringer J., Mercier J. (2001) Balance of substrate oxidation during submaximal exercise in lean and obese people. Diabetes Metabolism 27, 466-474 [PubMed] [Google Scholar]
- Phillips S. M., Tarnopolsky M. A., Heigenhauser G. J. F., Hill R. E., Grant S. M. (1996) Effect of training duration on substrate turnover and oxidation during exercise. Journal of Applied Physiology 81, 2182-2191. [DOI] [PubMed] [Google Scholar]
- Porcari J. P., Zedam J. M., Naser L., Miller M. (1998) Evaluation of an elliptical exerciser in comparison to treadmill walking and running, stationary cycling and stepping. Medicine and Science in Sport and Exercise 30, 168 [Google Scholar]
- Robergs R. A., Ghiasvand D., Parker D. (2004) Biochemistry of exercise-induced metabolic acidosis. American Journal of Physiology, Regulatory, Integrative Comparative Physiology 287, 502-516. [DOI] [PubMed] [Google Scholar]
- Secher N. H. (1993) Physiological and biomechanical aspects of rowing. Implications for training. Sports Medicine 15, 24-42. [DOI] [PubMed] [Google Scholar]
- Shaharudin S., Agrawal S. (2016) Muscle synergies during incremental rowing VO2max test of collegiate rowers and untrained subjects. Journal of Sports Medicine and Physical Fitness 56, 980-989. [PubMed] [Google Scholar]
- Sidossis L., Gastaldelli A., Klein S., Wolfe R. R. (1997) Regulation of plasma fatty acid oxidation during low- and high-intensity exercise. American Journal of Physiology 272, 1065-1070. [DOI] [PubMed] [Google Scholar]
- Sozen H. (2010) Comparison of muscle activation during elliptical trainer, treadmill and bike exercise. Biology of Sport 27, 203-206. [Google Scholar]
- Starritt E. C., Howlett R. A., Heigenhauser G. J., Spriet L. L. (2000) Sensitivity of CPT I to malonyl-CoA in trained and untrained human skeletal muscle. American Journal of Physiology Endocrinology and Metabolism 278, E462-8. [DOI] [PubMed] [Google Scholar]
- Street D., Bangsbo J., Juel C. (2001) Interstitial pH in human skeletal muscle during and after dynamic graded exercise. Journal of Physiology 537, 993-998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taivassalo T., Abbott A., Wyrick P., Haller R .G. (2002) Venous oxygen levels during aerobic forearm exercise: an index of impaired oxidative metabolism in mitochondrial myopathy. Annals of Neurology 51, 38-44. [DOI] [PubMed] [Google Scholar]
- Tarnopolsky M. A., Atkinson S. A., Phillips S. M., MacDougall J. D. (1995) Carbohydrate loading and metabolism during exercise in men and women. Journal of Applied Physiology 78, 1360-1368. [DOI] [PubMed] [Google Scholar]
- Vallerand A. L., Jacobs I. (1989) Rates of energy substrates utilization during human cold exposure. European Journal of Applied Physiology and Occupational Physiology 58, 873-878. [DOI] [PubMed] [Google Scholar]
- Venables M. C., Achten J., Jeukendrup A. E. (2005) Determinants of fat oxidation during exercise in healthy men and women: A cross-sectional study. Journal of Applied Physiology 98, 160-167. [DOI] [PubMed] [Google Scholar]
- Wells G. D., Noseworthy M. D., Hamilton J., Tarnopolski M., Tein I. (2008) Skeletal muscle metabolic dysfunction in obesity and metabolic syndrome. Canadian Journal of Neurological Sciences 35, 31-40. [DOI] [PubMed] [Google Scholar]
- Weltman A., Wood C. M., Womack C. J., Davis S. E., Blumer J. L., Alvarez J., Sauer K., Gaesser G. A. (1994) Catecholamine and blood lactate responses to incremental rowing and running exercise. Journal of Applied Physiology 76, 1144-1149. [DOI] [PubMed] [Google Scholar]
- Young A. J., Sawka M. N., Epstein Y., DeCristofano B., Pandolf K. B. (1986) Cooling different body surfaces during upper and lower body exercise. Journal of Applied Physiology 63, 1218-1223. [DOI] [PubMed] [Google Scholar]


