Abstract
Background
During the first wave of the coronavirus disease 2019 (COVID-19) pandemic, older patients had an increased risk of hospitalisation and death. Reports on the association of frailty with poor outcome have been conflicting.
Objective
The aim of the present study was to investigate the independent association between frailty and in-hospital mortality in older hospitalised COVID-19 patients in the Netherlands.
Methods
This was a multicentre retrospective cohort study in 15 hospitals in the Netherlands, including all patients aged ≥70 years, who were hospitalised with clinically confirmed COVID-19 between February and May 2020. Data were collected on demographics, co-morbidity, disease severity and Clinical Frailty Scale (CFS). Primary outcome was in-hospital mortality.
Results
A total of 1,376 patients were included (median age 78 years (interquartile range 74–84), 60% male). In total, 499 (38%) patients died during hospital admission. Parameters indicating presence of frailty (CFS 6–9) were associated with more co-morbidities, shorter symptom duration upon presentation (median 4 versus 7 days), lower oxygen demand and lower levels of C-reactive protein. In multivariable analyses, the CFS was independently associated with in-hospital mortality: compared with patients with CFS 1–3, patients with CFS 4–5 had a two times higher risk (odds ratio (OR) 2.0 (95% confidence interval (CI) 1.3–3.0)) and patients with CFS 6–9 had a three times higher risk of in-hospital mortality (OR 2.8 (95% CI 1.8–4.3)).
Conclusions
The in-hospital mortality of older hospitalised COVID-19 patients in the Netherlands was 38%. Frailty was independently associated with higher in-hospital mortality, even though COVID-19 patients with frailty presented earlier to the hospital with less severe symptoms.
Keywords: clinical frailty scale, COVID-19, frailty, SARS-CoV-2, older adults
Key points
Reports on the association of frailty with poor outcomes in older hospitalised COVID-19 patients have been conflicting.
Within this multicentre study in the Netherlands, the in-hospital mortality of older hospitalised COVID-19 patients was 38%.
COVID-19 patients with frailty presented earlier to the hospital with less severe symptoms.
Frailty was independently associated with higher in-hospital mortality.
These findings stress the importance of assessing frailty in older patients with COVID-19 to deliver appropriate care.
Introduction
During the first wave of the coronavirus disease 2019 (COVID-19) pandemic, older patients had an increased risk of hospitalisation and mortality. The first Chinese reports showed in-hospital mortality rates for older patients up to 34% [1]. Later European reports even showed in-hospital mortality rates up to 60% in patients aged 65 years or older [2].
Among older patients with COVID-19, pre-existing co-morbidity, disease severity and advanced age have been shown to be predictors of poor outcomes [1]. Interestingly, studies reporting the association of frailty with poor outcomes in the COVID-19 pandemic have shown conflicting results. Although frailty was associated with increased mortality in multiple studies [3–9], in some studies it was not [2, 10]. Potential explanations for these inconsistent findings may be of a methodological nature, including differences in patients’ selection (partially due to different healthcare systems), relatively small sample sizes and single-centre studies. Other explanations include the biological hypothesis that frail older patients have higher degrees of immunosenescence and hence a lower risk of inflammatory syndromes, which might cause complications and mortality [11].
The aim of the present study was to investigate the independent association between frailty and in-hospital mortality in older hospitalised COVID-19 patients in the Netherlands.
Methods
Study design
This was a retrospective multicentre cohort study among patients aged 70 years and older who were hospitalised with COVID-19 from 27 February to 14 May 2020 in the Netherlands: the ‘COVID-OLD’ study. Data were collected from 15 Dutch hospitals: Alrijne hospital, Amsterdam University Medical Center location AMC, Catharina Hospital, Elisabeth-TweeSteden Hospital, Erasmus Medical Center, Gelre Hospitals, Leiden University Medical Center, Maasstad Hospital, Medical Center Leeuwarden, OLVG Hospital, Reinier de Graaf Hospital, Spaarne Gasthuis location Haarlem, University Medical Center Groningen, VieCuri Medical Center and Zaans Medical Center. The study was initially set up as a prospective study in the first five hospitals (Erasmus Medical Center, Gelre Hospitals, Leiden University Medical Center, Reinier de Graaf Hospital and Spaarne Gasthuis) and started after the first person was diagnosed with COVID-19 in the Netherlands on 27 February 2020. An opt-out procedure was used to include eligible patients, which means chart data were available for scientific research unless a patient explicitly objected. The other 10 hospitals joined this study later and local researchers collected data retrospectively. The medical ethics committees of all hospitals waived the necessity for formal approval of the study, as data collection followed routine practice.
Setting
During the early months of the pandemic in the Netherlands, several national guidelines were published to address appropriateness of hospital care and intensive care unit (ICU) admittance for older patients with COVID-19 [12, 13]. Both guidelines advocated the use of the Clinical Frailty Scale (CFS), which ranges from 1 (very fit) to 9 (terminally ill) [14], and which was only sporadically used in Dutch hospitals prior to the pandemic. The guidelines for hospitalisation where implemented to assist physicians outside the hospital (general practitioners, nursing home physicians) regarding suspected benefit of hospitalisation for older people with COVID-19 (defined as aged ≥70 years), which was considered to be ‘doubtful’ for CFS 4–5 and ‘not enough physiological reserve’ for CFS 6–9. The guideline for ICU admission was not formally implemented and only communicated in preparation of a possible absolute shortage of ICU capacity. This guideline considered ‘undesirable’ for older people with CFS of 7 or higher. Although not formally implemented, the recommendations of this guideline may have influenced the selection of patients who were admitted to crowded ICU’s.
Study participants
The inclusion criteria were age ≥70 years and hospitalisation with diagnosed COVID-19. Patients were included if they tested positive for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) on reverse-transcriptase polymerase chain reaction (PCR) from an oropharyngeal and/or nasal swab or if they were diagnosed based on typical findings on computed tomography scan and/or chest X-ray. We also included patients with a clinical diagnosis of COVID-19 based on review of clinical, laboratory and radiological findings. Patients were excluded if they were not initially admitted with COVID-19 symptoms or PCR-proven infection, but were infected in the hospital during admission for another illness (patients with positive PCR ≥1 week after admission were excluded). Furthermore, patients who were transferred from another hospital were excluded because baseline information regarding the initial hospital admission was lacking.
Outcomes
The primary outcome of this study was in-hospital mortality. Secondary outcomes were ICU admission, presence of delirium during admission, hospital length of stay and discharge destination.
Data collection
Data were collected from the patients’ electronic health records. We collected demographic data on age, sex and living situation (at home or in an institution). The Charlson Comorbidity Index (CCI) was used to gain insight in the presence of comorbidity [15]. In addition, data on history of lung disease, presence of hypertension, smoking status and body mass index were collected.
Geriatric parameters were routinely collected with the Dutch National Safety Management System (Veiligheidsmanagementsysteem (VMS)) [16]. This risk assessment tool is routinely administered at hospital admission for all patients aged ≥70 years. The instrument consists of 13 questions regarding four domains: physical impairment, falls, delirium and malnutrition. First, the Katz Activities of Daily Living (ADL) Index is used to evaluate physical impairment ≥2 weeks prior to hospital admission (Katz score ≥2 meaning risk of physical impairment) [17]. Second, risk of falls is assessed with the question whether the patient has fallen in the last 6 months (yes/no). Third, a patient is considered to be at risk for delirium if one or more of three following questions are answered with ‘yes’: memory problems, need for help with self-care in the last 24 h and previously experienced confusion. Fourth, for evaluation of malnutrition, either the Short Nutritional Assessment Questionnaire (SNAQ) or Malnutrition Universal Screening Tool (MUST) is used (risk of malnutrition with SNAQ ≥3 or MUST ≥2) [18, 19].
Frailty was assessed with the CFS [14]. Data were collected both prospectively and retrospectively. The CFS was prospectively assigned at hospital admission according to the implemented guidelines (usually by a geriatric nurse). If not prospectively assigned, the CFS was determined retrospectively and was based on available chart data (which included the geriatric assessment from the VMS) and was scored by a geriatric specialist (geriatrician or internist–geriatrician). Data on CFS were considered missing if not prospectively assigned to the patient during admission or if information from the health record was not sufficient to determine the CFS retrospectively. According to the Dutch guidelines, the CFS was categorised in three groups: fit (CFS 1–3), pre-frail (CFS 4–5) and frail (CFS 6–9).
The date of admission subtracted from the date of onset of symptoms yielded the duration of COVID-19 symptoms until admission in days. Disease severity indicators were the first registered vital signs and laboratory results collected within 24 h of admission.
Collected in-hospital outcomes were ICU admission (including length of stay, ventilation and ICU mortality), hospital length of stay, in-hospital mortality and discharge destination (home, nursing home, other hospital, other). In addition, we collected data on documented presence of delirium during hospitalisation.
Statistical analyses
Continuous data are presented as means (standard deviation (SD)) if normally distributed, and as medians (interquartile range (IQR)) if skewed. Categorical data are presented as numbers (n (%)). Differences in patient characteristics and secondary outcomes between CFS categories (CFS 1–3 versus CFS 4–5 versus CFS 6–9) were assessed using one-way analysis of variance for normally distributed data, the Kruskal–Wallis test for skewed data and the χ2 test for categorical data.
Multivariable logistic regression analysis was used with in-hospital mortality as the dependent variable. Because of possible multicollinearity between patient characteristics and substantial numbers of missing numbers, the multivariable model included the following independent variables: demographics (age, sex), comorbidity (CCI), frailty (CFS) and disease severity indicators (duration of symptoms, oxygen amount needed and C-reactive protein (CRP)). Sensitivity analyses were executed with Katz ADL score in the model instead of CFS and several individual comorbidities (diabetes and myocardial infarction) instead of the CCI. In addition, as an exploration of predictive performance, the discriminatory properties of age, CFS and the combination of age and CFS were assessed with the area under the receiver operating characteristic curve (AUC) for the primary outcome in-hospital mortality.
Results are presented as odds ratios (ORs) with 95% confidence intervals (CIs). A P-value <0.05 was considered statistically significant. Data were collected using Castor Electronic Data Capture (2019). Statistical analyses were performed using IBM SPSS Statistics, version 25 (IBM Corp., Armonk, NY, USA).
Results
A total of 1,543 patients aged ≥70 years were hospitalised with COVID-19 infection in the participating hospitals, of whom 1,530 (99.2%) patients were eligible. We excluded 154 (10.1%) patients because of incomplete data due to transfer from other hospitals, resulting in a total of 1,376 patients for baseline analyses. For our primary outcome analyses, all patients discharged to other hospitals (n = 75) were additionally excluded because of incomplete follow-up, resulting in 1,301 patients available for analyses on in-hospital mortality (Figure 1). Of all 1,376 patients, CFS data was collected prospectively in 177 (12.9%) patients, retrospectively in 834 (60.6%) patients and data was missing in 365 (26.5%) patients. In 87.6% of cases, COVID-19 diagnosis was confirmed by PCR technique.
Figure 1 .
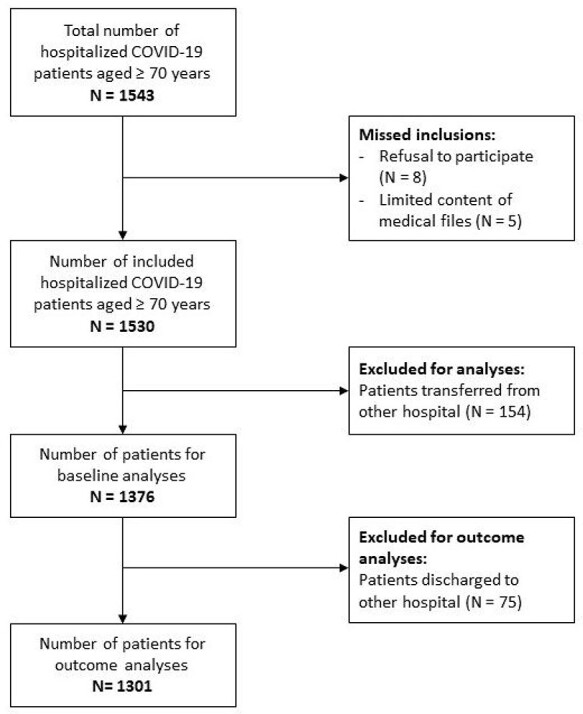
Flowchart of study population.
Baseline characteristics of the 1,376 included patients are shown in Table 1. The median age was 78 years (IQR 74–84), 60.4% of patients was male and the median CCI was 2 (IQR 1–3). In total, 515 (46.1%) patients were classified as CFS 1–3, 288 (25.8%) patients as CFS 4–5 and 313 (28.0%) patients as CFS 6–9. The median duration of COVID-19 symptoms until admission was 7 days (IQR 3–10). At hospital admission, patients had a mean temperature of 37.7°C (SD 1.1), required a median of 3 (IQR 1–5) L/min oxygen and had a median CRP of 79 (IQR 40–140) mg/L.
Table 1 .
Baseline characteristics for older hospitalised COVID-19 patients
| N = 1,376 | |
|---|---|
| Demographics | |
| Age (years), median (IQR) | 78 (74–84) |
| Male, n (%) | 830 (60.4) |
| Living at home, n (%) | 1,186 (89.9) |
| Co-morbidity | |
| Charlson Comorbidity Index, median (IQR) | 2 (1–3) |
| History of chronic lung diseasea, n (%) | 351 (25.5) |
| History of hypertension, n (%) | 776 (56.5) |
| History of diabetes, n (%) | 416 (30.3) |
| History of myocardial infarction, n (%) | 259 (18.8) |
| History of dementia, n (%) | 120 (8.7) |
| Smoking, n (%) | |
| Never | 438 (42.4) |
| Ex-smoker | 510 (49.4) |
| Current | 85 (8.2) |
| Body mass index, mean (SD) | 27.0 (6.2) |
| Geriatric measurements | |
| Katz ADL score, median (IQR) | 0 (0–3) |
| Risk of physical impairmentb, n (%) | 424 (35.5) |
| Risk of falling, n (%) | 323 (28.5) |
| Risk of delirium, n (%) | 536 (45.3) |
| Risk of malnutrition, n (%) | 221 (21.0) |
| Clinical Frailty Scale, n (%) | |
| 1–3 | 515 (46.1) |
| 4–5 | 288 (25.8) |
| 6–9 | 313 (28.0) |
| Disease severity indicators | |
| Duration of symptoms until admission (days), median (IQR) | 7 (3–10) |
| Temperature (°C), mean (SD) | 37.7 (1.1) |
| Respiratory rate (breaths/min), mean (SD) | 22 (8) |
| Oxygen amount needed (L/min), median (IQR) | 3 (1–5) |
| Lymphocytes (109/L), median (IQR) | 1.0 (0.6–1.7) |
| Creatinine (μmol/L), median (IQR) | 93 (74–130) |
| Lactic acid dehydrogenase (U/L), median (IQR) | 321 (257–415) |
| C-reactive protein (mg/L), median (IQR) | 79 (40–140) |
N, number. Missing: 2 sex, 57 living at home, 16 CCI, 2 hypertension, 4 diabetes, 1 myocardial infarction, 2 dementia, 343 smoking, 294 BMI, 180 Katz ADL, 245 risk of falling, 193 risk of delirium, 322 risk of malnutrition, 260 Clinical Frailty Scale, 146 duration of symptoms, 64 temperature, 78 respiratory rate, 151 oxygen, 251 lymphocytes, 85 creatinine, 274 LDH, 85 CRP.
aCOPD, asthma, interstitial lung disease or lung cancer.
bKatz ADL score ≥2.
Table 2 presents the baseline characteristics of the total population stratified per CFS category. With increasing CFS categories, patients were older (median 76 years (CFS 1–3) versus 79 years (CFS 4–5) and 83 years (CFS 6–9); P < 0.001) and less often male (70.0% male (CFS 1–3) versus 58.7% male (CFS 4–5) and 46.5% male (CFS 6–9); P < 0.001). Patients with higher CFS had more co-morbidities, represented by higher CCI scores (median 1 (CFS 1–3) versus median 2 (CFS 4–5) and median 3 (CFS 6–9); P < 0.001). The duration of COVID-19 symptoms until admission was significantly shorter for patients with the highest CFS classification (median 7 days (CFS 1–3) versus 7 days (CFS 4–5) and 4 days (CFS 6–9); P < 0.001) and their disease severity at admission was lower, represented by a lower amount of oxygen needed (median 3 L/min (CFS 1–3) versus 3 L/min (CFS 4–5) and 2 L/min (CFS 6–9); P = 0.008) and lower CRP levels (median 93 mg/L (CFS 1–3) versus 79 mg/L (CFS 4–5) and 63 mg/L (CFS 6–9); P < 0.001). Baseline characteristics stratified per age category are shown in Supplementary A1.
Table 2 .
Baseline characteristics for older hospitalised COVID-19 patients stratified by Clinical Frailty Scale
| Fit CFS 1–3 (N = 515) | Pre-frail CFS 4–5 (N = 288) | Frail CFS 6–9 (N = 313) | P-value* | |
|---|---|---|---|---|
| Demographics | ||||
| Age (years), median (IQR) | 76 (73–81) | 79 (75–85) | 83 (78–87) | <0.001 |
| Male, n (%) | 360 (70.0) | 169 (58.7) | 145 (46.5) | <0.001 |
| Living at home, n (%) | 496 (99.2) | 263 (94.9) | 213 (69.2) | <0.001 |
| Co-morbidity | ||||
| Charlson Comorbidity Index, median (IQR) | 1 (0–3) | 2 (1–4) | 3 (1–4) | <0.001 |
| History of chronic lung diseasea, n (%) | 96 (18.6) | 101 (35.1) | 84 (26.8) | <0.001 |
| History of hypertension, n (%) | 248 (48.2) | 183 (63.5) | 187 (59.7) | <0.001 |
| History of diabetes, n (%) | 127 (24.8) | 94 (32.8) | 109 (34.9) | 0.004 |
| History of myocardial infarction, n (%) | 73 (14.2) | 64 (22.2) | 61 (19.5) | 0.011 |
| History of dementia, n (%) | 5 (1.0) | 16 (5.6) | 85 (27.2) | <0.001 |
| Smoking, n (%) | ||||
| Never | 27 (7.0) | 16 (7.5) | 30 (12.6) | 0.052 |
| Ex-smoker | 204 (53.0) | 116 (54.7) | 101 (42.3) | |
| Current | 154 (40.0) | 80 (37.7) | 108 (45.2) | |
| Body mass index, mean (SD) | 26.9 (4.1) | 27.8 (5.4) | 26.5 (5.0) | 0.007 |
| Geriatric measurements | ||||
| Katz ADL score, median (IQR) | 0 (0–0) | 0 (0–2) | 4 (2–6) | <0.001 |
| Risk of physical impairmentb, n (%) | 35 (7.4) | 83 (31.8) | 238 (87.8) | <0.001 |
| Risk of falling, n (%) | 69 (15.4) | 76 (30.6) | 128 (52.0) | <0.001 |
| Risk of delirium, n (%) | 84 (18.7) | 116 (46.0) | 247 (87.3) | <0.001 |
| Risk of malnutrition, n (%) | 72 (17.1) | 48 (20.3) | 49 (21.2) | 0.369 |
| Disease severity indicators | ||||
| Duration of symptoms until admission (days), median (IQR) | 7 (5–12) | 7 (3–11) | 4 (2–7) | <0.001 |
| Temperature (°C), mean (SD) | 37.8 (1.1) | 37.7 (1.1) | 37.6 (1.1) | 0.125 |
| Respiratory rate (breaths/min), mean (SD) | 22 (8) | 22 (8) | 22 (8) | 0.931 |
| Oxygen amount needed (L/min), median (IQR) | 3 (2–5) | 3 (2–5) | 2 (1–5) | 0.008 |
| Lymphocytes (109/L), median (IQR) | 0.9 (0.6–1.8) | 1.1 (0.7–2.0) | 0.9 (0.6–1.7) | 0.132 |
| Creatinine (μmol/L), median (IQR) | 89 (74–111) | 103 (77–143) | 92 (70–142) | <0.001 |
| Lactic acid dehydrogenase (U/L), median (IQR) | 345 (274–442) | 306 (252–423) | 299 (241–388) | <0.001 |
| C-reactive protein (mg/L), median (IQR) | 93 (48–155) | 79 (37–137) | 63 (31–119) | <0.001 |
N, number.
aCOPD, asthma, interstitial lung disease or lung cancer.
bKatz ADL score ≥ 2.
* P-value for any difference between groups, calculated with one-way ANOVA for normally distributed data, the Kruskal–Wallis test for skewed data and the χ2 test for categorical data.
Because with increasing frailty, both a shorter duration of symptoms and lower CRP levels were found, an additional analysis was executed to explore the association between the duration of symptoms until admission and the level of CRP. CRP levels were higher for patients with a longer duration of symptoms until admission (median 63 mg/L (<5 days) versus 82 mg/L (5–7 days) and 87 (>7 days); P < 0.001).
In-hospital outcomes for the total population and stratified by CFS category are shown in Table 3. In total, 38.4% of all patients died in the hospital. In-hospital mortality increased with higher CFS categories, from 29.2% in the lowest CFS categories to 47.3% in patients with CFS 6–9 (P < 0.001). During hospitalisation, 179 (13.5%) patients were admitted to the ICU, with decreasing numbers of admissions for patients with higher CFS. Delirium during admission was documented for 24.3% of all patients and for 41.2% of patients with CFS 6–9. One-third of patients (32.1%) were discharged to a nursing home (either for temporary rehabilitation or permanent stay), of whom 20.4% were already nursing home residents. In-hospital outcomes stratified per age category are shown in Supplementary A2.
Table 3 .
In-hospital outcomes for older hospitalised COVID-19 patients stratified by Clinical Frailty Scale
| All (N = 1,376) | Fit CFS 1–3 (N = 515) | Pre-frail CFS 4–5 (N = 288) | Frail CFS 6–9 (N = 313) | P-value* | |
|---|---|---|---|---|---|
| ICU admission, n (%) | 179 (13.5) | 120 (24.2) | 16 (5.8) | 7 (2.3) | <0.001 |
| Invasive ventilation in ICU, n (%) | 152 (85.9) | 106 (88.3) | 11 (68.8) | 4 (57.1) | 0.015 |
| Documented delirium during hospital admission, n (%) | 315 (24.3) | 74 (15.2) | 60 (21.7) | 127 (41.2) | <0.001 |
| Hospital length of stay (days), median (IQR) | 6 (3–10) | 6 (3–11) | 6 (3–10) | 6 (3–10) | 0.487 |
| In-hospital mortality, n (%)a | 499 (38.4) | 135 (29.2) | 116 (41.4) | 148 (47.3) | <0.001 |
| Discharge destination, n (%)b | <0.001 | ||||
| Home | 484 (55.5) | 240 (63.7) | 91 (52.9) | 57 (34.5) | |
| Nursing home | 280 (32.1) | 75 (19.9) | 64 (37.2) | 96 (58.2) | |
| Other hospital | 75 (8.6) | 52 (13.8) | 8 (4.7) | 0 (0.0) | |
| Other | 33 (3.8) | 10 (2.7) | 9 (5.2) | 12 (7.3) |
N, number. Missing: 260 Clinical Frailty Scale, 48 ICU admission, 5 ICU length of stay, 2 ventilation, 32 days of ventilation, 80 delirium, 2 hospital length of stay, 5 in-hospital mortality, 5 discharge destination.
aIn-hospital mortality for patients who were not discharged to another hospital (N = 1301).
bDischarge destination of non-deceased patients (N = 877).
* P-value for any difference between groups, calculated with the Kruskal–Wallis test for skewed data and the χ2 test for categorical data.
Frail patients had a higher risk of in-hospital mortality independent of demographics, co-morbidities and COVID-19 symptoms (Table 4). Compared with patients with CFS 1–3, patients with CFS 4–5 had a two times higher risk of in-hospital mortality (OR 2.0 (95% CI 1.3–3.0)) and patients with CFS 6–9 had a three times higher risk of in-hospital mortality (OR 2.8 (95% CI 1.8–4.3)). Sensitivity analyses showed the same results with the Katz ADL score instead of CFS (Supplementary A3) and with separate co-morbidities instead of the CCI score (Supplementary A4). In addition, the results were similar for the selection of patients in whom the COVID diagnosis was confirmed by PCR technique (data not shown).
Table 4 .
Univariable and multivariable associations of baseline characteristics and in-hospital mortality for older hospitalised COVID-19 patients
| Univariable model | Multivariable model | ||||
|---|---|---|---|---|---|
| N | OR (95% CI) | P-value | OR (95% CI) | P-value | |
| Demographics | |||||
| Age (per year) | 1,301 | 1.1 (1.0–1.1) | <0.001 | 1.0 (1.0–1.1) | 0.056 |
| Male | 1,299 | 1.5 (1.2–1.8) | 0.002 | 1.9 (1.3–2.7) | <0.001 |
| Living in institution | 1,247 | 1.5 (1.1–2.2) | 0.025 | ||
| Co-morbidity | |||||
| Charlson Comorbidity Index (per point) | 1,285 | 1.1 (1.0–1.2) | 0.001 | 1.0 (0.9–1.1) | 0.583 |
| History of chronic lung diseasea | 1,301 | 1.1 (0.9–1.4) | 0.388 | ||
| History of hypertension | 1,299 | 1.2 (0.9–1.5) | 0.134 | ||
| History of diabetes | 1,297 | 1.5 (1.1–1.8) | 0.003 | ||
| History of myocardial infarction | 1,300 | 1.6 (1.2–2.1) | 0.001 | ||
| History of dementia | 1,299 | 1.4 (1.0–2.0) | 0.089 | ||
| Smoking | |||||
| Never | 413 | Ref | Ref | ||
| Ex-smoker | 478 | 1.1 (0.9–1.5) | 0.359 | ||
| Current | 81 | 1.4 (0.9–2.3) | 0.136 | ||
| Body mass index | |||||
| <25 | 400 | Ref | Ref | ||
| 25–30 | 392 | 1.1 (0.8–1.4) | 0.657 | ||
| >30 | 228 | 1.0 (0.7–1.4) | 0.909 | ||
| Geriatric measurements | |||||
| Katz ADL score ≥2b | 1,131 | 1.6 (1.2–2.0) | <0.001 | ||
| Risk of falling | 1,067 | 1.2 (0.9–1.6) | 0.123 | ||
| Risk of delirium | 1,119 | 1.7 (1.3–2.1) | <0.001 | ||
| Risk of malnutrition | 996 | 1.0 (0.7–1.4) | 0.998 | ||
| Clinical Frailty Scale | |||||
| 1–3 | 463 | Ref | Ref | Ref | Ref |
| 4–5 | 280 | 1.7 (1.2–2.3) | 0.001 | 2.0 (1.3–3.0) | 0.001 |
| 6–9 | 313 | 2.2 (1.6–2.9) | <0.001 | 2.8 (1.8–4.3) | <0.001 |
| Disease severity indicators | |||||
| Duration of symptoms till admission | |||||
| <5 days | 392 | 1.1 (0.8–1.5) | 0.457 | 1.0 (0.6–1.4) | 0.807 |
| 5–7 days | 329 | Ref | Ref | Ref | Ref |
| >7 days | 438 | 0.7 (0.5–0.9) | 0.012 | 0.7 (0.5–1.0) | 0.065 |
| Temperature (°C) | |||||
| <36.5 | 192 | 0.9 (0.6–1.3) | 0.526 | ||
| 36.5–38.5 | 779 | Ref | Ref | ||
| >38.5 | 266 | 1.6 (1.2–2.1) | 0.001 | ||
| Respiratory rate (breaths/min) | |||||
| <15 | 126 | 0.8 (0.5–1.3) | 0.335 | ||
| 15–20 | 477 | Ref | Ref | ||
| >20 | 620 | 2.0 (1.5–2.5) | <0.001 | ||
| Oxygen amount needed (L/min) | |||||
| 0 | 236 | Ref | Ref | Ref | Ref |
| 1–5 | 692 | 2.1 (1.5–3.0) | <0.001 | 1.8 (1.1–2.8) | 0.015 |
| >5 | 227 | 8.5 (5.6–13.0) | <0.001 | 8.2 (4.7–14.5) | <0.001 |
| Lymphocytes (109/L) | |||||
| <1.0 | 528 | 1.5 (1.1–1.9) | 0.008 | ||
| 1.0–3.5 | 357 | Ref | Ref | ||
| >3.5 | 175 | 1.7 (1.2–2.5) | 0.005 | ||
| Lactic acid dehydrogenase (U/L) | |||||
| 0–249 | 241 | Ref | Ref | ||
| >250 | 861 | 1.6 (1.2–2.2) | 0.004 | ||
| Creatinine (μmol/L) | |||||
| <60 | 99 | Ref | Ref | ||
| 61–100 | 596 | 1.3 (0.8–2.2) | 0.258 | ||
| 101–130 | 216 | 1.9 (1.1–3.2) | 0.018 | ||
| >130 | 306 | 4.1 (2.5–6.8) | <0.001 | ||
| C-reactive protein (mg/L) | |||||
| <10 | 79 | Ref | Ref | Ref | Ref |
| 10–100 | 662 | 3.1 (1.7–5.9) | <0.001 | 2.6 (1.1–6.0) | 0.024 |
| >100 | 479 | 5.0 (2.6–9.5) | <0.001 | 3.7 (1.6–8.6) | 0.003 |
N, number.
aCOPD, asthma, interstitial lung disease or lung cancer.
bRisk of physical impairment.
As an exploration of predictive performance, we additionally calculated the discriminative performance of age (AUC 0.59 (95% CI 0.56–0.62)), CFS (AUC 0.63 (95% CI 0.59–0.66)) and age and CFS combined (AUC 0.64 (95% CI 0.61–0.68)) on in-hospital mortality.
Discussion
In the present study among older hospitalised COVID-19 patients in the Netherlands, the in-hospital mortality was 38%. Patients with frailty were hospitalised after a shorter duration of symptoms prior to admission and with lower oxygen demand and CRP levels. Frailty, as measured with the CFS, was associated with in-hospital mortality independent of age, sex, co-morbidities and disease severity.
The mortality rate of almost 40% among older COVID-19 patients is within the range of earlier reported studies, reporting in-hospital mortality from 27 [5] to 60% [2]. This range may be attributable to differences in patient selection, such as differences in average level of frailty or socio-economic status. Furthermore, small sample size of some studies may have contributed to chance findings. Previous studies have reported unequivocal associations of the CFS with in-hospital mortality. In line with the large multicentre study, which was the first to be reported in the COVID-19 in Older PEople (COPE) study [5], we found an independent association of CFS with mortality, independent of age, CRP levels, co-morbidity index and COVID-19 symptom severity. Studies reporting no association were substantially smaller and single-centre studies [2, 10, 11], possibly resulting in selection bias. Future meta-analyses, using individual patient data, should study the overall effects and sources of heterogeneity.
In our study, patients with higher CFS scores had a shorter duration of COVID-19 symptoms prior to admission and had both a lower oxygen demand and lower CRP levels. These results suggest that frail older COVID-19 patients have more difficulties to cope with the infection and have to be hospitalised with less severe symptoms than fit patients. Previous studies have also shown that the same level of disease severity results in higher mortality among patients with frailty who present to the emergency department [20], which corresponds to our finding that—despite presenting earlier and having less disease severity—frail older patients have higher mortality rates. It has been hypothesised that frail older people might have a more modest and less destructive immune response (reflected by lower CRP levels) [11], but in our study, this did not outweigh the effect of frailty on mortality risk. Also, older patients may have presented with different symptoms than younger patients, such as syncope or delirium as a presenting symptom [21]. These findings stress the importance of assessing frailty in older patients with COVID-19 to inform advance care planning, deliver appropriate care and to initiate early comprehensive geriatric assessment.
Although the CFS is independently associated with mortality, our study does not provide evidence supporting treatment decisions based on CFS alone. The predictive value of the CFS for in-hospital mortality, although higher than calendar age, was weak. The CFS was not originally developed as a triage tool and its predictive performance should be studied further. Our observational data does not support any conclusion on whether that selection on frailty was justified either for hospitalisation as we have no comparison to those patients who did not present to the hospital, nor for ICU admission as we have very few and only observational data. Although there was no absolute shortage of ICU capacity and ICU guidelines were not formally implemented, the recommendations of the guidelines may have influenced the selection of patients who were admitted to crowded ICU’s and therefore impacted mortality rates. Our results show that only very few frail patients were admitted to the ICU. Furthermore, a quarter of older COVID-19 patients had a delirium during admission, which probably is an underestimation because delirium is often not well documented. Future research should study the prevalence, determinants and outcomes for delirium in this population.
Our study has some limitations. First, data were collected retrospectively for most patients, potentially introducing selection bias. However, registries on patients admitted with COVID-19 were made in all hospitals, suggesting we did not miss substantial numbers of patients. Second, for patients transferred to other hospitals, data upon baseline and follow-up were incomplete. However, since this procedure was random and the proportion of exclusions (10 and 5%, respectively) were relatively small, we do not think that this might have influenced our findings. Third, the CFS was predominantly determined retrospectively. This might introduce potential source of bias, as the assessor was not blinded for the outcome and because of possible systematic differences with the prospectively established CFS. However, a recent study showed good correlation between categories of CFS (fit (CFS 1–3), pre-frail (CFS 4–5) and frail (CFS 6–9)) when collected retrospectively versus prospectively [22]. Furthermore, the CFS was predominantly determined based on the prospectively collected VMS data from the Dutch National Safety Management System, which has previously been validated to predict mortality in older emergency department patients [23]. In addition, our sensitivity analyses found similar results for the Katz ADL as for the CFS, suggesting validity of the use of the CFS. More large-scale prospective studies using the CFS are needed to fully establish its predictive potential. Fourth, because of the retrospective and multicentre nature of the data collection in the middle of a pandemic, there were relatively high numbers of missing variables due to different protocols in the various hospitals. The CFS was missing in a quarter of all patients, which may have introduced selection bias. There are also several strengths. First, data were collected from 15 hospitals from various regions in the Netherlands, both academic hospitals and teaching hospitals, yielding a representative sample of COVID-19 patients in the Netherlands. Second, geriatric parameters were collected prospectively as part of the National Safety Management system. Third, data were available on the combination of demographics, physiological parameters, disease severity, co-morbidity and geriatric parameters and frailty.
In conclusion, the in-hospital mortality of older hospitalised COVID-19 patients in the Netherlands was 38%. Frailty was independently associated with higher in-hospital mortality, even though COVID-19 patients with frailty presented earlier to the hospital with less severe symptoms.
Supplementary Material
Acknowledgements
The authors are very grateful to J.G. den Hollander, MD, PhD, M.C. de Vogel, MD (Department of Internal Medicine, Maasstad Hospital, Rotterdam) and to G.J. Waverijn, PhD (Department of Business Intelligence, Maasstad Hospital, Rotterdam) for their equal and significant contribution in data acquisition and critically reviewing the (previous versions of this) manuscript. In addition, they have agreed on the journal to which the article will be submitted and agreed to be accountable for the contents of the article.
Contributor Information
Laura C Blomaard, Section Gerontology and Geriatrics, Department of Internal Medicine, Leiden University Medical Center, Leiden, the Netherlands.
Carolien M J van der Linden, Department of Geriatrics, Catharina Hospital, Eindhoven, the Netherlands.
Jessica M van der Bol, Department of Geriatrics, Reinier de Graaf Hospital, Delft, the Netherlands.
Steffy W M Jansen, Department of Geriatrics, Catharina Hospital, Eindhoven, the Netherlands.
Harmke A Polinder-Bos, Section Geriatrics, Department of Internal Medicine, Erasmus Medical Center, Rotterdam, the Netherlands.
Hanna C Willems, Section Geriatrics, Department of Internal Medicine, Amsterdam University Medical Center, location AMC, Amsterdam, the Netherlands.
Jan Festen, KBO-PCOB, Etten-Leur, the Netherlands.
Dennis G Barten, Department of Emergency Medicine, VieCuri Medical Center, Venlo, the Netherlands.
Anke J Borgers, Department of Geriatrics, Deventer Hospital, Deventer, the Netherlands.
Jeannet C Bos, Department of Internal Medicine, Reinier de Graaf Hospital, Delft, the Netherlands.
Frederiek van den Bos, Department of Geriatrics, University Medical Center Utrecht, Utrecht, the Netherlands.
Esther J M de Brouwer, Department of Geriatrics, Deventer Hospital, Deventer, the Netherlands.
Floor J A van Deudekom, Department of Internal Medicine and Geriatrics, OLVG Hospital, Amsterdam, the Netherlands.
Suzanne C van Dijk, Department of Geriatric Medicine, Franciscus Gasthuis & Vlietland, Rotterdam, the Netherlands.
Mariëlle H Emmelot-Vonk, Department of Geriatrics, University Medical Center Utrecht, Utrecht, the Netherlands.
Raya E S Geels, Section Gerontology and Geriatrics, Department of Internal Medicine, Leiden University Medical Center, Leiden, the Netherlands; Department of Geriatrics, Alrijne Hospital, Leiderdorp, the Netherlands.
Esther M M van de Glind, Department of Geriatrics, Alrijne Hospital, Leiderdorp, the Netherlands.
Bas de Groot, Department of Emergency Medicine, Leiden University Medical Center, Leiden, the Netherlands.
Liesbeth Hempenius, Department of Geriatric Medicine, Medical Center Leeuwarden, Leeuwarden, the Netherlands.
Ad M Kamper, Department of Internal Medicine, Isala Hospital, Zwolle, the Netherlands.
Linda M Kampschreur, Department of Internal Medicine, Medical Center Leeuwarden, Leeuwarden, the Netherlands.
Marre M M de Koning, Department of Geriatric Medicine, Franciscus Gasthuis & Vlietland, Rotterdam, the Netherlands.
Geert Labots, Department of Internal Medicine, Haga Teaching Hospital, The Hague, the Netherlands.
Roy Looman, Section Geriatrics, Department of Internal Medicine, Zaans Medical Center, Zaandam, the Netherlands.
Jacinta A Lucke, Department of Emergency Medicine, Spaarne Gasthuis, Haarlem, the Netherlands.
Huub A A M Maas, Department of Geriatric Medicine, Elisabeth-TweeSteden Hospital, Tilburg, the Netherlands.
Francesco U S Mattace-Raso, Section Geriatrics, Department of Internal Medicine, Erasmus Medical Center, Rotterdam, the Netherlands.
Rachida el Moussaoui, Department of Internal Medicine, Maasstad Hospital, Rotterdam, the Netherlands.
Barbara C van Munster, Department of Internal Medicine and Geriatrics, University Medical Center Groningen, Groningen, the Netherlands.
Cees van Nieuwkoop, Department of Internal Medicine, Haga Teaching Hospital, The Hague, the Netherlands.
Leanne (B L E) Oosterwijk, Section Geriatrics, Department of Internal Medicine, Erasmus Medical Center, Rotterdam, the Netherlands.
Marlies (E M) Regtuijt, Department of Geriatrics, Hospital Group Twente, Almelo, the Netherlands.
Sarah H M Robben, Department of Geriatric Medicine, Elisabeth-TweeSteden Hospital, Tilburg, the Netherlands.
Rikje Ruiter, Department of Internal Medicine, Maasstad Hospital, Rotterdam, the Netherlands; Department of Epidemiology, Erasmus Medical Center, Rotterdam, the Netherlands.
Aisha M Salarbaks, Department of Geriatrics, Hospital Group Twente, Almelo, the Netherlands.
Henrike J Schouten, Department of Geriatric Medicine, Gelre Hospitals, Apeldoorn, the Netherlands.
Orla M Smit, Section Geriatrics, Department of Internal Medicine, Zaans Medical Center, Zaandam, the Netherlands.
Rosalinde A L Smits, Section Gerontology and Geriatrics, Department of Internal Medicine, Leiden University Medical Center, Leiden, the Netherlands.
Petra E Spies, Department of Geriatric Medicine, Gelre Hospitals, Apeldoorn, the Netherlands.
Ralph Vreeswijk, Department of Geriatrics, Spaarne Gasthuis, Haarlem, the Netherlands.
Oscar J de Vries, Department of Internal Medicine and Geriatrics, OLVG Hospital, Amsterdam, the Netherlands.
Marjolein A Wijngaarden, Section Gerontology and Geriatrics, Department of Internal Medicine, Leiden University Medical Center, Leiden, the Netherlands.
Caroline E Wyers, Department of Internal Medicine, VieCuri Medical Center, Venlo, the Netherlands.
Simon P Mooijaart, Section Gerontology and Geriatrics, Department of Internal Medicine, Leiden University Medical Center, Leiden, the Netherlands.
Declaration of Conflict of Interest
None.
Declaration of Sources of Funding
None.
References
- 1. Chen T, Dai Z, Mo Pet al. Clinical characteristics and outcomes of older patients with coronavirus disease 2019 (COVID-19) in Wuhan, China (2019): a single-centered, retrospective study. J Gerontol A Biol Sci Med Sci 2020; 75: 1788–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Owen RK, Conroy SP, Taub Net al. Comparing associations between frailty and mortality in hospitalised older adults with or without COVID-19 infection: a retrospective observational study using electronic health records. Age Ageing 2020. doi: 10.1093/ageing/afaa167. Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Chinnadurai R, Ogedengbe O, Agarwal Pet al. Older age and frailty are the chief predictors of mortality in COVID-19 patients admitted to an acute medical unit in a secondary care setting- a cohort study. BMC Geriatr 2020; 20: 409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mendes A, Serratrice C, Herrmann FRet al. Predictors of in-hospital mortality in older patients with COVID-19: the COVIDAge study. J Am Med Dir Assoc 2020; 21: 1546–54.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Hewitt J, Carter B, Vilches-Moraga Aet al. The effect of frailty on survival in patients with COVID-19 (COPE): a multicentre, European, observational cohort study. Lancet Public Health 2020; 5: e444–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. De Smet R, Mellaerts B, Vandewinckele Het al. Frailty and mortality in hospitalized older adults with COVID-19: retrospective observational study. J Am Med Dir Assoc 2020; 21: 928–32.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Aw D, Woodrow L, Ogliari G, Harwood R. Association of frailty with mortality in older inpatients with COVID-19: a cohort study. Age Ageing 2020; 49: 915–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kundi H, Çetin E, Canpolat Uet al. The role of frailty on adverse outcomes among older patients with COVID-19. J Infect 2020; 81: 944–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Petermann-Rocha F, Hanlon P, Gray SRet al. Comparison of two different frailty measurements and risk of hospitalisation or death from COVID-19: findings from UK Biobank. BMC Med 2020; 18: 355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Miles A, Webb TE, McLoughlin BCet al. Outcomes from COVID-19 across the range of frailty: excess mortality in fitter older people. Eur Geriatr Med 2020; 11: 851–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Knopp P, Miles A, Webb TEet al. Presenting features of COVID-19 in older people: relationships with frailty, inflammation and mortality. Eur Geriatr Med 2020; 11: 1089–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Leidraad Triage thuisbehandeling versus verwijzen naar het ziekenhuis bij oudere patiënt met (verdenking op) COVID-19. Federatie Medisch Specialisten, NIV, Nederlandse Vereniging voor Klinische Geriatrie, NHG, Verenso, KNMG. 2020; Volume 1.2. https://www.demedischspecialist.nl/sites/default/files/Leidraad%20triage%20thuisbehandeling%20versus%20verwijzen%20oudere%20pati%C3%ABnt%20COVID-19%2027112020.pdf (9 September 2020, date last accessed). [Google Scholar]
- 13. Draaiboek Pandemie. Nederlandse Vereniging voor Intensive Care. 2020; Volume 1.3. https://www.demedischspecialist.nl/sites/default/files/Draaiboek%20pandemie%20deel%201.pdf (9 September 2020, date last accessed). [Google Scholar]
- 14. Rockwood K, Song X, MacKnight Cet al. A global clinical measure of fitness and frailty in elderly people. CMAJ 2005; 173: 489–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987; 40: 373–83. [DOI] [PubMed] [Google Scholar]
- 16. VMS. Praktijkgids `Kwetsbare Ouderen'. Den Haag: VMS Veiligheidsprogramma, 2009. 978-94-90101-04-6. https://www.vmszorg.nl/wp-content/uploads/2017/11/web_2009.0104_praktijkgids_kwetsbare_ouderen.pdf (9 September 2020, date last accessed). [Google Scholar]
- 17. Katz S, Ford AB, Moskowitz RW, Jackson BA, Jaffe MW. Studies of illness in the aged: the index of ADL: a standardized measure of biological and psychosocial function. JAMA 1963; 185: 914–9. [DOI] [PubMed] [Google Scholar]
- 18. Kruizenga HM, Seidell JC, Vet HC, Wierdsma NJ, van Bokhorst-de van der Schueren MA. Development and validation of a hospital screening tool for malnutrition: the short nutritional assessment questionnaire (SNAQ). Clin Nutr 2005; 24: 75–82. [DOI] [PubMed] [Google Scholar]
- 19. Stratton RJ, Hackston A, Longmore Det al. Malnutrition in hospital outpatients and inpatients: prevalence, concurrent validity and ease of use of the 'malnutrition universal screening tool' ('MUST') for adults. Br J Nutr 2004; 92: 799–808. [DOI] [PubMed] [Google Scholar]
- 20. Blomaard LC, Speksnijder C, Lucke JAet al. Geriatric screening, triage urgency, and 30-day mortality in older emergency department patients. J Am Geriatr Soc 2020; 68: 1755–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Martín-Sánchez FJ, Del Toro E, Cardassay Eet al. Clinical presentation and outcome across age categories among patients with COVID-19 admitted to a Spanish emergency department. Eur Geriatr Med 2020; 11: 829–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Stille K, Temmel N, Hepp J, Herget-Rosenthal S. Validation of the Clinical Frailty Scale for retrospective use in acute care. Eur Geriatr Med 2020; 11: 1009–15. [DOI] [PubMed] [Google Scholar]
- 23. Snijders BMG, Emmelot-Vonk MH, Souwer ETD, Kaasjager HAH, Bos F. Prognostic value of screening instrument based on the Dutch National VMS guidelines for older patients in the emergency department. Eur Geriatr Med 2020. doi: 10.1007/s41999-020-00385-0. Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


