Abstract
Bacillus Calmette-Guerin (BCG) vaccine is one of the most widely used vaccines in the world. It protects against many non-mycobacterial infections secondary to its nonspecific immune effects. The mechanism for these effects includes modification of innate and adaptive immunity. The alteration in innate immunity is through histone modifications and epigenetic reprogramming of monocytes to develop an inflammatory phenotype, a process called “trained immunity.” The memory T cells of adaptive immunity are also responsible for resistance against secondary infections after administration of BCG vaccine, a process called “heterologous immunity.” Bacillus Calmette-Guerin vaccine is known to not only boosts immune responses to many vaccines when they are co-administered but also decrease severity of these infections when used alone. The BCG vaccine by itself induces a TH1 type response, and its use as a vector has also shown promising results. This review article summarizes the studies showing effects of BCG vaccines on various viral infections, its role in enhancing vaccine responses, the mechanisms for this protective effect, and information on its effect on COVID-19.
Key Indexing Terms: Bacillus Calmette-Guerin vaccine, Recombinant BCG vaccine, Viral infections, Heterologous immunity, Trained immunity, Influenza
Introduction
A vaccine is a biological preparation that stimulates the body's immune system to recognize it as foreign, destroy it, and remember it so that the immune system can easily destroy it at any later encounters. Historically, vaccines have been known to have many non-disease- specific effects; for example, Vaccinia virus vaccine in the 17th century was noted to protect not only against small pox but also measles, scarlet fever, and syphilis. Bacillus Calmette-Guerin (BCG) vaccine is a live attenuated vaccine derived from Mycobacterium bovis. It is the most widely used vaccine in the world, and it reduces the risk of tuberculosis by approximately 50%.1 However, it also has been shown to decrease all cause neonatal mortality in several randomized trials done in West Africa, India, and Haiti.2 , 3 This effect is due to the prevention of neonatal sepsis and various upper respiratory tract infections (URTI) in these countries. BCG also has nonspecific protective effects against protozoan infections, including tegumentary leishmaniasis and malaria,4 and in animal models has provided protection against a diverse group of bacteria (Staphylococcus aureus, Mycobacterium fortuitum,5 Klebsiella pneumonia,6 Salmonella enteritidis), and viral infections.7 In addition to its effect on infectious diseases, BCG can induce potent nonspecific effects in the treatment of bladder cancer in humans8 and in topical therapy for common and genital warts and reduces the growth of subcutaneously transplanted tumors in mice.9, 10, 11
In this review article we aim to discuss the pathophysiologic mechanism for these effects and also the use of BCG in COVID-19 infection. It is important to understand that the BCG vaccine persists in skin for no more than 1 month and any protection against tuberculosis or any unrelated pathogen after that time period is due to modulation of immunity by this vaccine. The nonspecific effects of BCG vaccine in humans and animal models have been attributed to the two immunological mechanisms discussed below.
Heterologous immunity
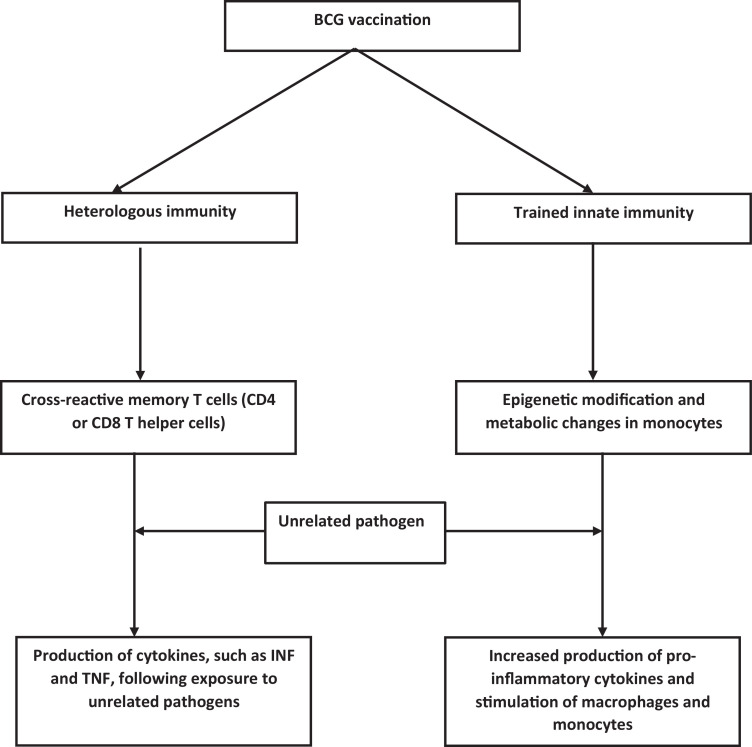
Adaptive immunity works through the interaction of T cells receptors (TCR) with an antigens presented by one of the antigen presenting cells (APC) coupled to a Major Histocompatibility Complex (MHC). The mechanism of adaptive immunity is specific, and this interaction leads to the production of memory T cells during the interaction of T cells with APC. The memory T cell produced during initial interaction of BCG epitopes cross react with unrelated antigens. Heterologous immunity implies that even after primary infection is over in response to exposure to an unrelated pathogen there is an enhanced and rapid production of the Th1/Th17 cytokines interferon-γ (IFN-γ) and tumor necrosis factor (TNF) which help clear the unrelated pathogen more quickly.12 This degenerate nature of T cell recognition in which an antigenic peptide is presented via MHC to T-cell receptor (TCR) enhances the probability of cross-reactivity to an unrelated pathogen is called heterologous immunity. These cross-reactive memory cells can be CD4 or CD8 T helper cells. The cross reactive T cells have developed between antigens of influenza and Epstein-Barr virus, hepatitis C (HCV) and human immunodeficiency virus (HIV), and human corona virus and Mycobacterium tuberculosis. This effect is clinically more obvious when these responses are produced in excess; for example, HCV-induced hepatitis or dengue virus-induced hemorrhagic fever are examples of this heterologous immunity.12
This concept of BCG vaccination resulting in immunity to unrelated pathogens was seen in vaccinia virus and herpes simplex virus type 2 (HSV-2) infections in murine studies through a mechanism dependent on increased T-cell receptor signaling and production of IFN-γ by memory CD4 T cells. Mice were immunized with BCG vaccine and were subsequently treated with isoniazid and rifampin to clear the bacteria from circulation. Mathurin et al. then tested the resting immunity in absence of any ongoing infection by challenging BCG immunized mice with Vaccinia virus and noted that even more than 6 months of BCG vaccination, memory CD4 were activated in mice in response to Vaccinia virus challenge and produced IFN-γ in substantial titers to give immunity to BCG immunized mice against Vaccinia virus.13 Thus, there was the creation of a memory T lymphocyte repertoire which was subsequently activated long after BCG immunization to provide benefits to other nonspecific infections. The mechanism in the study was postulated to be because of cross reactivity of T cell MHC to react with similar epitopes of Vaccinia virus. However, the study could not rule out the possibility of nonspecific activation of the memory T cells cytokines, such as IL-12 and IL-18 produced during secondary infections, independent of TCR signaling. More studies are needed to find the source of activation of these memory T cells, because even though the epitope spectrum of BCG and M tuberculosis is large with at least 4000 proteins, it is hard to speculate BCG epitopes being shared with all the pathogens described above.14 However, the end result remains that the memory cell generated in the initial encounter gets activated in the presence of a secondary unrelated infection by an unknown mechanism to produce Th1/Th17 cytokines to combat secondary infection. Another example of this immunity involves the administration of BCG vaccine for treatment of condyloma acuminatum and warts. Activation of CD 4 lymphocytes leads to increased TNF-α production which downregulates gene transcription of HPV.15
There are several questions which remain unanswered. This type of immunity cannot explain why the antiviral activity induced by BCG vaccine was more effective against localized dermal lesions than systemic involvement in HSV-2 infection, such as herpes encephalitis. Also, as noted previously the non-specific benefits f BCG vaccine against URTI were mostly found in studies done in developing countries, and these differences were not apparent in studies performed in Europe. It raises the question of whether these effects are dependent on socioeconomic status or geographical location or other factors related to infections and host defences (Fig. 1 ).16, 17
Fig. 1.
This figure outlines the possible changes in immunity following Bacillus Calmette-Guerin (BCG) vaccination.
Trained immunity
Heterologous immunity takes couple of weeks to develop it and could not explain the rapid decrease of perinatal mortality seen in several studies. Therefore, a second mechanism for the nonspecific effects of BCG involving innate immunity was thought to contribute to rapid effects. Innate immunity comprises of neutrophils, natural killer (NK) cells, dendritic cells, and monocytes/macrophages. However, neutrophils are terminally differentiated and short lived, leaving dendritic cells, monocytes, and NK cells to be capable of forming immunological memory called trained immunity. This trained immune memory is nonspecific and protects against unrelated secondary infections independent of T and B cell responses.18
Kleinnijenhuis et al. showed that monocytes can be functionally reprogrammed to induce protective effect against other infections.19 Mice lacking CD4 and CD8 cells were vaccinated with BCG and subsequently infected with lethal Candida albicans. These mice showed decreased mortality when compared to sham mice. Bacillus Calmette-Guerin vaccinated mice have an increase in monocyte population one-month post vaccination with changes in their phenotype leading to increased pro-inflammatory cytokine production and increased expression of pattern recognition receptors. These mice had increased methylation of histone 3 at lysine 4 (H3K4), a mechanism of epigenetic reprogramming at the promoter site that is associated with increased transcription of pro-inflammatory cytokine genes in monocytes. The genomic region associated with epigenetic reprogramming was upstream of the gene coding for nucleotide-binding oligomerization domain containing protein 2 (NOD2), which is the innate immune receptor-pattern recognition receptor recognizing muramyl dipeptide (MDP), a bacterial cell wall component, indicating that upregulation of NOD2 may also be responsible for training monocytes.19
In 2018, Arts et al. did a human study to see the effects of BCG in vaccinated volunteers and their response to an unrelated virus by administering yellow fever vaccine and subsequently comparing the viremia in BCG-immunized versus non immunized volunteers.20 Volunteers with prior BCG vaccination had significantly lower levels of viremia than volunteers vaccinated with placebo, even after three months, which was attributed to increased active chromatin state at gene promoters and enhancers of monocytes and increased pro-inflammatory cytokine production (IL-1B, TNF-α, IL-6) in this group. Arts et al. also showed that the PI3K/AKT pathway, another pathway for mediating immunity, was upregulated, and this was supported by the recent studies demonstrating its involvement in the induction of trained immunity by both BCG and b-glucan.20 These epigenetic changes in trained immune cell require changes in physiological activity with switching from oxidative phosphorylation to aerobic glycolysis with decreased O2 requirements, also known as the Warburg effect. It occurs in these trained inflammatory cells as an adaptation to work efficiently in hypoxic environments.21
In 2013, Kleinnijenhuis et al. tried to study the duration of these protective effects and found that at the end of one year heterologous immunity to an unrelated antigen was still active but cytokine production by monocytes in response to an unrelated pathogen decreased.19 However, there was still an increased expression of pattern recognition receptors on monocytes, thus concluding that long lasting nonspecific benefits of BCG vaccine were mainly because of heterologous immunity with some residual trained immunity long after the disappearance of BCG.
Enhanced vaccine responses
There were several studies done in 2002 to determine the effect of BCG vaccines on other vaccines in neonates based on the observations of decreased mortality in this population.22 BCC vaccine administered at birth augmented the pneumococcal vaccine titers and the response to hepatitis B vaccine and increased cytokines in response to tetanus toxoids at two months. The mechanisms underlying the influence of BCG on the priming of antibody responses to unrelated vaccine antigens likely involves enhanced activation of T lymphocytes by APC and markedly enhanced memory cell at 4.5 months. To determine the effect of BCG vaccine in adults, BCG immunized individuals were given the influenza vaccine 14 days after BCG immunization in a randomized controlled trial.23 It was found that BCG vaccinated subjects had markedly enhanced hemagglutinin-inhibition antibody responses against the H1N1/09 vaccine strain compared to the placebo-treated group, and there was sustained IL-B response (a marker of Th1 activation). The effects of non-live typhoid fever vaccine with BCG co-administration were also studied by Blok et al., and there was reduced production of IL-10 (an anti-inflammatory cytokine) and increased IL-22 by the typhoid fever vaccine suggesting that BCG produces a pro-inflammatory immune response instead of primary immune suppressive effect of typhoid fever vaccine.24 It was postulated that this opposing effect of BCG was due to its capacity to induce trained immunity and thus counteract immune tolerance. However, the study did not show any increase in humoral or cellular responses to Salmonella typhi.29 Similar to the typhoid vaccine, other inactivated vaccines like the influenza vaccine have an overall immune suppressive effect, especially in women, and BCG vaccination influenced the influenza vaccination-induced increase in TNF-α and IL-6 production after lipopolysaccharide stimulation. The beneficial effect of BCG on these vaccines makes it plausible for a role against these infections.
Reduction in severity of infection
Research done in 2017 showed that the BCG vaccine can actually decrease the severity of influenza infection. Influenza A virus infection targets the lung epithelial cells. Following infection these cells become apoptotic and necrotic and trigger excessive inflammation and cytokine storm. Alveolar macrophages reduce lung inflammation by ingesting apoptotic cells through a process called efferocytosis. Mukherjee et al. demonstrated that intranasal immunization with BCG increased efferocytosis by alveolar phagocytes and conferred protection against lethal influenza A virus pneumonia.25 They introduced the lethal influenza A virus two days after intranasal BCG delivery and found that all mice with prior BCG vaccination survived compared to no survivors in control animals. Since the mice were challenged with the virus only two days after BCG, this response demonstrated that the BCG strengthened innate immunity in lung. Subsequent viral loads were higher in control mice compared to BCG-immunized mice due to increased efferocytosis of influenza A virus infected apoptotic epithelial cell by alveolar phagocytes. Delivery of BCG into the alveolar space increased the expression of T-cell immunoglobulin and mucin domain containing-4 receptors and other phosphatidylserine receptors on macrophages. This study also showed that alveolar phagocytes from BCG-treated mice had significantly higher levels of peroxisome proliferator-activated nuclear receptors (PPARs) that have a role in inflammation and immune regulating properties leading to increased efferocytosis.30 This might suggest the use of BCG in patients with lung diseases with cells damaged by various pathogenetic mechanisms in which increased efferocytosis might decrease lung inflammation.
The Th1 milieu created by the BCG vaccine to control tuberculosis and the later heterologous immunity with non-specific effects were further studied by Wardhana et al.26 They reported that BCG vaccination in elderly subjects increases Th1 cells to produce IFN-γ and IL-2 which are protective against influenza and other acute upper respiratory tract infections, as opposed to the usual Th2 subtype predominance in this age group. Thus, in diseases whose pathophysiology involves increased Th2, such as allergic diseases, asthma, and Crohn's disease in which Th2 lymphocyte responses predominate, BCG vaccination has the potential to reverse this milieu to Th1 mediated cytokines (IL-1, IL-2, IFN-γ, TNF-α). Datau et al. reported that BCG vaccination shifted the Th2 mediated IL-4 milieu to Th1 mediated IFN-γ in asthmatics.27 The inhibition of Th2 responses by the BCG provides the rationale for its use in these diseases and is a current area of research.28
Because of the capacity to induce a T cell response which lasts for many years, recombinant BCG (rBCG) strains with BCG as vector are being used to facilitate viral clearance.29 rBCG technology refers to the genetic engineering of BCG bacteria to use them as a vector to express foreign genes of interest. This involves inserting the DNA encoding an immunogenic antigen (such as a bacterial surface protein) into M bovis bacteria and expressing the antigen in these cells. The rationale for using BCG as a delivery vector for heterologous antigens is based on its adjuvant activity and capacity to replicate inside antigen presenting cells (APC), such as macrophages and dendritic cells. Various antigen genes from M tuberculosis (e.g., Ag85) and other pathogens (e.g., HIV-1 env), tumor-associated antigen genes (e.g., MUC1), and genes that encode specific mammalian cell cytokines (e.g., IFN-γ, IL-2) have been introduced into BCG bacteria to construct rBCG strains that express those genes.
For example, during metapneumoviruses infections, host immunity is unable to generate protective immunological memory, and this allows recurrent infections. Also, the vaccine developed for RSV increased Th2 responses and was not effective. Both these problems can be fixed with recombinant BCG (rBCG) vaccine strains expressing respiratory syncytial viruses (RSV) or human metapneumoviruses as heterologous antigens on BCG vector to provide protection against these infections. The potential benefit with rBCG is also seen with strains of Mycobacterium smegmatis as a vaccine vector for HIV-1 envelope protein which induces CD8 T cells secreting IFN-γ and creates a stable immunological memory.30
BCG vaccination and COVID-19
Covian et al. analyzed the differences in the number of confirmed cases of COVID-19 per million in countries which had a BCG vaccination program and countries which did not.31 The number of confirmed cases was much lower in countries that used BCG immunization at birth. In addition, the number of deaths was lower, and the mortality rate was lower. Madan and colleagues reviewed information available on tuberculosis (TB) incidence, BCG coverage, and COVID-19 incidence in 174 countries. They found that countries with a low TB incidence and a low BCG coverage had the highest incidence of COVID-19 per 100,000 population. Countries with high BCG coverage, countries with a high TB incidence, and countries with high TB incidence and BCG coverage had much lower rates of COVID-19 infection. In addition, the case rate fatality rate appeared to be lower in countries with a high TB incidence with a high BCG coverage rate.32 Hamiel and coworkers studied COVID-19 infection rates in adults in the age range of 35–41 in Israel who presumably had different BCG vaccination rates.33 There was no significant difference in the proportion of positive test results in the BCG vaccinated group (11.7%) compared to the unvaccinated group (10.4%); the rates were 121 per 100,000 in the BCG vaccinated group and 100 per 100,000 in the BCG unvaccinated group. The number of cases with severe disease was small in both groups, and this study does not provide any information about the clinical benefit of BCG vaccination after infection. It is possible that cases with prior vaccination had less severe disease. It is also possible that the long interval between BCG vaccination and COVID-19 infection reduced any potential benefit associated with vaccination.
The rBCG vaccine has been shown to be protective for parasites like Plasmodium malariae and viruses like HCV. The inclusion of the coronavirus 2 spike protein in a recombinant BCG vaccine has a potential to induce specific immunity against the spike protein and increase trained innate immunity. However, the induction of immunity can have potential adverse effects. Tereza et al. analyzed an “optimal” immune response to Mycobacterium tuberculosis and argued that the factors involved in the development of a cavitary tuberculosis included the mycobacterial exposure level, the extent of the adaptive immune response, and the extent of the innate immune response.34 Consequently, in certain situations a vigorous immune response might cause more disease; this possibility greatly increases the complexity of vaccination trials.
Summary
Bacillus Calmette-Guerin vaccine can modify both innate and adaptive immunity and, thereby, provide immunity not only against Mycobacterium tuberculosis but also other pathogens. Heterologous immunity and trained immunity contribute to pathophysiologic mechanisms which explain how a vaccine against Mycobacterium tuberculosis protects against unrelated pathogens. The use of this vaccine as a vector to stimulate a milieu of adaptive response could be explored in the development of COVID vaccine. Randomized control trials are warranted in certain high-risk populations, such as healthcare workers and nursing home residents, to determine if BCG vaccination can reduce the incidence or severity of COVID-19.
Glossary
-
1
Epitope is fragment of an antigen equivalent to 5–15 amino acids or 3–4 sugar residues that reacts with receptors on B-lymphocytes and T-lymphocytes, as well as with free antibody molecules.
-
2
Antigen-presenting cells (APCs) are a heterogeneous group of immune cells that mediate the cellular immune response by processing and presenting antigens for recognition by certain lymphocytes, such as T cells. Classical APCs include dendritic cells, macrophages, Langerhans cells, and B cells.
-
3
Pattern recognition receptors (PRRs) are a group of receptors mainly expressed by antigen presenting cells, such as dendritic cells and macrophages, to recognize extracellular and intracellular invaders. The five PRR families include the Toll-like receptors (TLRs), the nucleotide-binding oligomerization domain (NOD)- Leucin Rich Repeats (LRR)-containing receptors (NLR), the retinoic acid-inducible gene 1 (RIG-1) -like receptors (RLR), and the C-type lectin receptors (CLRs). They usually differ in their ligand recognition, signal transduction, and sub-cellular localization.
-
4
Toll receptors: A type of PRR located on plasma membrane or endosome that recognizes a variety of pathogens like bacteria and work by signaling through a network of protein kinases to kill the pathogen.
-
5
Pathogen associated molecular patterns (PAMP) are molecules with conserved motifs that are associated with pathogen infection that serve as ligands for host pattern recognition molecules.
-
6
Efferocytosis is the process by which apoptotic cells with ‘eat me’ signals are removed by phagocytic cells limited not only to macrophages and dendritic cells but also by many other cell types, including epithelial cells and fibroblasts.
-
7
Cytokines are molecules that are used for cell signaling or cell-to-cell communication. Cytokines are similar to chemokines, wherein they can be used to communicate with neighboring or distant cells about initiating an immune response. Cytokines are also used to trigger cell trafficking, or movement, to a specific area of the body.
-
8
Chemokines are cytokines released by infected cell to initiate an immune response and to signal adjacent cells of the threat.
-
9
Major histocompatibility complex (MHC) is a group of genes that encode for cell surface glycoproteins (MHC molecule) whose main function is to present intracellular peptide fragments to T lymphocytes.
-
10
Innate immunity refers to nonspecific defense mechanisms that react immediately or within hours of an antigen's appearance in the body. The innate immune system includes physical and anatomical barriers as well as effector cells (highly phagocytic motile neutrophils, monocytes and tissue macrophages, eosinophils, and NK cells.), antimicrobial peptides, soluble mediators (cytokines/complement mediators), and cell receptors (PRR).
-
11
Adaptive immunity is characterized by specificity, immunological memory, and self/nonself recognition. The response involves clonal selection of lymphocytes that respond to a specific antigen. Adaptive immune responses are mediated by cellular (lymphocytes) effectors, soluble (cytokines) effectors, and humoral (plasma cell-derived antibodies) effectors.
-
12
Degeneracy (also referred to as cross reactivity or alloreactivity) is a feature of the immune response mechanism that permits effective T cell responses to a large number of potential peptide sequences complexed to MHC molecules with specificity sufficient to distinguish between self and foreign peptides and thus to avoid autoimmune disease.
-
13
Specificity is provided by CD8+ cytotoxic T lymphocytes (CTLs) recognizing 8–10-oligomer amino acid peptides and MHC class I molecular complexes on cells ubiquitously distributed in the body. CD4+ T helper cells (Th) recognize longer peptide fragments bound to MHC class II molecules present only on APC.
-
14
Memory T cells: Following exposure to an antigen, a small subset of effector T cells differentiate into memory cells and remain in peripheral lymphoid and non-lymphoid tissues for years. Memory T cells maintain their antigen specificity and help amplify the immune response during antigen re-exposure.
Financial support
None.
Author Contributions
All authors participated in this project and contributed to the final manuscript.
Conflict of interest statement
The author has no financial or other conflicts of interest to disclose.
References
- 1.Colditz G.A., Brewer T.F., Berkey C.S., et al. Efficacy of BCG vaccine in the prevention of tuberculosis. Meta-analysis of the published literature. JAMA. 1994;271(9):698–702. [PubMed] [Google Scholar]
- 2.Roth A., Gustafson P., Nhaga A., et al. BCG vaccination scar associated with better childhood survival in Guinea-Bissau. Int J Epidemiol. 2005;34(3):540–547. doi: 10.1093/ije/dyh392. [DOI] [PubMed] [Google Scholar]
- 3.Stensballe L.G., Sørup S., Aaby P., et al. BCG vaccination at birth and early childhood hospitalisation: a randomised clinical multicentre trial. Arch Dis Child. 2017;102(3):224–231. doi: 10.1136/archdischild-2016-310760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.dos Santos J.C., Teodoro Silva M.V., Ribeiro-Dias F., et al. Non-specific effects of BCG in protozoal infections: tegumentary leishmaniasis and malaria. Clin Microbiol Infect. 2019;25(12):1479–1483. doi: 10.1016/j.cmi.2019.06.002. [DOI] [PubMed] [Google Scholar]
- 5.Fox A.E., Evans G.L., Turner F.J., et al. Stimulation of nonspecific resistance to infection by a crude cell wall preparation from Mycobacterium phlei. J Bacteriol. 1966;92:1–5. doi: 10.1128/jb.92.1.1-5.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Weiss D.W., Bonhag R.S., Parks J.A. Studies on the heterologous immunogenicity of a methanol-insoluble fraction of attenuated tubercle bacilli (BCG). I antimicrobial protection. J Exp Med. 1964;119:53–70. doi: 10.1084/jem.119.1.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Moorlag S.J.C.F.M., Arts R.J.W., van Crevel R., et al. Non-specific effects of BCG vaccine on viral infections. Clin Microbiol Infect. 2019;25(12):1473–1478. doi: 10.1016/j.cmi.2019.04.020. [DOI] [PubMed] [Google Scholar]
- 8.Sylvester R.J., van der M.A., Lamm D.L. Intravesical bacillus Calmette-Guerin reduces the risk of progression in patients with superficial bladder cancer: a meta-analysis of the published results of randomized clinical trials. J Urol. 2002;168:1964–1970. doi: 10.1016/S0022-5347(05)64273-5. [DOI] [PubMed] [Google Scholar]
- 9.Metawea B., El-Nashar A.R., Kamel I., et al. Application of viable bacilleCalmette-Guerin topically as a potential therapeutic modality in condyloma acuminata: a placebo-controlled study. Urology. 2005;65:247–250. doi: 10.1016/j.urology.2004.09.025. [DOI] [PubMed] [Google Scholar]
- 10.Jaisinghani A.K., Dey V.K., Suresh M.S., et al. Bacillus calmette-guerin immunotherapy for recurrent multiple warts: an open-label uncontrolled study. Indian J Dermatol. 2019;64(2):164. doi: 10.4103/ijd.IJD_558_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Weiss D.W., Bonhag R.S., Deome K.B. Protective activity of fractions of tubercle bacilli against isologoustumours in mice. Nature. 1961;190:889–891. doi: 10.1038/190889a0. [DOI] [PubMed] [Google Scholar]
- 12.Benn C.S., Netea M.G., Selin L.K., et al. A small jab - a big effect: nonspecific immunomodulation by vaccines. Trends Immunol. 2013;34(9):431–439. doi: 10.1016/j.it.2013.04.004. [DOI] [PubMed] [Google Scholar]
- 13.Mathurin K.S., Martens G.W., Kornfeld H., et al. CD4 T-cell-mediated heterologous immunity between mycobacteria and poxviruses. J Virol. 2009;83(8):3528–3539. doi: 10.1128/JVI.02393-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Uthayakumar D., Paris S., Chapat L., et al. Non-specific effects of vaccines illustrated through the BCG example: from observations to demonstrations. Front Immunol. 2018;9:2869. doi: 10.3389/fimmu.2018.02869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Salem A., Nofal A., Hosny D. Treatment of common and plane warts in children with topical viable Bacillus Calmette-Guerin. Pediatr Dermatol. 2013;30:60–63. doi: 10.1111/j.1525-1470.2012.01848.x. [DOI] [PubMed] [Google Scholar]
- 16.Shann F. Nonspecific effects of vaccines and the reduction of mortality in children. Clin Ther. 2013;35:109e14. doi: 10.1016/j.clinthera.2013.01.007. [DOI] [PubMed] [Google Scholar]
- 17.Garly M.L., Martins CL, Bale C., et al. BCG scar and positive tuberculin reaction associated with reduced child mortality in West Africa. A non-specific beneficial effect of BCG? Vaccine. 2003;21(10):2782e90. doi: 10.1016/s0264-410x(03)00181-6. [DOI] [PubMed] [Google Scholar]
- 18.Arts R.J.W., Carvalho A., La Rocca C., et al. Immunometabolic pathways in BCG-induced trained immunity. Cell Rep. 2016;17(10):2562–2571. doi: 10.1016/j.celrep.2016.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kleinnijenhuis J., Quintin J., Preijers F., et al. Epigenetic reprogramming of monocytes by BCG. Proc Natl Acad Sci. 2012;109(43):17537–17542. doi: 10.1073/pnas.1202870109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Arts R.J.W., Moorlag S.J.C.F.M., Novakovic B., et al. BCG vaccination protects against experimental viral infection in humans through the induction of cytokines associated with trained immunity. Cell Host Microbe. 2018;23(1):89–100.e5. doi: 10.1016/j.chom.2017.12.010. [DOI] [PubMed] [Google Scholar]
- 21.Cheng S.C., Quintin J., Cramer R.A., et al. mTOR- and HIF-1α-mediated aerobic glycolysis as metabolic basis for trained immunity. Science. 2014;345(6204) doi: 10.1126/science.1250684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ota M.O.C., Vekemans J., Susanna E., et al. Influence of Mycobacterium bovis Bacillus Calmette-Guerin on antibody and cytokine responses to human neonatal vaccination. J Immunol. 2002;168:919–925. doi: 10.4049/jimmunol.168.2.919. [DOI] [PubMed] [Google Scholar]
- 23.Ritz N., Mui M., Balloch A., et al. Non-specific effect of Bacille Calmette-Guérin vaccine on the immune response to routine immunizations. Vaccine. 2013;31(30):3098–3103. doi: 10.1016/j.vaccine.2013.03.059. [DOI] [PubMed] [Google Scholar]
- 24.Blok B.A., Arts R.J.W., van Crevel R., et al. Differential effects of BCG vaccine on immune responses induced by vi polysaccharide typhoid fever vaccination: an explorative randomized trial. Eur J Clin Microbiol Infect Dis. 2020;39(6):1177–1184. doi: 10.1007/s10096-020-03813-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mukherjee S., Subramaniam R., Chen H., et al. Boosting efferocytosis in alveolar space using BCG vaccine to protect host against influenza pneumonia. PLoS ONE. 2017;12(7) doi: 10.1371/journal.pone.0180143. Published 2017 Jul 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wardhana, Datau E.A., Sultana A., et al. The efficacy of BacillusCalmette-Guerin vaccinations for the prevention of acute upper respiratory tract infection in the elderly. Acta Med Indones. 2011;43:185e90. [PubMed] [Google Scholar]
- 27.Datau E.A., Mewengkang H., Matheos J.C., et al. Clinical efficacy and laboratory improvement of bacillus Calmette Guerin vaccination on adults atopic asthma. World Allergy Organ J. 2008;14:63–69. doi: 10.1097/WOX.0b013e31816c8b85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rousseau M.C., Parent M.E., St-Pierre Y. Potential health effects from non-specific stimulation of the immune function in early age: the example of BCG vaccination. Pediatr Allergy Immunol. 2008;19(5):438–448. doi: 10.1111/j.1399-3038.2007.00669. [DOI] [PubMed] [Google Scholar]
- 29.Bueno S.M., González P.A., Cautivo K.M., et al. Protective T cell immunity against respiratory syncytial virus is efficiently induced by recombinant BCG. Proc Natl Acad Sci USA. 2008;105(52):20822–20827. doi: 10.1073/pnas.0806244105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cayabyab M.J., Hovav A.H., Hsu T., et al. Generation of CD8+ T-cell responses by a recombinant nonpathogenic Mycobacterium smegmatis vaccine vector expressing human immunodeficiency virus type 1 Env. J Virol. 2006;80(4):1645–1652. doi: 10.1128/JVI.80.4.1645-1652.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Covián C., Retamal-Díaz A., Bueno S.M, et al. Could BCG vaccination induce protective trained immunity for SARS-CoV-2? Front Immunol. 2020;11:970. doi: 10.3389/fimmu.2020.00970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Madan M., Pahuja S., Mohan A., et al. TB infection and BCG vaccination: are we protected from COVID-19? Public Health. 2020;185:91–92. doi: 10.1016/j.puhe.2020.05.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hamiel U., Kozer E., Youngster I. SAR-CoV-2 rates in BCG Vaccinated and unvaccinated young adults. JAMA. 2020;323:2340–2341. doi: 10.1001/jama.2020.8189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tezera L.B., Mansour S., Elkington P. Reconsidering the optimal immune response to mycobacterium tuberculosis. Am J Respir Crit Care Med. 2020;201(4):407–413. doi: 10.1164/rccm.201908-1506PP. [DOI] [PMC free article] [PubMed] [Google Scholar]