Abstract
Background
Mass drug administration (MDA) of ivermectin for onchocerciasis has been disrupted by the coronavirus disease 2019 (COVID-19) pandemic. Mathematical modelling can help predict how missed/delayed MDA will affect short-term epidemiological trends and elimination prospects by 2030.
Methods
Two onchocerciasis transmission models (EPIONCHO-IBM and ONCHOSIM) are used to simulate microfilarial prevalence trends, elimination probabilities and age profiles of Onchocerca volvulus microfilarial prevalence and intensity for different treatment histories and transmission settings, assuming no interruption, a 1-y (2020) interruption or a 2-y (2020–2021) interruption. Biannual MDA or increased coverage upon MDA resumption are investigated as remedial strategies.
Results
Programmes with shorter MDA histories and settings with high pre-intervention endemicity will be the most affected. Biannual MDA is more effective than increasing coverage for mitigating COVID-19’s impact on MDA. Programmes that had already switched to biannual MDA should be minimally affected. In high-transmission settings with short treatment history, a 2-y interruption could lead to increased microfilarial load in children (EPIONCHO-IBM) and adults (ONCHOSIM).
Conclusions
Programmes with shorter (annual MDA) treatment histories should be prioritised for remedial biannual MDA. Increases in microfilarial load could have short- and long-term morbidity and mortality repercussions. These results can guide decision-making to mitigate the impact of COVID-19 on onchocerciasis elimination.
Keywords: COVID-19, ivermectin, mass drug administration, mathematical modelling, onchocerciasis, remedial strategies
Introduction
The coronavirus disease 2019 (COVID-19) pandemic has led to severe disruptions in routine public health services on a global scale. These disruptions are expected to be particularly pronounced in low- and middle-income countries due to already underresourced healthcare systems. On 1 April 2020, the World Health Organization (WHO) advised that mass drug administration (MDA) and epidemiological surveys for neglected tropical diseases (NTDs) tackled by preventive chemotherapy and transmission control (PCT) should be postponed.1 Updated guidance was released on 27 July, which included a decision-making framework, allowing countries to restart routine MDA given careful risk assessment.2 Onchocerciasis is one such PCT disease, centred on ivermectin (Mectizan) MDA, delivered annually in the majority of endemic countries in Africa.
Although so-called lockdowns and delayed MDA might be effective in temporally reducing the transmission of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus, the implications for both short- and long-term onchocerciasis transmission are less clear. The impact of the Ebola outbreaks in West Africa (2013–2016) on morbidity due to other diseases (such as tuberculosis and human immunodeficiency virus) can provide some insight into the implications of withdrawing routine public health services.3,4 However, comparability is limited due to the stark differences in the scale and manifestations of Ebola and COVID-19. There are concerns that delaying MDA might increase onchocerciasis morbidity and undermine progress made towards the 2030 elimination of transmission (EOT) goals proposed in the recently launched WHO 2021–2030 Roadmap for NTDs.5 It is therefore important to quantify where (in terms of transmission setting and treatment history) the impact of postponements of ivermectin MDA for onchocerciasis will be most pronounced and to identify the most effective mitigation strategies to help affected programmes get back on track. This will allow better planning and prioritisation of ivermectin distribution/treatment upon safe MDA resumption.
Delayed ivermectin MDA, or reduced treatment coverage, could result not only from population-wide lockdowns to reduce COVID-19 transmission and the resulting redirection or disruption of health services, but also from shortages in drug availability (due to slower production and supply chains or exceeded drug shelf-life by the time MDA recommences).6,7 Deadlines for drug orders (typically by August for delivery in the following year), as required by the Mectizan Donation Program (MDP, the body providing oversight for ivermectin donation to endemic countries), might also be problematic. In addition to these challenges, programmes will have to be adaptive in the face of unforeseen setbacks that may emerge as MDA recommences, remedial strategies (such as increased MDA coverage and/or frequency) are attempted and as the COVID-19 pandemic progresses.
Mathematical models of onchocerciasis transmission provide a useful predictive tool for understanding the impacts of ivermectin MDA interruptions on the short-term (increases in transmission intensity) and long-term (elimination prospects), as well as the potential benefit of remedial mitigation strategies to help programmes get back on track. Although the resurgence of helminth transmission is typically slower than that of viral, bacterial and protozoan infections (due to differences in life history), increases in infection prevalence and intensity may increase onchocerciasis-associated morbidity.8 Delays in treating children who would otherwise receive ivermectin when turning 5 y of age could result in higher microfilarial loads experienced early in life, which may impact health outcomes in later years.9
In this article we use the EPIONCHO-IBM and ONCHOSIM transmission models to quantify where (in terms of transmission setting and treatment history) the impact of interruptions to ivermectin MDA for onchocerciasis will be most pronounced, allowing better planning and prioritisation of ivermectin distribution/treatment upon resumption, and investigate how mitigation strategies based on increased frequency (biannual MDA) of increased coverage can help affected programmes to get back on track. Both models contributed insights to inform the WHO 2021–2030 NTD Roadmap10 and provided preliminary but less comprehensive results for a report to the WHO on the impact of COVID-19 on NTD programmes.11
Methods
Models
We used two individual-based stochastic transmission models, namely EPIONCHO-IBM12 and ONCHOSIM13 to address seven questions concerning the effect of MDA disruptions due to COVID-19: (i) What is the impact of delaying treatment for 1 or 2 y on microfilarial prevalence trends and the probability of elimination by 2030?; (ii) Which pre-intervention endemicity levels are particularly vulnerable?; (iii) How much more vulnerable are programmes with shorter (annual) treatment histories (those with fewer years of MDA) than those with longer ones?; (iv) Which age groups will be most affected?; (v) Are remedial strategies based on increasing treatment frequency (to biannual MDA) or increasing treatment coverage during the early stages of MDA resumption useful for mitigating any setbacks?; (vi) How will programmes that had already switched to biannual MDA be impacted?; and (vii) What will be the effect on (West African) countries whose NTD programmes had already been disrupted by the Ebola outbreak in 2014?
In the context of a 1-y interruption or 1 y of missed MDA, the terms ‘interruption’ or ‘missed’ are used to describe a situation in which the MDA round planned to take place 12 months after a successful round pre-COVID-19 is not delivered (regardless of the calendar month in which treatment is usually distributed according to setting). ‘Remedial’ MDA implies that once MDA can safely resume,2 additional MDA (either an extra round or increased coverage within one round) is delivered. Hence ‘remedial’ biannual MDA can be interpreted as ‘delayed treatment’ if a round of annual MDA is not delivered when planned but given in addition to the planned round in the following (not necessarily calendar) year. Although, for simplicity, modelling results are presented as if MDA were delivered at the beginning of each year, what is important is the duration between two consecutive MDA rounds and whether MDA is delivered with increased frequency or coverage when treatment can recommence.
Supplementary Information A: Supplementary Methods provides a detailed description of the models. All (therapeutic) coverage levels refer to the percentage of people treated out of the total population, where total population includes children <5 y of age and individuals who never take treatment (non-participation). Figure S1 illustrates how elimination probabilities are calculated.
We followed the five principles of the Neglected Tropical Disease Modelling Consortium14 to advocate and adhere to using good practice for policy-relevant modelling. Supplementary Table S1 presents the Policy-Relevant Items for Reporting Models in Epidemiology of Neglected Tropical Diseases table.
Scenarios
Treatment histories and transmission settings
We simulated a range of scenarios reflecting pre-control endemicity and historical treatment durations in the former Onchocerciasis Control Programme in West Africa (OCP, 1975–2002), specifically areas in the western extension in which ivermectin MDA was implemented without vector control,15 and the African Programme for Onchocerciasis Control (APOC, 1995–2015).16
Pre-control endemicity
For the sake of completeness, we considered pre-intervention onchocerciasis endemicity levels given by a baseline Onchocerca volvulus microfilarial prevalence of 20–85% (i.e. from hypoendemic to highly hyperendemic settings), although the APOC prioritised treatment only in areas with microfilarial prevalence ≥40%.
History of ivermectin MDA
Annual ivermectin MDA programmes were simulated with start years 2000, 2003, 2006, 2009, 2012, 2014, 2017 and 2020, assuming that 65% of the total population is treated per round (i.e. approximately 80% coverage of eligible individuals ≥5 y of age). The level of systematic non-participation was assumed to be 5%.
Pre-existing biannual MDA programmes
We also modelled scenarios for pre-existing biannual programmes preceded by low-coverage annual MDA motivated by the situation in the Madi-Mid North focus in Uganda,17 where the main vector is Simulium damnosum sensu stricto (for which both models are parameterised). These pre-existing biannual programmes were assumed to have started in 1994 with annual MDA at 25% coverage (due, for instance, to internal conflict, as was the case of the Madi-Mid North focus) until 2012, when biannual treatment with 75% coverage of the total population (approximately 90% coverage of eligible individuals) began.17 A pre-intervention 50% microfilarial prevalence was motivated by the baseline values for Adjumani-Moyo16 and Kitgum18 in the Madi-Mid North focus.
Previous Ebola (2013–2014) outbreak
We modelled settings previously affected by the Ebola outbreak in western Africa in 2014, which were broadly motivated by the treatment histories in Sierra Leone, Liberia and Guinea.19–21 We assumed annual treatment from 2003 to 2013 (with coverage gradually increasing from 25% in 2003 to 65% in 2013), no treatment in 2014, 30% coverage in 2015, 65% coverage in 2016–2019, no treatment in 2020, 50% coverage in 2021 and 65% coverage in 2022–2030.
Modelling interruptions due to COVID-19
We projected microfilarial dynamics and estimated elimination probabilities assuming either a 1-y (2020) or 2-y (2020 and 2021) interruption to ivermectin MDA. Additionally, we tested various assumptions of delays in achieving pre-COVID-19 coverage upon treatment resumption, e.g. missing MDA in 2020 but gradually increasing coverage from 30% in 2021 to 50% in 2022 to 65% in 2023–2030. Levels of systematic non-participation were set at 5%.
Modelling remedial strategies
We modelled the effectiveness of increasing frequency (to biannual treatment at 65% coverage) or therapeutic coverage (to 75% for annual treatment) as strategies for mitigating the impact of missed treatment rounds (either in 2020 or in 2020 and 2021). For a selection of the scenarios described above, we investigated the capacity for remedial treatment to revert the microfilaridermia trends and restore elimination probabilities (with MDA ceasing in 2030) to those predicted without interruption. Typically MDP only approves ivermectin donations for biannual treatment if programmes can demonstrate sufficiently high coverage in preceding years. Therefore we assumed that remedial biannual MDA followed only after a year of annual 65% coverage once programmes restart.
Outcome measures
We present three model outputs to understand the implications of interruptions to MDA: temporal trends in microfilarial prevalence (%) to understand the short-term implications, probabilities of achieving elimination by 2030 to understand the long-term implications and age profiles for microfilarial prevalence (%) and intensity (microfilariae/skin snip) to identify the most affected age groups.
Results
Impact of interruptions to ivermectin MDA for annual programmes
Temporal microfilarial prevalence trends
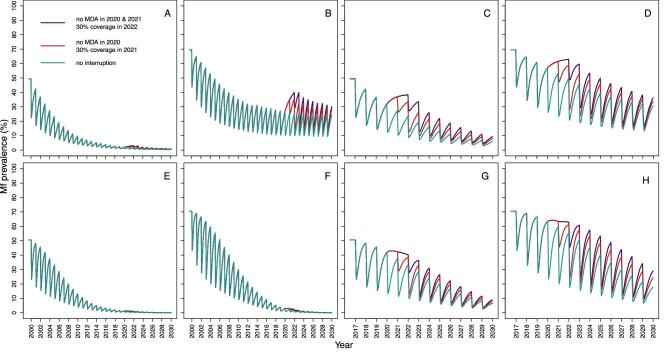
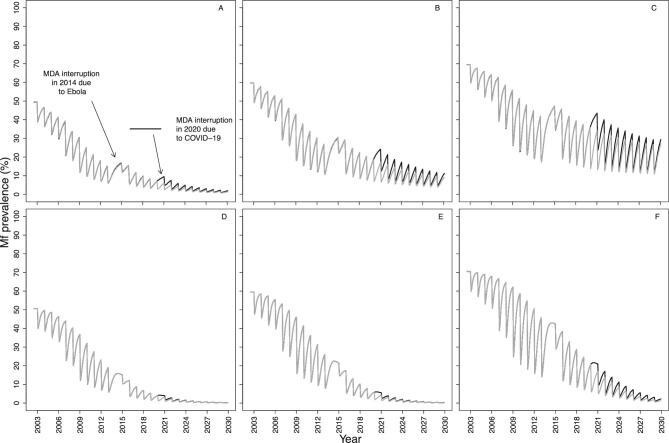
Figure 1 shows, for EPIONCHO-IBM and ONCHOSIM, the microfilarial prevalence dynamics for programmes with a long (starting in 2000) and short (starting in 2017) history of ivermectin MDA, comparing no interruption with no treatment during 2020 (and a 30% coverage at resumption in 2021) and with no treatment during 2020 and 2021 (with a 30% coverage at resumption in 2022). The models qualitatively agree on how the temporal microfilarial prevalence trends during an interruption differ from those for continuous MDA through 2020–2021 for MDA starting in 2017. Increases in microfilarial prevalence after a 2-y interruption (2020–2021) are more pronounced than after a 1-y (2020) interruption. How coverage, once treatment recommences, influences the temporal dynamics of microfilarial prevalence after a 1-y interruption is shown in Supplementary Information B: Supplementary Results, Figure S2 (EPIONCHO-IBM) and Figure S3 (ONCHOSIM). A detailed discussion of how the dynamics following an interruption differ from those without an interruption is presented in Supplementary Figure S4.
Figure 1.
Temporal dynamics of O. volvulus microfilarial prevalence assuming a 1-y (2020) and 2-y (2020–2021) interruption to annual MDA with ivermectin due to COVID-19 and no mitigation strategies, predicted by (A–D) EPIONCHO-IBM and (E–H) ONCHOSIM. The pre-intervention (baseline) microfilarial prevalence in individuals ≥5 y of age is 50% (A, C, E, G) and 70% (B, D, F, H). Annual MDA occurs from 2000 to 2030 (early-start programmes; A, B, E, F) or from 2017 to 2030 (late-start programmes; C, D, G, H), assuming no treatment in 2020 only (red lines), no treatment in both 2020 and 2021 (violet lines) and no remedial strategies subsequently. The temporal microfilarial dynamics with no interruption in MDA are shown as green lines. The therapeutic coverage (of the total population) is assumed to be 65%, with the exception of 30% in 2021 following a 1-y interruption and 30% in 2022 following a 2-y interruption to MDA. The proportion of systematic non-participation is set to 5% throughout all simulations.
Elimination probabilities
Although EPIONCHO-IBM generally predicts lower elimination probabilities than ONCHOSIM, the models agree qualitatively on the impact of MDA interruptions due to COVID-19. Both models predict that interruptions to MDA will reduce the prospects of onchocerciasis elimination by 2030 if no mitigation strategies are implemented. Both models also predict that programmes with shorter (annual) treatment histories will be more vulnerable to MDA disruptions than those that have distributed MDA for longer, particularly if treatment is delayed for 2 y (Figure 2). The impact of a 1- and 2-y interruption on elimination probabilities with start years ranging from 2000 to 2020 is presented in Supplementary Figures S5 and S6.
Figure 2.
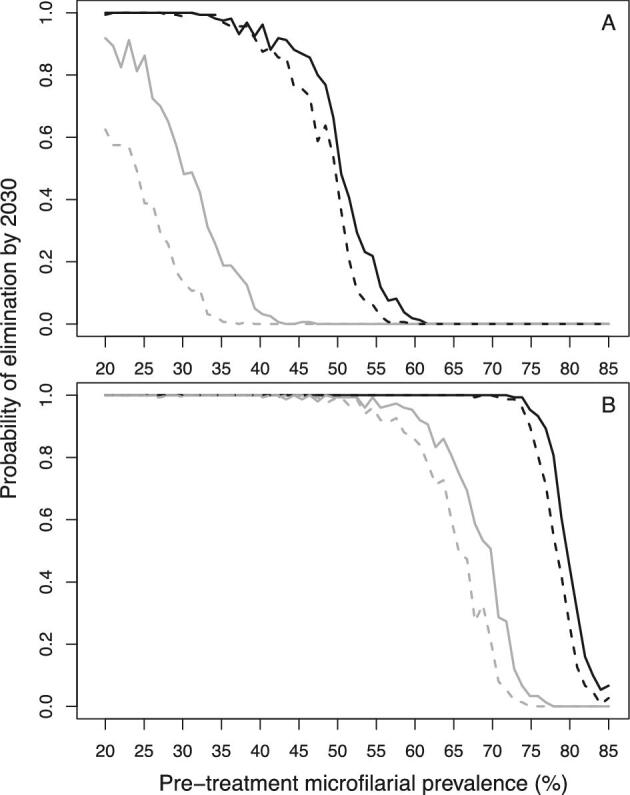
Elimination probabilities versus pre-intervention (baseline) endemicity (pre-treatment microfilarial prevalence) predicted by (A) EPIONCHO-IBM and (B) ONCHOSIM for early-start and late-start annual MDA with ivermectin with and without a 2-y (2020 and 2021) MDA interruption due to COVID-19 and no mitigation strategies. Annual MDA programmes start in 2000 (early start, black lines) or 2017 (late start, grey lines) and finish in 2030. Although in the African Programme for Onchocerciasis Control MDA treatment was prioritised for areas with a microfilarial prevalence ≥40%, the explored range of baseline microfilarial prevalences in individuals ≥5 y of age is 20%–85% for the sake of completeness. No interruption to MDA is represented by solid lines and a 2-y interruption is represented by dashed lines. The therapeutic coverage (of the total population) is assumed to be 65%, with the exception of 30% in 2022. The proportion of systematic non-participation is set to 5% throughout all simulations.
Mitigation strategies for annual programmes
Temporal microfilarial prevalence trends
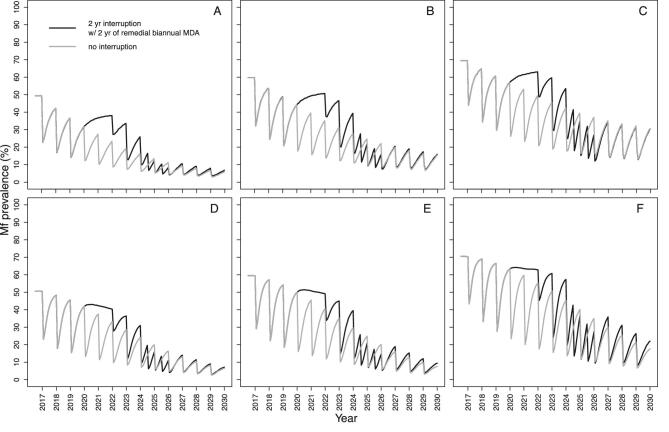
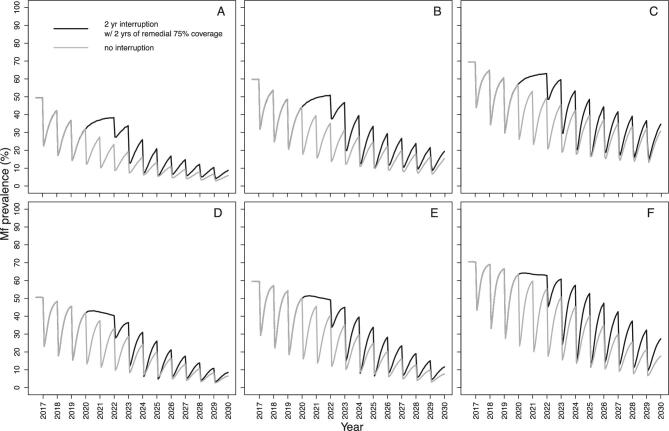
Both models suggest that for a programme with a short treatment history, increasing treatment frequency, i.e. implementing remedial biannual MDA at 65% coverage upon resumption of MDA, will be more effective at reducing the impact of treatment disruptions than increasing treatment coverage (i.e. implementing remedial 75% coverage of annual MDA). We illustrate these results after a 2-y interruption (2020–2021) when MDA initially resumes at low annual coverage (30%, 2022), returns to annual 65% coverage (2023) and then either increases to a 6-month frequency for 2 consecutive years (2024–2025) at 65% coverage (Figure 3) or increases to 75% annual MDA coverage also for 2 consecutive years (2024–2025) (Figure 4). Remedial biannual treatment results in lower microfilarial prevalence leading up to 2030 (Figure 4) than remedial increased coverage (Figure 3). The temporal microfilarial prevalence dynamics for a 1-y interruption with either remedial increased treatment frequency or coverage are shown in Supplementary Figures S7 and S8.
Figure 3.
Temporal dynamics of O. volvulus microfilarial prevalence predicted by (A–C) EPIONCHO-IBM and (D–F) ONCHOSIM with a 2-y (2020 and 2021) interruption in annual ivermectin MDA (2017–2030) due to COVID-19 and remedial biannual MDA. The pre-intervention (baseline) microfilarial prevalence (in individuals ≥5 y of age) is (A, D) 50%, (B, E) 60% and (C, F) 70%. Grey lines represent no interruption in MDA and black lines indicate no treatment in 2020 and 2021 but with 2 y of remedial biannual MDA in 2024 and 2025. The therapeutic coverage (of the total population) is assumed to be 65%, with the exception of 30% in 2022. The proportion of systematic non-participation is set to 5% throughout all simulations.
Figure 4.
Temporal dynamics of O. volvulus microfilarial prevalence predicted by (A–C) EPIONCHO-IBM and (D–F) ONCHOSIM with a 2-y (2020 and 2021) interruption due to COVID-19 of annual MDA with ivermectin (2017–2030) and remedial annual high-coverage MDA. The pre-intervention (baseline) microfilarial prevalence (in individuals ≥5 y of age) is (A, D) 50%, (B, E) 60%, and (C, F) 70%. Grey lines represent no interruption to MDA and black lines indicate no treatment in 2020 and 2021 but with 2 y of remedial high-coverage MDA in 2024 and 2025. The therapeutic coverage (of the total population) is assumed to be 65%, with the exception of 30% in 2022, 75% in 2024 and 75% in 2025. The proportion of systematic non-participation is set to 5% throughout all simulations.
Elimination probabilities with remedial strategies
For a 2-y interruption, although neither remedial strategy is sufficient to achieve the same elimination probabilities found in the absence of an interruption, 2 y of remedial biannual MDA results in higher elimination probabilities than a remedial increase in coverage (Supplementary Figure S9). For some treatment histories and pre-intervention endemicities (which do not result in 100% elimination probability without interruption), 1 y of missed MDA followed by 1 y of remedial biannual MDA gives similar elimination probabilities as those in the absence of MDA disruptions (Supplementary Figure S10).
Pre-existing biannual programmes and those previously affected by Ebola
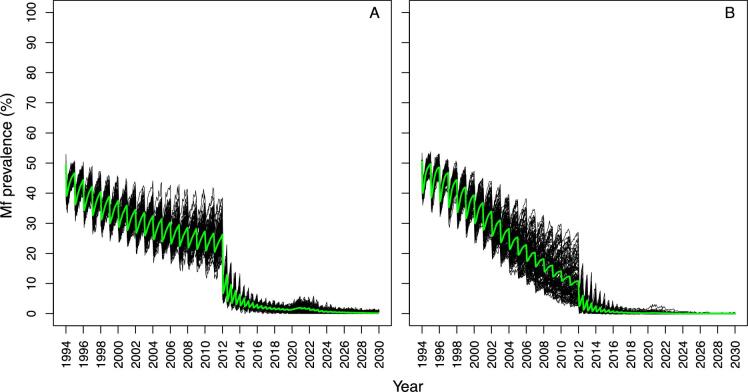
For the pre-existing biannual scenarios explored (which had achieved high pre-COVID-19 therapeutic coverage), a 1-y interruption did not result in pronounced microfilarial resurgence according to either model (Figure 5). For this scenario, it was considered that programmes would resume treatment in 2021 given their strong performance before COVID-19, so a 2-y interruption was not investigated.
Figure 5.
Temporal dynamics of O. volvulus microfilarial prevalence predicted by (A) EPIONCHO-IBM and (B) ONCHOSIM in programmes with pre-existing biannual MDA with ivermectin and a 1-y interruption (2020) due to COVID-19. Annual MDA assumes low (25%) therapeutic coverage (of the total population) from 1994 to 2011 and high (75%) biannual coverage (of the total population) from 2012 to 2019 (motivated by the Madi-Mid North focus in Uganda, where the main vector is S. damnosum sensu stricto), with a 50% pre-intervention (baseline) microfilarial prevalence (in individuals ≥5 y of age). Following resumption of MDA in 2021, coverage (of the total population) is assumed to reach 75% (same as pre-interruption). Black lines indicate individual simulations and green lines indicate the mean of all simulations. The proportion of systematic non-participation is set to 5% throughout all simulations.
EPIONCHO-IBM predicts that the impact of a 1-y interruption due to COVID-19 in 2020 on microfilarial prevalence dynamics in West African programmes that were affected by the Ebola outbreak in 2014 will depend (like in other scenarios) on the transmission setting (Figure 6). When assuming a 50% baseline microfilarial prevalence, there were small differences between no interruption and a 1-y interruption by 2030; however, for the 70% baseline microfilarial prevalence settings, clear differences between no interruption and a 1-y interruption were still evident by 2030 according to both models.
Figure 6.
Temporal dynamics of O. volvulus microfilarial prevalence predicted by (A–C) EPIONCHO-IBM and (D–F) ONCHOSIM during annual MDA with ivermectin, assuming a 1-y interruption (2014) due to the Ebola epidemic in West Africa, a 1-y interruption due to COVID-19 in 2020 and no remedial strategies. The pre-intervention (baseline) microfilarial prevalence in individuals ≥5 y of age is (A, D) 50%, (B, E) 60% and (C, F) 70%. The therapeutic coverage (of the total population) is assumed to be 25% in 2003 and 2004, 30% in 2005 and 2006, 50% in 2007 and 2008, 65% in 2009 to 2013, 30% in 2015 and 65% from 2016 onwards (motivated by the situation in West African countries that had experienced civil conflict and were affected by the Ebola epidemic), with the exception of 2021 (50%). Black and grey lines indicate, respectively, the microfilarial dynamics with and without an interruption due to COVID-19. The proportion of systematic non-participation is set to 5% throughout all simulations.
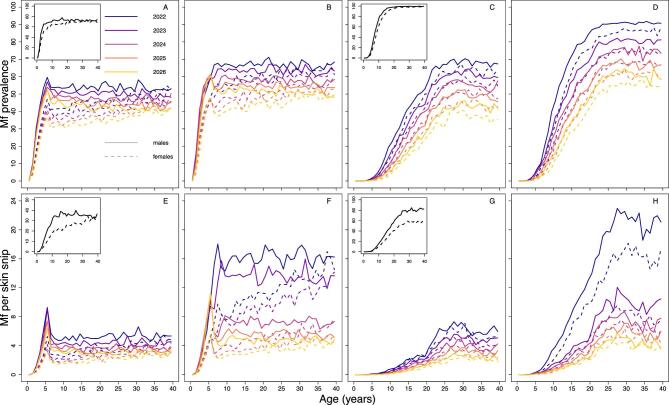
Age profiles of microfilarial prevalence and intensity
EPIONCHO-IBM and ONCHOSIM predict noticeably different age (and sex) patterns of microfilarial prevalence and intensity after three rounds of annual ivermectin MDA, with or without a 2-y (2020–2021) interruption (with MDA starting in 2017 and initial microfilarial prevalence of 70%) (Figure 7). EPIONCHO-IBM predicts higher prevalence and intensity in children <10 y of age following a 2-y MDA interruption than when there is no MDA interruption, and this is particularly pronounced in boys (Figure 7A and B for prevalence; Figure 7E and F for intensity). Following the interruption, ONCHOSIM predicts very low infection prevalence and intensity in children <5 y of age and an evident, but smaller, increase in 5- to 10-year-olds than in EPIONCHO-IBM (Figure 7C and D for prevalence; Figure 7G and H for intensity). In contrast, ONCHOSIM predicts a marked microfilarial prevalence and intensity increase in individuals ≥20 y of age compared with EPIONCHO-IBM. (The baseline profiles are shown as insets in Figure 7; individual-level microfilarial load is illustrated in Supplementary Figure S11.)
Figure 7.
Age- and sex-profiles of O. volvulus microfilarial (A–D) prevalence (%) and (E–H) mean intensity (mf/skin snip) predicted by (A, B, E, F) EPIONCHO-IBM and (C, D, G, H) ONCHOSIM assuming no interruption in MDA with ivermectin (A, C, E, G) and a 2-y interruption in MDA (B, D, F, H) in 2020 and 2021 due to COVID-19. The pre-intervention (baseline) microfilarial prevalence is 70% in individuals ≥5 y of age and the age profiles for (A, C) prevalence and (E, G) intensity at baseline are shown in the figure insets. Annual MDA starts in 2017 and the profiles (solid lines for males and dashed lines for females) are shown for 2022 (black), 2023 (violet), 2024 (purple), 2025 (red) and 2026 (yellow). The therapeutic coverage (of the total population) is assumed to be 65%, with the exception of 30% in 2022 (B, D, F, H). The proportion of systematic non-participation is set to 5% throughout all simulations.
Discussion
While in the grip of the COVID-19 pandemic in April 2020, the WHO recommended suspending all epidemiological surveys and MDA activities for NTDs to help curtail the transmission and spread of the SARS-CoV-2 virus.1 These disruptions to ivermectin MDA created concern in the NTD community regarding potential short-term increases in transmission and impacts on longer-term elimination prospects for onchocerciasis, particularly in the context of the EOT goals proposed by the WHO in its 2021–2030 NTD Roadmap.5 In response to these concerns, we used two stochastic onchocerciasis transmission models, EPIONCHO-IBM and ONCHOSIM, to investigate the impact of (1- and 2-y) interruptions to MDA in a variety of settings motivated by the epidemiological situation in endemic areas in Africa.
Generally the models suggest that programmes with short treatment histories of pre-existing annual MDA (late-start programmes) will be the most vulnerable to a 2-y (2020–2021) interruption. Programmes with longer treatment histories (early-start programmes) could also be adversely affected if initial endemicity levels indicate intense (hyperendemic) transmission. The influence of baseline microfilarial prevalence was more pronounced in EPIONCHO-IBM for both early- and late-start MDA programmes than in ONCHOSIM. Although for late-start programmes a 1-y interruption to MDA impacted the elimination probabilities and microfilarial prevalence in 2030, the effect would be more tolerable than for a 2-y interruption; early-start programmes with lower pre-intervention endemicities were mostly unaffected.
In July 2020, the WHO issued guidelines for the resumption of MDA provided it can be delivered ‘safely’ following a case-by-case risk–benefit assessment, with due consideration of a health system's capacity to conduct such modified activities effectively in the context of the ongoing COVID-19 pandemic.2 Therefore the implementation of mitigation strategies in the models allowed for a gradual scaling-up of therapeutic coverage once MDA resumes. Both models predict that remedial biannual MDA (in 2024 and 2025, after having demonstrated up-scaling to 65% coverage through 2022 and 2023) would be more effective at controlling increases in infection and decreases in elimination probabilities than remedial high (75%) coverage of annual MDA (also in 2024 and 2025).
Both models also predicted that pre-existing biannual MDA programmes—which had achieved a high treatment coverage for nearly a decade (in our simulations 75% pre-COVID-19 coverage from 2012 inclusive to reflect a 90% coverage of eligible population)—would not be adversely affected by a 1-y interruption. This was despite assuming a slow start of annual MDA at low coverage (before implementing biannual MDA) to capture initial difficulties in implementing treatment.17 A 1-y (rather than a 2-y) interruption was explored for these settings because of their pre-COVID-19 strong programmatic performance.17 It was also assumed that these programmes would be able to resume biannual MDA at the same high coverage they had recorded before the pandemic given their existing structure and commitment to achieving EOT.
Some West African countries (e.g. Guinea, Liberia, Sierra Leone) had already had their ivermectin MDA programmes interrupted in 2014 due to the Ebola epidemic. We therefore considered this situation in addition to a 1-y COVID-19 interruption in 2020. Both models predicted that in meso-endemic settings (50% baseline microfilarial prevalence), these two separate 1-y interruptions would not greatly impact a programmes’ performance, provided that coverage levels can catch up reasonably quickly following resumption of MDA (50% in 2021 and 65% in 2022 onwards). In hyperendemic and highly hyperendemic settings (60% and 70% baseline microfilarial prevalence) the impact would be more pronounced (particularly according to EPIONCHO-IBM).
Although programmes measure progress in terms of reducing mean infection prevalence and intensity at the population level, favourable infection trends may mask heterogeneities among different population subgroups. In areas of intense transmission and only recent MDA implementation, which could be exemplified by the situation of the Maridi Dam in South Sudan,22 children may reach their fifth birthday (when they become eligible for ivermectin treatment) with a sizeable microfilarial load. This has been shown to be a significant risk factor for the development of epilepsy later in life,9 ocular lesions23 and could also contribute to increasing the relative risk of mortality, which for a given microfilarial load is higher in children than in adults.24 In the Maridi villages, Ov-16 serology (a marker of exposure to infection) revealed a 20% (95% confidence interval 13 to 29) seroprevalence in 3- to 6-year-olds.22 Following a 2-y MDA interruption, children may not receive treatment until their seventh birthday, leading to a further microfilarial load build-up. Our modelling results indicate that in late-start (e.g. 2017) and high initial endemicity programmes (e.g. 70% microfilarial prevalence), there might be substantial increases in infection intensity in children <10 y of age, particularly according to EPIONCHO-IBM. In addition, both models predict that in these settings, and following a 2-y MDA interruption, there will also be increases in infection prevalence and intensity in older age groups (moderate in EPIONCHO-IBM but pronounced in ONCHOSIM). Although here we do not explicitly model morbidity, this increase could lead to exacerbation of other onchocerciasis-associated sequelae, such as troublesome itch and acute ocular lesions (due to microfilaria in the anterior eye chamber),25 according to epidemiological8 and modelling studies linking infection and disease.26,27 These results are mostly determined by the assumed patterns of age- and sex-dependent exposure to infection, which differ markedly between the two models.28
Although the models agree qualitatively on the role of treatment history in the outcome of interruptions to ivermectin MDA, and the differences between remedial strategies in mitigating the ensuing setbacks, ONCHOSIM predicts higher elimination probabilities than EPIONCHO-IBM. This is partly because EPIONCHO-IBM assumes strong regulatory processes operating on parasite establishment within humans that are relaxed as transmission declines during interventions, increasing the stability and resilience of the host–parasite system; these processes are not assumed in ONCHOSIM.12,28,29 In addition, the aforementioned differences in the assumed age- and sex-specific exposure patterns, and associated age and sex profiles of infection, results in ONCHOSIM predicting a lower microfilarial intensity in children <5 y of age, in EPIONCHO-IBM there exists a larger reservoir of infection in untreated children, which also contributes to lower elimination probabilities.
An assumption made in most of the modelled scenarios is that when MDA resumes, coverage will initially be lower than pre-pandemic levels and could take several years to recover. However, the usual drug distribution modality, by which members of the community present at a focal point to receive ivermectin, could be replaced by door-to-door drug distribution (as a result of social distancing measures). Depending on local circumstances, some programmes may achieve well-documented and high coverage levels, although the latter would also rely on well-stocked supply chains.
We did not consider the potential use of moxidectin and/or vector control as alternative remedial strategies to mitigate the impact of the current pandemic. This is because, while these approaches are very promising,30–32 they are currently not operationally implemented by national control programmes. However, where vector control is practicable, it should be considered as a complementary intervention when it is more conducive than chemotherapeutic approaches to implementation in a socially distanced manner.2
Conclusions
Both EPIONCHO-IBM and ONCHOSIM indicate that ivermectin MDA programmes with shorter (annual) treatment histories will be most vulnerable to MDA interruptions caused by the COVID-19 pandemic, particularly if treatment cannot be resumed safely in 2021. This impact may be noticeable in local infection resurgence in higher-endemicity settings and may reduce the probability of achieving EOT by 2030. Programmes with longer treatment histories of annual MDA that have achieved and maintained coverage of 65% are predicted to be less affected (but not totally impervious) by interruptions of ivermectin MDA, particularly if high initial endemicity indicates highly propitious transmission conditions. Young children have the potential to be negatively affected by increased levels of transmission resulting from missed MDA rounds, particularly for a 2-y interruption in highly endemic (≥70% pre-intervention microfilarial prevalence) settings recently incorporated into ivermectin MDA programmes, depending on local age-exposure patterns.
The impact of COVID-19 on progress towards the WHO 2021–2030 goals is best ameliorated by implementing biannual MDA as soon as pre-COVID-19 levels of therapeutic coverage are restored (in our simulations, to 65% of the total population in 2024–2025). This mitigation strategy is indicated as more effective than increasing annual MDA coverage (to 75%).
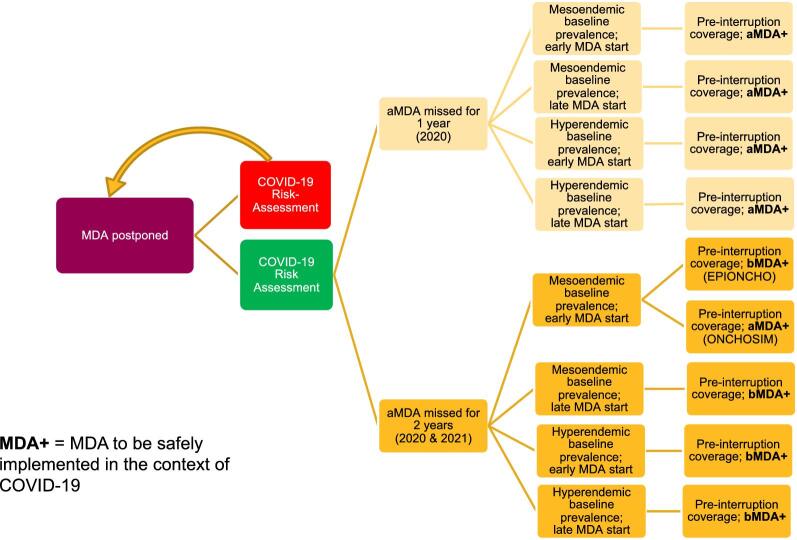
MDA programmes should promptly conduct the risk–benefit evaluations indicated by the WHO,2,33 so alternative modalities of MDA delivery can be put in place safely and effectively to resume MDA and regain pre-COVID-19 levels of coverage where possible (e.g. door-to-door distribution with household members measuring themselves with disinfected height poles and community drug distributors leaving tablets at the doorstep and observing household members swallowing the tablets with safe social distancing). Additionally, in August 2020, the United States Agency for International Development (USAID) released guidance on how best to implement MDA in the context of COVID-19.34 To complement the WHO and USAID documentation, we offer a decision-making tree to summarise our results and recommendations, which we hope will help programme managers to navigate the landscape of ivermectin MDA for onchocerciasis during the ongoing pandemic (Figure 8).
Figure 8.
Decision tree for implementing ivermectin MDA following a 1- or 2-y interruption in treatment programmes as a result of COVID-19. COVID-19 risk assessment conducted but risk deemed too high to safely implement MDA is represented by the red box, in which case MDA is postponed until subsequent risk assessments allow treatment activities to recommence. COVID-19 risk assessment conducted and deemed that MDA can resume safely is represented by the green box. MDA strategy upon resumption is based on the duration of the interruption, baseline prevalence and treatment history. MDA interruption (or missed MDA) is defined as no MDA in the period specified, i.e. 1 y (≥12 months since the last round of MDA) or 2 y (≥24 months since the last round of MDA). One year of missed annual MDA followed by a remedial biannual round in the following year can be interpreted as delayed MDA. Pre-interruption coverage assumes that before COVID-19 programmes had reached the minimum effective coverage of 65% of the total population and that this value will be reached following resumption of MDA. Early MDA start: annual programmes starting MDA in 2000; late MDA start: annual programmes starting MDA in 2017 (these start times were selected as they are at the two extremes of the treatment durations simulated); aMDA: annual MDA; bMDA: biannual MDA; MDA+: MDA implemented following the WHO guidelines for minimising the risk of COVID-19 transmission.2,34
Supplementary Material
Acknowledgements
None.
Contributor Information
Jonathan I D Hamley, London Centre for Neglected Tropical Disease Research, Department of Infectious Disease Epidemiology, School of Public Health, Faculty of Medicine (St Mary's campus), Imperial College London, Norfolk Place, London W2 1PG, UK; MRC Centre for Global Infectious Disease Analysis, Department of Infectious Disease Epidemiology, School of Public Health, Faculty of Medicine (St Mary's campus), Imperial College London, Norfolk Place, London W2 1PG, UK.
David J Blok, Department of Public Health, Erasmus MC, University Medical Center Rotterdam, P.O. Box 2040, 3000 CA Rotterdam, The Netherlands.
Martin Walker, London Centre for Neglected Tropical Disease Research, Department of Infectious Disease Epidemiology, School of Public Health, Faculty of Medicine (St Mary's campus), Imperial College London, Norfolk Place, London W2 1PG, UK; London Centre for Neglected Tropical Disease Research (LCNTDR), Department of Pathobiology and Population Sciences, Royal Veterinary College, University of London, Hatfield AL9 7TA, UK.
Philip Milton, London Centre for Neglected Tropical Disease Research, Department of Infectious Disease Epidemiology, School of Public Health, Faculty of Medicine (St Mary's campus), Imperial College London, Norfolk Place, London W2 1PG, UK; MRC Centre for Global Infectious Disease Analysis, Department of Infectious Disease Epidemiology, School of Public Health, Faculty of Medicine (St Mary's campus), Imperial College London, Norfolk Place, London W2 1PG, UK.
Adrian D Hopkins, Neglected and Disabling Diseases of Poverty Consultant, Kent, UK.
Louise C Hamill, Sightsavers, 35 Perrymount Road, Haywards Heath, RH16 3BW, UK.
Philip Downs, Sightsavers, 35 Perrymount Road, Haywards Heath, RH16 3BW, UK.
Sake J de Vlas, Department of Public Health, Erasmus MC, University Medical Center Rotterdam, P.O. Box 2040, 3000 CA Rotterdam, The Netherlands.
Wilma A Stolk, Department of Public Health, Erasmus MC, University Medical Center Rotterdam, P.O. Box 2040, 3000 CA Rotterdam, The Netherlands.
Maria-Gloria Basáñez, London Centre for Neglected Tropical Disease Research, Department of Infectious Disease Epidemiology, School of Public Health, Faculty of Medicine (St Mary's campus), Imperial College London, Norfolk Place, London W2 1PG, UK; MRC Centre for Global Infectious Disease Analysis, Department of Infectious Disease Epidemiology, School of Public Health, Faculty of Medicine (St Mary's campus), Imperial College London, Norfolk Place, London W2 1PG, UK.
Authors’ contributions
JIDH was responsible for conceptualization, methodology, investigation, formal analysis, software, visualization, writing the original draft and review and editing of the manuscript. DJB was responsible for conceptualization, formal analysis, software and review and editing of the manuscript. MW was responsible for conceptualization, methodology, supervision and review and editing of the manuscript. PM was responsible for methodology and review and editing of the manuscript. ADH and LCH were responsible for conceptualization and review and editing of the manuscript. PD was responsible for visualization and review and editing of the manuscript. SJdV was responsible for funding acquisition, supervision and review and editing of the manuscript. WAS was responsible for conceptualization, supervision and review and editing of the manuscript. MGB was responsible for conceptualization, methodology, funding acquisition, supervision, visualization, writing the original draft and review and editing of the manuscript.
Funding
This work was supported by the Bill and Melinda Gates Foundation via the NTD Modelling Consortium (grant OPP1184344), the UK Medical Research Council (MRC Doctoral Training Programme award to PM), MRC and the UK Department for International Development (DFID) Joint Centre Funding under the MRC/DFID concordat agreement of the European Union and European and Developing Countries Clinical Trials Partnership 2 (grant MR/R015600/1 to JIDH, PM and MGB).
Competing interests
None declared.
Data availability
No new data were used in this study. The code for EPIONCHO-IBM is available at https://github.com/jonathanhamley/EPIONCHO-IBM and the code for ONCHOSIM is available at https://gitlab.com/erasmusmc-public-health/wormsim.previous.versions/-/blob/master/wormsim-2.58Ap27.zip.
References
- 1. World Health Organization . COVID-19: WHO Issues Interim Guidance for Implementation of NTD Programmes. Available from: https://www.who.int/neglected_diseases/news/COVID19-WHO-interim-guidance-implementation-NTD-programmes/en/ [accessed 16 December 2020]. [Google Scholar]
- 2. World Health Organization . Considerations for implementing mass treatment, active case-finding and population-based surveys for neglected tropical diseases in the context of the COVID-19 pandemic. Available from: https://www.who.int/publications/i/item/WHO-2019-nCoV-neglected-tropical-diseases-2020-1 [accessed 16 December 2020]. [Google Scholar]
- 3. Parpia AS, Ndeffo-Mbah ML, Wenzel NSet al. Effects of response to 2014–2015 Ebola outbreak on deaths from malaria, HIV/AIDS, and tuberculosis, West Africa. Emerg Infect Dis. 2016;22(3):433–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Sochas L, Channon AA, Nam S.. Counting indirect crisis-related deaths in the context of a low-resilience health system: the case of maternal and neonatal health during the Ebola epidemic in Sierra Leone. Health Policy Plan. 2017;32(Suppl 3):iii32‒9. [DOI] [PubMed] [Google Scholar]
- 5. World Health Organization . Ending the Neglect to Attain the Sustainable Development Goals. A road map for neglected tropical diseases 2021–2030. Available from: https://www.who.int/neglected_diseases/Ending-the-neglect-to-attain-the-SDGs–NTD-Roadmap.pdf?ua = 1 [accessed 16 December 2020]. [Google Scholar]
- 6. Molyneux DH, Aboe A, Isiyaku Set al. COVID-19 and neglected tropical diseases in Africa: impacts, interactions, consequences. Int Health. 2020;12(5):367‒72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Chaumont C, Kamara K, Baring Eet al. The SARS-CoV-2 crisis and its impact on neglected tropical diseases: threat or opportunity? PLoS Negl Trop Dis. 2020;14(9):e0008680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Brieger WR, Awedoba AK, Eneanya CIet al. The effects of ivermectin on onchocercal skin disease and severe itching: results of a multicentre trial. Trop Med Int Health. 1998;3(12):951‒61. [DOI] [PubMed] [Google Scholar]
- 9. Chesnais CB, Nana-Djeunga HC, Njamnshi AKet al. The temporal relationship between onchocerciasis and epilepsy: a population-based cohort study. Lancet Infect Dis. 2018;18(11):1278‒86. [DOI] [PubMed] [Google Scholar]
- 10. NTD Modelling Consortium Onchocerciasis Group . The World Health Organization 2030 goals for onchocerciasis: insights and perspectives from mathematical modelling. Gates Open Res. 2019;3:1545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. NTD Modelling Consortium . The potential impact of programmes interruptions due to COVID-19 on 7 neglected tropical diseases: a modelling-based analysis. Available from: https://gatesopenresearch.org/documents/4-115 [accessed 16 December 2020]. [Google Scholar]
- 12. Hamley JID, Milton P, Walker Met al. Modelling exposure heterogeneity and density dependence in onchocerciasis using a novel individual-based transmission model, EPIONCHO-IBM: implications for elimination and data needs. PLoS Negl Trop Dis. 2019;13(12):e0007557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Coffeng LE, Stolk WA, Hoerauf Aet al. Elimination of African onchocerciasis: modeling the impact of increasing the frequency of ivermectin mass treatment. PLoS One. 2014;9(12):e115886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Behrend MR, Basáñez MG, Hamley JIDet al. Modelling for policy: the five principles of the Neglected Tropical Diseases Modelling Consortium. PLoS Negl Trop Dis. 2020;14(4):e0008033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Boatin B. The Onchocerciasis Control Programme in West Africa (OCP). Ann Trop Med Parasitol. 2008;102(Suppl 1):13–7. [DOI] [PubMed] [Google Scholar]
- 16. Tekle AH, Zouré HG, Noma Met al. Progress towards onchocerciasis elimination in the participating countries of the African Programme for Onchocerciasis Control: epidemiological evaluation results. Infect Dis Poverty. 2016;5:66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Katabarwa MN, Lakwo T, Habomugisha Pet al. After 70 years of fighting an age-old scourge, onchocerciasis in Uganda, the end is in sight. Int Health. 2018;10(Suppl 1):i79–i88. [DOI] [PubMed] [Google Scholar]
- 18. Carter Center, Ministry of Health Uganda . Insight Special 2011. Proceedings of Uganda Onchocerciasis Elimination Expert Advisory Committee. 4th Session, 15–17 August 2011, Kampala, Uganda. Available from: https://mhjapply.cartercenter.org/resources/pdfs/news/health_publications/river_blindness/insight-special-newsletter-2011.pdf [accessed 16 December 2020]. [Google Scholar]
- 19. Hodges ME, Koroma JB, Sonnie Met al. Neglected tropical disease control in post-war Sierra Leone using the Onchocerciasis Control Programme as a platform. Int Health. 2011;3(2):69‒74. [DOI] [PubMed] [Google Scholar]
- 20. Thomas BC, Kollie K, Koudou Bet al. Commentary: Restarting NTD programme activities after the Ebola outbreak in Liberia. Infect Dis Poverty. 2017;6(1):52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Koroma JB, Sesay S, Conteh Aet al. Impact of five annual rounds of mass drug administration with ivermectin on onchocerciasis in Sierra Leone. Infect Dis Poverty. 2018;7(1):30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Lakwo TL, Raimon S, Tionga Met al. The role of the Maridi Dam in causing an onchocerciasis-associated epilepsy epidemic in Maridi, South Sudan: an epidemiological, sociological, and entomological study. Pathogens. 2020;9(4):315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Renz A, Fuglsang H, Anderson J.. Studies on the dynamics of transmission of onchocerciasis in a Sudan-savanna area of North Cameroon IV. The different exposure to Simulium bites and transmission of boys and girls and men and women, and the resulting manifestations of onchocerciasis. Ann Trop Med Parasitol. 1987;81(3):253‒62. [DOI] [PubMed] [Google Scholar]
- 24. Walker M, Little MP, Wagner KSet al. Density-dependent mortality of the human host in onchocerciasis: relationships between microfilarial load and excess mortality. PLoS Negl Trop Dis. 2012;6(3):e1578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hopkins A, Boatin BA.. Onchocerciasis. In: Selendy JMH (ed). Water and sanitation-related diseases and the environment: challenges, interventions, and preventive measures. First edition. Hoboken, NJ: John Wiley & Sons;2011, p. 133‒49. [Google Scholar]
- 26. Coffeng LE, Stolk WA, Zouré HGet al. African Programme for Onchocerciasis Control 1995–2015: model-estimated health impact and cost. PLoS Negl Trop Dis. 2013;7(1):e2032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Turner HC, Walker M, Churcher TSet al. Modelling the impact of ivermectin on River Blindness and its burden of morbidity and mortality in African savannah: EpiOncho projections. Parasit Vectors. 2014;7:241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Basáñez MG, Walker M, Turner HCet al. River blindness: mathematical models for control and elimination. Adv Parasitol. 2016;94:247–341. [DOI] [PubMed] [Google Scholar]
- 29. Hamley JID, Walker M, Coffeng LEet al. Structural uncertainty in onchocerciasis transmission models influences the estimation of elimination thresholds and selection of age groups for seromonitoring. J Infect Dis. 2020;221(Suppl 5):S510–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Milton P, Hamley JID, Walker Met al. Moxidectin: an oral treatment for human onchocerciasis. Expert Rev Anti Infect Ther. 2020;18(11):1067–81. [DOI] [PubMed] [Google Scholar]
- 31. Jacob BG, Loum D, Lakwo TLet al. Community-directed vector control to supplement mass drug distribution for onchocerciasis elimination in the Madi mid-North focus of Northern Uganda. PLoS Negl Trop Dis. 2018;12(8):e0006702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Katabarwa MN, Habomugisha P, Khainza Aet al. Elimination of Simulium neavei-transmitted onchocerciasis in Wambabya-Rwamarongo focus of Western Uganda. Am J Trop Med Hyg. 2020;103(3):1135–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. World Health Organization . Rapid risk assessment of acute public health events. Available from: https://www.who.int/csr/resources/publications/HSE_GAR_ARO_2012_1/en/ [accessed 16 December 2020]. [Google Scholar]
- 34. United States Agency for International Development , Act to end NTDs East, Act to end NTDs West. Practical approaches to implementing WHO guidance for neglected tropical disease (NTD) programs in the context of COVID-19: mass drug administration (MDA). Available from: https://www.ntdtoolbox.org/toolbox-search/practical-approaches-implementing-who-guidance-neglected-tropical-disease-ntd [accessed 16 December 2020]. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No new data were used in this study. The code for EPIONCHO-IBM is available at https://github.com/jonathanhamley/EPIONCHO-IBM and the code for ONCHOSIM is available at https://gitlab.com/erasmusmc-public-health/wormsim.previous.versions/-/blob/master/wormsim-2.58Ap27.zip.