A 24-day-old female neonate was admitted to the paediatric cardiac intensive care unit with features of cardiogenic shock. She was born at term with a birthweight of 3·75 kg to a mother with a history of RT-PCR-positive, mild COVID-19 at 31 weeks gestation, which was managed with symptomatic and supportive measures. There were no fetal complications during the illness. The mother was RT-PCR-negative at delivery. Following an uncomplicated vaginal delivery, the neonate required supportive care for 5 days in a neonatal intensive care unit for mild tachypnoea and was subsequently discharged home with normal clinical, radiological, and laboratory parameters; RT-PCR for SARS-CoV-2 was not done. She stayed well at home until day 22 of life when she required admission to a local hospital with heart failure and systemic hypoperfusion. There was no preceding history of fever, rash, loose stools, abdominal distention, lethargy, or seizures. Following inadequate response to treatment, she was referred to our department (the Department of Pediatric Cardiology at the Amrita Institute of Medical Sciences and Research Centre, Kochi, India), with suspicion of a critical congenital heart disease.
At presentation (day 0) she was afebrile, drowsy, tachycardic (heart rate: 160 beats per minute), hypotensive (mean blood pressure: 25 mm Hg), and tachypnoeic (respiratory rate: 55 breaths per minute), with chest retractions, oxygen saturation of 97% on supplemental oxygen, cool peripheries, and delayed capillary refill. The precordium was hyperdynamic, with no cardiac murmurs; the chest was clear. Hepatomegaly was present. Neither neurological or musculoskeletal abnormalities nor dysmorphisms were noted. Erythema was noted at pressure points—over the occiput and at bilateral gluteal regions; no other skin or mucosal lesions were present. The patient had metabolic acidosis with an arterial pH of 7·173 and lactate concentration of 22 mmol/L. The electrocardiogram showed sinus rhythm and the chest x-ray showed cardiomegaly, with normal lung fields. The point-of-care echocardiogram showed severe biventricular dysfunction with a left ventricular ejection fraction of 10% and global hypokinesia. Coronary arteries were normal in origin and luminal dimensions, but appeared prominent and hyperechoic. Major congenital heart disease, including outflow tract obstructions, was ruled out. Features of primary cardiomyopathies or storage disorders were absent.
Initial management included mechanical ventilation, inotropic support (intravenous infusions of epinephrine [0·04 μg/kg per min] and milrinone [0·5 μg/kg per min]), diuretic (intravenous infusion of frusemide [2·0 mg/kg per day]), antimicrobial cover (intravenous infusion of parenteral cefoperazone plus sulbactam [50·0 mg/kg every 8 h]), and appropriate fluid and electrolyte management.
Cardiac markers were substantially elevated, with an N-terminal-pro-B-type natriuretic peptide concentration of 157 000 pg/mL, a creatine kinase myocardial band concentration of 14·7 ng/mL, and a troponin T concentration of 1·23 ng/mL (figure 1 ). The aspartate aminotransferase concentration was 2240 IU/L and the alanine aminotransferase concentration 866 IU/L, whereas the blood urea concentration was 75 mg/dL and the serum creatinine concentration 0·9 mg/dL. Inflammatory markers were also markedly elevated: the serum ferritin concentration was 56 400 ng/mL (reference range 12–327 ng/mL) and the serum lactate dehydrogenase concentration 6280 U/L (0–451 U/L). The C-reactive protein concentration was 6·5 mg/L and the D-Dimer concentration 20 μg/mL. There was mild leucocytosis: total leucocyte count was 16 400 cells per microlitre (75% neutrophils) and platelet count was 185 000 cells per microlitre. Serum electrolytes, thyroid hormones, and vitamin D concentrations were within normal ranges. Two blood cultures—at admission, and 5 days later—were sterile. Qualitative antibody assay (VIDAS, Biomerieux, Marcy-l'Étoile, France) detected IgG antibodies against SARS-CoV-2 spike protein in both maternal and neonatal serum samples, with a titre of 20·11 in the maternal serum sample and a titre of 10·00 in the neonatal serum sample; no IgM antibodies were detected. Nasopharyngeal swabs and rectal swab were negative for SARS-CoV-2 on RT-PCR. Cerebrospinal fluid was not tested. Qualitative RT-PCR panel of the nasal swab for common respiratory viruses detected human rhinovirus; none of the other viruses on the panel were detected.
Figure 1.
Timeline of clinical course and laboratory markers
The timeline from the point of diagnosis of COVID-19 in the mother through the presentation and clinical course of the patient, with trends of inflammatory markers (serum ferritin, serum dehydrogenase concentration), coagulopathy marker (D-Dimer), cardiac markers (NT-Pro BNP, CK-MB, troponin T), hepatic marker (SGOT), and renal parameters (blood urea, serum creatinine). CK-MB=creatine kinase myocardial band. IVIG=intravenous immune globulin. LDH=lactate dehydrogenase. NT-Pro BNP=N-terminal brain natriuretic peptide.
This clinical presentation with severe acute myocardial injury, multiorgan dysfunction, elevated inflammatory markers, temporal association with prenatal exposure to COVID-19, and laboratory evidence of IgG antibodies to SARS-CoV-2, in the absence of other plausible explanations, led us to consider the possibility of a hyperinflammatory response to prenatal exposure to COVID-19.
Immunomodulatory therapy was instituted on day 1 of hospitalisation, with a single dose of intravenous immunoglobulin (2 g/kg; slow infusion) as well as an injection of methylprednisolone once daily for 3 days (5 mg/kg; slow infusion). Continuous heparin infusion was added in a dose of 10 U/kg per h and titrated for optimal anticoagulation. Calcium (calcium gluconate intravenous infusion of 4 mg/kg per day), vitamin C (50 mg once daily, orally), vitamin D (400 IU once daily, orally), and zinc (10 mg once daily, orally) were supplemented. No antiviral agents were prescribed.
Clinical stability was attained within the next 48 h. Laboratory markers of inflammation and end-organ functions showed improvements starting 24 h after the initiation of treatment. Serial echocardiograms showed progressive improvement in ventricular systolic function. The dose of methylprednisolone was halved to 2·5 mg/kg per day on days 4 and 5. Following an unsuccessful attempt at extubation on day 5, mechanical ventilation and inotropic support were electively continued for the next 5 days. Oral prednisolone (2 mg/kg per day) was added after stopping methylprednisolone on day 6.
The patient was successfully extubated to non-invasive ventilation on day 11 of admission and to oxygen by nasal cannula on day 14. Inotropes were tapered off by day 13, and intravenous diuretic and heparin were stopped by day 15. Enalapril (initiated at 0·25 mg and gradually stepped up to 0·75 mg twice daily, orally), frusemide (1 mg/kg twice daily, orally) and aspirin (3 mg/kg once daily, orally) were added. Figure 1 shows the trends of the laboratory markers of inflammation, myocardial injury, and hepatic and renal functions during the clinical course. The trends for leucocyte counts, platelet counts, and C-reactive protein concentrations are depicted in the appendix (p 1).
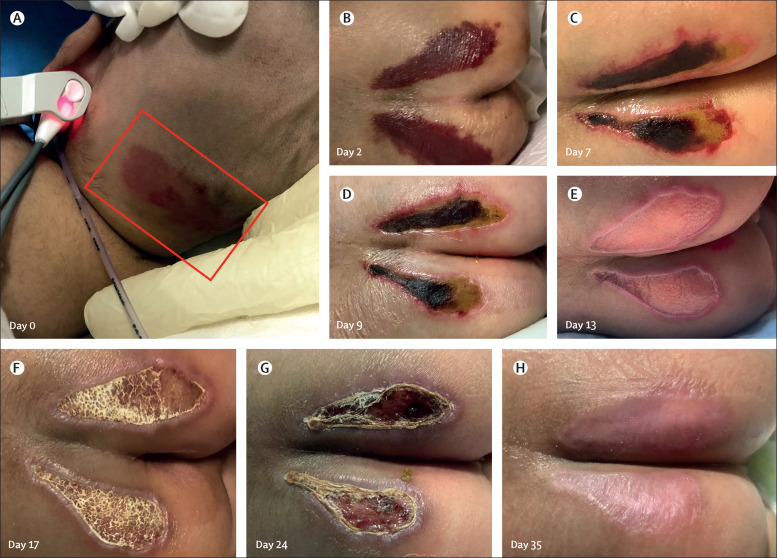
Erythema over the scalp (figure 2A ) spontaneously resolved. Gluteal skin lesions progressed into symmetrical, well demarcated purple patches by day 2 of admission (figure 2B). Superficial layers subsequently underwent necrosis, scab formation, and sloughing, leaving behind sharply marginated ulcers that finally healed with epithelialisation (figures 2B–2H). These ulcers were conservatively managed with topical antibiotic (mupirocin ointment [2·15%, twice daily]), zinc oxide ointment (15%, twice daily), and posture care.
Figure 2.
Skin manifestations in the patient
(A) Erythema (inside the box) over the occipital scalp region, noted at initial presentation. (B–H) Evolution of skin lesions over the buttocks during the course of illness.
Neurological status remained normal throughout the clinical course. In view of the modest fall in platelet counts, the elevated D-Dimer concentration, and the bruise-like gluteal skin lesion, point-of-care cranial ultrasound was done on day 3 of hospitalisation, which excluded intracranial bleed; no other neuroimaging was done. Patient was successfully transitioned out of the intensive care unit to the wards on day 20 of admission and discharged home on day 29. The echocardiogram at discharge showed a left ventricular ejection fraction of 58%, with mild dyskinesia of the apical-septal regions; coronaries appeared less hyperechoic than at presentation. Discharge medications included oral enalapril, aspirin, tapering doses of tablet prednisolone, and oral supplements (calcium, vitamin D, vitamin C, and zinc).
Acute COVID-19 is significantly less common and less severe in children, with few reports of neonates being affected.1, 2 This optimistic outlook has been altered by reports of multisystem inflammatory syndrome in children (MIS-C; also known as paediatric multisystem inflammatory syndrome temporally associated with SARS-CoV-2 [PIMS-TS]), a rare but potentially fatal hyperinflammatory response that can occur in children 2–6 weeks after SARS-CoV-2 exposure, with a median age of occurrence between 7 and 9 years.3, 4, 5 Immunomodulatory therapy—particularly intravenous immunoglobulin, corticosteroids, and interleukin-1-receptor antagonist (anakinra)—have been successfully used for treatment. Molecular mechanisms underlying MIS-C are not fully understood. Consiglio and colleagues6 showed that the pathophysiology of MIS-C is distinct from the cytokine storm of severe acute COVID-19 as well as from the inflammatory response of Kawasaki disease, in addition to finding evidence of autoantibody-mediated pathology.
There is debate on the possibility and effect of vertical transmission of SARS-CoV-2 infection to the fetus.7, 8, 9, 10, 11 A systematic review and meta-analysis found no conclusive evidence for in-utero transmission among 1316 pregnant women across 39 studies.9 Isolated case reports from China have supported the possibility of vertical transmission.10, 11 Vivanti and colleagues12 provided virological and pathological evidence for late-gestational maternal-fetal transmission of SARS-CoV-2 in one case. In a study on transplacental transfer of SARS-CoV-2 antibodies involving a cohort of 1471 mother–newborn dyads, Flannery and colleagues13 showed efficient transplacental transfer of IgG antibodies in 72 (87%) of 83 seropositive mothers. There is little information regarding the protection offered by maternal antibodies, as well as the possibility of hyperinflammatory responses in fetuses and neonates following antenatal exposure to SARS-CoV-2. One previous report indicated that potential MIS-C was suspected in a neonate presenting with persistent pulmonary hypertension, elevated inflammatory markers, and anti-SARS-CoV-2 IgG antibodies.14
To the best of our knowledge, this Case Report is the first description of severe hyperinflammatory syndrome in a neonate presenting catastrophically with myocarditis, shock, and multiorgan dysfunction following proven prenatal exposure to COVID-19. The diagnosis is supported by presence of anti-SARS-CoV-2 IgG antibodies and the excellent response to immunomodulatory therapy. Detection of human rhinovirus in nasal-swab RT-PCR is likely to be incidental because it has not been reported to be associated with such presentation in neonates. Additional clinical clues in our case were the coronary findings (appendix p 2) and skin manifestations. Coronary vasculitis has been described in association with MIS-C, although neither specific nor diagnostic. The morphology and evolution of the skin lesions over the buttocks might indicate acro-ischaemia analogous to so-called COVID toes seen in older children in association with the coagulopathy and microangiopathy of severe COVID-19.15, 16 Young and colleagues17 have described three adult patients presenting with ulcerations on the buttocks in the setting of COVID-19 coagulopathy.
MIS-C can occur in a neonate following in-utero exposure to SARS-CoV-2, resulting in multiple organ injury. This case highlights the need to better understand the effect of COVID-19 on the maternal–fetal dyad. It might have important implications for health-care professionals looking after peripartum women and their neonates, and also for the postnatal counselling and care of infants born with antenatal exposure to COVID-19. With pregnant women, globally, becoming more vulnerable to COVID-19 exposure because of relaxation of restrictions and setting in of pandemic fatigue, protecting the maternal–fetal dyad from SARS-CoV-2 through appropriate vaccination strategies and other measures might become an important public health need.
Acknowledgments
Acknowledgments
Parent of the patient provided informed written consent for publication of clinical information and photographs. This Case Report was approved by the Department of Pediatric Cardiology of the Amrita Institute of Medical Sciences and Research Centre, Kochi, India, with a waiver of informed ethical approval.
Contributors
MK and SB conceptualised and prepared the Case Report. SA, SM, SPL, SG, and JPJ contributed to the literature search, data collection, and data interpretation. SJ contributed to the literature search and manuscript preparation. AK helped with data collection, data interpretation, and microbiological investigations. MK, SB, and RKK had full access to all the data and verified it, and had final responsibility for the decision to submit for publication. AG and RKK supervised manuscript preparation and critically reviewed and edited the final manuscript.
Declaration of interests
We declare no competing interests.
Supplementary Material
References
- 1.De Rose DU, Piersigilli F, Ronchetti MP, et al. Novel coronavirus disease (COVID-19) in newborns and infants: what we know so far. Ital J Pediatr. 2020;46:56. doi: 10.1186/s13052-020-0820-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cook J, Harman K, Zoica B, Verma A, D'Silva P, Gupta A. Horizontal transmission of severe acute respiratory syndrome coronavirus 2 to a premature infant: multiple organ injury and association with markers of inflammation. Lancet Child Adolesc Health. 2020;4:548–551. doi: 10.1016/S2352-4642(20)30166-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Radia T, Williams N, Agrawal P, et al. Multi-system inflammatory syndrome in children & adolescents (MIS-C): a systematic review of clinical features and presentation. Paediatr Respir Rev. 2020 doi: 10.1016/j.prrv.2020.08.001. published online Aug 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alsaied T, Tremoulet AH, Burns JC, et al. Review of cardiac involvement in multisystem inflammatory syndrome in children. Circulation. 2021;143:78–88. doi: 10.1161/CIRCULATIONAHA.120.049836. [DOI] [PubMed] [Google Scholar]
- 5.Jain S, Sen S, Lakshmivenkateshiah S, et al. Multisystem inflammatory syndrome in children with COVID-19 in Mumbai, India. Indian Pediatr. 2020;57:1015–1019. doi: 10.1007/s13312-020-2026-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Consiglio CR, Cotugno N, Sardh F, et al. The immunology of multisystem inflammatory syndrome in children with COVID-19. Cell. 2020;183:968–981.e7. doi: 10.1016/j.cell.2020.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Karimi-Zarchi M, Neamatzadeh H, Dastgheib SA, et al. Vertical transmission of coronavirus disease 19 (COVID-19) from infected pregnant mothers to neonates: a review. Fetal Pediatr Pathol. 2020;39:246–250. doi: 10.1080/15513815.2020.1747120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Egloff C, Vauloup-Fellous C, Picone O, Mandelbrot L, Roques P. Evidence and possible mechanisms of rare maternal-fetal transmission of SARS-CoV-2. J Clin Virol. 2020;128 doi: 10.1016/j.jcv.2020.104447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Diriba K, Awulachew E, Getu E. The effect of coronavirus infection (SARS-CoV-2, MERS-CoV, and SARS-CoV) during pregnancy and the possibility of vertical maternal-fetal transmission: a systematic review and meta-analysis. Eur J Med Res. 2020;25:39. doi: 10.1186/s40001-020-00439-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dong L, Tian J, He S, et al. Possible vertical transmission of SARS-CoV-2 from an infected mother to her newborn. JAMA. 2020;323:1846–1848. doi: 10.1001/jama.2020.4621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zeng H, Xu C, Fan J, et al. Antibodies in infants born to mothers with COVID-19 pneumonia. JAMA. 2020;323:1848–1849. doi: 10.1001/jama.2020.4861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Vivanti AJ, Vauloup-Fellous C, Prevot S, et al. Transplacental transmission of SARS-CoV-2 infection. Nat Commun. 2020;11 doi: 10.1038/s41467-020-17436-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Flannery DD, Gouma S, Dhudasia MB, et al. Assessment of maternal and neonatal cord blood SARS-CoV-2 antibodies and placental transfer ratios. JAMA Pediatr. 2021 doi: 10.1001/jamapediatrics.2021.0038. published online Jan 29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Khaund Borkotoky R, Banerjee Barua P, Paul SP, Heaton PA. COVID-19-related potential multisystem inflammatory syndrome in childhood in a neonate presenting as persistent pulmonary hypertension of the newborn. Pediatr Infect Dis J. 2021 doi: 10.1097/INF.0000000000003054. published online Jan 12. [DOI] [PubMed] [Google Scholar]
- 15.Calvão J, Relvas M, Pinho A, Brinca A, Cardoso JC. Acro-ischaemia and COVID-19 infection: clinical and histopathological features. J Eur Acad Dermatol Venereol. 2020;34:e653–e754. doi: 10.1111/jdv.16687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fernandez-Nieto D, Jimenez-Cauhe J, Suarez-Valle A, et al. Characterization of acute acral skin lesions in nonhospitalized patients: a case series of 132 patients during the COVID-19 outbreak. J Am Acad Dermatol. 2020;83:e61–e63. doi: 10.1016/j.jaad.2020.04.093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Young S, Narang J, Kumar S, et al. Large sacral/buttocks ulcerations in the setting of coagulopathy: a case series establishing the skin as a target organ of significant damage and potential morbidity in patients with severe COVID-19. Int Wound J. 2020;17:2033–2037. doi: 10.1111/iwj.13457. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.