Abstract
COVID-19 is an infectious disease caused by the SARS-CoV-2 virus. Most people with COVID-19 have a mild to moderate respiratory illness; others experience severe illness, such as COVID-19 pneumonia. The first and most accessible diagnostic information is from symptoms and signs from clinical examination. Infected patients present with a variety of manifestations. Formal diagnosis requires laboratory analysis of nose and throat samples, or imaging tests like CT scans. Emerging data suggest that coronavirus disease 2019 (COVID-19) has extrapulmonary manifestations. Sometimes these extra-respiratory manifestations may be the initial or only symptom of COVID-19, prior to fever or respiratory manifestations. In summary, our concise review shows that there is a wide range of symptoms that can be presented by COVID-19 patients. Extra-respiratory manifestations of SARS-CoV-2 infection have recently been observed in the rapidly increasing number of COVID-19 cases. Considering the broad spectrum of clinical manifestations and the increasing worldwide burden of the disease, there is an urgent need to rapidly scale up the diagnostic capacity to detect COVID-19 and its complications. (www.actabiomedica.it)
Keywords: SARS-CoV-2 infection, COVID-19, signs and symptoms, extra-respiratory manifestations
Background
Coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has become a global health threat, infecting 27, 150, 797 cases 1, 844, 863 people and resulting in 889, 256 deaths 117, 021 deaths at the time of the last writing (September 7th, 2020). This new type of respiratory illness is characterized by rapid human-to-human transmission, having achieved pandemic spread (1). There are currently no therapeutics or vaccines available and no pre-existing immunity in the population.
Because COVID-19 is a new disease, our awareness and knowledge are gradually increasing based on ongoing research findings and clinical practice experience. A significant amount of collective work has been done to understand the prevention and treatment strategies of COVID-19. The disease has posed unparalleled challenges for health care communities, the general population, and in particular, for patients suffering from chronic diseases (such as: thalassemia and sickle cell disease) (2).
A wealth of data has been generated since its emergence in December 2019, and it is vital for clinicians to keep up with this data from across the world at a time of uncertainty and constantly evolving guidelines and clinical practice. Increasing knowledge about COVID-19 literature will aid in earlier recognition and more effective therapy.
Here, we provide a brief overview of recent knowledge, mainly focused on extrapulmonary manifestations reported in the literature in adult patients.
Search strategy
A systematic English-language literature search was carried out, from 1 January 2019 to 31 August 2020, using the main online databases (PubMed, Google Scholar, and MEDLINE) with the following keywords: ‘COVID-19’, ‘2019-nCoV’, ‘coronavirus’ and extrapulmonary manifestations. In addition, the references contained in the downloaded documents were examined for other sources of information that would be pertinent to our review. The literature search was updated until September 2020.
Structure of SARS-CoV-2
SARS-CoV-2 is an enveloped, positive-sense single-stranded RNA virus, from beta-coronavirus genera of coronaviridae family responsible for the 2019 coronavirus disease (COVID-19) pandemic. SARS-CoV-2 contains 29891 nucleotides encoding 9889 amino acids and four structural proteins, namely spike (S), envelope (E), membrane (M), and nucleocapsid (N) proteins of which S protein mediates the viral entry into host cells (Figure 1). The spikes (S) and the nucleocapsid (N) are the main proteins (antigens) which trigger an antibody response in humans. SARS-CoV-2 also contains a variable number of open reading frames (ORF), and these encode16 non-structural proteins, the remaining ORFs encode structural proteins (3-5).
Figure 1.
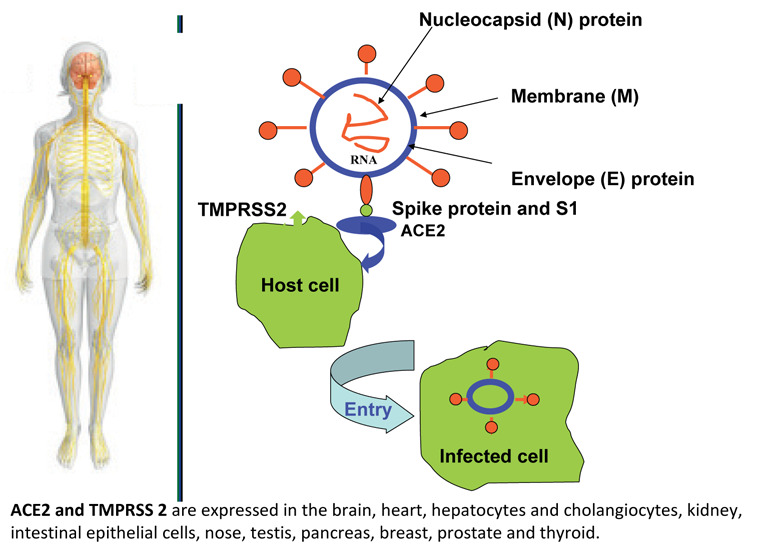
Structure of SARS-CoV-2. (A) There are four structural proteins as follows: spike (S) surface glycoprotein; membrane (M) protein; nucleocapsid (N) protein and envelope (E) protein. SARS-CoV-2 infected the host cells by the spike protein of the virus and the functions of ACE2 and TMPRSS2 in host cells.
SARS-CoV-2 and influenza primarily affect the respiratory system. SARS-CoV-2 gains access to the host cells using its surface spike (S) protein to bind human angiotensin-converting enzyme 2 receptor on host cells. The viral entry triggers a host immune response with a massive release of cytokines thought to be responsible for organ dysfunctions (6).
Pathophysiology
The pathogenesis of COVID-19 is largely unknown, but it may mimic SARS to some extent. SARS-CoV-2 attacks the alveolar epithelial cells via angiotensin-converting enzyme 2 (ACE2) via S-protein. ACE-2 is found on apical membranes of nasal, oral, nasopharyngeal and oropharyngeal mucosal epithelium, alveolar epithelium, endothelial cells of blood vessels and heart, kidneys, testes, brain, intestine, and colon, and liver (7,8).
ACE-2 has been proven to be a major receptor for SARS-CoV-2 and could mediate virus entry into cells. And transmembrane protease serine 2 (TMPRSS2) could cleave the spike (S) protein of SARS-CoV-2, which facilitates the fusion of SARS-CoV-2 and cellular membranes. The mRNA expressions of both ACE2 and TMPRSS2 were observed in the heart, digestive tract, liver, kidney, brain and other organs. SARS-CoV-2 may have a capacity to infect extrapulmonary organs due to the expressions of ACE2 and TMPRSS2 in the cells and tissues of these organs (9) (Figure 1).
Spike proteins on the exterior of SARS-CoV-2 anchor the virus to ACE-2 receptors on cells in the lower respiratory tract leading to endothelial and microvascular dysfunction, alveolar exudative inflammation, interstitial inflammation and fibrosis, and focal bleeding which causes severe respiratory manifestation of the disease (7).
ACE-2 is a counter-regulatory enzyme of renin-angiotensin-system which acts by converting angiotensin-2 to Ang-1–7 form. In healthy state, ACE-2 activity maintains homeostasis between angiotensin-2 (vasoconstriction, inflammation, fibrosis and proliferation) and Ang-1–7 pathways (vasodilatation, anti-apoptotic, anti-fibrosis, and anti-proliferation). After entering pneumocytes, SARS-CoV-2 downregulates ACE-2 expression, decreasing angiotensin-2 metabolism. Elevated angiotensin-2 increases pulmonary vascular permeability and inflammation, hence worsening of lung injur. Angiotensin-2 levels have been found to be increased in COVID-19 patients compared to healthy adults (7-9).
Routes of transmission and period of incubation
The disease is said to be transmitted through droplets from human saliva, eyes, and nose. A recent study found that SARS-CoV-2 lasts in aerosols for up to 3 hours and remains detectable for up to 72 hours on plastic and stainless-steel surfaces, 24 hours on cardboard, and 4 hours on copper. Fecal-oral transmission may also be possible but has not been verified to be clinically important (10,11).
Individuals of any age are susceptible to infection, including neonates and pregnant women.
The mean incubation period of SARS-CoV-2 is estimated to be 3–7 days (range, 2–14 days) (12).
This period is dependent on the age of the patient and status of the patient’s immune system. It was shorter among patients >70-years old compared with those under the age of 70 years. Figure 2 illustrates the immune response to viral infection. The SARS-CoV-2 virus persists in fecal samples for an average of 27 days, which is 10 days longer than its persistence in respiratory tract samples (13).
Figure 2.
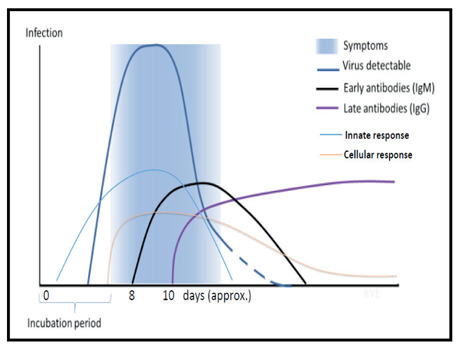
The immune response to viral infection [From: WHO. COVID-19 immunity and clinical manifestations. CORONAVIRUS (COVID-19) UPDATE NO. 24. May 1st, 2020]
Clinical features and Risk factors
It has been proposed that COVID-19 progresses through several stages in its disease course. The first stage is viral infection during which constitutional symptoms, such as fever and cough, predominate. The second stage is characterized by direct viral cytotoxic effects, particular those in the respiratory tract, leading to respiratory failure and potentially acute respiratory distress syndrome. The third and final stage is thought to be mediated by a hyperinflammatory response to the virus causing systemic effects (14).
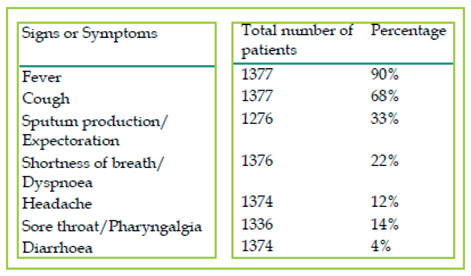
The symptoms of SARS-CoV-2 infection seem to be non-specific, as far as clinical manifestations are being concerned. They may be very similar to influenza (Table 1). Gastrointestinal symptoms such as nausea and vomiting (5%) and diarrhoea (3.8%) are relatively uncommon (10,12).
Table 1.
Common signs and symptoms of SARS-CoV-2 infected patients from four reports ( From: Zheng J et al. SARS-CoV-2: an Emerging Coronavirus that Causes a Global Threat. Int J Biol Sci. 2020;16:1678-1685, modified)
 |
The symptoms of COVID-19 initially begin with symptoms of fatigue, low-grade intermittent fever of prolonged duration, myalgia, dry cough and shortness of breath, which then either improves with early identification and conservative management or worsens and progresses to dyspnoea and productive cough. The median duration of fever was 12 days (8–13 days) and cough persisted for 19 days (12–23 days) in survivors. The median time to onset of dyspnoea from various cohorts was found to be 6 days following exposure. Serious cases can progress rapidly into acute respiratory distress syndrome (ARDS), septic shock, refractory metabolic acidosis, coagulation dysfunction and others such as acute kidney injury (AKI), acute cardiac injury (arrhythmias, heart failure, MI) (15,16). Complications developed at a median of 9-17 days.
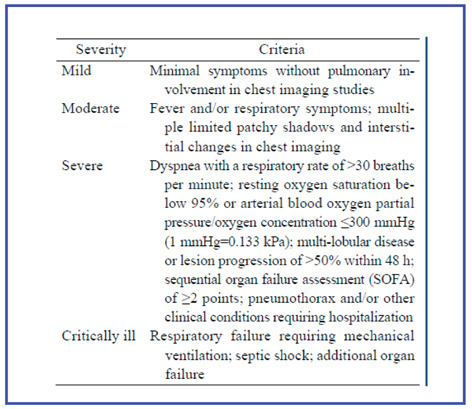
Given the wide clinical spectrum of COVID-19, a key challenge faced by frontline clinical staff is prioritisation of stretched resources. The clinical criteria for assessing the severity of COVID-19 are reported in Table 2. Furthermore, several evidences suggest that severe disease is more likely to take hold in individuals of older age, male sex, and in those with underlying co-morbidities, such as: diabetes, chronic obstructive pulmonary disease (COPD), hypertension, obesity (17).
Table 2.
Criteria for assessing the severity of COVID-19
 |
To explore the clinical and laboratory risk factors associated with disease severity, an univariate and multivariate logistic regression models was used by Liu et al. (18). In the univariate analysis, hypertension, lymphopenia, elevated neutrophil count, lactate dehydrogenase (LDH), C-reactive protein (CRP), and symptoms such as dyspnea, fatigue, and anorexia/lethargy were all associated with severe cases. In the multivariable logistic regression model, the Authors included 90 patients (56 pneumonia and 34 severe pneumonia). Hypertension, increased CRP and lymphocyte count as independent were identified as predictors of severe pneumonia.
Relationship between the ABO Blood Group and the COVID-19 Susceptibility published.
Blood group A was associated with a higher risk for acquiring COVID-19 compared with non-A blood groups, whereas blood group O was associated with a lower risk for the infection compared with non-O blood groups (19).
Laboratorial findings
Lymphopenia is a common finding in patients with COVID-19 infection, and is believed to represent a defective immune response to the virus (20).
Leukocytosis, is noted in a minority of COVID-19 infected patients and appears to herald bacterial infection or superinfection while neutrophilia is an expression of the cytokine storm and hyperinflammatory state which have an important pathogenetic role in COVID-19. Neutrophilia may also indicate superimposed bacterial infection (20). Neutrophil/lymphocyte ratio and peak platelet/lymphocyte ratio may also have prognostic value in determining severe cases (20,21).
Thrombocytopenia is an important indicator of severe disease in COVID-19 patients as well as D-dimer increases and progressive decrease of lymphocytes in peripheral blood. In quite a few patients, increase of liver enzymes, muscle enzymes, and myoglobin are observed (20,21).
Association of Autoimmune Haemolytic Anaemia (AHA) with COVID-19 infection has been rarely reported (22).
On initial presentation a majority of COVID-19 patients have procalcitonin levels in the normal range (20,22). Elevated procalcitonin levels may be seen in sepsis and are particularly associated with septic shock and organ dysfunction requiring intervention. Prothrombin time (PT), an assay used to evaluate the extrinsic and common coagulation pathways, and D-dimer are useful indicators of prognosis and severity of disease in COVID-19 (22).
Severely ill patients may have high levels of cytokine IL6 (>7.4 pg/mL), and tumor necrosis factor (TNF). Increased ferritin level (>300 μg/L) is an indicator of the imminent cytokine storm (20,21).
Early recognition of these parameters may be useful to predict the disease severity, to guide the therapy, and improve the patients’ clinical outcome (Table 3).
Table 3.
Hematologic and laboratory findings in patients with mild/moderate SARS-CoV-2 infection [Mean value and range (%) from 10 differente studies]
| Parameter | Clinical significance | Mild/moderate cases (%) |
| Lymphopenia (<1.5 × 109/L) | Defective host response | 47-54% |
| Leukocytosis ( >10×109/L) | Bacteria superinfection | 19–30% |
| Neutrophilia | Bacterial superinfection, cytokine storm | 1.4 % |
| Thrombocytopenia (<100 x 10 9/L) | Consumptive coagulopathy | 4% |
| Elevated CRP | Severe viral infection, including viremia | 45.5-56.4% |
| Elevated procalcitonin (≥0.25 to <0.5 ng/mL) | Bacterial superinfection | 7% |
| Elevated LDH (>245 U/L) | Pulmonary injury/ multiorgan damage | 63% |
| Prolonged prothrombin time (> 16 s) | Consumptive coagulopathy | 1.7% |
| Prolonged APTT (>37.0 s) | Consumptive coagulopathy | 9.6% |
| Elevated D-dimer (>1 μg/L) and/or FDP | Consumptive coagulopathy | 6%, |
Legend: Abbreviations = CRP-C reactive protein; LDH – lactate dehydrogenase; APTT-activated partial thromboplastin time; FDP-fibrin degradation product.
The three complete blood count findings of poor prognosis are: leukocytosis, thrombocytopenia, and lymphocytopenia. Whether poor prognosis is associated with lymphocytopenia below the reference interval or absolute count is unclear.
Differential diagnosis
On the basis of currently available data, neither absence nor presence of signs or symptoms are accurate enough to rule in or rule out COVID-19 disease. Signs of SARS-CoV-2 infection overlap with other viral infections (viral respiratory illnesses, respiratory syncytial virus, influenza, parainfluenza, adenovirus, and metapneumovirus) and bacterial pneumonia (streptococci, mycoplasma, chlamydia, legionella) that makes the clinical and radiological diagnosis very tricky (9, 12,15). Therefore, in the current epidemic time, when we face an inpatient with fever and/or new onset cough, with or without associated dyspnoea, all efforts should be directed at ruling out aetiologies other than SARS-CoV-2 through history, physical examination and routine laboratory tests. In Table 4 is reported the frequency of symptoms in COVID-19, flu and common cold.
Table 4.
Frequency of symptoms in COVID-19, flu and common cold [From: WHO. COVID-19 immunity and clinical manifestations. CORONAVIRUS (COVID-19) UPDATE NO. 24. May 1st, 2020]
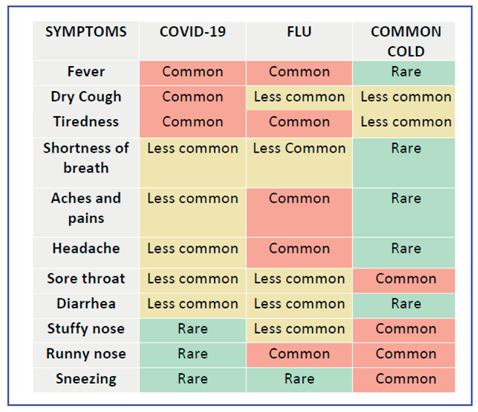 |
Influenza globally affects 5-10% of adults and 20-30% of children annually with most cases occurring during the winter months in the northern (November to April) and southern hemispheres (April to September) with no seasonal pattern in the tropical regions (23,24). Influenza-related respiratory diseases are responsible annually for an estimated 650,000 deaths globally (25).
Confirmation of COVID-19 diagnosis
Confirmation of coronavirus disease 2019 is by reverse transcriptase-polymerized chain reaction from upper airway swabs. A nucleic acid test (RT-PCR test) is currently accepted as the gold standard method to confirm diagnosis. RT-PCR testing is done on nasopharyngeal swabs. The period and type of specimen collected for RT-PCR play an important role in the diagnosis of COVID-19. In most individuals with symptomatic COVID-19 infection, viral RNA in the nasopharyngeal swab becomes detectable as early as day 1 of symptoms and peaks within the first week of symptom onset. The positivity decline by week 3 and subsequently becomes undetectable. In most studies of respiratory virus infections, serial sampling of nasopharyngeal or throat swabs is used for viral load monitoring (26).
Kucirka et al. (27) showed that the rate of false-negative RT-PCR results is highly dependent on the timing of nasopharyngeal sampling is being done: the false negative rate was 100% at 4 days before symptom onset and decreased to 20% 3 days after symptom onset. Another study found a false negative rate of 16.7% for RT-PCR in patients with a clinical suspicion of COVID-19 at initial clinical presentation (28). Reasons for such false-negative RT-PCR can be a viral load below the lower limit of detection of the employed assay, improper sampling of the nasopharyngeal swab, or decreased viral shedding at the anatomic sampling site (29). A potential cause of a lower specificity could be contamination with RNA from sources other than the patient under investigation during the testing process.
When the RT-PCR test result is negative in suspect cases, chest imaging should be considered. The standard image diagnosis tests for pneumonia are chest X-ray (CXR) and computed tomography (CT) scan. The CXR is the primary radiographic exam to evaluate pneumonia, but it is not as precise as the CT scan and has higher misdiagnosis rates. Nevertheless, the CXR is still useful because it is cheaper, faster, expose the patient to less radiation and is more widespread than CT scan (30,31)
Chest CT is highly recommended as the preferred imaging diagnosis method for COVID-19 due to its high density and high spatial resolution. The common CT manifestation of COVID-19 includes multiple segmental ground glass opacities (GGOs) distributed dominantly in extrapulmonary/ subpleural zones and along broncho-vascular bundles with crazy paving sign and interlobular septal thickening and consolidation. Pleural effusion or mediastinal lymphadenopathy is rarely seen (17,32,33).
Other viral pneumonia manifest generally diffuse large patches of GGOs in both lungs with thickening of interlobular septum, which makes it hard to differentiate them from COVID-19 pneumonia in images and clinically. COVID-19 ground-glass opacities are usually peripherally located with the lower lobes being commonly involved, while influenza has a central, peripheral, or random distribution usually affecting the five lobes (17,34) (Table 5).
Table 5.
Imaging characteristics of common causes of pneumonia similar to COVID-19 pneumonia (From: Li B et al. Diagnostic value and key features of computed tomography in Coronavirus Disease 2019. Emerg Microbes Infect. 2020;9:787-793; modified)
| Diseases | High-risk groups | CT imaging findings |
| COVID-19 | Elderly people; People with comorbidities |
Early stage: GGOs, Progressive stage: multiple GGOs, consolidation patches, crazy-pavement pattern Advanced stage: diffuse exudative lesions, white-out lung |
| Influenza pneumonia | Elderly people; Children under 5 years old | Small patch GGOs and consolidation with subpleural and or peribronchial distribution. Bilateral reticulonodular areas of opacity. |
| Mycoplasmal pneumonia | Children, adolescents and young adults | Interstitial infiltration or consolidations of segments or lobes manifested by patchy or fan-shaped infiltration. Thickening of the bronchial wall, centrilobular nodules, consolidations distributed along lobes, segments or subsegments of the lung, and enlargement of mediastinal lymph nodes. |
| Bacterial pneumonia | All ages | Bronchial or lobar pneumonia, bronchial wall thickening, multiple consolidation patches and centrilobular nodules |
Legend: GGO: ground-glass opacity.
Ultrasonography (US) has been employed in the detection of interstitial disorders, consolidation, and effusions (35). US is also a useful tool to support the ventilated patient in terms of assessing diaphragm mobility and supporting recruitment maneuvers (36).
Extrapulmonary manifestations
Although COVID-19 is well known to cause respiratory pathology, it can also result in several extrapulmonary manifestations of gastrointestinal system, cardiovascular system, liver, and kidneys (Figure 3). Key mechanisms that may have a role in the pathophysiology of multi-organ injury secondary to SARS-CoV-2 infection include direct viral toxicity, endothelial cell damage and thromboinflammation, deregulation of the immune response, and deregulation of the renin–angiotensin–aldosterone system (RAAS) (37,38).
Figure 3.
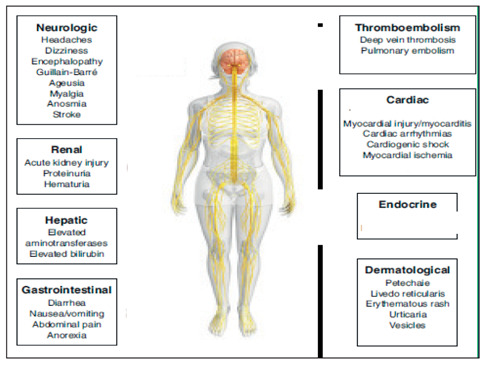
Extrapulmonary manifestations of COVID-19 (From: Aakriti Gupta et al. Nature Medicine. 2020;26:1017–1032; www.nature.com/naturemedicine, modified)
1. Dermatologic manifestations
Skin manifestations are seen in up to 20% of COVID-19 patients and are very heterogeneous. To date, reports have identified 5 main category of skin manifestations: acral lesions, vesicular rashes, urticarial rashes, maculopapular rashes, and livedoid and necrotic lesions. The presentation may vary in different population groups and are based on severity of disease.
The predominant places of dermatologic manifestations are the torso and body’s extremities and the latency period of the cutaneous symptoms vary from 1 to 30 days. However, some of them may emerge before the onset of COVID manifestations. The lesions’ progressive disappearance vary from four days to three weeks (39-41).
Cutaneous manifestations associated with COVID-19 probably reflect the activation of pathogenic pathways by the virus or a response to inflammatory processes, vascular or systemic complications, or even treatments (39-41).
A severe multisystem inflammatory syndrome associated with Kawasaki disease manifestations (MIS-C) has been recently reported in children with signs of recent infection with SARS-CoV-2. However. clinicians caring for adult patients must be aware that not only children but also young adults can be affected by a multisystem inflammatory syndrome with Kawasaki’s disease features associated with COVID-19 (42).
2. Cardiovascular manifestations
COVID-19 can be complicated by cardiac arrhythmias or clinical heart failure with or without associated hemodynamic instability, including shock (43). These cardiac complications can occur precipitously at any point during hospitalization and are increasingly being described as a late complication that can occur after improvements in a patient’s respiratory status (44,45).
The cardiac manifestations of COVID-19 might be related to the adrenergic drive, systemic inflammatory milieu and cytokine-release syndrome caused by SARS-CoV-2, direct viral infection of myocardial and endothelial cells, hypoxia due to respiratory failure, electrolytic imbalances, fluid overload, and side effects of certain COVID-19 medications (46).
Monitoring may include serial cardiac troponin and natriuretic peptides, along with fibrinogen, D-dimer, and inflammatory biomarkers. Management of acute COVID-19 cardiovascular syndrome should involve a multidisciplinary team including intensive care specialists, infectious disease specialists, and cardiologists.
3. Venous thromboembolic events (VTEs)
Patients with moderate and severe COVID-19 illness are more likely to have a hypercoagulable state placing them at high risk for venous thromboembolism (VTE) (47,48). VTE occurs approximately in 25% of severe COVID-19 patients (47).
Careful evaluation of laboratory indices at baseline and during the disease course (platelet count, PT, fibrinogen, D-dimer, antithrombin and protein C activity monitoring) can assist clinicians in formulating a tailored treatment approach and promptly provide intensive care to those who are in greater need (48).
Prophylactic low-molecular-weight heparin (LMWH) has been recommended by the International Society on Thrombosis and Haemostasis (ISTH) and the American Society of Hematology (ASH), but the best effective dosage is uncertain. As per recommendations by the American College of Chest Physicians, in the absence of contraindications, thrombotic prophylaxis is recommended in all moderate and severe COVID-19 patients, while low molecular weight heparin (LMWH) is preferred to oral anticoagulants (49). In patients requiring ICU admission, therapeutic treatment of LMWH can be effective in reducing in-hospital mortality.
4. Gastrointestinal (GI) and hepatic manifestations
The most common GI symptoms reported are diarrhea, nausea, vomiting, and abdominal discomfort, and few COVID-19 case reports have been reporting cases with GI symptoms preceding respiratory symptoms, with some patients only presenting with digestive symptoms in the absence of respiratory symptoms (50-52). Therefore, these uncommon GI presentation presentations can potentially lead to delay in diagnosis of COVID-19 (53).
SARS-CoV-2 infects the GI tract via its viral receptor angiotensin converting enzyme II, which is expressed on enterocytes of the ileum and colon.
An increasing number of COVID-19 patients experience hepatic injury, ranging on a spectrum of mild to severe damage. Most liver injuries are mild and transient, but severe liver damage can also occur in patients with severe COVID-19 and is associated with negative outcomes (54). Patients with chronic liver diseases, especially in those with pre-existing cirrhosis, there is an increased risk of worse outcomes in COVID-19, that have been attributed to their immunocompromised status (55).
The definitive mechanism by which liver injury occurs in COVID-19 patients is unclear. Liver injury in patients with COVID-19 might be due to viral infection in liver cells or due to other causes such as drug induced liver injury and systemic inflammation induced by cytokine storm or pneumonia-associated hypoxia. Furthermore, drug-induced hepatotoxicity may play a role in the elevation of liver enzymes, including medications such as remdesivir (an RNA polymerase inhibitor) and hydroxychloroquine (56).
5. Pancreas injury
In a recent study by Wang et al. (57) examining 52 patients with COVID-19 pneumonia, 17 % of patients experienced pancreatic injury, documented by an increase of amylase or lipase serum levels, in absence of clinical symptoms of pancreatitis.
Mechanisms by which pancreatic injury could occur include the direct cytopathic effects of SARS-CoV-2, or indirect systemic inflammatory and immune-mediated cellular responses, resulting in organ damage or secondary enzyme abnormalities. The ACE2 receptor is also highly expressed in pancreatic islet cells, therefore SARS-CoV-2 infection can theoretically cause islet damage resulting in acute diabetes (58).
6. Neurological manifestations
According to several studies, more than 35% of COVID-19 patients develop neurological symptoms sometimes as the initial presentations of the disease.
The neurological manifestations and complications of COVID-19 can be divided into central (encephalopathy, acute hemorrhagic necrotizing encephalopathy, acute myelitis, cerebrovascular accident, encephalitis, headaches and dizziness), and peripheral (anosmia and chemosensory dysfunction, Guillain Barrè syndrome, skeletal muscle damage) (59,60).
It is still unclear if these complications are directly due to the viral infestation or post-infectious auto-immune reactions, or hypoxic metabolic changes (59,60). Furthermore the hyperactivation of inflammatory factors may disrupt the coagulation system leading to D-dimer and platelet abnormalities, that may provoke the occurrence of cerebrovascular diseases (CVD) such as acute ischemic stroke (61).
6.1. Central nervous system (CNS) manifestations
The most common central nervous system (CNS) symptoms are dizziness (16.8%) and headache (13.1%), other less frequent symptoms included impaired consciousness (7.5%), acute cerebrovascular disease (2.8%), ataxia (0.5%), seizure (0.5%), vision impairment (1.4%) and nerve pain (2.7%). Headache, myalgia, and malaise are common initial neurological symptoms. Altered sensorium, in severe COVID-19, ranges from confusion, delirium, stupor to coma (59,60). The prevalence of neurological signs and symptoms is higher in patients with severe COVID-19 infection, which may be the result of cerebral hypoxia due to respiratory failure (59,60).
6.2. Peripheral nervous system (PNS) manifestations
PNS sign and symptoms of COVID-19 include hyposmia/anosmia, hypogeusia/ageusia, muscle pain, and Guillain-Barre syndrome (GBS). Spinal cord involvement is uncommon (62).
Anosmia and/or ageusia are the most common PNS manifestations of SARS-COV-2. In a multicenter European study among 417 mild-to-moderate COVID-19 patients, 85.6% and 88.0% of patients had olfactory and/or gustatory dysfunctions, respectively. Women were more likely to be affected, and there was an early olfactory recovery rate of 44%, while symptoms could last even 14 days after the resolution of symptoms (63). These symptoms occur suddenly, and usually with fewer nasal symptoms such as nasal obstruction or excessive nasal secretion. Anosmia and ageusia are mostly present in asymptomatic individuals or as the initial presentation of the disease with no other symptoms.
7. Ocular manifestations
Coronaviruses may induce a wide spectrum of ophthalmic manifestations, such as conjunctivitis, anterior uveitis, retinitis, or optic neuritis (64-67). The prevalence of ocular manifestations varies from 2% to 32% (67) and is associated with the severity of the COVID-19 (64-66).
Similarly, to other symptoms not related to the respiratory system, ophthalmic manifestations might appear as the first symptom without any other impairments.
Several mechanisms for the ocular transmission of the virus are proposed with highlight on the nasolacrimal system as a conduit between the eye and the respiratory tract, and the role of the lacrimal gland in hematogenous spread. However, it is likely that the virus does possess an ocular tropism, as do other respiratory viruses. Furthermore, given the high vascularity of conjunctiva, along with the expression of ACE2 on the surface of endothelial cells, it cannot be excluded that the ocular manifestation of SARS-CoV-2 infection may in fact manifest in the form of a local, transient vasculitis (68,69).
8. Renal manifestations
The kidneys are one of the most frequently affected extrapulmonary organs in patients infected with SARS-CoV-2; especially, in those patients who are severely ill (70). Hematuria has been reported in nearly half of patients with COVID-19, and proteinuria has been reported in up to 87% of critically ill patients with COVID-19. The occurrence of acute kidney injury (AKI) among patients with COVID-19 is not consistent across published studies, ranging from 0.1% to 29% (70-74). Recently, Sun et al (75) have reported the occurrence of subclinical AKI as reflected by increased urinary levels of β2-microglobulin, α1-microglobulin, N-acetyl-β-D-glucosaminidase, and retinol-binding protein (ie, all biomarkers of kidney tubular damage) in a sample of 32 confirmed COVID-19 cases without prior chronic kidney disease.
Although the mechanisms for the renal manifestations of COVID-19 are still elusive, a complex multifactorial pathway has been proposed and it includes: (a) direct viral involvement and replication in the kidneys leading to dysfunction; (b) local disruption in renin–angiotensin–aldosterone system (RAAS) homeostasis, and (c) as a result of a systemic inflammatory response “cytokine storm”(70,71,76).
Monitoring of markers of kidney function could help in the identification of patients who are at high risk for worse outcomes (76). However, more work is needed to help us better understand the pathophysiology underlying renal manifestations of COVID-19, to help in the identification of effective management strategies.
9. Reproductive health issues
A recent study using single-cell RNA sequencing found that the ACE2 receptor, a target for SARS-CoV-2 infection, is expressed in germ cells, Leydig cells, and Sertoli cells in the testis (77), suggesting the testis is potentially a tropism site and reservoir for the SARS-CoV-2 virus. Sertoli cells play a critical role in the homeostasis of seminiferous tubules and spermatogenesis and Leydig cells are involved in androgen production.
It has been reported that semen analysis following COVID-19 infection showed a low sperm concentration with low motility for up to three months post-infection (78). This might indicate that the effect of COVID-19 on male fertility might be only transient. Furthermore, a study conducted on 81 male patients with COVID-19 showed low testosterone level, high luteinizing hormone (LH) level and low testosterone/LH ratio indicating a possible viral testicular damage which subsequently affects the function of Leydig cells (79). Therefore, it has been suggested that the male survivors of COVID-19, especially those with reproductive needs, should be examined for testicular function and reproductive function after recovery (80).
An unusual presentation of a male case with severe external genital pain which was suspected to be the first clinical sign of COVID-19. has been reported by Özveri et al. (81)
In conclusion
COVID-19 probably represents the greatest pandemic event in modern human history. Our concise review shows that the disease presents a broad spectrum of clinical signs and symptoms. Extra-respiratory manifestations of SARS-CoV-2 infection have recently been observed in the rapidly increasing number of COVID-19 cases involving of vital organs such as: lungs, heart, gastrointestinal tract, liver, kidneys, central and peripheral system, and blood.
The main mechanism described is the high binding affinity of the virus with the ACE2 receptors that are widely expressed in most human cells.
Commonly, multisystemic involvement is associated with severe disease and might predict worse clinical outcomes and increased mortality. Therefore, a prompt high vigilance among primary care and emergency doctors is very important. Delineating the whole spectrum of symptoms of the disease can help with early diagnosis, prevention of the spread of the disease, and its treatment. In addition, it can help with the prevention of complications that may arise in the long term.
Acknowledgement
This project is supported and Co-Funded by the Erasmus and Programme of the European Union.
Project Title
Hemoglobinopathies for Immigrants. Education, Prevention, Genetic Diagnosis and Treatment Approach.

Declaration of interest
The authors report no conflicts of interest.
References
- 1.European Centre for Disease Prevention and Control. COVID-19 situation update worldwide, as of 7 September 2020. https://www.ecdc.europa.eu/en/geographical-distribution-2019-ncov-cases . [Google Scholar]
- 2.Wu Z, McGoogan JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA. 2020 doi: 10.1001/jama.2020.2648. 10.1001/jama.2020.2648. doi:10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 3.Nikolich-Zugich J, Knox KS, Rios CT, Natt B, Bhattacharya D, Fain MJ. SARS-CoV-2 and COVID-19 in older adults: what we may expect regarding pathogenesis, immune responses, and outcomes. GeroScience. 2020;42:505–514. doi: 10.1007/s11357-020-00186-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chen Y, Liu Q, Guo D. Emerging coronaviruses: Genome structure, replication, and pathogenesis. J Med Virol. 2020 doi: 10.1002/jmv.26234. 10.1002/jmv.26234. doi:10.1002/jmv.26234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Canatan D, De Sanctis V. The impact of MicroRNAs (miRNAs) on the genotype of coronaviruses. Acta Biomed. 2020;91:195–198. doi: 10.23750/abm.v91i2.9534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Du L, He Y, Zhou Y, Liu S, Zheng BJ, Jiang S. The spike protein of SARS-CoV--a target for vaccine and therapeutic development. Nat Rev Microbiol. 2009;7:226–236. doi: 10.1038/nrmicro2090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chen L, Li X, Chen M, Feng Y, Xiong C. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS-CoV-2. Cardiovasc Res. 2020;116:1097–1100. doi: 10.1093/cvr/cvaa078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Di Gennaro F, Pizzol D, Marotta C, et al. Coronavirus Diseases (COVID-19) Current Status and Future Perspectives: A Narrative Review. Int J Environ Res Public Health. 2020;17(8):2690. doi: 10.3390/ijerph17082690. Published 2020 Apr 14 doi:10.3390/ijerph17082690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dong M, Zhang J, Ma X, et al. ACE2, TMPRSS2 distribution and extrapulmonary organ injury in patients with COVID-19. Biomed Pharmacother. 2020 Aug 24;131 doi: 10.1016/j.biopha.2020.110678. 110678. doi: 10.1016/j.biopha.2020.110678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Xiao F, Tang M, Zheng X. Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterology. 2020;158:1831–1833. doi: 10.1053/j.gastro.2020.02.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang W, Xu Y, Gao R. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020;323:1843–1844. doi: 10.1001/jama.2020.3786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yang Y, Lu Q, Liu M, et al. Epidemiological and clinical features of the 2019 novel coronavirus outbreak in China. medRxiv. 2020 https://doi.org/10.1016/j.cmi.2020.02.005 . [Google Scholar]
- 13.Wu Y, Guo C, Tang L, et al. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol Hepatol. doi: 10.1016/S2468-1253(20)30083-2. doi:10.1016/S2468-1253(20)30083-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Atri D, Siddiqi HK, Lang J. COVID-19 for the cardiologist: a current review of the virology, clinical epidemiology, cardiac and other clinical manifestations and potential therapeutic strategies. JACC Basic Transl Sci. 2020;5:518–536. doi: 10.1016/j.jacbts.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Struyf T, Deeks JJ, Dinnes J, et al. Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19 disease. Cochrane Database Syst Rev. 2020;7(7) doi: 10.1002/14651858.CD013665. CD013665. doi:10.1002/14651858.CD013665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zheng Q, Lu Y, Lure F, Jaeger S, Lu P. Clinical and radiological features of novel coronavirus pneumonia. J Xray Sci Technol. 2020;28:391–404. doi: 10.3233/XST-200687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Han W, Quan B, Guo Y, et al. The course of clinical diagnosis and treatment of a case infected with coronavirus disease 2019. J Med Virol. 2020;92:461–463. doi: 10.1002/jmv.25711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liu X, Zhou H, Zhou Y, et al. Risk factors associated with disease severity and length of hospital stay in COVID-19 patients. J Infect. 2020;81:e95–e97. doi: 10.1016/j.jinf.2020.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhao J, Yang Y, Huang H, et al. Relationship between the ABO Blood Group and the COVID-19 Susceptibility Clin Infect Dis. 2020 Aug 4 doi: 10.1093/cid/ciaa1150. ciaa1150. doi: 10.1093/cid/ciaa1150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lippi G, Simundic AM, Plebani M. Potential preanalytical and analytical vulnerabilities in the laboratory diagnosis of coronavirus disease 2019 (COVID-19) Clin Chem Lab Med. 2020;58:1070–1076. doi: 10.1515/cclm-2020-0285. [DOI] [PubMed] [Google Scholar]
- 21.Frater JL, Zini G, d’Onofrio G, Rogers HJ. COVID-19 and the clinical hematology laboratory. Int J Lab Hematol. 2020;(42 Suppl 1):11–18. doi: 10.1111/ijlh.13229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lopez C, Kim J, Pandey A, Huang T, DeLoughery TG. Simultaneous Onset of COVID-19 and Autoimmune Haemolytic Anaemia. Br J Haematol. 2020;190:31–32. doi: 10.1111/bjh.16786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Russell CA, Jones TC, Barr IG, et al. Vaccine. 2008;26(Suppl 4):31–34. doi: 10.1016/j.vaccine.2008.07.078. [DOI] [PubMed] [Google Scholar]
- 24.World Health Organization. International travel and health: seasonal influenza and influenza A (H1N1) April 2020, https://www.who.int/ith/diseases/si_iAh1n1/en/ [Google Scholar]
- 25. World Health Organization. Up to 650,000 people die of respiratory diseases linked to seasonal flu each year. April 2020, http://www.who.int/mediacentre/news/releases/2017/seasonal-flu/en/ [Google Scholar]
- 26.Rothe C, Schunk M, Sothmann P, et al. Transmission of COVID-19 Infection from an asymptomatic contact in Germany. N Engl J Med. 2020;382:970–971. doi: 10.1056/NEJMc2001468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kucirka LM, Lauer SA, Laeyendecker O, Boon D, Lessler J. Variation in false-negative rate of reverse transcriptase polymerase chain reaction-based SARS-CoV-2 tests by time since exposure. Ann Intern Med. 2020;173:262–267. doi: 10.7326/M20-1495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Long C, Xu H, Shen Q, et al. Diagnosis of the Coronavirus disease (COVID-19): rRT-PCR or CT? Eur J Radiol. 2020;126:108961. doi: 10.1016/j.ejrad.2020.108961. doi:10.1016/j.ejrad.2020.108961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Watson J, Whiting PF, Brush JE. Interpreting a covid-19 test result. BMJ. 2020;369 doi: 10.1136/bmj.m1808. m1808. Published 2020 May 12 doi:10.1136/bmj.m1808. [DOI] [PubMed] [Google Scholar]
- 30.Self WH, Courtney DM, McNaughton CD, Wunderink RG, Kline JA. High discordance of chest x-ray and computed tomography for detection of pulmonary opacities in ed patients: implications for diagnosing pneumonia. Am J. Emerg Med. 2013;31:401–405. doi: 10.1016/j.ajem.2012.08.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rubin GD, Ryerson CJ, Haramati LB, et al. The Role of Chest Imaging in Patient Management During the COVID-19 Pandemic: A Multinational Consensus Statement From the Fleischner Society. Chest. 2020;158:106–116. doi: 10.1016/j.chest.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kanne JP, Little BP, Chung JH, Elicker BM, Ketai LH. Essentials for Radiologists on COVID-19: An Update-Radiology Scientific Expert Panel. Radiology. 2020;296:E113–E114. doi: 10.1148/radiol.2020200527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bernheim A, Mei X, Huang M, et al. Chest CT Findings in Coronavirus Disease-19 (COVID-19): Relationship to Duration of Infection. Radiology. 2020;295(3) doi: 10.1148/radiol.2020200463. 200463. doi:10.1148/radiol.2020200463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Onigbinde SO, Ojo AS, Fleary L, Hage R. Chest Computed Tomography Findings in COVID-19 and Influenza: A Narrative Review. Biomed Res Int. 2020;2020 doi: 10.1155/2020/6928368. 6928368. Published 2020 Jun 5 doi:10.1155/ 2020/6928368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Testa A, Soldati G, Copetti R, Giannuzzi R, Portale G, Gentiloni-Silveri N. Early recognition of the 2009 pandemic influenza A (H1N1) pneumonia by chest ultrasound. Crit Care. 2012 Feb 17;16(1):R30. doi: 10.1186/cc11201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sofia S, Boccatonda A, Montanari M, et al. Thoracic ultrasound and SARS-COVID-19: a pictorial essay. J Ultrasound. 2020;23:217–221. doi: 10.1007/s40477-020-00458-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zou X, Chen K, Zou J, et al. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front Med. 2020;14:185–192. doi: 10.1007/s11684-020-0754-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Martinez-Rojas MA, Vega-Vega O, Bobadilla NA. Is the kidney a target of SARS-CoV-2? Am J Physiol Renal Physiol. 2020;318:F1454–F1462. doi: 10.1152/ajprenal.00160.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Galván Casas C, Català A, Carretero Hernández G. Classification of the cutaneous manifestations of COVID-19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol. 2020 Apr 29 doi: 10.1111/bjd.19163. 10.1111/bjd.19163. doi: 10.1111/bjd.19163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Landa N, Mendieta-Eckert M, Fonda-Pascual P, Aguirre T. Chilblain-like lesions on feet and hands during the COVID-19 Pandemic. Int J Dermatol. 2020;59:739–743. doi: 10.1111/ijd.14937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tang K, Wang Y, Zhang H, Zheng Q, Fang R, Sun Q. Cutaneous manifestations of the Coronavirus Disease 2019 (COVID-19): A brief review. Dermatol Ther. 2020:e13528. doi: 10.1111/dth.13528. doi:10.1111/dth.13528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cogan E, Foulon P, Cappeliez O, Dolle N, Vanfraechem G, De Backer D. Multisystem Inflammatory Syndrome With Complete Kawasaki Disease Features Associated With SARS-CoV-2 Infection in a Young Adult. A Case Report. Front Med (Lausanne) 2020;7:428. doi: 10.3389/fmed.2020.00428. Published 2020 Jul 14 doi:10.3389/ fmed.2020.00428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fried JA, Ramasubbu K, Bhatt R, et al. The variety of cardiovascular presentations of COVID-19 [published online April 3, 2020] Circulation. doi: 10.1161/CIRCULATIONAHA.120.047164. doi: 10.1161/CIRCULATIONAHA.120.047164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lang JP, Wang X, Moura FA, Siddiqi HK, Morrow DA, Bohula EA. A current review of COVID-19 for the cardiovascular specialist. Am Heart J. 2020;226:29–44. doi: 10.1016/j.ahj.2020.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Boukhris M, Hillani A, Moroni F, et al. Cardiovascular Implications of the COVID-19 Pandemic: A Global Perspective. Can J Cardiol. 2020;36:1068–1080. doi: 10.1016/j.cjca.2020.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cui S, Chen S, Li X, Liu S, Wang F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J Thromb Haemost. 2020;18:1421–1424. doi: 10.1111/jth.14830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Moores LK, Tritschler T, Brosnahan S, et al. Prevention, diagnosis and treatment of venous thromboembolism in patients with COVID-19: CHEST guideline and expert panel report. Chest. 2020;3692:31625–31621. [Google Scholar]
- 50.Tian Y, Rong L, Nian W, He Y. Review article: gastrointestinal features in COVID-19 and the possibility of faecal transmission. Aliment Pharmacol Ther. 2020;51:843–851. doi: 10.1111/apt.15731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tran J, Glavis-Bloom J, Bryan T, Harding KT, Chahine C, Houshyar R. COVID-19 patient presenting with initial gastrointestinal symptoms. Eurorad. 2020 Case 16654. [Google Scholar]
- 53.Han C, Duan C, Zhang S, et al. Digestive Symptoms in COVID-19 Patients With Mild Disease Severity: Clinical Presentation, Stool Viral RNA Testing, and Outcomes. Am J Gastroenterol. 2020;115:916–923. doi: 10.14309/ajg.0000000000000664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. ACG News Team, Joint GI Society Message on COVID-19, American College of Gastroenterology, 2020, https://gi.org/2020/03/15/joint-gi-society-message-oncovid-19/ [Google Scholar]
- 55.Zhang C, Shi L, Wang FS. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020;5:428–30. doi: 10.1016/S2468-1253(20)30057-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cascella M, Rajnik M.M, Cuomo A.A, et al. Features, evaluation and treatment coronavirus (COVID-19), StatPearls [Internet], StatPearls Publishing, Treasure Island (FL) 2020 https://www.ncbi.nlm.nih.gov/books/NBK554776/ [PubMed] [Google Scholar]
- 57.Wang F, Wang H, Fan J, Zhang Y, Wang H, Zhao Q. Pancreatic Injury Patterns in Patients With Coronavirus Disease 19 Pneumonia. Gastroenterology. 2020;159:367–370. doi: 10.1053/j.gastro.2020.03.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Soliman AT, Al-Amri M, Alleethy K, Alaaraj N, Hamed N, De Sanctis V. Newly-onset type 1 diabetes mellitus precipitated by COVID-19 in an 8-month-old infant. Acta Biomed. 2020;91(3) doi: 10.23750/abm.v91i3.10074. Published 2020 Jul 13 doi:10.23750/abm.v91i3.10074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ahmad I, Rathore FA. Neurological manifestations and complications of COVID-19: A literature review. J Clin Neurosci. 2020;77:8–12. doi: 10.1016/j.jocn.2020.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Filatov A, Sharma P, Hindi F. Neurological complications of coronavirus disease (COVID-19): encephalopathy. Cureus. 2020;12(3):e7352. doi: 10.7759/cureus.7352. doi: 10.7759/cureus.7352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Baig A.M, Khaleeq A, Ali U, Syeda H. Evidence of the COVID-19 virus targeting the CNS: tissue distribution, host virus interaction, and proposed neurotropic mechanisms. ACS Chem Neurosci. 2020;11:995–998. doi: 10.1021/acschemneuro.0c00122. [DOI] [PubMed] [Google Scholar]
- 62.Mao L, Jin H, Wang M, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:1–9. doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lechien JR, Chiesa-Estomba CM, De Siati DR, et al. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study. Eur Arch Otorhinolaryngol. 2020;277:2251–2261. doi: 10.1007/s00405-020-05965-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Chen L, Deng C, Chen X, et al. Ocular manifestations and clinical characteristics of 535 cases of COVID-19 in Wuhan, China: a cross-sectional study. Acta Ophthalmol. 2020 doi: 10.1111/aos.14472. 10.1111/aos.14472. doi:10.1111/aos.14472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wu P, Duan F, Luo C, et al. Characteristics of Ocular Findings of Patients With Coronavirus Disease 2019 (COVID-19) in Hubei Province, China. JAMA Ophthalmol. 2020;138:575–578. doi: 10.1001/jamaophthalmol.2020.1291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Khavandi S, Tabibzadeh E, Naderan M, Shoar S. Corona virus disease-19 (COVID-19) presenting as conjunctivitis: atypically high-risk during a pandemic. Cont Lens Anterior Eye. 2020;43:211–212. doi: 10.1016/j.clae.2020.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ulhaq ZS, Soraya GV. The prevalence of ophthalmic manifestations in COVID-19 and the diagnostic value of ocular tissue/fluid. Graefe’s Arch Clin Exp Ophthalmol. 2020;258:1351–1352. doi: 10.1007/s00417-020-04695-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ho D, Low R, Tong L, Gupta V, Veeraraghavan A, Agrawal R. COVID-19 and the Ocular Surface: A Review of Transmission and Manifestations. Ocul Immunol Inflamm. 2020;28:726–734. doi: 10.1080/09273948.2020.1772313. [DOI] [PubMed] [Google Scholar]
- 69.Jiang G, Korteweg C. Pathology and pathogenesis of severe acute respiratory syndrome. Am J Pathol. 2007;170:1136–1147. doi: 10.2353/ajpath.2007.061088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kunutsor SK, Laukkanen JA. Renal complications in COVID-19: a systematic review and meta-analysis. Ann Med. 2020:1–9. doi: 10.1080/07853890.2020.1790643. doi:10.1080/07853890.2020.1790643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Hirsch JS, Ng JH, Ross DW, et al. Acute kidney injury in patients hospitalized with COVID-19. Kidney Int. 2020;98:209–218. doi: 10.1016/j.kint.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Cummings MJ, Baldwin MR, Abrams D, et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet. 2020;395:1763–1770. doi: 10.1016/S0140-6736(20)31189-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Farouk SS, Fiaccadori E, Cravedi P, Campbell KN. COVID-19 and the kidney: what we think we know so far and what we don’t. J Nephrol. 2020:1–6. doi: 10.1007/s40620-020-00789-y. doi:10.1007/s40620-020-00789-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Kunutsor SK, Laukkanen JA. Renal complications in COVID-19: a systematic review and meta-analysis. Ann Med. 2020:1–9. doi: 10.1080/07853890.2020.1790643. doi:10.1080/07853890.2020.1790643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sun DQ, Wang TY, Zheng KI, et al. Subclinical acute kidney injury in COVID-19 patients: a retrospective cohort study. Nephron. 2020:1–4. doi: 10.1159/000508502. https://doi.org/10.1159/000508502 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Diao B, Wang C, Wang R, et al. Human kidney is a target for novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. medRxiv. 2020 doi: 10.1038/s41467-021-22781-1. 2020.03.04.20031120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Shen Q, Xiao X, Aierken A, et al. The ACE2 expression in Sertoli cells and germ cells may cause male reproductive disorder after SARS-CoV-2 infection [published online ahead of print, 2020 Jun 28] J Cell Mol Med. 2020;24:9472–9477. doi: 10.1111/jcmm.15541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wang Z, Xu X. scRNA-seq profiling of human testes reveals the presence of the ACE2 receptor, a target for SARS-CoV-2 infection in Spermatogonia, Leydig and Sertoli cells. Cells. 2020;9(4):920. doi: 10.3390/cells9040920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ma L, Xie W, Li D. Effect of SARS-CoV-2 infection upon male gonadal function: a single center-based study. medRxiv. 2020 doi: 10.1101/2020.03.21.20037267. [Google Scholar]
- 80.Chen F. Rising Concern on Damaged Testis of COVID-19 Patients. Urology. 2020;142:42. doi: 10.1016/j.urology.2020.04.069. doi: 10.1016/j.urology.2020.04.069. [DOI] [PMC free article] [PubMed] [Google Scholar]


