Abstract
One in three Americans suffer from kidney diseases such as chronic kidney disease, and one of the etiologies is suggested to be long-term renal hypoxia. Interestingly, sympathetic nervous system activation evokes a renal vasoconstrictor effect that may limit oxygen delivery to the kidney. In this report, we sought to determine if sympathetic activation evoked by lower body negative pressure (LBNP) would decrease cortical and medullary oxygenation in humans. LBNP was activated in a graded fashion (LBNP; −10, −20, and −30 mmHg), as renal oxygenation was measured (T2*, blood oxygen level dependent, BOLD MRI; n = 8). At a separate time, renal blood flow velocity (RBV) to the kidney was measured (n = 13) as LBNP was instituted. LBNP significantly reduced RBV (P = 0.041) at −30 mmHg of LBNP (Δ−8.17 ± 3.75 cm/s). Moreover, both renal medullary and cortical T2* were reduced with the graded LBNP application (main effect for the level of LBNP P = 0.0008). During recovery, RBV rapidly returned to baseline, whereas medullary T2* remained depressed into the first minute of recovery. In conclusion, sympathetic activation reduces renal blood flow and leads to a significant decrease in oxygenation in the renal cortex and medulla.
NEW & NOTEWORTHY In healthy young adults, increased sympathetic activation induced by lower body negative pressure, led to a decrease in renal cortical and medullary oxygenation measured by T2*, a noninvasive magnetic resonance derived index of deoxyhemoglobin levels. In this study, we observed a significant decrease in renal cortical and medullary oxygenation with LBNP as well as an increase in renal vasoconstriction. We speculate that sympathetic renal vasoconstriction led to a significant reduction in tissue oxygenation by limiting oxygen delivery to the renal medulla.
Keywords: renal oxygenation, renal reflexes, sympathetic activation
INTRODUCTION
Chronic kidney disease is a major public health problem affecting one in three American adults and resulting in a significant socioeconomic burden (1, 2). This major health concern is not getting better, in fact, the number of patients with kidney diseases is steadily increasing (3, 4).
In physiological terms, the kidney plays an essential role in maintaining cardiovascular homeostasis by the handling of sodium and water. These factors determine blood volume, and are controlled by hormonal influences (i.e., aldosterone and antidiuretic hormone) and by direct and indirect actions of autonomic nervous system activation via renal sympathetic nerves. The actions of renal sympathetic nerve activation on fluid homeostasis include: 1) increase in renin secretion (5–8), 2) decrease in sodium excretion (9–12), and 3) vasoconstriction of the renal arterioles and interlobular arteries (6, 13).
The actions of renal sympathetic nerve activation are substantially influenced by sensory systems, with one of the most important regulators of renal sympathetic nerve activity being high pressure baroreceptors that are mechanoreceptors sensing blood pressure (14, 15). It is well documented that the sustained unloading of baroreflex via applying negative pressure to the lower body significantly increases renin release (16), and reduced sodium and water excretion without affecting the glomerular filtration rate (GFR) and renal hemodynamics (17) in humans. However, it is also known that short bouts of lower body negative pressure evoke renal vasoconstriction thereby reducing renal arterial blood flow (18). Moreover, tubular sodium reabsorption significantly depends on the tubular volume load, thus small changes in GFR will result in a significant change in sodium reabsorption by the kidney. However, it is unclear if the sympathetically mediated renal vasoconstriction and reduction in renal blood flow result in alterations in renal tissue deoxygenation. Understanding this relationship could be particularly important as we seek to better understand the impact of sympathetic activation and salt and water balance in diseases characterized by heightened sympathetic tone including congestive heart failure, hypertension, and obstructive sleep apnea (19–21). Moreover, although it is suggested that long-term renal hypoxia induces renal fibrosis and thus results in the progression of chronic kidney disease (22), the influence of sympathetic activation on renal oxygenation in humans has not been elucidated.
Blood oxygen level-dependent (BOLD) MRI is a noninvasive imaging technique that has been used to study oxygenation of the kidney. The quantitative BOLD-MRI yields a map of the time constant T2* that describes the exponential decay of the BOLD signal (23–27) which is sensitive to the ratio of oxyhemoglobin (oxyHb) to deoxyhemoglobin (deoxyHb) reflecting both tissue oxygen supply and demand (23, 28). With quantitative BOLD-MRI, relative markers of renal medullary and cortical oxygenation can be measured noninvasively. For this study, we have devised approaches with BOLD-MRI to assess renal oxygenation during sympathetic activation via LBNP in humans.
In this report, we examined the feasibility of using functional magnetic resonance imaging methods to characterize changes in hemoglobin deoxygenation in human subjects. We also examined the effects of acute sympathetic nervous system activation induced by graded LBNP on renal medullary oxygenation as determined by using BOLD MRI (medullary T2*). We hypothesize that the sympathetic activation would significantly reduce renal oxygenation. In separate studies conducted in a separate facility, we examined the effects of LBNP on renal blood flow velocity (RBV). We observed that LBNP had similar effects on RBV and medullary deoxygenation suggesting that sympathetic activation directed to the kidney acutely activates systems to both reduce the glomerular filtration rate and to enhance salt and water retention.
METHODS
Subjects
This study used a repeated-measures, within-subjects’ design to examine RBV and renal oxygenation during graded LBNP. Thirteen healthy young adults (28 ± 1 yr, 7 men, 6 women) were recruited from the local community. Interested volunteers provided written informed consent before enrollment. Medical screening was performed to determine eligibility. Participants were selected if they were normotensive, were free of overt chronic disease as assessed by a medical history questionnaire, and had completed a physical exam that showed no hematological, liver, or kidney abnormalities. All subjects met the following criteria: 1) ages between 21 and 35 yr, 2) resting blood pressure less than 140/90 mmHg, 3) nonsmoker, 4) not taking any medications, and 5) free of acute medical conditions. For the study visits, subjects were asked to fast 4 h and stay well hydrated for 24 h before experimental visits. The characteristics of participants who completed this study are shown in Table 1. All procedures were approved by the Institutional Review Board of the Penn State Health Milton S. Hershey Medical Center in agreement with the guidelines set forth by the Declaration of Helsinki.
Table 1.
Subject characteristics and resting hemodynamics
| Visit 1(Doppler visit) | Visit 2(MRI visit) | |
|---|---|---|
| Number of subjects (men/women) | 13 (7/6) | 8 (4/4) |
| Age, yr | 28 ± 1 | 28 ± 1 |
| Height, cm | 172 ± 3 | 173 ± 2 |
| Weight, kg | 70 ± 3 | 69 ± 4 |
| BMI, kg/m2 | 23.6 ± 0.7 | 23.2 ± 1.0 |
| SBP, mmHg | 116 ± 3 | 113 ± 2 |
| DBP, mmHg | 71 ± 3 | 70 ± 3 |
| MAP, mmHg | 86 ± 2 | 84 ± 3 |
| PP, mmHg | 44 ± 2 | 43 ± 2 |
| HR, beats/min | 60 ± 3 | 56 ± 2 |
| Cortical T2*, ms | - | 57.72 ± 0.24 |
| Medullary T2*, ms | - | 31.29 ± 0.51 |
Data are expressed as means ± SE. BMI, body mass index; DBP, diastolic blood pressure; HR, heart rate; MAP, mean arterial blood pressure; PP, pulse pressure; SBP, systolic blood pressure.
Study Design
This study consisted of two visits and the subjects performed the same study protocol (described below and in Fig. 1A) for both visits. Visit 1 was performed at the Clinical Research Center (CRC) for renal blood flow measurement, and visit 2 was performed at the Center for Nuclear Magnetic Resonance Research (CNMRR) for BOLD-MRI. Both centers are on the campus of Penn State Hershey Medical Center. Although all 13 volunteers participated in visit 1, and 5 of the 13 subjects did not participate in the MRI studies during visit 2 due to claustrophobia and other reasons (Table 1). For visit 1, the subjects were placed in the supine position. Their lower bodies were placed in a fabricated MRI-compatible LBNP chamber to the iliac crest chamber. This chamber was designed and built by one of the coauthors (M. Herr; Fig. 1C). Subjects were then instrumented with: 1) automated blood pressure oscillometry of the right brachial artery (Phillips Sure Signs VS3); 2) finger plethysmography for blood pressure (Finometer MIDI, Finapres Medical Systems); 3) 3-lead EKG electrodes (Cardiocap/5, GE Healthcare) for heart rate (HR) measurements; 4) a pulse oximeter (5250 RGM, Datex-Ohmeda Medical Equipment); and 5) an abdominal pneumographic belt to monitor respiration. During the study protocol, an investigator placed a Doppler ultrasound probe (Vivid 7, GE Healthcare) on the subject’s abdomen to obtain renal blood flow velocity (RBV). After instrumentation and device placement, subjects performed 12 voluntary breath-holds (∼22 s) to be familiarized with the breath-hold sound cue. RBV, BP, HR, and oxygen saturation were measured before, during, and after these breath-holds. After a short recovery following the breath-hold familiarization, a 7.5-min resting baseline occurred before LBNP was applied. Then LBNP was applied at −10, −20, and −30 mmHg for 5 min at each stage. This was followed by a 7.5-min recovery period. LBNP, RBV, BP, and HR were measured during baseline, at each stage of LBNP, and during recovery from LBNP. Similar to visit 1, during visit 2, subjects were placed in the supine position in the LBNP tank. They were then instrumented with an MRI compatible arm cuff for automated BP measurements and a pulse oximeter. The subjects were positioned within the magnet with the kidney MR coil placed over the kidney. Subjects were instructed to gently place their arms on their chest (Fig. 1B). Then, anatomical MRI scans were taken while the subjects performed voluntary breath-holds. BP, HR, and oxygen saturation parameters were measured throughout the scanning protocol. After these scans, the breath-hold and LBNP interventions were performed as in visit 1. MRI scans were taken during the breath-hold to minimize motion artifacts, and BP and HR were measured during baseline, at each stage of LBNP, and recovery from LBNP (Fig. 1A).
Figure 1.
A: experimental design. B: study model diagram. C: nonmagnetic LBNP tank. The subject was placed in the MRI compatible LBNP tank, which was then slid into the bore of the magnet. Participants’ placed their arms on the chest to avoid scanning the artifact. BH, breath hold; LBNP, lower body negative pressure; MRI, magnetic resonance imaging.
Lower Body Negative Pressure Using MRI Compatible Chamber
To employ LBNP during visits 1 and 2, we developed a nonmagnetic LBNP tank that allowed us to apply gradual suction. The LBNP tank was constructed using plywood and heavy-duty cardboard tubes, which were assembled with wood glue. The tank’s dimensions permitted entry into the magnet bore. Each volunteer wore a close-fitting neoprene “skirt” around the abdomen that was interfaced to the tank’s entrance to achieve a pneumatic seal (Fig. 1C). The air was withdrawn from the tank through a length of corrugated respirator tubing connected to a small canister vacuum cleaner located in the control room. A variable transformer controlled the level of suction generated by the vacuum cleaner. Tank pressure was remotely monitored via a custom pressure transducer amplifier connected to the tank with IV tubing.
Renal Vascular Imaging
RBV was recorded using Doppler ultrasound (Vivid 7, GE) via the anterior abdominal approach. The renal artery was scanned using a 2- to 5-MHz curved-array transducer with 2.5-MHz pulsed Doppler frequency. The insonation angle of the probe to the artery wall was 60° or less and the focal zone was set at the depth of the renal artery. The transducer was held in the same place for each trial and data were obtained during the voluntary breath-holds to ensure good image quality. Because of the low Doppler ultrasound frequency required to measure RBV and the resultant limited spatial resolution, it was not feasible to measure the renal artery diameter. However, it has been shown that the diameter of such a large conduit vessel does not change significantly in response to adenosine infusions, which evoke a large reduction in renal blood flow (29, 30). Thus, under conditions where renal blood flow falls, RBV should be an excellent surrogate for renal blood flow. All RBV data were collected and analyzed by one investigator, and to test the reproducibility the measurement was repeated on a different day (Supplemental Fig. S1; see https://doi.org/10.6084/m9.figshare.10042844). Lastly, renal vascular resistance (RVR) is calculated as MAP divided by RBV and reported to reflect renal vascular responses to sympathetic activation.
MRI Data Acquisition
Renal medullary and cortical oxygenation were measured noninvasively with quantitative blood oxygen level-dependent (BOLD) magnetic resonance imaging; a technique that yields a map of the time constant T2* that describes the exponential decay of the BOLD signal (23, 25, 27, 28, 31). This signal is sensitive to the ratio of oxyhemoglobin (oxyHb) to deoxyhemoglobin (deoxyHb) reflecting alterations in tissue oxygen extraction and utilization (23, 28).
Data were acquired using a 3 T PrismaFit (Siemens Healthineers, Erlangen, Germany). An 18-channel body flex array and 32-channel spine array were used for signal reception. A three-dimensional (3D) gradient-recalled sequence with a TR of 30 ms, a flip angle of 15°, and 6 echo times (ranging from 4.95 to 26.5 ms) was used to collect T2*-weighted data. The imaging volume consisted of a slab in the coronal orientation acquired with an in-plane voxel resolution of 1.6 × 1.6 mm, and 16 slices with a thickness of 2 mm per slice. A single 3D volume was acquired during a 22-s period as the subject was instructed to hold their breath. Breath-holds were interspersed with a 2-min and 8-s rest period. In this manner, 12 time points (3 in baseline, 2 in each LBNP stage, and 3 in recovery) were sampled during a 30-min interval. The ramping of the LBNP occurred during the intervals between scanning to allow for acclimation.
MRI Data Analysis
The multiecho image volumes were imported into IDL 8.6 (Exelis Visual, Inc, Boulder, CO) and parametric maps were calculated via a fit to the monoexponential equation: S(i) = S0 × exp[−TE/T2*] using an in-house script. The quantities S0, T2*, and a goodness of fit parameter were computed and stored for each voxel at all time points. Maps containing these quantities and the multiecho image volumes were then imported into a custom tool designed for kidney segmentation written in MATLAB 2017a (MathWorks, Natick, MA). A user manually outlined the kidney boundary on several slices at each time point. Care was taken to exclude slices suffering from susceptibility artifacts due to nearby air-tissue interfaces (Fig. 2). After the delineation of the kidney boundaries, each kidney slice was individually segmented into four distinct tissue compartments (pelvic, medulla, cortex, and arterial) with MATLAB’s k-means clustering algorithm. The segmentation was performed upon the first-echo image at each time point, respectively. The identified medulla compartment was then utilized to extract the corresponding voxels from the T2* maps, such that a pool of T2* values at each time point was formed. Further filtering of the pool was accomplished by the removal of voxels with a poor goodness of fit (<0.85) and outside a physiological range for medulla (10–40 ms) and cortex (45–70 ms). The renal cortex was processed in a similar manner, the only difference being the physiological filter range (45–70 ms). The signal timecourse was then formed by taking the mean of the filtered T2* pool at each time point.
Figure 2.
A: image of the left kidney taken in the coronal plane using the blood oxygen level-dependent (BOLD) magnetic resonance imaging technique in one subject at rest. B: segmented image of the region of interest (ROI). The darker region reflects the renal medulla. ROI is highlighted in red.
Data Collection and Statistical Analysis
All variables were measured continuously and analyzed offline. Primary outcome variables for this study were tested using one-way repeated-measure analysis of variance (ANOVA) to examine the effects of sympathetic activation via baroreceptor unloading on systemic hemodynamics and RBV. Changes in renal medullary and cortical oxygenation parameters during LBNP were analyzed using a two-way repeated ANOVA. Post hoc comparisons were performed when indicated using the Bonferroni method. Statistical analyses were performed using Prism 7 (GraphPad Software, Inc., La Jolla, CA). Recovery data points were compared to baseline using a paired t test. Alpha levels were set at P < 0.05 with appropriate adjustments made for multiple comparisons. Final summary data are expressed as means ± standard error.
RESULTS
Systemic Hemodynamic Responses during LBNP
No significant change in mean arterial blood pressure (MAP; P = 0.587) was observed with LBNP (Table 2; n = 13). On the other hand, systolic blood pressure (SBP; P = 0.013, effect size f = 0.143,) and pulse pressure (PP; P < 0.001, effect size f = 0.450,) fell and diastolic pressure tended to increase with LBNP at −30 mmHg of LBNP. HR increased with LBNP (P < 0.001, effect size f = 0.232,). Post hoc comparisons demonstrated that the HR values at −20 and −30 mmHg of LBNP were significantly higher than the baseline values by ∼5 beats/min (P < 0.001).
Table 2.
Hemodynamics during lower body negative pressure (LBNP) on visit 1 (n =13)
| Baseline | LBNP |
Recovery |
|||||
|---|---|---|---|---|---|---|---|
| −10 mmHg | −20 mmHg | −30 mmHg | 1 min | 2 min | 3 min | ||
| SBP, mmHg | 113 ± 3 | 113 ± 3 | 111 ± 2 | 110 ± 3* | 114 ± 2 | 115 ± 3 | 113 ± 2 |
| DBP, mmHg | 71 ± 2 | 71 ± 2 | 71 ± 2 | 73 ± 2 | 72 ± 2 | 71 ± 2 | 72 ± 2 |
| PP, mmHg | 42 ± 1 | 43 ± 2 | 39 ± 2 | 38 ± 1* | 42 ± 1 | 44 ± 2* | 41 ± 1 |
| MAP, mmHg | 83 ± 3 | 82 ± 3 | 83 ± 3 | 83 ± 3 | 83 ± 3 | 84 ± 3 | 83 ± 3 |
| HR, beats/min | 60 ± 3 | 59 ± 3 | 64 ± 3* | 65 ± 3* | 59 ± 3 | 57 ± 3* | 59 ± 3 |
For each LBNP stage, the second minute data are reported. One-way repeated ANOVA was performed for the statistical analysis. If the one-way repeated ANOVA revealed a significant main effect, post hoc comparisons (3 comparisons) were performed using the Bonferroni method. DBP, diastolic blood pressure; HR, heart rate; LBNP, lower body negative pressure; MAP, mean arterial blood pressure; PP, pulse pressure; SBP, systolic blood pressure.
Significant difference from baseline, P < 0.05. Data are expressed as means ± SE.
Renal Blood Flow Velocity Responses during LBNP and Recovery
Baroreceptor unloading with LBNP led to a decrease in RBV (P = 0.041, effect size f = 0.183, Fig. 3A, n = 13). Multiple comparisons demonstrated that the RBV was significantly reduced from the baseline (61.31 cm/s) at −30 mmHg of LBNP (53.14 cm/s; P = 0.019). Percent change in RVR rose with LBNP (P < 0.018, effect size f = 0.510, Fig. 3B). Mean RBVs of all 3 data points (R1, R2, and R3) during the recovery period were not statistically different from the baseline (Fig. 5A).
Figure 3.
Renal blood flow velocity (RBV) responses (A) and percent change in renal vascular resistance (RVR) (B) during lower body negative pressure (LBNP; −10 mmHg, −20 mmHg, −30 mmHg; n = 13). One-way repeated ANOVA was performed and showed significant main effect for both RBV (P = 0.041) and RVR (P = 0.018). For both variables, post hoc pairwise comparison (3 comparisons) with the Bonferroni method was followed. These comparisons revealed a significant decrease in RBV (P = 0.020) and increase in RVR (P = 0.006) at −30 mmHg of LBNP. Data are presented as means ± SE. *Significant difference from baseline, P < 0.05.
Figure 5.
Renal blood flow velocity (RBV; n = 13) (A) and renal medullary T2* (n = 8) (B) responses during recovery after termination of lower body negative pressure (LBNP). Post hoc paired t tests revealed significant reduction in medulla T2* 30 s into recovery (R1; P = 0.002 vs. baseline) and immediate return to baseline of RBV. R1, 30 s; R2, 3 min; R3, 5 min 30 s into the recovery period. Data are presented as means ± SE. *Significant difference from baseline, P < 0.05.
Renal Medullary and Cortical Oxygenation Responses During LBNP and Recovery
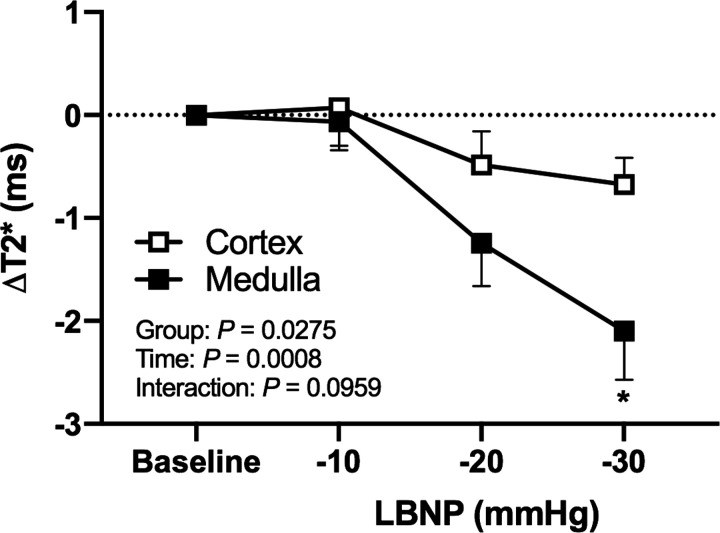
For the changes in renal medullary and cortical T2*, two-way repeated ANOVA revealed no interaction (P = 0.0959, effect size f = 2.256), but significant main effects for both vascular territories (P = 0.0275, effect size f = 6.054) and the level of LBNP (P = 0.0008, effect size f = 9.959; Fig. 4, n = 8) was observed. Specifically, renal medulla T2* was significantly decreased at −30 mmHg of LBNP (Δ−2.10 ± 0.47%, P = 0.009). Unlike the RBV recovery pattern, renal medulla and cortical T2* did not return to baseline until the second minute of recovery (Fig. 5B).
Figure 4.
Change from baseline for renal cortex T2* and renal medulla T2* response to lower body negative pressure (LBNP; −10 mmHg, −20 mmHg, −30 mmHg; n = 8). Two-way repeated ANOVA was performed and showed significant main effects for both the vascular territory (group; P = 0.0275) and the level of LBNP (time; P = 0.0008). Following post hoc pairwise comparison (3 comparisons) with the Bonferroni method for the significant main effects revealed a significant reduction at −30 mmHg of LBNP (P = 0.008 vs. baseline) for changes in medullary T2*. Data are presented as means ± SE. *Significant difference from baseline, P < 0.05.
DISCUSSION
Summary of Main Findings
The primary objective of this study was to use noninvasive and nonpharmacological methods to measure renal oxygenation of healthy human subjects as the sympathetic nervous system was activated with baroreceptor unloading. The findings from this study revealed that renal cortical and medullary oxygenation as well as renal arterial blood flow are significantly reduced during sympathetic activation with LBNP. These findings suggest that an acute moderate level of sympathetic activation reduces oxygen delivery to the renal cortex and medulla. Thus, we concluded that, in humans, sympathetic activation at relatively physiological levels of baroreceptor engagement acutely reduces renal blood flow and leads to tissue deoxygenation.
Potential Mechanisms
Similar to the previous findings (16, 18, 32), a significant reduction in renal arterial blood flow and a significant increase in renal vascular resistance are measured from this study which confirm that the sympathetic activation via LBNP results in robust renal vasoconstriction in healthy young adults. It is presumed that this is due to an increase in sympathetic activity directed to the kidney (16, 17, 33–36). Moreover, we suspect renal blood flow to the kidney does not fall significantly until the sympathetic activation level of −30 mmHg of LBNP because of powerful renal autoregulatory influences (37) preventing a reduction in blood flow at sympathetic stimulus less than that level, −10 and −20 mmHg of LBNP. Along with the renal vasoconstriction and the fall in tissue oxygenation, post hoc comparisons suggested a significant reduction in renal medullary oxygenation at −30 mmHg of LBNP. This observation suggests that the kidney becomes hypoxic during orthostatic stress. The most probable explanation for the significant reduction in renal oxygenation with LBNP is decreased oxygen delivery to the kidneys due to the increase in renal vasoconstriction and thus reduced GFR. Interestingly, significant changes in renal medullary oxygenation and renal arterial blood flow occur at the same level of sympathetic activation (−30 mmHg of LBNP) suggesting that physiological graded activation is not seen in humans. Prior work by DiBona and colleagues suggests that renal reflex effects occur as a function of the frequency of renal sympathetic nerve stimulation, called “staged renal reflex theory.” They reported that low frequencies of renal sympathetic nerve activation resulted in renin release, where higher frequencies resulted in increases in sodium reabsorption and the highest frequencies resulted in decreases in renal blood flow (38). In contrast to a staged renal reflex theory suggested by DiBona and colleagues in an animal model, our data in humans suggests that the sympathetically mediated activation timing of renal blood flow and medullary oxygenation changes are unlikely to occur in such a graded fashion.
Another finding from the current study is the recovery time course for RBV and medullary T2*. The time course of RBV and T2* during recovery from LBNP are different; immediately following termination of the LBNP application, RBV returned promptly to baseline whereas T2* stayed below baseline for an additional minute of recovery (R1: 30 s into the recovery, Fig. 5). This delayed recovery pattern of T2* may be explained, at least in part, by overall oxygen debt accumulated during the period of relative ischemia induced by the LBNP that has to be replenished. However, this study was not designed to investigate the magnitude of renal oxygen debt following relative ischemia, thus future research is needed to elucidate the underlying mechanisms for differences in the recovery time course of RBV and renal oxygenation.
Our heart rate findings during graded LBNP are consistent with previous literature (16, 18, 36). Our MAP and RBV responses are consistent with a previous report from this laboratory (36). However, Tidgren et al. (16) demonstrated a significant reduction in MAP at −27 mmHg of LBNP, and significant reductions in RBV were noted at −18 mmHg of LBNP. We did not note a fall in RBV at −20 mmHg or a fall in MAP at −30 mmHg. These differing results could be explained by differences in the samples studied (all men vs. both men and women) and the techniques used to measure renal blood flow (thermodilution vs. Doppler ultrasound).
Limitations
T2* reflects not only the absolute amount of deoxy-hemoglobin per tissue volume, but also blood vessel volume fraction and the local hematocrit. Due to the lower blood vessel volume fraction and hematocrit (39), and the higher water content in the medulla, it is more difficult to detect changes in cortical than medullary oxygenation (40, 41). However, despite the difficulty, we observed significant decreases in both cortical and medullary T2* with graded LBNP (main effects for the level of LBNP P = 0.0008). Although actual sodium excretion was not measured in our study, we used a surrogate noninvasive method that has excellent time resolution. Actual sodium excretion studies would require bladder catheterization, longer LBNP stages, and thus longer periods of investigation. Also, because of the complex measurements in our studies, the order of LBNP interventions could not be randomized. We acknowledge this limitation but note that previous work has also used a nonrandomized application at different levels of LBNP to study the effects of baroreceptor unloading (16, 42, 43). In addition, renal or vasoactive hormone, which may have contributed to the renal blood flow, renal blood flow distribution, and sodium reabsorption was not measured in the current study. Another limitation of this study is that no measure of sympathetic nerve activity was collected. With current clinical and research techniques, it is impossible to directly measure renal nerve activity in humans. However, the application of LBNP has been shown to increase muscle sympathetic nerve activity (44, 45). We also acknowledge that we do not have any kidney function measures before experimental testing. However, we studied healthy young human subjects who are unlikely to have renal dysfunction. Additionally, the focus of this study was to examine the effects of sympathetic activation on tissue oxygenation. Lastly, we acknowledge that RBV measured with Doppler ultrasound can be highly variable. To confirm the reproducibility of the measurement, a repeated visit was performed for RBV. The coefficient of variation for baseline RBV of visit 1 was 50.88%, whereas that of the repeated visit was 28.43%. More importantly, we performed the two-way repeated ANOVA to test the reproducibility of detecting changes in RBV with LBNP. The reproducibility test revealed that the RBV measured during the two different days were not significantly different (P = 0.762, Supplemental Fig. S1). Additionally, although the measured variables for renal flow velocity are somewhat variable, the general concept that LBNP lowers renal flow similarly on the two days of study is not in dispute.
In our studies, the subjects held their breath during MR imaging to enable acquiring high-quality images without motion artifacts. These breath-holds could influence sympathetic nervous system activation (46) and sympathetic effects on the kidney. However, we do not think that breath-holding affected the relationship between renal flow velocity and T2* as we were careful to perform identical breath-holds at each stage (baseline, LBNP, −10, −20, and −30) of the MR and Doppler imaging visits. Although RBV and medullary T2* were acquired on two different visits (visit 1, Doppler visit and visit 2, MRI visit), systemic hemodynamic response during LBNP was not different between visits (ΔMAP; main effect P = 0.429; Supplemental Fig. S2; see https://doi.org/10.6084/m9.figshare.10042916).
Conclusion
In this report, we demonstrate that −30 mmHg of LBNP, a stimulus that disengages aortic baroreceptors and enhances sympathetic outflow, led to a reduction in T2* and RBV. We suspect that the reduction in T2* is due to an increase in deoxyhemoglobin as sympathetic activation reduced oxygen delivery to the kidney by increasing renal vasoconstriction. Lastly, the recovery of renal medullary oxygenation following termination of LBNP did not occur immediately.
Implications
This report suggests that sympathetic activation alters renal oxygenation. Also, it confirmed the feasibility of a nonconventional and noninvasive renal function assessment method via the MRI BOLD technique during sympathetic activation. This study will provide a framework for future investigations to understand whether chronic activation of this system leads to: 1) a chronic salt retaining state and 2) a prediction to medullary ischemia and eventual fibrosis and chronic impairment in volume regulation. Additionally, this study will be a basis for future investigations to elucidate the link between sympathetic activation and renal hypoxia leading to renal complications in patients with heightened sympathetic activation (i.e., heart failure and hypertension).
GRANTS
This project is funded, in part, by a grant from the Pennsylvania Department of Health using Tobacco CURE Funds. The Department specifically disclaims responsibility for any analyses, interpretations, or conclusions (to L. I. Sinoway) and National Institutes of Health Grants P01 HL134609 and UL1 TR002014 (to L. I. Sinoway).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
AUTHOR CONTRIBUTIONS
D.J-K.K., R.C.D., C.T.S., Q.X.Y., M.D.H., and L.I.S. conceived and designed research; D.J-K.K., R.C.D., C.T.S., A.J.M., J.C., and M.D.H. performed experiments; D.J-K.K. and R.C.D. analyzed data; D.J-K.K., C.T.S., Q.X.Y., A.J.M., J.C., and L.I.S. interpreted results of experiments; D.J-K.K. prepared figures; D.J.K., C.T.S., and L.I.S. drafted manuscript; D.J-K.K., R.C.D., C.T.S., Q.X.Y., A.J.M., J.C., M.D.H., and L.I.S. edited and revised manuscript; D.J-K.K., R.C.D., C.T.S., Q.X.Y., A.J.M., J.C., M.D.H., and L.I.S. approved final version of manuscript.
ACKNOWLEDGMENTS
The authors thank Cheryl Blaha and Aimee Cauffman for nursing assistance and Jen Stoner and Kris Gray for the administrative support, Jeff Vesek for the MR imaging support, and the Center for NMR Research and the Heart and Vascular Institute at the Pennsylvania State University–College of Medicine for support.
REFERENCES
- 1.Crews DC, Bello AK, Saadi G. 2019 World Kidney Day Editorial – burden, access, and disparities in kidney disease. J Bras Nefrol 41: 1–9, 2019. doi: 10.1590/2175-8239-jbn-2018-0224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.GBD 2015 DALYs, HALE Collaborators. Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388: 1603–1658, 2016. doi: 10.1016/S0140-6736(16)31460-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Eckardt K-U, Coresh J, Devuyst O, Johnson RJ, Köttgen A, Levey AS, Levin A. Evolving importance of kidney disease: from subspecialty to global health burden. Lancet 382: 158–169, 2013. doi: 10.1016/S0140-6736(13)60439-0. [DOI] [PubMed] [Google Scholar]
- 4.Leung KC, Tonelli M, James MT. Chronic kidney disease following acute kidney injury-risk and outcomes. Nat Rev Nephrol 9: 77–85, 2013. doi: 10.1038/nrneph.2012.280. [DOI] [PubMed] [Google Scholar]
- 5.Barajas L, Liu L, Powers K. Anatomy of the renal innervation: intrarenal aspects and ganglia of origin. Can J Physiol Pharmacol 70: 735–749, 1992. doi: 10.1139/y92-098. [DOI] [PubMed] [Google Scholar]
- 6.Coote JH, Johns EJ, Macleod VH, Singer B. Effect of renal nerve stimulation, renal blood flow and adrenergic blockade on plasma renin activity in the cat. J Physiol 226: 15–36, 1972. doi: 10.1113/jphysiol.1972.sp009971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kopp U, Aurell M, Nilsson IM, Ablad B. The role of beta-1-adrenoceptors in the renin release response to graded renal sympathetic nerve stimulation. Pflugers Arch 387: 107–113, 1980. doi: 10.1007/BF00584260. [DOI] [PubMed] [Google Scholar]
- 8.Osborn JL, DiBona GF, Thames MD. Beta-1 receptor mediation of renin secretion elicited by low-frequency renal nerve stimulation. J Pharmacol Exp Ther 216: 265–269, 1981. [PubMed] [Google Scholar]
- 9.Bello-Reuss E, Colindres RE, Pastoriza-Muñoz E, Mueller RA, Gottschalk CW. Effects of acute unilateral renal denervation in the rat. J Clin Invest 56: 208–217, 1975.doi: 10.1172/JCI108069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hesse IF, Johns EJ. The effect of graded renal nerve stimulation on renal function in the anaesthetized rabbit. Comp Biochem Physiol A Comp Physiol 79: 409–414, 1984.doi: 10.1016/0300-9629(84)90536-X. [DOI] [PubMed] [Google Scholar]
- 11.Johns EJ, Manitius J. An investigation into the neural regulation of calcium excretion by the rat kidney. J Physiol 383: 745–755, 1987. doi: 10.1113/jphysiol.1987.sp016439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.La Grange RG, Sloop CH, Schmid HE. Selective stimulation of renal nerves in the anesthetized dog. Effect on renin release during controlled changes in renal hemodynamics. Circ Res 33: 704–712, 1973. doi: 10.1161/01.RES.33.6.704. [DOI] [PubMed] [Google Scholar]
- 13.Johns EJ, Lewis BA, Singer B. The sodium-retaining effect of renal nerve activity in the cat: role of angiotensin formation. Clin Sci Mol Med 51: 93–102, 1976. doi: 10.1042/cs0510093. [DOI] [PubMed] [Google Scholar]
- 14.Johns EJ. Angiotensin II in the brain and the autonomic control of the kidney. Exp Physiol 90: 163–168, 2005. doi: 10.1113/expphysiol.2004.029025. [DOI] [PubMed] [Google Scholar]
- 15.Johns EJ. The autonomic nervous system and pressure-natriuresis in cardiovascular-renal interactions in response to salt. Clin Auton Res 12: 256–263, 2002.doi: 10.1007/s10286-002-0050-x. [DOI] [PubMed] [Google Scholar]
- 16.Tidgren B, Hjemdahl P, Theodorsson E, Nussberger J. Renal responses to lower body negative pressure in humans. Am J Physiol 259: F573–F579, 1990. doi: 10.1152/ajprenal.1990.259.4.F573. [DOI] [PubMed] [Google Scholar]
- 17.Würzner G, Chiolero A, Maillard M, Nussberger J, Hayoz D, Brunner HR, Burnier M. Renal and neurohormonal responses to increasing levels of lower body negative pressure in men. Kidney Int 60: 1469–1476, 2001.doi: 10.1046/j.1523-1755.2001.00950.x. [DOI] [PubMed] [Google Scholar]
- 18.Clark CM, Monahan KD, Drew RC. Aging augments renal vasoconstrictor response to orthostatic stress in humans. Am J Physiol Regul Integr Comp Physiol 309: R1474–R1478, 2015. doi: 10.1152/ajpregu.00291.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Julius S, Nesbitt S. Sympathetic overactivity in hypertension. A moving target. Am J Hypertens 9: 113S–120S, 1996. doi: 10.1016/0895-7061(96)00287-7. [DOI] [PubMed] [Google Scholar]
- 20.Narkiewicz K, Somers VK. The sympathetic nervous system and obstructive sleep apnea: implications for hypertension. J Hypertens 15: 1613–1619, 1997. doi: 10.1097/00004872-199715120-00062. [DOI] [PubMed] [Google Scholar]
- 21.Triposkiadis F, Karayannis G, Giamouzis G, Skoularigis J, Louridas G, Butler J. The sympathetic nervous system in heart failure physiology, pathophysiology, and clinical implications. J Am Coll Cardiol 54: 1747–1762, 2009. doi: 10.1016/j.jacc.2009.05.015. [DOI] [PubMed] [Google Scholar]
- 22.Fu Q, Colgan SP, Shelley CS. Hypoxia: the force that drives chronic kidney disease. Clin Med Res 14: 15–39, 2016. doi: 10.3121/cmr.2015.1282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chavhan GB, Babyn PS, Thomas B, Shroff MM, Haacke EM. Principles, techniques, and applications of T2*-based MR imaging and its special applications. Radiographics 29: 1433–1449, 2009.doi: 10.1148/rg.295095034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Niendorf T, Pohlmann A, Arakelyan K, Flemming B, Cantow K, Hentschel J, Grosenick D, Ladwig M, Reimann H, Klix S, Waiczies S, Seeliger E. How bold is blood oxygenation level-dependent (BOLD) magnetic resonance imaging of the kidney? Opportunities, challenges and future directions. Acta Physiol (Oxf) 213: 19–38, 2015. doi: 10.1111/apha.12393. [DOI] [PubMed] [Google Scholar]
- 25.Prasad PV, Edelman RR, Epstein FH. Noninvasive evaluation of intrarenal oxygenation with BOLD MRI. Circulation 94: 3271–3275, 1996. doi: 10.1161/01.CIR.94.12.3271. [DOI] [PubMed] [Google Scholar]
- 26.Tumkur S, Vu A, Li L, Prasad PV. Evaluation of intrarenal oxygenation at 3.0 T using 3-dimensional multiple gradient-recalled echo sequence. Invest Radiol 41: 181–184, 2006. doi: 10.1097/01.rli.0000187166.43871.fb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vivier PH, Storey P, Chandarana H, Yamamoto A, Tantillo K, Khan U, Zhang JL, Sigmund EE, Rusinek H, Babb JS, Bubenheim M, Lee VS. Renal blood oxygenation level-dependent imaging: contribution of R2 to R2* values. Invest Radiol 48: 501–508, 2013. doi: 10.1097/RLI.0b013e3182823591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Niendorf T, Pohlmann A, Reimann HM, Waiczies H, Peper E, Huelnhagen T, Seeliger E, Schreiber A, Kettritz R, Strobel K, Ku M-C, Waiczies S. Advancing cardiovascular, neurovascular, and renal magnetic resonance imaging in small rodents using cryogenic radiofrequency coil technology. Front Pharmacol 6: 255, 2015. doi: 10.3389/fphar.2015.00255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Elkayam U, Mehra A, Cohen G, Tummala PP, Karaalp IS, Wani OR, Canetti M. Renal circulatory effects of adenosine in patients with chronic heart failure. J Am Coll Cardiol 32: 211–215, 1998. doi: 10.1016/S0735-1097(98)00191-0. [DOI] [PubMed] [Google Scholar]
- 30.Marraccini P, Fedele S, Marzilli M, Orsini E, Dukic G, Serasini L, L'Abbate A. Adenosine-induced renal vasoconstriction in man. Cardiovasc Res 32: 949–953, 1996. doi: 10.1016/s0008-6363(96)00128-9. [DOI] [PubMed] [Google Scholar]
- 31.Tumkur SM, Vu AT, Li LP, Pierchala L, Prasad PV. Evaluation of intra-renal oxygenation during water diuresis: a time-resolved study using BOLD MRI. Kidney Int 70: 139–143, 2006. doi: 10.1038/sj.ki.5000347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.van der Bel R, Verbree J, Gurney-Champion OJ, van Osch MJP, Stroes ESG, Nederveen AJ, Krediet CTP. Sympathetic activation by lower body negative pressure decreases kidney perfusion without inducing hypoxia in healthy humans. Clin Auton Res 30: 149–156, 2020. doi: 10.1007/s10286-018-0570-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Berdeaux A, Duranteau J, Pussard E, Edouard A, Giudicelli JF. Baroreflex control of regional vascular resistances during simulated orthostatism. Kidney Int Suppl 37: S29–S33, 1992. [PubMed] [Google Scholar]
- 34.Gilbert CA, Bricker LA, Springfield WT Jr, Stevens PM, Warren BH. Sodium and water excretion and renal hemodynamics during lower body negative pressure. J Appl Physiol 21: 1699–1704, 1966. doi: 10.1152/jappl.1966.21.6.1699. [DOI] [PubMed] [Google Scholar]
- 35.Miller JA, Floras JS, Skorecki KL, Blendis LM, Logan AG. Renal and humoral responses to sustained cardiopulmonary baroreceptor deactivation in humans. Am J Physiol 260: R642–R648, 1991. doi: 10.1152/ajpregu.1991.260.3.R642. [DOI] [PubMed] [Google Scholar]
- 36.Momen A, Thomas K, Blaha C, Gahremanpour A, Mansoor A, Leuenberger UA, Sinoway LI. Renal vasoconstrictor responses to static exercise during orthostatic stress in humans: effects of the muscle mechano- and the baroreflexes. J Physiol 573: 819–825, 2006.doi: 10.1113/jphysiol.2005.104612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Loutzenhiser R, Griffin K, Williamson G, Bidani A. Renal autoregulation: new perspectives regarding the protective and regulatory roles of the underlying mechanisms. Am J Physiol Regul Integr Comp Physiol 290: R1153–R1167, 2006. doi: 10.1152/ajpregu.00402.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.DiBona GF, Kopp UC. Neural control of renal function. Physiol Rev 77: 75–197, 1997.doi: 10.1152/physrev.1997.77.1.75. [DOI] [PubMed] [Google Scholar]
- 39.Edwards A, Silldforff EP, Pallone TL. The renal medullary microcirculation. Front Biosci 5: E36–E52, 2000. doi: 10.2741/edwards. [DOI] [PubMed] [Google Scholar]
- 40.Hueper K, Rong S, Gutberlet M, Hartung D, Mengel M, Lu X, Haller H, Wacker F, Meier M, Gueler F. T2 relaxation time and apparent diffusion coefficient for noninvasive assessment of renal pathology after acute kidney injury in mice: comparison with histopathology. Invest Radiol 48: 834–842, 2013. doi: 10.1097/RLI.0b013e31829d0414. [DOI] [PubMed] [Google Scholar]
- 41.Pohlmann A, Hentschel J, Fechner M, Hoff U, Bubalo G, Arakelyan K, Cantow K, Seeliger E, Flemming B, Waiczies H, Waiczies S, Schunck W-H, Dragun D, Niendorf T. High temporal resolution parametric MRI monitoring of the initial ischemia/reperfusion phase in experimental acute kidney injury. PLoS One 8: e57411, 2013. doi: 10.1371/journal.pone.0057411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Baily RG, Leuenberger U, Leaman G, Silber D, Sinoway LI. Norepinephrine kinetics and cardiac output during nonhypotensive lower body negative pressure. Am J Physiol 260: H1708–H1712, 1991. doi: 10.1152/ajpheart.1991.260.5.H1708. [DOI] [PubMed] [Google Scholar]
- 43.Baily RG, Prophet SA, Shenberger JS, Zelis R, Sinoway LI. Direct neurohumoral evidence for isolated sympathetic nervous system activation to skeletal muscle in response to cardiopulmonary baroreceptor unloading. Circ Res 66: 1720–1728, 1990. doi: 10.1161/01.res.66.6.1720. [DOI] [PubMed] [Google Scholar]
- 44.Khan MH, Sinoway LI, MacLean DA. Effects of graded LBNP on MSNA and interstitial norepinephrine. Am J Physiol Heart Circ Physiol 283: H2038–H2044, 2002.doi: 10.1152/ajpheart.00412.2001. [DOI] [PubMed] [Google Scholar]
- 45.Victor RG, Leimbach WN Jr.. Effects of lower body negative pressure on sympathetic discharge to leg muscles in humans. J Appl Physiol 63: 2558–2562, 1987. doi: 10.1152/jappl.1987.63.6.2558. [DOI] [PubMed] [Google Scholar]
- 46.Ebert TJ, Morgan BJ, Barney JA, Denahan T, Smith JJ. Effects of aging on baroreflex regulation of sympathetic activity in humans. Am J Physiol 263: H798–H803, 1992. doi: 10.1152/ajpheart.1992.263.3.H798. [DOI] [PubMed] [Google Scholar]