ABSTRACT
Antibiotics in childhood have been linked with diseases including asthma, juvenile arthritis, type 1 diabetes, Crohn’s disease and mental illness. The underlying mechanisms are thought related to dysbiosis of the gut microbiome. We conducted a systematic review of the association between antibiotics and disruption of the pediatric gut microbiome. Searches used MEDLINE, EMBASE and Web of Science. Eligible studies: association between antibiotics and gut microbiome dysbiosis; children 0–18 years; molecular techniques of assessment; outcomes of microbiome richness, diversity or composition. Quality assessed by Newcastle–Ottawa Scale or Cochrane Risk of Bias Tool. Meta-analysis where possible. A total of 4,668 publications identified: 12 in final analysis (5 randomized controlled trials (RCTs), 5 cohort studies, 2 cross-sectional studies). Microbiome richness was measured in 3 studies, species diversity in 6, and species composition in 10. Quality of evidence was good or fair. 5 studies found a significant reduction in diversity and 3 a significant reduction in richness. Macrolide exposure was associated with reduced richness for twice as long as penicillin. Significant reductions were seen in Bifidobacteria (5 studies) and Lactobacillus (2 studies), and significant increases in Proteobacteria such as E. coli (4 studies). A meta-analysis of RCTs of the effect of macrolide (azithromycin) exposure on the gut microbiome found a significant reduction in alpha-diversity (Shannon index: mean difference −0.86 (95% CI −1.59, −0.13). Antibiotic exposure was associated with reduced microbiome diversity and richness, and with changes in bacterial abundance. The potential for dysbiosis in the microbiome should be taken into account when prescribing antibiotics for children.
Systematic review registration number: CRD42018094188
KEYWORDS: Gut dysbiosis, Antibiotics, Children
Introduction
Research over recent years has emphasized the importance of the gut microbiome, and its association with health and the immune system. On the one hand, methods of enhancing the microbiome have proved effective. For example, probiotics have been used to reduce the incidence of severe necrotizing enterocolitis in preterm neonates as the gut microbiome is insufficiently developed to regulate the intestinal mucosa,1 and fecal microbial transplant (FMT) is being used successfully to treat patients with allergic colitis or Clostridium difficile infection.2,3 On the other hand, damage to the microbiome has been linked with conditions such as asthma,4–7 allergy,8 juvenile idiopathic arthritis,9,10 type 1 diabetes,11 obesity,12–17 celiac disease,18 mental illness,19 Crohn’s disease,20 and impaired neurocognitive outcomes.21
Although the mechanism of association for these diseases has not been fully explored, antibiotics, one of the most commonly prescribed drugs in children in western populations,22 appear to disrupt the normal maturation of the microbiome and destabilize it, altering basic physiological equilibria.23,24 Antibiotics also seem to affect gene expression, protein activity and overall metabolism of the gut microbiota which may directly influence major organ development and immune functioning.25 Antibiotic exposure has already been shown to alter the gut microbiome in adults and in neonates.26,27 This review sought to systematically examine the research into the association between antibiotic exposure and pediatric gut microbiome disruption.
Results
Study selection
The literature search identified 4,688 publications. The process of publication selection is described in Figure 1. Twelve studies met the eligibility criteria, were deemed good (nine studies) or fair (3 studies) in quality and were included in the final analysis. Meta-analysis was carried out on four RCTs that shared the Shannon Index as their outcome measure of the impact of antibiotics up to 14 days after administration. Quality assessments of RCTs are presented in Supplementary Data Figure S1 (Cochrane Risk of Bias Tool).28A high risk of bias was found in Wei et al.’s trial with respect to blinding of the outcome in the analysis done at 4 years but there was no such risk with respect to the analysis done at 14 days.29Quality assessments (Newcastle–Ottawa Scale) of observational cohort studies are presented in Supplementary Data Table S1 and of cross-sectional studies in Supplementary Data Table S2.30,31
Figure 1.
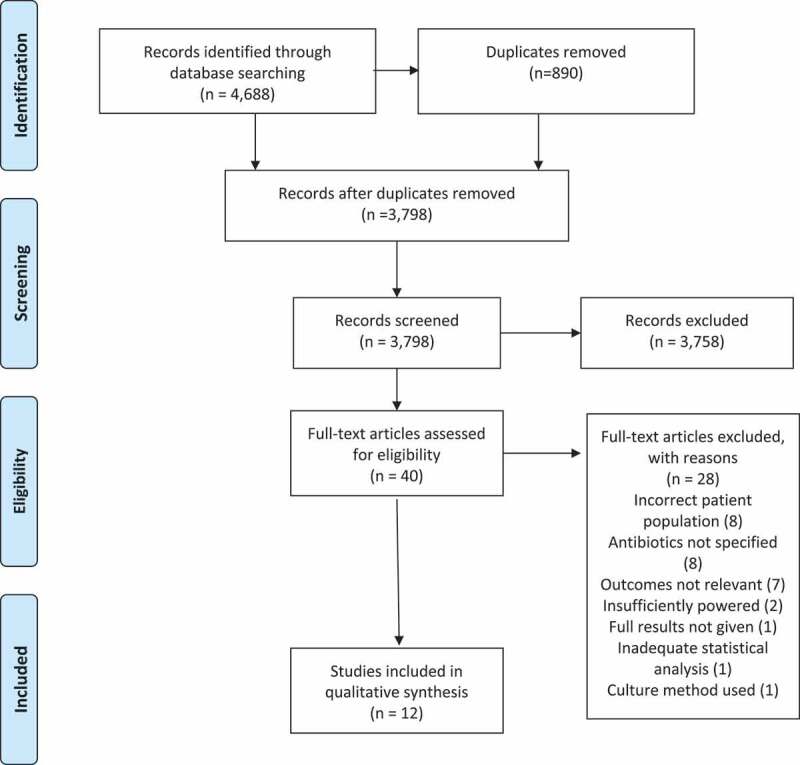
PRISMA flow chart. Preferred reporting items for systematic reviews and meta-analyses 2009
Included studies’ design and participant characteristics
The main characteristics of the included studies are summarized in Table 1. There were five randomized controlled trials (RCTs), five cohort studies and two cross-sectional studies. All studies detected changes in composition of the microbiome following antibiotic exposure in 3 main outcomes: reduction in microbiome species richness; reduction in species diversity; and change in taxonomic composition (change in a specific phylum, genus or species). The primary aim varied between studies. The age of participants ranged from new-born to 12 years old. Nearly all studies reported the short-term associations between antibiotic exposure (up to 1 month) and microbiome composition; some also reported longer-term outcomes up to 2 years and 4 years.33,37,29,42
Table 1.
Summary of included studies: author, year, location, design, aim, participants, antibiotic, duration, molecular techniques used, and outcomes
| Author, year, location | Design and reference group |
Aim | Study participants (n), age | Antibiotic exposure (route; duration) |
Exposure to outcome interval (sampling frequency) | Molecular technique | Key findings: Richness |
Key findings: Diversity |
Key findings: Taxonomical change |
|---|---|---|---|---|---|---|---|---|---|
| 1.Bai et al.32 China |
Cross-sectional with (control group) |
Added impact of antibiotics on microbiota changes in ALL | 33 healthy children; 10 received antibiotics 1–12 years |
Cephalosporin Penicillin (oral + IV: 10 days) |
1–4 weeks (x1) |
Next generation sequencing (NGS) | n/a |
Reduction in α-diversity Shannon index, ~2.75 (antibiotics) vs ~3.25 (control), p < .05 # Simpson index ~0.15 (antibiotics) vs ~0.09 (control) p < .05 # a |
Decreased Firmicutes/Bacteroidetes ratio by approximately one third (p < .05) No difference in abundances of Actinobacteria and Proteobacteria |
| 2. Bokulich et al,33 USA |
Cohort (baseline assessment) |
Microbiome development in first 2 years of life | 43 infants, 25 received antibiotics 0–2 years |
Cephalosporin Beta-lactams Macrolides Quinolones Nitrofurantoin (route/duration not specified) |
3–139 days (x25) |
PCR, 16s RNA gene amplification |
n/a |
No change in α-diversity after antibiotic exposure for median 52 days (13–139) Reduced β–diversity: UniFrac distance, Permutational MANOVA, R2 < 0.01, p < .001 |
No effect on Bifidobacterium abundance |
| 3.Brunser et al.34 Chile |
RCT (baseline assessment) |
Impact on microbiome of prebiotic supplement following antibiotic | 130 infants, before and after antibiotics 1–2 years |
Amoxicillin (oral; 7 days) |
1–3 weeks (x3) |
FISH* and flow cytometry | n/a | n/a |
Decrease in amoxicillin-associated fecal bacteria by 30% (p < .001) Increase in amoxicillin associated E. Coli Log count 4.77 ± 0.96 (baseline) vs 5.10 ± 1.39 after treatment (p = .015) No change in total counts of Bifidobacterium and Bacteroides |
| 4.Doan et al.35 Niger |
RCT (control group) |
Effects of azithromycin on gut microbiome diversity | 80 children, 40 received antibiotics 1–5 years |
Azithromycin (oral; single dose) |
5 days (x1) |
16S rRNA sequencing | n/a |
Reduction in α-diversity: Inverse Simpson’s α-diversity decreased (5.03 95% CI 4.08–6.14) vs placebo (6.91; 5.82 − 8.21) p = .03 Shannon’s α-diversity decreased (10.60; 95% CI 8.82–12.36) vs placebo (15.42; 13.24–17.80) p = .004 No change in β-diversity Decrease in Simpson’s community level γ diversity with azithromycin (10.10 95% CI 7.80–11.40) vs placebo (17.72;13.80–20.21) (p < .001) |
n/a |
| 5.Fouhy et al.36 Ireland |
Cohort study (control group) | Effect of antibiotics on gut microbiome | 18 children, 9 received antibiotics Newborn |
Ampicillin and gentamicin (IV:2–9 days) |
4 and 8 weeks (x2) |
High throughput sequencing of 16S rRNA | n/a | No change in α-diversity (Shannon Index) at 4 weeks (3.6) vs control (3.6) (p = .575) |
Decreased Bifidobacterium (5% vs 25%; p = .013) and Lactobacillus (1% vs 4%; p < .009) in treated group at 4 weeks vs control; no difference at 8 weeks Increased Proteobacteria (44% vs 23%; p < .005) and Enterobacteriaceae (50% vs 32%; p = .006) at 8 weeks vs control. Increased Clostridium in treated infants than controls at week 8 (7% vs 2%, p < .035) |
| 6.Korpela et al.37 Finland |
Retro-spective controlled cohort study | Antibiotic induced changes in microbiota composition | 142 children, 99 received antibiotics 2–7 years |
Macrolides Penicillins (route/duration not specified) |
Variable (<6 months to 2 years) (x1-2) |
DNA extraction, 16s rRNA gene sequencing | Macrolides: Reduced richness up to 2 years (p < .05)# Penicillins: Reduced richness over 6 months# (p < .001)resolved by 12 months |
n/a | Macrolides: Exposure over 6 months Reduced Bifidobacterium (0.23-fold change p < .004) and Lactobacillus (0.12-fold change p < .004) Increased Bacteroides (2.04-fold change p < .004) and Proteobacteria (1.96-fold change (p < .02). Penicillins: Decreased Lactobacillus (0.09 fold; p < .004) with exposure in previous 12 months |
| 7.Mangin et al.38 Chile |
Cohort (baseline assessment) |
Impact of amoxicillin on fecal bifidobacteria | 31 infants, all received antibiotics 12–24 months |
Amoxicillin (oral: 7 days) |
0 days (x2) |
Total DNA extraction, PCR | n/a | n/a |
No change in total Bifidobacteria Disappearance of Bifidobacterium adolescentis species (0% vs 36.4% (p < .001) |
| 8. Oldenburg et al.39 Burkina Faso |
RCT (baseline assessment) |
Investigate effect of 3 antibiotics on microbial diversity | 124 children, 93 received antibiotics 6–59 months |
Amoxicillin Azithromycin Cotrimoxazole (oral: 5 days) |
5 days (x2) |
DNA extraction, deep gene sequencing | n/a |
Reduced α- diversity with Azithromycin: Inverse Simpsons’ index decreased (6.6 95% CI 5.5–7.8) vs baseline (8.8 95% CI 7.5–10.1) (p < .001) Shannon index decreased (11.0 95% CI 9.3–12.7) vs baseline (14.6 95% CI 13.0–16.2) (p < .001) No reduction with Amoxicillin or Cotrimoxazole |
n/a |
| 9.Parker et al.40 India |
RCT (baseline assessment) |
Assess microbiota changes following azithromycin | 114 infants, 56 received antibiotics 6–11 months |
Azithromycin (oral: 3 days) |
12 days (x2) |
16s rRNA gene sequencing, DNA extraction PCR |
Lower OTU with azithromycin: (68.1 ± 15.4) vs placebo (73.6 ± 13.7) (linear regression p = .027) c. 7% less | No significant change in α-diversity (Shannon index) azithromycin (2.6 95%CI 2.47–2.73) vs placebo (2.8 95%CI 2.8(2.67–2.93) (p = .087) |
Decreased relative abundance of Proteobacteria (mainly Escherichia) mean % ± SD: 15.9 ± 13.2 vs 10.2 ± 15.4 FDR (p < .001) and Verrucomicrobia (genus Akkermansia muciniphilia) 0.5 ± 3.1 vs 0.0 ± 0.0 FDR (p < .012) No change in Actinobacteria, Bacteroides and Firmicutes, Bifidobacterium |
| 10.Penders et al.41 The Netherlands |
Cross Sectional (control group) |
Examine contribution of external influences to gut microbiota composition | 1032 infants, 28 received antibiotics 1 month |
Mainly Amoxicillin (oral: duration not specified) |
<1 month (x1) |
DNA isolation, PCR | n/a | n/a |
Decrease with antibiotics in Bifidobacteria. Antibiotics (10.29 CFU/g log10) vs Control (10.7 CFU/g log10 (p < .01) Decrease in Bacteroides fragilis. Antibiotics (6.39 CFU/g log10) vs Control (9.31 CFU/g log10) (p < .01) No change in Lactobacilli, Escherichia coli, Clostridium difficile |
| 11. Wei et al.29 Denmark |
RCT (control group) |
Examine short- and long-term impacts of azithromycin treatment on gut microbiota in children | 72 children, 33 episodes of asthma-like symptoms received antibiotics 12–36 months |
Azithromycin (oral: 3 days) |
14 days and up to 4 years (x3) |
DNA extraction and sequencing | Decrease in richness at 14 days: Observed richness: 23% reduction (177.8 ± 56.0 vs. 230.6 ± 61.2, p < .001); no difference by mean 233 days |
Reduced α-diversity: at 14 days: Shannon index: 13% reduction; 2.96 ± 0.80 (mean ± SD) vs control 3.41 ± 0.58, p = .009) Reduced β–diversity: UniFrac distance, treatment accounted for variance (R2 = 3.8%, p = .027 (weighted) and F2 = 4.2% p < .001 (un-weighted) |
Reduction, 50-fold, in genus Bifidobacterium at 14 days (p adjusted <0.011 (FDR p < .05) Long term (13–39 months) no differences seen between azithromycin and placebo groups |
| 12.Yassour et al.42 Finland |
Cohort (control group) |
Study development of infant gut microbiome and effect of antibiotics | 39 children, 20 received antibiotics 2–36 months |
Amoxicillin Cefalexin Clarithromycin Amoxicillin and clavulanic acid Trimethoprim and sulfadiazine Azithromycin Cefaclor Penicillin G Netilmicin (oral: duration not specified) |
<1 month (x28) |
16S rRNA gene and whole genome shotgun sequencing |
n/a | Reduced microbiome strain (subspecies) diversity (diversity index 0.0003 vs 0.55 (control) (p < .001) |
Decreased abundance of species from clostridium clusters IV and XIVa (T regulatory immune cells) at aged 3 (median abundance ~9% vs ~15% control)b (p = .037) Less stable gut microbiome following antibiotic treatment (Jaccard Index P = <0.001) |
Key: # – Approximate mean values taken from a Box and Whisker plot; ꝉ – statistical significance testing, confidence intervals, or standard deviations not given; a – higher the Simpson index, the lower the diversity; b -raw data not given, approximate values taken from graph
Abbreviations: n/a- data not available. ALL: acute lymphoblastic leukemia. CFU/g = Colony forming units per gram of sample. FISH: Fluorescent in-situ hybridization. FDR – False Discovery Rate correction. OTU count: operational taxonomical unit.
Microbiome richness
Microbiome richness (Table 6) data were available for 3 studies and are shown in Table 2. Microbiota richness in children exposed to antibiotics was statistically significantly reduced compared to that of children not exposed to antibiotics in all three studies.29,37,40 Measures of richness included Operational Taxonomic Unit (OTU) count (see Table 2) and a generic measure of ‘observed richness’. The time between exposure and analysis was ≤ 14 days in 2 studies,29,40 and ≤ 6 months in one study.37 The reduction in richness reported by Wei et al. had resolved by the time of a second analysis (mean of 223 days following exposure).29 Korpela et al., found that microbiome richness was reduced for up to 1 year following penicillin exposure and for up to 2 years following macrolide exposure.37 Parker reported that the significant reduction in species richness was driven through depletion of Proteobacteria (mainly the species Akkermansia mucinophilia) which were particularly susceptible to azithromycin.40 Three other authors also commented on richness but did not report raw data and hence are not included in Table 2.32,36,42
Table 6.
Definitions and examples of indices measuring microbiome richness and diversity
| Measures | Definition and example indices |
|---|---|
| Species Richness43 | Total number of bacterial species in sample Example indices: Operational Taxonomic Unit (OTU) count
|
| Alpha Diversity | The number of individual bacteria from each bacterial species present in sample Example indices: Shannon Index
|
| Beta diversity | Difference in microbial composition between two samples Example index:
|
| Gamma diversity | The overall total species diversity of a range of samples (incorporating the range of different species found in each sample) Example index
|
*Simpson’s Index is an inverse scale i.e. the higher the score the lower the diversity. It is therefore often reported as the Inverse Simpson Index so that higher scores indicate higher diversity.
Table 2.
Associations between antibiotic use and changes in microbiome richness in children up to 7 years
| Study | Type | Age group |
Country | Antibiotic | Duration of treatment | Time from exposure to analysis | Index of richness used | Placebo or Control (mean ± SD) | Intervention (mean ± SD) |
Percentage difference | Significance |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Wei et al.29 |
RCT | 1–3 years | Denmark | Azithromycin | 3 days | 14 days | Observed richness |
230.6 ± 61.2 | 177.8 ± 56.0 | −25.9% | p < .001 |
| Parker et al.40 | RCT | 6–11 months | India | Azithromycin | 3 days | 12 days | OTU count | 73.6 ± 13.6 | 68.1 ± 15.4 | −7.5% | p = .027 |
| Korpela et al.37 | Retro- spective cohort |
2–7 years | Finland | Macrolides (M) Penicillins (P) |
n/a | <6 months | OTU count | 230# | 175# (M) 180# (P) |
−23.91%(M) -21.74% (P) |
p < .001 p < .001 |
# Approximate value taken from bar chart. Confidence intervals or standard deviations not available. n/a = not available.
SD = Standard Deviation.
Microbiome diversity
Microbiome species diversity was reported by 8 studies.32,33,35,36,39,40,29,42 Data were available for 6.29,32,35,36,39,39,40 The main diversity outcome measure was alpha-diversity (Table 3). Antibiotic use was associated with a reduction in alpha-diversity (measured by Shannon or Simpson/Inverse Simpson indices) in 4 studies.29,32,35,39 Initial Shannon diversity indices varied substantially by geographical location (approximate index value of ‘3ʹ in studies in China, Denmark, India, and Ireland to approximate index value of ‘15ʹ in Burkina Faso and Niger). We carried out a meta-analysis of 4 RCTs examining the effect of azithromycin on the microbiome measured by the Shannon Index. We found a statistically significant overall reduction in alpha-diversity (mean difference −0.86 (−1.59 to −0.13, p < .001) (Figure 2).
Table 3.
Association between antibiotic use and changes in microbiome alpha-diversity (species level) in children up to 12 years
| Study authors, year, type and setting | Age group | Antibiotic | Days of therapy | Time between exposure and analysis | Indices of alpha-diversity used | Placebo/Control Group mean (95% CI) |
Intervention group mean (95% CI or SD) |
Percentage difference | Significance |
|---|---|---|---|---|---|---|---|---|---|
| Doan et al.35 RCT Niger |
1–5 years | Azithromycin | Single dose | 5 days | Shannon Inverse Simpson |
15.42 (13.24–17.80) 6.91 (5.82–8.21) |
10.60 (8.82–12.36) 5.03 (4.08–6.14) |
−31.25% -27.21% |
p = .004 p = .03 |
| Oldenburg et al.39 RCT Burkino Faso |
6–59 months | Azithromycin | 5 days | 5 days | Shannon Inverse Simpson |
14.6 (13.0–16.2) 8.80 (7.5–10.1) |
11.0 (9.3–12.7) 6.6 (5.5–7.8) |
−24.65% -25.0% |
p < .001 p < .001 |
| Wei et al.29 RCT Denmark |
1–3 years | Azithromycin | 3 days | 14 days | Shannon | 3.41 (3.23–3.59) | 2.96 (2.69–3.23) | −13.19% | P = .009 |
| Parker et al.40 RCT India |
6–11 months | Azithromycin | 3 days | 12 days | Shannon | 2.8 (2.67–2.93) | 2.6 (2.47–2.73) | −7.14% | P = .087 |
| Bai et al.32 Cross-sectional China |
1–12 years | Cephalosporin, Penicillin | 10 days | 1–4 weeks | Shannon Simpson |
3.25* | 2.75* | −15.38% | P < .05 |
| Fouhy et al.36 Cohort Ireland |
New-born | Ampicillin; gentamicin | 2–9 days | 4 weeks | Shannon | 3.8† | 3.6† | −5.26% | P = .575 |
† Confidence intervals or standard deviation not available*approximate values taken from box and whisker plot.
Figure 2.
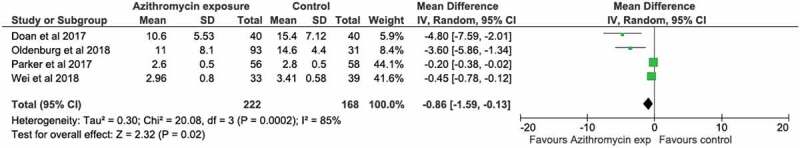
Meta-analysis of trials of azithromycin that used Shannon Index of microbiome alpha diversity as the outcome
Beta-diversity was reported in 3 studies and significantly reduced in 2 of those.29,33,35 Bokulich examined the impact of exposure to several different classes of antibiotics: cephalosporins, beta-lactams, macrolides, quinolones, and nitrofurantoin.33 They found that although microbiome alpha-diversity was unchanged following antibiotic exposure, beta-diversity differed significantly between children exposed to antibiotics and those unexposed (UniFrac distance, permutational MANOVA, R2 < 0.01 p < .001).33 This means that although the individual diversity index did not change (i.e. wide species variety and abundance) there was a significant change in the types of species found. With regards to azithromycin exposure alone, Wei reported associations with reduced alpha and beta diversity.29 Doan however found that beta diversity was unaffected (i.e., similar types of species in the two groups) following azithromycin exposure. But there was a 43% decrease in Simpson’s community-level gamma diversity (p < .001) which reflected the overall reduction in bacterial diversity of the treatment group compared to the placebo group.35 Six of eight studies reporting on species diversity found a significant association between antibiotic use and a reduction in species diversity.
Taxonomic composition
The major phyla reported in all studies were Actinobacteria, Bacteroidetes, Firmicutes, and Proteobacteria. One study reported the phylum Veruccomicrobia.40 A significant increase or decrease in the abundance of a particular phylum, genus or species was reported in 10 studies. These results are summarized in Table 4.
Table 4.
Associations between antibiotic use and changes in taxonomic composition of the microbiome in children up to 12 years
| Study Author, year, type of study, country, duration. |
Antibiotic (days of treatment where given) |
Time from exposure to analysis | Actinobacteria (includes genus Bifidobacteria) |
Bacteroidetes | Firmicutes (gram positive) |
Proteobacteria (gram negative) |
|---|---|---|---|---|---|---|
| Bai et al.32 Cross-Sectional China 1–12 years |
Cephalosporin, Penicillin 10 days |
1–4 weeks | No change in Actinobacteria | F/B ratio decreased by approx. 1/3(p < .05) * (increase in Bacteroidetes) |
F/B ratio decreased by approx. 1/3 (p < .05) * (decrease in Firmicutes) |
No change |
| Bokulich et al.33 Cohort USA 0–2 years |
Cephalosporin Beta lactams Macrolides Quinolones Nitrofurantoin |
3–139 days | No change in Bifidobacteria | Increased (p < .05) * | Clostridiales decreased from 3–9 months of age* | Increased (p < .05) * |
| Brunser et al.34 Cohort Chile 0–2 years |
Amoxicillin 7 days |
1–3 weeks | No change in Bifidobacteria | No change | n/a |
E. coli increased vs baseline (Log 4.77 ± 0.96 vs Log 5.10 ± 1.39 p = .015) |
| Fouhy et al.36 Cohort Ireland Neonates |
Ampicillin; gentamicin 2–9 days |
4 weeks | Lower levels of Bifidobacteria at 4 weeks vs control (5% vs 25%; p = .013); no difference at 8 weeks |
No change | Lower levels of Lactobacillus at 4 weeks vs control (1% vs 4%; p < .009); no difference at 8 weeks |
Higher proportions of Proteobacteria (44% vs 23%; p < .005) and Enterobacteriaceae (50% vs 32%; p = .006) at 8 weeks vs control |
| Korpela et al.37 Retrospective cohort Finland 2–7 years |
Macrolides (M) Penicillins (P) |
<6 months | Exposure in previous 6 months M: Bifidobacterium (0.23-fold decrease p < .004) P: Bifidobacterium: no change |
Exposure in previous 6 months M: Bacteroides increased (2.04-fold change p < .004) P: Bacteroides: no change |
Exposure in previous 6 months M: Lactobacillus decreased (0.12-fold change p < .004), Clostridium increased (2.68-fold change p < .004) P: Lactobacillus decreased (0.11-fold change p < .004), Clostridium: no change |
M: Proteobacteria increased (1.96-fold change p < .02) with exposure in previous 6 months P: no change |
| Mangin et al.38 Cohort Chile 6–59 months |
Amoxicillin 7 days |
0 days | Total Bifidobacterium concentrations not significantly altered but complete disappearance of Bifidobacterium adolescentis species (0% vs 36.4% (p < .001) |
n/a | n/a | n/a |
| Parker et al.40 RCT India 6–11 months |
Azithromycin 3 days |
12 days | No change in Actinobacteria or Bifidobacterium | No change | No change | Proteobacteria reduced (relative abundance mean % ± SD: 15.889 ±13.207 (placebo) vs 10.200± 15.401 (azithromycin), FDR p < .001) E.coli reduced (relative abundance mean % ± SD:12.087± 12.457 (placebo) vs 7.309 ±13.258 (azithromycin), FDR p < .013) |
| Penders et al.41 Cross sectional Netherlands 1 month |
Amoxicillin** | < 1 month | Bifidobacteria median count placebo log10 CFU/g feces 10.70 vs amoxicillin log CFU/g feces 10.29 (p < .01) | Bacteroides fragilis median count placebo log10 CFU/g feces 9.31 vs amoxicillin log10 CFU/g 6.39 (P < .01) | No change (lactobacilli) | No change in Clostridium difficile and E.Coli |
| Wei et al.29 RCT Denmark 1–3 years |
Azithromycin 3 days |
14 days | 50 x reduction (fold change) in genus Bifidobacterium at 14 days (p adjusted < 0.011 (FDR p < .05) | n/a | n/a | n/a |
| Yassour et al.42 Cohort Finland 2–36 months |
Amoxicillin Cefalexin Clarithromycin Amoxicillin and clavulanic acid Trimethoprim and sulfadiazine Azithromycin Cefaclor Penicillin G Netilmicin |
< 1 month | n/a | n/a | Decrease in abundance of species from clostridium clusters and 14XIVa IV (T regulatory immune cells) at aged 3 (median abundance ~9% vs ~15% control) b (p = .037) | n/a |
Key: †Confidence intervals or Standard Deviation not available. # Approximate value taken from bar chart. No confidence intervals or SD available. *no raw data. ** authors state that the antibiotics taken were ‘mainly amoxicillin’. b raw data not given; approximate values taken from graph.
Abbreviations: CFU/g = Colony forming unit/gram; F/B ratio = Firmicutes/Bacteroidetes ratio. FDR – false discovery rate correction. M = macrolides . P = penicillins. n/a = Data not available.
Actinobacteria
The association between antibiotics and the abundance of genus Bifidobacterium (phylum Actinobacteria) was examined in 9 studies (Table 4). In five studies, antibiotics were significantly associated with reduced abundance of Bifidobacteria.29,36–38,41 Both penicillins and macrolides were associated with a decrease in Bifidobacteria although in some studies there was no change. Comparing macrolides with penicillins, Korpela et al. found that exposure to macrolides was associated with a fourfold decrease in Bifidobacteria but that exposure to penicillins was not associated with Bifidobacteria levels.37 Fouhy et al. found that a combination of ampicillin and gentamicin was associated with reduced Bifidobacteria initially, but that by 8 weeks levels had returned to that of the control group.36 At species level, Mangin et al. found amoxicillin exposure was associated with complete disappearance of Bifidobacterium adolescentis but that overall concentrations of Bifidobacteria were not altered.38
Bacteroidetes
The association between antibiotics and the abundance of Bacteroidetes phylum (which includes the genus Bacteroides) was examined in seven studies (Table 4). There was a statistically significant change in 4 studies. The 3 studies that reported an increase in Bacteroidetes examined exposure to a combination of antibiotics including cephalosporins and macrolides.32,33,37 One study examining only amoxicillin exposure reported a decrease of the species Bacteroides fragilis.41 In 3 studies there was no change (studies examining amoxicillin, ampicillin/gentamicin and azithromycin).34,36,40
Firmicutes
The association between antibiotics and the abundance of Firmicutes phylum (which includes the genera Lactobacillus and Clostridium) was examined in seven studies (Table 4).32,33,36,40,41,42 A statistically significant decrease was seen in 4 studies following antibiotic exposure.32,36,37,42 Korpela et al. reported that Lactobacillus levels were reduced for up to 12 months following penicillin use and for up to 24 months following macrolide use.37 The same study found a nearly 3-fold increase in Clostridium within 6 months of exposure to macrolides only (details of specific species not given).37 Yassour et al reported a 40% decrease in Clostridium spp. belonging to clusters IV and XIVa (inducers of T regulatory immune cells) in children aged 3 who had had antibiotics.42
Proteobacteria
The abundance of Proteobacteria following antibiotic exposure was examined in six studies (Table 4). In 5 studies there was a statistically significant change in Proteobacteria following exposure to a variety of antibiotics, however the direction of association was not consistent. At phylum level, 4 studies reported an increase in Proteobacteria following exposure to different antibiotics including penicillins, cephalosporins and macrolides.33,34,36,37 One study reported a decrease in Proteobacteria following azithromycin exposure only.40 At species level, a statistically significant increase in E.coli was reported following amoxicillin exposure in children aged 1–2 years,34 but a statistically significant decrease in E.coli was reported following azithromycin exposure in children aged 6–11 months.40
Verrucomicrobia
The association between azithromycin and a reduction in the abundance of phylum Verrucomicrobia was examined in one study (Table 4). This phylum has relatively few species described. Parker et al. examined the association between azithromycin and the species Akkermansia mucinophila which completely disappeared with azithromycin use (p < .003).40
Discussion
Key findings
As far as we are aware this is the first systematic review to synthesize the evidence of the association between antibiotic exposure and changes in the microbiome specifically in children. We found evidence of microbiome disruption characterized by changes in richness, diversity, and taxonomic composition. We cannot be sure of the duration of these changes from the data presented as most studies only presented short-term data. The studies were heterogeneous, with variation between studies in participant age, setting, duration of antibiotic exposure, type of antibiotic given, mode of delivery, outcome measures and time between exposure and analysis. These factors may influence the association between antibiotic use and microbiome composition. Evidence of change in a wide range of microbiome characteristics associated with antibiotic exposure requires further investigation and explanation.
We found evidence that antibiotic exposure was associated with a reduction in both richness and diversity. In particular azithromycin exposure reduced microbiome alpha-diversity by a mean reduction in Shannon index of 0.86. The studies looked at a variety of antibiotics covering narrow to broad-spectrum antibiotics, with macrolides and penicillins representing the antibiotics most commonly studied. Although no specific change in richness or diversity emerged according to antibiotic class, we found evidence that macrolides were associated with more changes in the microbiome than penicillins and with effects that persisted for longer.37,39
We also found evidence that antibiotic use was associated with a reduced number of gut bacteria thought to be beneficial. Bifidobacteria (phylum Actinobacteria) and Lactobacilli (phylum Firmicutes) are producers of short-chain fatty acids which have positive effects on mammalian energy metabolism and form the basis of probiotic supplements.45 The majority of studies, however, did not report changes in these genera at species level which limits our appreciation of the changes in specific species associated with antibiotics. We also found evidence that changes in other beneficial bacteria were associated with antibiotic use. One study reported a decrease in Clostridium clusters IV and XIVa which are inducers of T regulatory immune cells which have a role in regulating or suppressing other cells in the immune system.42 A second found that Azithromycin was statistically significantly associated with reduced numbers of Akkermansia Mucinophilia.40 This species has previously been recognized as having anti-inflammatory and immunostimulant properties, and improving intestinal barrier function, endotoxinaemia and insulin sensitivity.46
We found evidence that antibiotics were associated with a rise in Bacteroidetes and Proteobacteria following antibiotic exposure. These phyla include species which have been implicated in serious infection. Although Bacteroides spp. may provide some level of protection from invasive pathogens as a gut commensal, Bacteroides have also been associated with bloodstream infections and abscess formation.47 However, it cannot be assumed that higher levels of Bacteroides in the gut are the source of these infections. E.coli (Proteobacteria) is a common cause of urinary tract infections and sepsis and a major source of antimicrobial resistance.48
Study strengths and limitations
Our review highlights important findings regarding the relationship between antibiotic exposure and microbiome disruption in children. A strength of our study is that we only included studies with named antibiotics which included specific details of antibiotic administration, rather than exposure to ‘antibiotics’ in general. However, several studies included more than one named antibiotic, so in these cases it was not possible to associate a particular change with a specific antibiotic or class. In the majority of studies, the indication for antibiotic use was infection. In one study there was no clinical indication for antibiotic use but associations with changes in the microbiome were still present. This supports the independent association between antibiotic exposure and microbiome disruption, although further studies of this relationship are required.400
The use of different outcome measures limited our ability to make comparisons between studies. Although the primary outcomes reported in the RCTs were similar, the applicability of the meta-analysis result may be limited by variation in initial Shannon index scores which in turn might reflect microbiome diversity by geographical location. We could find no evidence of agreement in the literature on the definition of a normal Shannon Index. This substantial difference in variation by geographical location does not seem to have been highlighted in the literature previously and may be worth further investigation. Outcomes in observational studies covered a number of indices of richness, diversity, and taxonomical changes which precluded meta-analysis of all studies. This variation is likely to reflect a lack of consensus among researchers about the most suitable outcome measures in addition to the complexity of the microbiome itself.
The majority of the studies included in the review focussed on microbiome changes over a short time following antibiotic prescription, i.e. less than 1 month. There was limited evidence therefore of the duration of the changes following exposure. Studies that examined effects over time, found that microbiome disruption lasted between 1 and 2 years,29,37 depending on the antibiotic studied. In this interval some children will receive a further course of antibiotics potentially disrupting microbiome recovery.37,49 Further studies are necessary to determine the duration of microbiome disruption.
Comparison/relation to existing literature
A systematic review of antibiotic prescribing in neonates (up to 44 weeks gestational age) looked at the effects of antibiotics on the neonatal microbiome and similarly found that antibiotic exposure was associated with reduced gut microbial diversity and reduced colonization rates of protective commensal bacteria, although the quality of evidence was low.27 A study looking at the gut microbiota of adults also found that antibiotic exposure was associated with a decrease in beneficial bacteria such as Bifidobacterium and butyrate producers and an increase in Enterobactericae (phylum Proteobacteria). The majority of the changes lasted for approximately 45 days, but the microbiome had not fully recovered by 180 days.26 Studies in mice support the findings of more reduced diversity following macrolide exposure compared to amoxicillin exposure. Cumulative effects on the microbiome of multiple antibiotic courses, delayed microbiome maturation following antibiotics and fewer changes associated with narrow-spectrum antibiotics have all been observed.50,51
Conclusion
In conclusion this review has gathered compelling evidence that antibiotic exposure in children is associated with a reduction in richness and/or diversity, and a change in the balance of species in the microbiome with reductions in the numbers of commensal bacteria thought to be beneficial. Studies that looked at the impact on the microbiome for more than 1 month were limited but there is evidence that antibiotics are associated with disruption to the microbiome for up to 2 years. Macrolide antibiotics cause immediate and longer term damage. More detailed understanding of the strength and duration of antibiotic-specific associations with microbiome dysbiosis in children is needed. Evidence should be sought of a causal relationship between antibiotic use in children, gut dysbiosis and subsequent risk of local or systemic pathological changes with repeated courses of antibiotics. In the meantime, healthcare practitioners should consider the potential for damage to the gut microbiome when prescribing antibiotics for children.
Methods
Procedures used in this review were consistent with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
Protocol and registration
A review protocol was submitted in advance to PROSPERO, a database of systematic review protocols (registration ID: CRD42018094188).
Eligibility criteria
Our inclusion criteria were: studies of any design-assessing change in the microbiome associated with named antibiotic exposure; participants aged from 0 to <18 years (excluding pre-term babies); assessment of composition and diversity of the microbiome using a genetic analysis technique; comparable reference group or baseline assessment and adequate statistical analysis. Our exclusion criteria were non-original research; studies investigating the impact of antibiotics in labor on neonates; studies investigating exposure to any intervention which was not a named antibiotic; studies assessing the impact of antibiotics on other systemic microflora only, e.g. skin, nasal; conference abstracts where insufficient data were given and where the study authors did not reply to further enquiries; and non-English language articles.
Information sources and search strategy
The literature search was performed in February 2019. The databases searched were MEDLINE, EMBASE and Web of Science. No restrictions were placed on the publication period. Search terms included both text words and MESH terms. The full search strategy can be viewed in Supplementary data Table S3.
Study Selection and data collection process
Papers were screened using Covidence software (Melbourne, Australia) to efficiently identify the most relevant and appropriate papers. The first reviewer (LM) conducted the literature search and imported the references. Duplicate articles were removed. Two reviewers (LM and AG) screened titles and abstracts with respect to eligibility criteria. Full-text articles of potentially relevant studies were independently assessed for eligibility by two reviewers (LM and VR). Any disagreements were reviewed by another reviewer (PW) and resolved through discussion.
Data extraction
Information was extracted from included studies on the study type, purpose, characteristics of study participants (age, co-morbidities), details of the antibiotic exposure (name, route of administration), time between exposure and microbiome analysis, molecular technique used and study outcomes. Molecular techniques used included Fluorescent in-situ hybridization (FISH) and flow cytometry, 16s RNA sequencing, DNA extraction, Next Generation Sequencing (NGS) and whole-genome shotgun sequencing (see Table 5). We excluded papers that did not name the antibiotic as we could not guarantee that the participants had been exposed to antibiotics.
Table 5.
Definition of molecular techniques used by studies in the review
| Technique name | Definition |
|---|---|
| Fluorescent in-situ hybridization (FISH) | Molecular cytogenic analysis using fluorescent probes to detect, quantify and map genetic material. |
| Flow cytometry | Analysis of the frequency and other properties of cells stained with specific fluorochrome conjugated antibodies to identify bacteria, their viability, and their DNA content. |
| 16s rRNA sequencing | Amplification of a piece of RNA (amplicon) and sequencing to identify and compare bacteria within a sample. |
| DNA extraction | Purification of DNA using physical and chemical methods |
| Next Generation Sequencing (NGS) | Sequencing of DNA and RNA with different technologies |
| Whole genome shotgun sequencing | Comprehensive sampling of all genes in all organisms present to evaluate diversity and study ‘difficult to culture’ microorganisms. |
Meta-analysis
eta-analysis was performed where studies shared the same outcome and where output data were available to include in the analysis. We performed a meta-analysis of four RCTs including 390 patients looking at the mean difference in Shannon Index before and after antibiotic exposure. Continuous outcomes were analyzed using an inverse variance model with a 95% CI. Values were reported as mean differences. P-values were two-tailed and statistically significant if p < .05. Statistical heterogeneity quantification was performed using the I2 statistic. Degrees of heterogeneity were defined as none (I2 0–20%), low (I2 25–49%), moderate (I2 50–74.9%) and high (I2 > 75%). When heterogeneity was quantified as low or above, a random-effects model was used. The meta-analysis was performed using review manager (Revman) for MAC (Version 5.3. Copenhagen: The Nordic Cochrane Center. The Cochrane Collaboration, 2014).
Quality assessment and risk of bias
Observational study quality (cohort and cross-sectional studies) was assessed using a modified version of the Newcastle–Ottawa scale.30,31 The Newcastle–Ottawa scale is used to assess quality and biases. Points are assigned on a nine-point scale. LM and PW independently assessed quality factors including: i) comparability of exposed and non-exposed groups; ii) evidence of microbiome assessment prior to exposure; iii) record of antibiotic exposure; iv) confounding factors; and v) statistical analysis. RCT quality was assessed using the Cochrane Risk of Bias Tool.28 LM and PW independently applied the risk of bias assessments to each RCT. Disagreement was resolved through discussion.
Additional quality features for RCTs included clear description of inclusion/exclusion criteria and of withdrawals/dropouts.
Summary measures
The primary outcome measure was the change in bacterial composition of the microbiome. This was measured as the changes in microbiome richness, alpha-diversity or taxonomic composition.29,33,35 Secondary outcome measures were beta- and gamma-diversity.29,33,35 Microbiome richness score measures the total number of species found in a single sample. Microbiome alpha-diversity score measures the number of individual bacteria from each of the bacterial species isolated from a single sample. Beta-diversity examines the differences in species composition between 2 samples.29,33,35 Gamma-diversity measures diversity across many samples taking into account the different species found in each sample.35 With regards to change in taxonomic composition, the four main phyla reported were Actinobacteria, Bacteroidetes, Firmicutes and Proteobacteria. The various different indices used by authors to quantify these measures are summarized in Table 6.
Supplementary Material
Acknowledgment
We are grateful to the library staff at King’s College London for their advice on the literature search strategy, and to Dr Iain Marshall for his advice on the quality assessment of included studies.
Funding Statement
LM is supported by a National Institute for Health Research (NIHR) In-Practice Fellowship. THH is supported by a National Institute for Health Research (NIHR) Doctoral Research Fellowship. This paper presents independent research. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Disclosure of potential conflicts of interest
The authors report no conflicts of interest
Author contributions
LM conceived the research. LM, DA, MA and PW all contributed to the design of the study. LM, AG, VR and TH reviewed the research papers identified at each stage. LM and PW reviewed the quality criteria. LM drafted the paper and all authors contributed to the writing of the paper.
Supplementary material
Supplemental data for this article can be accessed on the publisher’s website.
References
- 1.Sawh SC, Deshpande S, Jansen S, Reynaert CJ, Jones PM.. Prevention of necrotizing enterocolitis with probiotics: a systematic review and meta-analysis. PeerJ. 2016;4:e2429. doi: 10.7717/peerj.2429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liu SX, Li YH, Dai WK, Li X-S, Qiu C-Z, Ruan M-L, Zou B, Dong C, Liu Y-H, He J-Y, et al. Fecal microbiota transplantation induces remission of infantile allergic colitis through gut microbiota re-establishment. World J Gastroenterol. 2017;23(48):8570–18. doi: 10.3748/wjg.v23.i48.8570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Khan MY, Dirweesh A, Khurshid T, Siddiqui W. Comparing fecal microbiota transplantation to standard-of-care treatment for recurrent clostridium difficile infection: a systematic review and meta-analysis. Eur J Gastroenterol Hepatol. 2018;30(11):1309–1317. doi: 10.1097/MEG.000000000000124. [DOI] [PubMed] [Google Scholar]
- 4.Marra F, Marra CA, Richardson K, Lynd LD, Kozyrskyj A, Patrick DM, Bowie WR, Fitzgerald JM. Antibiotic use in children is associated with increased risk of asthma. Pediatrics. 2009;123(3):1003–1010. doi: 10.1542/peds.2008-1146. [DOI] [PubMed] [Google Scholar]
- 5.Stromberg Celind F, Wennergren G, Vasileiadou S, Alm B, Goksor E. Antibiotics in the first week of life were associated with atopic asthma at 12 years of age. Acta Paediatr. 2018;107(10):1798–1804. doi: 10.1111/apa.14332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yamamoto-Hanada K, Yang L, Narita M, Saito H, Ohya Y. Influence of antibiotic use in early childhood on asthma and allergic diseases at age 5. Ann Allergy Asthma Immunol. 2017;119(1):54–58. doi: 10.1016/j.anai.2017.05.013. [DOI] [PubMed] [Google Scholar]
- 7.Ahmadizar F, Vijverberg SJH, Arets HGM, de Boer A, Turner S, Devereux G, Arabkhazaeli A, Soares P, Mukhopadhyay S, Garssen J, et al. Early life antibiotic use and the risk of asthma and asthma exacerbations in children. Pediatr Allergy Immunol. 2017;28(5):430–437. doi: 10.1111/pai.12725. [DOI] [PubMed] [Google Scholar]
- 8.Kim DH, Han K, Kim SW. Effects of antibiotics on the development of asthma and other allergic diseases in children and adolescents. Allergy Asthma Immunol Res. 2018;10(5):457–465. doi: 10.4168/aair.2018.10.5.457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Horton DB, Scott FI, Haynes K, Putt ME, Rose CD, Lewis JD, Strom BL. Antibiotic exposure and juvenile idiopathic arthritis: a case-control study. Pediatrics. 2015;136(2):e333–43. doi: 10.1542/peds.2015-0036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Arvonen M, Virta LJ, Pokka T, Kröger L, Vähäsalo P. Repeated exposure to antibiotics in infancy: a predisposing factor for juvenile idiopathic arthritis or a sign of this group’s greater susceptibility to infections? J Rheumatol. 2015;42(3):521–526. doi: 10.1093/aje/kwx060. [DOI] [PubMed] [Google Scholar]
- 11.Clausen TD, Bergholt T, Bouaziz O, Arpi M, Eriksson F, Rasmussen S, Keiding N, Løkkegaard EC. Broad-spectrum antibiotic treatment and subsequent childhood type 1 diabetes: a nationwide danish cohort study. PLoS One. 2016;11(8):e0161654. doi: 10.1371/journal.pone.0161654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kelly D, Kelly A, O’Dowd T, Hayes C. Antibiotic use in early childhood and risk of obesity: longitudinal analysis of a national cohort. World J Pediatr. 2019;15(4):390–397. doi: 10.1007/s12519-018-00223-1. [DOI] [PubMed] [Google Scholar]
- 13.Block JP, Bailey LC, Gillman MW, Lunsford D, Daley MF, Eneli I, Finkelstein J, Heerman W, Horgan CE, Hsia DS. Early antibiotic exposure and weight outcomes in young children. Pediatrics. 2018;142(6):e20180290. doi: 10.1542/peds.2018-0290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Stark CM, Susi A, Emerick J, Nylund CM. Antibiotic and acid-suppression medications during early childhood are associated with obesity. Gut. 2019;68(1):62–69. doi: 10.1136/gutjnl-2017-314971. [DOI] [PubMed] [Google Scholar]
- 15.Miller SA, Wu RKS, Oremus M. The association between antibiotic use in infancy and childhood overweight or obesity: a systematic review and meta-analysis. Obes Rev. 2018;19(11):1463–1475. doi: 10.1111/obr.12717. [DOI] [PubMed] [Google Scholar]
- 16.Korpela K, Zijlmans MA, Kuitunen M, Kukkonen K, Savilahti E, Salonen A, de Weerth C, de Vos WM. Childhood BMI in relation to microbiota in infancy and lifetime antibiotic use. Microbiome. 2017;5(1):26. doi: 10.1186/s40168-017-0245-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Scott FI, Horton DB, Mamtani R, Haynes K, Goldberg DS, Lee DY, Lewis JD. Administration of antibiotics to children before age 2 years increases risk for childhood obesity. Gastroenterology. 2016;151(1):120–9.e5. doi: 10.1053/j.gastro.2016.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dydensborg Sander S, Nybo Andersen AM, Murray JA, Karlstad Ø, Husby S, Størdal K.. Association between antibiotics in the first year of life and celiac disease. Gastroenterology. 2019;156(8):2217–2229. doi: 10.1053/j.gastro.2019.02.039. [DOI] [PubMed] [Google Scholar]
- 19.Köhler-Forsberg O, Gasse C, Petersen L, Nierenberg AA, Mors O, Østergaard SD, Nationwide A. Study in Denmark of the association between treated infections and the subsequent risk of treated mental disorders in children and adolescents. JAMA Psychiatry. 2018;76(3):271–279. doi: 10.1016/j.jad.2018.12.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hviid A, Svanstrom H, Frisch M. Antibiotic use and inflammatory bowel diseases in childhood. Gut. 2011;60(1):49–54. doi: 10.1136/gut.2010.219683. [DOI] [PubMed] [Google Scholar]
- 21.Slykerman RF, Coomarasamy C, Wickens K, Thompson JMD, Stanley TV, Barthow C, Kang J, Crane J, Mitchell EA. Exposure to antibiotics in the first 24 months of life and neurocognitive outcomes at 11 years of age. Psychopharmacology (Ber). 2019;236(5):1573–1582. doi: 10.1007/s00213-019-05216-0. [DOI] [PubMed] [Google Scholar]
- 22.Hales CM, Kit BK, Gu Q, Ogden C. Trends in prescription medication use among children and adolescents-United States, 1999–2014. JAMA. 2018;319(19):2009–2020. doi: 10.1001/jama.2018.569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bhalodi AA, van Engelen TSR, Virk HS, Wiersinga WJ. Impact of antimicrobial therapy on the gut microbiome. J Antimicrob Chemother. 2019;74(Supplement_1):i6–i15. doi: 10.1093/jac/dky530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Francino MP. Antibiotics and the human gut microbiome: dysbioses and accumulation of resistances. Front Microbiol. 2015;6:1543. doi: 10.3389/fmicb.2015.01543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pérez-Cobas AE, Gosalbes MJ, Friedrichs A, Knecht H, Artacho A, Eismann K, Otto W, Rojo D, Bargiela R, von Bergen M, et al. Gut microbiota disturbance during antibiotic therapy: a multi-omic approach. Gut. 2013;62(11):1591–1601. doi: 10.1136/gutjnl-2012-303184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Palleja A, Mikkelsen KH, Forslund SK, Kashani A, Allin KH, Nielsen T, Hansen TH, Liang S, Feng Q, Zhang C. Recovery of gut microbiota of healthy adults following antibiotic exposure. Nat Microbiol. 2018;3(11):1255–1265. doi: 10.1038/s41564-018-0257-9. [DOI] [PubMed] [Google Scholar]
- 27.Fjalstad JW, Esaiassen E, Juvet LK, van den Anker JN, Klingenberg C. Antibiotic therapy in neonates and impact on gut microbiota and antibiotic resistance development: a systematic review. J Antimicrob Chemother. 2018;73(3):569–580. doi: 10.1093/jac/dkx088. [DOI] [PubMed] [Google Scholar]
- 28.Higgins JPT, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA, et al. The cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wei S, Mortensen MS, Stokholm J, Brejnrod AD, Thorsen J, Rasmussen MA, Trivedi U, Bisgaard H, Sørensen SJ. Short- and long-term impacts of azithromycin treatment on the gut microbiota in children: a double-blind, randomized, placebo-controlled trial. EBioMedicine. 2018;38:265–272. doi: 10.1016/j.ebiom.2018.11.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wells G, Shea B, O’Connell D, Peterson J, Welch V, Losos M, Tugwell P.. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. 2013. Accessed 06 January 2021. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
- 31.Margulis AV, Pladevall M, Rierz-Guardia N, Varas-Lorenzo C, Hazell L, Berkman ND, Viswanathan M, Perez-Gutthann S. Quality assessment of observational studies in a drug-safety systematic review, comparison of two tools: the newcastle-ottawa scale and the RTI item bank. Clin Epidemiol. 2014;6:359–368. doi: 10.2147/CLEP.S66677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bai L, Zhou P, Li D, Ju X. Changes in the gastrointestinal microbiota of children with acute lymphoblastic leukaema and its association with antibiotics in the short term. J Med Microbiol. 2017;66(9):1297–1307. doi: 10.1099/jmm.0.000568. [DOI] [PubMed] [Google Scholar]
- 33.Bokulich NA, Chung J, Battaglia T, Henderson N, Jay M, Li H, Lieber A D, Wu F, Perez-Perez GI, Chen Y, et al. Antibiotics, birth mode, and diet shape microbiome maturation during early life. Sci Transl Med. 2016;8(343):343ra82. doi: 10.1126/scitranslmed.aad7121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Brunser O, Gotteland M, Cruchet S, Figueroa G, Garrido D, Steenhout P. Effect of a milk formula with prebiotics on the intestinal microbiota of infants after an antibiotic treatment. Pediatr Res. 2006;59(3):451–456. doi: 10.1203/01.pdr.0000198773.40937.61. [DOI] [PubMed] [Google Scholar]
- 35.Doan T, Arzika AM, Ray KJ, Cotter SY, Kim J, Maliki R, Zhong L, Zhou Z, Porco TC, Vanderschelden B, et al. Gut microbial diversity in antibiotic-naive children after systemic antibiotic exposure: a randomized controlled trial. Clin Infect Dis. 2017;64(9):1147–1153. doi: 10.1093/cid/cix141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fouhy F, Guinane CM, Hussey S, Wall R, Ryan CA, Dempsey EM, Murphy B, Ross RP, Fitzgerald GF, Stanton C, et al. High-throughput sequencing reveals the incomplete, short-term recovery of infant gut microbiota following parenteral antibiotic treatment with ampicillin and gentamicin. Antimicrob Agents Chemother. 2012;56(11):5811–5820. doi: 10.1128/AAC.00789-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Korpela K, Salonen A, Virta LJ, Kekkonen RA, Forslund K, Bork P, de Vos WM. Intestinal microbiome is related to lifetime antibiotic use in Finnish pre-school children. Nat Commun. 2016;7:10410. doi: 10.1038/ncomms10410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mangin I, Suau A, Gotteland M, Brunser O, Pochart P. Amoxicillin treatment modifies the composition of Bifidobacterium species in infant intestinal microbiota. Anaerobe. 2010;16(4):433–438. doi: 10.1016/j.anaerobe.2010.06.005. [DOI] [PubMed] [Google Scholar]
- 39.Oldenburg CE, Sie A, Coulibaly B, Ouermi L, Dah C, Tapsoba C, Bärnighausen T, Ray KJ, Zhong L, Cummings S, et al. Effect of commonly used pediatric antibiotics on gut microbial diversity in preschool children in burkina faso: a randomized clinical trial. Open Forum Infect Dis. 2018;5(11). doi: 10.1093/ofid/ofz061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Parker EPK, Praharaj I, John J, Kaliappan SP, Kampmann B, Kang G, Grassly NC. Changes in the intestinal microbiota following the administration of azithromycin in a randomised placebo-controlled trial among infants in south India. Sci Rep. 2017;7(1):9168. doi: 10.1038/s41598-017-06862-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Penders J, Thijs C, Vink C, Stelma FF, Snijders B, Kummeling I, van den Brandt PA, Stobberingh EE. Factors influencing the composition of the intestinal microbiota in early infancy. Pediatrics. 2006;118(2):511–521. doi: 10.1542/peds.2005-2824. [DOI] [PubMed] [Google Scholar]
- 42.Yassour M, Vatanen T, Siljander H, Hämäläinen AM, Härkönen T, Ryhänen SJ, Franzosa EA, Vlamakis H, Huttenhower C, Gevers D, et al. Natural history of the infant gut microbiome and impact of antibiotic treatment on bacterial strain diversity and stability. Sci Transl Med. 2016;8(343):343ra81. doi: 10.1126/scitranslmed.aad0917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kim BR, Shin J, Guevarra R, Lee JH, Kim DW, Seol KH, Lee J-H, Kim HB, Isaacson RE. Deciphering diversity indices for a better understanding of microbial communities. J Microbiol Biotechnol. 2017;27(12):2089–2093. doi: 10.4014/jmb.1709.09027. [DOI] [PubMed] [Google Scholar]
- 44.Lozupone C, Lladser ME, Knights D, Stombaugh J, Knight R. UniFrac: an effective distance metric for microbial community comparison. Isme J. 2011;5(2):169–172. doi: 10.1038/ismej.2010.133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.King S, Tancredi D, Lenoir-Wijnkoop I, Gould K, Vann H, Connors G, Sanders ME, Linder JA, Shane AL, Merenstein D. Does probiotic consumption reduce antibiotic utilization for common acute infections? A systematic review and meta-analysis. Eur J Public Health. 2018;29(3):494–499. doi: 10.1093/eurpub/cky185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fujio-Vejar S, Vasquez Y, Morales P, Magne F, Vera-Wolf P, Ugalde JA, Navarrete P, Gotteland M. The gut microbiota of healthy chilean subjects reveals a high abundance of the phylum verrucomicrobia. Front Microbiol. 2017;8:1221. doi: 10.3389/fmicb.2017.01221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wexler HM. Bacteroides: the good, the bad, and the nitty-gritty. Clin Microbiol Rev. 2007;20(4):593–621. doi: 10.1128/CMR.00008-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Vihta KD, Stoesser N, Llewelyn MJ, Quan TP, Davies T, Fawcett NJ, Dunn L, Jeffery K, Butler CC, Hayward G. Trends over time in Escherichia coli bloodstream infections, urinary tract infections, and antibiotic susceptibilities in Oxfordshire, UK, 1998-2016: a study of electronic health records. Lancet Infect Dis. 2018;18(10):1138–1149. doi: 10.1016/S1473-3099(18)30353-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hicks LA, Bartoces MG, Roberts RM, Suda KJ, Hunkler RJ, Taylor TH Jr, Schrag SJ. US outpatient antibiotic prescribing variation according to geography, patient population, and provider specialty in 2011. Clin Infect Dis. 2015;60(9):1308–1316. doi: 10.1093/cid/civ076. [DOI] [PubMed] [Google Scholar]
- 50.Nobel YR, Cox LM, Kirigin FF, Bokulich NA, Yamanishi S, Teitler I, Chung J, Sohn J, Barber CM, Goldfarb DS. Metabolic and metagenomic outcomes from early-life pulsed antibiotic treatment. Nat Commun. 2015;6:7486. doi: 10.1038/ncomms8486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yao J, Carter RA, Vuagniaux G, Barbier M, Rosch JW, Rock CO. A pathogen-selective antibiotic minimizes disturbance to the microbiome. Antimicrob Agents Chemother. 2016;60(7):4264–4273. doi: 10.1128/AAC.00535-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


