Abstract
Nanotoxicology is argued and considered one of the emerging topics. In this study, polyaniline (PANI)/2-acrylamido-2-methylpropanesulfonic acid (AMPSA) capped silver nanoparticles (NPs)/graphene oxide (GO) quantum dots (QDs) nanocomposite (PANI/Ag (AMPSA)/GO QDs NC) as a nanoadsorbent has a potential for removal of toxic hexavalent chromium (Cr(VI)) ions from water. The acute toxicity of this NC was evaluated on Artemia salina and freshwater Ostracods (Cypridopsis vidua) larvae for 48 h. The measurements were made at 24 and 48 h with 3 repetitions. The 50% effective concentration (EC50) values of the NC were determined after the exposure of these organisms. According to the results of the optical microscope, it was found that both experimental organisms intake the NC. In the toxicity results of Ostracods, the NC had a highly toxic effect only at 250 mg/L after 48 h and the EC50 value was 157.6 ± 6.4 mg/L. For Artemia salina individuals, it was noted that they were less sensitive than the Ostracods and EC50 value was 476 ± 25.1 mg/L after 48 h. These results indicated that PANI/Ag (AMPSA)/GO QDs NC has low toxicity towards both investigated organisms.
Subject terms: Environmental sciences, Chemistry, Nanoscience and technology
Introduction
Engineered nanoparticles are defined as a material has a particle size ranged from 1 to 100 nm1. Nanoscience recently plays an important role by providing new materials with unique characteristics for remediation/treatment and sensing applications2–6. The research field called “nanoecotoxicology” explores the impacts of nanoparticles on the organisms and their ecosystems. This field has widely studied since some of the engineered nanoparticles reach water resources7.
Carbon nanostructures such as graphene, graphene oxide and carbon nanotubes have unique physical and chemical properties with wide potential applications in various fields8–14. Quantum dots (QDs) as colloidal nanocrystals have unique photophysical, electronic and chemical properties due to their large absorption coefficients, high emission and high photostability15. Carbon dots are a new type of carbon-based material class and their low-cost and easy preparation are resulting in their innumerable applications16,17. The green methods are encouraged to synthesize carbon dots because they produce harmless products. In addition, graphene oxide quantum dots (GO QDs) with a particle size ranging from 2 to 20 nm are attracted due to their chemical inertness, low toxicity, biocompatibility, eco-friendly, and good solubility18. It was reported that the 50% effective concentration (EC50) value of GO for Artemia salina is 368.18 mg/L19.
Silver nanoparticles (Ag NPs) have anti-microbial effect, high chemical stability, interested optical and electronic properties, high surface reactivity to enhance the adsorption capabilities and high surface to volume ratio20–22. However, Juganson et al. reported that Ag NPs have the highest toxicity among other nanomaterials23. Several studies investigated the toxicity of Ag NPs on Artemia salina and determined the values of EC50 after 24 h and 72 h to be 19.63 mg/L and 10.70 ± 1.3 mg/L, respectively24,25.
Polyaniline (PANI) and its nanocomposites (NCs) with high stability, high electroactivity, high conductivity and low cost are candidate to apply in the fields of sensors6,26–29 , energy storage10 and water treatment5,30–32. Yslas et al. reported that there is no toxicological effect of PANI nanofibers for Rhinella arenarum larvae exposed to PANI even at the highest concentration, 400 mg/L33. Ibarra et al. confirmed that the 50% lethal concentration (LC50) values of PANI NPs dispersed in polyvinylpyrrolidone and polyN-isopropylacrilamide on the larvae of Rhinella arenarum were 1500 mg/L and 1170 mg/L, respectively34.
Chromium compounds are one of the highest toxic materials and consider as environmental pollutants. High levels of these compounds affect cellular structures. Therefore, there are great efforts to develop selective sensors and adsorbents for chromium with various nanomaterials, nanocomposites or hybrids materials5,32,35–38.
Bioassay tests are important for determining the applicability of the synthesized materials in water treatment. The exposure to the nanomaterials and their possible effects should be evaluated in freshwater and saltwater. Cypridopsis vidua, also called Cypridopsis Müller are freshwater Ostracode. The most dominant species of freshwater Ostracods in Egypt is Cypridopsis vidua39. Ostracods have been used as one of freshwater crustaceans in the ecotoxicological studies and also for toxicity monitoring of water, soil and river sediment40–42.
Artemia salina is another crustacean from salt lakes and hypersaline ecosystems. Artemia salina is also considered as one of the test species by the United States Environmental Protection Agency (US-EPA) for acute toxicity testing43. Many studies have used Artemia salina as a model organism to study the ecotoxicity of some nanoparticles19,25,44–46.
The current work is engaged with testing of PANI/2-acrylamido-2-methylpropanesulfonic acid capped Ag NPs/GO QDs (PANI/Ag (AMPSA)/GO QDs) toxicity which have been prepared and used in the detection and removal of toxic hexavalent chromium (Cr(VI)) from polluted water6,32,47. This test is conducted on larvae of freshwater Ostracode (Cypridopsis vidua) and saltwater Artemia salina. The acute toxicity of different concentrations of PANI/Ag (AMPSA)/GO QDs is studied for 24 h and 48 h of exposure. Also, the uptake and accumulation of the NC in the Ostracode and Artemia larvae are observed and discussed after 24 h of exposure.
Materials and methods
Preparation and characterization techniques
GO QDs were synthesized by direct glucose (BDH Prolabo Chemicals) pyrolysis and dodecylbenzene sulfonic acid (El-Gomhoria Chemical Company, Egypt) doped PANI was synthesized by chemical oxidative polymerization method, respectively. An amount of glucose (2 g) was put in a beaker and heated to 250 °C for 20 min and the orange liquid was added drop by drop to 100 mL of 12.5% NH3 solution with stirring. This solution was heated at 70 °C for 3 h. 0.03 mL of aniline monomer (99.0%, Research Lab, India) was added to 10 mL deionized water and mixed with 10 mL of water (0.3 g DBSA and 0.1 g APS) through 1 h. Ag (AMPSA) NPs were prepared by the chemical reduction of silver nitrate (99.8%, PRS Panreac, Spain) using sodium borohydride (99.0%, Merck, Germany) as a reducing agent. 1.2 mL 10 mM sodium borohydride was inserted to 36.8 mL of deionized water in ice bath with stirring. Then, 0.4 mL 10 mM AgNO3 was mixed and added dropwise and 0.3 mL 10 mM AMPSA (97.0%, Acros Organics, Germany) was added dropwise to the mixture with stirring for 10 min. PANI/Ag (AMPSA) NC was prepared by in situ oxidative polymerization of aniline in with Ag (AMPSA) NPs. Aniline (0.03 mL) was dissolved in 10 mL previously prepared Ag (AMPSA) NPs47.
In addition, PANI/Ag (AMPSA)/GO QDs NC was prepared by in situ oxidative polymerization of aniline in presence of the nanoparticles. PANI/Ag (AMPSA)/GO QDs NC was prepared with the same procedure PANI/Ag (AMPSA) NC was prepared as above. The ternary NC was prepared by mixing 10 mL of AMPSA capped Ag NPs and 1 mL of the previously prepared GO QDs solution under magnetic stirring for 10 min47.
All details regarding the synthesis and characterization of PANI/Ag (AMPSA)/ GO QDs NC and its related materials in the present study can be found in our recent publications32,47. From our previous work47, it was noted that PANI/Ag (AMPSA)/GO QDs NC has the highest and sharpest PL intensity compared with each component of the NC of PANI, GO QDs and Ag (AMPSA) NPs. Therefore, PANI/Ag (AMPSA)/GO QDs NC was selected for Cr(VI) detection. PANI/Ag (AMPSA)/GO QDs NC was used as a sensitive fluorescence quenching probe for detecting Cr(VI). The explanation of the quenching mechanism of this NC is based on the synergistic effect of inner filter effect, the ground state compounds formation and ions exchange6.
Test organisms
The freshwater Ostracod was obtained from a local ornamental fish shop and was identified as Cypridopsis vidua. The identification of species was determined by the National Institute for Oceanography and Fisheries, Alexandria, Egypt. The Ostracods were reared in-house in 500 mL glass Jar at 28 ± 2 °C and fed with yeast powder and algae-containing water under the natural sunlight. The moderately hard synthetic freshwater (96 mg/L NaHCO3 (99%, Acros Organics, Germany), 60 mg/L Ca(NO3)2. 4H2O (99%, PRS Panreac, Spain), 123 mg/L MgSO4·7H2O (99.0%, Fisher Scientific, UK) and 4 mg/L KNO3 (98%, Fisher Scientific, UK)) was used as a test medium48. Adults of Ostracod were isolated in 50 mL beakers and allowed to produce offspring and the young Ostracods (neonates) produced were used in the experiments.
Commercially available dehydrated cysts of Artemia salina (origin: salt lake, U.S.) were obtained from Sera, Germany. Artemia life cycle begins as cysts, then emerged embryos, nauplii, finally larvae and adults. The hatching procedure followed the ARC-Test method49. The artificial seawater of 35 g/L was used for the hatching as well as a testing solution50.
Acute toxicity tests
Toxicity evaluation of PANI/Ag (AMPSA)/GO QDs NC was undertaken by determining the EC50 for both of Artemia salina and freshwater Ostracod (Cypridopsis vidua) and using an immobilization process as an acute endpoint. Forty-eight hour acute toxicity test on Artemia salina was performed according to ISO/TS 20787 standard operating procedure with a slightly modification51. For Cypridopsis vidua we used a modified version test of the OSTRACODTOXKIT F52. Two samples of PANI/Ag (AMPSA)/GO QDs NC (25 mg) were suspended in 25 mL of Ostracods or Artemia culture media to prepare two stock concentrations of 1000 mg/L. The two samples were then suspended and homogenized using an ultrasonic bath (Focus, Spain). Concentrations of 10, 50, 150, 250, 350, 450, 550 and 1000 mg/L of PANI/Ag (AMPSA)/GO QDs NC were tested to determine their acute toxicity on the Ostracods (24 h old) larva and Artemia (instar ӀӀ–ӀӀӀ) stages. Sorgeloos et al.53 reported that in the first Artemia larval stage (instar I), the digestive tract of the nauplius is not in contact yet with the external medium, and the larva only consumes its yolk. In the second larval stage (instar II), it starts the feeding on particulate matter. And, instar I larvae are significantly more resistant to chromic acid than instar II. So, they recommended that bioassays with Artemia larvae should be carried out as short-term toxicity tests with instar ӀӀ-ӀӀӀ stages53.
Groups of 10 Artemia (instar ӀӀ-ӀӀӀ) stages and Ostracods larvae were placed in wells (flat bottom with lid, pre-sterilized, Costar, US) contained 10 mL of the nanocomposite suspension. During the exposure, the tested larvae were not fed and kept under room temperature (28 °C). After 24 and 48 h, the numbers of immobilized larvae (completely motionless) were counted under a binocular microscope (PZ0, Poland) and the immobilization of each treatment was calculated. Immobilization percentage was calculated according to the following equation54:
| 1 |
Negative controls exposure without PANI/Ag (AMPSA)/GO QDs NC were performed in parallel for both the test organisms and in the positive controls, solution of 50 mg/L potassium dichromate (99.0%, Merck, Germany) was used according to the ISO/TS 20787 method51. Ostracods culture and Artemia hatching media containing the same number of organisms (ten organisms) were used. Results were recorded and ensured that the percentage of immobilization in negative control did not exceed 10%. Acute toxicity test was conducted in triplicates and EC50 values were calculated using the probit method of analysis in SPSS version 23 software.
Toxicity testing of the Cr(VI) treated water
The toxic effect of three different concentrations of Cr(VI) solutions (10, 30 and 60 mg/L) before and after treatment with 1 g/L of PANI/Ag (AMPSA)/GO QDs NC was investigated using 48 h acute toxicity bioassays expressed in immobilization percentage (%) for both Ostracod and Artemia salina. The Cr(VI) removal experiments and analyses methods using UV–Visible spectrophotometer (Evolution 300, Thermo scientific, USA) are reported in our previous publication32.
Uptake of PANI/Ag (AMPSA)/GO QDs NC by Ostracods and Artemia salina
The uptake and accumulation of PANI/Ag (AMPSA)/GO QDs NC by the Ostracods and Artemia salina were observed using an optical microscope (ZEISS Primo Star, Germany), and images were taken by an integrated digital camera (ZEISS, Germany).
Results and discussion
Acute toxicity of PANI/Ag (AMPSA)/GO QDs NC on the Ostracods
Table 1 shows the Ostracods mortality with different concentrations of the nanocomposite and the corresponding EC50. In addition, the toxicity unit (TU) values obtained after 24 and 48 h of exposure are depicted in Table 2. TU is equal to 100/EC50, which is the reciprocal of the concentration that causes 50% of the organisms to immobilize by the end of the acute exposure period. Results obtained from Table 1 indicate that the exposure to 10 mg/L of PANI/Ag (AMPSA)/GO QDs has not any toxic effects on the Ostracods. It is noted that further increasing of the NC concentration raises the immobilization. After 24 and 48 h of exposure, 100% immobilization is observed at concentration higher than 450 mg/L NC. Ag NPs and GO QDs dissolution and their aggregation are assumed to control the toxicity. The release of dissolved Ag+ ions and GO from the NC surface to the media can be responsible for the toxicity55.
Table 1.
Ostracods mean immobilization % after 24 and 48 h of exposure to different concentrations of PANI/Ag (AMPSA)/GO QDs NC.
| Concentration (mg/L) | 24 h immobilization % | 48 h immobilization % |
|---|---|---|
| 10 | 0 | 0 |
| 50 | 7 | 10 |
| 150 | 37 | 43 |
| 250 | 60 | 70 |
| 350 | 73 | 80 |
| 450 | 87 | 93 |
| 550 | 100 | 100 |
| 1000 | 100 | 100 |
Table 2.
EC50 and TU of PANI/Ag (AMPSA)/GO QDs NC after 24 and 48 h of exposure to Ostracods.
| EC50 | Mean values (mg/L) | Toxicity level | 95% confidence limits (mg/L) |
|---|---|---|---|
| EC50 at 24 h | 187.1 ± 15.7 | Not acutely toxic | 121.599–251.037 |
| EC50 at 48 h | 157.6 ± 6.4 | (EC50 ˃100) | 99.361–213.655 |
| TU at 24 h | 0.53 | Low toxic | |
| TU at 48 h | 0.63 | (TU˂1) |
The results in Table 2 display that the 48 h EC50 value is lesser than 24 h EC50. The mean values of EC50 and TU for PANI/Ag (AMPSA)/GO QDs after 24 and 48 h are (187.1 and 157.6 mg/L) and (0.53 and 0.63), respectively. Based on the general criteria proposed by Canton et al. and Tonkes et al. for the acute toxicity classification of effluent, PANI/Ag (AMPSA)/GO QDs NC is not acutely toxic (EC50 ˃100 mg/L) and with respect to TU is low toxic (TU˂1)56,57.
Immobilization values are the mean values of three replicates ± standard deviation
Ag NPs have a toxic effect on aquatic organisms and this toxicity is highly depending on the particle size and different surface coatings. Several publications reported that polymer-coated Ag NPs significantly decreased the toxicity of the bare or citrate-coated Ag NPs. Moreover, they found that surface coating was the major factor that determines the toxicity compared with particle size58–60. On the other hand, it was found that there is no acute toxicity for the graphene derivatives demonstrated on the crustaceans although the optical microscope images showed the presence of these graphene derivatives aggregated in the gut19,61.
The main acute toxicity mechanisms governed the toxicity of the nanoparticles in aquatic organisms are the ion regulatory disturbance and competitive inhibition of K or Na ion-dependent adenosine triphosphate (Na+, K+-ATPase) activity62–64. For aquatic organisms in general, including invertebrates, there are very few studies which have addressed this issue. Meanwhile, they reported that after penetrating these NPs into the living cells, it produced reactive oxygen species and induced toxic effects such as membrane lipid peroxidation, mitochondrial damage, DNA damage, and consequently cell apoptosis65.
The low toxicity of PANI/Ag (AMPSA)/GO QDs NC is attributed to the presence of the nontoxic and good environmental stable PANI coating layer of the nanocomposite which decreases the dissolution of the Ag NPs and the release of ions33,34. Moreover, The AMPSA capping agent protects and preserves the Ag NPs from the dissolution.
The results regarding exposure of Artemia salina to different concentrations of PANI/Ag (AMPSA)/GO QDs are expressed in Table 3. A concentration of 250 mg/L of PANI/Ag (AMPSA)/GO QDs has a negligible acute toxic effect (0% immobilization) for Artemia salina after 24 h exposure and 20% immobilization is observed after 48 h. The concentration resulting in 100% immobilization of Artemia salina after 48 h is 1000 mg/L. The mean values of EC50 and TU for PANI/Ag (AMPSA)/GO QDs after 48 h are 476 mg/L and 0.21, respectively as shown in Table 4.
Table 3.
Artemia salina mean immobilization % after 24 and 48 h of exposure to different concentrations of PANI/Ag (AMPSA)/GO QDs NC.
| Concentration (mg/L) | 24 h immobilization % | 48 h immobilization % |
|---|---|---|
| 10 | 0 | 0 |
| 50 | 0 | 0 |
| 150 | 0 | 7 |
| 250 | 0 | 20 |
| 350 | 10 | 30 |
| 450 | 20 | 43 |
| 550 | 40 | 63 |
| 1000 | 80 | 100 |
Table 4.
EC50 and TU of PANI/Ag (AMPSA)/GO QDs NC after 48 h of exposure to Artemia salina.
| EC50 | Mean values (mg/L) | Toxicity level | 95% confidence limits (mg/L) |
|---|---|---|---|
| EC50 at 48 h | 476 ± 25.1 | Not acutely toxic (EC50 ˃100) | 121.599–251.037 |
| TU at 48 h | 0.21 | Low toxic (TU˂1) |
It is found that EC50 values of the nanocomposite for the Ostracods are lower than that for Artemia salina. This is may be due to the high aggregation of PANI/Ag (AMPSA)/GO QDs NC in saltwater to form micro-scale particles. The size distribution of the aggregated nanoparticles is usually not unimodal and the aggregated size increases as the pH approaches the point of zero charge66. The pH of PANI/Ag (AMPSA)/GO QDs suspension in the artificial seawater is observed to be 8 ± 0.5, while the point of zero charge of PANI/Ag (AMPSA)/GO QDs NC is at about pH 10.332.
It is reported that nanoparticles suspended in the seawater tend to aggregate in the range from 400 nm up to several microns in diameter44,67 and the Artemia salina larva are able to ingest them. Artemia salina are nonselective filter feeders, and they can readily ingest particles of up to 50 μm in diameter68. Several studies have reported that the uptake of nanoparticles by Artemia larvae is influenced by the NPs concentration and the time of the exposure while the size of the NPs was not a major factor44,67.
Toxicity of the Cr(VI) treated water
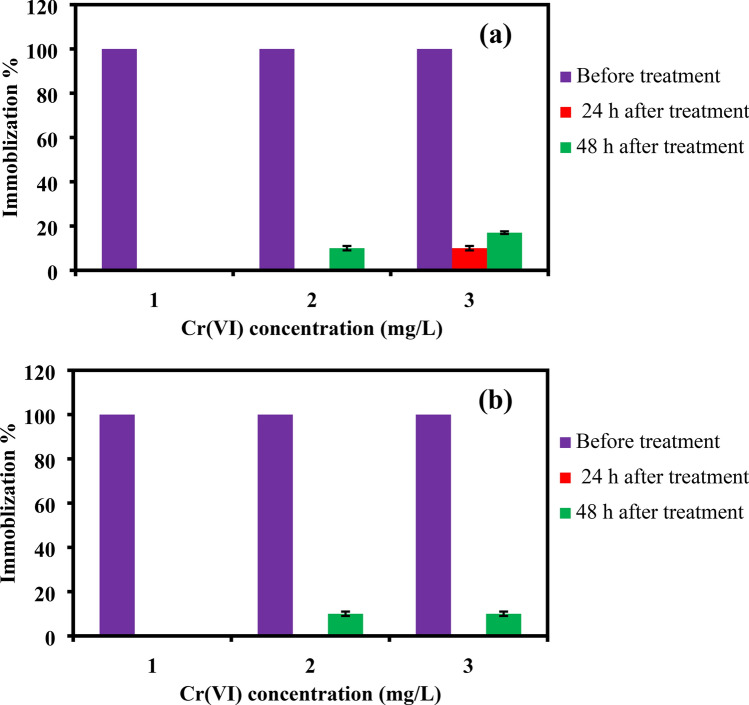
In our recent publication32, PANI/Ag (AMPSA)/GO QDs NC was applied for the water purification of two water samples containing 60 mg L−1 Cr(VI) ions. The results demonstrated that more than 98% of the Cr(VI) ions were removed from the water samples. It was also found that presence of 60 mg L−1 of Cr(VI) with multiions did not significantly affect the removal % by PANI/Ag (AMPSA)/GO QDs NC. The toxicity expressed in immobilization % of Cr(VI) solutions with different concentrations of 10, 30, 60 mg/L before and after treatment using PANI/Ag (AMPSA)/GO QDs NC on the Ostracods and Artemia salina larvae after 24 and 48 h is evaluated as shown in Fig. 1a,b. It is evident that Cr(VI) at these concentrations are completely toxic to both Ostracods and Artemia. However, using PANI/Ag (AMPSA)/GO QDs NC as Cr(VI) adsorbent plainly caused a significant reduction in the immobilization for the tested concentrations. The NC has reduced the immobilization by 100% for Cr(VI) of 10 mg/L, and 90 and 83% for Cr(VI) of 30 and 60 mg/L, respectively for the Ostracods after 48 h exposure as shown in Fig. 1a. For Artemia, up to 90% reduction in the immobilization % is observed in 48 h exposure for 60 mg/L Cr(VI) solution after treatment with 1 g/L PANI/Ag (AMPSA)/GO QDs (Fig. 1b). These immobilization reductions are due to the removal of toxic Cr(VI) occurred by the Cr(VI) adsorption onto the NC surface. The chemical interaction between the NC and Cr(VI) is governed by the ion exchange between the dopant (–SO3–) ions in the adsorbent’s structure and the monovalent bichromate (HCrO4−) ions in the solutions. Also, the electron donating groups on the NC surface reduce the Cr(VI) ions to the low toxic Cr(III) ions. In addition, the electrostatic attraction can be occurred between the positively charged of polaronic charges of NC and Cr(VI) anions5,32.
Figure 1.
Comparison between the immobilization % of Ostracods (a) and Artemia salina (b) larvae exposed to different concentrations of Cr(VI) before and after treatment with PANI/Ag (AMPSA)/GO QDs NC for 24 and 48 h.
Uptake of PANI/Ag (AMPSA)/GO QDs by Ostracods and Artemia salina
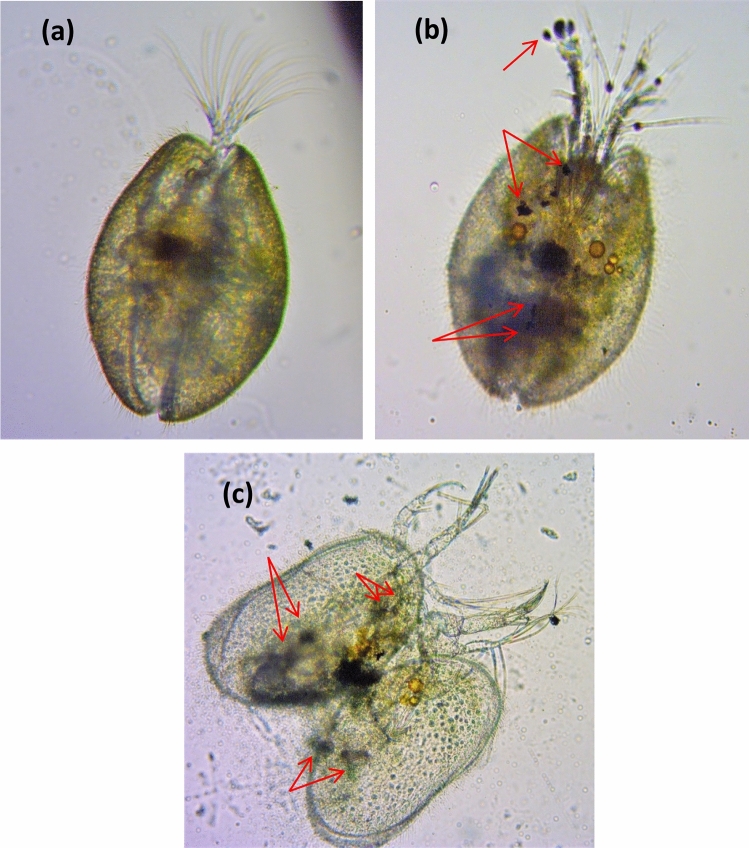
Organisms used for the acute toxicity tests were collected and observed by an optical microscope to study the nanocomposite ingestion and external deposition. Figure 2 shows changes suffered by Ostracods exposed to a solution of 150 mg/L PANI/Ag (AMPSA)/GO QDs NC for 24 h. As can be seen in comparison to the control (Fig. 2a), the dark coloration observed in the Ostracod indicates that these organisms ingested the solution of PANI/Ag (AMPSA)/GO QDs NC. It is noted that there are PANI/Ag (AMPSA)/GO QDs agglomerates impregnated in the carapace, antennules and other parts of the body (Fig. 2b). The shells of Ostracods are composed of low-magnesium calcite, and in some groups are semi-transparent so that the internal parts can be seen through the carapace69. Similar results were reported by using Heterocypris incongruens Ostracods as test organism for hazard evaluation of polystyrene nanoplastic and GO, respectively70,71. Figure 2c displays the dead Ostracods that contains PANI/Ag (AMPSA)/GO QDs aggregates. Dead crustaceans are usually colorless, have their carapaces fully opened, and sometimes they are ripped apart and have their guts spilled over72.
Figure 2.
Optical microscope images of the ingestion of PANI/Ag (AMPSA)/GO QDs (red arrows) by Ostracods (×40 magnification) after 24 h exposure to 150 mg/L PANI/Ag (AMPSA)/GO QDs. The control is clear of PANI/Ag (AMPSA)/GO QDs (a), PANI/Ag (AMPSA)/GO QDs is visible as dark aggregates inside the body of treatment (b) and a dead Ostracod was ingested PANI/Ag (AMPSA)/GO QDs (c).
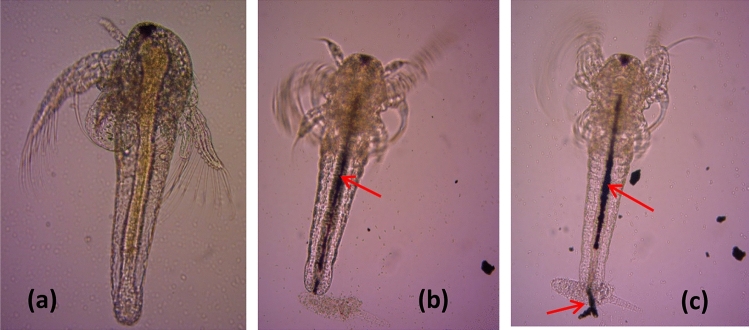
Ingestion of 150 mg/L PANI/Ag (AMPSA)/GO QDs after 24 h by Artemia salina is visually verified also under optical microscope as shown in Fig. 3. The gut is empty in the control (Fig. 3a), after exposure to PANI/Ag (AMPSA)/GO QDs, Artemia salina larvae ingest the nanocomposite and the gut is almost entirely filled as manifested by a dark line inside the gut (Fig. 3b). Finally, the accumulated PANI/Ag (AMPSA)/GO QDs NC is excreted by Artemia salina (Fig. 3c). The accumulation of NPs inside the gut of Artemia salina has already been reported for the ecotoxicity of other NPs19,44.
Figure 3.
Optical microscope images of the ingestion of PANI/Ag (AMPSA)/GO QDs (red arrows) by Artemia salina larvae (×10 magnification) after 24 h exposure to 150 mg/L PANI/Ag (AMPSA)/GO QDs. The gut is empty in the control (a), PANI/Ag (AMPSA)/GO QDs is visible as a dark line inside the gut of treatment (b) and PANI/Ag (AMPSA)/GO QDs is excreted by Artemia salina larvae (c).
Conclusions
The acute toxicity effect of PANI/Ag (AMPSA)/GO QDs NC on the aquatic environment was studied using two organisms, freshwater Ostracods (Cypridopsis vidua) and saltwater Artemia salina larvae for 48 h. The mean values of EC50 of PANI/Ag (AMPSA)/GO QDs NC powder after 48 h of exposure to the Ostracods and Artemia salina were 157.6 ± 6.4 and 476 ± 25.1 mg/L, respectively. PANI/Ag (AMPSA)/GO QDs NC was found to be not acutely toxic for both organisms (EC50 ˃100), although the nanocomposite was accumulated inside the organisms. The Ostracods were appeared to be more sensitive towards PANI/Ag (AMPSA)/GO QDs than Artemia salina. It is recommended to fabricate this NC as a filter and exposed the polluted water for this filter to control the contamination of the NC and the adsorbed pollutants.
Author contributions
S.E. and A.S. contributed to ideas, experiment execution and interpretation of the data and writing of the manuscript. M.K. and H.I. share in the characterization of the prepared nanocomposite and explaining the results of characterization. M.S. supervised the experiments and wrote and edited the manuscript.
Data availability
All data generated or analysed during this study are included in this published article.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Zanker H, Schierz A. Engineered nanoparticles and their identification among natural nanoparticles. Annu. Rev. Anal. Chem. 2012;5:107–132. doi: 10.1146/annurev-anchem-062011-143130. [DOI] [PubMed] [Google Scholar]
- 2.Khan A, et al. Synthesis, characterization of silver nanoparticle embedded polyaniline tungstophosphate-nanocomposite cation exchanger and its application for heavy metal selective membrane. Compos. B. Eng. 2013;45:1486–1492. doi: 10.1016/j.compositesb.2012.09.023. [DOI] [Google Scholar]
- 3.Alam MK, et al. In-situ synthesis of gold nanocrystals anchored graphene oxide and its application in biosensor and chemical sensor. J. Electroanal. Chem. 2019;835:329–337. doi: 10.1016/j.jelechem.2019.01.023. [DOI] [Google Scholar]
- 4.Chen Q, et al. Synthesis and structure study of copolymers from thiadiazole fused indolocarbazole and dithienosilole. Polymer. 2013;54:223–229. doi: 10.1016/j.polymer.2012.11.006. [DOI] [Google Scholar]
- 5.Ebrahim S, Shokry A, Ibrahim H, Soliman M. Polyaniline/akaganéite nanocomposite for detoxification of noxious Cr(VI) from aquatic environment. J. Polym. Res. 2016;23:79–89. doi: 10.1007/s10965-016-0977-6. [DOI] [Google Scholar]
- 6.Ebrahim S, Shokry A, Khalil MMA, Ibrahim H, Soliman M. Polyaniline/Ag nanoparticles/graphene oxide nanocomposite fluorescent sensor for recognition of chromium (VI) ions. Sci. Rep. 2020;10:13617–13627. doi: 10.1038/s41598-020-70678-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Borase HP, Patil SV, Singhal RS. Moina macrocopa as a non-target aquatic organism for assessment of ecotoxicity of silver nanoparticles: Effect of size. Chemosphere. 2019;219:713–723. doi: 10.1016/j.chemosphere.2018.12.031. [DOI] [PubMed] [Google Scholar]
- 8.Shakeel N, et al. Functionalized magnetic nanoparticle-reduced graphene oxide nanocomposite for enzymatic biofuel cell applications. Int. J. Hydrogen Energy. 2019;44:28294–28304. doi: 10.1016/j.ijhydene.2019.09.037. [DOI] [Google Scholar]
- 9.Ghann W, et al. Synthesis and characterization of reduced graphene oxide and their application in dye-sensitized solar cells. ChemEngineering. 2019;3:7–19. doi: 10.3390/chemengineering3010007. [DOI] [Google Scholar]
- 10.ul Haque S, Nasar A, Rahman MM. Applications of chitosan (CHI)-reduced graphene oxide (rGO)-polyaniline (PAni) conducting composite electrode for energy generation in glucose biofuel cell. Sci. Rep. 2020;10:10428–10439. doi: 10.1038/s41598-020-67253-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Karim MR, Rahman MM, Asiri AM. Termination of structural deformation and proton-electron conductive inflection of graphene oxide in six years. ACS Appl. Electron. Mater. 2020;2:1304–1312. doi: 10.1021/acsaelm.0c00101. [DOI] [Google Scholar]
- 12.Alam MK, et al. Highly sensitive and selective detection of Bis-phenol A based on hydroxyapatite decorated reduced graphene oxide nanocomposites. Electrochim. Acta. 2017;241:353–361. doi: 10.1016/j.electacta.2017.04.135. [DOI] [Google Scholar]
- 13.Alam MK, et al. Ultra-sensitive 2-nitrophenol detection based on reduced graphene oxide/ZnO nanocomposites. J. Electroanal. Chem. 2017;788:66–73. doi: 10.1016/j.jelechem.2017.02.004. [DOI] [Google Scholar]
- 14.Rahman MM, Balkhoyor HB, Asiri AM. Phenolic sensor development based on chromium oxide-decorated carbon nanotubes for environmental safety. J. Environ. Manag. 2017;188:228–237. doi: 10.1016/j.jenvman.2016.12.008. [DOI] [PubMed] [Google Scholar]
- 15.Adel R, Ebrahim S, Shokry A, Soliman M, Khalil M. Nanocomposite of CuInS/ZnS and nitrogen-doped grapheme quantum dots for cholesterol sensing. ACS Omega. 2021;6:2167–2176. doi: 10.1021/acsomega.0c05416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mohammadi M, Rezaei A, Khazaei A, Xuwei S, Huajun Z. Targeted development of sustainable green catalysts for oxidation of alcohols via tungstate-decorated multifunctional amphiphilic carbon quantum dots. ACS Appl. Mater. 2019;11:33194–33206. doi: 10.1021/acsami.9b07961. [DOI] [PubMed] [Google Scholar]
- 17.Hadian-Dehkordi L, et al. Amphiphilic carbon quantum dots as a bridge to a pseudohomogeneous catalyst for selective oxidative cracking of alkenes to aldehydes: A nonmetallic oxidation system. ACS Appl. Mater. 2020;12:31360–31371. doi: 10.1021/acsami.0c05025. [DOI] [PubMed] [Google Scholar]
- 18.Karimi B, Ramezanzadeh BA. Comparative study on the effects of ultrathin luminescent graphene oxide quantum dot (GOQD) and graphene oxide (GO) nanosheets on the interfacial interactions and mechanical properties of an epoxy composite. J. Colloid Interface Sci. 2017;493:62–76. doi: 10.1016/j.jcis.2017.01.013. [DOI] [PubMed] [Google Scholar]
- 19.Zhu S, Luo F, Chen W, Zhu B, Wang G. Toxicity evaluation of graphene oxide on cysts and three larval stages of Artemia salina. Sci. Total Environ. 2017;595:101–109. doi: 10.1016/j.scitotenv.2017.03.224. [DOI] [PubMed] [Google Scholar]
- 20.Rahman MM, Khan A, Marwani HM, Asiri AM. Hydrazine sensor based on silver nanoparticle-decorated polyaniline tungstophosphate nanocomposite for use in environmental remediation. Microchim. Acta. 2016;183:1787–1796. doi: 10.1007/s00604-016-1809-4. [DOI] [Google Scholar]
- 21.Deng H, Zhang Y, Yu H. Nanoparticles considered as mixtures for toxicological research. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2018;36:1–20. doi: 10.1080/10590501.2018.1418792. [DOI] [PubMed] [Google Scholar]
- 22.El-Daly SA, Rahman MM, Alamry KA, Asiri AM. Fluorescence quenching of N,N-bis(2,5-di-tert-butylphenyl)-3,4:9,10-perylenebis(dicarboximide) (DBPI) by silver nanoparticles. J. Lumin. 2014;148:303–306. doi: 10.1016/j.jlumin.2013.12.026. [DOI] [PubMed] [Google Scholar]
- 23.Juganson K, Ivask A, Blinova I, Mortimer M, Kahru A. NanoE-Tox: New and in-depth database concerning ecotoxicity of nanomaterials. Beilstein J. Nanotechnol. 2015;6:1788–1804. doi: 10.3762/bjnano.6.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lacave JM, et al. Acute toxicity, bioaccumulation and effects of dietary transfer of silver from brine shrimp exposed to PVP/PEI-coated silver nanoparticles to zebrafish. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2017;199:69–80. doi: 10.1016/j.cbpc.2017.03.008. [DOI] [PubMed] [Google Scholar]
- 25.An HJ, Sarkheil M, Park HS, Yu IJ, Johari SA. Comparative toxicity of silver nanoparticles (AgNPs) and silver nanowires (AgNWs) on saltwater microcrustacean, Artemia salina. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2019;218:62–69. doi: 10.1016/j.cbpc.2019.01.002. [DOI] [PubMed] [Google Scholar]
- 26.Khan A, et al. Preparation and characterization of PANI@G/CWO nanocomposite for enhanced 2-nitrophenol sensing. Appl. Surf. Sci. 2018;433:696–704. doi: 10.1016/j.apsusc.2017.09.219. [DOI] [Google Scholar]
- 27.Khan A, Khan AAP, Rahman MM, Asiri AM. High performance polyaniline/vanadyl phosphate (PANI–VOPO4) nano composite sheets prepared by exfoliation/intercalation method for sensing applications. Eur. Polym. J. 2016;75:388–398. doi: 10.1016/j.eurpolymj.2016.01.003. [DOI] [Google Scholar]
- 28.Karim MR, et al. The fabrication of a chemical sensor with PANI-TiO2 nanocomposites. RSC Adv. 2020;10:12224–12233. doi: 10.1039/C9RA09315J. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Karim MR, et al. Fabrication of 1,4-dioxane sensor based on microwave assisted PAni-SiO2 nanocomposites. Talanta. 2018;193:64–69. doi: 10.1016/j.talanta.2018.09.100. [DOI] [PubMed] [Google Scholar]
- 30.Ebrahim SM, Soliman MM, Abd El-Latif MM. Blend of nylon 6 and polyaniline doped with sulfanilic acid and its schottky diode. High Perform. Polym. 2010;22:377–391. doi: 10.1177/0954008309103796. [DOI] [Google Scholar]
- 31.Shokry A, El-Tahan A, Ibrahim H, Soliman M, Ebrahim S. Polyaniline/akaganéite superparamagnetic nanocomposite for cadmium uptake from polluted water. Desalin. Water Treat. 2019;171:205–215. doi: 10.5004/dwt.2019.24835. [DOI] [Google Scholar]
- 32.Shokry A, El-Tahan A, Ibrahim H, Soliman M, Ebrahim S. The development of ternary nanocomposite for the removal of Cr(VI) ions from aqueous solutions. RSC Adv. 2019;9:39187–39200. doi: 10.1039/C9RA08298K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yslas IE, et al. Polyaniline nanofibers: Acute toxicity and teratogenic effect on Rhinella arenarum embryos. Chemosphere. 2012;87:1374–1380. doi: 10.1016/j.chemosphere.2012.02.033. [DOI] [PubMed] [Google Scholar]
- 34.Ibarra EL, et al. Assessment of polyaniline nanoparticles toxicity and teratogenicity in aquatic environment using Rhinella arenarum model. Ecotoxicol. Environ. Saf. 2015;114:84–92. doi: 10.1016/j.ecoenv.2015.01.013. [DOI] [PubMed] [Google Scholar]
- 35.Khan A, Parwaz Khan AA, Rahman MM, Asiri AM, Alamry KA. Preparation of polyaniline grafted graphene oxide–WO3 nanocomposite and its application as a chromium(iii) chemi-sensor. RSC Adv. 2015;5:105169–105178. doi: 10.1039/C5RA17925D. [DOI] [Google Scholar]
- 36.Hussain MM, Asiri AM, Arshad MN, Rahman MM. Synthesis, characterization, and crystal structure of (E)-Nʹ-(4-bromobenzylidene)-benzenesulfonohydrazide and its application as a sensor of chromium ion detection from environmental samples. J. Mol. Struct. 2020;1207:127810–127818. doi: 10.1016/j.molstruc.2020.127810. [DOI] [Google Scholar]
- 37.Asiri AM, Khan SB, Alamry KA, Marwani HM, Rahman MM. Growth of Mn3O4 on cellulose matrix: Nanohybrid as a solid phase adsorbent for trivalent chromium. Appl. Surf. Sci. 2013;270:539–544. doi: 10.1016/j.apsusc.2013.01.083. [DOI] [Google Scholar]
- 38.El-Shishtawy RM, et al. A new Cr3+ electrochemical sensor based on ATNA/nafion/glassy carbon electrode. Materials. 2020;13:2695–2713. doi: 10.3390/ma13122695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hussein MA, Obuid-AIlah AH, Mahmoud AA, Fangary HM. Ecology of eight species of freshwater ostracods (crustacea) from Qena governorate, Upper Egypt. Egypt J. Aquat. Biol. Fish. 2004;8:107–122. doi: 10.21608/ejabf.2004.1811. [DOI] [Google Scholar]
- 40.El-Temsah SY, Joner JE. Effects of nano-sized zero-valent iron (nZVI) on DDT degradation in soil and its toxicity to collembola and ostracods. Chemosphere. 2013;92:131–137. doi: 10.1016/j.chemosphere.2013.02.039. [DOI] [PubMed] [Google Scholar]
- 41.Ruiz F, González-Regalado ML, Borrego J, Abad MJ, Pendón JG. Ostracoda and foraminifera as short-term tracers of environmental changes in very polluted areas: The Odiel Estuary (SW Spain) Environ. Pollut. 2014;129:49–61. doi: 10.1016/j.envpol.2003.09.024. [DOI] [PubMed] [Google Scholar]
- 42.Plachtová P, et al. Iron and iron oxide nanoparticles synthesized with green tea extract: Differences in ecotoxicological profile and ability to degrade malachite green. ACS Sustain. Chem. Eng. 2018;6:8679–8687. doi: 10.1021/acssuschemeng.8b00986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nunes BS, Carvalho FD, Guilhermino LM, Stappen GV. Use of the genus Artemia in ecotoxicity testing. Environ. Pollut. 2006;144:453–462. doi: 10.1016/j.envpol.2005.12.037. [DOI] [PubMed] [Google Scholar]
- 44.Sarkheil M, et al. Acute toxicity, uptake, and elimination of zinc oxide nanoparticles (ZnO NPs) using saltwater microcrustacean, Artemia franciscana. Environ. Toxicol. Pharmacol. 2018;57:181–188. doi: 10.1016/j.etap.2017.12.018. [DOI] [PubMed] [Google Scholar]
- 45.Lish RAD, Johar SA, Sarkheil M, Yu IJ. On how environmental and experimental conditions affect the results of aquatic nanotoxicology on brine shrimp (Artemia salina): A case of silver nanoparticles toxicity. Environ. Pollut. 2019;255:113358–113365. doi: 10.1016/j.envpol.2019.113358. [DOI] [PubMed] [Google Scholar]
- 46.Demarchi CA, et al. Nanoecotoxicology study of the response of magnetic O-Carboxymethylchitosan loaded silver nanoparticles on Artemia salina. Environ. Toxicol. Pharmacol. 2020;74:103298–103322. doi: 10.1016/j.etap.2019.103298. [DOI] [PubMed] [Google Scholar]
- 47.Shokry A, Khalil MMA, Ibrahim H, Soliman M, Ebrahim S. Highly luminescent ternary nanocomposite of polyaniline, silver nanoparticles and graphene oxide quantum dots. Sci. Rep. 2019;9:16984–16995. doi: 10.1038/s41598-019-53584-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Blinova I, et al. Assessment of the hazard of nine (doped) lanthanides-based ceramic oxides to four aquatic species. Sci. Total Environ. 2018;612:1171–1176. doi: 10.1016/j.scitotenv.2017.08.274. [DOI] [PubMed] [Google Scholar]
- 49.Vanhaecke, P. & Persoone, G. The ARC-test: a standardized short-term routine toxicity test with Artemia nauplii: methodology and evaluation (eds. Persoone, G. et al.) 143–157 (State University of Ghent and Institute Marine Scientific Research, 1984).
- 50.Hadjispyrou S, Kungolos A, Anagnostopoulos A. Toxicity, bioaccumulation, and interactive effects of organotin, cadmium, and chromium on Artemia franciscana. Ecotoxicol. Environ. Saf. 2001;49:179–186. doi: 10.1006/eesa.2001.2059. [DOI] [PubMed] [Google Scholar]
- 51.Johari SA, et al. Introducing a new standardized nanomaterial environmental toxicity screening testing procedure, ISO/TS 20787: Aquatic toxicity assessment of manufactured nanomaterials in saltwater lakes using Artemia sp. nauplii. Toxicol. Mech. Methods. 2018;2:95–109. doi: 10.1080/15376516.2018.1512695. [DOI] [PubMed] [Google Scholar]
- 52.Muna M, et al. Combined effects of test media and dietary algae on the toxicity of CuO and ZnO nanoparticles to freshwater microcrustaceans Daphnia magna and Heterocypris incongruens: Food for thought. Nanomaterials. 2018;9:23–36. doi: 10.3390/nano9010023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sorgeloos P, Remiche-Van Der Wielen C, Persoone G. The use of Artemia nauplii for toxicity tests—A critical analysis. Ecotoxicol. Environ. Saf. 1978;2:249–255. doi: 10.1016/S0147-6513(78)80003-7. [DOI] [PubMed] [Google Scholar]
- 54.Vanhaek P, Persoone G, Claus C, Sorgeloos P. Proposal for a short term toxicity test with Artemia nauplii. Ecotoxicol. Environ. Saf. 1981;5:382–387. doi: 10.1016/0147-6513(81)90012-9. [DOI] [PubMed] [Google Scholar]
- 55.Petersen JE, et al. Adapting OECD aquatic toxicity tests for use with manufactured nanomaterials: Key issues and consensus recommendations. Environ. Sci. Technol. 2015;49:9532–9547. doi: 10.1021/acs.est.5b00997. [DOI] [PubMed] [Google Scholar]
- 56.Canton, J. H. et al. Catch-up operation on old pesticides: An integration. (Rijksinstituut voor Volksgezondheid en Milieu RIVM, National Institute for Public Health and the Environment, 1991).
- 57.Tonkes M, de Graaf PJF, Graansma J. Assessment of complex industrial effluents in the Netherlands using a whole effluent toxicity (or wet) approach. Water Sci. Technol. 1999;39:55–61. doi: 10.2166/wst.1999.0630. [DOI] [Google Scholar]
- 58.Hou J, Zhou Y, Wang C, Li S, Wang X. Toxic effects and molecular mechanism of different types of silver nanoparticles to the aquatic crustacean daphnia magna. Environ. Sci. Technol. 2017;51:12868–12878. doi: 10.1021/acs.est.7b03918. [DOI] [PubMed] [Google Scholar]
- 59.Huynh KA, Chen KL. Aggregation kinetics of citrate and polyvinylpyrrolidone coated silver nanoparticles in monovalent and divalent electrolyte solutions. Environ. Sci. Technol. 2011;45:5564–5571. doi: 10.1021/es200157h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Colman BP, et al. Emerging contaminant or an old toxin in disguise? Silver nanoparticle impacts on ecosystems. Environ. Sci. Technol. 2014;48:5229–5236. doi: 10.1021/es405454v. [DOI] [PubMed] [Google Scholar]
- 61.Pretti C, et al. Ecotoxicity of pristine graphene to marine organisms. Ecotoxicol. Environ. Saf. 2014;101:138–145. doi: 10.1016/j.ecoenv.2013.11.008. [DOI] [PubMed] [Google Scholar]
- 62.Bianchini A, Grosell M, Gregory SM, Wood CM. Acute silver toxicity in aquatic animals is a function of sodium uptake rate. Environ. Sci. Technol. 2002;36:1763–1766. doi: 10.1021/es011028t. [DOI] [PubMed] [Google Scholar]
- 63.Bianchini A, Wood CM. Mechanism of acute silver toxicity in Daphnia magna. Environ. Toxicol. Chem. 2003;22:1361–1367. doi: 10.1002/etc.5620220624. [DOI] [PubMed] [Google Scholar]
- 64.Khoshnamvand M, Ashtiani S, Liu J. Acute toxicity of gold nanoparticles synthesized from macroalga Saccharina japonica towards Daphnia magna. Environ. Sci. Pollut. Res. 2020;27:22120–22126. doi: 10.1007/s11356-020-08770-9. [DOI] [PubMed] [Google Scholar]
- 65.Choi JE, et al. Induction of oxidative stress and apoptosis by silver nanoparticles in the liver of adult zebrafish. Aquat. Toxicol. 2010;100:151–159. doi: 10.1016/j.aquatox.2009.12.012. [DOI] [PubMed] [Google Scholar]
- 66.Tso C, et al. Stability of metal oxide nanoparticles in aqueous solutions. Water Sci. Technol. 2010;61:127–133. doi: 10.2166/wst.2010.787. [DOI] [PubMed] [Google Scholar]
- 67.Ates M, Daniels J, Arslan Z, Farah IO, Rivera HFE. Comparative evaluation of impact of Zn and ZnO nanoparticles on brine shrimp (Artemia salina) larvae: Effects of particle size and solubility on toxicity. Environ. Sci. Process Impacts. 2013;15:225–233. doi: 10.1039/C2EM30540B. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gambardella C, et al. Effects of selected metal oxide nanoparticles on Artemia salina larvae: Evaluation of mortality and behavioural and biochemical responses. Environ. Monit. Assess. 2014;186:4249–4259. doi: 10.1007/s10661-014-3695-8. [DOI] [PubMed] [Google Scholar]
- 69.Kornicker SL. Benthic marine Cypridinacea from Hawaii (Ostracoda) Smithson Contrib. Zool. 1976;231:1–24. [Google Scholar]
- 70.Heinlaan M, et al. Hazard evaluation of polystyrene nanoplastic with nine bioassays did not show particle-specific acute toxicity. Sci. Total Environ. 2020;707:136073–136079. doi: 10.1016/j.scitotenv.2019.136073. [DOI] [PubMed] [Google Scholar]
- 71.Malina T, Maršálková E, Holá K, Zbořil R, Maršálek B. The environmental fate of graphene oxide in aquatic environment—Complete mitigation of its acute toxicity to planktonic and benthic crustaceans by algae. J. Hazard. Mater. 2020;399:123027–123036. doi: 10.1016/j.jhazmat.2020.123027. [DOI] [PubMed] [Google Scholar]
- 72.Sánchez-Bayo F, Goka K. Influence of light in acute toxicity bioassays of imidacloprid and zinc pyrithione to zooplankton crustaceans. Aquat. Toxicol. 2006;78:262–271. doi: 10.1016/j.aquatox.2006.03.009. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analysed during this study are included in this published article.