Abstract
Studies of heart function and metabolism have been used to predict the impact of global warming on fish survival and distribution, and their susceptibility to acute and chronic temperature increases. Yet, despite the fact that hypoxia and high temperatures often co-occur, only one study has examined the effects of hypoxia on fish thermal tolerance, and the consequences of hypoxia for fish cardiac responses to acute warming have not been investigated. We report that sablefish (Anoplopoma fimbria) did not increase heart rate or cardiac output when warmed while hypoxic, and that this response was associated with reductions in maximum O2 consumption and thermal tolerance (CTmax) of 66% and approximately 3°C, respectively. Further, acclimation to hypoxia for four to six months did not substantially alter the sablefish's temperature-dependent physiological responses or improve its CTmax. These results provide novel, and compelling, evidence that hypoxia can impair the cardiac and metabolic response to increased temperatures in fish, and suggest that some coastal species may be more vulnerable to climate change-related heat waves than previously thought. Further, they support research showing that cross-tolerance and physiological plasticity in fish following hypoxia acclimation are limited.
Keywords: cardiac function, hypoxia, thermal tolerance, temperature, haematology, climate change
1. Introduction
More frequent and extreme warming events (i.e. heat waves) are a consequence of climate change [1–3]. The ability of fish to survive exposure to high temperatures depends to a large extent on the capacity of the heart to deliver more O2 to the tissues to meet elevated metabolic demands (i.e. oxygen consumption, ṀO2) [4,5]. For example, heart rate (fH) and cardiac output (Q̇; the amount of blood pumped per minute) increase with temperature, and cardiac collapse [6–10] and neural impairment [11] appear to be key factors in determining the upper thermal limit of fishes. The occurrence and severity of hypoxia are also increasing with climate change [1,12], and hypoxia often coincides with high temperatures in coastal environments [12,13], including at aquaculture cage-sites [14–16]. This could severely limit fish thermal tolerance [17]. This is because low O2 levels result in a regulated decrease in fH (a response known as bradycardia [18]) that, theoretically, may prevent necessary increases in cardiac function (fH and Q̇) when fish are exposed to high temperatures. The effect of hypoxia on thermal tolerance has only been examined in two fish species [17], and this condition's impact on temperature-dependent cardiorespiratory responses is unknown. Further, despite evidence that acclimation (long-term exposure) to hypoxia modifies the cardiorespiratory response to warming at high O2 levels (i.e. under normoxic conditions) considerably [19], there is conflicting information about whether hypoxic acclimation improves thermal tolerance, and vice versa (i.e. that there is ‘cross-tolerance’ between these two oxygen-limiting conditions [19–24]). Specifically, the hypothesis that hypoxia acclimation increases upper thermal tolerance when O2 levels are low has not been experimentally tested. These are important questions given the need to understand how physiological plasticity is related to a fish's tolerance to environmental change, and have implications for fish survival and potential shifts in species' distribution with global warming.
In this study, we conducted two experiments on sablefish (Anoplopoma fimbria) with the goal of providing insights into the potential consequences of hypoxia on the cardiorespiratory response and susceptibility of fish to acute warming events. The sablefish is a widely distributed species across the North Pacific, that lives in coastal waters as juveniles but successfully inhabits oxygen minimum zones (OMZs; areas with O2 levels of less than 2 mg l−1) as adults [25,26]. Given that this species encounters, and tolerates [27], a wide range of temperatures and O2 levels, it is an ideal model in which to test the above questions/hypotheses with respect to the relationship between acute hypoxia, adaptation to hypoxia and thermal tolerance. In the first experiment, we acclimated sablefish to either 100% (normoxia) or 40% air saturation (hypoxia) at 10°C for four to six months. This is a moderate hypoxic level for the sablefish at this temperature, as it is well above the critical and lethal O2 thresholds (15.8% and 5.4% air saturation, respectively) for this species [27], but also low enough to potentially constrain O2 delivery and trigger a plastic response. Then, we performed surgery to allow for in vivo cardiovascular measurements and repeated blood sampling (see below), and recovered both acclimation groups under normoxia for 2 days before exposing them to acute hypoxia (a decrease to 40% air saturation over 2 h) followed by warming (at 2°C h−1) until their critical thermal maximum (CTmax; the temperature at which the fish could no longer maintain equilibrium). Throughout the experiment, we recorded Q̇, and analysed blood O2-carrying capacity (haemoglobin and haematocrit), stress hormone and glucose levels, and lactate (an index of anaerobic metabolism) (electronic supplementary material, figure S1). We also measured ṀO2 using intermittent-flow respirometry, and used ṀO2/Q̇ (the amount of O2 consumed per millilitre of blood pumped) to estimate O2 extraction by the tissues based on rearranging the Fick equation. It should be noted that during warming, 40% air saturation likely becomes a more severe hypoxia level for sablefish, given that a fish's critical O2 threshold (Pcrit) increases with temperature in other species [17,28]. Subsequently, we performed a follow-up experiment involving the same cardiorespiratory measurements, but where warming occurred under normoxia, to better understand the responses observed under hypoxia.
2. Methods
(a). Experimental animals and hypoxia acclimation
Juvenile sablefish (1 g) were acquired from NOAA's Manchester Research Station (Port Orchard, WA, USA) and reared for approximately 1 year at the Dr Joe Brown Aquatic Research Building (JBARB) of MUN in tanks supplied with 10°C seawater with an O2 level of approximately 100% air saturation (157 mmHg; 9.3 mg l−1), and a 12 h light : 12 h dark photoperiod. Procedures used to acclimate the sablefish to hypoxia (approx. 40% air saturation; 63 mmHg; 3.7 mg l−1) have been described previously [29] and are detailed in the electronic supplementary material. The hypoxic acclimation period was four to six months, depending on when the fish were used in the experiments. Water PO2 and temperature in the hypoxic and normoxic tanks during this period were 42.4 ± 0.1% air saturation and 10.2 ± 0.0°C, and 106.2 ± 0.5% air saturation and 10.3 ± 0.0°C, respectively. The fish were fed a marine fish diet (Europa, Skretting) at 0.65% of their body weight day−1, and on days when the hypoxia-acclimated fish did not consume their full ration, the normoxia-acclimated fish were fed the same amount. This ensured that there were no nutritional or morphometric differences between the two experimental groups. The weight (approx. 1320 g), length, condition factor, cardiac and splenic masses of the normoxia- and hypoxia-acclimated sablefish at the time of the experiments were similar (see electronic supplementary material, table S4).
(b). Surgery and recovery
Sablefish were initially anaesthetized by immersion in seawater containing 0.1 g l−1 tricaine methanesulfonate (MS-222; AquaLife TMS, Syndel Laboratories Ltd, Nanaimo, BC, Canada) until ventilatory movements ceased. After weight and fork length were recorded, the fish were transferred to a surgery table where their gills were continuously irrigated with chilled (4°C) and oxygenated seawater containing a maintenance dose of MS-222 (0.05 g l−1). To measure in vivo cardiac function, a Transonic® flow probe (2.0–2.5 S) was placed around the ventral aorta (electronic supplementary material, figure S1), following the procedures described for Atlantic cod [30], but with some modifications for sablefish. To allow for repeated blood sampling, a polyethylene cannula (PE 50; Intramedic®, Becton Dickinson and Co., NJ, USA) filled with heparinized 0.9% saline (100 U ml−1) was subsequently inserted into the afferent branchial artery of the second or third gill arch (electronic supplementary material, figure S1).
Fish were recovered in aerated anaesthetic-free seawater at 12°C. When ventilation commenced, the fish were individually placed into 19.8 l cylindrical respirometers (20.3 cm in diameter × 61.0 cm long) for 42–46 h to recover from surgery under normoxic conditions (electronic supplementary material, figure S1). The respirometers were submersed in a water table supplied with seawater from a temperature- and PO2-controlled reservoir, which initially kept water in the respirometers at 12°C and approximately 100% air saturation. Surgery was always performed on one fish from each acclimation group (the respirometer they were placed into randomized), so that these two fish could be tested in parallel. More details on surgery and recovery are provided in the electronic supplementary material.
(c). Hypoxic warming challenge
On the experimental day, the flow probe leads were connected to the Transonic® flow meter, after which the fish were left to settle for 2 h before performing any measurements. To investigate the cardiorespiratory response to warming under acute hypoxia, the water PO2 was first gradually decreased over the course of 2 h to 40% air saturation. Subsequently, the temperature was increased at 2°C h−1 until the fish reached its CTmax. This rate of heating in CTmax tests is widely used in the field of fish physiology to assess acute thermal tolerance, and is considered to be ecologically relevant (e.g. [17]). Cardiac function and ṀO2 were measured at the following points during the protocol: (i) under normoxia (100% air saturation) at 12°C; (ii) under hypoxia (40% air saturation) at 12°C; and (iii) at every 2°C increase thereafter. Cardiac function and ṀO2 were always recorded simultaneously and measured twice at each time point. The temperature at which the heartbeat became arrhythmic/irregular (Tarrhythmia) was also recorded. Blood (approx. 0.7 ml) was sampled from the cannula immediately after the cardiac function and ṀO2 measurements were taken at four points during the experiment: (i) under normoxia at 12°C; (ii) under hypoxia at 12°C; (iii) under hypoxia at 18°C; (iv) under hypoxia at CTmax. At each sampling, this volume was replaced by an equal volume of 0.9% saline to help maintain the fish's blood volume. The entire protocol took approximately 11 h to complete. Upon reaching their CTmax, fish were euthanized as quickly as possible inside their respirometer (by injecting MS-222 into their respirometer through a tube while in ‘recirculation’ mode: final concentration 0.3 g l−1) [27]. This was important as the CTmax of the paired fish often differed. After removing the fish from their respirometer, correct placement of the flow probe was verified. Then, the heart and spleen were dissected out, rinsed in saline, blotted dry and weighted to determine the relative atrial, ventricular, bulbus and splenic masses [(tissue mass/body mass) × 100].
(d). Measurements of cardiorespiratory function
Cardiac function was recorded at 20 Hz by interfacing the flow meter with an MP100A-CE data acquisition system and a laptop running AcqKnowledge software (BIOPAC Systems Inc., Goleta, CA, USA). We calculated fH (in beats min−1) manually by determining the time required for 20 systolic peaks in the blood flow recording. This time period was also used to measure Q̇ (in ml min−1), which was expressed relative to the fish's body mass (in ml min−1 kg−1). We calculated VS (in ml beat−1 kg−1) as Q̇/fH. Sometimes, small peaks were seen in the recordings (e.g. electronic supplementary material, figure S2). These were not counted as systolic peaks, as they resulted from the Transonic® flow probes picking up ventilatory movements. However, it is interesting that ventilation appeared to remain regular after the heart went arrhythmic (see electronic supplementary material, figure S2d,h), and this is consistent with recent data suggesting that arrhythmias at high temperatures are not caused by nervous dysfunction, but by ionic disturbance at the level of the ventricular myocytes [31]. The Transonic® flow probes were calibrated prior to the experiments (see electronic supplementary material).
We measured ṀO2 (in mg O2 min−1 kg−1) with the automated intermittent-flow respirometry system described previously [27,32]. The durations of the ‘flushing’ and ‘recirculation’ periods were adjusted throughout the protocol, to ensure an R2 > 0.90 for each measurement, and to avoid a decline in PO2 inside the respirometers of more than approximately 5% air saturation. The respirometers were cleaned regularly to prevent background bacterial respiration. This background was considered negligible based on overnight measurements and experiments using respirometers without fish. In these tests, PO2 declined by less than 1% air saturation during the measurement period. We used ṀO2 per unit of Q̇ (ṀO2/Q̇; mg O2 l−1 blood) as a measure of tissue O2 extraction, by rearranging the Fick equation:
where CaO2 and CvO2 are the O2 content of the arterial and venous blood, respectively. We are aware that ṀO2/Q̇ is an indirect measure of O2 extraction and has limitations (i.e. the equation does not account for potential O2 uptake through cutaneous respiration, see [33]), and that for a direct assessment CaO2 and CvO2 would need to be measured. However, this approach is used by various research groups (see [19,34–36]), and we measured blood haemoglobin content so that changes in blood O2-carrying capacity could be accounted for in our interpretation of the data. Blood O2 content was not estimated from haemoglobin content, given that the combined effects of temperature, pH and CO2 on Hb–O2 affinity and maximum saturation of sablefish blood are still unknown. For each fish, the routine and maximum ṀO2 were determined as the average of the ṀO2 measurements at 42–46 h post-surgery, and as the highest ṀO2 recorded during exposure to hypoxic warming, respectively. The scope for ṀO2 was calculated as maximum–routine ṀO2. We are aware that aerobic scope is most commonly determined using swimming-flumes and/or chase protocols, but the use of ‘temperature-induced’ aerobic scope (AST) is appropriate for this thermal tolerance study and provides equivalent data to these traditional methods (e.g. [27,37,38]). Resting, and maximum values, and values for scope for cardiac parameters (fH, Q̇, VS) and ṀO2/Q̇ were determined in the same way as for ṀO2.
(e). Blood sampling and analyses
The collected blood samples were immediately aliquoted for the analysis of various haematological parameters. Blood was first drawn into microhaematocrit tubes and these were centrifuged at 10 000g for 5 min to determine Hct (%). An aliquot of 50 µl of blood was collected for the measurement of blood Hb concentration. Then, the remaining blood was centrifuged for 1 min at 10 000g in a mini-centrifuge (05-090-128, Fisher Scientific) and 300 µl of plasma was pipetted into a 1.5 ml brown (opaque) Eppendorf tube containing 15 µl of 0.2 M EDTA and 15 µl of 0.15 M glutathione for later measurement of circulating catecholamine levels. The rest of the plasma was divided into 50 µl aliquots for the measurement of cortisol, lactate and glucose. The RBC pellet was used for the analysis of RBC protein content. All samples were immediately frozen in liquid N2 and stored at −80°C. See electronic supplementary material for further details on the blood sampling and analyses.
(f). Additional experiment
The primary aim of this experiment was to verify that sablefish have the capacity to enhance fH and Q̇ when warmed under normoxia, like other teleosts [9,10]. The sablefish used for this experiment were acquired from Golden Eagle Sable Fish (Vancouver Island, British Columbia, Canada) as young juveniles, and reared for approximately 1.5 years at the JBARB at the same temperature, PO2 and light regime, and with the same diet and ration, as the sablefish used in the initial experiment. The sablefish were also of a comparable age (less than two months older), and had similar body morphometrics (e.g. the same body mass), although length, condition factor, and atrial and ventricular mass differed (see electronic supplementary material, table S4). The surgical procedures in the additional experiment were exactly the same as in the initial experiment, except that fish were not cannulated, given that this research focused on measuring the fish's cardiac response and the presence/absence of blood sampling did not influence thermal tolerance (electronic supplementary material, figure S3). Furthermore, surgical recovery was only for 1 day (22–23 h) instead of 2 days (42–46 h), because it was shown in the initial experiment that 1 day was adequate for the cardiorespiratory variables to stabilize (see electronic supplementary material). Measurements of cardiac function and ṀO2, and all other procedures, were performed as described for the initial experiment. To maintain normoxia (PO2 at approx. 100% air saturation) throughout the thermal challenge, including the ‘recirculation’ periods of ṀO2 measurements, O2 was bubbled into the water as required using the control system (OXY-REG, Loligo Systems) that was described previously [27]. More details on the additional experiment are provided in the electronic supplementary material.
(g). Data statistical analyses
All statistical analyses were performed using Rstudio v. 1.2.5033 with R v. 3.6.2 unless mentioned otherwise. Cardiorespiratory and haematological data from the initial experiment shown in figures 1, 4 and S4 (electronic supplementary material) were analysed using a general linear mixed model (lmer function), which included fish as a random factor, and acute condition (hypoxia and warming), acclimation condition and their interaction, as fixed effects (see electronic supplementary material for further details). Main effects were assessed using ANOVAs (anova function) with type III sums of squares. If the model indicated a significant fixed effect, then differences between the categories among that effect were analysed using post hoc least-squared means comparisons, with the fdr method (Bonferroni-based) for multiplicity p-value adjustment (emmeans and pairs functions). Cardiorespiratory data from the additional experiment shown in figure 1 were analysed in the same way, except that the model did not include a factor for the effect of acclimation. For the cardiorespiratory, thermal tolerance and morphometric variables shown in figure 2 and electronic supplementary material, figure S3 and tables S3–S5, comparisons were made using two-sided Student's t-tests or two-way ANOVAs, and these analyses were done in GraphPad Prism 8. Statistical significance was set at p < 0.05 and all data are shown as means ± s.e.m.
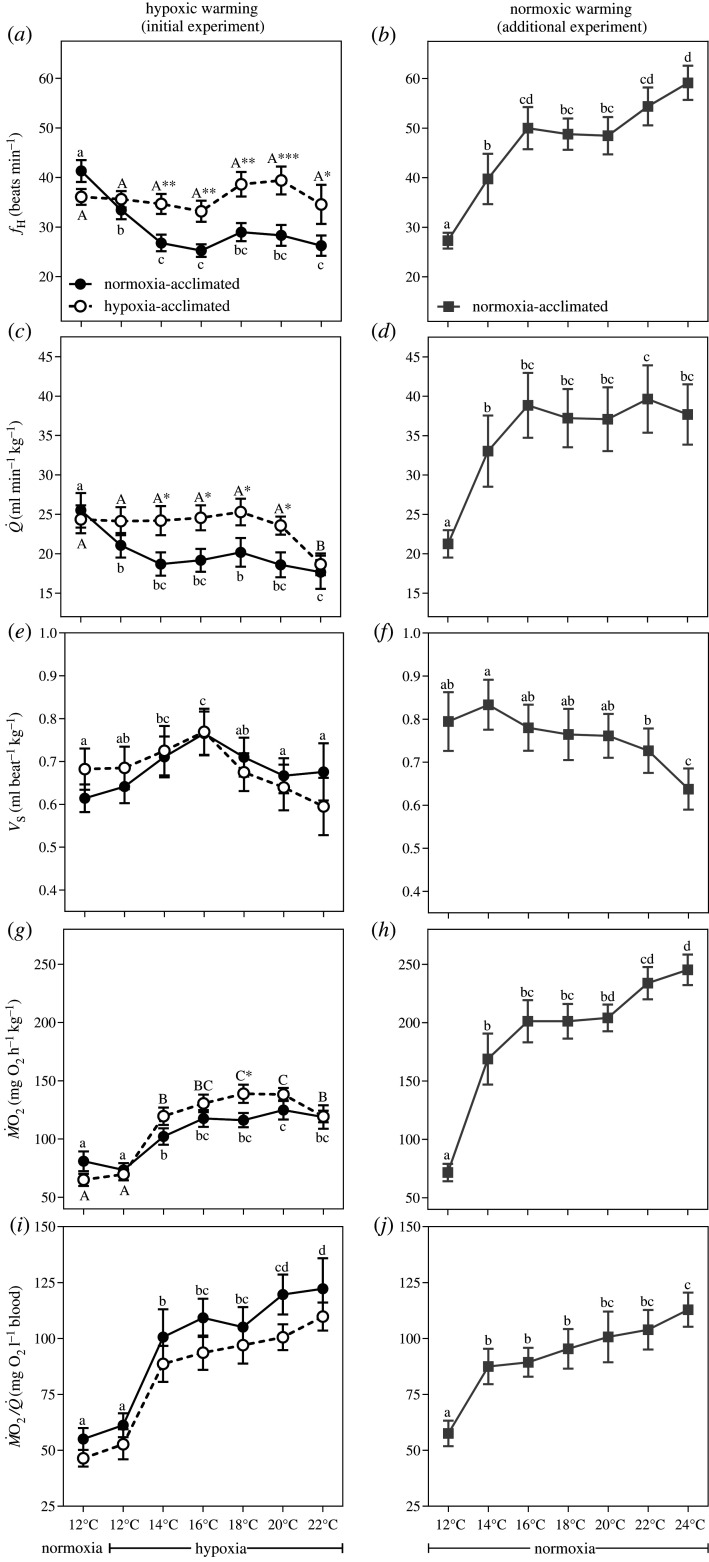
Figure 1.
Cardiorespiratory responses of normoxia- and hypoxia-acclimated sablefish exposed to hypoxic or normoxic warming. Shown are (a,b) heart rate (ƒH), (c,d) cardiac output (Q̇), (e,f) stroke volume (VS), (g,h) O2 consumption (ṀO2) and (i,j) O2 extraction (ṀO2/Q̇). Hypoxic warming, left panels; normoxic warming, right panels. Symbols without a letter in common are significantly different across the sampling points/temperatures (p < 0.05). In the case of a significant acclimation effect and/or acclimation × sampling interaction, lower and uppercase letters indicate differences within the normoxia- and hypoxia-acclimated groups, respectively. Asterisks indicate significant differences between acclimation groups at a particular sampling point (*p < 0.05; **p < 0.01; ***p < 0.001). Values are means ± s.e.m. with n = 14–15 per group for the initial experiment (except at 22°C where n = 9–10 per group, because 4–6 fish had already reached their CTmax), and with n = 9 for the additional experiment (except at 24°C where n = 8, because one fish had already reached its CTmax).
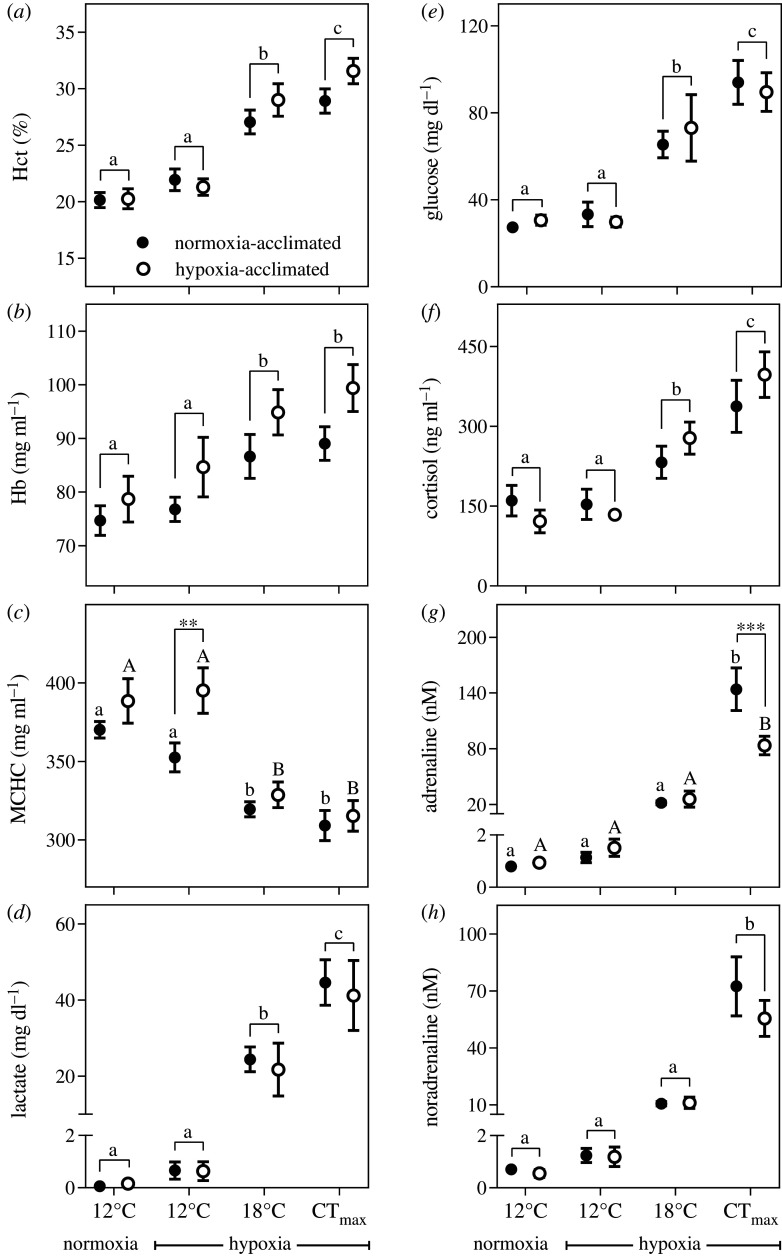
Figure 4.
Haematological changes in normoxia- and hypoxia-acclimated sablefish when exposed to hypoxic warming. Shown are blood/plasma levels of (a) haematocrit (Hct), (b) haemoglobin (Hb), (c) mean cellular haemoglobin concentration (MCHC), (d) lactate, (e) glucose, (f) cortisol, (g) adrenaline and (h) noradrenaline. CTmax, critical thermal maximum. Symbols without a letter in common are significantly different across the sampling points/temperatures (p < 0.05). In the case of a significant acclimation effect and/or acclimation × sampling interaction, lower and uppercase letters indicate differences within the normoxia- and hypoxia-acclimated groups, respectively. Asterisks indicate significant differences between acclimation groups at a particular sampling point (**p < 0.01; ***p < 0.001). Values are means ± s.e.m. with n = 9–10 per group.
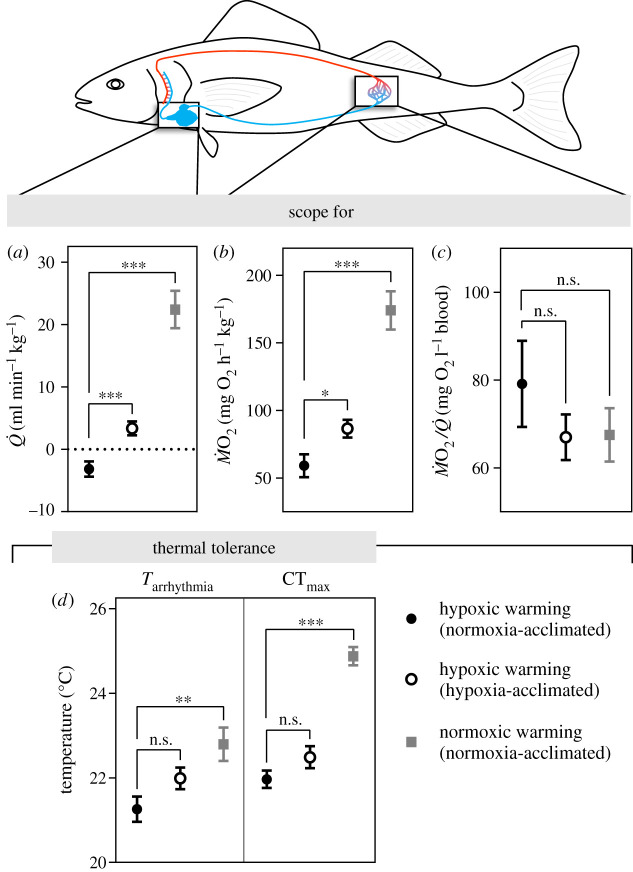
Figure 2.
The capacity of normoxia- and hypoxia-acclimated sablefish to increase cardiorespiratory function (i.e. heart function and oxygen consumption/extraction by the tissues) when exposed to hypoxic and normoxic warming, and parameters related to thermal tolerance. Values for scope were calculated as maximum—resting values for (a) cardiac output (Q̇), (b) O2 consumption (ṀO2) and (c) O2 extraction (ṀO2/Q̇). (d) Parameters for thermal tolerance are the onset temperature of cardiac arrhythmias (Tarrhythmia) and the critical thermal maximum (CTmax). The normoxia-acclimated fish tested under hypoxia are compared to the other two groups (n.s. = p > 0.05; *p < 0.05; **p < 0.01; ***p < 0.001). Values are means ± s.e.m. with n = 14–15 and n = 9 per group for hypoxic and normoxic warming, respectively. (Online version in colour.)
3. Results
(a). Cardiorespiratory response to warming when hypoxic
In normoxia-acclimated sablefish, acute hypoxia resulted in a significant decrease in fH (from 41.3 ± 2.2 to 33.4 ± 1.8 beats min−1, p < 0.01), and surprisingly, fH declined even further during warming (to 25.3 ± 1.3 beats min−1, p < 0.0001) (figure 1a; electronic supplementary material, figure S2 and tables S1 and S2). Stroke volume (VS; the amount of blood pumped per heartbeat) increased slightly with temperature (figure 1e), but this was not sufficient to compensate for the decrease in fH. Thus, Q̇ fell during hypoxia (from 25.5 ± 2.2 to 21.1 ± 1.6 ml min−1 kg−1, p < 0.001) and remained at this level until it declined further at 22°C (i.e. fish approached their CTmax) (figure 1c); this response resulting in a negative scope for Q̇ (figure 2a; electronic supplementary material, table S3). Hypoxia-acclimated sablefish did not experience bradycardia when exposed to acute hypoxia at 12°C (figure 1a; electronic supplementary material, figure S2 and tables S1 and S2), and this likely contributed to their higher aerobic scope (AST; maximum–routine ṀO2) when warmed (p < 0.05; figure 2b; electronic supplementary material, table S3). Nonetheless, fH and Q̇ also failed to increase with temperature in this group (figure 1a,c), and hypoxia acclimation did not change the temperature at which cardiac arrhythmias began (Tarrhythmia, approx. 21.6°C) or CTmax (approx. 22.2°C) (p = 0.078 and p = 0.124, respectively) (figure 2d).
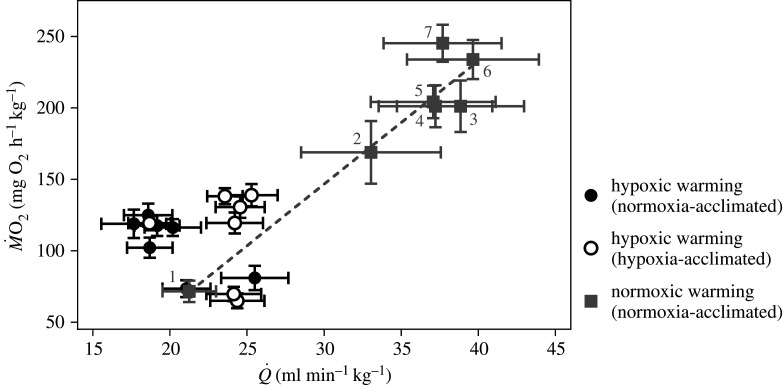
There was an unusual relationship between ṀO2 and Q̇ in sablefish exposed to hypoxic warming (figure 3). Normally, ṀO2 and Q̇ are positively correlated in fishes (e.g. [19]). However, we show that as ṀO2 increased, Q̇ either declined (in normoxia-acclimated fish) or remained the same (in hypoxia-acclimated fish) (figure 1c,g), and that there was no significant (negative or positive) correlation between ṀO2 and Q̇ in the normoxia- and hypoxia-acclimated groups (p = 0.064 and p = 0.884, respectively; figure 3). To raise ṀO2 during hypoxic warming without increasing Q̇, both groups relied solely, and to the same extent, on enhanced ṀO2/Q̇, which increased from approximately 45–55 to 115–135 mg O2 l−1 blood pumped (p < 0.0001; figure 1i; electronic supplementary material, table S3). This was partially mediated by increases in blood haemoglobin levels (by 25%; figure 4a,b), which occurred despite cell swelling that resulted in a decrease in the mean cellular haemoglobin concentration (MCHC; figure 4c) and RBC protein levels (electronic supplementary material, figure S4). However, it is clear that the enhancement of ṀO2/Q̇ was primarily due to augmented O2 uptake by the tissues. Further, while hypoxia-acclimated sablefish had significantly lower plasma levels of adrenaline at CTmax, other stress hormone (cortisol and noradrenaline) and glucose levels increased during warming to a similar degree in both groups (figure 4f–h). The capacity for anaerobic metabolism was considerable in this species (plasma lactate increased from less than 0.2 to approx. 45 mg dl−1) and was initiated at 18°C; indicating that the sablefish experienced insufficient O2 delivery well before its CTmax. However, this parameter was also not affected by hypoxia acclimation (figure 4d).
Figure 3.
The relationship between ṀO2 and Q̇ in normoxia- and hypoxia-acclimated sablefish exposed to hypoxic and normoxic warming. No significant linear regression could be fitted to the data for the normoxia and hypoxia acclimation groups tested under hypoxia (p = 0.064 and p = 0.884, respectively), whereas a significant linear regression was fitted to the data for the fish tested under normoxia (p < 0.001, r2 = 0.918, y = 8.6x–112.0). The symbols for the latter group are labelled with grey numbers, which indicate the following conditions: 1, 12°C; 2, 14°C; 3, 16°C; 4, 18°C; 5, 20°C; 6, 22°C; 7, 24°C at normoxia. Values are means ± s.e.m. with n = 14–15 per group for the hypoxic warming experiment (except at 22°C where n = 9–10 per group, because 4–6 fish had already reached their CTmax), and with n = 9 for the normoxic warming experiment (except at 24°C where n =8, because one fish had already reached its CTmax).
(b). Cardiorespiratory response to warming when normoxic
The finding that fH and cardiac function were severely constrained during warming when the sablefish was hypoxic is unprecedented. Therefore, we performed an additional experiment with normoxia-acclimated sablefish to verify that this species has the capacity to elevate fH and Q̇ when warmed under normoxia, as is typical for other fishes [9,10]. Indeed, sablefish were able to increase fH and Q̇ by twofold (up to 59.1 ± 3.5 beats min−1 and 39.7 ± 4.3 ml beat−1 kg−1, respectively, p < 0.0001) (figures 1 and 2; electronic supplementary material, figure S2 and tables S1 and S3). Further, Q̇ and ṀO2 were positively correlated (p < 0.001; figure 3), which is consistent with the relationship that is normally observed for fishes (e.g. [19]). These findings suggest that acute hypoxia prevented the sablefish's normal cardiac response to warming, and that this limited their AST and thermal tolerance. For example, CTmax was approximately 3°C lower in fish exposed to acute hypoxia when compared with those tested under normoxia (figure 2d), and this reduction in CTmax was associated with a much lower AST (approx. 59 versus 174 mg O2 h−1 kg−1) (figure 2b); although values for ṀO2/Q̇ were similar (figures 1i,j and 2c; electronic supplementary material, table S3).
4. Discussion
Our results (i) provide additional evidence that cardiac function is linked with temperature-induced aerobic scope and thermal tolerance (figure 2), and further emphasize the role of the heart in delivering O2 to the tissues to meet the fish's metabolic demands when exposed to increased temperatures [4–10]; and (ii) show that hypoxia strongly limits the capacity of the cardiorespiratory system to respond to an acute thermal challenge. However, our study also offers other valuable physiological insights, brings up several questions and has major ecological implications.
(a). Possible reasons why fH did not increase during hypoxic warming
A key question, which future studies will need to explore, is why hypoxic sablefish were unable to/did not increase fH when exposed to rising temperatures (figure 1a; electronic supplementary material, table S3). One of the potential explanations is that adenosine (which is produced by the heart muscle when O2 supply is limited) depressed fH [39,40]. However, because hypoxic bradycardia in fish is induced by an increase in cholinergic nervous tone [9,18] on the heart, it is most likely that the sablefish was unable to remove (or reduce) this nervous tone as temperature rose and that this prevented increases in fH. Thus, it appears that while cholinergic inhibition of the heart (i.e. which prevents fH from getting too high, and delays the onset of arrhythmias) may be beneficial for upper temperature tolerance in species such as rainbow trout (Oncorhynchus mykiss) while normoxic [41], it may reduce the thermal tolerance of at least some fishes during hypoxia. This is not only because fH does not increase in hypoxic sablefish with temperature, but also that VS increased only slightly or did not change (figure 1e), and this prevented Q̇ from increasing with temperature. This finding is in contrast to Keen et al. [42] who used the pharmacological agent zatebradine to prevent fH increases in normoxic rainbow trout during a CTmax test, and showed that increases in VS completely compensated for the inability to elevate fH. This difference between studies is likely due to the direct negative effects that low O2 levels have on the contractility of the fish heart [43,44].
(b). New perspectives on fish cardiorespiratory physiology and thermal tolerance
Currently, the literature on fish cardiorespiratory physiology and thermal tolerance suggests that increases in fH (and thus Q̇) are primarily responsible for increases in ṀO2 with temperature and key to a fish's ability to tolerate warming events [4–10], while the importance of enhancing O2 extraction is seldom acknowledged/reported. One reason for this may be that salmonid species are often used as a model for the teleost fish's response to increasing temperatures. For example, the increase in ṀO2 in rainbow trout with temperature is due to a 154% enhancement of Q̇ but only a 16% increase in ṀO2/Q̇ (see figs 2 and 4 in Motyka et al. [19]). Similarly, Eliason et al. [45] reported that O2 extraction contributed little to the swimming and metabolic performance of sockeye salmon (Oncorhynchus nerka) at high temperatures. By contrast, the rise in the sablefish's ṀO2 during normoxic warming was due to comparable increases in Q̇ and ṀO2/Q̇ (i.e. both by approx. twofold; figure 1; electronic supplementary material, table S3), and when Q̇ did not increase under hypoxia, sablefish relied solely on enhanced ṀO2/Q̇. Therefore, this finding fundamentally changes our understanding of fish cardiorespiratory function and the drivers of O2 consumption at high temperatures. This important physiological adaptation for enhancing ṀO2 in the sablefish (and probably other fishes, e.g. European eel [34]) versus salmonids (trout, char, salmon) may be related to differences in their maximum fH and scope for increases in fH, and thus Q̇. Salmonids acclimated to 10°C, and warmed to their CTmax, have a maximum fH and scope for fH of approx. 120–130 and 70–80 beats min−1, respectively [19,46], whereas these values in sablefish are approx. 64 and 37 beats min−1 (figure 1a; electronic supplementary material, table S3). What mechanisms allow for the large enhancement in O2 extraction in the sablefish (and possibly other fishes) is not known. However, the sablefish represents one of the five teleostean groups where red blood cells appear to lack the β-adrenergic Na+/H+ exchanger (β-NHE) that protects against intracellular pH reductions [47], and this may allow for enhanced root effect-mediated O2 offloading from haemoglobin at the tissues. Interestingly, some red cell swelling during hypoxic warming was observed in this study (figure 4c; electronic supplementary material, figure S4), although this could have been due to passive/osmotic water influx and increased membrane fluidity/permeability, and is not necessarily indicative of β-NHE [48]. Recently, research has highlighted the potential role of plasma-accessible carbonic anhydrase (PaCA) in tissue O2 extraction in fishes, as this enzyme can acidify the blood as it passes through the tissues, and result in enhanced offloading of O2 from haemoglobin [36]. This mechanism may also play a role in facilitating O2 extraction in sablefish.
(c). No cross-tolerance and limited plasticity
Motyka et al. [19] showed that acclimation of steelhead trout to hypoxia (40% air saturation) does not affect this species' temperature-induced aerobic scope (AST) or CTmax when tested under normoxic conditions, and this largely agrees with the data that we report for the sablefish. Collectively, these data support the majority of research showing that there is no or very little ‘cross-tolerance’ between these two O2-limiting conditions (i.e. acclimation to hypoxia does not enhance tolerance to high temperatures, and vice versa) [19–21,23,24]. Further, the data from these two experiments fit very well with the ‘plastic floors and concrete ceilings' hypothesis [49] which was originally formulated based on the relationship between acclimation temperature and a fish's thermal tolerance. The hypoxia-intolerant rainbow trout in Motyka et al. [19] could increase tissue O2 extraction (ṀO2/Q̇) by 74% when acclimated to hypoxia, given that this parameter contributes little to AST in normoxia-acclimated fish warmed to their CTmax. By contrast, the sablefish (which has adapted to the OMZ conditions that they live in as adults [25,26]) has no plasticity to enhance ṀO2/Q̇ further through hypoxic acclimation because it is already a major contributor (i.e. it increased by 117%) to this fish's AST (figures 1 and 2; electronic supplementary material, table S3). This suggests that hypoxia-tolerant species may have no, or limited, scope for enhancing their cardiorespiratory capacity upon exposure to oxygen-limiting conditions because it has already been fully exploited as a result of their evolutionary adaptation to environmental hypoxia.
(d). Concluding remarks and ecological implications of the research
By explicitly testing the effect of an acute warming event on cardiac function when fish are already experiencing moderate hypoxia, we were able to show that prior exposure to acute hypoxia prevents the sablefish from increasing fH when subsequently exposed to high temperatures (figure 1). These data suggest that the ‘physiological chain of command’ (in this case nervous control of fH) may be detrimental under certain environmental conditions, or when they are experienced in a particular order (e.g. hypoxia before acute warming). In other words, the depression of fH by hypoxia appears to dominate over the requirement for increasing fH during warming, and this severely limits the fish's cardiac response to the latter stressor, and ultimately, appears to constrain thermal tolerance (figure 2); a finding that has recently been confirmed in the Atlantic salmon (Salmo salar) (Gamperl et al. [50]). Further, our results indicate that the importance of O2 extraction in determining the thermal tolerance of fishes has been underappreciated, and raise important questions (and testable hypotheses) about: the control of heart rate (function) when high temperatures and hypoxia occur simultaneously; and the role of PaCA in O2 extraction in fishes under varying conditions.
Importantly, these findings also confirm the findings of Ern et al. [17] that hypoxia limits fish acute thermal tolerance (figure 2), and draw attention to the possible role of hypoxia in determining whether fish species/populations survive in the current era of climate change. Coastal areas are experiencing more severe and frequent heat waves [1–3], and periods of hypoxia [1,12], and it is very likely that fish species in some of these areas will suffer a greater loss of biomass than the 5–17% estimated by Lotze et al. [51] for higher trophic level organisms in the oceans. This is because many fisheries and marine ecosystem models do not incorporate such periodic events (e.g. [51,52]), and the interactive effects of temperature and hypoxia on organisms in coastal ecosystems are only starting to be addressed [53,54]. Thus, climate change-induced declines in fish populations may be more severe than predicted to date. In addition, there is little evidence that acclimation to hypoxia substantially improves the tolerance of fishes to high temperatures (i.e. there is no cross-tolerance at the whole animal level) [19–21,24]. Although there are instances where the capability for plasticity in fish is considerable (e.g. [55]), it appears that many species of fish (and other ectothermic animals) have limited plasticity to respond to these climate-related challenges (present study, and [19,49,56,57]). Finally, while transgenerational plasticity has been shown to improve some aspects of fish physiology (e.g. aerobic scope) following acclimation to hypoxia or high temperatures, it is unlikely that this will translate into an increased ability to tolerate higher temperatures [56–59]. Thus, it is uncertain whether cross-tolerance, developmental plasticity or adaptation will allow coastal fish populations to persist across their current ranges as marine heat waves and hypoxic zones become more common, and extreme, with climate change.
Supplementary Material
Acknowledgements
We thank Rick Goetz (NOAA, Manchester Research Station, Port Orchard, WA, USA) and Golden Eagle Sablefish for supplying the sablefish for this study, the staff of the Cold-Ocean and Deep-Sea Research Facility and the Dr Joe Brown Aquatic Research Building for help with fish care, and Drs Doug Syme, Tony Farrell and Amanda Bates for providing valuable comments on earlier drafts of this manuscript.
Contributor Information
Robine H. J. Leeuwis, Email: rhjleeuwis@mun.ca.
A. Kurt Gamperl, Email: kgamperl@mun.ca.
Ethics
All experimental procedures were performed in compliance with the guidelines of the Canadian Council on Animal Care and were approved by the Memorial University of Newfoundland (MUN) Animal Care Committee under the protocol no. 16-92-KG.
Data accessibility
All data are provided within the manuscript itself or the electronic supplementary material, and full datasets supporting the main findings of this study are openly available at Zenodo via http://doi.org/10.5281/zenodo.3934340.
Authors' contributions
A.K.G. and R.H.J.L. conceptualized the study. R.H.J.L., E.F.C.P. and F.S.Z. performed the experiments and collected the data. A.K.G., R.H.J.L. and F.S.Z. contributed to data analysis and interpretation, and to the writing of the manuscript.
Competing interests
Authors declare no competing interests.
Funding
This research was supported by an NSERC Discovery Grant to A.K.G. (2016-0448), and research funding to A.K.G. from the Ocean Frontier Institute, through an award from the Canada First Research Excellence Fund. R.H.J.L. was also supported by a Memorial University of Newfoundland, School of Graduate Studies, fellowship.
References
- 1.IPCC. 2014. Climate change 2014: synthesis report. Geneva, Switzerland: IPCC. [Google Scholar]
- 2.Oliver ECJ, et al. 2018. Longer and more frequent marine heatwaves over the past century. Nat. Commun. 9, 1324. ( 10.1038/s41467-018-03732-9) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Frölicher TL, Fischer EM, Gruber N. 2018. Marine heatwaves under global warming. Nature 560, 360-364. ( 10.1038/s41586-018-0383-9) [DOI] [PubMed] [Google Scholar]
- 4.Pörtner HO. 2010. Oxygen- and capacity-limitation of thermal tolerance: a matrix for integrating climate-related stressor effects in marine ecosystems. J. Exp. Biol. 213, 881-893. ( 10.1242/jeb.037523) [DOI] [PubMed] [Google Scholar]
- 5.Pörtner HO, Bock C, Mark FC. 2017. Oxygen- and capacity-limited thermal tolerance: bridging ecology and physiology. J. Exp. Biol. 220, 2685-2696. ( 10.1242/jeb.134585) [DOI] [PubMed] [Google Scholar]
- 6.Wang T, Overgaard J. 2007. The heartbreak of adapting to global warming. Science 315, 49-50. ( 10.1126/science.1137359) [DOI] [PubMed] [Google Scholar]
- 7.Farrell AP, Eliason EJ, Sandblom E, Clark TD. 2009. Fish cardiorespiratory physiology in an era of climate change. Can. J. Zool. 87, 835-851. ( 10.1139/Z09-092) [DOI] [Google Scholar]
- 8.Eliason EJ, et al. 2011. Differences in thermal tolerance among sockeye salmon populations. Science 332, 109-112. ( 10.1126/science.1199158) [DOI] [PubMed] [Google Scholar]
- 9.Farrell AP, Smith F. 2017. Cardiac form, function and physiology in fish physiology, Vol. 36A, pp. 155-264. Amsterdam, The Netherlands: Elsevier. [Google Scholar]
- 10.Eliason EJ, Anttila K. 2017. Temperature and the cardiovascular system in fish physiology, Vol. 36B, pp. 235-297. Amsterdam, The Netherlands: Elsevier. [Google Scholar]
- 11.Andreassen AH, Hall P, Khatibzadeh P, Jutfelt F, Kermen F. 2020. Neural dysfunction at the upper thermal limit in the zebrafish. BioRxiv preprint. ( 10.1101/2020.12.28.424529) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Breitburg D, et al. 2018. Declining oxygen in the global ocean and coastal waters. Science 359, 7240. ( 10.1126/science.aam7240) [DOI] [PubMed] [Google Scholar]
- 13.Kemp WM, et al. 2005. Eutrophication of Chesapeake Bay: historical trends and ecological interactions. Mar. Ecol. Prog. Ser. 303, 1-29. ( 10.3354/meps303001) [DOI] [Google Scholar]
- 14.Burt K, Hamoutene D, Mabrouk G, Lang C, Puestow T, Drover D, Losier R, Page F. 2012. Environmental conditions and occurrence of hypoxia within production cages of Atlantic salmon on the south coast of Newfoundland. Aquac. Res. 43, 607-620. ( 10.1111/j.1365-2109.2011.02867.x) [DOI] [Google Scholar]
- 15.Stehfest KM, Carter CG, McAllister JD, Ross JD, Semmens JM. 2017. Response of Atlantic salmon Salmo salar to temperature and dissolved oxygen extremes established using animal-borne environmental sensors. Sci. Rep. 7, 4545. ( 10.1038/s41598-017-04806-2) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Burke H, Gardner I, Farrell A.. 2020. A review of the 2019 Newfoundland and Labrador South Coast cultured Atlantic Salmon mortality event. Special Studies Reports, Department of Fisheries and Land Resources, Government of Newfoundland and Labrador.
- 17.Ern R, Norin T, Gamperl AK, Esbaugh AJ. 2016. Oxygen dependence of upper thermal limits in fishes. J. Exp. Biol. 219, 3376-3383. ( 10.1242/jeb.143495) [DOI] [PubMed] [Google Scholar]
- 18.Farrell AP. 2007. Tribute to P. L. Lutz: a message from the heart—why hypoxic bradycardia in fishes? J. Exp. Biol. 210, 1715-1725. ( 10.1242/jeb.02781) [DOI] [PubMed] [Google Scholar]
- 19.Motyka R, Norin T, Petersen LH, Huggett DB, Gamperl AK. 2017. Long-term hypoxia exposure alters the cardiorespiratory physiology of steelhead trout (Oncorhynchus mykiss), but does not affect their upper thermal tolerance. J. Therm. Biol. 68, 149-161. ( 10.1016/j.jtherbio.2016.03.007) [DOI] [PubMed] [Google Scholar]
- 20.Burleson ML, Silva PE. 2011. Cross tolerance to environmental stressors: effects of hypoxic acclimation on cardiovascular responses of channel catfish (Ictalurus punctatus) to a thermal challenge. J. Therm. Biol. 36, 250-254. ( 10.1016/j.jtherbio.2011.03.009) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Anttila LM, Prokkola JM, Kanerva M, Seppänen E, Kolari I, Nikinmaa M. 2015. Warm acclimation and oxygen depletion induce species-specific responses in salmonids. J. Exp. Biol. 218, 1471-1477. ( 10.1242/jeb.119115) [DOI] [PubMed] [Google Scholar]
- 22.McBryan TL, Healy TM, Haakons KL, Schulte PM. 2016. Warm acclimation improves hypoxia tolerance in Fundulus heteroclitus. J. Exp. Biol. 219, 474-484. ( 10.1242/jeb.133413) [DOI] [PubMed] [Google Scholar]
- 23.Levesque KD, Wright PA, Bernier NJ. 2019. Cross talk without cross tolerance: effect of rearing temperature on the hypoxia response of embryonic zebrafish. Physiol. Biochem. Zool. 92, 349-364. ( 10.1086/703178) [DOI] [PubMed] [Google Scholar]
- 24.McDonnell LH, Reemeyer JE, Chapman LJ. 2019. Independent and interactive effects of long-term exposure to hypoxia and elevated water temperature on behavior and thermal tolerance of an equatorial cichlid. Physiol. Biochem. Zool. 92, 253-265. ( 10.1086/702712) [DOI] [PubMed] [Google Scholar]
- 25.Mason JC, Beamish RJ, McFarlane GA. 1983. Sexual maturity, fecundity, spawning, and early life history of sablefish (Anoplopoma fimbria) off the Pacific coast of Canada. Can. J. Fish. Aquat. Sci. 40, 2126-2134. ( 10.1139/f83-247) [DOI] [Google Scholar]
- 26.Doya C, Chatzievangelou D, Bahamon N, Purser A, De Leo FC, Juniper SK, Thomsen L, Aguzzi J. 2017. Seasonal monitoring of deep-sea megabenthos in Barkley Canyon cold seep by internet operated vehicle (IOV). PLoS ONE 12, e0176917. ( 10.1371/journal.pone.0176917) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Leeuwis RHJ, Nash GW, Sandrelli RM, Zanuzzo FS, Gamperl AK. 2019. The environmental tolerances and metabolic physiology of sablefish (Anoplopoma fimbria). Comp. Biochem. Physiol. A 231, 140-148. ( 10.1016/j.cbpa.2019.02.004) [DOI] [PubMed] [Google Scholar]
- 28.Remen M, Oppedal F, Imsland AK, Olsen RE, Torgersen T. 2013. Hypoxia tolerance thresholds for post-smolt Atlantic salmon: dependency of temperature and hypoxia acclimation. Aquaculture 416–417, 41-47. ( 10.1016/j.aquaculture.2013.08.024) [DOI] [Google Scholar]
- 29.Gerber L, Clow KA, Katan T, Emam M, Leeuwis RHJ, Parrish CC, Gamperl AK. 2019. Cardiac mitochondrial function, nitric oxide sensitivity and lipid composition following hypoxia acclimation in sablefish. J. Exp. Biol. 222, 208074. ( 10.1242/jeb.208074) [DOI] [PubMed] [Google Scholar]
- 30.Gollock MJ, Currie S, Petersen LH, Gamperl AK. 2006. Cardiovascular and haematological responses of Atlantic cod (Gadus morhua) to acute temperature increase. J. Exp. Biol. 209, 2961-2970. ( 10.1242/jeb.02319) [DOI] [PubMed] [Google Scholar]
- 31.Haverinen J, Vornanen M. 2020. Reduced ventricular excitability causes atrioventricular block and depression of heart rate in fish at critically high temperatures. J. Exp. Biol. 223, jeb225227. ( 10.1242/jeb.225227) [DOI] [PubMed] [Google Scholar]
- 32.Zanuzzo F, Urbinati E, Nash G, Gamperl A. 2015. Steelhead trout Oncorhynchus mykiss metabolic rate is affected by dietary Aloe vera inclusion but not by mounting an immune response against formalin-killed Aeromonas salmonicida. J. Fish Biol. 87, 43-53. ( 10.1111/jfb.12690) [DOI] [PubMed] [Google Scholar]
- 33.Farrell AP, Eliason EJ, Clark TD, Steinhausen MF. 2014. Oxygen removal from water versus arterial oxygen delivery: calibrating the Fick equation in Pacific salmon. J. Comp. Physiol. B 184, 855-864. ( 10.1007/s00360-014-0839-7) [DOI] [PubMed] [Google Scholar]
- 34.Claësson D, Wang T, Malte H. 2016. Maximal oxygen consumption increases with temperature in the European eel (Anguilla anguilla) through increased heart rate and arteriovenous extraction. Conserv. Physiol. 4, cow027. ( 10.1093/conphys/cow027) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Joyce W, Axelsson M, Egginton S, Farrell AP, Crockett EL, O'Brien KM. 2018. The effects of thermal acclimation on cardio-respiratory performance in an Antarctic fish (Notothenia coriiceps). Conserv. Physiol. 6, coy069. ( 10.1093/conphys/coy069) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Harter TS, Zanuzzo FS, Supuran CT, Gamperl AK, Brauner CJ. 2019. Functional support for a novel mechanism that enhances tissue oxygen extraction in a teleost fish. Proc. R. Soc. B 286, 20190339. ( 10.1098/rspb.2019.0339) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Paschke K, et al. 2018. Comparison of aerobic scope for metabolic activity in aquatic ectotherms with temperature related metabolic stimulation: a novel approach for aerobic power budget. Front. Physiol. 9, phys.2018.01438. ( 10.3389/fphys.2018.01438) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Norin T, Canada P, Bailey JA, Gamperl AK. 2019. Thermal biology and swimming performance of Atlantic cod (Gadus morhua) and haddock (Melanogrammus aeglefinus). PeerJ 7, e7784. ( 10.7717/peerj.7784) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sundin L, Axelsson M, Davison W, Forster ME. 1999. Cardiovascular responses to adenosine in the Antarctic fish Pagothenia borchgrevinki. J. Exp. Biol. 202, 2259-2267. [DOI] [PubMed] [Google Scholar]
- 40.Stecyk JA, Stensløkken KO, Nilsson GE, Farrell AP. 2007. Adenosine does not save the heart of anoxia-tolerant vertebrates during prolonged oxygen deprivation. Comp. Biochem. Physiol. A 147, 961-973. ( 10.1016/j.cbpa.2007.03.002) [DOI] [PubMed] [Google Scholar]
- 41.Gilbert MJH, Rani V, McKenzie SM, Farrell AP. 2019. Autonomic cardiac regulation facilitates acute heat tolerance in rainbow trout: in situ and in vivo support. J. Exp. Biol. 222, 194365. ( 10.1242/jeb.194365) [DOI] [PubMed] [Google Scholar]
- 42.Keen AN, Gamperl AK. 2012. Blood oxygenation and cardiorespiratory function in steelhead trout (Oncorhynchus mykiss) challenged with an acute temperature increase and zatebradine-induced bradycardia. J. Therm. Biol. 37, 201-210. ( 10.1016/j.jtherbio.2012.01.002) [DOI] [Google Scholar]
- 43.Tikkanen E, Haverinen J, Egginton S, Hassinen M, Vornanen M. 2017. Effects of prolonged anoxia on electrical activity of the heart in crucian carp (Carassius carassius). J. Exp. Biol. 220, 445-454. ( 10.1242/jeb.145177) [DOI] [PubMed] [Google Scholar]
- 44.Carnevale C, Roberts JC, Syme DA, Gamperl AK. 2020. Hypoxic acclimation negatively impacts the contractility of steelhead trout (Oncorhynchus mykiss) spongy myocardium. Am. J. Physiol. Regul. Integr. Comp. Physiol. 318, 214-226. ( 10.1152/ajpregu.00107.2019) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Eliason EJ, Clark TD, Hinch SG, Farrell AP. 2013. Cardiorespiratory collapse at high temperature in swimming adult sockeye salmon. Conserv. Physiol. 1, cot008. ( 10.1093/conphys/cot008) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Penney CM, Nash GW, Gamperl AK. 2014. Cardiorespiratory responses of seawater-acclimated adult Arctic char (Salvelinus alpinus) and Atlantic salmon (Salmo salar) to an acute temperature increase. Can. J. Fish. Aquat. Sci. 71, 1096-1105. ( 10.1139/cjfas-2013-0569) [DOI] [Google Scholar]
- 47.Rummer JL, Roshan-Moniri M, Balfry SK, Brauner CJ. 2010. Use it or lose it? Sablefish, Anoplopoma fimbria, a species representing a fifth teleostean group where the βNHE associated with the red blood cell adrenergic stress response has been secondarily lost. J. Exp. Biol. 213, 1503-1512. ( 10.1242/jeb.038844) [DOI] [PubMed] [Google Scholar]
- 48.Nikinmaa M. 1992. How does environmental pollution affect red cell function in fish? Aquat. Toxicol. 22, 227-238. ( 10.1016/0166-445X(92)90042-L) [DOI] [Google Scholar]
- 49.Sandblom E, et al. 2016. Physiological constraints to climate warming in fish follow principles of plastic floors and concrete ceilings. Nat. Commun. 7, 11447. ( 10.1038/ncomms11447) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gamperl AK, Clow KA, Sandrelli RM, Gerber L, Peroni EFC. In preparation. Hypoxia-induced bradycardia prevents temperature-dependent increases in heart rate and greatly reduces the thermal tolerance of Atlantic salmon (Salmo salar).
- 51.Lotze HK, et al. 2019. Global ensemble projections reveal trophic amplification of ocean biomass declines with climate change. Proc. Natl. Acad. Sci USA 116, 12 907-12 912. ( 10.1073/pnas.1900194116) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bell JD, et al. 2013. Mixed responses of tropical Pacific fisheries and aquaculture to climate change. Nat. Clim. Chang. 3, 591-599. ( 10.1038/NCLIMATE1838) [DOI] [Google Scholar]
- 53.Roman MR, Brandt SB, Houde ED, Pierson JJ. 2019. Interactive effects of hypoxia and temperature on coastal pelagic zooplankton and fish. Front. Mar. Sci. 6, 139. ( 10.3389/fmars.2019.00139) [DOI] [Google Scholar]
- 54.Howard EM, et al. 2020. Climate-driven aerobic habitat loss in the California Current System. Sci. Adv. 6, eaay31882020. ( 10.1126/sciadv.aay3188) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Anttila K, Couturier CS, Øverli Ø, Johnsen A, Marthinsen G, Nilsson GE, Farrell AP. 2014. Atlantic salmon show capability for cardiac acclimation to warm temperatures. Nat. Commun. 5, 4252. ( 10.1038/ncomms5252) [DOI] [PubMed] [Google Scholar]
- 56.Muñoz NJ, Farrell AP, Heath JW, Neff BD. 2015. Adaptive potential of a Pacific salmon challenged by climate change. Nat. Clim. Change 5, 163-166. ( 10.1038/NCLIMATE2473) [DOI] [Google Scholar]
- 57.Kielland ØN, Bech C, Einum S. 2019. Warm and out of breath: thermal phenotypic plasticity in oxygen supply. Funct. Ecol. 33, 2142-2149. ( 10.1111/1365-2435.13449) [DOI] [Google Scholar]
- 58.Donelson JM, Munday PL, McCormick MI, Pitcher CR. 2012. Rapid transgenerational acclimation of a tropical reef fish to climate change. Nat. Clim. Change 2, 30-32. ( 10.1038/NCLIMATE1323) [DOI] [Google Scholar]
- 59.White DP, Wahl DH. 2020. Growth and physiological responses in largemouth bass populations to environmental warming: effects of inhabiting chronically heated environments. J. Therm. Biol. 88, 102467. ( 10.1016/j.jtherbio.2019.102467) [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data are provided within the manuscript itself or the electronic supplementary material, and full datasets supporting the main findings of this study are openly available at Zenodo via http://doi.org/10.5281/zenodo.3934340.