Abstract
Postural orthostatic tachycardia syndrome (POTS), a disorder of the autonomic nervous system characterized by a rise in heart rate of at least 30 bpm from supine to standing position, has been traditionally viewed as a dysfunction of the peripheral nervous system. However, recent studies and evidence from overlapping conditions suggest that in addition to being considered a disorder of the peripheral nervous system, POTS should be viewed also as a central nervous system (CNS) disorder given (1) significant CNS symptom burden in patients with POTS; (2) structural and functional differences found on neuroimaging in patients with POTS and other forms of orthostatic intolerance; (3) evidence of cerebral hypoperfusion and possible alteration in cerebrospinal fluid volume, and (4) positive response to medications targeting the CNS and non-pharmacologic CNS therapies. This review outlines existing evidence of POTS as a CNS disorder and proposes a hypothetical model combining key mechanisms in the pathophysiology of POTS. Redefining POTS as a CNS disorder can lead to new possibilities in pharmacotherapy and non-pharmacologic therapeutic interventions in patents affected by this disabling syndrome.
Keywords: Postural orthostatic tachycardia syndrome, Central nervous system, Brain, Brainstem, Pathophysiology
Introduction
In the past 30 years, postural orthostatic tachycardia syndrome (POTS), one of the most common disorders of the autonomic nervous system (ANS) [1], has undergone metamorphosis from an obscure and rarely diagnosed disorder to a fairly prevalent and recognized syndrome, generating interest among clinicians and researchers. Today the interest in understanding, diagnosing and treating POTS is especially prominent as COVID-19 pandemic left millions of people worldwide disabled with post-COVID syndrome, including post-COVID dysautonomia.
POTS is defined by the following diagnostic criteria: (1) A sustained heart rate elevation of at least 30 bpm in adults and at least 40 bpm in teens 12–19 years of age from supine to standing position during a 10-min stand test or a tilt table test. (2) Absence of orthostatic hypotension. (3) Symptoms of orthostatic intolerance must be present for at least 6 months [2]. Although POTS is characterized by orthostatic tachycardia and orthostatic intolerance, central nervous system (CNS) symptoms, such as headache, fatigue, chronic dizziness, cognitive dysfunction and sleep disturbance, affect a large number of patients. These symptoms are often more disabling and difficult to treat than the hallmark feature of postural tachycardia. Traditionally, POTS has been viewed as a syndrome affecting the peripheral nervous system via autonomic nerve fibers, with 50% of patients exhibiting small fiber neuropathy on quantitative sudomotor axon reflex test (QSART) [3]. However, central nervous system pathophysiology involvement has been emerging as a possible mechanism in POTS and will be discussed in this paper.
The pathophysiology of POTS has been deemed as largely heterogeneous and traditionally classified as neuropathic, hypovolemic and hyperadrenergic [4]. Recent exploration of the etiology of POTS implicated autoimmunity as one of its major mechanisms. Patients with POTS display a higher prevalence of various non-specific autoimmune markers, including anti-nuclear antibodies, and comorbid autoimmune disorders than the general population [5]. More specifically to the autonomic nervous system, ganglionic N-type and P/Q-type acetylcholine receptor antibodies, alpha 1, beta 1 and beta 2 adrenergic antibodies, muscarinic M2 and M4 antibodies, angiotensin II type 1 receptor antibodies and opioid-like 1 receptor antibodies have been identified in patients with POTS [6–11]. Many of these antibodies have been also identified in patients with chronic fatigue syndrome, small fiber neuropathy, complex regional pain syndromes and cardiovascular disorders—conditions that share some clinical features with POTS.
Interestingly, several of these and other comorbid and overlapping syndromes were recently reframed as possible central nervous system disorders. Migraine, the most common comorbidity of POTS, is viewed as a cortical brain disorder [12]; fibromyalgia is considered a neurosensory disorder with central sensitization as the main mechanism [13, 14], and chronic fatigue syndrome is being reframed as a central nervous system disorder of neuroinflammation and abnormal neurovascular coupling [15, 16]. Applying these novel concepts to the existing evidence of CNS involvement in POTS, it is reasonable to consider POTS as a central nervous system disorder.
ANS in the CNS
Although POTS is considered a disorder of peripheral nervous system [17], evidence that it affects the CNS has been mounting over the past 2 decades. In 1998, Low et al. suggested that although hyperadrenergic state and distal neuropathy are involved in the pathophysiology of POTS, “certain features suggest brainstem dysregulation” [18]. While subsequent studies concentrated on researching the hyperadrenergic state and distal neuropathy, the notion that POTS is associated with brainstem dysregulation has gone largely unexplored.
ANS consists of peripheral and central autonomic pathways, with the central input originating from the hypothalamus and descending through the nuclei in the rostral medulla and caudal pons to peripheral target organs via the sympathetic nervous system (SNS) and parasympathetic nervous system (PNS), which in turn send sensory signals back to the brain [19]. Limbic system, amygdala and insular cortex are important central regions that are involved in regulating emotions, behavior, and homeostasis utilizing the ANS [18]. The dorsal medulla is the main location of the autonomic nuclei responsible for blood pressure regulation and for integrating vagal afferent and efferent pathways [20]. At the level of dorsal medulla, higher total choline and myoinositol were found on magnetic resonance (MR) spectroscopy in adolescents with POTS, syncope and orthostatic hypotension suggestive of neuroinflammation [21].
Neuroimaging studies in POTS, other autonomic disorders and CFS
More recently, studies utilizing neuroimaging with various techniques uncovered noteworthy findings in patients with POTS, as well as other disorders of the orthostatic intolerance. Wagoner et al. utilized proton MR spectroscopy in 11 children and teens with orthostatic intolerance, 5 of whom had POTS confirmed by a tilt table test, and found that patients with orthostatic intolerance had a higher myoinositol and total choline in the dorsal medulla than healthy controls suggesting neuroinflammation as an underlying cause or consequence of the autonomic dysfunction [21].
In a study by Umeda et al. involving 11 patients with POTS and 23 controls, MRI of the brain with voxel-based morphometry and DARTEL procedure demonstrated lower white matter volume beneath the precentral gyrus and paracentral lobule, right pre- and post-central gyrus, paracentral lobule and superior frontal gyrus in POTS patients [22]. There was also a significant negative correlation between left insula volume and trait anxiety and depression scores, which suggested a link between dysregulated physiological reactions arising from compromised central autonomic control and increased vulnerability to psychiatric symptoms in POTS patients [22].
In a study of 22 patients with orthostatic hypotension, defined as a sustained reduction in blood pressure greater than 20 mm HG systolic or greater than 10 mm HG diastolic within 3 min of standing or a tilt table test [2], and 8 patients with POTS using T1-weighted MRI, the cortical thickness in the right hemisphere, including the medial orbitofrontal, peri-calcarine, post-central, inferior temporal, and lateral occipital cortex, and in the peri-calcarine cortex of the left hemisphere was thinned in patients with orthostatic hypotension, but not POTS, compared to normal controls [23].
Neurocardiogenic syncope (NCS) is caused by an abrupt drop in blood pressure, heart rate and cerebral perfusion accompanied by transient loss of consciousness [2]. When MRI of the brain using voxel-based morphometry was compared between 32 patients with NCS proven by a tilt table test and healthy controls, a right insular atrophy was found in patients with NCS [24]. Furthermore, a study of 291 patients with migraine, a common comorbidity with POTS, demonstrated that frequent syncope (OR 2.7) and orthostatic intolerance (OR 2.0) were independent risk factors for high load of deep white matter lesions seen on MRI of the brain [25]. Additionally, the study determined that individuals with orthostatic intolerance had higher prevalence of high periventricular white matter lesion load (OR = 1.9), but there was no increased prevalence of orthostatic hypotension or POTS in that cohort of migraineurs [25].
Two recent case reports also support a CNS etiology of POTS. Kim et al., described an 18-year-old woman with narcolepsy and POTS, whose MRI of the brain demonstrated two non-enhancing lesions in the right thalamus and amygdala [26]. The authors speculate that the amygdala contains nuclei that project to autonomic centers in the hypothalamus and brainstem that regulate physiological responses to fear, stress, and emotion [26]. In another case report, Gadze et al. described a 29-year-old woman with intractable epilepsy and POTS who was treated with vagal nerve stimulation (VNS) for seizures and whose POTS symptoms disappeared and tilt table test became normal with VNS therapy [27]. The authors suggest the afferent parasympathetic nerve fibers, which originate in the end organs and project to the nucleus tractus solitarius first, then to the brain, are likely responsible for the therapeutic effect of VNS since it reduces sympathetic nervous system and increases parasympathetic nervous system activity. The authors speculate that increasing parasympathetic input to the heart via VNS appears to be possibly cardioprotective [27].
Abnormal neuroimaging has been demonstrated in patients with chronic fatigue syndrome (CFS), a syndrome that is comorbid with POTS and shares a significant clinical overlap. CFS is defined as profound fatigue lasting at least 6 months, unalleviated by rest and associated with post-exertional malaise and unrefreshed sleep [28]. Indeed, the autonomic dysfunction is so vital to the pathophysiology of CFS that the presence of orthostatic intolerance has been included as one of the additional diagnostic criteria in the Center for Disease Control case definition of chronic fatigue syndrome [28]. In a study of autonomic correlates of steady state blood pressure and heart rate using MRI of the brain in 25 patients with CFS and 25 healthy controls, vasomotor center, midbrain and hypothalamus correlations were found to be abnormal in CFS, with impaired signaling between brainstem/midbrain regulatory nuclei in patients with CFS [29]. Finkelmeyer et al. also found larger GM volume and lower WM volume in 42 pts with CFS, including increased GM volume in the amygdala and insula, and a reduction in WM volume in the midbrain, pons and right temporal lobe [30].
Cerebrospinal fluid findings in patients with autonomic disorders and CFS
In a study of 32 patients with persistent neurologic symptoms after HPV vaccine, 8 of whom had confirmed autonomic disorders, researchers found increased cerebrospinal fluid (CSF) proinflammatory cytokines and antibodies to GluN2B-NT2, GluN2B-CT and GluN1-NT receptors in patients compared to controls, suggesting T helper 2 immune-mediated response [31]. Similarly, in CFS, CSF proinflammatory cytokines, chemokines and other immunologic markers have been found consistently in at least 30% of patients vs. healthy controls (0%) [32, 33]. Neuroinflammation has been implicated in the pathogenesis of CFS, and an initiative to study the biological mechanisms of neuroinflammation using various neuroimaging and CSF analysis techniques has been set in motion [15].
No studies on CSF analysis in patients with POTS have been conducted to date, but such studies may be necessary to ascertain whether proinflammatory cytokines and neuronal antibodies are also present in patients with POTS. Unfortunately, studies involving a spinal tap are more invasive than those utilizing neuroimaging and may carry a risk of post-lumbar puncture headache in patients with POTS, especially if they have comorbid Ehlers–Danlos syndrome [34], which can make these patients particularly vulnerable to dural leaks.
POTS and mechanical compression of brainstem
There have been several reports in the media and scientific literature implicating compression of the brainstem as a possible mechanism of orthostatic intolerance and describing resolution of POTS, syncope or orthostatic intolerance after surgical intervention [35]. Since Ehlers–Danlos syndrome is comorbid with POTS, and Ehlers–Danlos syndrome is associated with Chiari malformation, a triad of EDS, POTS and Chiari type I malformation is not uncommon in clinical practice. However, one study of 23 women with POTS found that herniation of the cerebellar tonsils is not a common cause of orthostatic intolerance [36]. Nevertheless, the authors admitted that hindbrain compression may still be present and that a single measurement of tonsillar depression might underestimate the number of patients with hindbrain compression [36]. Indeed, some patients may have cranio-cervical instability, which manifests with cerebrospinal fluid flow obstruction and compression of the medulla or pons where central autonomic nervous system networks are located. To date, there have been no formal research studies that investigated mechanical compression of the brainstem as a possible etiology of the autonomic dysfunction outside of the anecdotal reports of resolution of dysautonomia following surgery that have been circulating on social media. A literature search revealed a case series of ten patients with Chiari type I malformation, seven of whom had improved or resolved syncope following the surgical decompression, and another report of a patient whose POTS resolved after surgery for Chiari I malformation [37, 38].
POTS and cerebral blood flow
Abnormal cerebral blood flow has been at the core of POTS pathophysiology with findings of reduced cerebral perfusion, impaired cerebral autoregulation, oscillatory cerebral blood flow, which is linked to impaired cognitive function, and altered EEG amplitude modulation that may reflect abnormal brainstem physiology [39–41]. As a result of cerebral hypoperfusion, cerebral tissue oxygenation, assessed using non-invasive near infrared spectroscopy, is found to decrease during orthostatic provocation in patients with POTS [42]. However, at this time, it is unknown whether lower cerebral tissue oxygenation in POTS patients is a consequence of or a partial cause of POTS symptoms [42].
Van Campen et al. studied 429 patients with CFS with 247 having a normal blood pressure and heart response to a tilt table test, 62 with delated orthostatic hypotension and 120 with POTS using Doppler flow imaging. Abnormal CBF was found in 100% of patients with POTS, 98% of patients with OH and 82% of CFS patients with normal tilt table test, and there was a correlation between orthostatic intolerance symptoms and reduction in the CBF at mid-tilt [43]. The study revealed that 90% of CFS patients had abnormal CBF reduction during the orthostatic stress, regardless of the tilt table test pattern, and that orthostatic intolerance symptoms correlate with CBF reduction.
POTS and intracranial hypotension
Spontaneous intracranial hypotension (SIH) is a relatively rare condition characterized by lower than average intracranial pressure, most commonly caused by a spinal fluid leak at the level of the spine. Orthostatic headache is the hallmark of SIH and is also a symptom in almost 30% of patients with POTS. There is a significant overlap in symptoms of SIH and symptoms of POTS, which raised a possibility of shared etiology. In a case series of 48 patients with POTS and 9 patients with SIH, all patients with SIH fulfilled the clinical criteria of POTS leading the authors to conclude that patients with POTS and orthostatic headache should be screened with further diagnostic tests for SIH [44]. Kato et al. reported a patient with POTS and SIH whose SIH and POTS resolved after treatment with an epidural blood patch, suggesting that POTS may be secondary to SIH [45]. Using dynamic ultrasound of the optic nerve may differentiate between SIH and POTS based on the decreasing optic nerve sheath diameter upon standing in SIH, but not POTS [46].
Although the mechanism of orthostatic headache in POTS is not entirely clear, there are suggestions that a relative CSF hypotension, in terms of reduction in spinal venous pressure and in CSF volume, might be the underlying cause [47]. It is also conceivable that CSF volume may be reduced in patients with POTS, given plasma hypovolemia and possible increased compliance of the dural tube, especially if a connective tissue disorder such as EDS is present. Studies have shown that the role of angiotensin in central neural mechanisms of fluid balance in dehydrated animals depends primarily on the systemic renin–angiotensin system and not an endogenous brain renin-angiotensin system [48], which raises a possibility that aberration in systemic renin–angiotensin–aldosterone may translate into impaired CSF volume regulation in patients with POTS.
POTS and the blood–brain barrier
The blood–brain barrier (BBB) provides an integral protection of the brain from the circulating toxins, immune complexes and infections and has been considered an important substrate in the etiologies of CFS, traumatic brain injury, hypertension and other conditions [49]. Disruption of the BBB is also observed in many different neurologic disorders including multiple sclerosis, stroke, Alzheimer’s disease, epilepsy, and traumatic brain injuries. In most CNS pathologies, the BBB is affected as a result of the inflammation, injury, or degenerative processes specific to the pathology [49].
A recent study demonstrated that a protein component of SARS-CoV-2 virus increased the permeability of the blood–brain barrier, potentially disrupting the delicate neural networks within the brain [50]. Altered function of the BBB can resulted in neuroinflammation and a multitude of neurologic symptoms, such as headache, dizziness, cognitive dysfunction, sleep disturbance and mood alterations that are now seen in patients with post-COVID syndrome—symptoms that are identical to those of POTS. The integrity of BBB needs to be explored using a POTS animal model, which may offer insights into the possible alteration of the blood–brain barrier in patients with POTS.
CNS-targeted therapy
Therapeutically, CNS pharmacologic agents, such as stimulants, anti-depressants and benzodiazepines, have been utilized for treatment of various POTS symptoms, including fatigue, cognitive disturbance, neuropathic pain, sleep disturbance, and mood disorders. Central alpha agonists that work on the CNS, such as clonidine and methyldopa, have been beneficial for sympathetic overactivity, which manifests with episodes of elevated blood pressure, tachycardia, diaphoresis, and anxiety [20]. Immunomodulatory therapy, such as intravenous immunoglobulin, subcutaneous immunoglobulin, and plasmapheresis, used for treatment-refractory POTS with positive autoimmune markers [51–53], may be effective by reducing neuroinflammation in the central nervous system.
Additionally, it has been observed that some patients with POTS improve with biofeedback and rehabilitation programs that focus on cognitive behavioral therapy, exercise therapy, group therapy, physical therapy, occupational therapy, guided meditation and yoga [54]. In a study of seven teens with POTS, a beneficial effect of electronecephalic mirroring, (HIRREM®), a non-invasive neurotechnology, on sympathetic nervous system and symptoms was observed [55]. A plausible explanation of this improvement in some patients may be that neuroplasticity as a result of these therapies could have led to the reorganization of certain networks in the insula, limbic system and other cortical autonomic centers, thereby reducing sympathetic overactivity—the key pathophysiologic mechanism of POTS.
Since deep brain stimulation of substantia nigra was found to improve postural hypotension in patients with Parkinson’s [56], neuromodulation could present a potential treatment avenue for the autonomic disorders. A non-invasive transdermal vagus nerve stimulation for treatment of POTS is currently in clinical trials [57].
POTS as a CNS disorder: proposed model
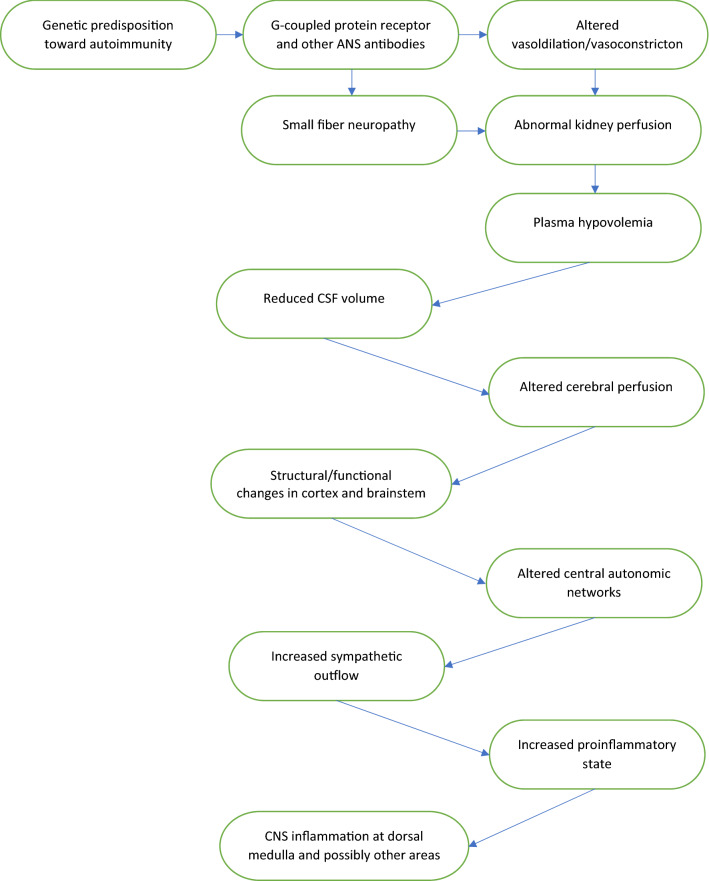
Combining the key mechanisms that have been identified as essential to the pathophysiology of POTS, it is plausible to consider the following hypothetical model involving both peripheral and central autonomic nervous systems (Fig. 1): circulating G-coupled protein receptor and/or other unidentified central or peripheral neuronal or vascular antibodies may result in abnormal vasodilation and vasoconstriction, small fiber neuropathy and impaired renin–angiotensin–aldosterone system via abnormal kidney perfusion or sympathetic denervation, which in turn can lead to reduced circulating plasma volume. Subsequently, systemic hypovolemia may cause or contribute to reduced CSF volume and impaired cerebral blood flow, which can result in structural and functional changes in the cortex and brainstem, altering the central autonomic networks and resulting in increased sympathetic outflow in response to central autonomic activation. Sympathetic overactivity is known to be associated with proinflammatory state, which may in turn result in low-grade CNS inflammation, including or exclusively occurring at the dorsal medulla [21] and possibly in other areas of the brain and/or brainstem. Genetic predisposition toward autoimmunity and/or the presence of hereditary connective tissue disorders may raise an individual’s risk for the formation of antibodies targeting both peripheral and central ANS and vasculature in response to an environmental trigger, such as infection, trauma, surgery, or pregnancy. Similarly, predisposition toward autoimmunity and/or connective tissue disorders could result in weakening of the blood–brain barrier that may be further compromised by the components of the viral particles either via direct invasion of the virus or via autoimmune complex formation with cross-reacting antibodies. Variations of this model, complex interplay between peripheral and central autonomic nervous system and the effect of various unidentified antibodies would need to be further delineated via future research.
Fig. 1.
Pathophysiology of POTS: proposed model
Conclusion
In summary, in addition to being considered a disorder of the peripheral nervous system, POTS should be viewed also as a central nervous system disorder given (1) significant CNS symptom burden in patients with POTS, (2) structural and functional differences found on neuroimaging in patients with POTS and other forms of orthostatic intolerance, (3) evidence of cerebral hypoperfusion and possible alteration in CSF volume and (4) positive response to medications targeting the CNS, non-pharmacologic CNS therapies and in rare cases, surgical interventions that arguably reverse the brainstem compression and restores brainstem perfusion and normal CSF flow.
Large case–control studies utilizing functional MRI, PET scan, MR spectroscopy and other structural and functional neuroimaging modalities, as well as evaluation of neuroinflammatory markers in the cerebrospinal fluid, are needed to (1) confirm whether POTS is disorder of the central nervous system and (2) determine if it is based in neuroinflammation. Redefining POTS as a CNS disorder can in turn lead to new possibilities in pharmacotherapy and non-pharmacologic therapeutic interventions in patents affected by this disabling syndrome.
Declarations
Conflicts of interest
None.
References
- 1.Bhatia R, Kizilbash S, Ahren SP, et al. Outcomes of adolescent-onset postural orthostatic tachycardia syndrome. J Pediatr. 2016;173:149–153. doi: 10.1016/j.jpeds.2016.02.035. [DOI] [PubMed] [Google Scholar]
- 2.Freeman R, Weiling W, Axelrod F, et al. Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Clin Auton Res. 2011;21:69–72. doi: 10.1007/s10286-011-0119-5. [DOI] [PubMed] [Google Scholar]
- 3.Peltier AC, Garland A, Raj SR. Distal sudomotor findings in postural tachycardia syndrome. ClinAuton Res. 2010;20:93–99. doi: 10.1007/s10286-009-0045-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Low PA, Sandroni P, Joyner M, et al. Postural tachycardia syndrome (POTS) J Cardiovasc Electrophysiol. 2009;20:352–358. doi: 10.1111/j.1540-8167.2008.01407.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Blitshteyn S. Autoimmune markers and autoimmune disorders in patients with postural tachycardia syndrome (POTS) Lupus. 2015;24:1364–1369. doi: 10.1177/0961203315587566. [DOI] [PubMed] [Google Scholar]
- 6.Kharraziha I, Axelsson J, Ricci F, et al. Serum activity against G protein-coupled receptors and severity of orthostatic symptoms in postural orthostatic tachycardia syndrome. J Am Heart Assoc. 2020;9:e015989. doi: 10.1161/JAHA.120.015989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gunning WT, 3rd, Kvale H, Kramer PM, et al. Postural orthostatic tachycardia syndrome is associated with elevated G-protein coupled receptor autoantibodies. J Am Heart Assoc. 2019;8:e013602. doi: 10.1161/JAHA.119.013602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Watari M, Nakane S, Mukaino A, et al. Autoimmune postural orthostatic tachycardia syndrome. Ann Clin Transl Neurol. 2018;5:486–492. doi: 10.1002/acn3.524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yu X, Li H, Murphy TA, Nuss Z, et al. Angiotensin II type 1 receptor autoantibodies in postural tachycardia syndrome. J Am Heart Assoc. 2018;7:e008351. doi: 10.1161/JAHA.117.008351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Thieben MJ, Sandroni P, Sletten DM, et al. Postural orthostatic tachycardia syndrome: the Mayo Clinic experience. Mayo Clin Proc. 2007;82:308–313. doi: 10.4065/82.3.308. [DOI] [PubMed] [Google Scholar]
- 11.Li H, Yu X, Liles C, et al. Autoimmune basis for postural tachycardia syndrome. J Am Heart Assoc. 2014;3:e000755. doi: 10.1161/JAHA.113.000755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Barbanti P, Brighina F, Egeo G, et al. Migraine as a cortical brain disorder. Headache. 2020 doi: 10.1111/head.13935. [DOI] [PubMed] [Google Scholar]
- 13.Burgmer M, Pogatzki-Zahn E, Gaubitz M, et al. Altered brain activity during pain processing in fibromyalgia. Neuroimage. 2009;44(2):502–508. doi: 10.1016/j.neuroimage.2008.09.008. [DOI] [PubMed] [Google Scholar]
- 14.Yunus MB. Fibromyalgia and overlapping disorders: the unifying concept of central sensitivity syndromes. Semin Arthritis Rheum. 2007;36(6):339–356. doi: 10.1016/j.semarthrit.2006.12.009. [DOI] [PubMed] [Google Scholar]
- 15.VanElzakker MB, Brumfield SA, Mejia P. Neuroinflammation and cytokines in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): a critical review of research methods. Front Neurol. 2018;9:1033. doi: 10.3389/fneur.2018.01033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shan ZY, Barnden LR, Kwiatek RA, et al. Neuroimaging characteristics of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): a systematic review. J Transl Med. 2020;18:335. doi: 10.1186/s12967-020-02506-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gibbons CH, Bonyhay I, Benson A, et al. Structural and functional small fiber abnormalities in the neuropathic postural tachycardia syndrome. PLoS ONE. 2013;8:e84716. doi: 10.1371/journal.pone.0084716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Novak V, Novak P, Opfer-Gehrking TL, et al. Clinical and laboratory indices that enhance the diagnosis of postural tachycardia syndrome. Mayo Clinic Proc. 1998;73:1141–1150. doi: 10.4065/73.12.1141. [DOI] [PubMed] [Google Scholar]
- 19.Critchley HD, Nagai Y, Gray MA, et al. Dissecting axes of autonomic control in humans: Insights from neuroimaging. Auton Neurosci. 2011;161:34–42. doi: 10.1016/j.autneu.2010.09.005. [DOI] [PubMed] [Google Scholar]
- 20.Benarroch E. The central autonomic network: functional organization, dysfunction, and perspective. Mayo Clinic Proc. 1993;68:988–1001. doi: 10.1016/s0025-6196(12)62272-1. [DOI] [PubMed] [Google Scholar]
- 21.Wagoner AL, Olson JD, Westwood BM, et al. Children with orthostatic intolerance exhibit elevated markers of inflammation in the dorsal medulla. Am J Physiol Heart Circ Physiol. 2019;317:H323–H329. doi: 10.1152/ajpheart.00680.2018. [DOI] [PubMed] [Google Scholar]
- 22.Umeda S, Harrison NA, Gray MA. Structural brain abnormalities in postural tachycardia syndrome: a VBM-DARTEL study. Front Neurosci. 2015;9:34. doi: 10.3389/fnins.2015.00034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shin KJ, Kim TH, Han YH, et al. Cortical morphology in patients with orthostatic intolerance. EurNeurol. 2015;73:264–270. doi: 10.1159/000381540. [DOI] [PubMed] [Google Scholar]
- 24.Kim JB, Suh S, Seo WK, et al. Right insular atrophy in neurocardiogenic syncope: a volumetric MRI study. Am J Neuroradiol. 2014;35:113–118. doi: 10.3174/ajnr.A3611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kruit MC, Thijs RD, Ferrari MD, et al. Syncope and orthostatic intolerance increase risk of brain lesions in migraineurs and controls. Neurology. 2013;80:1958–1965. doi: 10.1212/WNL.0b013e318293e1c7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kim P, During E, Miglis M. A case of narcolepsy type 2 and postural tachycardia syndrome secondary to lesions of the thalamus and amygdala. J Clin Sleep Med. 2018;14:479–481. doi: 10.5664/jcsm.7006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gadze ZP, Kovac AB, Adamec I, et al. Vagal nerve stimulation is beneficial in postural orthostatic tachycardia syndrome and epilepsy. Seizure. 2018;57:11–13. doi: 10.1016/j.seizure.2018.03.001. [DOI] [PubMed] [Google Scholar]
- 28.Institute of Medicine. Beyond myalgic encephalomyelitis/chronic fatigue syndrome: redefining an illness; 2015. https://www.cdc.gov/me-cfs/healthcare-providers/diagnosis/iom-2015-diagnostic-criteria.html
- 29.Barnden LR, Kwiatek R, Crouch B, et al. Autonomic correlations with MRI are abnormal in the brainstem vasomotor centre in Chronic Fatigue Syndrome. Neuroimage Clin. 2016;11:530–537. doi: 10.1016/j.nicl.2016.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Finkelmeyer A, He J, Maclachlan L, et al. Grey and white matter differences in Chronic Fatigue Syndrome—a voxel-based morphometry study. Neuroimage Clin. 2017;17:24–30. doi: 10.1016/j.nicl.2017.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Takahashi Y, Matsudaira T, Nakano H, Nasu H, Ikeda H, Nakaoka K. Immunological studies of cerebrospinal fluid from patients with CNS symptoms after human papillomavirus vaccination. J Neuroimmunol. 2016;298:71–78. doi: 10.1016/j.jneuroim.2016.07.003. [DOI] [PubMed] [Google Scholar]
- 32.Hornig M, Gottschalk G, Peterson DL, et al. Cytokine network analysis of cerebrospinal fluid in myalgic encephalomyelitis/chronic fatigue syndrome. Mol Psychiatry. 2016;21:261–269. doi: 10.1038/mp.2015.29. [DOI] [PubMed] [Google Scholar]
- 33.Natelson BH, Weaver SA, Tseng CL, et al. Spinal fluid abnormalities in patients with chronic fatigue syndrome. Clin Diagn Lab Immunol. 2005;12:52–55. doi: 10.1128/CDLI.12.1.52-55.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shaw BH, Stiles LE, Bourne K, et al. The face of postural tachycardia syndrome—insights from a large cross-sectional online community-based survey. JIM. 2019;286:438–448. doi: 10.1111/joim.12895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Garland EM, Robertson D. Chiari I malformation as a cause of orthostatic intolerance symptoms: a media myth? AJM. 2001;111:546–552. doi: 10.1016/s0002-9343(01)00922-6. [DOI] [PubMed] [Google Scholar]
- 36.Garland EM, Anderson JC, Black BK, Kessler RM, Konrad PE, Robertson D. No increased herniation of the cerebellar tonsils in a group of patients with orthostatic intolerance. Clin Auton Res. 2002;12:472–476. doi: 10.1007/s10286-002-0051-9. [DOI] [PubMed] [Google Scholar]
- 37.Straus D, Foster K, Zimmerman F, Frim D. Chiari drop attacks: surgical decompression and the role of tilt table testing. Pediatr Neurosurg. 2009;45:384–389. doi: 10.1159/000260909. [DOI] [PubMed] [Google Scholar]
- 38.Prilipko O, Dehdashti AR, Zaim S, Seeck MJ. Orthostatic intolerance and syncope associated with Chiari type I malformation. Neurol Neurosurg Psychiatry. 2005;76:1034–1036. doi: 10.1136/jnnp.2004.048330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Low PA, Novak V, Spies JM, et al. Cerebrovascular regulation in the postural orthostatic tachycardia syndrome (POTS) Am J Med Sci. 1999;317:124–133. doi: 10.1097/00000441-199902000-00007. [DOI] [PubMed] [Google Scholar]
- 40.Stewart JM, Del Pozzi AT, Pandey A, et al. Oscillatory cerebral blood flow is associated with impaired neurocognition and functional hyperemia in postural tachycardia syndrome during graded tilt. Hypertension. 2015;65:636–643. doi: 10.1161/HYPERTENSIONAHA.114.04576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lagerlund TD, Low PA, Novak V, et al. Spectral analysis of slow modulation of EEG amplitude and cardiovascular variables in subjects with postural tachycardia syndrome. Auton Neurosci. 2005;117:132–142. doi: 10.1016/j.autneu.2004.11.010. [DOI] [PubMed] [Google Scholar]
- 42.Kharraziha I, Holm H, Bachus E, et al. Cerebral oximetry in syncope and syndromes of orthostatic intolerance. Front Cardiovasc Med. 2019;6:171. doi: 10.3389/fcvm.2019.00171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Van Campen C, Verheugt F, Rowe PC, et al. Cerebral blood flow is reduced in ME/CFS during head-up tilt testing even in the absence of hypotension or tachycardia: a quantitative, controlled study using Doppler echography. ClinNeurophysPract. 2020;5:50–58. doi: 10.1016/j.cnp.2020.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Graf N, Santos A, Ulrich C, et al. Clinical symptoms and results of autonomic function testing overlap in spontaneous intracranial hypotension and postural tachycardia syndrome: a retrospective study. Cephalalgia Rep. 2018;1:1–6. [Google Scholar]
- 45.Kato Y, Hayashi T, Arai N, et al. Spontaneous intracranial hypotension associated with Postural Tachycardia Syndrome. Intern Med. 2019;58:2569–2571. doi: 10.2169/internalmedicine.2587-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cipriani D, Rodriguez B, Häni L, et al. Postural changes in optic nerve and optic nerve sheath diameters in postural orthostatic tachycardia syndrome and spontaneous intracranial hypotension: a cohort study. PLoS ONE. 2019;14:e0223484. doi: 10.1371/journal.pone.0223484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mokri B, Low PA. Orthostatic headaches without CSF leak in postural tachycardia syndrome. Neurology. 2003;61:980–982. doi: 10.1212/01.wnl.0000085868.37963.7d. [DOI] [PubMed] [Google Scholar]
- 48.Bell FR, Doris PA, Wood TJ. The coincidental effects of dehydration and rehydration on plasma and cerebrospinal fluid angiotensin II levels in unrestrained steers. Brain Res. 1985;325:143–150. doi: 10.1016/0006-8993(85)90310-5. [DOI] [PubMed] [Google Scholar]
- 49.Daneman R, Prat A. The blood-brain barrier. Cold Spring Harb Perspect Biol. 2015;7:a020412. doi: 10.1101/cshperspect.a020412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Buzhdygan TP, DeOre BJ, Baldwin-Leclair A, et al. The SARS-CoV-2 spike protein alters barrier function in 2D static and 3D microfluidic in-vitro models of the human blood-brain barrier. Neurobio Dis. 2020 doi: 10.1016/j.nbd.2020.105131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Schofield JR, Chemali KR. Intravenous immunoglobulin therapy in refractory autoimmune dysautonomias: a retrospective analysis of 38 patients. Am J Ther. 2019;26:570–582. doi: 10.1097/MJT.0000000000000778. [DOI] [PubMed] [Google Scholar]
- 52.Kesterson K. Blitshteyn S. Subcutaneous immunoglobulin as an effective therapy for POTS. Auton Neurosci 2020; Abstract
- 53.Wells R, Hissaria P, Elliott AD, et al. Plasma exchange therapy in postural tachycardia syndrome: a novel long-term approach? Am J Med. 2020;133:e157–159. doi: 10.1016/j.amjmed.2019.10.016. [DOI] [PubMed] [Google Scholar]
- 54.Bruce BK, Harrison TE, Bee SM, et al. Improvement in functioning and psychological distress in adolescents with postural orthostatic tachycardia syndrome following interdisciplinary treatment. ClinPediatr (Phila) 2016;55:1300–1304. doi: 10.1177/0009922816638663. [DOI] [PubMed] [Google Scholar]
- 55.Fortunato JE, Tegeler CL, Gerdes L, et al. Use of an allostatic neurotechnology by adolescents with postural orthostatic tachycardia syndrome (POTS) is associated with improvements in heart rate variability and changes in temporal lobe electrical activity. Exp Brain Res. 2016;234:791–798. doi: 10.1007/s00221-015-4499-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Stemper B, Beric A, Welsch G, et al. Deep brain stimulation improves orthostatic regulation of patients with Parkinson disease. Neurology. 2006;67:1781–1785. doi: 10.1212/01.wnl.0000244416.30605.f1. [DOI] [PubMed] [Google Scholar]
- 57.https://clinicaltrials.gov/ct2/show/NCT02281097