Abstract
Background
Understanding risk factors of symptomatic coronavirus disease 2019 (COVID-19) vis-à-vis asymptomatic severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, severe disease and death is important.
Methods
An unmatched case–control study was conducted through telephonic interviews among individuals who tested positive for SARS-CoV-2 in Jodhpur, India from 23 March to 20 July 2020. Contact history, comorbidities and tobacco and alcohol use were elicited using standard tools.
Results
Among 911 SARS-CoV-2-infected individuals, 47.5% were symptomatic, 14.1% had severe COVID-19 and 41 (4.5%) died. Older age, working outside the home, cardiac and respiratory comorbidity and alcohol use were found to increase the risk of symptomatic disease as compared with asymptomatic infection. Current tobacco smoking (odds ratio [OR] 0.46 [95% confidence interval {CI} 0.26 to 0.78]) but not smokeless tobacco use (OR 0.81 [95% CI 0.55 to 1.19]) appeared to reduce the risk of symptomatic disease. Age ≥60 y and renal comorbidity were significantly associated with severe COVID-19. Age ≥60 y and respiratory and cardiac comorbidity were found to predispose to mortality.
Conclusions
The apparent reduced risk of symptomatic COVID-19 among tobacco smokers could be due to residual confounding owing to unknown factors, while acknowledging the limitation of recall bias. Cross-protection afforded by frequent upper respiratory tract infection among tobacco smokers could explain why a similar association was not found for smokeless tobacco use, thereby being more plausible than the ‘nicotinic hypothesis’. Those with comorbidities and age ≥60 y should be prioritized for hospital admission.
Keywords: alcohol, case-control, COVID-19, cross-protection, nicotine hypothesis, tobacco
Introduction
Coronavirus disease 2019 (COVID-19) has emerged as a pandemic with 83.3 million confirmed cases and 1.8 million deaths worldwide as on 5 January 2021.1 The humanitarian and economic toll of this disease has been unprecedented. It emerged in China, where the first case was reported from Wuhan in early December 2019.2 With >10 million cumulative COVID-19 cases, India is now the second most affected country in the world after the United States.1
Asymptomatic infection has been implicated in undetected transmission and the large size of COVID-19 outbreaks.3 The risk factors for severe COVID-19 disease resulting in the requirement for ventilation and death have been studied among patients admitted to hospitals.4,5 These include comorbidities such as diabetes, heart disease, chronic obstructive pulmonary disease and age >65 y.4,5 Similarly, it is important to find the risk factors that lead to an individual developing symptomatic COVID-19 disease vis-à-vis those who are infected but remain completely asymptomatic. It could be useful to identify those contacts who are at a higher risk of developing symptomatic disease and would be more likely to benefit from focused monitoring and follow-up in the community. This could also help in developing better strategies of symptomatic screening of COVID-19. Therefore we aimed to study the factors predisposing to symptomatic disease among the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)-infected individuals, especially with regards to age, gender, comorbidities, tobacco smoking, smokeless tobacco use, alcohol use and other epidemiological factors.
Methods
Study setting and eligibility for inclusion
Our institute covers Jodhpur and its adjoining districts in the western part of Rajasthan state in India (Figure 1). As per national guidelines, all symptomatic individuals with influenza-like illness who had a history of foreign travel within the past 14 d, a history of domestic migration within the past 7 d, were residing in hotspot or containment zones, had contact with a confirmed COVID-19 case and health workers involved in the care of COVID-19 cases were considered as suspected COVID-19 and were eligible for SARS-CoV-2 testing.6 Those with severe acute respiratory infection and asymptomatic contacts of COVID-19 cases were also eligible for testing.6 These individuals suspected of COVID-19 were being tested at our institute with nasopharyngeal and oropharyngeal swab real-time reverse transcription polymerase chain reaction (RT-PCR) for SARS-CoV-2 prior to admission.6 Additionally, field-level swab samples from among inbound travellers, contacts of confirmed COVID-19 cases and from areas designated by administration as having COVID-19 transmission (hotspot areas) were also being sent for testing at our institute.
Figure 1.
Location of the All India Institute of Medical Sciences and the approximate catchment area of Jodhpur and surrounding districts in western Rajasthan, India. (Modified from source file https://commons.wikimedia.org/wiki/File:India_districts_map.svg, Creative Commons Attribution-Share Alike 4.0 International license).
Study design and data collection procedure
A prospective observational study with unmatched case–control design was conducted among those individuals who had tested positive for SARS-CoV-2 and were admitted at our institute from 23 March to 20 July 2020. Owing to the introduction of guidelines for home isolation, we expanded our eligibility criteria to all those testing positive instead of only those being admitted during 16 May–8 June 2020. In order to minimize the risk of infection to interviewers, all eligible individuals were contacted by telephone and were interviewed upon providing verbal informed consent, usually 2–3 d after real-time RT-PCR test results were available. Telephone interviews provided a uniform data collection procedure for all study participants, whether admitted at our institute or undergoing isolation at home or an institutional facility. Information pertaining to individuals <18 y of age was provided by parents after they provided verbal informed consent. The study was approved by the institutional ethics committee (AIIMS/IEC/2020-21/1099).
Definition of cases, controls and severe COVID-19
All individuals detected as infected with SARS-CoV-2 and reporting at least one symptom were taken as symptomatic and were enrolled as ‘cases’ in the case–control study. A standard case investigation form developed by the National Centre for Disease Control, New Delhi, India was used for recording symptoms, comorbidities and epidemiological data of COVID-19 cases.7 Symptoms specifically assessed as per the format were fever, cough, sore throat, running nose, breathlessness, general weakness, headache, pain (muscular, chest, abdominal, joint), nausea/vomiting, diarrhoea and irritability/confusion. Any other symptoms reported by the patient but not specified in the format, such as anosmia, were also recorded and were considered for ascertainment as ‘cases’. Those ascertained as asymptomatic SARS-CoV-2-infected individuals were taken as ‘controls’.
Of the SARS-CoV-2-infected individuals, those admitted with clinical signs of pneumonia plus respiratory rate >30 breaths/min, severe respiratory distress or peripheral capillary oxygen saturation <90% on room air were defined as having severe COVID-19 as per in national clinical management protocol.8 Comorbidities were self-reported by study participants at the time of telephonic interview, which reflected diagnoses prior to admission or isolation as well as those made during the initial days of hospital admission. Additional data related to plausible risk factors such as tobacco smoking, smokeless tobacco and alcohol use were collected using a structured questionnaire adopted from the World Health Organization (WHO) STEPwise Approach to Surveillance instrument.9 Smoking was further quantified into cigarette pack-years. Bidi smoking was also converted to equivalent cigarette pack-years using a conversion factor of 2.64 bidis equalling 1 commercial filtered cigarette, based on nicotine content.10 Occupational history was obtained and was broadly classified as those requiring movement outside the home and those mostly staying home, such as homemakers and retirees.
Outcome assessment
The outcomes of the patients were subsequently obtained from the official report provided on a daily basis to the state government by our institute. Additionally, all individuals, whether admitted or in isolation, were followed up by telephone 30 d after detection of SARS-CoV-2 infection to have their final outcome updated and to cross-check their history of tobacco smoking, smokeless tobacco use and alcohol use. Death in the study was defined as death in a SARS-CoV-2-infected individual within 30 d of diagnosis. The availability of tobacco products and alcohol remained restricted after the first countrywide lockdown of 25 March 2020. Therefore a 1-month period prior to lockdown was taken as the reference period for assessing current tobacco or alcohol use.
Sample size calculation
We considered a proportion of approximately 60% asymptomatic individuals among those testing positive, yielding an asymptomatic: symptomatic ratio of 1.5, based on a previous report.11 Related to the primary outcome of interest, a tobacco use prevalence of 28.6% was used for controls, similar to that of the Indian population as per the Global Adult Tobacco Survey 2016–17.12 In the absence of prior estimates, we assumed that tobacco use increased the odds of symptomatic disease among SARS-CoV-2-infected individuals by 1.5 times. This yielded a total sample size of 900 individuals with 80% power and an α error of 5% using the OpenEpi tool.13 We attempted to universally sample each eligible individual until the required sample size was reached.
Data analysis
Continuous variables were described using mean and standard deviation (SD) or median and interquartile range (IQR), where appropriate. Categorical variables related to risk factors were tabulated and odds ratios (ORs) were calculated both using univariate analysis and multivariate logistic regression in order to adjust for potential confounding. Analysis was done separately for all three dependent variables—symptomatic vs asymptomatic, severe COVID-19 vs mild or moderate COVID-19 and dead vs alive outcome. Multivariate analysis was done with model 1 taking all plausible risk factors together and model 2 taking all risk factor together with tobacco smoking categorized in pack-years instead of a binary variable. For multivariate models, similar variables were not analysed together to avoid collinearity. Data analysis was conducted using Stata version 16 (StataCorp, College Station, TX, USA). A p-value <0.05 was considered significant.
Results
Clinical and epidemiological characteristics
Of a total of 1203 eligible SARS-CoV-2-infected individuals whom we attempted to contact, 911 provided consent and could be interviewed. Around half (433 [47.5%]) were symptomatic and 128 (14.1%) were classified as having severe COVID-19. From among the symptomatic COVID-19 patients, fever was the most common self-reported symptom in almost two-thirds (280 [64.7%]), followed by cough (146 [33.7%]), sore throat (89 [20.6%]), breathlessness (72 [16.6%]), generalized weakness (59 [13.6%]), myalgia (34 [7.9%]), runny nose (32 [7.4%]), headache (22 [5.1%]), anosmia (14 [3.2%]) and other symptoms (32 [7.4%]).
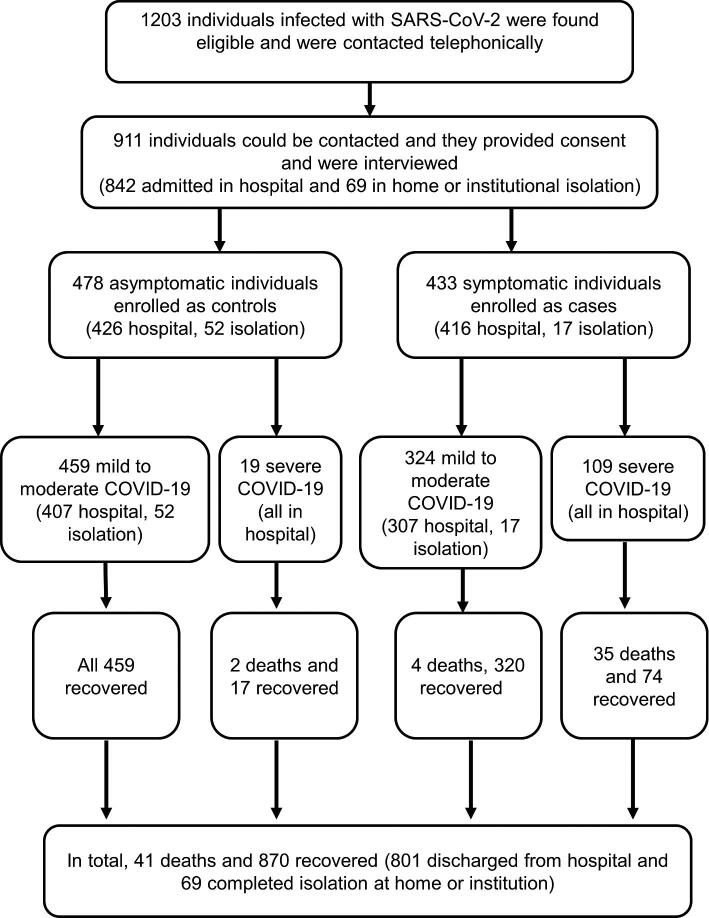
Of the 911 study participants, 41 (4.9%) died of COVID-19-related complications and 870 (95.1%) recovered and were alive at the 1-month follow-up (Figure 2). Among the recovered, 801 patients were discharged from the hospital and 69 successfully completed isolation at their home or institutional facility (Table 1 and Figure 2). The age and gender of symptomatic and asymptomatic individuals were similarly distributed (Table 1). The median duration from symptom onset to COVID-19 testing was 2 d and testing to admission was 1 d. The median duration from COVID-19 testing to interview was 5 d (IQR 3–7) and 5 d (IQR 4–7) among asymptomatic and symptomatic individuals, respectively (Table 1).
Figure 2.
Flowchart of recruitment of SARS-CoV-2-infected individuals for the case–control study, their clinical severity and outcomes.
Table 1.
Demographic and clinical characteristics of asymptomatic and symptomatic SARS-CoV-2-infected individuals
| Asymptomatic | Symptomatic | Total | |
|---|---|---|---|
| Characteristics | (n=478) | (n=433) | (N=911) |
| Demographic details | |||
| Age (years), mean (SD) | 42.1 (19.8) | 44.2 (18.4) | 43.1 (19.2) |
| Age group (years), n (%) | |||
| 0–9 | 22 (4.6) | 12 (2.8) | 34 (3.7) |
| 10–19 | 39 (8.2) | 18 (4.2) | 57 (6.3) |
| 20–29 | 93 (19.5) | 77 (17.8) | 170 (18.7) |
| 30–39 | 70 (14.6) | 70 (16.2) | 140 (15.4) |
| 40–49 | 57 (11.9) | 79 (18.2) | 136 (14.9) |
| 50–59 | 80 (16.7) | 70 (16.2) | 150 (16.5) |
| 60–69 | 66 (13.8) | 73 (16.9) | 139 (15.3) |
| ≥70 | 51 (10.7) | 34 (7.9) | 85 (9.3) |
| Gender, n (%) | |||
| Female | 170 (34.6) | 151 (34.9) | 321 (35.2) |
| Male | 308 (64.4) | 282 (65.1) | 590 (64.8) |
| Comorbidities, n (%) | |||
| Any comorbidity | 134 (28.0) | 165 (38.1) | 299 (32.8) |
| Diabetes | 64 (13.4) | 66 (15.2) | 130 (14.3) |
| Hypertension | 70 (14.6) | 79 (18.2) | 149 (16.4) |
| Diabetes or hypertension | 105 (22.0) | 116 (26.8) | 221 (24.3) |
| Diabetes and hypertension | 29 (6.1) | 29 (6.7) | 58 (6.4) |
| Respiratory | 5 (1.0) | 22 (5.1) | 27 (3.0) |
| Cardiac | 14 (2.9) | 28 (6.5) | 42 (4.6) |
| Renal | 3 (0.6) | 6 (1.4) | 9 (1.0) |
| Gastrointestinal | 1 (0.2) | 2 (0.5) | 3 (0.3) |
| Malignancy | 3 (0.6) | 7 (1.6) | 10 (1.1) |
| Health services and outcomes | |||
| Facility of care, n (%) | |||
| Home isolation | 31 (6.5) | 10 (2.3) | 41 (4.5) |
| Non-hospital institutional isolation | 21 (4.4) | 7 (1.6) | 28 (3.1) |
| Hospital | 426 (89.1) | 416 (96.1) | 842 (92.4) |
| Symptom onset to COVID-19 testing (days), median (IQR) | – | 2 (0–3) | 2 (0–3) |
| Testing to admission (days), median (IQR) | 1 (0–2) | 1 (0–2) | 1 (0–2) |
| Symptom onset to interview (days), median (IQR) | – | 7 (5–10) | 7 (5–10) |
| Testing to interview (days), median (IQR) | 5 (3–7) | 5 (4–7) | 5 (3–7) |
| COVID-19 severity, n (%) | |||
| Mild to moderate | 459 (96.0) | 324 (74.8) | 783 (85.1) |
| Severe | 19 (4.0) | 109 (25.2) | 128 (14.9) |
| Outcomes, n (%) | |||
| Completed isolation | 52 (10.9) | 17 (3.9) | 69 (7.6) |
| Discharged from hospital | 424 (88.7) | 377 (87.1) | 801 (87.9) |
| Death | 2 (0.4) | 39 (9.0) | 41 (4.5) |
| Time to discharge (days), median (IQR) | 8 (6–11) | 8 (7–10) | 8 (6–10) |
Epidemiological history revealed that those having contact with a confirmed COVID-19 case, belonging to a known cluster or hotspot of COVID-19 cases and having a history of travel were more likely to contribute to detection of asymptomatic SARS-CoV-2 infection as compared with symptomatic disease (Table 2). Nearly four-fifths of the SARS-CoV-2-infected individuals did not report any contact with a confirmed COVID-19 case (Table 2). The most common contact with another confirmed COVID-19 case was within the household setting for both symptomatic and asymptomatic individuals. Current smoking, the use of smokeless tobacco and the use of any form of tobacco was 8.1%, 15.3% and 22.1%, respectively, among all infected individuals. Current alcohol use was reported by 3.8%. The full study data set is also provided (supplementary file 1).
Table 2.
Epidemiological characteristics of asymptomatic and symptomatic SARS-CoV-2-infected individuals
| Asymptomatic | Symptomatic | Total | |
|---|---|---|---|
| Characteristics | (n=478) | (n=433) | (N=911) |
| Epidemiological history, n (%) | |||
| Occupation requiring movement outside the home | 250 (52.3) | 268 (61.9) | 518 (56.9) |
| Contact with a laboratory-confirmed COVID-19 case | 107 (22.4) | 72 (16.6) | 179 (19.6) |
| Type of contact with a confirmed COVID-19 case (among N=179), n (%) | |||
| From another member of the same household | 84 (78.5) | 39 (54.2) | 123 (68.7) |
| From neighbourhood | 10 (9.3) | 18 (25.0) | 28 (15.6) |
| During clinical care or visit to a place of sample collection | 7 (6.5) | 6 (8.3) | 13 (7.3) |
| At workplace or with a friend | 3 (2.8) | 8 (11.1) | 11 (6.1) |
| During travel | 3 (2.8) | 1 (1.4) | 4 (2.2) |
| Self-reporting of belonging to a COVID-19 cluster or hotspot area | 124 (25.9) | 85 (19.6) | 209 (22.9) |
| Attended mass gathering in the past month | 5 (1.0) | 11 (2.5) | 16 (1.8) |
| Any travel history in the past month | 100 (20.9) | 57 (13.2) | 157 (17.2) |
| Domestic travel outside district in the past month | 43 (9.0) | 52 (12.0) | 95 (10.4) |
| Foreign travel in the past month | 57 (11.9) | 5 (1.2) | 62 (6.8) |
| Tobacco and alcohol use, n (%) | |||
| Ever tobacco smoker (both current and past smokers) | 50 (10.5) | 30 (6.9) | 80 (8.8) |
| Past tobacco smokers | 3 (0.6) | 3 (0.7) | 6 (0.7) |
| Median duration of stopping tobacco smoking for past smokers (years) | 20 | 20 | 20 |
| Current tobacco smoker (both daily and occasional) | 47 (9.8) | 27 (6.2) | 74 (8.1) |
| Daily tobacco smoker | 44 (9.2) | 25 (5.8) | 69 (7.6) |
| Occasional tobacco smoker (less than daily) | 3 (0.6) | 2 (0.5) | 5 (0.5) |
| Type of product smoked among current tobacco smokers (N=74), n (%) | |||
| Cigarette | 26 (55.2) | 17 (63.0) | 43 (58.1) |
| Bidi | 20 (42.6) | 10 (37.0) | 30 (40.5) |
| Chillum | 1 (2.1) | 0 (0) | 1 (1.4) |
| Pack-years among current smokers (N=74), n (%) | |||
| <1 | 18 | 10 | 28 |
| 1–5 | 16 | 10 | 26 |
| >5 | 12 | 6 | 18 |
| Not applicable (for chillum)/data missing | 1 | 1 | 2 |
| Current user of smokeless tobacco | 74 (15.5) | 65 (15.0) | 139 (15.3) |
| Current user of any form of tobacco | 113 (23.6) | 87 (20.1) | 201 (22.1) |
| Currently smoker as well as using smokeless tobacco | 8 (1.7) | 4 (0.9) | 12 (1.3) |
| Current use of alcohol (at least once in the past month) | 12 (2.5) | 23 (5.3) | 35 (3.8) |
| Current regular use of alcohol (at least once a week) | 10 (2.1) | 21 (4.8) | 31 (3.4) |
| Current user of both tobacco and alcohol | 9 (1.9) | 9 (2.1) | 18 (2.0) |
Risk factors of symptomatic COVID-19
Upon univariate analysis, having an occupation requiring outdoor movement, the presence of any comorbidity, the presence of respiratory or cardiac comorbidity and the use of alcohol were found to be significantly associated with symptomatic COVID-19 as compared with asymptomatic SARS-CoV-2 infection. On the other hand, residing in a cluster or hotspot area and having a history of travel in the past month were associated with greater asymptomatic infection as compared with symptomatic disease.
Upon multivariate analysis with model 1, older age, especially the 40–49 and 60–69 y age groups, occupation requiring movement outside the home, respiratory comorbidity and alcohol use were found to increase the risk of symptomatic disease as compared with asymptomatic infection (Table 3). A history of travel in the past month and tobacco smoking appeared to reduce the risk of symptomatic disease, while the apparently protective association with residing in a cluster or hotspot area disappeared completely (Table 3). Multivariate analysis with model 2 additionally showed an association of asymptomatic course among those with >5 pack-years of tobacco smoking as compared with non-smokers. No significant association was found for an asymptomatic course among those smoking <1 pack-year or 1–5 pack-years as compared with non-smokers.
Table 3.
Univariate and multivariate analysis of plausible risk factors for developing symptomatic COVID-19 as compared with asymptomatic SARS-CoV-2 infection (N=911)
| Univariate | Multivariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| analysis | analysis (model 1) | analysis (model 2) | ||||
| OR | Adjusted OR | Adjusted OR | ||||
| Characteristics | (95% CI) | p-Value | (95% CI) | p-Value | (95% CI) | p-Value |
| Demographic and epidemiological characteristics | ||||||
| Male gender (vs female) | 1.02 (0.78 to 1.34) | 0.865 | 0.78 (0.56 to 1.11) | 0.401 | 0.79 (0.56 to 1.12) | 0.185 |
| Age (years) | 1.006 (0.999 to 1.012) | 0.111 | – | – | – | – |
| Age group (years) | ||||||
| 0–9 | Reference | – | Reference | – | Reference | – |
| 10–19 | 0.85 (0.34 to 2.08) | 0.715 | 0.68 (0.27 to 1.69) | 0.401 | 0.67 (0.27 to 1.69) | 0.400 |
| 20–29 | 1.52 (0.71 to 3.26) | 0.285 | 1.58 (0.72 to 3.45) | 0.250 | 1.58 (0.72 to 3.44) | 0.253 |
| 30–39 | 1.83 (0.84 to 3.99) | 0.127 | 2.11 (0.95 to 4.66) | 0.066 | 2.10 (0.95 to 4.66) | 0.067 |
| 40–49 | 2.54 (1.16 to 5.55) | 0.019 | 2.48 (1.11 to 5.88) | 0.028 | 2.45 (1.09 to 5.48) | 0.029 |
| 50–59 | 1.60 (0.74 to 3.48) | 0.231 | 1.83 (0.81 to 4.13) | 0.145 | 1.83 (0.81 to 4.13) | 0.145 |
| 60–69 | 2.03 (0.93 to 4.42) | 0.075 | 2.55 (1.11 to 5.88) | 0.028 | 2.58 (1.12 to 5.96) | 0.026 |
| ≥70 | 1.22 (0.53 to 2.79) | 0.064 | 1.86 (0.77 to 4.53) | 0.170 | 1.88 (0.77 to 4.59) | 0.163 |
| Residing in a cluster or hotspot area | 0.70 (0.51 to 0.96) | 0.029 | 1.00 (0.67 to 1.50) | 0.983 | 1.00 (0.67 to 1.49) | 0.996 |
| Any travel in the past month | 0.58 (0.40 to 0.82) | 0.002 | 0.57 (0.36 to 0.90) | 0.015 | 0.57 (0.36 to 0.89) | 0.015 |
| Occupation requiring movement outside the home | 1.48 (1.14 to 1.93) | 0.004 | 2.02 (1.41 to 2.90) | <0.0001 | 2.01 (1.40 to 2.89) | <0.0001 |
| Comorbidities | ||||||
| Any comorbidity | 1.61 (1.22 to 2.12) | 0.001 | – | – | – | – |
| Diabetes | 1.19 (0.82 to 1.73) | 0.359 | 1.06 (0.69 to 1.63) | 0.775 | 1.08 (0.70 to 1.66) | 0.725 |
| Hypertension | 1.33 (0.93 to 1.89) | 0.113 | 1.09 (0.71 to 1.68) | 0.702 | 1.07 (0.69 to 1.65) | 0.765 |
| Diabetes or hypertension | 1.32 (0.98 to 1.79) | 0.071 | – | – | – | – |
| Respiratory | 5.1 (1.91 to 13.55) | 0.001 | 4.43 (1.62 to 12.11) | 0.004 | 4.68 (1.69 to 12.95) | 0.003 |
| Cardiac | 2.30 (1.20 to 4.43) | 0.013 | 2.08 (0.99 to 4.35) | 0.052 | 2.10 (1.00 to 4.42) | 0.050 |
| Renal | 2.23 (0.56 to 8.99) | 0.258 | 1.69 (0.39 to 7.22) | 0.481 | 1.66 (0.39 to 7.12) | 0.495 |
| Tobacco and alcohol use | ||||||
| Current tobacco smoking | 0.61 (0.37 to 1.00) | 0.051 | 0.46 (0.26 to 0.78) | 0.005 | – | – |
| Pack-years of tobacco smoking among current smokers | ||||||
| 0 (non-smoker) | Reference | – | Reference | – | ||
| <1 | 0.59 (0.27 to 1.29) | 0.187 | 0.50 (0.21 to 1.15) | 0.101 | ||
| 1–5 | 0.66 (0.30 to 1.48) | 0.316 | 0.51 (0.21 to 1.22) | 0.130 | ||
| >5 | 0.53 (0.20 to 1.43) | 0.209 | 0.31 (0.10 to 0.95) | 0.041 | ||
| Current smokeless tobacco use | 0.97 (0.68 to 1.39) | 0.745 | 0.81 (0.55 to 1.19) | 0.284 | 0.81 (0.55 to 1.19) | 0.277 |
| Current use of any form of tobacco | 0.83 (0.60 to 1.14) | 0.242 | – | – | – | – |
| Current alcohol use | 2.19 (1.08 to 4.45) | 0.031 | 2.42 (1.12 to 5.24) | 0.024 | 2.39 (1.10 to 5.17) | 0.027 |
Model 1 parameters: log likelihood=593.75, minus 2 log likelihood difference vs intercept=73.19, df=19, p<0.0001, pseudo-R2=0.0581. Model 2 parameters: log likelihood=593.45, minus 2 log likelihood difference vs intercept=73.79, df=21, p<0.0001, pseudo-R2=0.0585.
Risk factors for severe COVID-19
Older age and comorbidities such as diabetes, hypertension and cardiac and renal disease were significantly associated with severe COVID-19 on univariate analysis. Furthermore, older age, especially the 60–69 and ≥70 y age groups, and renal comorbidity remained as risk factors for severe COVID-19 upon multivariate analysis with model 1 (Table 4). Residing in a cluster or hotspot area was associated with reduced severity of COVID-19. The magnitude of association of tobacco smoking with reduced severity of COVID-19 became non-significant (OR 0.47 [95% CI 0.21 to 1.08], p=0.074) as compared its significant association with reduced symptomatic COVID-19 (OR 0.46 [95% CI 0.26 to 0.78], p=0.008). The association of smokeless tobacco use with severe COVID-19 (OR 0.98 [95% CI 0.57 to 1.70], p=0.952) remained close to 1 as well as statistically non-significant, which was nearly similar to that for its association with symptomatic COVID-19 (OR 0.81 [95% CI 0.55 to 1.19], p=0.284). Model 2 showed similar results while also showing a marginally significant association of reduced severity of COVID-19 for those with tobacco smoking of ≥1 pack-years. Reduced severity of COVID-19 was not significantly associated with those smoking <1 pack-year.
Table 4.
Univariate and multivariate analysis of plausible risk factors for developing severe COVID-19 as compared with mild or moderate disease (N=911)
| Univariate | Multivariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| analysis | analysis (model 1) | analysis (model 2) | ||||
| OR | Adjusted OR | Adjusted OR | ||||
| Characteristics | (95% CI) | p-Value | (95% CI) | p-Value | (95% CI) | p-Value |
| Demographic and epidemiological characteristics | ||||||
| Male gender (vs female) | 1.28 (0.86 to 1.92) | 0.224 | 1.59 (0.93 to 2.71) | 0.087 | 1.61 (0.94 to 2.74) | 0.082 |
| Age (years) | 1.051 (1.039 to 1.064) | <0.001 | - | - | - | - |
| Age group (years) | ||||||
| 0–9 | Reference | – | Reference | – | Reference | – |
| 10–19 | 0.59 (0.36 to 9.74) | 0.900 | 0.61 (0.04 to 10.24) | 0.734 | 0.60 (0.04 to 10.08) | 0.725 |
| 20–29 | 0.80 (0.09 to 7.34) | 0.840 | 0.90 (0.10 to 8.38) | 0.927 | 0.87 (0.09 to 8.10) | 0.903 |
| 30–39 | 2.27 (0.28 to 18.53) | 0.445 | 2.55 (0.31 to 21.00) | 0.385 | 2.50 (0.30 to 20.64) | 0.395 |
| 40–49 | 4.71 (0.60 to 36.74) | 0.139 | 4.36 (0.55 to 34.52) | 0.163 | 4.20 (0.53 to 33.28) | 0.174 |
| 50–59 | 7.24 (0.95 to 55.30) | 0.056 | 7.61 (0.97 to 59.64) | 0.053 | 7.48 (0.95 to 58.66) | 0.055 |
| 60–69 | 16.32 (2.16 to 123.11) | 0.007 | 16.02 (2.05 to 125.00) | 0.008 | 16.15 (2.07 to 126.15) | 0.008 |
| ≥70 | 12.24 (1.58 to 94.73) | 0.016 | 12.18 (1.51 to 98.49) | 0.019 | 12.33 (1.52 to 99.77) | 0.019 |
| Residing in a cluster or hotspot area | 0.28 (0.15 to 0.53) | <0.001 | 0.38 (0.18 to 0.83) | 0.015 | 0.37 (0.17 to 0.81) | 0.012 |
| Any travel in the past month | 0.28 (0.14 to 0.59) | 0.001 | 0.42 (0.17 to 1.03) | 0.058 | 0.42 (0.17 to 1.02) | 0.056 |
| Occupation requiring movement outside the home | 0.61 (0.42 to 0.88) | 0.009 | 1.09 (0.64 to 1.86) | 0.737 | 1.08 (0.64 to 1.84) | 0.767 |
| Comorbidities | ||||||
| Any comorbidity | 3.32 (2.05 to 5.37) | <0.001 | - | - | - | - |
| Diabetes | 3.17 (2.05 to 4.91) | <0.001 | 1.45 (0.87 to 2.42) | 0.149 | 1.48 (0.89 to 2.47) | 0.135 |
| Hypertension | 3.70 (2.44 to 5.62) | <0.001 | 1.34 (0.80 to 2.24) | 0.259 | 1.35 (0.81 to 2.25) | 0.253 |
| Diabetes or hypertension | 3.41 (2.31 to 5.02) | <0.001 | - | - | - | - |
| Respiratory | 2.21 (0.91 to 5.33) | 0.078 | 1.67 (0.63 to 4.39) | 0.300 | 1.75 (0.66 to 4.64) | 0.259 |
| Cardiac | 4.16 (2.16 to 8.00) | < 0.001 | 1.52 (0.69 to 3.35) | 0.301 | 1.52 (0.68 to 3.37) | 0.305 |
| Renal | 12.79 (3.16 to 51.80) | < 0.001 | 6.34 (1.32 to 30.49) | 0.021 | 5.93 (1.19 to 29.47) | 0.030 |
| Tobacco and alcohol use | ||||||
| Current tobacco smoking | 0.84 (0.41 to 1.72) | 0.626 | 0.47 (0.21 to 1.08) | 0.074 | - | - |
| Pack-years of tobacco smoking among current smokers | - | - | ||||
| 0 (non-smoker) | Reference | – | Reference | – | ||
| <1 | 0.73 (0.22 to 2.44) | 0.605 | 1.26 (0.29 to 5.53) | 0.761 | ||
| 1–5 | 1.82 (0.71 to 4.61) | 0.210 | 0.37 (0.14 to 0.97)* | 0.044 | ||
| >5 | 0 | - | NA | - | ||
| Current smokeless tobacco use | 1.18 (0.72 to 1.95) | 0.513 | 0.98 (0.57 to 1.70) | 0.952 | 0.99 (0.57 to 1.71) | 0.970 |
| Current use of any form of tobacco | 1.10 (0.70 to 1.71) | 0.686 | - | - | - | - |
| Current alcohol use | 0.78 (0.27 to 2.26) | 0.650 | 0.56 (0.17 to 1.77) | 0.322 | 0.51 (0.16 to 1.65) | 0.264 |
*For model 2, pack-year categories 1–5 and >5 were combined and the OR for the combined category is presented here as ≥1 pack-years. Model 1 parameters: log likelihood=295.84, minus 2 log likelihood difference vs intercept=147.83, df=19, p<0.0001, pseudo-R2=0.1999.
Model 2 parameters: log likelihood=295.19, minus 2 log likelihood difference vs intercept=149.13, df=20, p<0.0001, pseudo-R2=0.2017.
Risk factors for mortality in COVID-19
Univariate analysis revealed that older age, especially ≥60 y, hypertension, the presence of diabetes or hypertension and respiratory, cardiac and renal comorbidity were associated with mortality (Table 5). Upon multivariate analysis, only older age and respiratory and cardiac comorbidity were found to be the risk factors of mortality among COVID-19 patients (Table 5). No significant association of tobacco smoking either as a binary variable or categorized as pack-years, smokeless tobacco use or alcohol use was seen with risk of mortality on either univariate or multivariate analysis. A significant association of those working outside the home and a history of travel in the past month with a lower risk of death disappeared on multivariate analysis, probably owing to confounding by older individuals staying at home and also being at risk of a fatal outcome.
Table 5.
Univariate and multivariate analysis of plausible risk factors for mortality in COVID-19 (N=911)
| Univariate | Multivariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| analysis | analysis (model 1) | analysis (model 2) | ||||
| OR | Adjusted OR | Adjusted OR | ||||
| Characteristics | (95% CI) | p-Value | (95% CI) | p-Value | (95% CI) | p-Value |
| Demographic and epidemiological characteristics | ||||||
| Male gender (vs female) | 1.05 (0.54 to 2.04) | 0.881 | 1.15 (0.50 to 2.64) | 0.736 | 1.15 (0.50 to 2.65) | 0.737 |
| Age (years) | 1.079 (1.055 to 1.103) | <0.001 | 1.078 (1.051 to 1.107) | <0.001 | 1.079 (1.051 to 2.648) | <0.001 |
| Age group (years) | ||||||
| 0–9 | 0 | |||||
| 10–19 | 0 | |||||
| 20–29 | 0 | |||||
| 30–39 | 0 | |||||
| 40–49 | 1 (reference) | - | ||||
| 50–59 | 1.38 (0.38 to 4.98) | 0.63 | ||||
| 60–69 | 5.55 (1.84 to 16.69) | 0.002 | ||||
| ≥70 | 4.91 (1.51 to 15.95) | 0.008 | ||||
| Residing in a cluster or hotspot area | 0.45 (0.18 to 1.17) | 0.102 | 0.76 (0.24 to 2.37) | 0.634 | 0.76 (0.24 to 2.39) | 0.642 |
| Any travel in the past month | 0.24 (0.06 to 0.99) | 0.048 | 0.28 (0.05 to 1.50) | 0.138 | 0.28 (0.05 to 1.50) | 0.137 |
| Occupation requiring movement outside the home | 0.38 (0.20 to 0.73) | 0.004 | 0.91 (0.39 to 2.13) | 0.827 | 0.91 (0.39 to 2.13) | 0.823 |
| Comorbidities | ||||||
| Any comorbidity | 3.79 (1.98 to 7.27) | <0.001 | - | - | - | - |
| Diabetes | 2.02 (0.96 to 4.22) | 0.063 | 1.18 (0.52 to 2.70) | 0.693 | 1.16 (0.51 to 2.68) | 0.720 |
| Hypertension | 3.55 (1.84 to 6.82) | <0.001 | 0.91 (0.41 to 2.02) | 0.812 | 0.95 (0.42 to 2.12) | 0.892 |
| Diabetes or hypertension | 3.17 (1.68 to 5.97) | <0.001 | - | - | - | - |
| Respiratory | 3.98 (1.31 to 12.10) | 0.015 | 3.62 (1.08 to 12.12) | 0.037 | 3.65 (1.08 to 12.34) | 0.037 |
| Cardiac | 7.90 (3.46 to 18.02) | <0.001 | 3.46 (1.28 to 9.38) | 0.015 | 3.41 (1.25 to 9.29) | 0.017 |
| Renal | 6.87 (1.38 to 34.26) | 0.019 | 1.96 (0.27 to 14.30) | 0.507 | 1.64 (0.19 to 13.81) | 0.651 |
| Tobacco and alcohol use | ||||||
| Current tobacco smoking | 1.24 (0.43 to 3.57) | 0.696 | 0.60 (0.18 to 2.00) | 0.407 | - | - |
| Pack-years of tobacco smoking among current smokers | ||||||
| 0 (non-smoker) | 1 (reference) | - | 1 (reference) | - | ||
| <1 | 0.80 (0.11 to 6.07) | 0.831 | 1.28 (0.09 to 17.88) | 0.856 | ||
| 1–5 | 0.87 (0.11 to 6.57) | 0.890 | 0.34 (0.04 to 2.91) | 0.325 | ||
| >5 | 2.71 (0.60 to 12.22) | 0.195 | 0.74 (0.14 to 4.01) | 0.730 | ||
| Current smokeless tobacco use | 1.37 (0.62 to 3.03) | 0.440 | 1.30 (0.55 to 3.10) | 0.549 | 1.31 (0.55 to 3.11) | 0.543 |
| Current use of any form of tobacco | 1.49 (0.75 to 2.98) | 0.258 | - | - | - | - |
| Current alcohol use | 0.61 (0.08 to 4.60) | 0.636 | 0.80 (0.10 to 6.71) | 0.840 | 0.81 (0.09 to 6.91) | 0.847 |
Model 1 parameters: log likelihood=127.19, minus 2 log likelihood difference vs intercept=80.02, df=13, p<0.0001, pseudo-R2=0.2393.
Model 2 parameters: log likelihood=126.88, minus 2 log likelihood difference vs intercept=80.64, df=13, p<0.0001, pseudo-R2=0.2411.
Discussion
Epidemiological factors
Prompt symptom onset to diagnosis in COVID-19 was in contrast to the long delay observed in the diagnosis of centuries-old infectious diseases such as tuberculosis and visceral leishmaniasis.14,15 This could be attributed to widespread awareness of COVID-19 symptoms in the community as well as rapid deployment of testing facilities. The short duration from testing to admission could be explained by admission of a substantial number of suspected COVID-19 cases while awaiting test results.
The present study adds to the evidence that older age and comorbidities increase the risk of severe COVID-19 and mortality, which appears biologically plausible.4,5 Greater risk of severe disease and death was associated with comorbidities even after adjusting for age and other possible confounders. The relative abundance of asymptomatic SARS-CoV-2-infected individuals among travellers, those having contact with lab-confirmed COVID-19 and those residing in a cluster or hotspot area could be attributed to aggressive household contact testing of confirmed COVID-19 cases, community testing in hotspot areas and testing among travellers. This was in accordance with the prevailing COVID-19 case-finding strategy.6 Thus a larger number of asymptomatic individuals were detected among these groups as compared with the general population, who would have likely approached the health facility only upon emergence of symptoms.
Tobacco and alcohol use
We also found alcohol use as a risk factor for developing symptomatic disease, which was retained on multivariate analysis. This appears plausible given the harmful effect of alcohol in supressing immunity and thereby predisposing to a greater risk of infectious respiratory diseases.16
Current tobacco smoking was found to be associated with reduced symptomatic COVID-19, even after adjusting for possible confounders on multivariate analysis. Further, some evidence of a dose–response relationship was seen with greater pack-years of tobacco smoking associated with an apparently reduced risk of symptomatic and severe disease. Earlier studies using the approach of comparing smoking prevalence among hospitalized patients with that of population surveys found a protective effect of smoking on the risk of testing positive for SARS-CoV-2 and being hospitalized with COVID-19.17,18 However, this approach could lead to information bias due to the better effectiveness of smoking-related information collection in population-level surveys as compared with hospital settings.19 In the present study we attempted to eliminate this information bias by collecting the smoking-related data in the asymptomatic and symptomatic groups in a similar manner through telephone interviews.
‘Nicotine hypothesis’ of protection against COVID-19
The reduced risk of SARS-CoV-2 infection among tobacco smokers has been hypothesized to be due to the effect of nicotine on the nicotinic acetylcholine receptor (nAChR). This has been proposed to act through a reduction of potential sites for viral entry in the pulmonary alveolar epithelium and through inhibition of pro-inflammatory cytokines.17,19–21 As seen in other viral diseases such as cytomegalovirus, factors reducing the risk of infection could also reduce the possibility of symptomatic disease due to lesser primary viremia.22
The nAChR receptors are located in the brain, where nicotine dissolved in blood crosses the blood–brain barrier.23 It could reach the bloodstream through absorption in the lung, gut or transdermally.23 Theoretically, any association of tobacco smoking with reduced SARS-CoV-2 infection or severity through the nicotinic mechanism should also be seen with regard to smokeless tobacco use. In this regard, India provides an appropriate opportunity to test this hypothesis, as smokeless tobacco use is twice as prevalent as tobacco smoking.12 Since we did not observe an association of smokeless tobacco use with either reduced symptomatic COVID-19 or reduced severity of COVID-19, the ‘nicotine hypothesis’ may need to be re-examined. Further, if we consider smokeless tobacco use in the present study as a proxy for pharmaceutical nicotine patches, suggestions regarding their use to explore protection against COVID-19 seem implausible.17,19
Several studies have found that tobacco smoking increases the risk of progression to severe disease, the requirement for mechanical ventilation and mortality among COVID-19 patients.24,25 Further, the biological plausibility of respiratory pathogenesis suggests that smoking leads to severe lung disease in influenza and tuberculosis.26,27 However, we did not find a statistically convincing effect of tobacco use on severe disease and death, possibly because our study was underpowered to detect these effects due to the lower overall proportion of severe disease and deaths among all infected individuals.
Alternative explanations
We suggest an alternative hypothesis for explaining the possible association of tobacco smoking with the low risk of SARS-CoV-2 infection. A recent immunological study detected SARS-CoV-2-reactive CD4+ T cells among 40–60% of unexposed individuals. This suggests cross-reactivity between commonly circulating low-pathogenic coronaviruses causing the common cold and SARS-CoV-2, mainly targeting the viral 1AB polyprotein and S protein regions.28 Genetically, these regions also consistently show high sequence similarity between the common cold coronaviruses (229E, NL63, OC43 or HKU1) and SARS-CoV-2.29 Further, cross-reactivity of SARS-CoV-2 antibodies has been shown with the full spike proteins of common cold coronaviruses using enzyme-linked immunosorbent assays.30 Exposure to cigarette smoke has been known to lead to more frequent and serious upper respiratory tract infections, including the common cold.26,31,32 Therefore we propose that cross-protection afforded by frequent upper respiratory tract infection among tobacco smokers could explain the association of tobacco smoking with the lower risk of SARS-CoV-2 infection. Studies assessing cell-mediated and humoral cross-reactivity of SARS-CoV-2 with common cold coronaviruses among tobacco smokers as compared with non-smokers may be conducted to further explore this hypothesis.
Further, variations in host genetic factors are known to modulate susceptibility to SARS-CoV-2 and can influence the severity of symptoms.33 Single-nucleotide polymorphisms of a variety of genes, including ACE2, TMPRSS2, HLA, CD147, MIF, IFNG and IL6, have been implicated in the pathological and immunological response to COVID-19.33 The pseudo-R2 value of only 5% in the multivariate analysis suggests that the 13 plausible risk factors taken together failed to provide a substantial association explaining the risk of symptomatic COVID-19 vis-à-vis asymptomatic infection. Therefore the presence of residual confounding factors such as host genetic or environmental factors should also be considered to fully explain the risk of development of symptomatic COVID-19.
Due to the harmful effect of smoking on respiratory and cardiac health and its effect on increasing mortality, we support the WHO guidance that tobacco smoking should be discouraged during the COVID-19 pandemic and afterwards.34 Also, owing to the presence of silent hypoxemia in a subset of patients, an asymptomatic course of disease does not necessarily mean the absence of pathogenesis in COVID-19.35 Contradictory and confusing findings emphasizing the protective effect of smoking on COVID-19 could potentially reverse the gains made in anti-tobacco messaging. Consequently, smokers on the verge of quitting could delay or abandon such plans owing to the perception of a reduction in potential harm.
The majority of infected individuals diagnosed at our institute had no history of contact with a laboratory-confirmed COVID-19 case. Similar epidemiological data are being collected country-wide by the Integrated Disease Surveillance Programme through the Integrated Health Information Platform portal. Basic information related to the age and gender distribution of COVID-19 patients is already being displayed in an online dashboard.36 If similar findings emerge from the country-level data of India, then the current transmission classification of ‘clusters of cases’ might need to be revised to ‘community transmission’.1
Limitations
We elicited the symptomatic history only once after the lab confirmation of SARS-CoV-2 infection. This could lead to misclassification of cases and controls owing to the study design for some of the individuals who might have been in a pre-symptomatic phase of their illness during the telephone interview. We interviewed individuals a median of 5 d (IQR 3–7) after testing, whereas the median pre-symptomatic phase is known to be <1–4 d.37 This could have allowed sufficient time to elapse for most of the pre-symptomatic individuals to develop symptoms, thus the effect of misclassification based on symptom elicitation only once is not expected to be large. However, some amount of misclassification due to recall errors from the patients’ side was possible.
Furthermore, around 25% of the eligible individuals could not be contacted for inclusion in the study. This could have led to a possible selection bias in the study population. We cross-checked the distribution of key risk factors in the study population, such as the prevalence of tobacco use, which was found similar to that of population estimates for Rajasthan in the Global Adult Tobacco Survey 2016–17.12 In the worst-case scenario, this suggested a non-differential bias with regard to tobacco use–related risk factors, which could have influenced the key OR estimates to move towards null. This could have some implication with regard to the validity of our findings that smokeless tobacco use was not associated with either a protective or harmful effect for symptomatic or severe COVID-19. The possibility of underrepresentation of some other plausible risk factors such as comorbidities also remained. Further, the possibility of some recall bias existed for alcohol and tobacco use, especially when their availability had been reduced recently due to lockdown. We also did not collect data on household size, factors influencing social mixing and other predisposing factors for respiratory illness.
Conclusions
Earlier and more aggressive case-finding among household contacts of confirmed cases and travellers resulted in a greater representation of asymptomatic cases. Older age and the presence of comorbidities were associated with symptomatic disease, severe symptomatic disease and death. Alcohol use was found to be associated with symptomatic disease. Tobacco smoking was found to be associated with reduced risk of symptomatic disease and severe disease to some extent, while no association was found with mortality. However, the lack of a similar association with smokeless tobacco use goes against the ‘nicotine hypothesis’ of protection against COVID-19. Both smoking and smokeless tobacco use should be strongly discouraged during the COVID-19 pandemic due to the known effect of increasing morbidity and mortality through multiple pathways. We propose that cross-protection afforded by frequent upper respiratory tract infections among tobacco smokers could possibly explain its association with a lower risk of symptomatic COVID-19. Genetic factors influencing host immunity should be further explored for symptom development and severity of COVID-19. The evidence of risk factors of symptomatic and severe symptomatic disease could be useful for prioritizing measures of prevention in the community and clinical care in the hospital. We recommend that older SARS-CoV-2-infected individuals, especially those ≥60 y of age, and those with comorbidities be prioritized for hospital admission due to a greater risk of severe disease and fatal outcome.
Supplementary Material
Acknowledgements
We acknowledge the help of personnel involved in the testing and clinical care of SARS-CoV-2-infected individuals at the All India Institute of Medical Sciences. We also thankfully acknowledge the Masters of Public Health scholars at the All India Institute of Medical Sciences: Jay Shree Shekhawat, Prasannajeet Bal, Ipsa Kutlehrria, Nainsi Gupta, Neelam Kumari, Mahima Choudhary, Musarrat Siddiqui, Sonali Bhattacharya, Himani Sharma, Uplabdhi Sahu, Shubham Rai, Oshi Chaturvedi, Ashu Ranga, Premlata Meghwal, Chunni Lal, Rupali Gupta, Zeba Bano, Diksha Mahajan, Abhilipsa Pradhan, Jinesh Saini, Neha Mantri and Nishant Soni for supporting the epidemiological data collection. The views expressed in this article are those of the authors alone and do not necessarily represent the views of their organizations.
Contributor Information
Suman Saurabh, Department of Community Medicine and Family Medicine, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Mahendra Kumar Verma, Department of Community Medicine and Family Medicine, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Vaishali Gautam, Department of Community Medicine and Family Medicine, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Nitesh Kumar, Department of Community Medicine and Family Medicine, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Vidhi Jain, Department of Microbiology, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Akhil Dhanesh Goel, Department of Community Medicine and Family Medicine, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Manoj Kumar Gupta, Department of Community Medicine and Family Medicine, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Prem Prakash Sharma, Department of Community Medicine and Family Medicine, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Pankaj Bhardwaj, Department of Community Medicine and Family Medicine, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Kuldeep Singh, Department of Paediatrics, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Vijaya Lakshmi Nag, Department of Microbiology, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Mahendra Kumar Garg, Department of Medicine, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Sanjeev Misra, Director, All India Institute of Medical Sciences, Jodhpur, Rajasthan 342005, India.
Authors’ contributions
MKV, VG, NK and SS collected, curated and entered the data. SS conducted the analysis with input from PPS and wrote the draft manuscript with input from MKV, VG, ADG, VJ, PPS, MKGupta, KS, PB and MKGarg. VJ and VLN coordinated the laboratory testing and MKGarg coordinated the clinical care of COVID-19 patients. SM provided overall supervision of the laboratory testing, clinical care and research related to COVID-19 at the All India Institute of Medical Sciences. All authors approved the final manuscript.
Funding
Department of Health Research/Indian Council of Medical Research, Government of India is acknowledged for funding the operationalization of Regional -Viral Diagnosis & Research Laboratory (VDRL) at AIIMS Jodhpur, which was utilized for laboratory testing for COVID-19. The authors declare that no other funding was received from any source for the study and preparation of this article.
Competing interests
The authors declare that there are no competing interests for publication of this article. The views expressed in this article are those of the authors alone and do not necessarily represent the view of their organization.
Ethical approval
The study was approved by the Institutional Ethics Committee of the All India Institute of Medical Sciences (AIIMS/IEC/2020-21/1099).
Data availability
Anonymized data on which the study is based have been included as a supplementary file (Supplementary File 1) along with the article.
References
- 1. World Health Organization . Weekly epidemiological update for COVID-19 – 5 January 2021. Geneva: World Health Organization; 2020. Available from: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20210105_weekly_epi_update_21.pdf?sfvrsn=15359201_5 [accessed January 2021]. [Google Scholar]
- 2. Chen N, Zhou M, Dong X et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Gandhi M, Yokoe DS, Havlir DV. Asymptomatic transmission, the Achilles’ heel of current strategies to control Covid-19. N Engl J Med. 2020;382(22):2158–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Li Q, Guan X, Wu P et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med. 2020;382(13):1199–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Centers for Disease Control . Groups at higher risk of severe illness with COVID-19. Atlanta, GA: Centers for Disease Control. Available from: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/groups-at-higher-risk.html [accessed January 2021]. [Google Scholar]
- 6. Indian Council of Medical Research . Strategy for COVID-19 Testing in India – version 5 (18 May 2020). New Delhi: Indian Council of Medical Research; 2020. Available from: https://www.icmr.gov.in/pdf/covid/strategy/Testing_Strategy_v5_18052020.pdf [accessed January 2021]. [Google Scholar]
- 7. National Centre for Disease Control . Revised case investigation format for COVID-19. New Delhi: National Centre for Disease Control; 2020. Available from: https://ncdc.gov.in/showfile.php?lid=530 [accessed January 2021]. [Google Scholar]
- 8. Ministry of Health and Family Welfare . Clinical management protocol for COVID-19 (Version 5, Dated 3 July 2020). New Delhi: Ministry of Health and Family Welfare; 2020. Available from: https://www.mohfw.gov.in/pdf/UpdatedClinicalManagementProtocolforCOVID19dated03072020.pdf [accessed January 2021]. [Google Scholar]
- 9. World Health Organization . WHO STEPS questionnaire for non-communicable diseases and their risk factors. Geneva: World Health Organization; 2002. Available from: https://www.who.int/ncds/surveillance/steps/instrument/en/ [accessed January 2021]. [Google Scholar]
- 10. Malson JL, Sims K, Murty R et al. Comparison of the nicotine content of tobacco used in bidis and conventional cigarettes. Tob Control. 2001;10(2):181–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Gudbjartsson DF, Helgason A, Jonsson H et al. Spread of SARS-CoV-2 in the Icelandic population. N Engl J Med. 2020;382(24):2302–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. World Health Organization . Global adult tobacco survey factsheet for India 2016–17. Geneva: World Health Organization; 2020. Available from: https://www.who.int/tobacco/surveillance/survey/gats/ind/en/ [accessed January 2021]. [Google Scholar]
- 13. OpenEpi: open source epidemiologic statistics for public health. Atlanta, GA: Rollins School of Public Health, Emory University; 2020. Available from: http://openepi.com/Menu/OE_Menu.htm [accessed January 2021]. [Google Scholar]
- 14. Kumar A, Saurabh S, Jamil S et al. Intensely clustered outbreak of visceral leishmaniasis (kala-azar) in a setting of seasonal migration in a village of Bihar, India. BMC Infect Dis. 2020;20(1):10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Das D, Dwibedi B. Delay in diagnosis among pulmonary tuberculosis patients of Rayagada District, Odisha, India. Int J Mycobacteriol. 2016;5(Suppl 1):S172–3. [DOI] [PubMed] [Google Scholar]
- 16. Simet SM, Sisson JH. Alcohol's effects on lung health and immunity. Alcohol Res. 2015;37(2):199–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Farsalinos K, Barbouni A, Poulas K et al. Current smoking, former smoking, and adverse outcome among hospitalized COVID-19 patients: a systematic review and meta-analysis. Ther Adv Chronic Dis. 2020;11:2040622320935765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Grundy EJ, Suddek T, Filippidis FT et al. SARS-CoV-2 and COVID-19: a review of reviews considering implications for public health policy and practice. Tob Induc Dis. 2020;18:58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Vaduganathan M, Vardeny O, Michel T et al. Renin–angiotensin–aldosterone system inhibitors in patients with Covid-19. N Engl J Med. 2020;382(17):1653–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Changeux J-P, Amoura Z, Rey FA et al. A nicotinic hypothesis for Covid-19 with preventive and therapeutic implications. C R Biol. 2020;343(1):33–9. [DOI] [PubMed] [Google Scholar]
- 21. Boutou AK, Pitsiou G, Kontakiotis T et al. Nicotine treatment and smoking cessation in the era of COVID-19 pandemic: an interesting alliance. ERJ Open Res. 2020;6(3):00306–2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Smiljkovic M, Le Meur J-B, Malette B et al. Blood viral load in the diagnostic workup of congenital cytomegalovirus infection. J Clin Virol. 2020;122:104231. [DOI] [PubMed] [Google Scholar]
- 23. Benowitz NL. Pharmacology of nicotine: addiction, smoking-induced disease, and therapeutics. Annu Rev Pharmacol Toxicol. 2009;49:57–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Vardavas CI, Nikitara K. COVID-19 and smoking: a systematic review of the evidence. Tob Induc Dis. 2020;18:20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Patanavanich R, Glantz SA. Smoking is associated with COVID-19 progression: a meta-analysis. Nicotine Tob Res. 2020;22(9):1653–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Feldman C, Anderson R. Cigarette smoking and mechanisms of susceptibility to infections of the respiratory tract and other organ systems. J Infect. 2013;67(3):169–84. [DOI] [PubMed] [Google Scholar]
- 27. Lawrence H, Hunter A, Murray R et al. Cigarette smoking and the occurrence of influenza – systematic review. J Infect. 2019;79(5):401–6. [DOI] [PubMed] [Google Scholar]
- 28. Grifoni A, Weiskopf D, Ramirez SI et al. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell 2020;181(7):1489–501.e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Ma Z, Li P, Ji Y et al. Cross-reactivity towards SARS-CoV-2: the potential role of low-pathogenic human coronaviruses. Lancet Microbe. 2020;1(4):e151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Hicks J, Klumpp-Thomas C, Kalish H et al. Serologic cross-reactivity of SARS-CoV-2 with endemic and seasonal Betacoronaviruses. medRxiv 2020; 10.1101/2020.06.22.20137695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Zhou G, Liu H, He M et al. Smoking, leisure-time exercise and frequency of self-reported common cold among the general population in northeastern China: a cross-sectional study. BMC Public Health. 2018;18(1):294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Putri PD, Susanto AD, Hudoyo A et al. Correlation between domestic cigarette smoke exposure and respiratory complaints, hospitalization and school absence due to respiratory complains in the Indonesian elementary school-aged children. Int J Appl Basic Med Res. 2018;8(4):244–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Ovsyannikova IG, Haralambieva IH, Crooke SN et al. The role of host genetics in the immune response to SARS-CoV-2 and COVID-19 susceptibility and severity. Immunol Rev. 2020;296(1):205–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. World Health Organization . Smoking and COVID-19: Scientific Brief (dated 30 June 2020). Geneva: World Health Organization; 2020. Available from: https://www.who.int/news-room/commentaries/detail/smoking-and-covid-19 [accessed January 2021]. [Google Scholar]
- 35. Wilkerson RG, Adler JD, Shah NG et al. Silent hypoxia: a harbinger of clinical deterioration in patients with COVID-19. Am J Emerg Med. 2020;38(10):2243.e5–e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. National Centre for Disease Control . Integrated health information platform – graphical illustration of data from COVID-19 cases in India. New Delhi: National Centre for Disease Control; 2020. Available from: https://ncdc.gov.in/dashboard.php [accessed January 2021]. [Google Scholar]
- 37. Byrne AW, McEvoy D, Collins AB et al. Inferred duration of infectious period of SARS-CoV-2: rapid scoping review and analysis of available evidence for asymptomatic and symptomatic COVID-19 cases. BMJ Open. 2020;10(8):e039856. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Anonymized data on which the study is based have been included as a supplementary file (Supplementary File 1) along with the article.