Abstract
The most important characteristics of coronavirus disease 2019 (COVID-19) transmission that makes it difficult to control are 1) asymptomatic and presymptomatic transmission, 2) low incidence or lack of dominant systemic symptoms such as fever, 3) airborne transmission that may need a high infectious dose, and 4) super-spread events (SSEs). Patients with COVID-19 have high viral loads at symptom onset or even a few days prior to symptom onset, and most patients with COVID-19 have only mild respiratory symptoms or merely pauci-/null-symptoms. These characteristics of the virus enable it to easily spread to the community because most patients are unaware of their potential infectivity, and symptom-based control measures cannot prevent this type of transmission. Furthermore, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is also capable of airborne transmission in conditions such as aerosol-generating procedures, under-ventilated indoor spaces, and over-crowded areas. In this context, universal mask-wearing is important to prevent both outward and inward transmission until an adequate degree of herd immunity is achieved through vaccination. Lastly, the SSEs of SARS-CoV-2 transmission emphasize the importance of reducing contacts by limiting social gatherings. The above-mentioned transmission characteristics of SARS-CoV-2 have culminated in the failure of long-lasting quarantine measures, and indicate that only highly effective vaccines can keep the communities safe from this deadly, multifaceted virus.
Keywords: SARS-CoV-2, COVID-19, Transmission
INTRODUCTION
In late December of 2019, the first pneumonia cases of unknown cause were reported in Wuhan, China, and the pathogen was later identified and named as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). SARS-CoV-2 quickly spread to other countries and the World Health Organization declared this ongoing outbreak of coronavirus disease 2019 (COVID-19) as a pandemic on March 11, 2020. As of December 31, 2020, more than 83 million individuals worldwide have been infected with SARS-CoV-2, of whom more than 1.8 million (2.2%) died. In this review, we summarize the most important characteristics of COVID-19 transmission: 1) asymptomatic and presymptomatic transmission, 2) low incidence or lack of dominant systemic symptoms such as fever, 3) airborne transmission that may need a high infectious dose, and 4) super-spread events (SSEs).
ASYMPTOMATIC AND PRESYMPTOMATIC TRANSMISSION
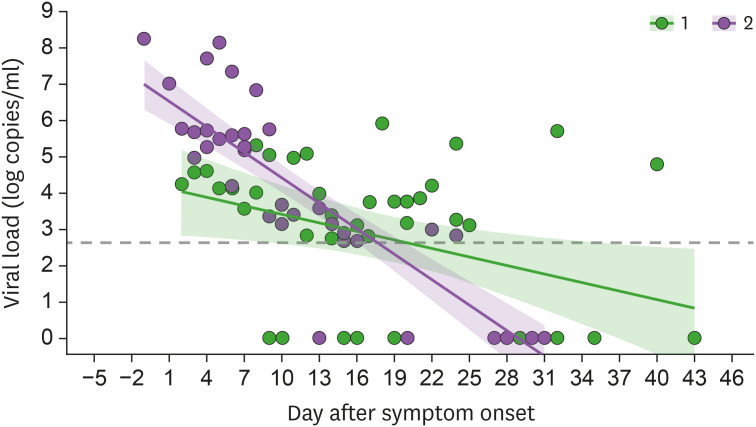
In the case of severe acute respiratory syndrome (SARS), the maximal viral shedding (i.e., high infectivity) occurred late in the disease course (e.g., 7–10 days after symptom onset) and infectivity was limited in the early course of the disease (1). As such, SARS could be successfully controlled by early case identification and strict quarantine measures without the need for a vaccine. The viral load of Middle East respiratory syndrome coronavirus (MERS-CoV) in patients with Middle East respiratory syndrome (MERS) was reported to peak during the second week of illness, thus resembling the disease course of SARS (2). Therefore, early case identification also sufficed for MERS and a vaccine was not necessary for successful control. In contrast, patients with COVID-19 have a high viral load at symptom onset or even a few days prior to symptom onset in some cases (Fig. 1). As such, the spread of COVID-19 cannot be readily controlled using the quarantine measures used in SARS or MERS.
Figure 1. Viral load kinetics in the nasopharyngeal swab samples of patients with severe (group 1, n=3) and mild-to-moderate (group 2, n=4) symptoms of COVID-19 who were admitted to Asan Medical Center between February 2020 and June 2020. SARS-CoV-2 viral loads were quantified by in-house real-time RT-PCR assay targeting the S gene and the N gene of SARS-CoV-2.
Presymptomatic transmission is a unique feature of SARS-CoV-2 that significantly hinders the effective control of this pandemic. A previous study reported that viable virus was isolated 6 days prior to symptom onset (3), and other experts also stated that presymptomatic transmission occurred 1–3 days before symptom onset (4,5). Our analysis of nosocomial transmission clusters revealed that presymptomatic transmission occurred 1 to 4 days prior to symptom onset (manuscript under consideration elsewhere). In addition, a household transmission study from Wuhan, China revealed that the transmissibility of SARS-CoV-2 was 1.4-fold higher in the presymptomatic period than in the postsymptomatic period (6). Timely quarantine of contacts with a known confirmed patient may prevent further presymptomatic transmission, but every new patient with presymptomatic transmission theoretically leads to secondary cases before symptom-based identification and isolation. Therefore, forward tracing after disease confirmation has a limited effect on reducing transmission, and backward tracing before disease confirmation poses significant burdens on epidemiologic investigations. In addition, it is yet unclear as to how many patients with SARS-CoV-2 infection have presymptomatic viral shedding, how contagious they are during the presymptomatic period, and how long prior to symptom onset are the patients able to spread the virus. Such uncertainty inevitably results in unidentified transmitters that generate further transmission chains.
Furthermore, one study showed that 20%–30% of patients with SARS-CoV-2 infection were classified as persistently asymptomatic, and not presymptomatic (7). During the early phase of this pandemic, many experts believed that asymptomatic individuals could not play a major role in the transmission of the disease because of low infectivity. However, accumulating data suggest that asymptomatic individuals are indeed contributing to the ongoing community spread of COVID-19. Epidemiologic findings indicate that the secondary attack rate was indeed lower in contacts of people with asymptomatic infection than those with symptomatic infection (relative risk 0.35) (7). However, asymptomatic individuals with SARS-CoV-2 infection often go unnoticed; instead, they are usually discovered through contact tracing during outbreak investigations, and are immediately isolated and have less chance to spread to others. Accordingly, the presently available epidemiologic data may be largely underestimating the infectivity of asymptomatic individuals with SARS-CoV-2 infection.
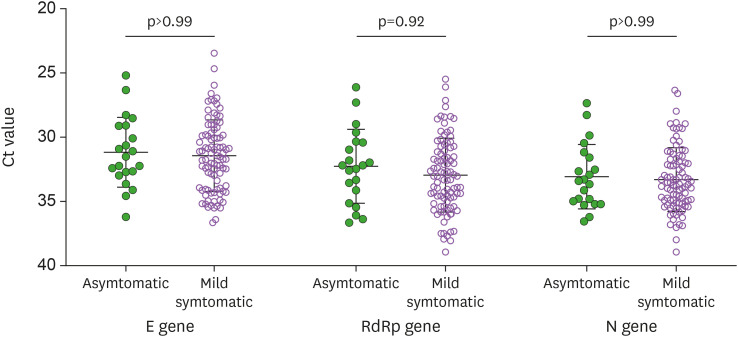
Instead of epidemiologic data, we can infer the infectivity of asymptomatic individuals from the viral shedding kinetics of asymptomatic individuals compared with those of symptomatic individuals. Previous studies have repeatedly shown that asymptomatic individuals had a comparable viral load compared with that of symptomatic patients (8,9). Likewise, we also found that the mean Ct values of SARS-CoV-2 genes were highly similar between asymptomatic individuals and symptomatic patients (Fig. 2) (10). Therefore, asymptomatic individuals with SARS-CoV-2 infection seem to have a comparable potential for spreading the virus as much as symptomatic patients, thus supporting the possibility that asymptomatic individuals are contributing to the ongoing community spread of COVID-19. A recent analytic model that assessed the proportion of SARS-CoV-2 transmissions in the community revealed that at least 50% of transmissions came from asymptomatic cases (11). However, while asymptomatic individuals with SARS-CoV-2 infection have comparable viral shedding and more active social interaction than symptomatic COVID-19 patients, asymptomatic infected persons exhibited a short duration of viral shedding that may result in less chance of transmission (12). One post-outbreak study from Wuhan reported that there was no evidence of transmission from a total of 1,174 close contacts of the asymptomatic individuals who had positive SARS-CoV-2 PCR results (12). The important characteristics of SARS-CoV-2 infection including viral shedding and transmissibility between symptomatic and asymptomatic individuals are shown in Table 1. Further studies are urgently needed on a more detailed viral shedding kinetics and transmission rate of asymptomatic individuals.
Figure 2. Comparisons of viral loads between asymptomatic and mild symptomatic individuals with SARS-CoV-2 infection for the E gene, RdRp gene, and N gene of SARS-CoV-2 (10). The nasopharyngeal and oropharyngeal swab samples were obtained from asymptomatic individuals and mild symptomatic individuals at a median of 13 days from the diagnosis. SARS-CoV-2 viral loads (Ct values) were quantified by a commercial real-time RT-PCR using the Allplex 2019-nCoV Assay kit (Seegene, Seoul, Korea).
Table 1. Comparison of the important characteristics of SARS-CoV-2 transmission between symptomatic, presymptomatic, and asymptomatic individuals.
| Characteristics | Symptomatic | Presymptomatic | (Persistent) Asymptomatic |
|---|---|---|---|
| Proportion | 30%–40%? | 20%–30%? | 20%–30% (7,11) |
| Viral load by PCR | Reference | Comparable but peak before symptom onset | Comparable (8,9,10) |
| Viable viral shedding | Up to 10 days after symptom onset (4,5) | Up to 4–6 days before symptom onset and up to 10 days after symptom onset (3,4,5) | Up to 7–10 days after diagnosis or viral replication (4,5) |
| Transmissibility | Reference | Higher before symptom onset (1.4-fold higher) (6) | 1/3–1/5 lower (6,7) |
| Social activity (average rate of contact) | Low | High before symptom onset | High |
| Difficulty for control using quarantine policy | Reference | High | High |
| Contribution to transmission | 30%–40% (11) | 35%–45% (11) | 25% (11) |
Parentheses indicate the reference.
LOW INCIDENCE OR LACK OF DOMINANT SYSTEMIC SYMPTOMS SUCH AS FEVER
Mild respiratory symptoms or pauci-/null-symptoms are common in patients with COVID-19. Such lack or absence of symptoms unfortunately facilitate the spread of the virus to the community because the infected individuals are not aware of their ability to transmit the viruses, and symptom-based infection control measures cannot prevent this type of transmission. Most patients with SARS or MERS had a fever (13,14), which enabled their detection and subsequent isolation; in contrast, only 10% to 30% of patients with SARS-CoV-2 infection have a fever (15,16), suggesting that fever screening, which is widely used in hospitals and community settings, may only detect no more than one-third of patients with SARS-CoV-2 infection. Therefore, in addition to fever screening, screening questionnaires including other non-specific symptoms (e.g., sore throat, myalgia, anosmia) and epidemiologic links should be used for the early detection of SARS-CoV-2 infection.
AIRBORNE TRANSMISSION THAT MAY NEED A HIGH INFECTIOUS DOSE
SARS-CoV and MERS-CoV were both capable of airborne transmission. There have been at least 3 examples of SARS outbreaks due to airborne transmissions including that in the Amoy Garden in Hong Kong, in the Prince of Wales Hospital in Hong Kong, and in airplanes (17,18,19). Roy and Milton (20) proposed that the transmission mode of SARS-CoV would lie somewhere between droplet transmission and airborne transmission. As for MERS-CoV, experimental aerosolization of MERS-CoV did not decrease its stability at 20oC and 40% relative humidity (21). Bin et al. (22) showed that at the entrance to air-ventilating equipment in the room of a patient with MERS, MERS-CoV was detected by RT-PCR but not by viral culture, and suggested the existence of airborne-virus particles. Our previous study also demonstrated viable MERS-CoV from air samples and swabbed surfaces such as the ventilator exit and the top of televisions, which were inaccessible areas that are remote from the patients as well as those easily missed in daily cleaning, suggesting the potential airborne and contact transmission routes of MERS-CoV (23).
Like SARS-CoV and MERS-CoV, SARS-CoV-2 was reported to have stability in the air (24). An epidemiology study demonstrated that social distancing measures for interrupting droplet transmission with hand hygiene for interrupting contact transmission were ultimately unsuccessful in controlling the community spread of SARS-CoV-2, whereas mandatory mask-wearing was effective in interrupting the transmission (25). This epidemiologic observation indicates that airborne transmission is the dominant route even in community settings without aerosol-generating procedures, especially those that are under-ventilated or crowded (25). In addition, large outbreaks in under-ventilated areas (26,27) and the transmission of SARS-CoV-2 at a distance of beyond 2 meters from index patients (28,29) are more easily explained by airborne transmission rather than droplet or contact transmissions. Some may argue against airborne transmission because more explosive outbreaks or high basic reproduction numbers are expected if SARS-CoV-2 is frequently transmitted in an airborne manner. As such, we hypothesize that SARS-CoV-2 might need a higher infectious dose for airborne transmission than measles, although the infectious dose for SARS-CoV-2 is largely unknown. The recent paper reviewed the common “myths” on the airborne transmission to clarify and dispel them based on the scientific updated data (30).
Therefore, the importance of mask-wearing cannot be overemphasized because mask-wearing likely reduces the number of infectious particles for outward transmission, and even the wearing of less efficient masks may also reduce the inhaled number of infectious particles in inward transmission. In this context, our previous data on the superiority of highly efficient masks such as KF94 masks over surgical masks in reducing outward viral load (31) suggest that wearing more efficient masks would be more helpful to reduce the community transmission, especially in terms of outward blocking of SARS-CoV-2. However, a well-designed experimental study showed that the outward reduction of SARS-CoV-2 load was not significantly different depending on the type of masks (32). Therefore, further experimental and epidemiologic studies are needed on which types of masks may be more useful in reducing the outward and inward transmission of SARS-CoV-2.
SSEs
Previous studies on the SARS pandemic reported that about 20% of infected individuals were responsible for 80% of transmission (33). During the MERS outbreak in South Korea, a 35-year-old man stayed at the emergency room in a tertiary hospital for 58 h and caused 82 secondary cases (34). Similarly, a recent study on COVID-19 reported that whereas about 70% of patients with SARS-CoV-2 infection do not lead to secondary cases, 10% to 20% of patients induce SSEs and account for 80% of all SARS-CoV-2 transmissions (35). The factors of COVID-19 that cause SSEs are yet unclear, but it can be assumed that the important characteristics of transmission described above as well as some host factors might contribute to the SSEs. For example, active social gathering of presymptomatic or asymptomatic individuals, a large degree of aerosol production by singing or talking without masks, and airborne transmission by a high cumulative number of aerosols enough to infect a large number of individuals in under-ventilated areas are likely to lead to SSEs, and thus emphasize the importance of reducing the numbers of contacts by limiting the number of social gatherings.
CONCLUSIONS
The above-mentioned transmission characteristics of SARS-CoV-2 are leading to the failure of traditional quarantine measures learned from SARS or MERS outbreaks. Early identification and isolation with extensive contact tracing may be possible and effective only against the countable number of SARS-CoV-2 cases entering through immigration. Conversely, the unrealistically massive testing for more than millions of people during a period of few weeks and subsequent isolation shown in China might be effective against limited importation into the community (36). Otherwise, the transmission characteristics of SARS-CoV-2 that makes it difficult to be under control would inevitably allow undetected cases to cause further transmission. This has led many European countries and many of the states in the USA to eventually go into lockdown to control the spread of this highly transmissible virus. Unfortunately, lockdown is not a long-lasting quarantine measure, so only a highly effective vaccine can bring the communities under control from this deadly multifaceted virus.
ACKNOWLEDGMENTS
This review was supported by a grant from the Korea Advanced Research Program through the National Research Foundation of Korea (NRF), which is funded by the Ministry of Science and ICT, Republic of Korea (grant No. 2020M3H8A1115041).
Abbreviations
- COVID-19
coronavirus disease 2019
- MERS
Middle East respiratory syndrome
- MERS-CoV
Middle East respiratory syndrome coronavirus
- SARS
severe acute respiratory syndrome
- SARS-CoV-2
severe acute respiratory syndrome coronavirus 2
- SSE
super-spread event
Footnotes
Conflict of Interest: The authors declare no potential conflicts of interest.
- Conceptualization: Lim JS, Kim SH.
- Formal analysis: Bae S, Lim JS, Kim JY, Jung J, Kim SH.
- Funding acquisition: Kim SH.
- Investigation: Kim SH.
- Methodology: Kim JY, Jung J.
- Supervision: Kim SH.
- Validation: Bae S.
- Writing - original draft: Kim SH.
- Writing - review & editing: Bae S, Lim JS, Kim JY, Jung J.
References
- 1.Peiris JS, Chu CM, Cheng VC, Chan KS, Hung IF, Poon LL, Law KI, Tang BS, Hon TY, Chan CS, et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003;361:1767–1772. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Oh MD, Park WB, Choe PG, Choi SJ, Kim JI, Chae J, Park SS, Kim EC, Oh HS, Kim EJ, et al. Viral load kinetics of MERS coronavirus infection. N Engl J Med. 2016;375:1303–1305. doi: 10.1056/NEJMc1511695. [DOI] [PubMed] [Google Scholar]
- 3.Arons MM, Hatfield KM, Reddy SC, Kimball A, James A, Jacobs JR, Taylor J, Spicer K, Bardossy AC, Oakley LP, et al. Presymptomatic SARS-CoV-2 infections and transmission in a skilled nursing facility. N Engl J Med. 2020;382:2081–2090. doi: 10.1056/NEJMoa2008457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wei WE, Li Z, Chiew CJ, Yong SE, Tho MP, Lee VJ. Presymptomatic transmission of SARS-CoV-2 — Singapore, January 23–March 16, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:411–415. doi: 10.15585/mmwr.mm6914e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.WHO. Transmission of SARS-CoV-2: implications for infection prevention precautions [Internet] [accessed on 31 December 2020]. Available at https://www.who.int/news-room/commentaries/detail/transmission-of-sars-cov-2-implications-for-infection-prevention-precautions.
- 6.Li F, Li YY, Liu MJ, Fang LQ, Dean NE, Wong GW, Yang XB, Longini I, Halloran ME, Wang HJ, et al. Household transmission of SARS-CoV-2 and risk factors for susceptibility and infectivity in Wuhan: a retrospective observational study. Lancet Infect Dis. 2021:S1473-3099(20)30981-6. doi: 10.1016/S1473-3099(20)30981-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Buitrago-Garcia D, Egli-Gany D, Counotte MJ, Hossmann S, Imeri H, Ipekci AM, Salanti G, Low N. Occurrence and transmission potential of asymptomatic and presymptomatic SARS-CoV-2 infections: a living systematic review and meta-analysis. PLoS Med. 2020;17:e1003346. doi: 10.1371/journal.pmed.1003346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Long QX, Tang XJ, Shi QL, Li Q, Deng HJ, Yuan J, Hu JL, Xu W, Zhang Y, Lv FJ, et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat Med. 2020;26:1200–1204. doi: 10.1038/s41591-020-0965-6. [DOI] [PubMed] [Google Scholar]
- 9.Lee S, Kim T, Lee E, Lee C, Kim H, Rhee H, Park SY, Son HJ, Yu S, Park JW, et al. Clinical course and molecular viral shedding among asymptomatic and symptomatic patients with SARS-CoV-2 infection in a community treatment center in the Republic of Korea. JAMA Intern Med. 2020;180:1447. doi: 10.1001/jamainternmed.2020.3862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ra SH, Lim JS, Kim GU, Kim MJ, Jung J, Kim SH. Upper respiratory viral load in asymptomatic individuals and mildly symptomatic patients with SARS-CoV-2 infection. Thorax. 2021;76:61–63. doi: 10.1136/thoraxjnl-2020-215042. [DOI] [PubMed] [Google Scholar]
- 11.Johansson MA, Quandelacy TM, Kada S, Prasad PV, Steele M, Brooks JT, Slayton RB, Biggerstaff M, Butler JC. SARS-CoV-2 transmission from people without COVID-19 symptoms. JAMA Netw Open. 2021;4:e2035057. doi: 10.1001/jamanetworkopen.2020.35057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cao S, Gan Y, Wang C, Bachmann M, Wei S, Gong J, Huang Y, Wang T, Li L, Lu K, et al. Post-lockdown SARS-CoV-2 nucleic acid screening in nearly ten million residents of Wuhan, China. Nat Commun. 2020;11:5917. doi: 10.1038/s41467-020-19802-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hui DS, Memish ZA, Zumla A. Severe acute respiratory syndrome vs. the Middle East respiratory syndrome. Curr Opin Pulm Med. 2014;20:233–241. doi: 10.1097/MCP.0000000000000046. [DOI] [PubMed] [Google Scholar]
- 14.Memish ZA, Perlman S, Van Kerkhove MD, Zumla A. Middle East respiratory syndrome. Lancet. 2020;395:1063–1077. doi: 10.1016/S0140-6736(19)33221-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim GU, Kim MJ, Ra SH, Lee J, Bae S, Jung J, Kim SH. Clinical characteristics of asymptomatic and symptomatic patients with mild COVID-19. Clin Microbiol Infect. 2020;26:948.e1–948.e3. doi: 10.1016/j.cmi.2020.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sung HK, Kim JY, Heo J, Seo H, Jang YS, Kim H, Koh BR, Jo N, Oh HS, Baek YM, et al. Clinical course and outcomes of 3,060 patients with coronavirus disease 2019 in Korea, January-May 2020. J Korean Med Sci. 2020;35:e280. doi: 10.3346/jkms.2020.35.e280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yu IT, Li Y, Wong TW, Tam W, Chan AT, Lee JH, Leung DY, Ho T. Evidence of airborne transmission of the severe acute respiratory syndrome virus. N Engl J Med. 2004;350:1731–1739. doi: 10.1056/NEJMoa032867. [DOI] [PubMed] [Google Scholar]
- 18.Olsen SJ, Chang HL, Cheung TY, Tang AF, Fisk TL, Ooi SP, Kuo HW, Jiang DD, Chen KT, Lando J, et al. Transmission of the severe acute respiratory syndrome on aircraft. N Engl J Med. 2003;349:2416–2422. doi: 10.1056/NEJMoa031349. [DOI] [PubMed] [Google Scholar]
- 19.Wong TW, Lee CK, Tam W, Lau JT, Yu TS, Lui SF, Chan PK, Li Y, Bresee JS, Sung JJ, et al. Cluster of SARS among medical students exposed to single patient, Hong Kong. Emerg Infect Dis. 2004;10:269–276. doi: 10.3201/eid1002.030452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Roy CJ, Milton DK. Airborne transmission of communicable infection--The elusive pathway. N Engl J Med. 2004;350:1710–1712. doi: 10.1056/NEJMp048051. [DOI] [PubMed] [Google Scholar]
- 21.van Doremalen N, Bushmaker T, Munster VJ. Stability of Middle East respiratory syndrome coronavirus (MERS-CoV) under different environmental conditions. Euro Surveill. 2013;18:20590. doi: 10.2807/1560-7917.es2013.18.38.20590. [DOI] [PubMed] [Google Scholar]
- 22.Bin SY, Heo JY, Song MS, Lee J, Kim EH, Park SJ, Kwon HI, Kim SM, Kim YI, Si YJ, et al. Environmental contamination and viral shedding in MERS patients during MERS-CoV outbreak in South Korea. Clin Infect Dis. 2016;62:755–760. doi: 10.1093/cid/civ1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kim SH, Chang SY, Sung M, Park JH, Kim HB, Lee H, Choi JP, Choi WS, Min JY. Extensive viable Middle East respiratory syndrome (MERS) coronavirus contamination in air and surrounding environment in MERS isolation wards. Clin Infect Dis. 2016;63:363–369. doi: 10.1093/cid/ciw239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.van Doremalen N, Bushmaker T, Morris DH, Holbrook MG, Gamble A, Williamson BN, Tamin A, Harcourt JL, Thornburg NJ, Gerber SI, et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med. 2020;382:1564–1567. doi: 10.1056/NEJMc2004973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhang R, Li Y, Zhang AL, Wang Y, Molina MJ. Identifying airborne transmission as the dominant route for the spread of COVID-19. Proc Natl Acad Sci U S A. 2020;117:14857–14863. doi: 10.1073/pnas.2009637117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Park SY, Kim YM, Yi S, Lee S, Na BJ, Kim CB, Kim JI, Kim HS, Kim YB, Park Y, et al. Coronavirus disease outbreak in call center, South Korea. Emerg Infect Dis. 2020;26:1666–1670. doi: 10.3201/eid2608.201274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shen Y, Li C, Dong H, Wang Z, Martinez L, Sun Z, Handel A, Chen Z, Chen E, Ebell MH, et al. Community outbreak investigation of SARS-CoV-2 transmission among bus riders in Eastern China. JAMA Intern Med. 2020;180:1665–1671. doi: 10.1001/jamainternmed.2020.5225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kwon KS, Park JI, Park YJ, Jung DM, Ryu KW, Lee JH. Evidence of long-distance droplet transmission of SARS-CoV-2 by direct air flow in a restaurant in Korea. J Korean Med Sci. 2020;35:e415. doi: 10.3346/jkms.2020.35.e415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hwang SE, Chang JH, Oh B, Heo J. Possible aerosol transmission of COVID-19 associated with an outbreak in an apartment in Seoul, South Korea, 2020. Int J Infect Dis. 2020;104:73–76. doi: 10.1016/j.ijid.2020.12.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tang JW, Bahnfleth WP, Bluyssen PM, Buonanno G, Jimenez JL, Kurnitski J, Li Y, Miller S, Sekhar C, Morawska L, et al. Dismantling myths on the airborne transmission of severe acute respiratory syndrome coronavirus (SARS-CoV-2) J Hosp Infect. 2021:S0195-6701(21)00007-4. doi: 10.1016/j.jhin.2020.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kim MC, Bae S, Kim JY, Park SY, Lim JS, Sung M, Kim SH. Effectiveness of surgical, KF94, and N95 respirator masks in blocking SARS-CoV-2: a controlled comparison in 7 patients. Infect Dis (Lond) 2020;52:908–912. doi: 10.1080/23744235.2020.1810858. [DOI] [PubMed] [Google Scholar]
- 32.Ueki H, Furusawa Y, Iwatsuki-Horimoto K, Imai M, Kabata H, Nishimura H, Kawaoka Y. Effectiveness of face masks in preventing airborne transmission of SARS-CoV-2. mSphere. 2020;5:e00637-20. doi: 10.1128/mSphere.00637-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shen Z, Ning F, Zhou W, He X, Lin C, Chin DP, Zhu Z, Schuchat A. Superspreading SARS events, Beijing, 2003. Emerg Infect Dis. 2004;10:256–260. doi: 10.3201/eid1002.030732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Oh MD, Park WB, Park SW, Choe PG, Bang JH, Song KH, Kim ES, Kim HB, Kim NJ. Middle East respiratory syndrome: what we learned from the 2015 outbreak in the Republic of Korea. Korean J Intern Med. 2018;33:233–246. doi: 10.3904/kjim.2018.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Adam DC, Wu P, Wong JY, Lau EH, Tsang TK, Cauchemez S, Leung GM, Cowling BJ. Clustering and superspreading potential of SARS-CoV-2 infections in Hong Kong. Nat Med. 2020;26:1714–1719. doi: 10.1038/s41591-020-1092-0. [DOI] [PubMed] [Google Scholar]
- 36.Xing Y, Wong GW, Ni W, Hu X, Xing Q. Rapid response to an outbreak in Qingdao, China. N Engl J Med. 2020;383:e129. doi: 10.1056/NEJMc2032361. [DOI] [PMC free article] [PubMed] [Google Scholar]