Abstract
Background
A combination of programmed cell death protein‐1 (PD‐1)/programmed cell death ligand‐1 (PD‐L1) inhibitors and radiotherapy (RT) is increasingly being used to treat non‐small‐cell lung cancer (NSCLC). However, the safety and efficacy of this approach remains controversial. We performed a systematic review and meta‐analysis to summarize the related research.
Methods
We searched the China Biology Medicine, EMBASE, Cochrane Library, and PubMed databases for all the relevant studies. The Stata software, version 12.0 was used for the meta‐analysis.
Results
The study included 20 clinical trials that enrolled 2027 patients with NSCLC. Compared with non‐combination therapy, combination therapy using PD‐1/PD‐L1 inhibitors and RT was associated with prolonged overall survival (OS) (1‐year OS: odds ratio [OR] 1.77, 95% confidence interval [CI] 1.35–2.33, p = 0.000; 2‐year OS: OR 1.77, 95% CI 1.35–2.33, p = 0.000) and progression‐free survival (PFS) (0.5‐year PFS: OR 1.83, 95% CI 1.13–2.98, p = 0.014; 1‐year PFS: OR 2.09, 95% CI 1.29–3.38, p = 0.003; 2‐year PFS: OR 2.47, 95% CI 1.13–5.37, p = 0.023). Combination therapy also improved the objective response rate (OR 2.76, 95% CI 1.06–7.19, p = 0.038) and disease control rate (OR 1.80, 95% CI 1.21–2.68, p = 0.004). This meta‐analysis showed that compared with non‐combination therapy, combination therapy using PD‐1/PD‐L1 inhibitors and RT did not increase the serious adverse event rates (≥grade 3); however, this approach increased the rate of grade 1–2 immune‐related or radiation pneumonitis. Subgroup analyses revealed that the sequence of PD‐1/PD‐L1 inhibitors followed RT outperformed in which concurrent PD‐1/PD‐L1 inhibitor and RT followed PD‐1/PD‐L1 inhibitor. Combination of stereotactic body RT or stereotactic radiosurgery with PD‐1/PD‐L1 inhibitors may be more effective than a combination of conventional RT with PD‐1/PD‐L1 inhibitors in patients with advanced NSCLC.
Conclusion
Combination therapy using PD‐1/PD‐L1 inhibitors and RT may improve OS, PFS, and tumor response rates without an increase in serious adverse events in patients with advanced NSCLC. However, combination therapy was shown to increase the incidence of mild pneumonitis.
Keywords: combined radio‐immunotherapy, meta‐analysis, non‐small‐cell lung cancer, programmed cell death protein‐1/programmed cell death ligand‐1 inhibitors, radiotherapy, systematic review
Radiotherapy combined with PD‐1/PD‐L1 inhibitors in the treatment of advanced NSCLC showed clinical long‐term survival benefit and short‐term efficacy benefit, and might not increase the serious adverse events.

1. INTRODUCTION
Non‐small‐cell lung cancer (NSCLC) is a common malignant tumor associated with significantly high morbidity and mortality rates globally. Latest statistical data show that there will be 228,820 new cases of lung cancer and 135,720 deaths in 2020. 1 Conventional treatments for NSCLC include surgery, chemotherapy, and radiotherapy (RT). Despite the progress in treatment of locally advanced and metastatic NSCLC using the aforementioned combination treatments over the past few decades, 2 survival rates and local control rates of this malignancy remain unsatisfactory. Currently, immune checkpoint inhibitors (ICIs) have shown promising results as novel anti‐cancer drugs. ICIs are monoclonal antibodies that can eliminate T‐cell inhibitory pathways. 3 Programmed cell death protein‐1/programmed cell death ligand‐1 (PD‐1/PD‐L1) inhibitors represent the most important and frequently used class of ICIs. Clinical trials are increasingly being performed in recent times to investigate the role of combination therapy using PD‐1/PD‐L1 inhibitors and RT for NSCLC. 4 This combination is referred to as “combined radio‐immunotherapy.” However, conflicting conclusions have been drawn regarding the efficacy and toxicity profile of combined radio‐immunotherapy in patients with NSCLC. Some clinical studies support the administration of combined radio‐immunotherapy; for example, the PACIFIC study (a phase III, double‐blind multicenter clinical study with a 2:1 randomized control design) 5 included patients with locally advanced NSCLC treated with durvalumab (a PD‐L1 inhibitor) followed by radiochemotherapy and reported significantly better overall survival (OS) and progression‐free survival (PFS) in this patient population than in those treated with radiochemotherapy alone. Adverse events (AEs) were similar between the groups. The KEYNOTE‐001 trial (a single‐center retrospective study) reported the same trend. A history of RT administered to any site can prolong PFS and OS in patients with metastatic NSCLC treated with pembrolizumab (a PD‐1 inhibitor) with acceptable toxicity. 6 Two retrospective studies have shown that combination treatment with RT and nivolumab (a PD‐1 inhibitor) in patients with advanced NSCLC may improve OS and PFS without an increase in acute toxicity rates. 7 , 8 However, a few studies have reported contrasting results. For example, the PEMBRO‐RT trial showed that stereotactic body radiotherapy (SBRT) preceding pembrolizumab treatment for metastatic lesions did not improve long‐term survival compared with pembrolizumab alone in patients with metastatic NSCLC. 9 Cho et al. 10 reported that RT‐induced lymphopenia in patients with advanced NSCLC can reduce the efficacy of ICIs. A study performed by Bang et al. 11 described higher overall toxicity following RT administration within 14 days of ICI treatment. A review by Li et al. 12 suggests a high incidence of immune‐related or radiation pneumonitis in patients with advanced NSCLC who receive combination radio‐immunotherapy. As mentioned earlier, the efficacy and safety of combination therapy using PD‐1/PD‐L1 inhibitors and RT in patients with NSCLC remains controversial. The results of high‐quality meta‐analyses are increasingly being considered high‐quality evidence. 13 , 14 , 15 Therefore, we performed a systematic review and meta‐analysis of the safety and efficacy of combination therapy using RT and PD‐1/PD‐L1 inhibitors for the treatment of patients with NSCLC.
2. MATERIALS & METHODS
This study was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta‐Analyses guidelines. 16 , 17 , 18 The protocol is available online at PROSPERO. A MeaSurement Tool to Assess systematic Reviews was used to assess methodological quality. 19 , 20
2.1. Search strategy
Two investigators independently searched for articles in the China Biology Medicine, EMBASE, Cochrane Library, and PubMed databases from inception until April 28, 2020, and no language restrictions were applied. We used the following search terms: “Carcinoma, Non‐Small‐Cell Lung,” “Programmed death‐1,” “Programmed death ligand‐1,” “Radiotherapy,” and “Chemoradiotherapy.” All relevant keyword variants for these terms were used. Figure 1 shows PRISMA flow diagram. Supplement 1 shows the detailed search strategy for each database.
FIGURE 1.
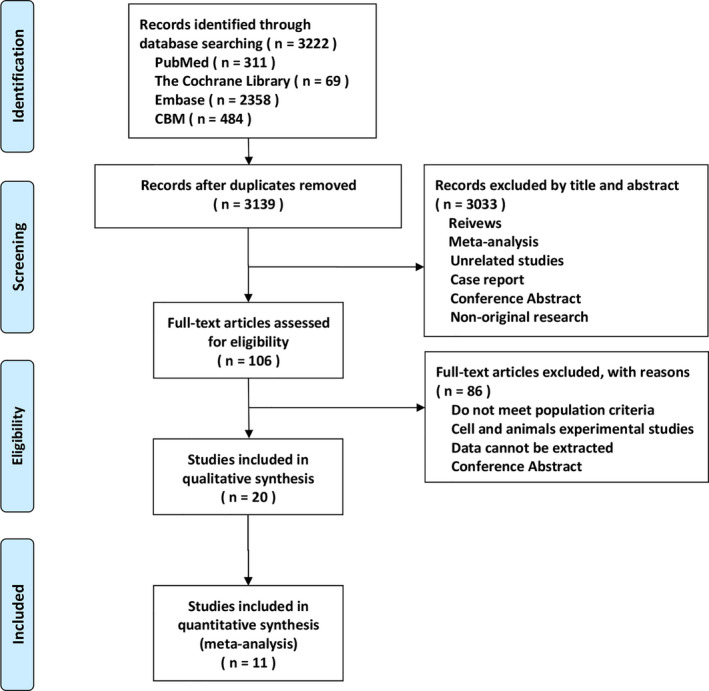
Flow diagram of the identification of eligible studies.
2.2. Inclusion and exclusion criteria
The inclusion criteria were as follows: Original papers of human clinical trials that reported the outcomes of combination therapy using PD‐1/PD‐L1 inhibitors and RT in patients with NSCLC. There were no restrictions on tumor stage, publication date, study population, language, or study design.
The exclusion criteria were as follows: (i) Studies that reported NSCLC concomitant with other malignant neoplasms, (ii) studies from which data could not be extracted, (iii) duplicate reports (only the latest or the parent study was included), (iv) studies that reported only protocols, and (v) abstracts for which the complete text was unavailable.
2.3. Study selection
After the literature search, two investigators discarded all duplicate studies obtained from various databases and independently screened the titles and abstracts of all remaining articles to exclude manuscripts that did not meet the inclusion criteria. Furthermore, the investigators carefully checked the complete texts of articles that were deemed potentially eligible for inclusion in this research. Finally, a third investigator discussed the selections with the two investigators who performed the screening. Any disagreement regarding study eligibility that persisted after this discussion was resolved by consulting a fourth investigator.
2.4. Data extraction
The two aforementioned investigators independently extracted the following information from each trial: (i) the characteristics of the included trials, including the country, year of publication, author names, baseline characteristics, number of patients, smoking status, PD‐L1 expression status, oncogene driver mutation status, disease stage, PD‐1/PD‐L1 inhibitors and dose, RT dose (Gy)/fraction, the sequence of administration of PD‐1/PD‐L1 inhibitors and RT, RT target sites, RT techniques, and follow‐up duration. (ii) Long‐term outcomes represented by the OS and PFS. (iii) Short‐term outcomes represented by the objective response rate (ORR) and the disease control rate (DCR). (iv) AEs (regardless of grade).
2.5. Quality assessment
The two aforementioned investigators independently assessed the methodological quality of the randomized controlled trials (RCTs) using the Cochrane Handbook for Systematic Reviews of Interventions, version 5.1.0 (Cochrane Collaboration Risk of Bias Tool). 21 The Newcastle‐Ottawa Scale was used for cohort or case‐control studies. The Case Series Report Quality Evaluation Form was used for single‐arm studies. 22 The details of the quality assessment are display in Tables S1–S3 (Supplement 2).
2.6. Statistical analysis
We used the Stata software, version 12.0 (2011, Stata Corp, College Station, TX, USA) for data analyses. Odds ratios (ORs) and their 95% confidence intervals (CIs) were used as summary statistics for dichotomous data. A two‐tailed p value of 0.05 was considered statistically significant. Data were combined using a random‐effects model, and statistical heterogeneity was evaluated using the I2 test. A Higgins I2 statistic <50% represented low heterogeneity and >50% represented high heterogeneity. Subgroup analyses were performed to determine the source of heterogeneity and the factors associated with clinical benefits, if any.
3. RESULTS
3.1. Study characteristics
Approximately 2027 patients were enrolled in the 20 studies included in this research. With regard to the study design, nine studies were non‐randomized controlled trials (NRCT), 6 , 7 , 8 , 23 , 24 , 25 , 26 , 27 , 28 nine were single‐arm studies, 29 , 30 , 31 , 32 , 33 , 34 , 35 , 36 , 37 and two were RCTs. 5 , 9 All studies included at least one arm treated with combination therapy using PD‐1/PD‐L1 inhibitors and RT. Six studies compared the administration of RT plus PD‐1/PD‐L1 inhibitors with PD‐1/PD‐L1 inhibitors alone, and five studies compared the administration of RT plus PD‐1/PD‐L1 inhibitors with RT alone. Of the 20 studies, 13 reported chemotherapy regimens used and seven did not describe these regimens. Most chemotherapy regimens were platinum‐doublet or platinum‐based; however, the exact number of chemotherapy cycles administered was unknown. Table 1 summarizes the baseline characteristics of the included studies (the RT technique, RT dose, the sequence of PD‐1/PD‐L1 inhibitors and RT administered, and types of PD‐1/PD‐L1 inhibitors used). Table 2 summarizes the characteristics of patients included in the studies (age, sex, histopathological features, smoking status, PD‐L1 status, oncogene driver mutation status, Eastern Cooperative Oncology Group status, or the Karnofsky Performance Status). Tables S1–S3 (Supplement 2) show the results of the quality analysis.
TABLE 1.
Characteristics of the included studies.
| Author | Year | Nation | Study type | Group | N | Tumor stage | CT status, N (%) | RT target | RT technique | RT dose/fraction | ICIs sequencing | ICIs type |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Shaverdian | 2017 | America | NRCT | RT+ICI | 42 | IV | Not clear | MRT |
Con‐RT SRS SBRT |
Not clear | Sequential (After RT) |
Anti‐PD−1 Pembrolizumab |
| ICI | 55 | |||||||||||
| Tamiya | 2017 | Japan | NRCT | RT+ICI | 50 | IV | Not clear | TRT | Not clear | Not clear | Sequential (After RT) |
Anti‐PD−1 Nivolumab |
| ICI | 151 | |||||||||||
| Fiorica | 2018 | Italy | NRCT | RT+ICI | 15 | IIIB‐IV |
Prior CT≧ 1cycle platinum‐based |
MRT |
3DCRT IMRT SBRT |
36 Gy/12F 8–16 Gy/1‐2F |
Sequential (After RT) |
Anti‐PD−1 Nivolumab |
| ICI | 20 | |||||||||||
| Theelen | 2019 | Netherland |
RCT Phase II |
RT+ICI | 36 | IV |
Prior CT≧ 1cycle Regimen not clear |
MRT | SBRT | 24 Gy/3F | Sequential (After RT) |
Anti‐PD−1 Pembrolizumab |
| ICI | 40 | |||||||||||
| Yamaguchi | 2019 | Japan | NRCT | RT+ICI | 66 | III‐IV |
Prior CT ≧ 1cycle Most platinum‐based |
MRT | Not clear |
Bone 8–30 Gy Thorax 30–60 Gy Brain 30–50 Gy |
Sequential (After RT) |
Anti‐PD−1 Nivolumab |
| ICI | 58 | |||||||||||
| Ratnayake | 2020 | Australia | NRCT | RT+ICI | 65 | IV |
Prior CT platinum doublet |
MRT | Not clear |
30 Gy (8–66 Gy) 20 Gy (6–50 Gy) |
Sequential (After RT) Concurrent |
Anti‐PD−1 Nivolumab |
| ICI | 20 | |||||||||||
| Hubbeling | 2018 | America | NRCT | RT+ICI | 50 |
IV With BM |
Prior CT platinum doublet |
MRT |
WBRT PBI SRS |
Not clear WBRT and PBI 10‐15F SRS 1‐5F |
Sequential (After and Before RT) Concurrent |
Anti‐PD−1 Anti‐PD‐L1 |
| RT | 113 | |||||||||||
| Shepard | 2019 | America | NRCT | RT+ICI | 17 |
IV With BM |
Prior CT Most platinum doublet |
MRT | SRS | 18.4 Gy±2.3 Gy | Concurrent |
Nivolumab Pembrolizumab Atezolizumab |
| RT | 34 | 19.3 Gy±2.5 Gy | ||||||||||
| Fukui | 2020 | Japan | NRCT | RT+ICI | 18 | IIIA‐IIIB |
Prior CT ≧ 2cycle platinum‐based |
TRT | 3DCRT | 60 (10–66)Gy | Sequential (After RT) |
Nivolumab Pembrolizumab Atezolizumab |
| RT | 90 | |||||||||||
| Gray | 2020 | Multicenter |
RCT Phase III |
RT+ICI | 476 | III |
Prior CT ≧ 2cycle platinum‐based |
TRT | Not clear |
<54 Gy ≥54 Gy to ≤66 Gy >66 Gy to ≤74 Gy |
Sequential (After RT) |
Anti‐PD‐L1 Durvalumab |
| RT | 237 | |||||||||||
| Singh | 2020 | America | NRCT | RT+ICI | 39 |
IV With BM |
Prior CT Regimen and cycle not clear |
MRT | SRS | 18 Gy (12–24 Gy) |
Sequential Concurrent |
Nivolumab Pembrolizumab Ipilimumab nivolumab Atezolimumab |
| RT | 46 | |||||||||||
| Ahmed | 2017 | America |
NRCT Single arm |
RT+ICI | 17 |
IV With BM |
Most prior CT Platinum‐based |
MRT |
SRS FSRT |
18 Gy/1F, 20 Gy/1F, 21 Gy/1F, 24 Gy/1F, 25 Gy/5F |
Sequential Concurrent |
Anti‐PD−1 Anti‐PD‐L1 |
| Lesueur | 2018 | France |
NRCT Single arm |
RT+ICI | 104 | IV | Not clear | MRT |
3DCRT SRS IMRT Other |
30 Gy (29.6–30.0 Gy) |
Sequential Concurrent |
Anti‐PD−1 Nivolumab |
| Schapira | 2018 | America |
NRCT Single arm |
RT+ICI | 37 |
IV With BM |
Prior CT Regimen and cycle not clear |
MRT | SRS | 25 Gy/5F, 22 Gy/2F, 21 Gy/3F, 20 Gy/2F, 18 Gy/1F, 17 Gy/1F, 16 Gy/1F, 15 Gy/1F |
Sequential Concurrent |
Nivolumab Pembrolizumab Atezolizumab |
| Miyamoto | 2019 | Japan |
NRCT Single arm |
RT+ICI | 6 | IV |
Prior CT Regimen and cycle not clear |
MRT | SRT | 33 Gy/3F, 30 Gy/3F, 38 Gy/4F, 48 Gy/4F, 25.5 Gy/3F | Sequential (After RT) |
Anti‐PD−1 Nivolumab |
| Qin | 2019 | America |
NRCT Single arm |
RT+ICI | 12 | IV |
Prior CT platinum doublet |
MRT | HIGRT | 12–50 Gy, 6–8 Gy/F, 3F−5F | Concurrent |
Anti‐PD‐L1 Atezolizumab |
| Amino | 2020 | Japan |
NRCT Single arm |
RT+ICI | 20 | III |
Prior CT platinum doublet |
TRT | Not clear | 54–60 Gy | Sequential (After RT) |
Anti‐PD−1 Nivolumab Pembrolizumab |
| Chu | 2020 | China |
NRCT Single arm |
RT+ICI | 31 | III |
Prior CT platinum doublet |
TRT | Not clear |
66–70 Gy/32‐35F 60–66 Gy >66 Gy |
Sequential (After RT) |
Anti‐PD‐L1 Durvalumab |
| Jabbour | 2020 | America |
NRCT Single arm |
RT+ICI | 21 | III |
Prior CT Platinum doublet |
TRT |
IMRT VMAT Proton |
60 Gy 40 Gy |
Concurrent |
Anti‐PD−1 Pembrolizumab |
| Miura | 2020 | Japan |
NRCT Single arm |
RT+ICI | 41 | III |
Prior CT platinum‐based |
TRT |
4D‐CT IGRT |
60 Gy/30F 54 Gy/25F |
Sequential (After RT) |
Anti‐PD‐L1 Durvalumab |
BM, brain metastasis; CT, chemotherapy; RT, radiotherapy; ICIs, immune checkpoint inhibitors; TRT, thoracic radiotherapy; MRT, radiotherapy of metastases; Co‐RT, conventional radiotherapy; 3DCRT, 3‐dimensional‐conformal radiation therapy; IMRT, intensity modulated radiation therapy; SRS, stereotatic radiosurgery; SBRT, stereotactic body radiation therapy; PBI, partial brain irradiation; FSRT, fractionated stereotactic radiotherapy; HIGRT, hypofractionated image‐guided radiation therapy; VMAT, volumetric modulated arc therapy; IGRT, image‐guided radiotherapy; IMRT, intensity modulated radiation therapy.
TABLE 2.
Patients characteristics of the included studies.
| Author | Year | Group | N | Age median (range) | SEX, n (%) | Histology, n (%) | ECOG, n (%) | Smoking status, n (%) | PD‐L1 status | Oncogene driver mutation | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Male | Female | Sq | Ad | Oth | 0–1 | ≧2 | Yes | No | EGFR | KRAS | Oth | ||||||
| Shaverdian | 2017 | RT+ICI | 42 | 65 (36.0–77.0) | 21 (50.0) | 21 (50.0) | 11 (20.0) | 31 (74.0) | 42 (100.0) | 0 | 23 (55.0) | 19 (45.0) |
P 30 (71.0) N 5 (12.0) U 7 (17.0) |
‐ | |||
| ICI | 55 | 66 (32.0–83.0) | 29 (53.0) | 26 (47.0) | 8 (15.0) | 47 (85.0) | 55 (100.0) | 0 | 30 (55.0) | 25 (45.0) |
P 44 (80.0) N 6 (11.0) U 5 (9.0) |
‐ | |||||
| Tamiya | 2017 | RT+ICI | 50 | 68 (27–87) | 135 (67.2) | 66 (32.8) | 42 (20.9) | 142 (70.6) | 17 (8.5) | 153 (76.1) | 48 (23.9) | 44 (21.9) | 157 (78.1) | ‐ |
P 36 (17.9)/N 128 (63.7) U 37 (18.4) |
||
| ICI | 151 | 68 (27–87) | |||||||||||||||
| Fiorica | 2018 | RT+ICI | 15 | 70 (44–81) | 11 (73.0) | 4 (27.0) | 9 (60.0) | 6 (40.0) | 9 (60.0) | 6 (40.0) | 14 (93.0) | 1 (7.0) | ‐ | ‐ | |||
| ICI | 20 | 69 (53–77) | 19 (95.0) | 1 (0.5) | 10 (50.0) | 10 (50.0) | 14 (70.0) | 6 (30.0) | 18 (90.0) | 2 (10.0) | |||||||
| Theelen | 2019 | RT+ICI | 36 | 62 (35–78) | 20 (55.6) | 16 (44.4) | 5 (13.9) | 31 (86.1) | 36 (100.0) | 0 | 36 (100.0) | 0 |
0: 18 (50.0) 1–49%: 8 (22.2) ≥50%: 10 (27.8) |
‐ | |||
| ICI | 40 | 23 (57.5) | 17 (42.5) | 4 (10.0) | 36 (90.0) | 40 (100.0) | 0 | 40 (100.0) | 0 |
0: 25 (62.5) 1–49%:8 (20.0) ≥50%: 5 (12.5) |
|||||||
| Yamaguchi | 2019 | RT+ICI | 66 | 69 (31–85) | 51 (77.3) | 15 (22.7) | 35 (53.0) | 31 (47.0) | 0: 31 (47.0) | 1–3: 35 (53.0) | 52 (78.8) | 14 (21.2) | ‐ | P 10 (15.2)/N 51 (77.3) | |||
| ICI | 58 | 42 (72.4) | 16 (27.6) | 24 (41.4) | 34 (58.6) | 0: 18 (31.0) | 1–3: 40 (69.0) | 47 (81.0) | 11 (19.0) | P 4 (6.9)/N 53 (91.4) | |||||||
| Ratnayake | 2020 | RT+ICI | 65 | 65 (42–84) | 44 (67.7) | 21 (32.3) | 20 (30.8) | 41 (63.1) | 4 (6.2) | 40 (61.5) | 23 (35.4) | 58 (89.2) | 7 (10.8) | ‐ | ‐ | ||
| ICI | 20 | 9 (45.0) | 11 (55.0) | 8 (40.0) | 12 (60.0) | 0 | 13 (65.0) | 7 (35.0) | 17 (85.0) | 3 (15.0) | |||||||
| Hubbeling | 2018 | RT+ICI | 50 | 61 (35–82) | 16 (32.0) | 34 (68.0) | 8 (16.0) | 38 (76.0) | 4 (8.0) | M(R):1 (0–3) | ‐ | ‐ | ‐ | ||||
| RT | 113 | 62 (31–97) | 42 (37.0) | 71 (63.0) | 11 (10.0) | 99 (88.0 | 3 (3.0) | M(R):1 (0–4) | |||||||||
| Shepard | 2019 | RT+ICI | 17 | 64.4 ± 8.6 | 11 (64.7) | 6 (35.3) | ‐ | ‐ | ‐ | ‐ | ‐ | ||||||
| RT | 34 | 64.1 ± 10.2 | 20 (58.8) | 14 (41.2) | |||||||||||||
| Fukui | 2020 | RT+ICI | 18 | 65 (36–76) | 81 (75.0) | 27 (25.0) | 38 (35.0) | 46 (43.0) | 24 (22.0) | 106 (98.0) | 2 (2.0) | 97 (90.0) | 11 (10.0) | ‐ |
P 2 (2.0)/N 69 (62.0) U 32 (30.0) |
‐ |
ALK 4 (4.0) ROS1 1 (1.0) |
| RT | 90 | ||||||||||||||||
| Gray | 2020 | RT+ICI | 476 | 64 (31–84) | 334 (70.2) | 142 (29.8) | 224 (47.1) | 252 (52.9) | 476 (100.0) | ‐ | 433 (91.0) | 43 (9.0) |
≥25%: 115 (24.2) <25%: 187 (39.3) |
P 29 (6.1)/N 315 (66.2) U 132 (27.7) |
|||
| RT | 237 | 64 (23–90) | 166 (70.0) | 71 (30.0) | 102 (43.0) | 135 (57.0) | 237 (100.0) | ‐ | 216 (91.1) | 21 (8.9) |
≥25%: 44 (18.6) <25%: 105 (44.3) |
P 14 (5.9)/N 165 (69.6) U 58 (24.5) |
|||||
| Singh | 2020 | RT+ICI | 39 | 61.9 (28–87.5) | 12 (30.7) | 23 (50.0) | 6 (15.0) | 28 (72.5) | ‐ | KPS:80 (50–100) | ‐ | P 39 (100.0) | ‐ | ||||
| RT | 46 | 62.5 (31.8–79.4) | 27 (69.2) | 23 (50.0) | 3 (6.5) | 29 (63.0) | ‐ | KPS:90 (60–100) | ‐ | ||||||||
| Ahmed | 2017 | RT+ICI | 17 | 60 (44–79) | 10 (58.8) | 7 (41.2) | ‐ |
KPS:90: 9 (53.0) 80: 5 (29.0) 70: 3 (18.0) |
‐ | ‐ | 2 (12.0) | 3 (18.0) | Both EGFR and KRAS:1 (6.0) | ||||
| Lesueur | 2018 | RT+ICI | 104 | 60.3 (54.5–67.1) | 67 (64.4) | 37 (35.6) | 65 (62.5) | 34 (32.7) | 5 (4.8) | 69 (66.4) | 35 (33.5) | 96 (92.3) | 8 (7.7) | ‐ | 2 (1.9) | 22 (21.2) |
ALK:2 (1.9) MET:5 (4.8) Others:3 (2.9) |
| Schapira | 2018 | RT+ICI | 37 | 63 (42–84) | 13 (35.1) | 24 (64.9) | ‐ |
KPS:90–100:24 (64.9) 70–80:12 (32.4) 60:1 (2.7) |
‐ | ‐ | ‐ | ||||||
| Miyamoto | 2019 | RT+ICI | 6 | 58 (45–72) | 4 (66.7) | 2 (33.3) | ‐ | 5 (83.3) | 1 (16.7) | 0–2: 6 (100.0) | 4 (66.7) | 2 (33.3) | ≥50%:4 (66.7) | 2 (33.3) | 6 (100.0) | ‐ | |
| Qin | 2019 | RT+ICI | 12 | 64 (55–77) | 8 (66.7) | 4 (33.3) | 2 (16.7) | 7 (58.3) | 3 (25.0) | 0–2: 12 (100.0) | 1 (91.7) | 1 (8.3) |
P: 9 (75.0) U: 3 (25.0) |
‐ | |||
| Amino | 2020 | RT+ICI | 20 | 59 (42–73) | 18 (90.0) | 2 (10.0) | 14 (70.0) | ‐ | 6 (30.0) |
0: 8 (40.0) 1: 11 (55.0) 2: 1 (5.0) |
19 (95.0) | 1 (5.0) |
<1%: 1 (5.0) 1–49%: 4 (20.0) ≥50%: 5 (25.0) NE: 10 (50.0) |
‐ | |||
| Chu | 2020 | RT+ICI | 31 | 64 (52–74) | 26 (83.9) | 5 (16.1) | 8 (25.8) | 20 (64.5) | 3 (9.7) |
0: 25 (80.7) 1: 5 (16.1) 2: 1 (3.2) |
23 (74.2) | 8 (25.8) |
P: 14 (45.2) N: 6 (19.3) U: 11 (35.5) |
4 (12.9) | ‐ | ALK:1 (3.3) | |
| Jabbour | 2020 | RT+ICI | 21 | 69.5 (53–85) | 10 (48.0) | 11 (52.0) | 10 (48.0)* | 11 (52.0) | 21 (100.0) | ‐ | 20 (95.0) | 1 (5.0) |
<1%: 4 (21.0) 1–49%: 10 (53.0) ≥50%: 5 (26.0) |
‐ | |||
| Miura | 2020 | RT+ICI | 41 | 72 (51–80) | 33 (80.0) | 8 (20.0) | 15 (37.0) | 21 (51.0) | 5 (12.0) |
0: 24 (58.0) 1: 17 (42.0) |
‐ | 33 (80.0) | 8 (20.0) |
<1%: 12 (29.0) 1–49%: 11 (27.0) ≥50%: 9 (22.0) NE: 9 (22.0) |
5 (12.0) |
ROS1:1 (3.0) Unknown:35 (85.0) |
|
RT, radiotherapy; ICI, immune checkpoint inhibitors; Sq, squamous; Ad, adenocarcinoma; Oth, other; ECOG, Eastern Cooperative Oncology Group; M(R), median(range); P, positive; N, negative; U, unknown; KPS, Karnofsky; NE, not evaluate.
3.2. Long‐term efficacy outcomes: Overall survival and progression‐free survival
Survival outcomes were the primary endpoints in our systematic review and meta‐analysis. Eight studies 5 , 6 , 7 , 8 , 9 , 24 , 25 , 26 were included in this meta‐analysis. Compared with non‐combination therapy, combination radio‐immunotherapy using PD‐1/PD‐L1 inhibitors and RT was significantly associated with longer 1‐year OS (I2 = 0.0%, OR 1.77, 95% CI 1.35–2.33, p = 0.000) and 2‐year OS (I2 = 56.3%, OR 1.77, 95% CI 1.35–2.33, p = 0.000) in patients with advanced NSCLC. Subgroup analysis based on the study design (RCT or NRCT) showed differences in 2‐year OS (RCT: OR 1.60, 95% CI 1.16–2.20, p = 0.004, vs NRCT: I2 = 70.6%, OR 2.50, 95% CI 0.81–7.66, p = 0.110). We also investigated the effect of the sequence of treatment rendered and the RT techniques. Subgroup analyses showed longer survival time in the sequential RT‐first group than in the concurrent or sequential ICI‐first group. With regard to RT techniques, combination therapy using SBRT or SRS and PD‐1/PD‐L1 inhibitors was associated with better survival benefits than combination therapy using conventional RT and PD‐1/PD‐L1 inhibitors. Figure 2 shows the results of the meta‐analysis and subgroup analyses of the 1‐ and 2‐year OS rates depicted as forest plots.
FIGURE 2.
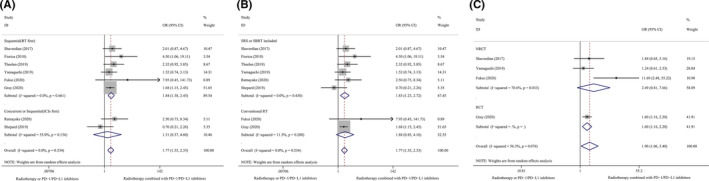
Subgroup analyses of 1–2 year OS. (A), 1‐year OS, Sequence of RT and ICIs. (B), 1‐year OS, RT techniques. (C), 2‐year OS, Study design. RT, radiotherapy; ICIs, immune checkpoint inhibitors; OS, overall survival; SBRT, stereotactic body radiotherapy; SRS, stereotactic radiosurgery; RCT, randomized controlled trial; NRCT, non‐randomized controlled trial.
Compared with non‐combination therapy, combination radio‐immunotherapy using PD‐1/PD‐L1 inhibitors and RT was significantly associated with a longer 0.5‐year PFS (I2 = 43.1%, OR 1.83, 95% CI 1.13–2.98, p = 0.014), 1‐year PFS (I2 = 45.9%, OR 2.09, 95% CI 1.29–3.38, p = 0.003), and 2‐year PFS (I2 = 0.0%, OR 2.47, 95% CI 1.13–5.37, p = 0.023) in patients with advanced NSCLC. Subgroup analysis based on the study design (RCT or NRCT) also showed differences in the 1‐year PFS (RCT: I2 = 0.0%, OR 2.22, 95% CI 1.63–3.01, p = 0.000 and NRCT: I2 = 65.1%, OR 2.14, 95% CI 0.84–5.49, p = 0.112). Figure 3 shows the results of the meta‐analysis and subgroup analyses of the 0.5‐, 1‐, and 2‐year PFS depicted as forest plots.
FIGURE 3.
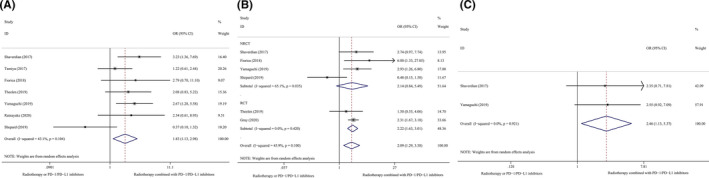
Subgroup analyses of 0.5–2 year PFS. (A), 0.5‐year PFS, Overall population. (B), 1‐year PFS, Study design. (C), 2‐year PFS, Overall population. PFS, progression‐free survival; RCT, randomized controlled trial; NRCT, non‐randomized controlled trial.
Among the eight single‐arm studies that investigated patients who received combination therapy using PD‐1/PD‐L1 and RT, the 0.5‐year OS was 88.1% (median) (range 81.0%–95.2%), the 1‐year OS was 50.5% (48.8%–85.2%), and the 2‐year OS was 27.1% (25.0%–29.1%). The median OS duration was 21.6 months (6.9–29.4 months). Only one study reported a 0.5‐year PFS rate of 81.0%. The 1‐year PFS was 42.5% (20.9%–67.9%), and only one study reported 2‐year PFS of 10.1%. The median PFS was 8.4 months (2.3–18.7 months) (Table 3).
TABLE 3.
Outcomes of single‐arm studies.
| Author | Year | N | ORR (%) | DCR (%) | 6‐month OS (%) | 1‐year OS (%) | 2‐year OS (%) | 6‐month PFS (%) | 1‐year PFS (%) | 2‐year PFS (%) | Median OS (month) | Median PFS (month) | AE<G3 (%) | AE≧G3 (%) | p<G3 (%) | P≧G3 (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ahmed | 2017 | 17 | ‐ | ‐ | 81.0 | 51.0 | ‐ | ‐ | ‐ | ‐ | 17.9 | ‐ | ‐ | 11.8 | ‐ | ‐ |
| Lesueur | 2018 | 104 | ‐ | ‐ | ‐ | 48.8 | 29.1 | ‐ | 20.9 | 10.1 | 25.2 | 2.7 | 85.6 | 14.4 | 3.3 | 1.1 |
| Schapira | 2018 | 37 | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ |
| Miyamoto | 2019 | 6 | 50.0 | 66.7 | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | 4.6 | ‐ | 16.7 | 0 | 16.7 |
| Qin | 2019 | 12 | 25.0 | 50.0 | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | 6.9 | 2.3 | 8.3 | 41.7 | 0 | 8.3 |
| Amino | 2020 | 20 | 45.0 | 65.0 | ‐ | 50.0 | 29.1 | ‐ | 28.5 | ‐ | 26.2 | 8.4 | 30.0 | 5.0 | 5.0 | 0 |
| Chu | 2020 | 31 | 25.8 | 80.6 | ‐ | ‐ | ‐ | ‐ | 56.4 | ‐ | 12 | 58.6 | 13.8 | 10.3 | 6.9 | |
| Jabbour | 2020 | 21 | ‐ | ‐ | 95.2 | 85.2 | ‐ | 81.0 | 69.7 | ‐ | 29.4 | 18.7 | ‐ | ‐ | 28.0 | 5.0 |
| Miura | 2020 | 41 | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | ‐ | 15.4 | 14.1 | ‐ | ‐ | 58.5 | 2.4 |
ORR, objective response rate; DCR, disease control rate; OS, overall survival; PFS, progression‐free survival; AE, adverse events; P, pneumonitis.
3.3. Short‐term efficacy outcomes: Evaluation of response to treatment
The tumor response rate, which was determined using the Response Evaluation Criteria in Solid Tumors guideline, version 1.1 was the secondary endpoint in this meta‐analysis. One study used the Immune‐related Response Criteria 6 and another used the Response Assessment in Neuro‐oncology tool. 26 Results of the meta‐analysis showed that combination therapy using PD‐1/PD‐L1 inhibitors and RT improved the ORR (I2 = 78.1%, OR 2.76, 95% CI 1.06–7.19, p = 0.038) and DCR (I2 = 0.0%, OR 1.80, 95% CI 1.21–2.68, p = 0.004).
Subgroup analyses based on the study design (RCT vs NRCT) showed differences in ORRs (RCT: OR 3.08, 95% CI 1.15–8.29, p = 0.026 vs NRCT: I2 = 83.5%, OR 2.77, 95% CI 0.79–9.76, p = 0.112). However, the DCR did not significantly differ (RCT: OR 2.87, 95% CI 1.10–7.49, p = 0.031 vs NRCT: I2 = 0.0%, OR 1.64, 95% CI 1.06–2.53, p = 0.026). Additionally, this meta‐analysis showed that the DCRs were significantly higher in patients who received combination therapy using PD‐1/PD‐L1 inhibitors and RT than in patients who received ICIs alone (I2 = 0.0%, OR 1.93, 95% CI 1.25–2.97, p = 0.003); however, no significant differences were observed in the ORRs. Moreover, the ORRs were significantly higher in patients who received combination therapy using PD‐1/PD‐L1 inhibitors and RT than in patients who received RT alone (I2 = 20.0%, OR 7.04, 95% CI 2.27–21.87, p = 0.001); however, no significant differences were observed in the DCRs. The median ORR and DCR were 35.4% (25.0%–50.0%) and 65.9% (50.0%–80.6%), respectively in single‐arm studies (Table 3). Figure 4 shows the results of the meta‐analysis and subgroup analyses of the ORR and DCR depicted as forest plots.
FIGURE 4.
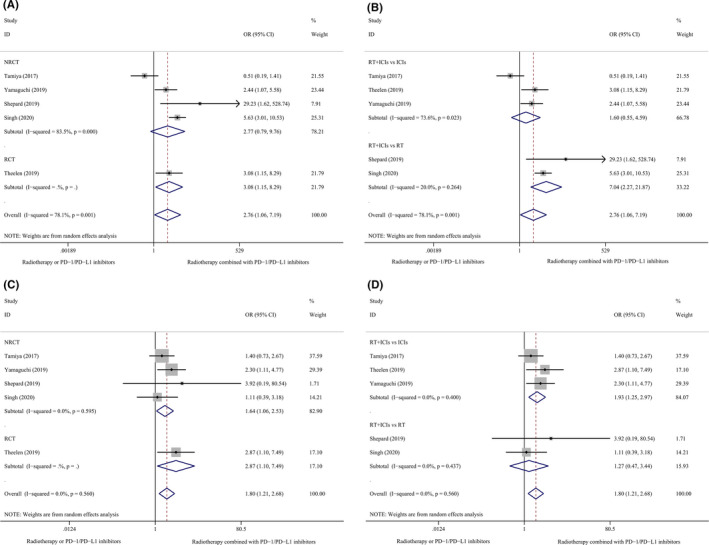
Subgroup analyses of ORR and DCR. (A), ORR, Study design. (B), ORR, Different groups. (C), DCR, Study design. (D), DCR, Different groups. RT, radiotherapy; ICIs, immune checkpoint inhibitors; ORR, objective response rate; DCR, disease control rate; RCT, randomized controlled trial; NRCT, non‐randomized controlled trial.
3.4. Adverse events
AEs were defined using the National Cancer Institute‐Common Terminology Criteria for Adverse Events (NCI‐CTCAE), version 4.0. One study used the NCI‐CTCAE, version 5.0 31 and another used the NCI‐CTCAE, version 3.0. 7 The included studies primarily reported the total AE rates and immune‐related or radiation pneumonitis rates associated with combination therapy using PD‐1/PD‐L1 inhibitors and RT. The AEs described included fatigue, dermatitis, skin rash, diarrhea, nausea, constipation, anemia, neutropenia, leukopenia, thrombocytopenia, hypothyroidism, and abnormal liver function.
Results of the meta‐analysis showed that combination therapy using PD‐1/PD‐L1 inhibitors and RT did not increase the serious AE rates (≥grade 3) (I2 = 0.0%, OR 1.24, 95% CI 0.88–1.74, p = 0.222) or the mild AE rates (grades 1 and 2) (I2 = 0.0%, OR 0.97, 95% CI 0.71–1.34, p = 0.858) compared with administration of PD‐1/PD‐L1 inhibitors or RT rendered alone. Notably, combination therapy using PD‐1/PD‐L1 inhibitors and RT was not associated with a high risk of serious (≥grade 3) immune‐related or radiation pneumonitis, although rates of mild (grade 1–2) pneumonitis were increased in patients who received combination therapy. Subgroup analyses revealed that thoracic RT (OR 1.47, 95% CI 1.02–2.12, p = 0.040) was associated with a higher incidence of grade 1–2 pneumonitis than RT administered to metastatic lesions (I2 = 0.0%, OR 5.09, 95% CI 0.86–30.24, p = 0.074, Cross p = 0.180). The risk of grade 1–2 pneumonitis was higher in those who received PD‐L1 inhibitors (OR 1.47, 95% CI 1.02–2.12, p = 0.040) than in those who received PD‐1 inhibitors (I2 = 0.0%, OR 5.09, 95% CI 0.86–30.24, p = 0.074, Cross p = 0.180).
Three studies that reported combination therapy using SRS and PD‐1/PD‐L1 inhibitors 23 , 26 , 27 compared the incidence of cerebral radiation‐induced necrosis in patients with brain metastases. Notably, combination therapy using PD‐1/PD‐L1 inhibitors and RT did not significantly increase the incidence of cerebral radiation‐induced necrosis One study showed no significant differences in the progression of brain peritumoral edema between patients with NSCLC treated with and without combination therapy. 26 The median incidence rate of mild AEs was 44.3% (8.3%–85.6%), and the median incidence rate of serious AEs was 14.1% (5.0%–41.7%) in single‐arm studies included in our research. The incidence rate of mild pneumonitis was 5% (0.0%–58.5.0%), and the median incidence rate of serious pneumonitis was 5% (0.0%–16.7%) (Table 3). Figure 5 shows the results of the meta‐analysis and subgroup analyses of the AEs and mild pneumonitis depicted as forest plots.
FIGURE 5.

Subgroup analyses of AEs and mild pneumonitis. (A), AEs ≥grade 3, Overall population. (B), AEs grade 1–2, Overall population. (C), Pneumonitis grade 1–2, The site of RT. (D), Pneumonitis grade 1–2, PD‐1 vs PDL1. RT, radiotherapy; AEs, adverse events.
4. DISCUSSION
Our results suggest that combination therapy using PD‐1/PD‐L1 inhibitors and RT may improve OS, PFS, and tumor response rates in patients with advanced NSCLC. Moreover this therapeutic approach did not significantly increase the risk of serious AEs.
We observed that combination therapy using PD‐1/PD‐L1 inhibitors and RT was associated with prolonged 1‐ and 2‐year OS, improved 0.5, 1‐, and 2‐year PFS, and improved ORRs and DCRs. The higher survival benefit and better tumor response rates associated with combined radio‐immunotherapy could be attributed to the following mechanisms: The PD‐1 receptor is highly expressed on activated infiltrating T cells induction by the tumor microenvironment, and PD‐L1 is expressed on the surface of tumor cells. Tumor cells can trigger the PD‐1 pathway to escape the anti‐tumor immune response, and PD‐1 or PD‐L1 inhibitors can restore T‐cell signaling, with subsequent reactivation of the antitumor activity of specific CD8+T cells. Recruitment and activation of CD8+T cells are the key processes involved in the therapeutic actions of PD‐1/PD‐L1 inhibitors. 3 (i) RT induces and enhances the immunogenicity of tumors by increasing the expression of tumor‐associated antigens, the major histocompatibility complex, and damage‐associated molecular patterns. 38 CD8+T and dendritic cells subsequently recruit and activate accelerately, inducing the anti‐tumor immunological effect in the body. 38 , 39 , 40 , 41 (ii) RT also regulates the tumor microenvironment, increases the levels of chemokines CXCL10 and CXCL16 in the tumor microenvironment, and promotes the migration of CD8+T cells to the tumor site. 42 (iii) RT activates the cGAS/STING pathway to produce an effective immune response. 43 (iv) ICIs not only activate CD8+T cells but also normalize the tumor vasculature, reduce the state of tumor hypoxia, and increase tumor radiosensitivity. 44 (v) “Abscopal effects” have been detected at distant tumor sites that do not receive radiation during RT. Therefore, combination therapy using PD‐1/PD‐L1 inhibitors and RT may promote “abscopal effects” and enhance the effectiveness of cancer immunotherapy. 45 The aforementioned mechanisms could contribute to the short‐ and long‐term benefits of combination therapy using PD‐1/PD‐L1 inhibitors and RT in patients with advanced NSCLC.
The sequence of administration of combination therapy using PD‐1/PD‐L1 inhibitors and RT is the focus of investigation in this field; however, currently, the optimal sequence remains unclear. Subgroup analyses performed in our study showed that PD‐1/PD‐L1 inhibitor administration following RT outperformed in which concurrent PD‐1/PD‐L1 inhibitor and RT followed PD‐1/PD‐L1 inhibitor therapy. This result is consistent with the theory established by preclinical studies, which proposes that RT upregulates PD‐L1 expression through DNA double‐strand breaks and increased CD8+T cell infiltration. 46 , 47 Higher PD‐L1 expression was shown to improve the efficacy of PD‐1/PD‐L1 inhibitors and increase CD8+T cell infiltration. Similar findings have also been reported in association with other diseases and particle therapy for cancer treatment. A study performed by Iijima et al. 48 reported that carbon ion radiotherapy (CIRT) can upregulate PD‐L1 expression in adenocarcinoma of the uterine cervix. Moreover PD‐L1 expression in adenocarcinoma of the uterine cervix after CIRT was favorably associated with PFS. However, the time interval between RT and ICI administration should not be prolonged. The PACIFIC study showed that the OS benefit was significantly greater in patients who were administered immunotherapy within 14 days after concurrent chemoradiotherapy than in patients who were administered immunotherapy after 14–42 days. 5
Subgroup analyses also revealed that combination therapy using SBRT or SRS and PD‐1/PD‐L1 inhibitors may be more effective than combination therapy using conventional RT and PD‐1/PD‐L1 inhibitors in patients with advanced NSCLC. The PEMBRO‐RT (a phase II RCT) reported that the long‐term OS benefit associated with SBRT was observed only in the PD‐L1‐negative subgroup, 9 which indicates that RT, particularly SBRT, can alter PD‐L1 expression, thereby improving the efficacy of ICIs. The combination of immunotherapy and stereotactic ablative radiotherapy is referred to as ISABR. 49 Notably, in addition to activation of the immune system, RT may cause lymphopenia, which consequently affects the efficacy of ICI therapy. Reportedly, the main factors that affect radiation‐induced lymphopenia are multicourse RT, multisite irradiation, high‐dose RT, and variations in the RT techniques utilized (SBRT tends to reduce this risk 10 ). The favorable results of combination therapy using SBRT or SRS and PD‐1/PD‐L1 inhibitors can be attributed to the aforementioned advantage offered by SBRT. SBRT and particle beam radiotherapy would be useful to reduce the volume of normal tissue irradiation and also activate the immune system and would therefore be a better radiotherapeutic choice when combined with PD‐1/PD‐L1 inhibitors. Preclinical studies have investigated the effects of low‐dose irradiation or precision radiotherapy on the immune system, 50 , 51 and this subject will be the focus of future research.
Our results prove that combination therapy using PD‐1/PD‐L1 inhibitors and RT did not increase the rate of serious AEs (≥grade 3), although rates of mild (grade 1–2) pneumonitis were higher in patients who received combination therapy comprising PD‐1/PD‐L1 inhibitors and RT. Radiation or immune pneumonitis associated with combination therapy using RT and ICIs, particularly thoracic RT combined with ICIs, has attracted particular attention. Our subgroup analyses revealed that the incidence of pneumonitis was higher in those who underwent thoracic RT than in those who received RT for metastatic lesions. Previous studies have reported that the higher incidence rate of pneumonitis may be associated with the race, type of histopathological presentation, performance score (PS), and dose‐volume indices of the lungs. Multivariate analysis of the data obtained in the PACIFIC study showed that among the patients treated with durvalumab, Asian patients with non‐squamous tumors and poor PS were more likely to develop pneumonitis. 52 With regard to patients treated with chemoradiotherapy combined with ICIs, the management of lung volume receiving ≥20 Gy (V20) may appear relatively more important; however, it is unclear whether further randomized studies with larger sample sizes are necessary to investigate low‐dose effects such as V5 and V10 on the lungs. 29 Our study results concur with those of other studies; mild pneumonitis did not affect the primary outcome such as OS. Exploratory analysis of the PACIFIC study reported at the 2019 European Society for Medical Oncology (ESMO) meeting 53 showed pneumonitis did not affect treatment completion with durvalumab. No significant difference was observed in the percentage of patients who completed 12‐month treatment with durvalumab between patients with or without pneumonitis (49.5% vs 48.1%), and the incidence of grade 3–4 AEs and AE‐induced mortality rates were not associated with pneumonitis in patients treated with durvalumab. Moreover, although pneumonitis was more common in patients in the durvalumab group, the mortality rate of patients with pneumonitis in the durvalumab group was lower than that of patients with pneumonitis in the placebo group, and pneumonitis did not affect the survival benefit conferred by consolidation therapy using durvalumab. AEs associated with combination therapy using RT and ICIs reported by the U.S. Food and Drug Administration were discussed at the 2020 American Society of Clinical Oncology (ASCO) meeting. 54 Analysis of pooled data from 66 prospective trials of ICIs (which included 25,836 patients) showed that the incidence of hematological toxicity and pneumonitis associated with combined radio‐immunotherapy may be slightly higher than that of RT alone, whereas the incidence of colitis, hepatitis, and myocarditis may be slightly lower, although these differences were statistically nonsignificant. We observed that some of the included single‐arm studies reported a relatively high incidence of AEs or mild pneumonitis. 29 , 31 , 33 , 36 The method of administration of combined radio‐immunotherapy 33 and the small sample size of some of the included studies (which is likely to have introduced a risk of bias) could have contributed to these results. Additionally, the AEs observed in the included single‐arm studies were similar to those in the PACIFIC study. It is reasonable to conclude that the safety profile of combination therapy using PD‐1/PD‐L1 inhibitors and RT is satisfactory.
Interestingly, multiple studies have suggested that multisystem immune‐related adverse events (iRAEs), such as immune pneumonitis, thyroiditis, and dermatitis, may prolong the PFS and OS or increase the ORR and reduce the progressive disease rate in patients with NSCLC treated with ICIs, 28 , 33 , 36 , 55 , 56 , 57 which suggests that iRAEs may serve as a marker of immune system activation. The role of multisystem iRAEs as possible predictive markers of the response to ICIs should be investigated in future studies. 58 Our results also show this tendency. Although we observed a high incidence of mild pneumonitis, the long‐term survival and tumor response rates were significantly improved. However, achieving a balance between safety and efficacy of combined radio‐immunotherapy is a concern that should be addressed by future research.
Our subgroup analyses revealed that the incidence of mild pneumonitis was higher in patients who received PD‐L1 inhibitors than in those who received PD‐1 inhibitors; however, no significant intergroup difference was observed (Cross p = 0.180). Two meta‐analyses performed in patients with NSCLC showed a higher incidence of pneumonitis in patients treated with PD‐1 inhibitors than in those treated with PD‐L1 inhibitors. 59 , 60 A possible mechanism that could explain this finding is the fact that PD‐1 inhibitors can block PD‐1 binding to PD‐L2 leading to increased PD‐L2 binding to the receptor repulsive guidance molecule b, resulting in local T‐cell clonal expansion, which breaks the balance of respiratory tolerance and consequently increases the risk of pneumonitis. In our view, this difference in outcomes may be secondary to the design limitations of NRCTs 21 (some of the included studies were small‐size studies) and the differences in treatment modalities, which may introduce an element of bias. No AEs were observed in the sequence/concurrent subgroup or the SBRT/conventional RT subgroup.
The 2020 ASCO meeting also reported the efficacy of combination therapy using RT and pembrolizumab in patients with metastatic NSCLC. The results of the pooled analysis of two randomized studies suggested that RT combined with ICIs could significantly improve the ORRs of lesions in the radiation field and significantly improve PFS. Furthermore, the partial remission rate was significantly higher in patients with metastatic NSCLC treated with SBRT than in patients treated with conventional RT 61 ; this finding was similar to our results. Several clinical trials of radiochemotherapy combined with ICIs are ongoing 62 ; the complete texts of these studies are unavailable and were not included in this meta‐analysis. We will update this systematic review and meta‐analysis in the future.
Following are the strengths of this systematic review and meta‐analysis: (a) We analyzed the sequence of administration of RT and PD‐1/PD‐L1 inhibitors and observed that RT administered before PD‐1/PD‐L1 inhibitors may be beneficial and that this approach could be useful in real‐world clinical practice in the future. (b) We investigated the effect of RT techniques such as SBRT or SRS; in our opinion, improvements in RT techniques and precision are important factors that would determine the efficacy of combination therapy. Knowledge regarding the aforementioned advantages of SBRT or SRS might change the RT dose fractionation in combination therapy using RT and ICIs. (c) To our knowledge, this is the first study to assess the efficacy and safety profile of combination therapy using PD‐1/PD‐L1 inhibitors and RT in patients with NSCLC.
Following are the limitations of this study: (a) The study included only 20 relevant RCTs; the small sample size is a drawback of this research. (b) We selected both RCTs and NRCTs in this study. Although subgroup analyses were performed, we were unable to eliminate the effects of the NRCT design on the results. (c) Most studies did not use blinding or random allocation. (d) Although we intended to perform subgroup analyses for smoking status, the dose of RT and ICIs, PD‐L1 status, oncogene driver mutation status, and different irradiated sites, current data do not support such analyses. (e) The chemotherapeutic regimens and the number of cycles administered were unclear; therefore, despite subgroup analyses, we could not eliminate the effect of chemotherapy.
5. CONCLUSION
Combination therapy using PD‐1/PD‐L1 inhibitors and RT for the treatment of advanced NSCLC may improve OS, PFS, and tumor response rates without an increase in ≥grade 3 AEs. However, this combination therapy increased the incidence of mild (grade 1–2) pneumonitis. The sequence of administration of PD‐1/PD‐L1 inhibitors after RT and precision radiotherapy techniques such as SBRT may offer greater benefit in patients who receive this combination therapy. Further large‐scale RCTs are warranted confirm these results.
6. DECLARATION
The data used to support the findings of this study are available from the corresponding author upon request. No ethic approval was required as all data originated from previously published studies.
CONFLICTS OF INTEREST
The authors have no conflict of interest.
AUTHORS’ CONTRIBUTIONS
Xiaohu Wang and Bing Lu contributed to choose research directions.Yichao Geng finished the manuscript. Qiuning Zhang and Zheng Li assessed the level of evidence. Ruifeng Liu and Hongtao Luo searched electronic database. Shuangwu Feng, Chengcheng Li, Lina Wang, Xueshan Zhao and Zhen Yang contributed to screened literatures. All authors read and approved the final manuscript.
Supporting information
Supplement 1
Supplement 2
ACKNOWLEDGEMENT
We thank all authors for their contributions to this study.
Funding information
The article were supported by the Lanzhou Innovation and Entrepreneurship Talent Project (award number: 2017‐RC‐23/2020‐RC‐113) and the Science and Technology Plan Project of Chengguan District, Lanzhou (award number: no. 2020‐2‐2‐5).
DATA AVAILABILITY STATEMENT
I confirm that my article contains a Data Availability Statement even if no data is available (list of sample statements) unless my article type does not require one. I confirm that I have included a citation for available data in my references section, unless my article type is exempt.
REFERENCES
- 1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70(1):7‐30. [DOI] [PubMed] [Google Scholar]
- 2. Su S, Li T, Lu B, et al. Three‐dimensional radiation therapy to the primary tumor with concurrent chemotherapy in patients with stage IV non‐small cell lung cancer: results of a multicenter phase 2 study from PPRA‐RTOG, China. Int J Radiat Oncol Biol Phys. 2015;93(4):769‐777. [DOI] [PubMed] [Google Scholar]
- 3. Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science. 2018;359(6382):1350‐1355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Johnson CB, Win SY. Combination therapy with PD‐1/PD‐L1 blockade: an overview of ongoing clinical trials. OncoImmunology. 2018;7(4):e1408744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Gray JE, Villegas A, Daniel D, et al. Three‐year overall survival with durvalumab after chemoradiotherapy in stage III NSCLC—update from PACIFIC. J Thorac Oncol. 2020;15(2):288‐293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Shaverdian N, Lisberg AE, Bornazyan K, et al. Previous radiotherapy and the clinical activity and toxicity of pembrolizumab in the treatment of non‐small‐cell lung cancer: a secondary analysis of the KEYNOTE‐001 phase 1 trial. Lancet Oncol. 2017;18(7):895‐903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Fiorica F, Belluomini L, Stefanelli A, et al. Immune checkpoint inhibitor nivolumab and radiotherapy in pretreated lung cancer patients: efficacy and safety of combination. Am J Clin Oncol. 2018. [DOI] [PubMed] [Google Scholar]
- 8. Ratnayake G, Shanker M, Roberts K, et al. Prior or concurrent radiotherapy and nivolumab immunotherapy in non‐small cell lung cancer. Asia Pac J Clin Oncol. 2020;16(1):56‐62. [DOI] [PubMed] [Google Scholar]
- 9. Theelen WSME, Peulen HMU, Lalezari F, et al. Effect of pembrolizumab after stereotactic body radiotherapy vs pembrolizumab alone on tumor response in patients with advanced non‐small cell lung cancer: results of the PEMBRO‐RT phase 2 randomized clinical trial. JAMA Oncol. 2019;5(9):1276‐1282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Cho Y, Park S, Byun HK, et al. Impact of treatment‐related lymphopenia on immunotherapy for advanced non‐small cell lung cancer. Int J Radiat Oncol Biol Phys. 2019;105(5):1065‐1073. [DOI] [PubMed] [Google Scholar]
- 11. Bang A, Wilhite TJ, Pike LRG, et al. Multicenter evaluation of the tolerability of combined treatment with PD‐1 and CTLA‐4 immune checkpoint inhibitors and palliative radiation therapy. Int J Radiat Oncol Biol Phys. 2017;98(2):344‐351. [DOI] [PubMed] [Google Scholar]
- 12. Li M, Gan L, Song A, Xue J, Lu Y. Rethinking pulmonary toxicity in advanced non‐small cell lung cancer in the era of combining anti‐PD‐1/PD‐L1 therapy with thoracic radiotherapy. Biochim Biophys Acta Rev Cancer. 2019;1871(2):323‐330. [DOI] [PubMed] [Google Scholar]
- 13. Xiu‐xia L, Ya Z, Yao‐long C, Ke‐hu Y, Zong‐jiu Z. The reporting characteristics and methodological quality of Cochrane reviews about health policy research. Health Policy. 2015;119(4):503‐510. [DOI] [PubMed] [Google Scholar]
- 14. Yao L, Sun R, Chen Y‐L, et al. The quality of evidence in Chinese meta‐analyses needs to be improved. J Clin Epidemiol. 2016;74:73‐79. [DOI] [PubMed] [Google Scholar]
- 15. Tian J, Zhang J, Ge L, Yang K, Song F. The methodological and reporting quality of systematic reviews from China and the USA are similar. J Clin Epidemiol. 2017;85:50‐58. [DOI] [PubMed] [Google Scholar]
- 16. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta‐analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Ge L, Tian J‐H, Li Y‐N, et al. Association between prospective registration and overall reporting and methodological quality of systematic reviews: a meta‐epidemiological study. J Clin Epidemiol. 2018;93:45‐55. [DOI] [PubMed] [Google Scholar]
- 18. Wang X, Chen Y, Yao L, et al. Reporting of declarations and conflicts of interest in WHO guidelines can be further improved. J Clin Epidemiol. 2018;98:1‐8. [DOI] [PubMed] [Google Scholar]
- 19. Pieper D, Buechter RB, Li L, Prediger B, Eikermann M. Systematic review found AMSTAR, but not R(evised)‐AMSTAR, to have good measurement properties. J Clin Epidemiol. 2015;68(5):574‐583. [DOI] [PubMed] [Google Scholar]
- 20. Yan P, Yao L, Li H, et al. The methodological quality of robotic surgical meta‐analyses needed to be improved: a cross‐sectional study. J Clin Epidemiol. 2019;109:20‐29. [DOI] [PubMed] [Google Scholar]
- 21. Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA. Cochrane Handbook for Systematic Reviews of Interventions version 6.0 (updated July 2019). Cochrane. 2019. [DOI] [PMC free article] [PubMed]
- 22. Moga C, Guo B, Schopflocher D, Harstall C. Development of a quality appraisal tool for case series studies using a modified Delphi technique. 2012.
- 23. Singh C, Qian JM, Yu JB, Chiang VL. Local tumor response and survival outcomes after combined stereotactic radiosurgery and immunotherapy in non‐small cell lung cancer with brain metastases. J Neurosurg. 2020;132(2):512‐517. [DOI] [PubMed] [Google Scholar]
- 24. Fukui T, Hosotani S, Soda I, et al. Current status and progress of concurrent chemoradiotherapy in patients with locally advanced non‐small cell lung cancer prior to the approval of durvalumab. Thorac Cancer. 2020;11(4):1005‐1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Yamaguchi OU, Kaira K, Hashimoto K, et al. Radiotherapy is an independent prognostic marker of favorable prognosis in non‐small cell lung cancer patients after treatment with the immune checkpoint inhibitor, nivolumab. Thorac Cancer. 2019;10(4):992‐1000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Shepard MJ, Xu Z, Donahue J, et al. Stereotactic radiosurgery with and without checkpoint inhibition for patients with metastatic non‐small cell lung cancer to the brain: a matched cohort study. J Neurosurg. 2019;1‐8. [DOI] [PubMed] [Google Scholar]
- 27. Hubbeling HG, Schapira EF, Horick NK, et al. Safety of combined PD‐1 pathway inhibition and intracranial radiation therapy in non‐small cell lung cancer. J Thorac Oncol. 2018;13(4):550‐558. [DOI] [PubMed] [Google Scholar]
- 28. Tamiya A, Tamiya M, Nakahama K, et al. Correlation of radiation pneumonitis history before nivolumab with onset of interstitial lung disease and progression‐free survival of patients with pre‐treated advanced non‐small cell lung cancer. Anticancer Res. 2017;37(9):5199‐5205. [DOI] [PubMed] [Google Scholar]
- 29. Miura YU, Mouri A, Kaira K, et al. Chemoradiotherapy followed by durvalumab in patients with unresectable advanced non‐small cell lung cancer: management of adverse events. Thorac Cancer. 2020;11(5):1280‐1287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Jabbour SK, Berman AT, Decker RH, et al. Phase 1 trial of pembrolizumab administered concurrently with chemoradiotherapy for locally advanced non‐small cell lung cancer: a nonrandomized controlled trial. JAMA Oncology. 2020;6(6):848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Chu C‐H, Chiu T‐H, Wang C‐C, et al. Consolidation treatment of durvalumab after chemoradiation in real‐world patients with stage III unresectable non‐small cell lung cancer. Thorac Cancer. 2020;11(6):1541‐1549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Amino Y, Kitazono S, Uematsu S, et al. Efficacy of anti‐PD‐1 therapy for recurrence after chemoradiotherapy in locally advanced NSC LC. Int J Clin Oncol. 2020;25(1):67‐73. [DOI] [PubMed] [Google Scholar]
- 33. Qin A, Rengan R, Lee S, et al. A pilot study of atezolizumab plus hypofractionated image‐guided radiotherapy for the treatment of advanced non‐small cell lung cancer. Int J Radiat Oncol Biol Phys. 2020;108:170‐177. [DOI] [PubMed] [Google Scholar]
- 34. Miyamoto S, Nomura R, Sato K, et al. Nivolumab and stereotactic radiation therapy for the treatment of patients with Stage IV non‐small‐cell lung cancer. Jpn J Clin Oncol. 2019;49(2):160‐164. [DOI] [PubMed] [Google Scholar]
- 35. Schapira E, Hubbeling H, Yeap BY, et al. Improved overall survival and locoregional disease control with concurrent PD‐1 pathway inhibitors and stereotactic radiosurgery for lung cancer patients with brain metastases. Int J Radiat Oncol Biol Phys. 2018;101(3):624‐629. [DOI] [PubMed] [Google Scholar]
- 36. Lesueur P, Escande A, Thariat J, et al. Safety of combined PD‐1 pathway inhibition and radiation therapy for non‐small‐cell lung cancer: a multicentric retrospective study from the GFPC. Cancer Med. 2018;7(11):5505‐5513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Ahmed KA, Kim S, Arrington J, et al. Outcomes targeting the PD‐1/PD‐L1 axis in conjunction with stereotactic radiation for patients with non‐small cell lung cancer brain metastases. J Neurooncol. 2017;133(2):331‐338. [DOI] [PubMed] [Google Scholar]
- 38. Galluzzi L, Buqué A, Kepp O, Zitvogel L, Kroemer G. Immunogenic cell death in cancer and infectious disease. Nat Rev Immunol. 2017;17(2):97‐111. [DOI] [PubMed] [Google Scholar]
- 39. Durante M, Reppingen N, Held KD. Immunologically augmented cancer treatment using modern radiotherapy. Trends Mol Med. 2013;19(9):565‐582. [DOI] [PubMed] [Google Scholar]
- 40. Sukkurwala AQ, Adjemian S, Senovilla L, et al. Screening of novel immunogenic cell death inducers within the NCI Mechanistic Diversity Set. OncoImmunology. 2014;3:e28473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Reits EA, Hodge JW, Herberts CA, et al. Radiation modulates the peptide repertoire, enhances MHC class I expression, and induces successful antitumor immunotherapy. J Exp Med. 2006;203(5):1259‐1271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Matsumura S, Wang B, Kawashima N, et al. Radiation‐induced CXCL16 release by breast cancer cells attracts effector T cells. J Immunol. 2008;181(5):3099‐3107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Vanpouille‐Box C, Alard A, Aryankalayil MJ, et al. DNA exonuclease Trex1 regulates radiotherapy‐induced tumour immunogenicity. Nat Commun. 2017;8:15618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Tian L, Goldstein A, Wang H, et al. Mutual regulation of tumour vessel normalization and immunostimulatory reprogramming. Nature. 2017;544(7649):250‐254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Reynders K, Illidge T, Siva S, Chang JY, De Ruysscher D. The abscopal effect of local radiotherapy: using immunotherapy to make a rare event clinically relevant. Cancer Treat Rev. 2015;41(6):503‐510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Sato H, Niimi A, Yasuhara T, et al. DNA double‐strand break repair pathway regulates PD‐L1 expression in cancer cells. Nat Commun. 2017;8(1):1751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Choe E‐A, Cha YJ, Kim J‐H, et al. Dynamic changes in PD‐L1 expression and CD8+ T cell infiltration in non‐small cell lung cancer following chemoradiation therapy. Lung Cancer. 2019;136:30‐36. [DOI] [PubMed] [Google Scholar]
- 48. Iijima M, Okonogi N, Nakajima NI, et al. Significance of PD‐L1 expression in carbon‐ion radiotherapy for uterine cervical adeno/adenosquamous carcinoma. J Gynecol Oncol. 2020;31(2):e19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Bernstein MB, Krishnan S, Hodge JW, Chang JY. Immunotherapy and stereotactic ablative radiotherapy (ISABR): a curative approach? Nat Rev Clin Oncol. 2016;13(8):516‐524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Klug F, Prakash H, Huber P, et al. Low‐dose irradiation programs macrophage differentiation to an iNOS⁺/M1 phenotype that orchestrates effective T cell immunotherapy. Cancer Cell. 2013;24(5):589‐602. [DOI] [PubMed] [Google Scholar]
- 51. Vanpouille‐Box C, Formenti SC, Demaria S. Toward precision radiotherapy for use with immune checkpoint blockers. Clin Cancer Res. 2018;24(2):259‐265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Antonia SJ, Villegas A, Daniel D, et al. Durvalumab after chemoradiotherapy in stage III non‐small‐cell lung cancer. N Engl J Med. 2017;377(20):1919‐1929. [DOI] [PubMed] [Google Scholar]
- 53. Vansteenkiste JF, Naidoo J, Faivre‐Finn C, et al. 1459PDEfficacy of durvalumab in patients with stage III NSCLC who experience pneumonitis (PACIFIC). Ann Oncol. 2019;30:v592‐v593. 10.1093/annonc/mdz259.002. [DOI] [Google Scholar]
- 54. Anscher MS, Arora S, Weinstock C, et al. Impact of radiotherapy on risk of adverse events in patients receiving immunotherapy: a U.S. Food and Drug Administration pooled analysis. J Clin Oncol. 2020;38:3018. [Google Scholar]
- 55. Shankar B, Zhang J, Naqash AR, et al. Multisystem immune‐related adverse events associated with immune checkpoint inhibitors for treatment of non‐small cell lung cancer. JAMA Oncol. 2020;6(12):1952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Hwang WL, Niemierko A, Hwang KL, et al. Clinical outcomes in patients with metastatic lung cancer treated with PD‐1/PD‐L1 inhibitors and thoracic radiotherapy. JAMA Oncol. 2018;4(2):253‐255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Lisberg A, Tucker DA, Goldman JW, et al. Treatment‐related adverse events predict improved clinical outcome in NSCLC patients on KEYNOTE‐001 at a single center. Cancer Immunol Res. 2018;6(3):288‐294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Komiya K, Nakamura T, Hirakawa H, et al. irAE, possible predictive markers for immune checkpoint inhibitors. J Thorac Oncol. 2018;13(10):S479‐S480. [Google Scholar]
- 59. Pillai RN, Behera M, Owonikoko TK, et al. Comparison of the toxicity profile of PD‐1 versus PD‐L1 inhibitors in non‐small cell lung cancer: a systematic analysis of the literature. Cancer. 2018;124(2):271‐277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Khunger M, Rakshit S, Pasupuleti V, et al. Incidence of pneumonitis with use of programmed death 1 and programmed death‐ligand 1 inhibitors in non‐small cell lung cancer: a systematic review and meta‐analysis of trials. Chest. 2017;152(2):271‐281. [DOI] [PubMed] [Google Scholar]
- 61. Welsh JW, Chen D, Baas P, et al. Radiotherapy to augment pembrolizumab responses and outcomes in metastatic non‐small cell lung cancer: Pooled analysis of two randomized trials. J Clin Oncol. 2020;38(15_suppl):9548. 10.1200/JCO.2020.38.15_suppl.9548. [DOI] [Google Scholar]
- 62. Cushman TR, Caetano MS, Welsh JW, Verma V. Overview of ongoing clinical trials investigating combined radiotherapy and immunotherapy. Immunotherapy. 2018;10(10):851. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplement 1
Supplement 2
Data Availability Statement
I confirm that my article contains a Data Availability Statement even if no data is available (list of sample statements) unless my article type does not require one. I confirm that I have included a citation for available data in my references section, unless my article type is exempt.


