Abstract
Objective:
The study was designed to assess the impact of socioeconomic barriers on the rate of prenatal diagnosis of critical congenital heart disease (CCHD).
Methods:
This was a retrospective review of the Medicaid analytic extract (MAX) dataset, a national Medicaid administrative claims database with linked maternal-infant claims, from 2007 to 2012. Infants with CCHD were identified by searching for International Classification of Diseases (ICD) 9 codes and Procedural Coding System (PCS) codes for CCHD within the first 6 months after the delivery date. Multivariate logistic regression was used to evaluate the effect of maternal and socioeconomic factors on the prenatal diagnosis rate.
Results:
There were 4702 mother-infant dyads included in the analysis. The prenatal diagnosis rate of CCHD was 27.9%. Factors independently associated with odds of prenatal diagnosis of CCHD were presence of maternal diabetes (OR, 2.055; P < .001), ZIP code level median household income (OR, 1.005; P = .015), sonographer labor quotient (OR, 1.804; P = .047), the year of the delivery (OR, 1.155; P < .001), and needing a view other than a 4 chamber or outflow tract view to obtain the diagnosis (OR, 0.383; P < .001).
Conclusion:
Maternal health, diabetes, socioeconomic factors, and access to sonographers impacts prenatal diagnosis of CCHD.
1 |. INTRODUCTION
Prenatal diagnosis of critical congenital heart disease (CCHD) enables safe delivery planning, with access to appropriate cardiac therapies shortly after birth. Additionally, it is associated with improved postnatal stability with lower preoperative acidosis and in certain defects lower preoperative mortality.1,2 Prenatal diagnosis of CCHD is made by fetal ultrasound, and for many pregnancies, the optimal chance to identify CCHD is during the mid-gestation ultrasound, commonly obtained between 18 to 22 weeks gestation. Certain types of CCHD, such as hypoplastic left heart syndrome, are readily identifiable on a 4 chamber view of the heart, and therefore have high rates of prenatal detection. Other types of CCHD, such as transposition of the great arteries or tetralogy of Fallot, require more complex imaging planes that include the outflow tracts to diagnose and therefore are more difficult to identify prenatally.3
There is wide variation in the prenatal diagnosis rate of CCHD across regions, although the underlying reasons for this variation are not well understood.3 Despite multiple single center studies investigating this dilemma, there is no consensus on the reasons for the well described variation in rates of prenatal diagnosis.4–7 One study found that insurance type and socioeconomic status were the strongest predictor of prenatal diagnosis, while another study found that insurance type did not play a role, but that lower socioeconomic status and rural patients had lower prenatal diagnosis rates.4,5 Given this lack of consensus, our primary goal was to determine the impact of socioeconomic factors on prenatal diagnosis of CCHD using data from multiple states and hospital systems. We hypothesized that fetuses born in lower income, more rural areas, with lower proportions of obstetricians and sonographers, will have lower rates of prenatal diagnosis of CCHD. Additionally, we hypothesized that being born in an earlier year, and having a fetus with a type of CCHD that is not readily diagnosed by a standard 4 chamber view will also be associated with a missed prenatal diagnosis. Because of the recommendation for mothers with diabetes to obtain a fetal echocardiogram, we hypothesized that mothers of fetuses with diabetes will have higher prenatal diagnosis rate compared to low risk pregnancies.
2 |. METHODS
2.1 |. Data sources
The study used a national Medicaid administrative claims database, specifically the Medicaid analytic extract (MAX dataset), with linked maternal-infant claims. Medicaid is a form of public health insurance in the United States that is available to individuals based on their income or medical need. Medicaid is a major source of healthcare financing for pregnant women and deliveries.8 Nationally, nearly half of all births are financed by Medicaid, making this a particularly robust database for pregnancy related outcomes.8 This database has been previously utilized to study risks of maternal medications on infant outcomes.9 Using ICD-9 codes and PCS codes to identify congenital cardiac anomalies within the MAX database has previously been validated, and has a positive predictive value between 75 to 85%.10 The database was used with information from January 1, 2007 to December 31, 2012. The data include inpatient and outpatient claims, pharmacy claims, and demographic information, such as maternal zip code, race, and maternal health conditions. The MAX dataset was reviewed by the IRB at the Children’s Hospital of Philadelphia (IRB 15–012247) and determined to be exempt from IRB oversight as it did not meet criteria as human subjects research.
2.2 |. Inclusion/exclusion criteria
States with greater than 80% maternal-infant linkage of the Centers for Medicare and Medicaid Services (CMS) database were included in the study (N = 15). Infants with CCHD were identified in the database by searching both maternal and infant claims for ICD-9 codes (745–747) and PCS codes (35–39) for congenital heart disease within the first 6 months after the delivery date. Infants whose only ICD-9 code was for a secundum atrial septal defect (745.5) or patent ductus arteriosus (747.0), or whose only PCS code was for patent ductus arteriosus closure (38.85) or atrial septal defect repair (35.61, 35.71) were excluded, as these structures are normal components of the fetal circulation. Twin pregnancies were excluded. Each Medicaid enrollee is given a unique identifier (ID), which is either the enrollee’s social security number or a state assigned ID. Using the infant’s ID, the maternal ID was then identified using the linkage. Mothers who did not have any visits in the 9 months preceding the delivery were excluded, as it was unclear if these mothers had only enrolled in Medicaid at delivery. Maternal codes were included from 9 months before delivery to half a month prior to delivery, and were searched for Current Procedural Terminology (CPT) codes for fetal echocardiograms (76,825–76,828, 93,325), ultrasounds after 14 weeks gestation (76,805, 76,810–76,812), and ICD-9 codes for congenital heart disease (745–747) or fetal anomaly affecting the pregnancy (655.8, 655.9). If a mother’s claims in the 9 months preceding delivery included a code for a fetal echocardiogram and an ICD-9 code for either fetal congenital heart lesion, or a fetal anomaly affecting the pregnancy, the infant was considered to have been prenatally diagnosed.
2.3 |. Socioeconomic data
The patient-level data were linked to ZIP code level data from the American Community Survey, which includes median household income and percent of the population in poverty at the ZIP code level. Median household income is recorded in inflation adjusted 2012 dollars. The economic research service provides a ZIP code level urban– rural continuum score. Maternal race was included with the Medicaid claims dataset. The Bureau of Labor Statistics publishes location quotients for diagnostic medical sonographers and obstetricians. A location quotient is the ratio of the area concentration of the occupational employment to the national average concentration, with a value greater than one indicating a higher share of employment than average.
2.4 |. Infant factors
Certain types of CCHD are easier to diagnose on ultrasound.5 For instance, hypoplastic left heart syndrome is readily identified on the 4 chamber view, which is the most commonly obtained obstetrical view.3,5 Transposition of the great arteries, however, appears normal on the 4 chamber view, but can only be identified by the outflow tract view. The echocardiographic view that would typically be required to diagnose the type congenital heart lesion was based on the postnatal ICD diagnosis of the infant, and was assigned to either four chamber view, outflow tract view, or other, based on the categorization described by Hill et al.5
2.5 |. Statistical analysis
Initially, univariate logistic regression was performed on each factor to evaluate for association with prenatal diagnosis. Years were treated as a continuous variable. Due to the collinearity of median household income and percent in poverty, percent in poverty was removed from the multivariate regression model. A multivariate logistic regression was performed on the remainder of the variables, with robust standard errors to account for heteroskedacity. The continuous variables were tested for nonlinearity with a Box-Tidwell test, and they did not violate the linearity assumption. All analyzes were performed using Stata (StataCorp. 2017. Stata Statistical Software: Release 15.1 revision Feb 2020. College Station, TX: StataCorp LLC).
3 |. RESULTS
The database included 15 states with greater than 80% linkage, which had 4,830,332 Medicaid-financed births over the period of study. The states were: AK, CA, CO, DE, IN, LA, MI, NH, NY, OR, SD, TN, VA, VT, WY. Initially, 6039 mother-infant dyads met inclusion based on predefined ICD-9 and PCS codes. Exclusions are shown in Figure 1, and ultimately 4702 mother-baby dyads were included. Patient demographics are shown in Table 1. To estimate the validity of case ascertainment, the incidence of CCHD per live births was calculated. The overall prenatal diagnosis rate in the sample was 27.9%, out of a sample of 4702 infants. There was a steady increase in the rate of prenatal diagnosis rate through the years of the study, which correlated with an increase in the percentage of mothers obtaining routine ultrasounds (Figure 2). On multivariate logistic regression, median household income, year of delivery, maternal diabetes, sonographer location quotient, and needing a view other than a 4-chamber or outflow tract view were statistically significant (Table 2). The odds of prenatal diagnosis increased by a multiplicative factor of 1.005, 95% CI [1.001, 1.008] for each thousand dollar increase in median household income. The unadjusted prenatal diagnosis rate by income decile is shown in Figure 3. The odds of prenatal diagnosis increased by a multiplicative factor of 1.16, 95% CI [1.11, 1.20], for each increasing year, compared with the baseline of 2007. If the mother had a diagnosis code for diabetes mellitus, the odds of being prenatally diagnosed with a congenital heart defect increased by a multiplicative factor of 2.06 95% CI [1.39, 3.04]. Having a fetal cardiac diagnosis needing an outflow tract view did not change the odds of prenatal diagnosis rate, compared with needing a 4 chamber view. However, having a fetal diagnosis that cannot be diagnosed by the standard 4 chamber view or outflow tract view decreased the odds of having a prenatal diagnosis, by a multiplicative odds of 0.38, 95% CI [0.26, 0.56].
FIGURE 1.
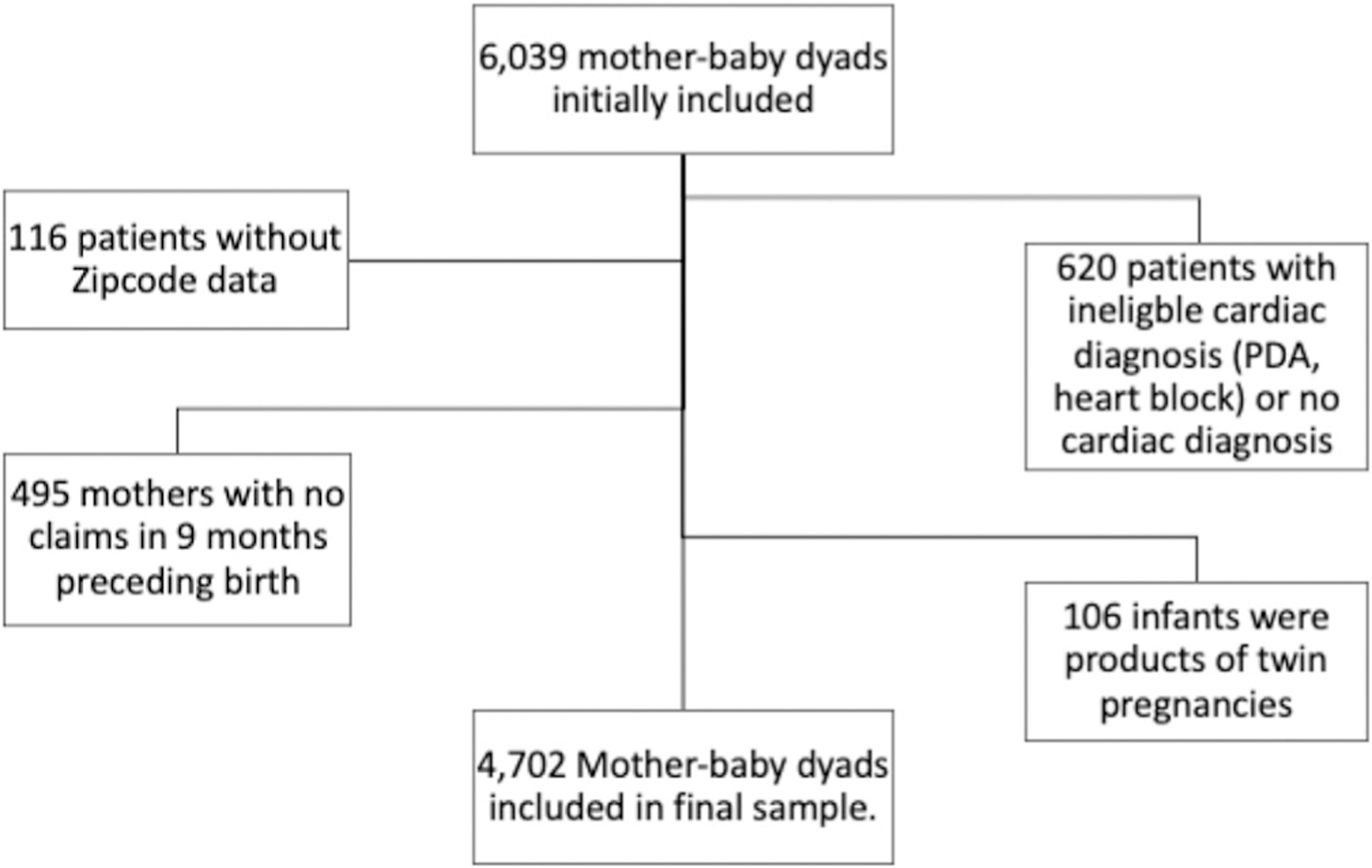
Inclusions and exclusions
TABLE 1.
Demographics
| Variable | n (%) | Median | IQR |
|---|---|---|---|
| Race | |||
| White | 1734 (36%) | — | — |
| Black | 859 (18%) | — | — |
| Hispanic | 1645 (34%) | — | — |
| Other | 538 (11%) | — | — |
| Maternal diabetes | 109 (2.3%) | — | — |
| Household income (thousands) | — | 45.585 | 36.9–56.75 |
| Rural urban continuum code | — | 1 | 1–1 |
| OB location quotienta | — | 1.02 | 0.886–1.441 |
| Sonographer location quotienta | — | 0.904 | 0.904–1.081 |
The location quotient is the ratio of the area concentration of occupational employment (ie, for sonographers or obstetricians) to the national average concentration.
FIGURE 2.
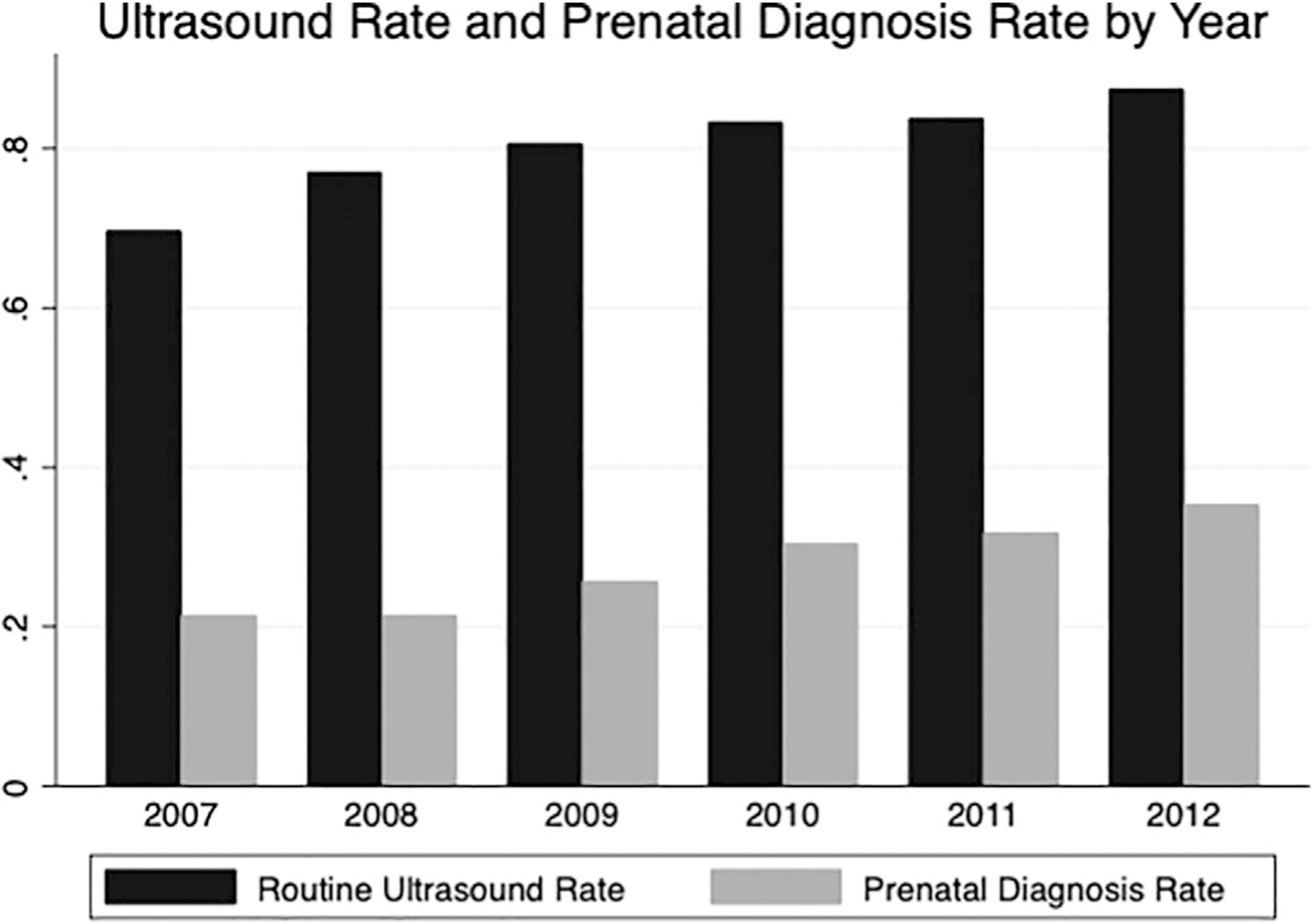
Routine ultrasound rate and prenatal diagnosis rate. The increasing rate of prenatal diagnosis of critical congenital heart disease correlated with increasing rates of routine ultrasounds in pregnancy
TABLE 2.
Multivariate logistic regression of maternal, fetal, and socioeconomic factors on likelihood of prenatal diagnosis of congenital heart disease
| Variable | Odds ratio | 95% CI (P value) |
|---|---|---|
| Rural urban score | 0.99 | [0.95, 1.02] (.45) |
| Median household income (thousands of dollars) | 1.005 | [1.001, 1.008] (.02) |
| Race | ||
| White | 1 | — |
| Black | 0.86 | [0.70, 1.05] (.14) |
| Hispanic | 1.09 | [0.92, 1.30] (.31) |
| Other | 0.91 | [0.71, 1.15] (.42) |
| OB location quotient | 0.92 | [0.70, 1.22] (.57) |
| Sonographer location quotient | 1.80 | [1.01, 3.23] (.047) |
| Year | 1.16 | [1.11, 1.20] (<.001) |
| Maternal diabetes mellitus | 2.06 | [1.39, 3.04] (<.001) |
| View neededa | ||
| 4 chamber | 1 | — |
| Outflow tract | 1.09 | [0.94, 1.26] (.25) |
| Other | 0.38 | [0.26, 0.56] (<.001) |
View needed is the standard ultrasound view that would typically be required to make the diagnosis of the specific type of critical congenital heart disease the patient had.
FIGURE 3.
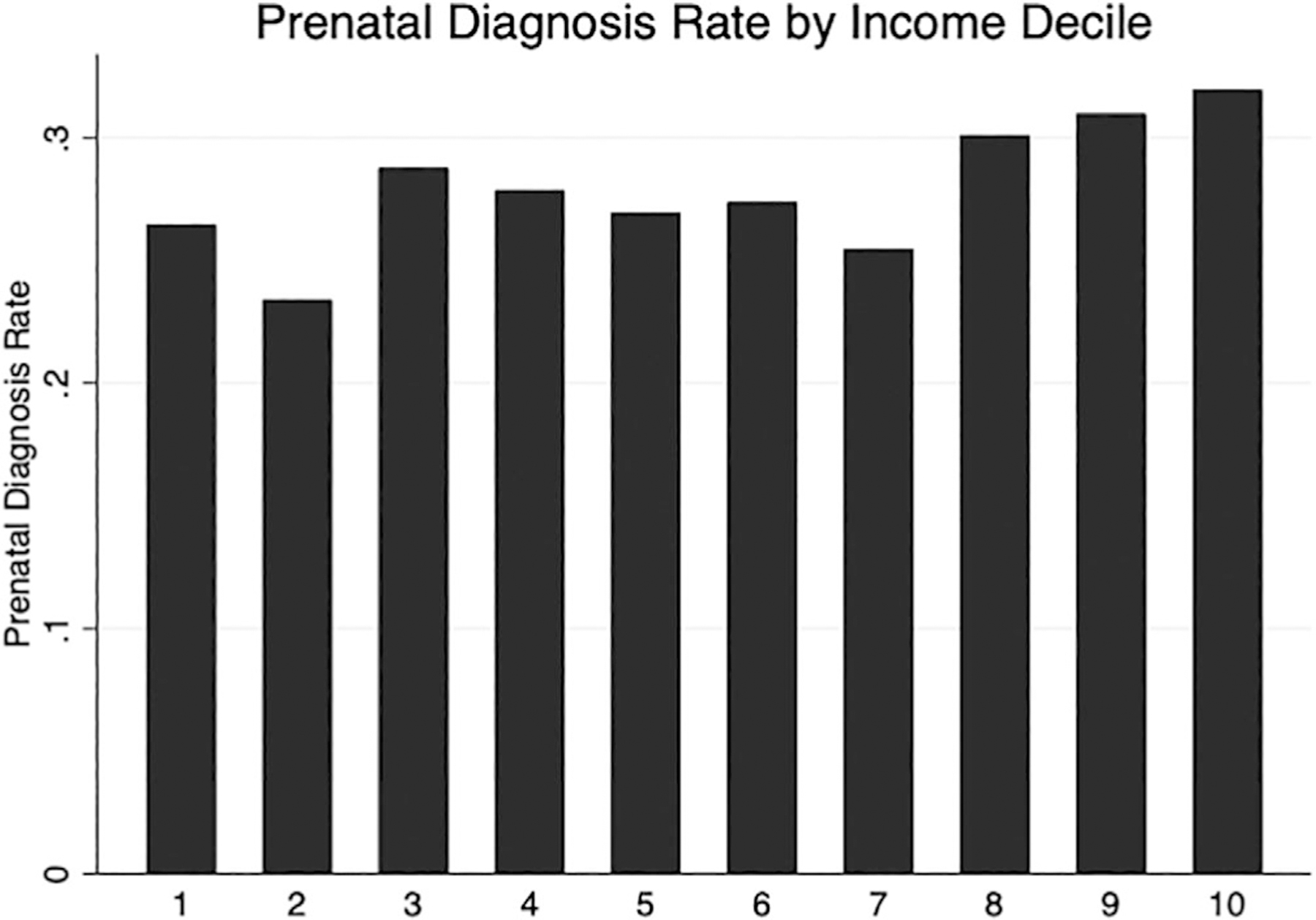
The Effect of Median Household Income on Prenatal Diagnosis Rate. The unadjusted prenatal diagnosis rate is shown based on increasing median income decile. The prenatal diagnosis rate increases with increasing median income decile
Because this study relies on assumptions about prenatal diagnosis based on claims data, alternate formulations of the model were constructed for sensitivity analyzes. When prenatal diagnosis was defined as any pregnant mother of a child with CCHD who was billed for a fetal echocardiogram, the prenatal diagnosis increased to 30%. The same variables of median household income, sonographer location quotient, year of delivery, view required, and presence of maternal diabetes were again independently associated with prenatal diagnosis (Table S1). With the most restrictive definition, that only infants with a fetal echocardiogram and an ICD diagnosis for CHD were considered prenatally diagnosed, the prenatal diagnosis rate fell to 15%. Sonographer location quotient was no longer statistically significantly associated with the outcome, but median income, year of delivery, view required, and presence of maternal diabetes remained statistically significantly associated with the outcome (Table S2). In an additional formulation of the model, percent in poverty was used instead of median household income, and was statistically significantly associated with prenatal diagnosis rate (OR, 0.993, P = .049). The relationships of the additional factors did not significantly change by replacing median household income with percent in poverty in the model (Table S3). When year of birth was treated as a categorical value, later years continued to be statistically significantly associated with prenatal diagnosis rate, and the relationships of additional factors did not significantly change (Table S4). In subgroup analysis, the increase in rate of ultrasounds was consistent across all races.
4 |. DISCUSSION
This large, national study of a select population of women enrolled in Medicaid demonstrated that women living in areas with lower median household income were less likely to have a prenatal diagnosis of CCHD. Additionally, this study shows that increased density of sonographers in a state is associated with increased prenatal diagnosis rate, raising concerns about adequate access to sonographers. These findings are alarming because Medicaid is designed to address disparities, but those disparities still exist despite all of the women in this study being insured. Encouragingly, the prenatal diagnosis rate increased over the years of the study, from 21.9% to 35.3%. This increase coincided with a concomitant increase in use of routine mid-gestation ultrasounds, further suggesting the importance of routine ultrasonography in prenatal detection of CCHD. Maternal conditions such as diabetes, and the type of CCHD in the infant also play important roles in determining prenatal diagnosis rate.
This study helps quantify the impact of socioeconomic factors that were identified by smaller, single center studies. In a retrospective review from 2003 to 2006, Peiris et al. demonstrated that private insurance was the single greatest predictor of prenatal diagnosis, although insurance status was highly correlated with socioeconomic status.4 Hill et al found that living in a higher poverty area was associated with lower prenatal diagnosis rate.5 While this retrospective study cannot assess causality, the association of lower median household income and lower prenatal diagnosis of congenital heart disease may be mediated through neighborhood characteristics. Prior studies have shown that neighborhood characteristics impact the type of care individuals seek, with lower socioeconomic neighborhoods more likely to seek care at safety net centers, such as community health centers, rather than physician offices.11 Additionally, having an ultrasound at a community clinic is associated with a lower prenatal diagnoses rate of congenital heart disease than at a university-based setting, hospital or high risk clinic.6,7 The differences in prenatal diagnosis rate by median household income level among mothers enrolled in Medicaid may indicate a neighborhood level disparity in the quality of care received.
This study also demonstrates that a major factor associated with the increase in prenatal diagnosis rate from 2007 to 2012 was the increase in utilization of mid-gestation ultrasounds over the course of the study. While the overall prenatal diagnosis rate was low, at just 27.9%, it increased steadily from 21.9% to 35.3%. This temporal trend in prenatal diagnosis rate is consistent with prior literature, but for the first time is shown to have coincided with an increase in patients obtaining routine ultrasounds, from 69.7% in 2007 to 87.6% in 2012.3,5 The reasons for the increased utilization of mid-gestation ultrasounds is unclear, although access to prenatal ultrasounds may have been an important factor, as a higher sonographer quotient was associated with higher prenatal diagnosis rate. Additionally, access to fetal echocardiograms may have been an important factor, as only 57.8% of mothers with maternal diabetes were billed for a fetal echocardiogram, despite the American Society of Echocardiography and American Institute of Ultrasound Medicine guideline that all pregnant women with diabetes undergo a fetal echocardiogram.12,13 The increase in mid-gestation ultrasound rate, which may have been related to increased access to sonographers, likely drove the concurrent increase in prenatal diagnosis rate of CCHD.
As noted in prior studies, the type of fetal congenital heart disease is also important in determining prenatal diagnosis rate, because certain diseases are easier to detect by ultrasound.3,5,7 Types of CCHD that can be readily identified on a standard 4 chamber view, such as hypoplastic left heart syndrome, have a much higher rate of prenatal diagnosis than CCHD, such as coarctation of the aorta, that requires nonstandard views. While this study did not find a difference between diagnoses likely requiring an outflow tract view vs a 4 chamber view, that may have been due to the limited ability of administrative data to discern subtle differences in complex congenital heart disease diagnoses.14 Unless additional views are added to the standard mid-gestation obstetrical ultrasound, certain types of CCHD, such as coarctation of the aorta, will continue to be missed. The study reinforces the difficulty of routine ultrasound at detecting certain types of CCHD that are not readily diagnosed on standard ultrasound views.
This study has several limitations. The study relies on retrospectively collected claims data. Due to the limitation of claims data, we cannot differentiate between a fetus who was correctly diagnosed with congenital heart disease from a fetus who had a congenital anomaly and a fetal echocardiogram that missed a congenital heart lesion. We believe the incidence of the latter to be a rare occurrence, because presence of an anomaly in another organ system greatly increases the odds of prenatal diagnosis of CCHD.5 The study is also unable to account for unmeasured confounders, such as obesity. The association of poverty with obesity has long been described, and obesity is associated with decreased quality of fetal ultrasound imaging, which could in turn lead to lower prenatal diagnosis rate.15–18 It is also important to note that this study relied on prenatal diagnosis rate of CCHD in liveborn infants who underwent an intervention within the first 6 months. Infants with congenital heart disease that did not undergo an intervention in the first 6 months of life were not included in the dataset. Therefore, the study could not account for the effects of intrauterine demise, elective termination of pregnancy, or mortality shortly after birth but prior to an intervention. Despite these limitations, the study offers a unique analysis of the factors leading to prenatal diagnosis of CCHD.
5 |. CONCLUSIONS
In the selected Medicaid population, median household income, year of birth, echocardiographic view needed for diagnosis, and maternal diabetes were all independently associated with more frequent prenatal diagnosis of CCHD. Additionally, at the state level lower numbers of sonographers were associated with lower rates of prenatal diagnosis. The steady rise in the prenatal diagnosis of CCHD from 2007 to 2013 was correlated with a concomitant increase in the percentage of patients obtaining screening ultrasounds, which was consistent across race and states. Future increases to the prenatal diagnosis rate will rely on increasing the yield of routine ultrasound screening, and on increasing the percentage of at-risk pregnancies who obtain routine fetal echocardiograms.
Supplementary Material
What’s already known on this topic?
Single center studies have demonstrated that lower socioeconomic status is linked with lower rates of prenatal diagnosis of CCHD. There is wide regional variation in prenatal diagnosis rate of CCHD.
What does this study add?
This large, national study quantified the effect of socioeconomic factors on the likelihood of prenatal diagnosis of CCHD, as well as demonstrated a rise in prenatal diagnosis rate coinciding with an increase in rate of routine mid-gestation ultrasounds.
ACKNOWLEDGEMENT
This publication was made possible by an NIH-funded fellowship to MJC (NIH 5 T32 HL007915).
Funding information
Foundation for the National Institutes of Health, Grant/Award Number: NIH 5T32 HL007915
Footnotes
CONFLICT OF INTEREST
The authors have no conflicts of interest relevant to this article to disclose.
SUPPORTING INFORMATION
Additional supporting information may be found online in the Supporting Information section at the end of this article.
DATA AVAILABILITY STATEMENT
Research Data will not be shared from this study.
REFERENCES
- 1.Verheijen PM, Lisowski LA, Stoutenbeek P, et al. Prenatal diagnosis of congenital heart disease affects preoperative acidosis in the newborn patient. J Thorac Cardiovasc Surg. 2001;121(4):798–803. [DOI] [PubMed] [Google Scholar]
- 2.Holland BJ, Myers JA, Woods CR Jr. Prenatal diagnosis of critical congenital heart disease reduces risk of death from cardiovascular compromise prior to planned neonatal cardiac surgery: a meta-analysis. Ultrasound Obstet Gynecol. 2015;45(6):631–638. [DOI] [PubMed] [Google Scholar]
- 3.Quartermain MD, Pasquali SK, Hill KD, et al. Variation in prenatal diagnosis of congenital heart disease in infants. Pediatrics. 2015;136 (2):e378–e385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Peiris V, Singh TP, Tworetzky W, Chong EC, Gauvreau K, Brown DW. Association of socioeconomic position and medical insurance with fetal diagnosis of critical congenital heart disease. Circ Cardiovasc Qual Outcomes. 2009;2(4):354–360. [DOI] [PubMed] [Google Scholar]
- 5.Hill GD, Block JR, Tanem JB, Frommelt MA. Disparities in the prenatal detection of critical congenital heart disease. Prenat Diagn. 2015;35 (9):859–863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Friedberg MK, Silverman NH, Moon-Grady AJ, et al. Prenatal detection of congenital heart disease. J Pediatr. 2009;155(1):26–31. [DOI] [PubMed] [Google Scholar]
- 7.Pinto NM, Keenan HT, Minich LL, Puchalski MD, Heywood M, Botto LD. Barriers to prenatal detection of congenital heart disease: a population-based study. Ultrasound Obstet Gynecol. 2012;40(4):418–425. [DOI] [PubMed] [Google Scholar]
- 8.Markus AR, Andres E, West KD, Garro N, Pellegrini C. Medicaid covered births, 2008 through 2010, in the context of the implementation of health reform. Womens Health Issues. 2013;23(5):e273–e280. [DOI] [PubMed] [Google Scholar]
- 9.Huybrechts KF, Broms G, Christensen LB, et al. Association between methylphenidate and amphetamine use in pregnancy and risk of congenital malformations: a cohort study from the international pregnancy safety study consortium. JAMA Psychiat. 2018;75(2):167–175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Palmsten K, Huybrechts KF, Kowal MK, Mogun H, Hernandez-Diaz S. Validity of maternal and infant outcomes within nationwide Medicaid data. Pharmacoepidemiol Drug Saf. 2014;23(6):646–655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hussein M, Diez Roux AV, Field RI. Neighborhood socioeconomic status and primary health care: usual points of access and temporal trends in a major US urban area. J Urban Health. 2016;93(6): 1027–1045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.AIUM practice guideline for the performance of fetal echocardiography. J Ultrasound Med. 2011;30(1):127–136. [DOI] [PubMed] [Google Scholar]
- 13.Rychik J, Ayres N, Cuneo B, et al. American Society of Echocardiography guidelines and standards for performance of the fetal echocardiogram. J Am Soc Echocardiogr. 2004;17(7):803–810. [DOI] [PubMed] [Google Scholar]
- 14.Khan A, Ramsey K, Ballard C, et al. Limited accuracy of administrative data for the identification and classification of adult congenital heart disease. J Am Heart Assoc. 2018;7(2):e007378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sobal J, Stunkard AJ. Socioeconomic status and obesity: a review of the literature. Psychol Bull. 1989;105(2):260–275. [DOI] [PubMed] [Google Scholar]
- 16.McLaren L Socioeconomic status and obesity. Epidemiol Rev. 2007; 29:29–48. [DOI] [PubMed] [Google Scholar]
- 17.Uhden M, Knippel AJ, Stressig R, et al. Impact of maternal obesity and maternal overweight on the detection rate of fetal heart defects and the image quality of prenatal echocardiography. Ultraschall Med. 2011;32(Suppl 2):E108–E114. [DOI] [PubMed] [Google Scholar]
- 18.Phatak M, Ramsay J. Impact of maternal obesity on procedure of mid-trimester anomaly scan. J Obstet Gynaecol. 2010;30(5):447–450. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Research Data will not be shared from this study.


