Abstract
Background/aims
Isoalantolactone (IATL) is one of multiple isomeric sesquiterpene lactones and is isolated from inula helenium. IATL has multiple functions such as antibacterial, antihelminthic and antiproliferative activities. IATL also inhibits pancreatic cancer proliferation and induces apoptosis by increasing ROS production. However, the detailed mechanism of IATL-mediated pancreatic cancer apoptosis remains largely unknown.
Methods
In current study, pancreatic carcinoma cell lines (PANC-1, AsPC-1, BxPC-3) and a mouse xenograft model were used to determine the mechanism of IATL-mediated toxic effects.
Results
IATL (20μM) inhibited pancreatic adenocarcinoma cell lines proliferation in a time-dependent way; while scratch assay showed that IATL significantly inhibited PANC-1 scratch closure (P<0.05); Invasion assays indicated that IATL significantly attenuated pancreatic adenocarcinoma cell lines invasion on matrigel. Signal analysis showed that IATL inhibited pancreatic adenocarcinoma cell proliferation by blocking EGF-PI3K-Skp2-Akt signal axis. Moreover, IATL induced pancreatic adenocarcinoma cell apoptosis by increasing cytosolic Caspase3 and Box expression. This apoptosis was mediated by inhibition of canonical wnt signal pathway. Finally, xenograft studies showed that IATL also significantly inhibited pancreatic adenocarcinoma cell proliferation and induced pancreatic adenocarcinoma cell apoptosis in vivo.
Conclusions
IATL inhibits pancreatic cancer proliferation and induces apoptosis on cellular and in vivo models. Signal pathway studies reveal that EGF-PI3K-Skp2-Akt signal axis and canonical wnt pathway are involved in IATL-mediated cellular proliferation inhibition and apoptosis. These studies indicate that IATL may provide a future potential therapy for pancreatic cancer.
Introduction
Pancreatic cancer is a pandemic disease and has increased all over the world in the past decades. Pancreatic cancer has been classified into exocrine and endocrine groups. The exocrine group is the majority (95%), in which ductal adenocarcinoma is the most common type. The endocrine group is characterized by hormone secretion, such as insulin, gastrin and glucagon from neuroendocrine tumor cells [1]. Pancreatic cancer is associated with a high death rate, poor outcome and rapid developments to later stages [2].
Pancreatic cancer is believed to be related to various epidemic factors, such as obesity, heavy alcoholic consumption, genetic causes, etc [3]. The tumorigenesis processes of pancreatic cancer may involve many signal pathways, such as TGFβ, PI3K-Akt, Wnt/β-Catenin, ROS, and cyclin-Dependent Kinase related signal pathways [4–7]. Recent studies indicate that not only genetic mutations, but also epigenetic modifications are involved in pancreatic tumorigenesis [8, 9].
Based on these signaling pathways, many novel therapeutic targets of pancreatic cancer have been developed. Current adjuvant chemotherapies include gemcitabine or 5-FU [10] while a combination chemotherapy such as FOLFIRINOX (folinic acid plus fluorouracil) has been found more effective than gemcitabine but has more side-effects [11]. Moreover, surgery with radiotherapy is also an option for pancreatic cancer patients [12]. Although more new medicines are being developed for clinical use, an ideal therapy does not yet exist for treating pancreatic cancer.
Sesquiterpene lactones are a group of chemicals containing lactone ring and normally exist in plants of asteraceae such as daisies and asters. Sesquiterpene lactones can be divided in several main classes: germacranolides, heliangolides, guaianolides, pseudoguaianolides, hypocretenolides and eudesmanolides. Isoalantolactone (IATL) is one of the isomeric sesquiterpene lactones isolated from inula helenium, which has shown multiple functions as an antibacterial, antihelminthic and antiproliferative agent [13]. IATL has also been reported to induce apoptosis in a pancreatic cancer cellular model by increasing ROS [14]. A recent study confirmed the anticancer effect of IATL by using combinations of alloalantolactone, alantolactone and IATL [15]. However, the detailed mechanism of IATL remains largely unknown.
In the current study, we used in vitro cellular and in vivo xenograft models to determine the effect of IATL on the fate of pancreatic cancer. Our study shows that IATL induces pancreatic cancer cell apoptosis by inhibition of the Wnt-β-catenin signal pathway. Furthermore, IATL inhibits pancreatic cancer cell proliferation by AMPK- Skp2-Akt signal pathway. Most importantly, the xenograft model shows that IATL inhibits pancreatic carcinoma cell growth and induces apoptosis in vivo.
Material and methods
Cell culture and IATL
Human pancreatic ductal adenocarcinoma (PDAC) cell lines (PANC-1, AsPC-1 and BxPC-3) were purchased from American Type culture collection (ATCC, Manassas, VA). All cell lines used in this study were authenticated and characterized, and the mycoplasma testing of these cell lines are negative. The cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum containing 100 units/mil penicillin and streptomycin. The cells were incubated at 37°C in 5%CO2 and were used at passages 5–20. All experiments and measurements were carried out in accordance with the relevant guidelines and regulations of the universities. IATL were purchased from Sigma-Aldrich (PHL89228, St. Louis, MO. USA).
Cell viability assay (MTT)
PANC-1, AsPC-1 and BxPC-3 cells were seeded in 96 wells plate (104/ well). The following day, the cells were treated with 5% DMSO or IATL for 72 hours. Cell proliferation was determined by MTT assay kit (Sigma, St. Louis, MO). The total live cells were also stained by trypan blue and then counted by TC 10 automated cell counter (Bio-Rad Inc., Hercules, CA) [16].
Wound closure assay and invasion assay
PANC-1 Cells were seeded in 6 wells plate and scratched by a sterilized 200μl pipette tip. After that, cells were treated with DMSO or IATL for 24 hours. Phase contrast images were taken at beginning and after 24 hours and 48 hours. The cell migration effects were measured by the closure area of scratch [17]. Invasion assays were performed on an 8μm pore size transwell (BD Falcon cell culture insert, BD Bioscience) with modifications. Invasion assays were performed in 24 well transwell inserts which were coated with 100 μL matrigel, 1×105 PANC-1, AsPC-1 and BxPC-3 cells were transfected with pLV-GFP virus (vectorbuilder, Chicago, Illinois). After 3 days incubation, more than 90% of each of the three cell lines were GFP-positive. The cells then were seeded into the insert. The cells were cultured in 10% FBS complete DMEM medium in presence or absence of ITAL. The bottom chambers contained 0.5ml 10% FBS complete DMEM medium. After 24 hours, the cells in the inserts were removed using a cotton swab. The cells on the bottom side of insert were covered by prolong Gold Antifade mountant with coverslides (Therm Fisher Scientific, Waltham, MA). The GFP positive cells were counted using a Nikon fluorescent microscope (Nikon, Eclipse TE2000-S, Nikon Instrument Inc, Melville, NY) [18].
Xenograft models
A total 36 BALB/c nude male mice (4–5 weeks old) were purchased and were divided into 6 groups (Beijing HFK Bioscience Co., Ltd., Beijing, China). The six groups included PANC-1, PANC-1/ IATL; AsPC-1, AsPC-1/ IATL; BxPC-3, BxPc-3/IATL treatment groups. All animal experiments were reviewed and approved by the Institutional Animal Care and Use committee at Sichuan University (protocol number: 20190089). The BALB/c nude male mice were kept in mouse cage (CAVENS, Changzhou, China) located in the mouse room with 24°C temperature, 50% humidity, the food for mice were purchased from CAVENS, the autoclaved drinking water were provided. Nestlets were provided for environmental enrichment. Once the mice were anesthetized by ketamine and xylazine (The used dose of ketamine/xylazine for this experiment 100 mg/kg and 10 mg/kg body weight. Here 1.6mg ketamine and 0.16mg xylazine were diluted in 100 μl PBS for intraperitoneal injection), PANC-1, AsPC-1 or BxPC-3 cells (1x106 cells in 100μl PBS) were injected into left flank of the mice subcutaneously. Mice were kept warm using heat lamp and be checked frequently 15 min until wake. For the health of mice, these mice were checked and food and water intake were monitored daily. IATL or PBS were given to the mice (0.5mg/kg, IP) once a week for 5 weeks. The tumor sizes were recorded every week. We believed that large volume tumor may be a better indictor to reflect the effect of IATL on tumor growth. Institutional Animal Care and Use Committee at Sichuan University specifically reviewed and believed that large tumor volumes do not bring additional and unnecessary discomfort to these mice. This Committee approved the use of large tumor volumes this study. The largest (a) and smallest diameters (b) of each xenograft were measured twice. Volume (V) was estimated using the formula V = 0.52×a2×b. The mean tumor volume were recorded as tumor growth curves for each group of mice. Both tumor weights and volume were also measured after the mice were euthanized by anesthesia of CO2 and dislocation of cervical spine at the end of experiment. The tumor growth was evaluated as the mean volume± SE. At the end of experiment, the IATL-mediated apoptosis in tumors was evaluated using flow cytometry [19, 20].
Apoptosis flow cytometry assay
The xeonograft cell apoptosis was also evaluated by flow cytometric detection of phosphotidyl serine externalization using the FITC annexin V apoptosis detection kit I (cat.no. 556547, BD Biosciences, San Jose, Ca). After the mice were treated with vehicle or IATL, xenografts were minced and washed once with cold PBS. The cells were then digested with collagenase and dispase for 30min at 37°C. The suspensions were filtered using a cell strainer (70μm). The filtered solution was centrifuged at 300g for 10min. The pellet was washed with PBS once. The cell pellets were resuspended in 1×binding buffer and incubated with 5μl FITC annexin V and 5 μl PI solution. After incubation for 15 minutes at room temperature, binding buffer was added. FITC annexin V and propidium iodide (PI) positive cells were determined by FACS caliber flow cytometry (BD Bioscience, San Jose, CA) [21].
Western blotting
Samples were harvested with RIPA buffer (cat.no. ab15634, Abcam) containing proteinase inhibitors and phosphatase inhibitors as per standard protocols. After sonication and centrifugation, the supernatant was collected, Laemmli sample buffer was added. And then the samples were boiled and subsequently analyzed by SDS–PAGE. After transfer to a nitrocellulose membrane (Bio-Rad, Inc., Hercules, CA), Western blotting was performed using appropriate primary antibodies and horseradish peroxidase-conjugated secondary antibodies prior to visualization via chemiluminescence (Amersham Biosciences, Piscataway, NJ). The used antibodies were listed as followed: Anti-AMPK (phosphor T183+T172) antibody (1:1500, cat.no. ab133448, Abcam, Cambridge, UK), anti-AMPK alpha 1 antibody (1:2000, cat.no. ab133448, Abcam), the customed phosphor-Skp2 S256 antibody is from Jiaxing Xinda Biotechnology Co. Ltd (1:1000), anti-Skp2 antbody (1:1000, cat.no. ab19877, Abcam), anti-AKT antibody (1:1000, cat.no. ab179463, Abcam), anti-AKT1 (phosphor S473) antibody (1:1000, cat.no. ab81283, Abcam), anti-caspase-3 antibody (1:2000, cat.no. ab813847, Abcam), anti-bax antibody (1:1000, cat.no. ab32503, Abcam), anti-phospho-GSK-3β (Ser9) antibody (1:1000, cat.no. #9336, Cell Signaling TECHNOLOGY, Danvers, Massachuset), anti-GSK-3β antibody (1:1000, cat.no. #9315, Cell Signaling TECHNOLOGY,), anti-β catenin antibody (1:1000, cat.no. 16051, Abcam), anti-β catenin phosphor Y142 antibody (1:1000, cat.no. 27798, Abcam), anti-Lamin B1 antibody (1:1000, cat.no.16048, Abcam). Anti-β-actin antibody (1:2000, cat.no.8227, Abcam). Blot density was determined by Alpha Imager software (Alpha Innotech, San Leandro, CA) [22].
Statistical analysis
All data were analyzed using SPSS software version 16.0 (SPSS, Inc.) Student’s t-test was used to compare the means of data from two experimental groups while significant differences (p < 0.05) amongst multiple group comparisons were confirmed by one-way ANOVA and Tukey’s post hoc multiple comparisons testing. Results are expressed as means ± SE [22].
Results
IATL inhibited pancreatic adenocarcinoma cell proliferation
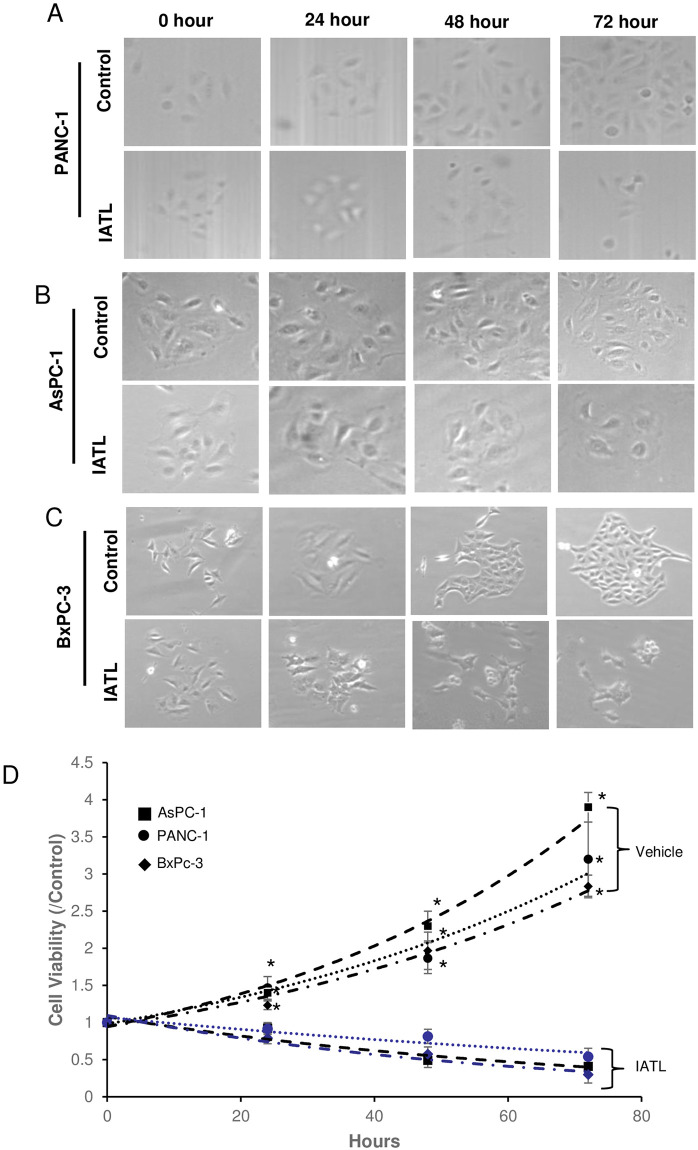
IATL has been reported to induce apoptosis of pancreatic carcinoma in both cellular and in vivo models. The underlying mechanism of IATL-mediated apoptosis has been reported as a ROS-induced toxic effect [14]. However, ROS-induced pancreatic carcinoma apoptosis did not account for these complicated processes. In this study, three different pancreatic cancer cell lines, PANC-1, AsPC-1 and BxPC-3 were cultured in 96 wells plates in the presence of 20μM IATL and proliferation was tested at various time. The colony and cell count showed that IATL significantly lowered both colonies (Fig 1A–1C, Table 1) and total live cell numbers compared to control (Table 2). The MTT assay showed that IATL time-dependently inhibited PANC-1s proliferation after 24 hours treatment (Fig 1D; P<0.05). IATL also showed a significant inhibitory effect on cell proliferation on both AsPC-1 and BxPC-3 after 24 hours treatment (Fig 1D; P<0.01).
Fig 1. IATL inhibits pancreatic cancer cell proliferation.
The PANC-1, AsPC-1 or BxPC-3 cells were treated with 20μM IATL at the indicated times. After the cells were digested with trypsin, the cellular viability was determined by MTT assay. (A) representative images of PANC-1 were treated with IATL; (B) The morphological characteristic changes in AsPC-1 when treated with IATL; (C) The morphological characteristic changes in BxPc-3 when treated with IATL; (D) MTT assay indicated that IATL significantly inhibited cell viability in each cell line (*P<0.05, n = 4).
Table 1. IATL inhibited pancreatic carcinoma cells colony development.
| Time (Hrs) | PANC-1 | AsPC-1 | BxPC-3 | |||
|---|---|---|---|---|---|---|
| Vehicle | IATL | Vehicle | IATL | Vehicle | IATL | |
| 0 | 14.7±0.57 | 15.0±1.0 | 15.7±1.15 | 15.0±1.0 | 15.3±1.52 | 16.0±1.0 |
| 24 | 17.6±0.53 | 16.7±0.58 | 19.7±1.58 | 16.3±1.15 | 19.7±2.52 | 17.7±1.52 |
| 48 | 24.0± 1.0 | 9.0±1.0* | 25.3±1.52 | 12.6±0.52* | 22.6±2.08 | 11.0±1.0* |
| 72 | 31.3±1.5 | 7.6±0.57* | 32.3±2.12 | 9.67±1.52* | 36.3±2.51 | 7.33±0.57* |
The pancreatic carcinoma cells were seeded into 12 wells plate. The colony number was counted under microscope. Each well was counted 5 independent visual fields. The number of colonies was represented as mean±SE.
Colony number/visual field (* P<0.05).
Table 2. IATL inhibited pancreatic carcinoma cells proliferation.
| Time (Hrs) | PANC-1 | AsPC-1 | BxPC-3 | |||
|---|---|---|---|---|---|---|
| Vehicle | IATL | Vehicle | IATL | Vehicle | IATL | |
| 0 | 1.0±0.10 | 1.0±0.1 | 1.0±0.15 | 1.0±0.10 | 1.0±0.1 | 1.0±0.1 |
| 24 | 1.30±0.10 | 1.41±0.1 | 1.6±0.1 | 1.43±0.15 | 1.4±0.10 | 1.1±0.26 |
| 48 | 2.96±0.25 | 0.8±0.1* | 3.26±0.21 | 0.56±0.11* | 3.26±0.21 | 0.56±0.06* |
| 72 | 4.30±0.26 | 0.6±0.1* | 5.13±0.21 | 0.43±0.05* | 3.80±0.1 | 0.38±0.05* |
The pancreatic carcinoma cells were seeded into 12 wells plate and were collected at indicated time. After the cells centrifuged, the cells were resuspended in 1ml PBS and were counted by TC 10 automated cell counter.
Trypan Blue staining negative cell (×105/mL, * P<0.05).
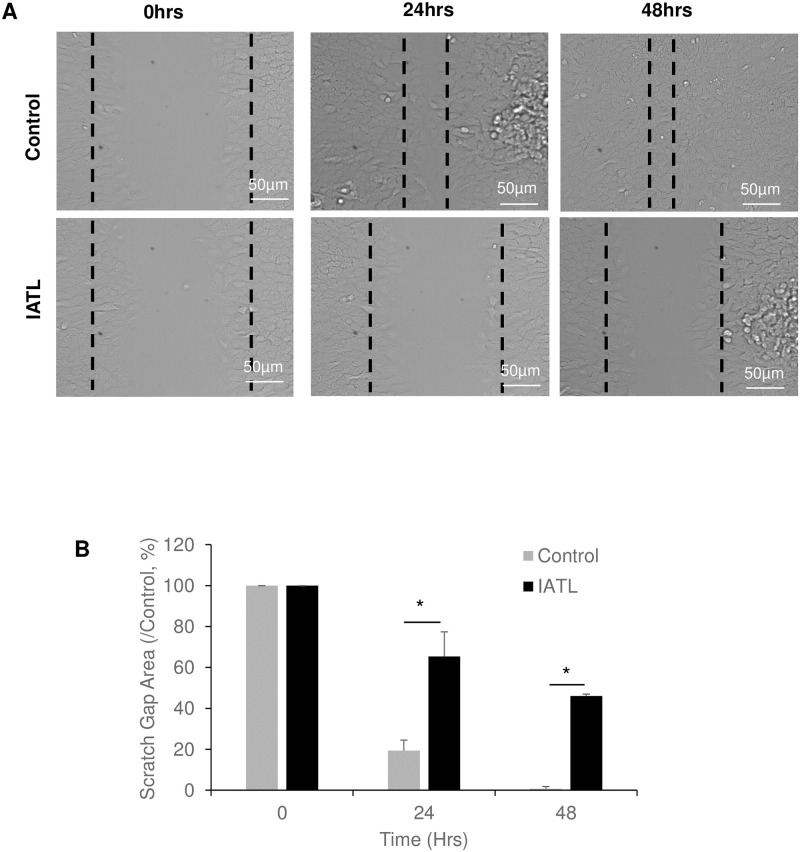
IATL inhibited pancreatic adenocarcinoma cell migration
The cell scratch closure assay showed that the scratch gap was significantly closed in PANC-1s control group after 24 hours incubation and completely closed at 48 hours (Fig 2A). However, IATL-treated PANC-1s significantly delayed the gap closures at both 24hours and 48 hours incubations (Fig 2A and 2B; P<0.05). These results indicated that IATL-mediated PANC-1 toxic effects may include inhibition of cellular differentiation and migration.
Fig 2. IATL inhibits pancreatic cancer cell migration.
The confluent PANC-1 cells were scratched followed by treatment with 20 μM IATL at the indicated times. (A) Representative scratch images of PANC-1 in the presence or absence of IATL; (B) IATL significantly inhibited scratch closure of PANC-1 as compared to vehicle treatment. The PANC cell closure areas were represented as percentage of 0 hours, respectively (P<0.05, n = 3).
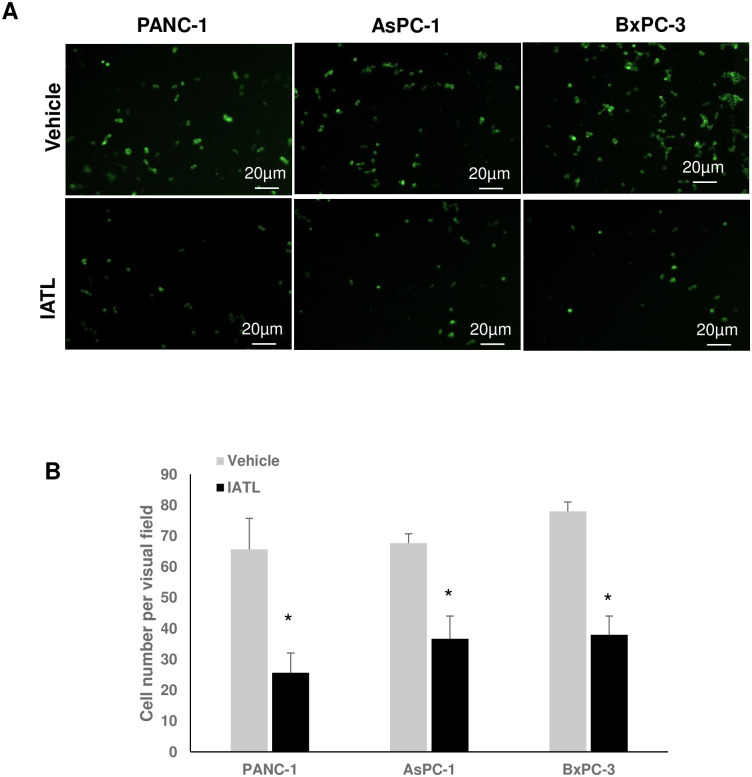
IATL inhibited pancreatic cancer cell invasion
PANC-1s, AsPC-1s or BxPC-3s (1×105) expressed GFP by transfecting pLV-GFP virus. Cells were then cultured on matrigel coated transwell insert. After incubation for 16 hours, the GFP positive cells on the bottom side of the transwell insert member were counted using fluorescent microscopy (Fig 3A). In the control groups, the average number of invaded PANC-1s, AsPC-1s or BxPC-3s under an individual field were 65.67±10.1, 66.67±3.1, 78.0±3.1, respectively. However, 20μM IATL treatment for 16 hours significantly inhibited pancreatic cancer cell invasion. The average invaded number of PANC-1s, AsPC-1s or BxPC-3s under individual field were 25.67±6.4, 36.67±7.37, 38.0±6.08, respectively (Fig 3B; P<0.01).
Fig 3. IATL inhibits pancreatic cancer cell invasion.
The GFP-expressing PANC-1, AsPC-1 or BxPC-3 cells were seeded into transwell inserts, which were previously coated with matrigel. The PANC-1, AsPC-1 or BxPC-3 cells (1×105) were treated with 20 μM IATL or vehicle for 24hours. The insert membranes were cut and mounted onto coverslides and the images were taken using fluorescent microscopy. (A) Representative cell images on the insert member after IATL or vehicle treatment for 24 hours; (B) Compared to vehicle, IATL treatment significantly inhibited invasion in all three different pancreatic cancer cell lines (P<0.05, n = 4).
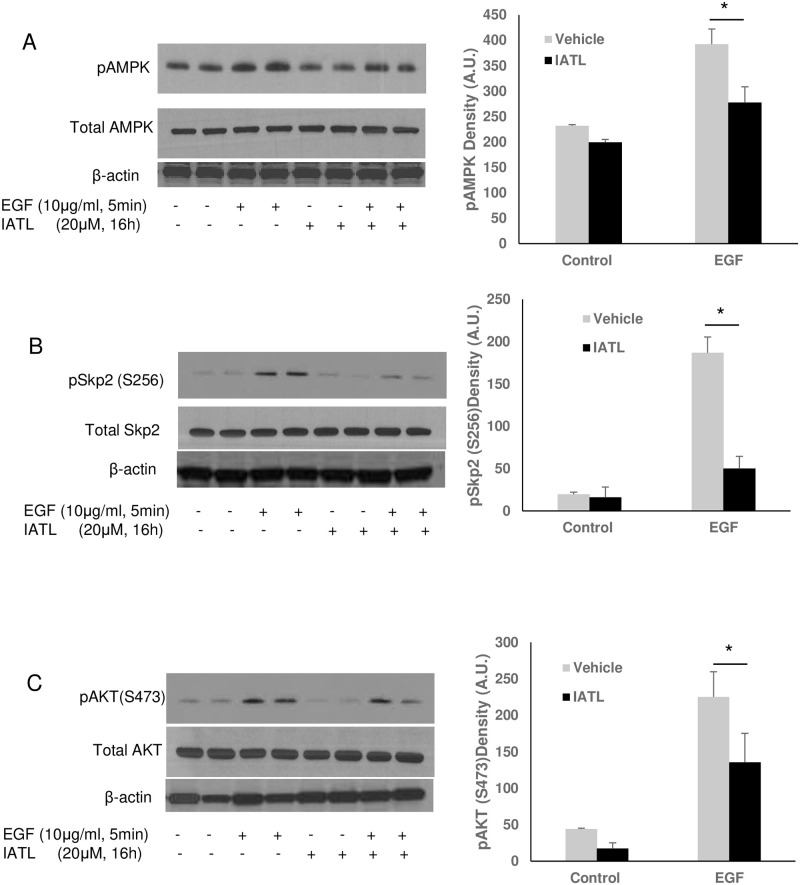
IATL inhibited AMPK-SKP2-Akt signal pathway in pancreatic cancer cells
Previous studies indicated that multiple signal pathways regulate pancreatic carcinoma cell apoptosis or proliferation, including PI3K-akt-mTOR signal pathway [23], AMPK-Akt signal pathway [24, 25] or Wnt signal pathway [26]. To investigate the detailed mechanism of IATL-mediated pancreatic carcinoma cell apoptosis, we first tested the effect of IATL on AMPK-Skp2-Akt signal pathway in pancreatic cancer cells. As shown in Fig 4A, EGF caused a significant phosphorylation of AMPK (Fig 4A), Skp2 (Fig 4B)- and Akt (Fig 4C) in PNAC-1s. However, EGF-mediated activation of AMPK, Skp2, and Akt were significantly inhibited by IATL (Fig 4).
Fig 4. IATL inhibits EGF signal pathway through Skp2 inactivation in pancreatic cancer cells.
The PANC-1 cells were treated with 20μM IATL followed by EGF (10μg/mL) treatment for 5minutes. After the cell lysates were harvested, the target protein phosphorylation was determined by western blotting. EGF caused AMPK (A), Skp2 (B) and Akt (C) phosphorylation increases; but IATL significantly inhibited EGF-mediated EGF (A)-SKp2 (B) and Akt (C) phosphorylation (P<0.05, n = 3).
IATL inhibited canonical Wnt signal pathway in PANC-1
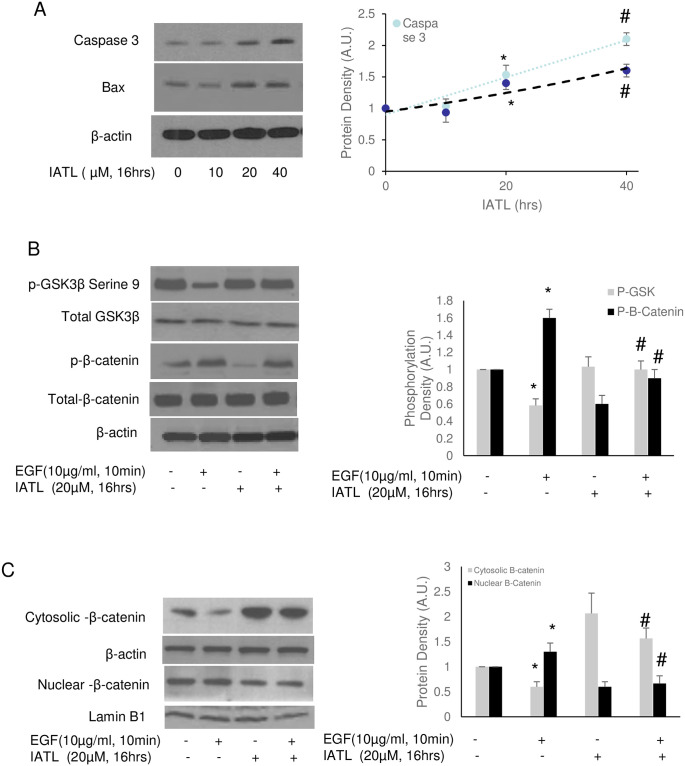
By regulation of β-catenin, the Wnt signal pathway regulates the development of various cancers, such as breast, colorectal, melanoma, prostate cancer and pancreatic cancer [27]. The regulatory mechanisms of Wnt include cell proliferation, migration, and cell specification [28]. Therefore, we then investigated whether Wnt signal pathways regulated IATL-mediated pancreatic carcinoma cell apoptosis. Western blots showed that IATL induced release of capase-3 and Bax dose-dependently in PANC-1(Fig 5A). 20μM IATL was enough to induce PANC-1 apoptosis according to caspase-3 and Bax release. Serine 9 phosphorylation of GSK-3β, an inhibitory index of GSK was decreased by EGF but was reversed by IATL. In addition, IATL caused decrease in both basal and EGF-mediated β-catenin phosphorylation (Fig 5B). Interestingly, IATL decreased β-catenin accumulation in cellular nucleus (Fig 5C).
Fig 5. IATL inhibits Wnt signal pathway and induces pancreatic cancer cells apoptosis.
The PANC-1 cells were treated with 20μM IATL alone or in combination with EGF (5μg/mL) at the indicated time. The cell lysates were harvested for western blotting. (A) IATL induced both Caspase 3 and Bax in PANC-1 cells in a dose-dependent fashion; (B) EGF activated the Wnt signal pathway by decreasing serine 9 phosphorylation of GSK-3β (*P<0.05, n = 3), however, IATL increased GSK-3β phosphorylation (#P<0.05, n = 3); EGF increased β-catenin phosphorylation (*P<0.05) but IATL reversed this regulation (#P<0.05, n = 3); (C) EGF caused β-catenin translocation into cellular nucleus (*P<0.05, n = 3) but IATL inhibited EGF-mediated β-catenin translocation (#P<0.05, n = 3).
IATL inhibited proliferation of pancreatic cancer cells xenograft in vivo
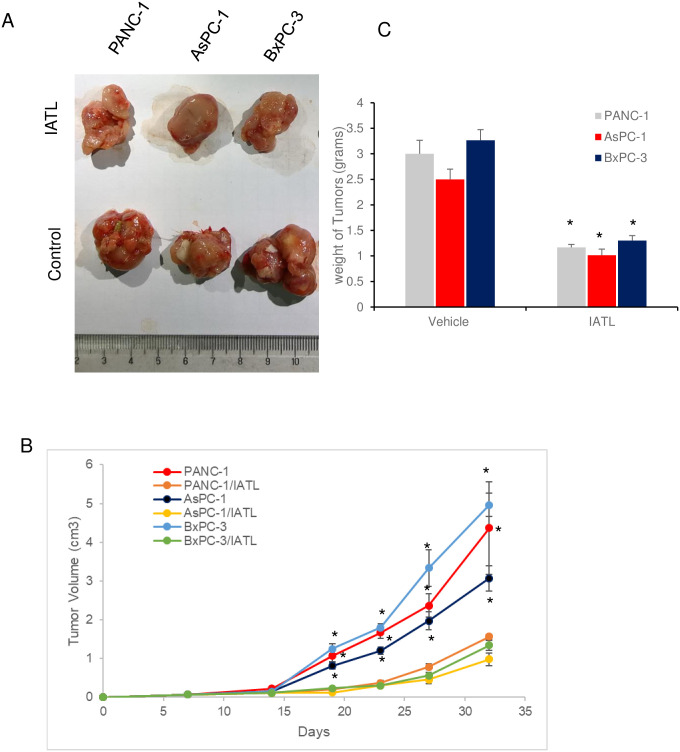
The three different pancreatic cancer cell line, PANC-1s, AsPC-1s or BxPC-3s (1×106) were injected into left frontier axilla subcutaneously for 5 weeks and the tumor growth was monitored at indicated time (Fig 6B). Once the cells were injected into mice, the mice were treated with IATL (0.5mg/kg, once/week) or vehicle for 5 weeks. Compared to the vehicle treatment group, IATL treatment significantly reduced the tumor sizes of PANC-1s xenografts (4.3±1.2 vs 1.3±0.07 cm3; vehicle vs ITAL, Fig 6A and 6B, P<0.05). Similar toxic effects were also observed with AsPC-1s (3.07±0.32vs 0.97±0.17 cm3; vehicle vs ITAL, Fig 6A and 6B, P<0.05) and BxPC-3s xenografts treated with IATL (4.96±0.30 vs 1.34±0.127 cm3; vehicle vs ITAL, Fig 6A and 6B, P<0.05).
Fig 6. IATL inhibits pancreatic cancer cells growth in vivo.
The PANC-1, AsPC-1 or BxPC-3 cells were subcutaneously xenografted onto BALB/c nude mice flank. Once the cells were injected, the mice were treated with IATL (0.5mg/kg) or vehicle once a week for 5 weeks. At the end of experiment, the xenografts were removed for assessing weight or for imaging. (A) Images of representative tumors from PANC-1, AsPC-1 or BxPC-3 cell xenografts (n = 6). (B) Growth curve of tumors xenografts in the presence or absence of IATL over 5 weeks; (C) IATL significantly decreased the weight of tumors generated from PANC-1, AsPC-1 or BxPC-3 cells in vivo (*, P<0.05).
Consistently, IATL also significantly decreased the tumor weight of PANC-1s xenografts (3.0±0.26 vs 1.16±0.06 g; vehicle vs ITAL, Fig 6C, P<0.05). Similar toxic effects were also observed with AsPC-1s (2.5±0.2 vs 1.01±0.12 g; vehicle vs ITAL, Fig 6C, P<0.05) and BxPC-3s xenografts treated with IATL (3.26±0.21 vs 1.29 ±0.10 g; vehicle vs ITAL, Fig 6C, P<0.05).
IATL induced apoptosis of PANC-1 xenograft in vivo
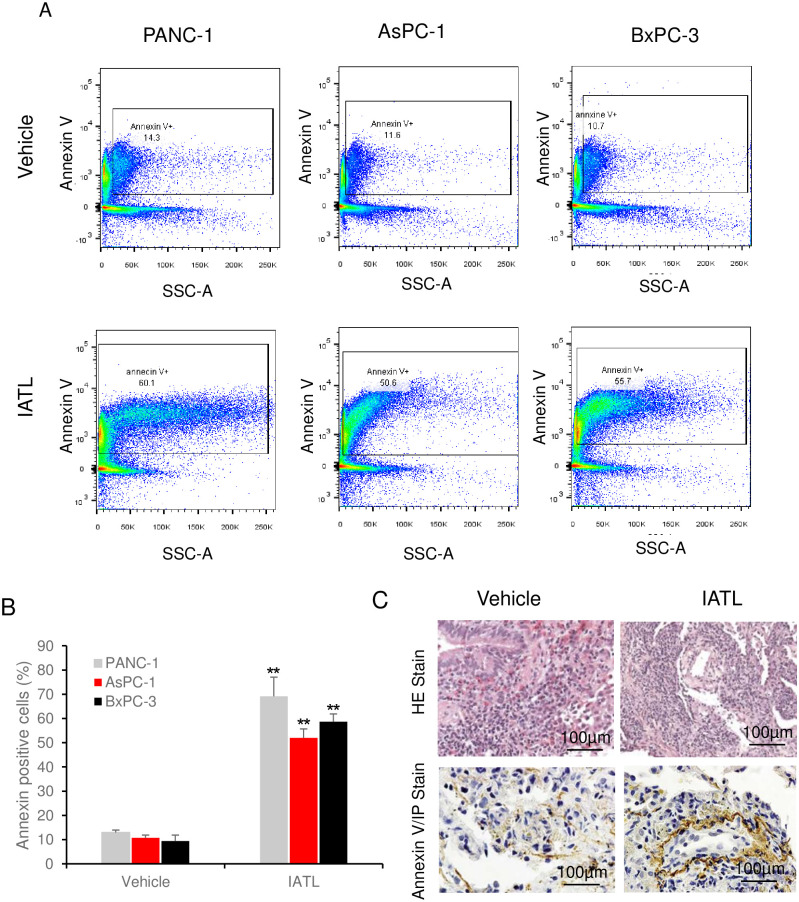
To further evaluate the effect of IATL on pancreatic cancer in vivo, flow cytometry was performed on the cells isolated from xenograft. Staining cells isolated from PANC-1 xenograft with Annexin V-Propidium iodide (PI) showed that IATL treatment caused a significantly higher annexin-V positive cell ratio comparing to vehicle groups (Fig 7A). The annexin-V positive cell ratio in PANC-1 xenograft were 13.0 ±1.1 vs 69.13±7.94%; vehicle vs ITAL (Fig 7B, P<0.05). A similar toxic effect of IATL against AsPC-1s (10.78±1.15 vs 52.3±3.7%; vehicle vs ITAL, Fig 7B, P<0.05) and BxPC-3s (9.4±2.49 vs 58.67±3.22%; vehicle vs ITAL, Fig 7B, P<0.05) in respective xenografts was also observed.
Fig 7. IATL induces pancreatic cancer cells apoptosis in vivo.
The PANC-1, AsPC-1 or BxPC-3 cells were xenografted on BALB/c nude mice and were treated with IATL. In the end of experiment, the xenografts on nude mice were removed for flowcytometry or histology assay. (A) Representative images of flow cytometry assay showing detection of annexin-V positive cells from PANC-1, AsPC-1 or BxPC-3 xenografts for both vehicle and IATL treatment groups; (B) Compared to vehicle, IATL treatment significantly increased annexin-V positive cells in PANC-1, AsPC-1 or BxPC-3 cell xenografts (*, P<0.05; **, <0.01). (C) Representative images of H&E staining (upper panel) and annexin V staining (bottom panel) of PANC-1s xenografts (n = 6).
Moreover, H&E stain and immunohistological assays were performed after PANC-1 xenografts were isolated from mice. The samples from both vehicles and IATL treatment groups showed typical carcinoma characteristics, such as lower level organized tissue, poor blood vessel development and dark stain of cellular nucleus (Fig 7C upper panel). However, annexin-V stain showed that the annexin-V positive cell number in IATL groups was significantly higher than those in vehicle groups (P<0.05, Fig 7C, bottom panel).
Discussion
Discussion
IATL demonstrated significant inhibitory effects on pancreatic carcinoma cell proliferation, migration and invasion. Xenograft studies also showed that IATL significantly inhibited pancreatic carcinoma cell transplants growth in vivo. Signal pathway studies demonstrated that IATL induces pancreatic carcinoma cell apoptosis by inhibition of the non-canonical wnt signal pathway. IATL inhibits pancreatic carcinoma cells proliferation by inhibition of AMPK-Skp2-Akt signal pathway. S-phase kinase associated protein 2 (Skp2) is a member of the F-box family, which regulates the cell cycle and is highly expressed in various carcinomas, such as pancreatic [29], breast [24] and kidney carcinoma [30]. Skp2 recruits Skp1, cullin-1 and Rbx1E3 ligase to form a SCFSkp2 complex, which targets cell cycle elements, such as p27, p21 and further regulates cell cycle entry and G1/S transition [31–33]. Silence of Skp2 or inhibition Skp2 by pharmaceutic inhibitors significantly improves breast cancer cell sensitivity to the anti-breast cancer drug, Gefitinib. Further studies indicate that Skp2 plays a central role in EGF-mediated signal activation, such as phosphorylation and ubiquitination of Akt. Akt ubiquitination further causes tumorigenesis and drug resistance [24]. In the current study, IATL significantly inhibits EGF-mediated Skp2 serine phosphorylation. Moreover, AMPK-Skp2-Akt signal pathway appears to be involved in IATL-mediated effects as both AMPK and Akt were dephosphorylated as Skp2 was simultaneously dephosphorylated. These results are consistent with previous observations in different models [24, 34].
The Wnt signal pathway is another signal pathway regulating many cell fates, including apoptosis, cell death and inflammatory responses. The Wnt signal pathway includes non-canonical and canonical Wnt signal pathways [35]. Cadmium inhibited the canonical wnt signal pathway, which in turn induces non-small cell lung carcinoma cell apoptosis [26]. Early gene profiling studies indicate that Wnt5a, a marker of the non-canonical wnt molecule is up-regulated in pancreatic ductal adenocarcinoma cells [36]. DKK1 is also involved in pancreatic ductal carcinoma and is a potential biomarker [37]. Wnt inhibitors significantly reduce human pancreatic carcinoma cell viability in cellular and in vivo model [38]. Frizzled family receptor 8 inhibitor, OMP-54F28 inhibits advanced solid tumor cells, including pancreatic carcinoma cell proliferation [39]. In the current study, we showed that IATL inhibited the canonical wnt signal pathway in pancreatic carcinoma cells and induced apoptosis.
PRI-724 is the specific Wnt pathway inhibitor, which binds to the coactvator CBP, inhibiting its interaction with β-catenin [40]. Here we showed that IATL deceased β-catenin phosphorylation and β-catenin accumulation in cellular nucleus. The detail of molecular interaction need further investigation.
As a most commonly diagnosed cancer worldwide, anti-pancreatic carcinoma medicines and their mechanisms have been intensively investigated. By inhibiting DNA damage repairing mechanism, poly (ADP-ribose) polymerase inhibitors caused accumulation of double stranded DNA breaks and induced pancreatic ductal adenocarcinoma cell death [41]. By inhibiting some of the specific signal pathways, such as the Src signal pathway or the KRas signal pathway, these medicines also appear to work efficiently on pancreatic carcinoma [42, 43]. The manipulation of ROS as in the drug-mediated apoptosis in pancreatic carcinoma has also been widely investigated [44]. However, ROS-induced apoptosis in pancreatic carcinoma was mediated by specific target pathway or down-stream target transfactors or proteins [45, 46]. Previous reports have also shown that ROS functions as a mediator in IATL-induced toxic effects of pancreatic carcinoma [14, 47]. Meanwhile, the studies also showed that specific signal pathways, such as STAT3, MAPK may also play critical roles in the process of apoptosis [14, 47]. However, the regulatory relationship between ROS and these signal pathways remain unclear. To our knowledge, the current study characterized for the first time the involvement of the EGF- AMPK-Skp2-Akt signal pathway and canonical Wnt signal pathway in IATL-mediated apoptosis of pancreatic carcinoma. Considering the importance of EGF- AMPK-Skp2-Akt signal pathway in tumorigenesis, metastasis and invasion, IATL may provide a promising medicine for pancreatic cancer. Moreover, Skp2 is a central signal protein related to both EGF and drug resistance [29, 48]. The inhibitory effect of IATL on both EGF and SKP2 may offer a novel option for pancreatic cancer.
Conclusion
In summary, our present studies indicate that IATL inhibits pancreatic carcinoma by inhibition of the EGF- AMPK-Skp2-Akt signal pathway. IATL induces pancreatic carcinoma apoptosis by inhibition of the canonical wnt signal pathway. Moreover, IATL shows a significant inhibitory effect of pancreatic carcinoma in an in vivo xenograft model. These studies of IATL on pancreatic cancer may offer opportunities for both pharmaceutic and clinical purposes in the future.
Supporting information
(PDF)
Acknowledgments
We thank Dr. Ibra Fancher for assistance in writing and discussion.
Data Availability
All relevant data are within the manuscript and its Supporting information files.
Funding Statement
The authors received no specific funding for this work.
References
- 1.Collisson EA, Bailey P, Chang DK, Biankin AV. Molecular subtypes of pancreatic cancer. Nat Rev Gastroenterol Hepatol. 2019;16(4):207–20. 10.1038/s41575-019-0109-y [DOI] [PubMed] [Google Scholar]
- 2.Kota J, Hancock J, Kwon J, Korc M. Pancreatic cancer: Stroma and its current and emerging targeted therapies. Cancer Lett. 2017;391:38–49. 10.1016/j.canlet.2016.12.035 [DOI] [PubMed] [Google Scholar]
- 3.Rawla P, Thandra KC, Sunkara T. Pancreatic cancer and obesity: epidemiology, mechanism, and preventive strategies. Clin J Gastroenterol. 2019. [DOI] [PubMed] [Google Scholar]
- 4.Garcia-Reyes B, Kretz AL, Ruff JP, von Karstedt S, Hillenbrand A, Knippschild U, et al. The Emerging Role of Cyclin-Dependent Kinases (CDKs) in Pancreatic Ductal Adenocarcinoma. Int J Mol Sci. 2018;19(10). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Song M, Bode AM, Dong Z, Lee MH. AKT as a Therapeutic Target for Cancer. Cancer Res. 2019;79(6):1019–31. 10.1158/0008-5472.CAN-18-2738 [DOI] [PubMed] [Google Scholar]
- 6.Cheng X, Xu X, Chen D, Zhao F, Wang W. Therapeutic potential of targeting the Wnt/beta-catenin signaling pathway in colorectal cancer. Biomed Pharmacother. 2019;110:473–81. 10.1016/j.biopha.2018.11.082 [DOI] [PubMed] [Google Scholar]
- 7.Baldini N, Avnet S. The Effects of Systemic and Local Acidosis on Insulin Resistance and Signaling. Int J Mol Sci. 2018;20(1). 10.3390/ijms20010126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li L, Bi Z, Wadgaonkar P, Lu Y, Zhang Q, Fu Y, et al. Metabolic and epigenetic reprogramming in the arsenic-induced cancer stem cells. Semin Cancer Biol. 2019. 10.1016/j.semcancer.2019.04.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Juiz NA, Iovanna J, Dusetti N. Pancreatic Cancer Heterogeneity Can Be Explained Beyond the Genome. Front Oncol. 2019;9:246. 10.3389/fonc.2019.00246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Seufferlein T, Bachet JB, Van Cutsem E, Rougier P, Group EGW. Pancreatic adenocarcinoma: ESMO-ESDO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23 Suppl 7:vii33–40. 10.1093/annonc/mds224 [DOI] [PubMed] [Google Scholar]
- 11.Borazanci E, Von Hoff DD. Nab-paclitaxel and gemcitabine for the treatment of patients with metastatic pancreatic cancer. Expert Rev Gastroenterol Hepatol. 2014;8(7):739–47. 10.1586/17474124.2014.925799 [DOI] [PubMed] [Google Scholar]
- 12.Wolfgang CL, Herman JM, Laheru DA, Klein AP, Erdek MA, Fishman EK, et al. Recent progress in pancreatic cancer. CA Cancer J Clin. 2013;63(5):318–48. 10.3322/caac.21190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pal HC, Sehar I, Bhushan S, Gupta BD, Saxena AK. Activation of caspases and poly (ADP-ribose) polymerase cleavage to induce apoptosis in leukemia HL-60 cells by Inula racemosa. Toxicol In Vitro. 2010;24(6):1599–609. 10.1016/j.tiv.2010.06.007 [DOI] [PubMed] [Google Scholar]
- 14.Khan M, Ding C, Rasul A, Yi F, Li T, Gao H, et al. Isoalantolactone induces reactive oxygen species mediated apoptosis in pancreatic carcinoma PANC-1 cells. Int J Biol Sci. 2012;8(4):533–47. 10.7150/ijbs.3753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yan YY, Zhang Q, Zhang B, Yang B, Lin NM. Active ingredients of Inula helenium L. exhibits similar anti-cancer effects as isoalantolactone in pancreatic cancer cells. Nat Prod Res. 2019:1–6. 10.1080/14786419.2018.1543676 [DOI] [PubMed] [Google Scholar]
- 16.Zhang X, Wu D, Aldarouish M, Yin X, Li C, Wang C. ETS-1: A potential target of glycolysis for metabolic therapy by regulating glucose metabolism in pancreatic cancer. Int J Oncol. 2017;50(1):232–40. 10.3892/ijo.2016.3770 [DOI] [PubMed] [Google Scholar]
- 17.Ren R, Yu J, Zhang Y, Wang SF, Guo X, Shen M, et al. Inflammation Promotes Progression of Pancreatic Cancer Through WNT/beta-Catenin Pathway-Dependent Manner. Pancreas. 2019;48(8):1003–14. 10.1097/MPA.0000000000001386 [DOI] [PubMed] [Google Scholar]
- 18.Rybarczyk P, Vanlaeys A, Brassart B, Dhennin-Duthille I, Chatelain D, Sevestre H, et al. The Transient Receptor Potential Melastatin 7 Channel Regulates Pancreatic Cancer Cell Invasion through the Hsp90alpha/uPA/MMP2 pathway. Neoplasia. 2017;19(4):288–300. 10.1016/j.neo.2017.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dosch AR, Dai X, Gaidarski AA Iii, Shi C, Castellanos JA, VanSaun MN, et al. Src kinase inhibition restores E-cadherin expression in dasatinib-sensitive pancreatic cancer cells. Oncotarget. 2019;10(10):1056–69. 10.18632/oncotarget.26621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Luan YP, Li QF, Wu SG, Mao DC, Deng YY, Chen RW. Tsoong induces apoptosis and inhibits proliferation, migration and invasion of pancreatic ductal adenocarcinoma cells. Mol Med Rep. 2018;17(3):3527–36. 10.3892/mmr.2017.8328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hu W, Liu Q, Pan J, Sui Z. MiR-373-3p enhances the chemosensitivity of gemcitabine through cell cycle pathway by targeting CCND2 in pancreatic carcinoma cells. Biomed Pharmacother. 2018;105:887–98. 10.1016/j.biopha.2018.05.091 [DOI] [PubMed] [Google Scholar]
- 22.Lederer PA, Zhou T, Chen W, Epshtein Y, Wang H, Mathew B, et al. Attenuation of murine acute lung injury by PF-573,228, an inhibitor of focal adhesion kinase. Vascul Pharmacol. 2018;110:16–23. 10.1016/j.vph.2018.06.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Totiger TM, Srinivasan S, Jala VR, Lamichhane P, Dosch AR, Gaidarski AA 3rd, et al. Urolithin A, a Novel Natural Compound to Target PI3K/AKT/mTOR Pathway in Pancreatic Cancer. Mol Cancer Ther. 2019;18(2):301–11. 10.1158/1535-7163.MCT-18-0464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Han F, Li CF, Cai Z, Zhang X, Jin G, Zhang WN, et al. The critical role of AMPK in driving Akt activation under stress, tumorigenesis and drug resistance. Nat Commun. 2018;9(1):4728. 10.1038/s41467-018-07188-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yang Y, Lu Y, Han F, Chang Y, Li X, Han Z, et al. Saxagliptin regulates M1/M2 macrophage polarization via CaMKKbeta/AMPK pathway to attenuate NAFLD. Biochem Biophys Res Commun. 2018;503(3):1618–24. 10.1016/j.bbrc.2018.07.090 [DOI] [PubMed] [Google Scholar]
- 26.Park CH, Lee BH, Ahn SG, Yoon JH, Oh SH. Serine 9 and tyrosine 216 phosphorylation of GSK-3beta differentially regulates autophagy in acquired cadmium resistance. Toxicol Sci. 2013;135(2):380–9. 10.1093/toxsci/kft158 [DOI] [PubMed] [Google Scholar]
- 27.Butti R, Gunasekaran VP, Kumar TVS, Banerjee P, Kundu GC. Breast cancer stem cells: Biology and therapeutic implications. Int J Biochem Cell Biol. 2019;107:38–52. 10.1016/j.biocel.2018.12.001 [DOI] [PubMed] [Google Scholar]
- 28.Mirabelli CK, Nusse R, Tuveson DA, Williams BO. Perspectives on the role of Wnt biology in cancer. Sci Signal. 2019;12(589). 10.1126/scisignal.aay4494 [DOI] [PubMed] [Google Scholar]
- 29.Gao J, Wang G, Wu J, Zuo Y, Zhang J, Chen J. Arsenic trioxide inhibits Skp2 expression to increase chemosensitivity to gemcitabine in pancreatic cancer cells. Am J Transl Res. 2019;11(2):991–7. [PMC free article] [PubMed] [Google Scholar]
- 30.Lu H, Cao X, Zhang H, Sun G, Fan G, Chen L, et al. Imbalance between MMP-2, 9 and TIMP-1 promote the invasion and metastasis of renal cell carcinoma via SKP2 signaling pathways. Tumour Biol. 2014;35(10):9807–13. 10.1007/s13277-014-2256-7 [DOI] [PubMed] [Google Scholar]
- 31.Jung D, Khurana A, Roy D, Kalogera E, Bakkum-Gamez J, Chien J, et al. Quinacrine upregulates p21/p27 independent of p53 through autophagy-mediated downregulation of p62-Skp2 axis in ovarian cancer. Sci Rep. 2018;8(1):2487. 10.1038/s41598-018-20531-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shen L, Qu X, Li H, Xu C, Wei M, Wang Q, et al. NDRG2 facilitates colorectal cancer differentiation through the regulation of Skp2-p21/p27 axis. Oncogene. 2018;37(13):1759–74. 10.1038/s41388-017-0118-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lanctot AA, Guo Y, Le Y, Edens BM, Nowakowski RS, Feng Y. Loss of Brap Results in Premature G1/S Phase Transition and Impeded Neural Progenitor Differentiation. Cell Rep. 2017;20(5):1148–60. 10.1016/j.celrep.2017.07.018 [DOI] [PubMed] [Google Scholar]
- 34.Frau M, Feo F, Pascale RM. Pleiotropic effects of methionine adenosyltransferases deregulation as determinants of liver cancer progression and prognosis. J Hepatol. 2013;59(4):830–41. 10.1016/j.jhep.2013.04.031 [DOI] [PubMed] [Google Scholar]
- 35.Pashirzad M, Shafiee M, Khazaei M, Fiuji H, Ryzhikov M, Soleimanpour S, et al. Therapeutic potency of Wnt signaling antagonists in the pathogenesis of prostate cancer, current status and perspectives. J Cell Physiol. 2019;234(2):1237–47. 10.1002/jcp.27137 [DOI] [PubMed] [Google Scholar]
- 36.Pilarsky C, Ammerpohl O, Sipos B, Dahl E, Hartmann A, Wellmann A, et al. Activation of Wnt signalling in stroma from pancreatic cancer identified by gene expression profiling. J Cell Mol Med. 2008;12(6B):2823–35. 10.1111/j.1582-4934.2008.00289.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Igbinigie E, Guo F, Jiang SW, Kelley C, Li J. Dkk1 involvement and its potential as a biomarker in pancreatic ductal adenocarcinoma. Clin Chim Acta. 2019;488:226–34. 10.1016/j.cca.2018.11.023 [DOI] [PubMed] [Google Scholar]
- 38.Wall I, Schmidt-Wolf IG. Effect of Wnt inhibitors in pancreatic cancer. Anticancer Res. 2014;34(10):5375–80. [PubMed] [Google Scholar]
- 39.Le PN, McDermott JD, Jimeno A. Targeting the Wnt pathway in human cancers: therapeutic targeting with a focus on OMP-54F28. Pharmacol Ther. 2015;146:1–11. 10.1016/j.pharmthera.2014.08.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sebio A, Kahn M, Lenz H-J. The potential of targeting Wnt/β-catenin in colon cancer. Taylor & Francis; 2014. [DOI] [PubMed] [Google Scholar]
- 41.Gupta M, Iyer R, Fountzilas C. Poly(ADP-Ribose) Polymerase Inhibitors in Pancreatic Cancer: A New Treatment Paradigms and Future Implications. Cancers (Basel). 2019;11(12). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Parkin A, Man J, Timpson P, Pajic M. Targeting the complexity of Src signalling in the tumour microenvironment of pancreatic cancer: from mechanism to therapy. FEBS J. 2019;286(18):3510–39. 10.1111/febs.15011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhu Z, Xiao S, Hao H, Hou Q, Fu X. Kirsten Rat Sarcoma Viral Oncogene Homologue (KRAS) Mutations in the Occurrence and Treatment of Pancreatic Cancer. Curr Top Med Chem. 2019;19(23):2176–86. 10.2174/1568026619666190828160804 [DOI] [PubMed] [Google Scholar]
- 44.Buchholz M, Majchrzak-Stiller B, Hahn S, Vangala D, Pfirrmann RW, Uhl W, et al. Innovative substance 2250 as a highly promising anti-neoplastic agent in malignant pancreatic carcinoma—in vitro and in vivo. BMC Cancer. 2017;17(1):216. 10.1186/s12885-017-3204-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rabi T, Catapano CV. Aphanin, a triterpenoid from Amoora rohituka inhibits K-Ras mutant activity and STAT3 in pancreatic carcinoma cells. Tumour Biol. 2016;37(9):12455–64. 10.1007/s13277-016-5102-2 [DOI] [PubMed] [Google Scholar]
- 46.Moore Z, Chakrabarti G, Luo X, Ali A, Hu Z, Fattah FJ, et al. NAMPT inhibition sensitizes pancreatic adenocarcinoma cells to tumor-selective, PAR-independent metabolic catastrophe and cell death induced by beta-lapachone. Cell Death Dis. 2015;6:e1599. 10.1038/cddis.2014.564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chen W, Li P, Liu Y, Yang Y, Ye X, Zhang F, et al. Isoalantolactone induces apoptosis through ROS-mediated ER stress and inhibition of STAT3 in prostate cancer cells. J Exp Clin Cancer Res. 2018;37(1):309. 10.1186/s13046-018-0987-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Schuler S, Diersch S, Hamacher R, Schmid RM, Saur D, Schneider G. SKP2 confers resistance of pancreatic cancer cells towards TRAIL-induced apoptosis. Int J Oncol. 2011;38(1):219–25. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PDF)
Data Availability Statement
All relevant data are within the manuscript and its Supporting information files.