Abstract
The phytopathogenic smut fungus Ustilago maydis is a versatile model organism to study plant pathology, fungal genetics, and molecular cell biology. Here, we report several strategies to manipulate the genome of U. maydis by the CRISPR/Cas9 technology. These include targeted gene deletion via homologous recombination of short double-stranded oligonucleotides, introduction of point mutations, heterologous complementation at the genomic locus, and endogenous N-terminal tagging with the fluorescent protein mCherry. All applications are independent of a permanent selectable marker and only require transient expression of the endonuclease Cas9hf and sgRNA. The techniques presented here are likely to accelerate research in the U. maydis community but can also act as a template for genome editing in other important fungi.
Keywords: Ustilago maydis, CRISPR/Cas9, genetic manipulation, deletion, point mutation
1. Introduction
The biological function of the CRISPR (clustered regularly interspaced short palindromic repeats)-Cas (CRISPR-associated protein)-system was identified in archaea in 2007. It helps to acquire immunity against phages [1,2]. RNA guided recognition results in sequence-specific nucleic acid cleavage to introduce double strand breaks (DSBs) in target DNA, can be programmed and can be used outside of prokaryotes [3,4,5,6,7]. The discovery that the CRISPR/Cas system can be used for genome editing at a desired target site was awarded with the Noble prize in 2020 [8]. The CRISPR/Cas9-system and many of its variations have been used in basic research to manipulate genomes of diverse organisms [9,10,11].
The basidiomycetous fungus Ustilago maydis infects maize plants and has been established as a genetic model organism for studying plant infection, DNA repair, intracellular long distance transport of proteins and mRNA, secondary metabolism, and translational recoding [12,13,14,15,16,17,18,19]. Haploid U. maydis cells divide by budding and are highly amenable to genetic analysis. Genetic manipulation can be achieved by transformation of protoplasts using gene cassettes with antibiotic resistance markers [20,21]. U. maydis has a very efficient homologous recombination machinery which allows for high levels of gene replacement. This strategy is so far limited to a maximum of five independent modifications per cell by the number of available resistance markers (hygromycin, carboxin, phleomycin, gentamycin, and nourseothricin) [20,22]. In 2016, CRISPR-Cas9 editing was described for U. maydis to generate mutants by non-homologous end joining (NHEJ) [23]. This system was further modified for multiplex gene disruption, generation of complete deletion mutants by using 2000 bp long donor DNA for gene replacement, and by introduction of high fidelity Cas9hf enzyme to reduce potential off-target effects [24,25,26].
Here, we used the power of the CRISPR/Cas9 technology for the creation of different types of mutants in U. maydis. This included the generation of marker-free deletion strains but also the introduction of point mutations by site directed mutagenesis. Furthermore, CRISPR/Cas9 was used to replace a gene by its homologous gene from the related fungus Ustilago hordei and to generate an endogenous N-terminal fusion with the fluorescent mCherry marker.
2. Materials and Methods
2.1. Strains and Growth Conditions
Fungal strains were grown in liquid YEPSL (1% yeast extract (Roth, Karlsruhe, Germany), 0.4% peptone (Neogen, Lansing, MI, USA), 0.4% sucrose (Roth, Karlsruhe, Germany)) or on solid potato dextrose broth (Difco, Detroit, MI, USA) containing 1.5% Bacto agar (Roth, Karlsruhe, Germany) at 28 °C and 23 °C, respectively. The U. maydis strains used and generated in this study are listed in Supplementary Table S1 and are derivatives of the U. maydis strains Bub8 [27] and MB215 [28]. U. hordei (Uh4857-4) was a kind gift from Regine Kahmann (MPI Marburg, Germany). To induce glycolipid production U. maydis strains were grown to stationary phase in YEPS-L and then transferred to nitrogen starvation medium (OD = 0.1) containing 0.17% YNB (yeast nitrogen base without ammonia, Difco, Detroit, MI, USA) and 1% glucose (Roth, Karlsruhe, Germany) as carbon source. Glycolipid production was then allowed for 6 days at 23 °C under rotation. Escherichia coli strain Top10 (Invitrogen, Carlsbad, CA, USA) was used for transformation according to Hanahan et al., and amplification of plasmid DNA [29].
2.2. Molecular Cloning and Nucleic Acid Procedures
Standard procedures were followed for generation of pSM2 and its derivatives [30,31]. Plasmids are based on the plasmid pCas9_sgRNA_0 [23]. PAM motifs were identified using www.e-crisp.org [32]. Genomic DNAs of U. maydis and U. hordei cells were prepared according to an established protocol [33]. Donor DNAs were amplified by two rounds of PCRs as illustrated in Supplementary Figures S1–S3. Primer sequences are listed in supplementary Table S2 and plasmids are listed in Supplementary Table S3. Plasmids were verified by sequencing.
2.3. Strain Construction
Transformation of U. maydis was conducted as described [34]. Briefly, 100 ng of autonomously replicating pSM2 plasmids were co-transformed with either 0.2–0.6 pmol of PCR products or 65 pmol of hybridized oligonucleotides of donor DNA. Selection was performed on carboxin (cbx, Sigma Aldrich, Buchs, Switzerland) containing plates. Transformants were verified by PCR and sequencing. Positive clones were then streaked out on cbx-free plates unless the plasmid had been lost [23].
2.4. Analysis of Glycolipids
Extracellular glycolipids were extracted as previously described [28]. Glycolipids (MELs and CLs) were separated by thin-layer chromatography (TLC) on silica gel 60 F254 plates (Merck, Darmstadt, Germany) first with a solvent system consisting of chloroform–methanol–water (65:25:4, v/v/v) (Roth, Karlsruhe, Germany) for 5 min followed by a second solvent system consisting of chloroform–methanol (9:1, v/v; 2 × 18 min) [35]. The plates were dried, and sugar containing compounds were visualized by application of a mixture of ethanol:sulfuric acid:p-anisaldehyde (18:1:1, v/v) (Roth, Karlsruhe, Germany; Merck, Darmstadt, Germany; Sigma Aldrich, Steinheim, Germany) followed by heating at 150 °C for 2 min [36].
2.5. Microscopy
U. maydis cells from logarithmically growing cultures were placed on agarose cushions. Cells were visualized by differential interference contrast (DIC), phase contrast (PC) and epifluorescence microscopy using a Zeiss Axiophot 200 microscope. Images were taken using a cooled Hamamatsu Orca-ER CCD camera with an exposure time of 50–300 ms. Image acquisition and deconvolution were performed using Improvision Volocity software and processing was done with ImageJ [37].
2.6. Kinase Inhibition
The activity of the engineered kinase Cdk5F78G was inhibited by addition of the indicated amounts of NA-PP1 (Calbiochem, San Diego, CA, USA). Inhibition of Cdk5F78G in logarithmically growing cells blocked cell growth immediately. The essential function of Cdk5 for normal cell growth was measured in growth assays using a micro plate reader (Synergy Mix; BioTek). 100 µl of fungal cells of OD = 0.1 were grown in YEPSL medium in the presence of NA-PP1. Growth was recorded every 30 min for 12 h in three independent experiments. Data were analyzed with Microsoft Excel software.
3. Results
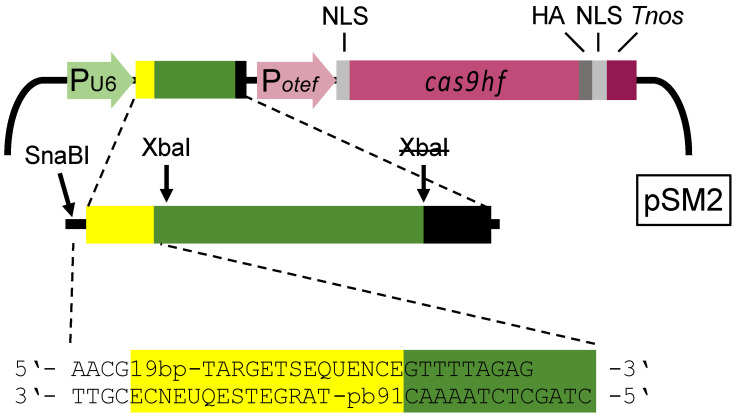
The CRISPR/Cas system previously used in U. maydis is based on plasmid pCas9_sgRNA_0, which contains the gene for the Cas9 endonuclease and can be equipped with sequences encoding single guide RNA (sgRNA) for specific gene disruption in U. maydis [23]. Each gene disruption requires synthesis of a specific 175 bp DNA fragment for a Gibson cloning strategy [23,30]. We first modified the vector to enable direct cloning of short double stranded DNA (dsDNA) fragments containing the 19 bp target sequence of the sgRNA and some flanking sequences into pSM1 linearized with SnaBI/XbaI (Figure 1). Note that both restriction sites become inactive in the final construct. To generate plasmid pSM2, we introduced three point mutations (R661A, Q695A, and Q926A) into Cas9 to gain the high fidelity variant of Cas9, which reduces off-target cleavage [23,38].
Figure 1.
Schematic drawing of the Cas9 expression cassette and the RNA polymerase III U6 promoter (light green; PU6) driven sgDNA (pSM2) according to Schuster et al., 2016 [23]. Yellow: 19 bp target sequence; dark green: scaffold sequence; black: U6 terminator; light purple: constitutive otef-promoter (Potef); light gray: nuclear localization signal (NLS); purple: open reading frame (ORF) of high fidelity cas9; dark gray: HA-tag; dark purple: nos terminator. The former XbaI-site inside of the U6 terminator was inactivated by a point mutation. The 32/36 bp flanking sequences for the oligonucleotides are given.
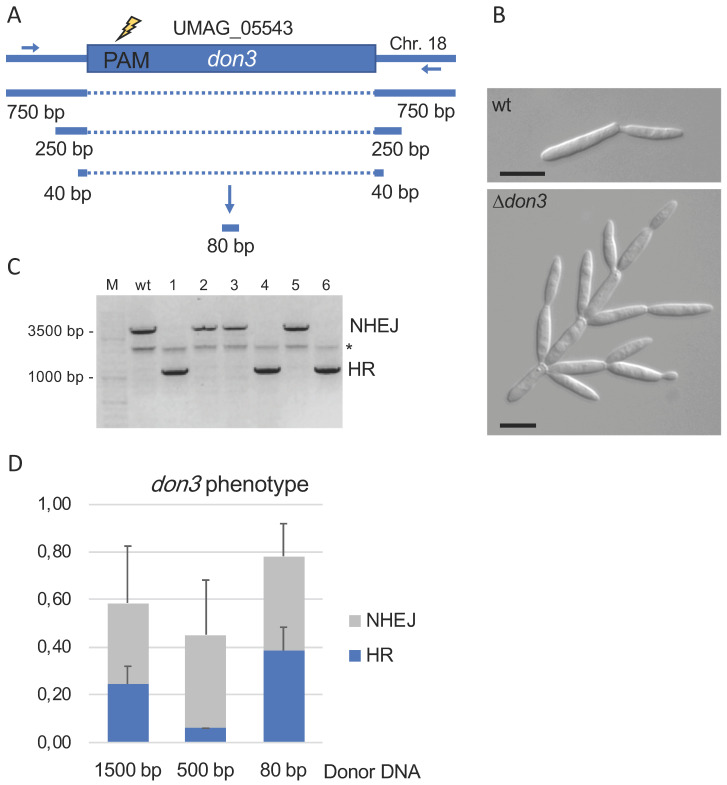
To allow the efficient and defined deletion of genes we next determined the minimal length of donor dsDNA required for efficient homologous recombination. As a target gene we used don3 encoding a germinal center kinase required for cell separation [39,40] because of the remarkable tree-like deletion phenotype (Figure 2A,B). To study the length of the donor dsDNA required for efficient repair of double strand breaks and introduced via Cas9, we used flanking regions of 750 bp, 250 bp, and 40 bp, respectively (Figure 2A). dsDNA was generated by two step PCR (1500 bp and 500 bp fragments containing both the left and right borders) or by hybridization of oligonucleotides (80 bp fragment) (Figure 2A). Transformants were screened for the don3-phenotype and further characterized by PCR (Figure 2B,C). Generation of deletion mutants was confirmed irrespective of the donor DNA applied. The use of high concentrated oligonucleotides providing only short homology was most efficient; approx. 50% of all mutants exhibiting the don3 phenotype were deletions (Figure 2D). This strategy was also used successfully for additional genes like rua1, emt1, and mat1 [Fabienne Becker and Björn Sandrock; unpublished data].
Figure 2.
Generation of don3 deletion mutants via donor DNA of different length. (A): Schematic drawing of the don3 gene indicating the protospacer adjacent motif (PAM) in the 5′-region of the ORF, which was used. Flanking sequences of 1500 bp and 500 bp, respectively, were amplified by a two-step PCR approach. The 80 bp dsDNA was generated by hybridization of oligonucleotides. (B): Microscopic image (DIC) of the don3 phenotype used for identification of deletion mutants (Scale bar 10 µm). (C): PCR test of six don3 mutants either resulting from non-homologous end joining (NHEJ) or homologous recombination using donor DNA (HR). Wildtype Bub8 (wt) was used as a control. * unspecific PCR product. (D): Summery of three independent transformation experiments using 0.2–0.6 pmol for the PCR based donor DNAs 1500 bp and 500 bp and using 65–70 pmol hybridized oligonucleotides (80 bp).
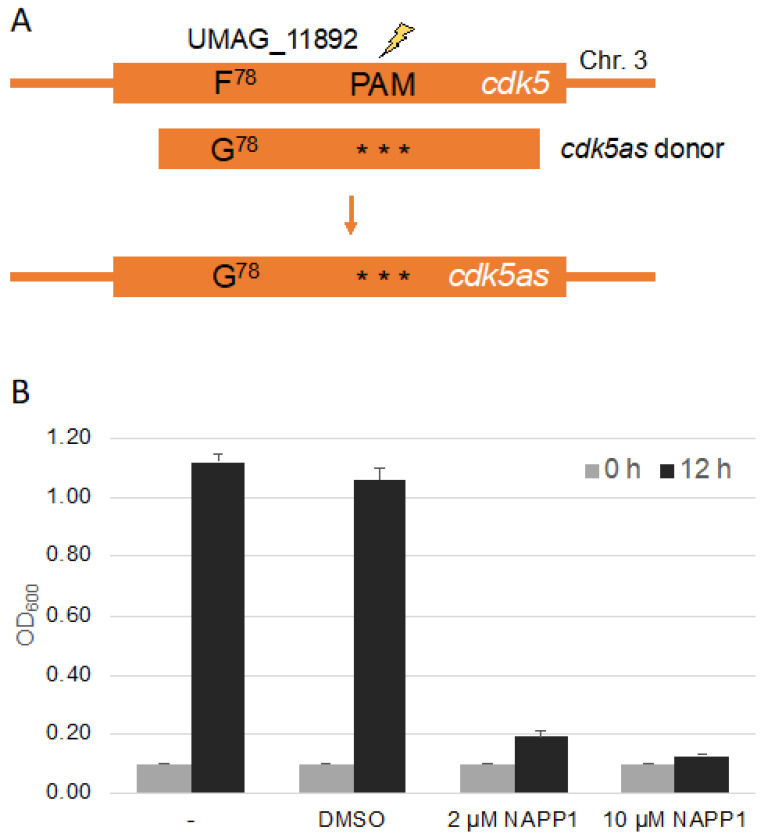
Chemical genetics is a powerful tool to study protein kinases in vivo, and is regularly used by our lab to characterize protein kinases during cytokinesis as well as for systematic mining for the function of protein kinases not yet characterized in U. maydis, together with undergraduate researchers [39,40,41,42,43,44]. Specific mutations in the binding domain allows for inhibition of the kinase by an external chemical. We decided to use CRISPR/Cas9 to test for the introduction of point mutations at the genomic locus, which render a kinase repressible by the ATP analog NA-PP1. As an example we decided to mutate the gene umag_11892 encoding UmCdk5, a homolog of the cyclin dependent kinase Pho85 from Saccharomyces cerevisiae [45,46]. The gatekeeper in the kinase domain of UmCdk5 is located at F78 and the mutation for the chemical genetics analysis is F78G (Figure 3A). CRISPR/Cas9 dependent cleavage requires a protospacer adjacent motif(PAM) in vicinity of the cleavage site [32]. This can hamper the introduction of point mutations in regions devoid of a PAM. We searched for a PAM at a distance of around 500 bp of the codon encoding the gatekeeper amino acid [41,47]. To create donor DNA for recombination, we set up a two step-PCR with three overlapping fragments (Figure 3A and Figure S1A). To avoid donor DNA disruption by Cas9 we inserted three silent mutations within the sequence corresponding to the sgRNA. For easy detection of positive transformants, a silent mutation resulting in the restriction site HindIII in the gatekeeper primers was introduced. The donor DNA was transformed together with the corresponding pSM2-Cdk5 plasmid and transformants were identified by PCR, restriction digest and sequencing (Figure S1B). We succeeded in generating cdk5 point mutations (3 of 18 transformants) and found that the kinase, which is essential in U. maydis [46], can be inhibited by NA-PP1 leading to an arrest of cell growth (Figure 3B).
Figure 3.
Site directed mutagenesis to generate an analog sensitive Cdk5 kinase. (A): Schematic drawing of the cdk5 gene highlighting the position of the region encoding the gatekeeper and the PAM site. The donor DNA encoded the F78G mutation and contained three silent mutations within the sequence corresponding to the sgRNA. Further details are depicted in Figure S1 and Table S3. (B): Quantification of growth assays of Bub8 cdk5as in the absence or presence of NA-PP1 inhibitor starting with an OD600 of 0.1. DMSO is the solvent of NA-PP1 and served as control. Three independent experiments were analyzed.
These data demonstrate that in U. maydis CRISPR/Cas9 can be used to generate marker free strains with desired point mutations. In collaboration with undergraduate researchers we have already generated and tested mutated variants of the following proteins: Elm1as (L437G, UMAG_15093, 1 of 12), Mkk1as (M295G, UMAG_10855, 2 of 12), and Yak1as (L684G, UMAG_06306, 3 of 12). Strains are available upon request.
In recent years, we have characterized a gene cluster for biosynthesis of mannosylerythritol lipids (MELs) in U. maydis and related fungi [48,49,50]. Five similar genes are responsible for the production of these amphiphilic compounds, which differ in their acetylation and acylation patterns [51,52]. We have shown, that expression of the acetyl-transferase Mat1 from Ustilago hordei in U. maydis ∆mat1 cells resulted in the exclusive production of mono-acetylated MEL-C molecules [50]. Therefore, we concluded that cross species complementation might be a valuable approach to customize MEL biosynthesis. For this approach marker free gene replacement would be of particular interest.
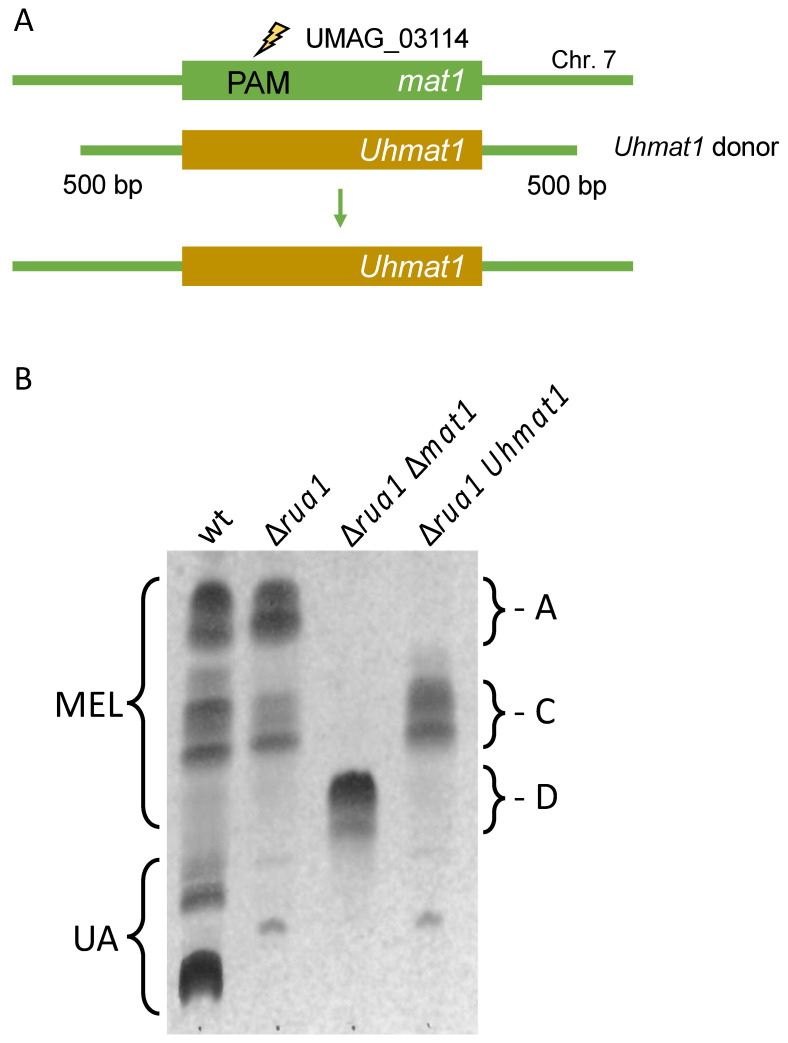
Thus, we constructed donor DNA consisting of the open reading frame (ORF) U. hordei mat1 and 500 bp fragments from the U. maydis mat1 gene to trigger homologous recombination (Figure 4A and Figure S2A). Donor DNA (0.3 pmol) was co-transformed with pSM2-Mat1 in strain MB215 ∆rua1, which cannot produce the related glycolipid ustilagic acid [53]. Candidates were tested for MEL biosynthesis by growth in minimal medium without a nitrogen source. The majority of transformants produced MEL-D, the non-acetylated variant indicating disruption of the mat1 gene. Around 5% showed the desired MEL-C production (Figure 4B). The successful exchange of the mat1 gene was verified by PCR and sequencing (Figure S2B). This strategy may also be performed for mat1 gene from Pseudozyma tsukubaensis to exclusively produce MEL-B variants [54]. These findings showcase the potential of CRISPR/Cas9 for heterologous complementation in U. maydis in general and its potential to aid with customization of metabolic pathways.
Figure 4.
Heterologous complementation at the genomic locus. (A): Schematic drawing of the U. maydis (Um) mat1 gene indicating the position of the PAM site. Donor DNA contained the ORF for Mat1 from U. hordei (Uh) and 500 bp upstream and downstream flanking sequence of the U. maydis ORF. Further details are depicted in Figure S2 and Table S3. (B): Thin layer chromatography of glycolipids from MB215 (wt), ∆rua1, ∆rua1 ∆mat1 and ∆rua1 Uhmat1. MEL variants (A,C,D) can be separated according to their acetylation patterns.
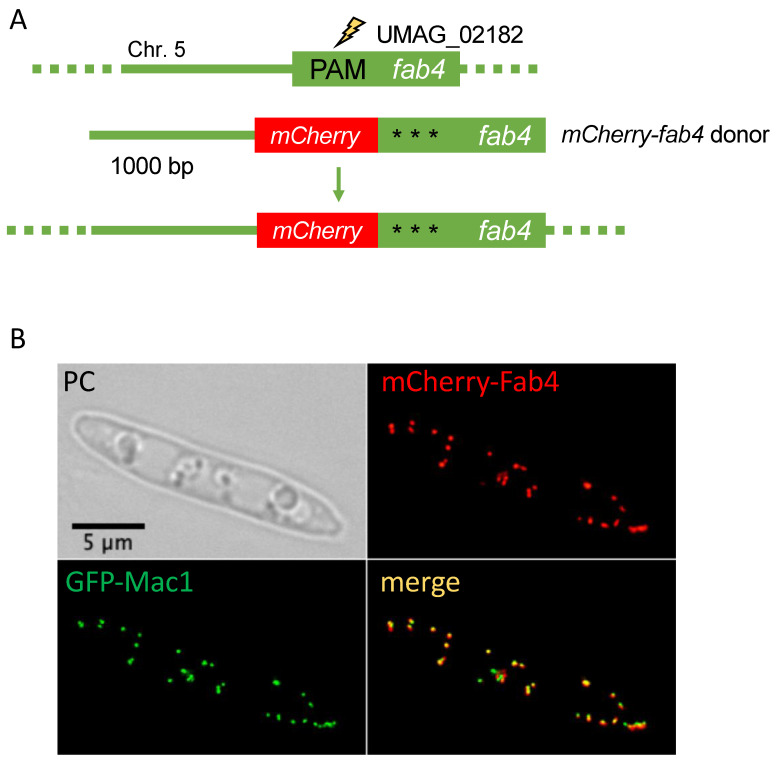
Intracellular localization of proteins is often determined by signal sequences residing at the very N-terminus or very C-terminus of proteins [55]. While production of C-terminally tagged versions of proteins expressed from their endogenous loci is a standard procedure in U. maydis, N-terminal tagging usually also involves the substitution of the endogenous promoter [20,56]. Targeting of soluble proteins to cellular organelles called peroxisomes regularly depends on C-terminal extensions termed peroxisomal targeting signal type I (PTS1) [57]. Hence, PTS1 proteins might be suitable candidates to demonstrate N-terminal tagging with a fluorescent protein tag with the help of CRISPR/Cas9. Fab4 (UMAG_02182) is predicted to localize in peroxisomes and is expressed throughout the cell and the life cycle of U. maydis [58]. Fab4 is a putative NADPH-dependent beta-ketoacyl carrier protein reductase. It was shown that FabG, an ortholog of Fab4, performs the NADPH-dependent reduction of beta-ketoacyl-ACP substrates to beta-hydroxyacyl-ACP products, the first reductive step in the elongation cycle of fatty acid biosynthesis [59]. We engineered a donor DNA composed of four fragments fused by overlapping PCR (Figure 5A and Figure S3A). Donor DNA included 1000 bp flanking sequence and the mCherry sequence upstream of the Fab4 ORF to express mCherry-Fab4. For the construction of the pSM2-Fab4 we used a PAM close to the 5′end of the ORF. Again, three silent mutations were introduced to avoid disruption of the donor DNA by Cas9 cleavage.
Figure 5.
N-terminal tagging at the endogenous locus (A): Schematic drawing of the fab4 gene indicating the position of the PAM site. Donor DNA consisted of 1000 bp upstream and downstream flanking sequence, the ORF for mCherry. Three silent mutations were introduced in the sequences corresponding to the sgRNA. For further details see Figure S3 and Table S3. (B): Phase contrast (PC) and fluorescent imaging of mCherry-fab4 expressing cells. mCherry-Fab4 co-localized with GFP-Mac1 in peroxisomes [17,49]. Scale bar: 5 µm.
One positive mCherry-Fab4 expressing strain with clear peroxisomal localization was recovered from 36 candidates indicating that N-terminal tagging is possible albeit with low efficiency (Figure 5B and Figure S3B). This demonstrates that in U. maydis N-terminal tagging under endogenous expression conditions is now possible with only one round of transformation.
4. Discussion
Here, we have shown different applications of the CRISPR/Cas9 system, which are likely to enhance the molecular characterization of U. maydis. A major limitation of the U. maydis system compared to S. cerevisiae was the requirement for very long homologous sequences to achieve deletion of genes [20,60]. This can now be compensated by the combination of CRISPR/Cas9 and highly concentrated dsDNA oligonucleotides, which can be easily generated from commercially available stocks.
Methods described here may not only be a benefit for research in U. maydis but may be also applicable in related fungi such as U. hordei, Sporisorium reilianum, and Ustilago bromivora [61,62,63]. It is possible that CRISPR may also allow for the genetic manipulation of pathogenic rust fungi, which have an enormous impact on agriculture [64].
A major limitation of the methods presented here is the strict requirement of suitable PAM sites in the vicinity to the sites of the genome for a desired genetic manipulation. While this is usually not problematic for creation of deletion mutants, other interventions such as N-terminal tagging and the creation of point mutations will strongly profit from the engineered Cas9 enzymes or alternative endonucleases [65].
Overall our study showcases different applications of CRISPR/Cas9 base genome editing, which might ultimately override many of the disadvantages researchers had, if they decided to tackle the biology of organisms outside of the traditional model systems. As multiplexing is possible with CRISPR/Cas9 systems [66,67] questions in such systems can now even be addressed systematically.
Acknowledgments
We thank Marisa Piscator for excellent technical assistance. We express thanks to the MSc lab courses “Molecular genetics” in the WS2017/18, 2018/19 and 2019/20 at the Philipps-University Marburg (Germany). We are grateful to Regine Kahmann for support and resources.
Supplementary Materials
The following are available online at https://www.mdpi.com/2309-608X/7/2/149/s1, Figure S1: A: Two-step PCR strategy to generate donor DNA for cdk5as using indicated primers. B: Sequencing result of the genomic DNA from positive transformants. GK: DNA region of the gatekeeper sequence with the HindIII silent mutation in red and the gatekeeper codon mutation in green. PAM: region including the PAM site, three silent mutations in the sgRNA region in red. Figure S2: A: Two-step PCR strategy to generate donor DNA for Uhmat1 using indicated primers. B: Sequencing result of genomic DNA from a positive transformant. DNA sequence around the start codon showing the U. maydis sequence in green and the U. hordei sequence in brown. Figure S3: A: Two-step PCR strategy to generate donor DNA for mCherry-fab4 using indicated primers. B: PCR results of genomic DNA from wt and the positive transformant containing mCherry (mC) using different primer combinations (Table S2). Table S1: Strains used in the study. Table S2: Oligonucleotides used in this study. Table S3: Plasmids used in this study.
Author Contributions
Conceptualization, B.S., J.F, M.B.; Methodology, S.-M.W., K.G., F.B., B.S.; Validation, S.-M.W., K.G., F.B., B.S.; Formal Analysis, S.-M.W., K.G., F.B., M.B., J.F., B.S.; Investigation, B.S.; Resources, M.B.; Writing, Review and Editing, B.S., J.F., M.B. Visualization, B.S.; Supervision, B.S., J.F., M.B.; Project Administration, B.S.; Funding Acquisition, M.B. All authors have read and agreed to the published version of the manuscript.
Funding
We acknowledge funding by the German Research Foundation (Sonderforschungsbereich SFB987 and grant nr. BO2094-5).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Accession numbers from National Center for Biotechnology Information (NCBI; www.ncbi.nlm.nih.gov): U. maydis: Don3 (UMAG_05543; XP_011391855), Pho85 (UMAG_11892; XP_011388110), Mat1 (UMAG_03114; XP_011389465), Fab4 (UMAG_02182; XP_011388532). U. hordei: Mat1 (UHOR_04871; CCF52714).
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Barrangou R., Fremaux C., Deveau H., Richards M., Boyaval P., Moineau S., Romero D.A., Horvath P. CRISPR Provides Acquired Resistance Against Viruses in Prokaryotes. Science. 2007;315:1709–1712. doi: 10.1126/science.1138140. [DOI] [PubMed] [Google Scholar]
- 2.Horvath P., Barrangou R. CRISPR/Cas, the immune system of bacteria and archaea. Science. 2010;327:167–170. doi: 10.1126/science.1179555. [DOI] [PubMed] [Google Scholar]
- 3.Haurwitz R.E., Jinek M., Wiedenheft B., Zhou K., Doudna J.A. Sequence-and structure-specific RNA processing by a CRISPR endonuclease. Science. 2010;329:1355–1358. doi: 10.1126/science.1192272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Garneau J.E., Dupuis M.-È., Villion M., Romero D.A., Barrangou R., Boyaval P., Fremaux C., Horvath P., Magadán A.H., Moineau S. The CRISPR/Cas bacterial immune system cleaves bacteriophage and plasmid DNA. Nature. 2010;468:67–71. doi: 10.1038/nature09523. [DOI] [PubMed] [Google Scholar]
- 5.Jinek M., Chylinski K., Fonfara I., Hauer M., Doudna J.A., Charpentier E. A programmable dual-RNA--guided DNA endonuclease in adaptive bacterial immunity. Science. 2012;337:816–821. doi: 10.1126/science.1225829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ran F.A., Hsu P.D., Wright J., Agarwala V., Scott D.A., Zhang F. Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 2013;8:2281–2308. doi: 10.1038/nprot.2013.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Doudna J.A., Charpentier E. The new frontier of genome engineering with CRISPR-Cas9. Science. 2014;346:1258096. doi: 10.1126/science.1258096. [DOI] [PubMed] [Google Scholar]
- 8.Cohen J. A cut above: Pair that developed CRISPR earns historic award. Science. 2020;370:271–272. doi: 10.1126/science.370.6514.271. [DOI] [PubMed] [Google Scholar]
- 9.Zhu H., Li C., Gao C. Applications of CRISPR--Cas in agriculture and plant biotechnology. Nat. Rev. Mol. Cell Biol. 2020;21:661–677. doi: 10.1038/s41580-020-00288-9. [DOI] [PubMed] [Google Scholar]
- 10.Wright A.V., Nuñez J.K., Doudna J.A. Biology and applications of CRISPR systems: Harnessing nature’s toolbox for genome engineering. Cell. 2016;164:29–44. doi: 10.1016/j.cell.2015.12.035. [DOI] [PubMed] [Google Scholar]
- 11.Barrangou R., Doudna J.A. Applications of CRISPR technologies in research and beyond. Nat. Biotechnol. 2016;34:933–941. doi: 10.1038/nbt.3659. [DOI] [PubMed] [Google Scholar]
- 12.Kämper J., Kahmann R., Bölker M., Ma L.J., Brefort T., Saville B.J., Banuett F., Kronstad J.W., Gold S.E., Müller O., et al. Insights from the genome of the biotrophic fungal plant pathogen Ustilago maydis. Nature. 2006;444:97–101. doi: 10.1038/nature05248. [DOI] [PubMed] [Google Scholar]
- 13.Lanver D., Tollot M., Schweizer G., Presti L.L., Reissmann S., Ma L.-S., Schuster M., Tanaka S., Liang L., Ludwig N., et al. Ustilago maydis effectors and their impact on virulence. Nat. Rev. Microbiol. 2017;15:409. doi: 10.1038/nrmicro.2017.33. [DOI] [PubMed] [Google Scholar]
- 14.Holloman W.K. Unraveling the mechanism of BRCA2 in homologous recombination. Nat. Struct. Mol. Biol. 2011;18:748–754. doi: 10.1038/nsmb.2096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.König J., Baumann S., Koepke J., Pohlmann T., Zarnack K., Feldbrügge M. The fungal RNA-binding protein Rrm4 mediates long-distance transport of ubi1 and rho3 mRNAs. EMBO J. 2009;28:1855–1866. doi: 10.1038/emboj.2009.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Feldbrügge M., Kellner R., Schipper K. The biotechnological use and potential of plant pathogenic smut fungi. Appl. Microbiol. Biotechnol. 2013;97:3253–3265. doi: 10.1007/s00253-013-4777-1. [DOI] [PubMed] [Google Scholar]
- 17.Freitag J., Ast J., Bölker M. Cryptic peroxisomal targeting via alternative splicing and stop codon read-through in fungi. Nature. 2012;485:522–525. doi: 10.1038/nature11051. [DOI] [PubMed] [Google Scholar]
- 18.Stiebler A.C., Freitag J., Schink K.O., Stehlik T., Tillmann B.A.M., Ast J., Bölker M. Ribosomal Readthrough at a Short UGA Stop Codon Context Triggers Dual Localization of Metabolic Enzymes in Fungi and Animals. PLoS Genet. 2014;10:1–9. doi: 10.1371/journal.pgen.1004685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pohlmann T., Baumann S., Haag C., Albrecht M., Feldbrügge M. A FYVE zinc finger domain protein specifically links mRNA transport to endosome trafficking. Elife. 2015;4:e06041. doi: 10.7554/eLife.06041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Brachmann A., König J., Julius C., Feldbrügge M. A reverse genetic approach for generating gene replacement mutants in Ustilago maydis. Mol. Genet. Genom. 2004;272:216–226. doi: 10.1007/s00438-004-1047-z. [DOI] [PubMed] [Google Scholar]
- 21.Kämper J. A PCR-based system for highly efficient generation of gene replacement mutants in Ustilago maydis. Mol. Genet. Genom. 2004;271:103–110. doi: 10.1007/s00438-003-0962-8. [DOI] [PubMed] [Google Scholar]
- 22.Kojic M., Holloman W.K. Shuttle vectors for genetic manipulations in Ustilago maydis. Can. J. Microbiol. 2000;46:333–338. doi: 10.1139/w00-002. [DOI] [PubMed] [Google Scholar]
- 23.Schuster M., Schweizer G., Reissmann S., Kahmann R. Genome editing in Ustilago maydis using the CRISPR--Cas system. Fungal Genet. Biol. 2016;89:3–9. doi: 10.1016/j.fgb.2015.09.001. [DOI] [PubMed] [Google Scholar]
- 24.Schuster M., Schweizer G., Kahmann R. Comparative analyses of secreted proteins in plant pathogenic smut fungi and related basidiomycetes. Fungal Genet. Biol. 2018;112:21–30. doi: 10.1016/j.fgb.2016.12.003. [DOI] [PubMed] [Google Scholar]
- 25.Becker J., Hosseinpour Tehrani H., Gauert M., Mampel J., Blank L.M., Wierckx N. An Ustilago maydis chassis for itaconic acid production without by-products. Microb. Biotechnol. 2020;13:350–362. doi: 10.1111/1751-7915.13525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zuo W., Depotter J.R.L., Doehlemann G. Cas9HF1 enhanced specificity in Ustilago maydis. Fungal Biol. 2020;124:228–234. doi: 10.1016/j.funbio.2020.02.006. [DOI] [PubMed] [Google Scholar]
- 27.Weinzierl G., Leveleki L., Hassel A., Kost G., Wanner G., Bölker M. Regulation of cell separation in the dimorphic fungus Ustilago maydis. Mol. Microbiol. 2002;45:219–231. doi: 10.1046/j.1365-2958.2002.03010.x. [DOI] [PubMed] [Google Scholar]
- 28.Hewald S., Josephs K., Bölker M. Genetic analysis of biosurfactant production in Ustilago maydis. Appl. Environ. Microbiol. 2005;71:3033–3040. doi: 10.1128/AEM.71.6.3033-3040.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hanahan D., Jessee J., Bloom F.R. Plasmid transformation of Escherichia coli and other bacteria. Methods Enzymol. 1991;204:63–113. doi: 10.1016/0076-6879(91)04006-a. [DOI] [PubMed] [Google Scholar]
- 30.Gibson D.G., Young L., Chuang R.-Y., Venter J.C., Hutchison C.A., III, Smith H.O. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods. 2009;6:343. doi: 10.1038/nmeth.1318. [DOI] [PubMed] [Google Scholar]
- 31.Sambrook J., Fritsch E.F., Maniatis T. In: Molecular Cloning: A Laboratory Manual. Argentine J., editor. Vol. 3. Cold Spring Harbor Laboratory Press; Cold Spring Harbor, NY, USA: 1989. Molecular Cloning a Laboratory Manual. [Google Scholar]
- 32.Semenova E., Jore M.M., Datsenko K.A., Semenova A., Westra E.R., Wanner B., Van Der Oost J., Brouns S.J.J., Severinov K. Interference by clustered regularly interspaced short palindromic repeat (CRISPR) RNA is governed by a seed sequence. Proc. Natl. Acad. Sci. USA. 2011;108:10098–10103. doi: 10.1073/pnas.1104144108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hoffman C.S., Winston F. A ten-minute DNA preparation from yeast efficiently releases autonomous plasmids for transformaion of Escherichia coli. Gene. 1987;57:267–272. doi: 10.1016/0378-1119(87)90131-4. [DOI] [PubMed] [Google Scholar]
- 34.Schulz B., Banuett F., Dahl M., Schlesinger R., Schäfer W., Martin T., Herskowitz I., Kahmann R. The b alleles of U. maydis, whose combinations program pathogenic development, code for polypeptides containing a homeodomain-related motif. Cell. 1990;60:295–306. doi: 10.1016/0092-8674(90)90744-Y. [DOI] [PubMed] [Google Scholar]
- 35.Kurz M., Eder C., Isert D., Li Z., Paulus E.F., Schiell M., Toti L., Vertesy L., Wink J., Seibert G. Ustilipids, acylated beta-D-mannopyranosyl D-erythritols from Ustilago maydis and Geotrichum candidum. J. Antibiot. 2003;56:91–101. doi: 10.7164/antibiotics.56.91. [DOI] [PubMed] [Google Scholar]
- 36.Frautz B., Lang S., Wagner F. Formation of cellobiose lipids by growing and resting cells of Ustilago maydis. Biotechnol. Lett. 1986;8:757–762. doi: 10.1007/BF01020817. [DOI] [Google Scholar]
- 37.Schneider C.A., Rasband W.S., Eliceiri K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods. 2012;9:671–675. doi: 10.1038/nmeth.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kleinstiver B.P., Pattanayak V., Prew M.S., Tsai S.Q., Nguyen N.T., Zheng Z., Joung J.K. High-fidelity CRISPR--Cas9 nucleases with no detectable genome-wide off-target effects. Nature. 2016;529:490–495. doi: 10.1038/nature16526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sandrock B., Böhmer C., Bölker M. Dual function of the germinal centre kinase Don3 during mitosis and cytokinesis in Ustilago maydis. Mol. Microbiol. 2006;62:655–666. doi: 10.1111/j.1365-2958.2006.05405.x. [DOI] [PubMed] [Google Scholar]
- 40.Böhmer C., Ripp C., Bölker M. The germinal centre kinase Don3 triggers the dynamic rearrangement of higher-order septin structures during cytokinesis in Ustilago maydis. Mol. Microbiol. 2009;74:1484–1496. doi: 10.1111/j.1365-2958.2009.06948.x. [DOI] [PubMed] [Google Scholar]
- 41.Bishop A.C., Shah K., Liu Y., Witucki L., Kung C., Shokat K.M. Design of allele-specific inhibitors to probe protein kinase signaling. Curr. Biol. 1998;8:257–266. doi: 10.1016/S0960-9822(98)70198-8. [DOI] [PubMed] [Google Scholar]
- 42.Böhmer C., Böhmer M., Bölker M., Sandrock B. Cdc42 and the Ste20-like kinase Don3 act independently in triggering cytokinesis in Ustilago maydis. J. Cell Sci. 2008;121:143–148. doi: 10.1242/jcs.014449. [DOI] [PubMed] [Google Scholar]
- 43.Freitag J., Lanver D., Böhmer C., Schink K.O., Bölker M., Sandrock B. Septation of infectious hyphae is critical for appressoria formation and virulence in the smut fungus Ustilago maydis. PLoS Pathog. 2011;7:e1002044. doi: 10.1371/journal.ppat.1002044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sandrock B. Chemical Genetics—A Versatile Method to Combine Science and Higher Level Teaching in Molecular Genetics. Molecules. 2012;17:11920–11930. doi: 10.3390/molecules171011920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Carroll A.S., O’Shea E.K. Pho85 and signaling environmental conditions. Trends Biochem. Sci. 2002;27:87–93. doi: 10.1016/S0968-0004(01)02040-0. [DOI] [PubMed] [Google Scholar]
- 46.Castillo-Lluva S., Alvarez-Tabarés I., Weber I., Steinberg G., Pérez-Martin J. Sustained cell polarity and virulence in the phytopathogenic fungus Ustilago maydis depends on an essential cyclin-dependent kinase from the Cdk5/Pho85 family. J. Cell Sci. 2007;120:1584–1595. doi: 10.1242/jcs.005314. [DOI] [PubMed] [Google Scholar]
- 47.Heigwer F., Kerr G., Boutros M. E-CRISP: Fast CRISPR target site identification. Nat. Methods. 2014;11:122–123. doi: 10.1038/nmeth.2812. [DOI] [PubMed] [Google Scholar]
- 48.Hewald S., Linne U., Scherer M., Marahiel M.A., Kämper J., Bölker M. Identification of a gene cluster for biosynthesis of mannosylerythritol lipids in the basidiomycetous fungus Ustilago maydis. Appl. Environ. Microbiol. 2006;72:5469–5477. doi: 10.1128/AEM.00506-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Freitag J., Ast J., Linne U., Stehlik T., Martorana D., Bölker M., Sandrock B. Peroxisomes contribute to biosynthesis of extracellular glycolipids in fungi. Mol. Microbiol. 2014;93:24–36. doi: 10.1111/mmi.12642. [DOI] [PubMed] [Google Scholar]
- 50.Deinzer H.-T., Linne U., Xie X., Bölker M., Sandrock B. Elucidation of substrate specificities of decorating enzymes involved in mannosylerythritol lipid production by cross-species complementation. Fungal Genet. Biol. 2019;130:91–97. doi: 10.1016/j.fgb.2019.05.003. [DOI] [PubMed] [Google Scholar]
- 51.Bölker M., Basse C.W., Schirawski J. Ustilago maydis secondary metabolism—From genomics to biochemistry. Fungal Genet. Biol. 2008;45:S88–S93. doi: 10.1016/j.fgb.2008.05.007. [DOI] [PubMed] [Google Scholar]
- 52.Saika A., Koike H., Fukuoka T., Morita T. Tailor-made mannosylerythritol lipids: Current state and perspectives. Appl. Microbiol. Biotechnol. 2018;102:6877–6884. doi: 10.1007/s00253-018-9160-9. [DOI] [PubMed] [Google Scholar]
- 53.Teichmann B., Linne U., Hewald S., Marahiel M.A., Bölker M. A biosynthetic gene cluster for a secreted cellobiose lipid with antifungal activity from Ustilago maydis. Mol. Microbiol. 2007;66:525–533. doi: 10.1111/j.1365-2958.2007.05941.x. [DOI] [PubMed] [Google Scholar]
- 54.Fukuoka T., Morita T., Konishi M., Imura T., Kitamoto D. A basidiomycetous yeast, Pseudozyma tsukubaensis, efficiently produces a novel glycolipid biosurfactant. The identification of a new diastereomer of mannosylerythritol lipid-B. Carbohydr. Res. 2008;343:555–560. doi: 10.1016/j.carres.2007.11.023. [DOI] [PubMed] [Google Scholar]
- 55.Lodish H., Berk A., Zipursky S.L., Matsudaira P., Baltimore D., Darnell J. Molecular Cell Biology. 4th ed. WH Freeman; New York, NY, USA: 2000. Organelles of the Eukaryotic cell. [Google Scholar]
- 56.Terfrüchte M., Joehnk B., Fajardo-Somera R., Braus G.H., Riquelme M., Schipper K., Feldbrügge M. Establishing a versatile Golden Gate cloning system for genetic engineering in fungi. Fungal Genet. Biol. 2014;62:1–10. doi: 10.1016/j.fgb.2013.10.012. [DOI] [PubMed] [Google Scholar]
- 57.Smith J.J., Aitchison J.D. Peroxisomes take shape. Nat. Rev. Mol. Cell Biol. 2013;14:803–817. doi: 10.1038/nrm3700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lanver D., Müller A.N., Happel P., Schweizer G., Haas F.B., Franitza M., Pellegrin C., Reissmann S., Altmüller J., Rensing S.A., et al. The biotrophic development of Ustilago maydis studied by RNA-seq analysis. Plant Cell. 2018;30:300–323. doi: 10.1105/tpc.17.00764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Lai C.-Y., Cronan J.E. Isolation and characterization of β-ketoacyl-acyl carrier protein reductase (fabG) mutants of Escherichia coli and Salmonella enterica serovar Typhimurium. J. Bacteriol. 2004;186:1869–1878. doi: 10.1128/JB.186.6.1869-1878.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Holloman W.K., Schirawski J., Holliday R. The homologous recombination system of Ustilago maydis. Fungal Genet. Biol. 2008;45(Suppl. 1):S31–S39. doi: 10.1016/j.fgb.2008.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Schirawski J., Mannhaupt G., Münch K., Brefort T., Schipper K., Doehlemann G., Di Stasio M., Rössel N., Mendoza-Mendoza A., Pester D., et al. Pathogenicity determinants in smut fungi revealed by genome comparison. Science. 2010;330:1546–1548. doi: 10.1126/science.1195330. [DOI] [PubMed] [Google Scholar]
- 62.Laurie J.D., Ali S., Linning R., Mannhaupt G., Wong P., Güldener U., Münsterkötter M., Moore R., Kahmann R., Bakkeren G., et al. Genome comparison of barley and maize smut fungi reveals targeted loss of RNA silencing components and species-specific presence of transposable elements. Plant Cell. 2012;24:1733–1745. doi: 10.1105/tpc.112.097261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Rabe F., Bosch J., Stirnberg A., Guse T., Bauer L., Seitner D., Rabanal F.A., Czedik-Eysenberg A., Uhse S., Bindics J., et al. A complete toolset for the study of Ustilago bromivora and Brachypodium sp. as a fungal-temperate grass pathosystem. Elife. 2016;5:e20522. doi: 10.7554/eLife.20522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Bakkeren G., Szabo L.J. Progress on Molecular Genetics and Manipulation of Rust Fungi. Phytopathology. 2020;110:532–543. doi: 10.1094/PHYTO-07-19-0228-IA. [DOI] [PubMed] [Google Scholar]
- 65.Kleinstiver B.P., Prew M.S., Tsai S.Q., Topkar V.V., Nguyen N.T., Zheng Z., Gonzales A.P.W., Li Z., Peterson R.T., Yeh J.-R.J., et al. Engineered CRISPR-Cas9 nucleases with altered PAM specificities. Nature. 2015;523:481–485. doi: 10.1038/nature14592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Cong L., Ran F.A., Cox D., Lin S., Barretto R., Habib N., Hsu P.D., Wu X., Jiang W., Marraffini L.A., et al. Multiplex genome engineering using CRISPR/Cas systems. Science. 2013;339:819–823. doi: 10.1126/science.1231143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zetsche B., Heidenreich M., Mohanraju P., Fedorova I., Kneppers J., DeGennaro E.M., Winblad N., Choudhury S.R., Abudayyeh O.O., Gootenberg J.S., et al. Multiplex gene editing by CRISPR--Cpf1 using a single crRNA array. Nat. Biotechnol. 2017;35:31–34. doi: 10.1038/nbt.3737. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Accession numbers from National Center for Biotechnology Information (NCBI; www.ncbi.nlm.nih.gov): U. maydis: Don3 (UMAG_05543; XP_011391855), Pho85 (UMAG_11892; XP_011388110), Mat1 (UMAG_03114; XP_011389465), Fab4 (UMAG_02182; XP_011388532). U. hordei: Mat1 (UHOR_04871; CCF52714).