Abstract
Liver fibrosis should be assessed in all individuals with chronic liver disease as it predicts the risk of future liver-related morbidity and thus need for treatment, monitoring and surveillance. Non-invasive fibrosis tests (NITs) overcome many limitations of liver biopsy and are now routinely incorporated into specialist clinical practice. Simple serum-based tests (eg, Fibrosis Score 4, non-alcoholic fatty liver disease Fibrosis Score) consist of readily available biochemical surrogates and clinical risk factors for liver fibrosis (eg, age and sex). These have been extensively validated across a spectrum of chronic liver diseases, however, tend to be less accurate than more ‘complex’ serum tests, which incorporate direct measures of fibrogenesis or fibrolysis (eg, hyaluronic acid, N-terminal propeptide of type three collagen). Elastography methods quantify liver stiffness as a marker of fibrosis and are more accurate than simple serum NITs, however, suffer increasing rates of unreliability with increasing obesity. MR elastography appears more accurate than sonographic elastography and is not significantly impacted by obesity but is costly with limited availability. NITs are valuable for excluding advanced fibrosis or cirrhosis, however, are not sufficiently predictive when used in isolation. Combining serum and elastography techniques increases diagnostic accuracy and can be used as screening and confirmatory tests, respectively. Unfortunately, NITs have not yet been demonstrated to accurately reflect fibrosis change in response to treatment, limiting their role in disease monitoring. However, recent studies have demonstrated lipidomic, proteomic and gut microbiome profiles as well as microRNA signatures to be promising techniques for fibrosis assessment in the future.
INTRODUCTION
Despite the considerable regenerative capacity of the liver, chronic injury leads to the development of hepatic fibrosis. Fibrosis occurs as a gradient of severity, which increases in the presence of continuing insult, but may also reverse with removal of the injurious agent or infection.1 The degree of liver fibrosis in patients with chronic liver disease (CLD) predicts the likelihood of developing liver-related morbidity and death.2 Thus, assessment of liver fibrosis is an essential part of the evaluation of any patient with CLD in order to prognosticate, stratify therapeutic and surveillance strategies and evaluate response to treatment over time. In addition, fibrosis has been used as a key surrogate end point for clinical trials in patients with CLD allowing expedited approval of efficacious drug treatment.
The optimal method for evaluating liver fibrosis should be accurate (precise in its measurement), reproducible (providing the same result on repeated measurements) and dynamic (responsive to change in fibrosis levels over time). Additional important characteristics include acceptability to the patient and physician, accessibility and cost-effectiveness. Liver biopsy provides a direct measure of liver fibrosis, however, has well-described limitations of invasiveness with limited patient and physician acceptability, interobserver and intraobserver variability and cost. The potential complications of the procedure include pain, infection, bleeding, perforation of the organs near the liver and extremely rarely, even death. In addition, current histopathology fibrosis staging systems provide only a semiquantitative measure of fibrosis, which may not be sensitive to subtle changes in fibrosis over time. Non-invasive tests (NITs) including blood-based biomarkers and imaging techniques, such as elastography, overcome a number of these limitations and are generally preferred by patients and physicians in routine clinical practice.
NITs have been available for nearly two decades, and are already routinely incorporated into clinical practice in many centres. Nonetheless, data are still emerging regarding the optimal way to use these tests (screening vs diagnosis, single vs multiple, combination of tests together or sequential). Many tests continue to be refined and their pitfalls and limitations as well as their role in monitoring fibrosis over time or in response to treatment are currently being defined. In addition, recent innovations in elastography, imaging and omics methods offer the potential for increased diagnostic accuracy and will be discussed in this review.
GENERAL PRINCIPLES
Non-invasive fibrosis methods provide a continuous measure from which a cut-off is chosen to predict a binary degree of liver fibrosis, such as advanced (bridging) fibrosis or cirrhosis. Under standardised conditions, the accuracy of these methods is typically robust for advanced fibrosis/cirrhosis, however, the diagnostic characteristics vary significantly depending on the cut-off value. Typically, a ‘high’ cut-off value provides greater specificity for advanced fibrosis and cirrhosis, and a ‘low’ cut-off provides greater sensitivity for no or minimal fibrosis. Due to the generally low prevalence (or pretest probability) of advanced fibrosis and cirrhosis in the population being tested, the positive predictive value (PPV) of a result above the high cut-off is typically modest, and often not sufficient to be diagnostic in the absence of additional supportive clinical information. In contrast, the negative predictive value (NPV) of NITs is generally very high, allowing the clinician to be confident that advanced fibrosis or cirrhosis has been excluded.
BLOOD-BASED BIOMARKERS
Simple biomarker blood tests (eg, Fibrosis Score 4 (FIB-4), AST-Platelet Ratio Index (APRI), non-alcoholic fatty liver disease (NAFLD)) incorporate ‘indirect’ markers of liver fibrosis such as liver aminotransaminases, often with clinical parameters such as age and sex, to increase accuracy. ‘Complex’ biomarker blood tests (eg, Enhanced Liver Fibrosis Score (ELF), Hepascore, Fibrospect II) incorporate some of the direct markers of fibrogenesis and fibrinolysis (eg, serum tissue metalloproteinases and hyaluronic acid) and require specialist laboratory assessment but are generally more accurate than ‘simple’ biomarkers in predicting advanced fibrosis and cirrhosis (table 1).3–6
Table 1.
Comparison of blood-based biomarkers of liver fibrosis
| Biomarker | Components | Disease specificity | Validation | Accuracy* | Indeterminate Cases† | Prognosticability* |
|---|---|---|---|---|---|---|
| APRI | AST, platelets | CHC, NAFLD | +++ | +/++ | 50%–60% | + |
| FIB-4 | AST, ALT, platelets, age | CHC, NAFLD | +++ | +++ | 20%–30% | ++ |
| NAFLD fibrosis score | Age, BMI, IFG/diabetes, AST, ALT, platelets, albumin | NAFLD | +++ | +++ | 20%–35% | ++ |
| Fibrotest/fibrosure | Age, sex, bilirubin, GGT, α2M, haptoglobin, apo-A1 | CHC, CHB, ALD, NAFLD | +++ | +++‡ | 0%–35% | ++++ |
| Hepascore | Age, sex, bilirubin, GGT, α2M, HA. | CHC, CHB, ALD, NAFLD | +++ | ++++ | 0%–30% | ++++ |
| Fibrospect (CHC) | α2M, HA, TIMP-1 | CHC | ++ | +++‡ | 0% | NA |
| Fibrospect (NASH) | α2M, HA, TIMP-1 | NAFLD | + | ++++ | 0%–40% | NA |
| FibroMeterv2G (virus) | Age, sex, platelets, ALT, AST, GGT, PTI, urea, α2M, | CHC, CHB | ++ | +++ | 0% | ++++ |
| FibroMeter (SNAFFLED) | Age, sex, weight, platelets, ALT, AST, ferritin, glucose | NAFLD | ++ | ++ | 0%–35% | NA |
| Enhanced liver fibrosis score | HA, TIMP-1, PNPIII | CHC, CHB, ALD, NAFLD, PSC | +++ | ++++/+++++ | 0%–40% | ++++ |
AUC ≥0.90
AUC 0.85–0.89
AUC 0.80–0.84
AUC 0.75–0.79
AUC <0.75 (Harrell’s c index substituted for AUC for prognostic category).
Accuracy for determining advanced (bridging) fibrosis abstracted from meta-analyses where available7,9,12,118 or from large cohorts of chronic liver disease patients.26,59,74,119–123
Proportion of cases falling within an indeterminate range for the prediction of advanced fibrosis.6,38,72,119,120,124–127
AUC for significant (F2–4) fibrosis.
ALD, alcoholic liver disease; ALT, alanine aminotransferase; Apo-A1, apolipoprotein-A1; APRI, AST-Platelet Ratio Index; AST, aspartate aminotransferase; AUC, area under the curve; BMI, body mass index; CHB, Chronic hepatitis B; CHC, chronic hepatitis C; HA, hyaluranic acid; IFG, impaired fasting glucose; α2-M, α2-macroglobulin; NA, not applicable; NAFLD, non-alcoholic fatty liver disease; PNPIII, procollagen N-terminal peptide III; PSC, primary sclerosing cholangitis; PTI, prothrombin index; TIMP-1, tissue inhibitor of matrix metalloproteinase-1.
The accuracy of blood-based NITs varies according to the underlying aetiology of CLD. Several serum tests are specific for particular aetiologies of CLD; FibroMeter and Fibrospect II, have hepatitis virus and NAFLD specific algorithms, and the NAFLD Fibrosis Score is specific for NAFLD. Other tests have been developed in patients with chronic hepatitis C (CHC) (eg, Fibrotest, Hepascore) but are accurate in other liver disease groups.7 APRI was developed in CHC patients and consists of readily available parameters (aspartate aminotransaminase, platelet count) and has good accuracy for advanced fibrosis (summary area under the curve (AUC) 0.80) in this population,8 but performs modestly in NAFLD (summary AUC 0.77),9 and is less reliable in chronic hepatitis B (CHB)10 and alcoholic liver disease (ALD).11 FIB-4 is also composed of aminotransaminase levels and platelets and was developed in CHC/HIV coinfected patients and has been validated in CHC and NAFLD. The ELF test is composed of three direct markers of fibrogensis/lysis and has been validated as an accurate predictor (AUC >0.85) of advanced fibrosis in patients with mixed aetiologies of CLD with the exception of patients with CHB.12,13
Confounding factors need to be excluded when interpreting blood-based NITs, particularly significant liver and systemic inflammation, which may increase blood biomarker levels independently of fibrosis stage.14 Biomarkers incorporating bilirubin (Hepascore, Fibrotest) may be falsely increased in the setting of Gilbert or haemolysis. In addition, FIB-4 and NAFLD Fibrosis Score may be less accurate in individuals <35 years (though the prevalence of advanced fibrosis is low in this group) and become less specific with increasing age, with higher cut-offs proposed to exclude advanced fibrosis in those >65 years.15 ELF also increases with age, although revised cut-offs are not recommended at this time.16
ELASTOGRAPHY
Elastography techniques take advantage of the physical properties of liver fibrosis that make the liver ‘stiffer’ by quantifying ‘sheer wave’ velocity or tissue displacement generated by an ultrasonic or physical impulse. Vibration-controlled transient elastography (VCTE or Fibroscan) and MR elastography (MRE) use a mechanical driver to generate the sheer wave and measure its velocity using sonographic Doppler or MR techniques, respectively. Point sheer wave elastography (pSWE or acoustic radiation force impulse) and two-dimensional SWE (2D-SWE) use high frequency sonographic impulses for sheer wave generation. pSWE measures the shear wave generated from one sonographic frequency in metres/second whereas 2D-SWE measures sonographic waves in multiple frequencies in real-time using 2D ultrasound in kilopascals (kPa). Lastly, real-time (strain) elastography uses standard ultrasound to measure liver tissue displacement (or strain) induced by a sonographic probe or by cardiac impulse. Due to the different methodology used between technologies, elastography values between different techniques are not comparable.
Sonographic elastography techniques
VCTE was the first elastography technique to be commercialised and thus has had extensive validation and evaluation of its strengths and limitations in comparison with other methods (tables 2 and 3). Liver stiffness measurement (LSM) by VCTE may be increased by acute hepatitis and cholestasis, respiration, congestive cardiac failure, recent food and excess alcohol ingestion and increasing body mass index (BMI).17–19 Confounding factors for other elastography techniques are less defined, however, are likely to be similar. Using VCTE with the obesity-specific (XL) probe, inability to scan (ie, scan failure) or unreliable scans occur in 3%–14% and 1%–9% of patients, respectively, and are more likely with significantly obese patients and inexperienced operators.20–24 Approximately 30% of obese patients had either unreliable or invalid scans in a prospectively evaluated cohort of 291 patients with NAFLD irrespective of whether VCTE, p-SWE or 2D-SWE was used.23 Intraobserver agreement for VCTE is excellent (intraclass correlation coefficient 0.98), though is lower with lesser degrees of fibrosis, increasing steatosis and BMI.25 pSWE has a very low scan failure rate (0%–1%), however, is unreliable in 16%–24% of subjects24,26,27 and has a learning curve, with intraobserver agreement increasing after 130 examinations.28 2D-SWE does not have validated reliability criteria and thus invalid scans are typically not reported though has a failure rate of 1%–13%, being lower in patients with CHB and higher in patients with NAFLD.23,24,29,30 2D-SWE also requires a degree of radiological expertise compared with VCTE, with greater intraobserver variability noted in less experienced operators.31 In the absence of an obesity-specific probe, increasing BMI appears to be a significant limitation for both point and 2D SWE techniques, with unreliable or invalid scans being reported in approximately 30% of obese (BMI >30 kg/ m2) patients with NAFLD and unreliable pSWE scans reported in >50% of patients when the skin to liver capsule distance is ≥30 mm.23,32,33 Increasing BMI also reduces accuracy of VCTE with AUC values for determining advanced fibrosis falling to <0.80 in morbidly obese (BMI ≥35 kg/m2).17 Real-time elastography has been criticised for observer variability stemming from its qualitative nature and has limited validation.34
Table 2.
Comparison of elastography techniques in the prediction of liver fibrosis
| Elastography technique | Validated disease groups | Accuracy† | Reliability (ICC) | Prognostic ability* | Reliability criteria | Indeterminate cases† |
|---|---|---|---|---|---|---|
| VCTE | CHC, CHB, NAFLD, ALD, PBC, PSC | +++/++++ | 0.99 | NAFLD, CHC, PBC, PSC | Yes | 30%–40% |
| pSWE | HCV, NAFLD | ++/++++ | 0.98 | NA | Yes | NA |
| 2D-SWE | HCV, HBV, NAFLD | +++++ | 0.98–1.0 | NA | No | NA |
| 2D-MRE | CHC, CHB, NAFLD | +++++ | 0.99 | PSC, cirrhosis | Yes | NA |
+++++AUC ≥0.90
++++AUC 0.85–0.89
+++AUC 0.80–0.84
++AUC 0.75–0.79
+AUC <0.75 (Harrell’s c index substituted for AUC for prognostic category).
Accuracy for determining advanced (bridging) fibrosis abstracted from meta-analyses where available or large patient cohorts. 24,27,54,55,129
Proportion of cases falling within an indeterminate range for the prediction of advanced fibrosis.124,130
ALD, alcoholic liver disease; AUC, area under the curve; CHB, Chronic hepatitis B; CHC, chronic hepatitis C; 2D-MRE, 2-dimensional MR elastography; 2D-SWE, 2-dimensional shear wave elastography; HBV, hepatitis B virus infection; HCV, hepatitis C virus infection; ICC, Intraclass correlation coefficient; NA, not applicable; NAFLD, nonalcoholic fatty liver disease; PBC, primay biliary cholangitis; pSWE, point shear wave elastography; VCTE, Vibration-controlled transient elastography.
Table 3.
Limitations of blood-based biomarkers and elastography techniques for the prediction of liver fibrosis
| Fibrosis marker | Failure rate | Factors related to failure | Invalid/unreliable result rate | Confounders |
|---|---|---|---|---|
| Indirect blood-based biomarkers | Negligible | - | 30% Indeterminate (FIB-4, NAFLD Fibrosis Score) | Acute hepatitis, cholestasis, systemic inflammation, Gilberts/hemolysis (scores with bilirubin) |
| Direct blood-based biomarkers | Negligible | - | ? | Acute hepatitis, systemic inflammation |
| VCTE | 3%–14% | Obesity (less with XL probe), ascites | 1%–9% | Acute hepatitis, cholestasis, beta-blockers, food ingestion, obesity, cardiac congestion. |
| pSWE | 0%–1% | Obesity | 16%–24% | Acute hepatitis, food ingestion, obesity* |
| 2D-SWE | 1%–13% | Obesity | 0% | Acute hepatitis, food ingestion* |
| 2D-MRE | <5% | Claustraphobia, inability to fit in MRI or breath hold, | Negligible | Iron overload, acute hepatitis, massive ascites |
Additional confounding factors for VCTE also likely to impact SWE.
2D-MRE, 2-dimensional MR elastography; 2D-SWE, 2-dimensional shear wave elastography; FIB-4, Fibrosis Score 4; NAFLD, non-alcoholic fatty liver disease; pSWE, point shear wave elastography; VCTE, Vibration-controlled transient elastography.
Cut-offs are variable between aetiologies of liver disease and not universally accepted within causes of liver disease, however, low readings (VCTE <6.0 kPa or Aixplorer 2D-SWE <7.1 kPa) reliably exclude advanced fibrosis and cirrhosis.35 Elevated readings may be falsely high and repeating VCTE within 6 months of a high reading can increase the certainty of advanced fibrosis or cirrhosis.36 Nonetheless, the predictive value of VCTE increases as LSM increases, with readings >20 kPa highly suggestive of cirrhosis and raising the possibility of significant portal hypertension. The current Baveno VI consensus suggests an LSM threshold of 20 kPa or platelet count <150 for endoscopic screening for varices.37
Overall, 2D-SWE appears to have comparative accuracy to VCTE in ALD and NAFLD, but greater accuracy in other aetiologies, particularly among patients with CHB.9,23,24,35,38 Prospective studies comparing VCTE and pSWE are limited, however, suggest no significant difference in accuracy.23,24,28,39 Further prospective comparative studies are required to confirm the relative strengths and limitations of these elastography techniques, although one advantage of the SWE techniques is their ready application to conventional ultrasound machines, whereas VCTE can be used as a point-of care test.
MR elastography
MRE examines whole sections of liver and thus is less prone to sampling error and has a low technical failure rate (≤5%), although is higher in patients with massive ascites.40–42 A newly developed spin-echo echo-planar sequence overcomes previous difficulties caused by significant hepatic iron.43 Reports of the impact of obesity on successful MRE acquisition are conflicting,40,44 however, it appears less problematic in comparison with VCTE with successful scans recorded in 96% and 88%, respectively, of patients with severe obesity (BMI ≥35 kg/m2).45 MRE can be performed on different MRI machine models and tesla strengths46 and has robust reproducibility between radiologists.47 Experience with MRE is limited in comparison to VCTE, however, studies in patients with predominately chronic viral hepatitis or NAFLD, have demonstrated it has excellent accuracy for the prediction of significant fibrosis, advanced fibrosis and cirrhosis with AUC values consistently above 0.90.48 Obesity, hepatic inflammation and degree of steatosis does not impact on accuracy in NAFLD, however, increased LSMs are observed with hepatic inflammation in chronic viral hepatitis B and C.49–52 The number of comparative studies examining MRE and sonographic elastography techniques is also limited, however, data to date demonstrates MRE to have significantly greater accuracy than VCTE and pSWE in NAFLD,53 and is more accurate than VCTE in CHB.9,54,55 Other MR techniques using diffusion weighted imaging or contrast have also been assessed as diagnostic tests for liver fibrosis, however, appear to be less accurate than MRE.56,57
BLOOD-BASED BIOMARKERS VERSUS ELASTOGRAPHY Accuracy
Overall, ultrasound elastography techniques have greater accuracy than simple ‘direct’ blood biomarkers (APRI, FIB-4, BARD) for the prediction of cirrhosis in chronic viral hepatitis, ALD and NAFLD.6,58–62 Blood-based markers have the advantage of a negligible failure rate and reliability that is not impacted by increasing BMI (table 3). However, in the setting of a reliable scan, VCTE and 2D-SWE have greater accuracy than ‘direct’ blood-based biomarkers (ELF, Fibrotest, Hepascore) for the prediction of advanced fibrosis and cirrhosis across a range of CLDs.38,62–65 When patients with unreliable scans are included on an ‘intention to diagnose’ basis, the accuracy to predict advanced fibrosis in patients with ALD is similar between ELF and Fibrotest, VCTE and 2D-SWE.5,38 MRE has greater accuracy than indirect blood markers in NAFLD and chronic viral hepatitis.66–69 Meta-analyses of NAFLD cohorts have demonstrated MRE to have the highest accuracy for fibrosis prediction, however, few studies with direct comparisons were available.54 Further prospective comparative studies involving MRE, sonographic elastography methods and direct blood biomarkers are required.
COMBINATION ASSESSMENT
In general, blood-based biomarkers and elastography techniques are excellent at excluding advanced fibrosis and cirrhosis with high NPVs (>85%) but have modest ability to diagnose cirrhosis with PPVs between 40% and 70%.6,58 In addition, NITs may have upper and lower cut-offs which are optimised to predict or exclude fibrosis, meaning some results fall within an indeterminate or grey zone (table 1). Approximately one-third of results of serum-based tests and VCTE may be indeterminate with indeterminate ranges for other elastography techniques yet to be well validated. Using concurrent serum NITs (Fibrotest, FIB-4, NFS) with VCTE increases diagnostic accuracy and specificity, with concordant results reliably excluding or confirming cirrhosis (NPV and PPV >90%), however, discordant results requiring a liver biopsy occur in 25%–70%.60,70–72 Sequential serum NITs, where a second test is used when the first is in the ‘grey zone’, have been examined in CHC, where the ‘SAFE’ algorithm (APRI followed by Fibrotest) avoids more biopsies but at the expense of lower accuracy and PPV (56%–78%) for the diagnosis of cirrhosis.70,71,73 Lastly, the combination of a serum NIT (FibroMeter second generation) and VCTE into one propriety algorithm (FibroMeterVCTE2G) has been demonstrated to have a high degree of accuracy (AUC >0.9) for the prediction of cirrhosis in cohorts of predominately viral hepatitis patients, although has added complexity and requires independent validation.74
POPULATION-BASED SCREENING
Using a serum NIT as an initial screening test followed by VCTE is an attractive algorithm for screening large populations. Serum NITs are widely available, inexpensive, applicable in obese patients and lend themselves into incorporation into clinical decision support systems and point-of-care testing where elastography techniques are not available.75 Blood-based biomarkers also predict risk of liver related death in the general population supporting their suitability as a screening test for liver disease.76 Nonetheless, their strength is excluding (rather than diagnosing) advanced fibrosis with poor agreement between serum NITs in predicting advanced fibrosis.77
Elastography techniques have greater accuracy for the diagnosis of cirrhosis than blood-based NITs and thus are suitable as confirmatory diagnostic tests (figure 1). Sonographic elastography is increasingly available in commercial radiology practices, although is less accurate than MRE which is expensive and limited to specialist centres. VCTE has been implemented in primary care screening programmes of subjects with or at risk of CLD and can identify patients with cirrhosis and those at risk of liver decompensation, however, dedicated machines, operator and specialist interpretation is required.21,78 One study screening 1358 subjects undergoing a community-based medical check-up with VCTE revealed 7.5% had elevated LSM (>8 kPa), of whom all had liver disease and 0.6% with previously undiagnosed cirrhosis.78 However, 12 of every 13 subjects in this population did not have liver disease (defined by VCTE) and 166 subjects were scanned for every cirrhotic found, suggesting implementation of a risk factor stratification strategy and/or initial serum NIT screening test is sensible. The optimal serum screening NIT is unclear, however, most experience is with non-propriety tests such as APRI, FIB-4 and NAFLD Fibrosis Score. Unfortunately, the sensitivity of these markers is typically <80%,8,54,74 raising the question whether more accurate ‘direct’ serum NITs would be a preferable first-line test at the cost of extra expense. In addition, age impacts the accuracy of simple indirect markers such as FIB-4 and NAFLD Fibrosis Score, limiting their utility in subjects >60 years.75,79
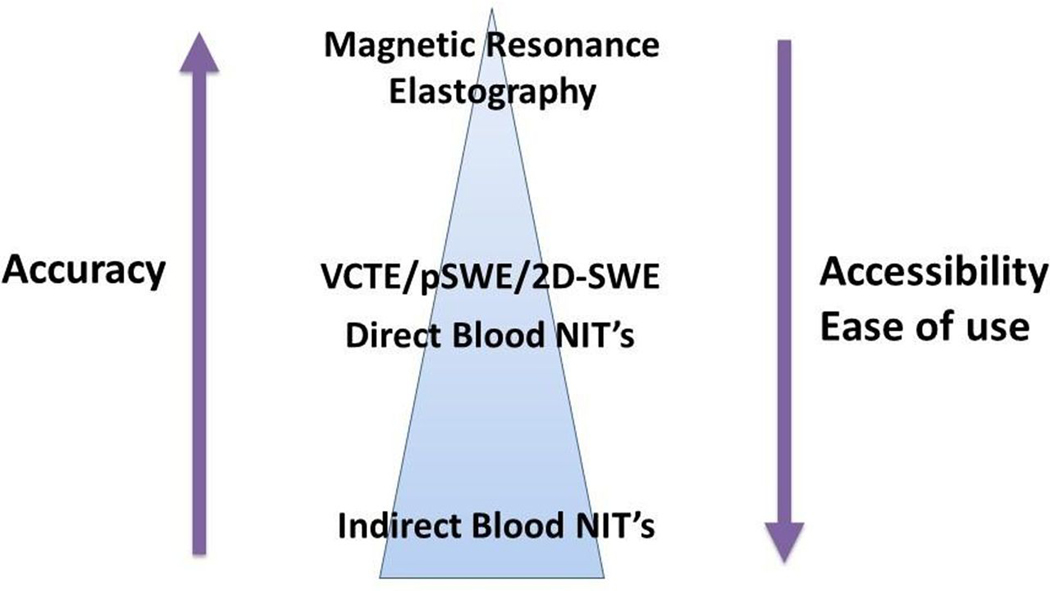
Figure 1.
Comparative accuracy and accessibility of non-invasive fibrosis tests (NITs). 2D-SWE, two-dimensional sheer wave elastography; pSWE, pulse shear wave elastography; VCTE, vibration-controlled transient elastography.
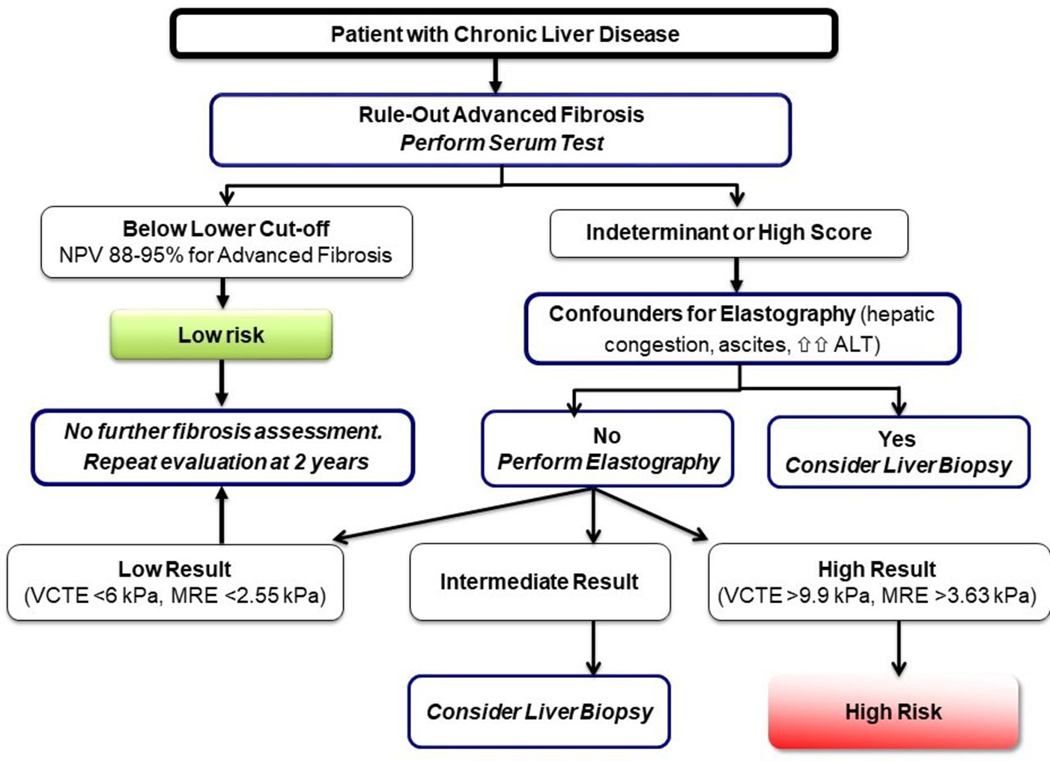
A serial screening strategy in general practice revolving around identification of risk factors for liver disease (hazardous alcohol use, type 2 diabetes or elevated ALT), followed by a simple indirect blood biomarker (BARD or AST/ALT ratio), found a normal blood biomarker excluded 12% of patients with liver disease risk factors from further investigation with VCTE. Following VCTE, a 3% prevalence of cirrhosis was found. Notably, APRI and FIB-4 would have missed 100% and 82% of the cirrhosis patients if substituted for VCTE highlighting the limitations of these simple serum NITs as confirmatory diagnostic tests.80 A similar stepwise approach has been suggested for general population based screening with a Spanish study of 3076 subjects recommending VCTE only in those who had risk factors for liver disease (58% of the population) and subsequently an elevated (≥60) fatty liver index (33% of the whole population). The prevalence of LSM ≥9.2 kPa was 8.7% in this subset (representing 2.8% of the whole population) suggesting that further refinement with a screening serum NIT may be beneficial.81 A suggested algorithm for fibrosis assessment in individuals with CLD using serial blood-based biomarkers and elastography is outlined in figure 2.
Figure 2.
Algorithm for assessment of advanced fibrosis in patients with chronic liver disease. A liver biopsy can be considered in the correct clinical context following an indeterminate or high serum test result in conjunction with a high elastography result as the positive predictive value for advanced fibrosis may be less than 80%. MRE, MR elastography; NPV, negative predictive value; VCTE, vibration-controlled transient elastography. ALT, alanine aminotransferase.
PREDICTION OF PROGNOSIS
Blood-based biomarkers predict hepatic decompensation and liver-related death in a range of CLD supporting their validity as diagnostic tests. ‘Complex’ blood biomarkers are generally more accurate than ‘simple’ biomarkers,59,82 however, these tests have limited discriminative ability for these long-term outcomes at an individual patient level. VCTE is more accurate in predicting outcomes than serum NITs (such as FIB-4), although when patients with unreliable scans are included on an ‘intention to diagnose’ basis, the accuracy to predict future liver-related events is similar.5,38 Emerging data suggest that ELF ≥9.8 is associated with higher risk of progression to cirrhosis in patients with bridging fibrosis due to NAFLD, and an ELF ≥11.3 is associated with higher risk of hepatic decompensation in patients with cirrhosis due to NAFLD.83 MRE also predicts future decompensation and survival in cirrhosis patients independently of MELD score, demonstrating its utility beyond diagnosing cirrhosis alone.84,85
Non-invasive assessment of antifibrotic treatment response
Experience in evaluating NITs in response to antifibrotic treatment is limited. Treatment of inflammatory CLD is typically associated with improvement of liver inflammation, which may in turn lead to reduction in liver elasticity and blood biomarkers. Consequently, NIT values tend to improve independently of fibrosis regression leading to a tendency to underestimate fibrosis stage, thereby reducing the utility of currently available NITs for assessment of short-term fibrosis response to treatment (table 4).86,87
Table 4.
Assessment of antifibrotic response by non-invasive tests in comparison to paired liver biopsies
| Study | Liver disease | Treatment | Non-invasive test | N, (range of fibrosis) | Follow-up | AUC for post-treatment fibrosis assessment | Comment |
|---|---|---|---|---|---|---|---|
| Kim et al10 | CHB | Tenofovir | Blood biomarkers | 298 | 4.6 years | NR | No correlation between change in APRI or FIB-4 and change in fibrosis over time. |
| Liang et al131 | CHB | Telbivudine | VCTE | 164 (FO-4) | 2 years | NR | At 2 years; LSM >9.0kPa had 22% PPV for advanced fibrosis; LSM <6.0 kPa had 100% NPV for advanced fibrosis |
| Dong et al132 | CHB | Entecavir | VCTE | 182 (FO-6*) | 1.5 years | For advanced fibrosis 0.85; for cirrhosis, 0.88 | Decline in LSM equivalent between those with fibrosis regression and stabilisation. |
| D’Ambrosio et al94 | CHC | IFN | Blood biomarkers | 38 (F4) | 5 years | For cirrhosis: APRI 0.58, FIB-4 0.59, Forns 0.56, ELF 0.63 | At Syears; PPV for cirrhosis 47%−100%. NPV 66%−78%. |
| Tachi et al133 | CHC | IFN | Blood biomarkers | 115 (FO-4) | 5 years | For advanced fibrosis: APRI 0.89, FIB-4 0.81, Forns 0.86 | At 5years, PPV for advanced fibrosis 29%−45%, NPV 96%−98%. |
| Tachi et al134 | CHC | IFN and DAA | pSWE | 140 (FO-4) | 5 years | NR | Follow-up LSM was lower in patients with baseline FI or 2 who had fibrosis regression, but not in those with advanced fibrosis who had fibrosis regression. |
| Pan et al99 | CHC | DAA | VCTE | 15 (F3–4) | 3 years | NR | At 3years; LSM >9.5 kPa had 100% PPV for advanced fibrosis, LSM <9.5 had 31 % NPV for advanced fibrosis |
| D’Ambrosio et al93 | CHC | IFN | VCTE | 33 (F4) | 5 years | For cirrhosis: 0.77 | At 5years; LSM &12.0kPa had 89% PPV for cirrhosis, LSM <12.0 had 79% NPV for cirrhosis. |
| Mauro et al87 | CHC post-LT | IFN and DAA | VCTE ELF | 112 (FI −4) | 1.5–2 years | For advanced fibrosis: VCTE 0.90, ELF 0.76 | At 2 years; 50% reduction in baseline LSM had 75% PPV and 44% NPV to predict fibrosis regression. AUC 0.65 |
| Chalasani et al88 | NAFLD | Obeticholic acid | Blood biomarkers | 200 (F0–3) | 1.5 years | NR | AUC for predicting fibrosis improvement was 0.72 (APRI), 0.68 (FIB-4) and 0.65 (NFS) when combined with baseline fibrosis stage, change in biomarker and treatment arm. PPV’s 36%−43%, NPV’s 89%−91%. |
| Vilar-Gomez et al90 | NAFLD | Lifestyle intervention | Blood biomarkers | 261 (F0–3) | lyear | NR | AUC for change in biomarker predicting fibrosis improvement was 0.65 (APRI), 0.65 (FIB-4) and 0.69 (NFS) |
| Jayakumar et al89 | NAFLD | Selonsertib | MRE | 54 (F2,3) | 24 weeks | For advanced fibrosis: 0.80 | Any reduction in MRE had PPV 48% and NPV 79% for fibrosis improvement. |
Ishak staging system.
APRI, AST-Platelet Ratio Index; AUC, area under the curve; CHB, chronic hepatitis B; CHC, chronic hepatitis C; DAA, direct acting antivirals; DAA, direct acting anti-viral treatment; ELF, enhanced liver fibrosis; FIB-4, Fibrosis Score 4; IFN, interferon; LSM, liver stiffness measurement; MRE, MR elastography; NAFLD, non-alcoholic fatty liver disease; NFS, NAFLD Fibrosis Score; NPV, negative predictive value; NR, nonresponder; post-LT, postliver transplant; PPV, positive predictive value; pSWE, point-sheer wave elastography; VCTE, vibration-controlled transient elastography.
In NAFLD, serum biomarkers including ELF, NFS, FIB-4 and APRI have poor to modest accuracy (AUC <0.75) in predicting response of liver fibrosis to drug treatment88,89 or lifestyle intervention90 and cannot be recommended to monitor for short- term (≤1 year) treatment response. In a 24-week trial of selonsertib in NAFLD, MRE and VCTE had poor accuracy for predicting fibrosis improvement (AUC <0.65), however, the accuracy of MRE increased to 0.79 when combined with baseline MRE value, suggesting utility for monitoring antifibrotic treatment response.89 MRE may also be useful for monitoring fibrosis progression over time in the absence of treatment, with a minimum 15% increase in value over 1.4 years associated with a 3.4-fold higher risk of fibrosis progression in a cohort of 102 patients with NAFLD.91 An algorithm combining platelet count, ALT normalisation and change in HbA1c had high accuracy (AUC 0.96) for predicting fibrosis improvement following 1 tear of life style intervention in patients with NASH, however, requires further validation.90 In the absence of intervention, FIB-4, APRI and NAFLD Fibrosis Score have poor to modest accuracy in detecting any fibrosis progression (AUCs <0.75), though increases (AUC 0.80–0.82) for the prediction of progression to advanced fibrosis.92 These simple parameters do not improve with fibrosis regression in NAFLD, and their strength is confirming absence of progression (NPVs 89%–90%) rather than diagnosing progression to advanced fibrosis (PPVs 44%–49%).
In CHC, diagnostic accuracy of VCTE postviral eradication appears to fall (AUC <0.80) and the accuracy of blood-based biomarkers is poor (AUC <0.70) up to 5 years post-treatment.93,94 Beyond 5 years, however, the accuracy of elastography and simple blood-based NIT seems to improve though revised cut-offs are required.95 One study of 84 CHC postliver transplant patients found VCTE but not ELF, remained accurate at predicting advanced fibrosis following successful antiviral treatment.87
Data from cohorts of cirrhotic CHC and CHB patients achieving viral eradication or control, suggests that while liver stiffness and serum-based NITs improve with successful treatment, neither are sufficiently reliable in excluding risk of future hepatocellular carcinoma (HCC).96,97 Furthermore, LSM98 cirrhosis,93,94,99 thus, ceasing HCC surveillance in cirrhotic patients with improving NITs on antiviral therapy cannot be currently recommended.
EMERGING TECHNOLOGIES
Imaging-based techniques
Three-dimensional (3D) MRE evaluates sheer wave propagation in multiple planes and avoids mathematical assumptions inherent to 3D techniques. Although 3D-MRE has been demonstrated to be more accurate in predicting advanced fibrosis in patients with CHB, CHC and NAFLD compared with 2D-MRE,67,100 further validation is required to understand the incremental benefit of this technique. New methods using multiparametric MRI incorporate damping ratio at a lower frequency using 3D MRE along with shear wave stiffness on MRE and these may further help refine the detection of NASH and NASH-related fibrosis. 101,102
Collagen synthesis markers
The deposition of fibrosis is a dynamic process reflecting an imbalance of fibrogenesis and fibrinolysis. The rate of turnover of hepatic collagen in liver biopsies can be determined by isotope techniques and is highly correlated with the fractional synthesis rate of plasma lumican, which is a peptide mediator of collagen synthesis which is overexpressed in the presence of liver fibrosis.98,103 Thus, the plasma kinetics of lumican can provide a real-time estimate of the dynamics of fibrosis turnover within the liver and appears as an attractive technique for rapid assessment of drug efficacy and determination of potential for fibrosis progression as well as regression in early phase trials.104,105
During extracellular matrix formation, the N-terminal propeptide of type 3 collagen (Pro-C3) is cleaved from procollagen of type III collagen, reflecting fibrogenic activity. Serum Pro-C3 levels correlate with liver fibrosis and offers promise as an accurate fibrosis biomarker in NAFLD patients when combined with simple clinical parameters.106,107
Genetic prediction models
Genetic variability between individuals leads to differential susceptibility towards the development of liver fibrosis and is estimated to account for half of the phenotypic variance in CLDs such as NAFLD.108 Genetic variants related to single nucleotide polymorphisms within genes or epigenetic changes such as differential DNA methylation, have been associated with fibrosis in CHC and NAFLD.109–111 Although most DNA methylation studies have characterised changes in liver biopsies, plasma levels of cell-free circulating DNA methylation of PP AR-gamma may be a promising and accessible diagnostic marker.111 It is likely that the inclusion of clinical risk factors such as age are still likely to be required in order to develop accurate predictive models.112 Validation across different ethnicities remains important to demonstrate the generalizability of gene-based scores.
Microbiome
The gut microbiome has been implicated in the genesis of liver injury and fibrosis in CLD. Proof-of-principle studies using different sequencing technology have demonstrated that the bacterial composition in stool varies according to fibrosis stage in patients with NAFLD.113,114 Emerging data have shown that a metagenomic signature of gut microbiome along with age, BMI and ethnicity can be used to detect presence of advanced fibrosis with high accuracy among patients with biopsy-proven NAFLD.114 Using a familial study design, a recent study demonstrated that a 16S signature of gut microbiome was able to differentiate family members who had NAFLD cirrhosis from those who did not with a high diagnostic accuracy (AUC >0.9).113 Further studies are underway to validate these findings in independent external validation cohorts.
‘Omics including miRNA
Characterisation of the phenome associated with liver fibrosis offers a hypothesis free approach to identify novel markers of fibrosis. Metabolomic and proteomic approaches using mass spectroscopy screening have identified numerous molecules associated with advanced fibrosis in NAFLD, viral hepatitis and ALD.115,116 Complicated methodology and lack of independent validation has limited translation into clinical practice. MicroRNA’s (miRNAs) are non-coding RNA molecules which regulate gene expression and have been illustrated to be differentially expressed in the liver of NAFLD patients according to the degree of fibrosis. Less work has been done examining circulating plasma miRNA levels, however, miRNA 122a has been associated with NASH and liver fibrosis, but has limited accuracy (AUC 0.71 and 0.61, respectively).117 Large multicentre collaborations (non-invasive biomarkers of metabolic liver disease (NIMBLE), LITMUS and NASH-CRN) are exploring promising biomarkers of fibrosis in NAFLD and are likely to lead to the discovery of clinically relevant panels.
CONCLUSIONS
Non-invasive assessment of liver fibrosis has become part of routine clinical care for patients with CLD. Accurate serum and imaging methods are now available, along with increased understanding of their limitations which is required for correct interpretation and application. Serum markers are valuable for screening due their ease and cost, whereas imaging-based techniques lend themselves as confirmatory tests. Advances in imaging techniques and the promise of novel markers discovered by ‘omic’ approaches mean the accuracy and clinical utility of NITs is likely to increase further in the future.
Key messages.
Within the general population, only patients at risk of liver disease should be screened using non-invasive tests (NITs) due to the potential for false positive results.
Serum-based tests can reliably exclude advanced fibrosis/cirrhosis and can be integrated into point-of care testing in the community.
Sonographic elastography techniques are more accurate at diagnosing cirrhosis than serum NITs and can be confirmatory of a high serum test result.
Using concurrent serum and vibration-controlled transient elastography increases diagnostic accuracy and specificity for determining advanced fibrosis.
Obesity significantly impacts scan success rate and validity of newer sonographic elastography methods such as point and shear wave elastography.
Serum and sonographic elastography tests have limited accuracy in monitoring fibrosis change in response to therapy.
In patients with non-alcoholic fatty liver disease (NAFLD), stable AST-Platelet Ratio Index, Fibrosis Score 4 and NAFLD Fibrosis Scores confirm absence of fibrosis progression, however, score increase is poorly predictive of fibrosis progression.
MR elastography is currently the most accurate NIT across the spectrum of liver fibrosis and offers promise in the assessment of response to antifibrotic drugs.
Acknowledgments
Funding RL receives funding support from NIEHS (5P42ES010337), NCATS (5UL1TR001442), NIDDK (R01DK106419, P30DK120515) and DOD PRCRP (CA170674P2).
Footnotes
Competing interests LAA holds patents for Hepascore and his employer (University of Western Australia) has a licensing agreement with Quest Diagnostics. RL serves as a consultant or advisory board member for Arrowhead Pharmaceuticals, AstraZeneca, Bird Rock Bio, Boehringer Ingelheim, Bristol-Myer Squibb, Celgene, Cirius, CohBar, Conatus, Eli Lilly, Galmed, Gemphire, Gilead, Glympse bio, GNI, GRI Bio, Intercept, Ionis, Janssen, Merck, Metacrine, NGM Biopharmaceuticals, Novartis, Novo Nordisk, Pfizer, Prometheus, Sanofi, Siemens, and Viking Therapeutics. In addition, his institution has received grant support from Allergan, BoehringerIngelheim, Bristol-Myers Squibb, Cirius, Eli Lilly and Company, Galectin Therapeutics, Galmed Pharmaceuticals, GE, Genfit, Gilead, Intercept, Grail, Janssen, Madrigal Pharmaceuticals, Merck, NGM Biopharmaceuticals, NuSirt, Pfizer, pH Pharma, Prometheus, and Siemens. He is also cofounder of Liponexus.
Patient consent for publication Not required.
Provenance and peer review Commissioned; externally peer reviewed.
Contributors Both authors are corresponding authors.
REFERENCES
- 1.Zoubek ME, Trautwein C, Strnad P. Reversal of liver fibrosis: from fiction to reality. Best Pract Res Clin Gastroenterol 2017;31:129–41. [DOI] [PubMed] [Google Scholar]
- 2.Huang Y, de Boer WB, Adams LA, et al. Image analysis of liver biopsy samples measures fibrosis and predicts clinical outcome. J Hepatol 2014;61:22–7. [DOI] [PubMed] [Google Scholar]
- 3.Sebastiani G, Castera L, Halfon P, et al. The impact of liver disease aetiology and the stages of hepatic fibrosis on the performance of non-invasive fibrosis biomarkers: an international study of 2411 cases. Aliment Pharmacol Ther 2011;34:1202–16. [DOI] [PubMed] [Google Scholar]
- 4.Adams LA, George J, Bugianesi E, et al. Complex non-invasive fibrosis models are more accurate than simple models in non-alcoholic fatty liver disease. J Gastroenterol Hepatol 2011;26:1536–43. [DOI] [PubMed] [Google Scholar]
- 5.Bertrais S, Boursier J, Ducancelle A, et al. Prognostic durability of liver fibrosis tests and improvement in predictive performance for mortality by combining tests. J Gastroenterol Hepatol 2017;32:1240–9. [DOI] [PubMed] [Google Scholar]
- 6.Zarski J-P, Sturm N, Guechot J, et al. Comparison of nine blood tests and transient elastography for liver fibrosis in chronic hepatitis C: the ANRS HCEP-23 study. J Hepatol 2012;56:55–62. [DOI] [PubMed] [Google Scholar]
- 7.Huang Y, Adams LA, Joseph J, et al. The ability of Hepascore to predict liver fibrosis in chronic liver disease: a meta-analysis. Liver Int 2017;37:121–31. [DOI] [PubMed] [Google Scholar]
- 8.Lin Z-H, Xin Y-N, Dong Q-J, et al. Performance of the aspartate aminotransferase-to-platelet ratio index for the staging of hepatitis C-related fibrosis: an updated meta-analysis. Hepatology 2011;53:726–36. [DOI] [PubMed] [Google Scholar]
- 9.Xiao G, Zhu S, Xiao X, et al. Comparison of laboratory tests, ultrasound, or magnetic resonance elastography to detect fibrosis in patients with nonalcoholic fatty liver disease: a meta-analysis. Hepatology 2017;66:1486–501. [DOI] [PubMed] [Google Scholar]
- 10.Kim WR, Berg T, Asselah T, et al. Evaluation of APRI and FIB-4 scoring systems for non-invasive assessment of hepatic fibrosis in chronic hepatitis B patients. J Hepatol 2016;64:773–80. [DOI] [PubMed] [Google Scholar]
- 11.Parkes J, Guha IN, Harris S, et al. Systematic review of the diagnostic performance of serum markers of liver fibrosis in alcoholic liver disease. Comp Hepatol 2012;11:5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Xie Q, Zhou X, Huang P, et al. The performance of enhanced liver fibrosis (ELF) test for the staging of liver fibrosis: a meta-analysis. PLoS One 2014;9:e92772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wong GL-H, Chan HL-Y, Choi PC-L, et al. Non-invasive algorithm of enhanced liver fibrosis and liver stiffness measurement with transient elastography for advanced liver fibrosis in chronic hepatitis B. Aliment Pharmacol Ther 2014;39:197–208. [DOI] [PubMed] [Google Scholar]
- 14.Fagan KJ, Pretorius CJ, Horsfall LU, et al. ELF score ≥9.8 indicates advanced hepatic fibrosis and is influenced by age, steatosis and histological activity. Liver Int 2015;35:1673–81. [DOI] [PubMed] [Google Scholar]
- 15.McPherson S, Hardy T, Dufour J-F, et al. Age as a confounding factor for the accurate non-invasive diagnosis of advanced NAFLD fibrosis. Am J Gastroenterol 2017;112:740–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lichtinghagen R, Pietsch D, Bantel H, et al. The enhanced liver fibrosis (ELF) score: normal values, influence factors and proposed cut-off values. J Hepatol 2013;59:236–42. [DOI] [PubMed] [Google Scholar]
- 17.Siddiqui MS, Vuppalanchi R, Van Natta ML, et al. Vibration-Controlled transient elastography to assess fibrosis and steatosis in patients with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol 2019;17:156–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.European Association for Study of Liver, Asociacion Latinoamericana para el Estudio del Higado. EASL-ALEH clinical practice guidelines: non-invasive tests for evaluation of liver disease severity and prognosis. J Hepatol 2015;63:237–64. [DOI] [PubMed] [Google Scholar]
- 19.Boursier J, de Ledinghen V, Sturm N, et al. Precise evaluation of liver histology by computerized morphometry shows that steatosis influences liver stiffness measured by transient elastography in chronic hepatitis C. J Gastroenterol 2014;49:527–37. [DOI] [PubMed] [Google Scholar]
- 20.Vuppalanchi R, Siddiqui MS, Van Natta ML, et al. Performance characteristics of vibration-controlled transient elastography for evaluation of nonalcoholic fatty liver disease. Hepatology 2018;67:134–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bloom S, Kemp W, Nicoll A, et al. Liver stiffness measurement in the primary care setting detects high rates of advanced fibrosis and predicts liver-related events in hepatitis C. J Hepatol 2018;69:575–83. [DOI] [PubMed] [Google Scholar]
- 22.Puigvehí M, Broquetas T, Coll S, et al. Impact of anthropometric features on the applicability and accuracy of FibroScan® (M and XL) in overweight/obese patients. J Gastroenterol Hepatol 2017;32:1746–53. [DOI] [PubMed] [Google Scholar]
- 23.Cassinotto C, Boursier J, de Lédinghen V, et al. Liver stiffness in nonalcoholic fatty liver disease: a comparison of supersonic shear imaging, FibroScan, and ARFI with liver biopsy. Hepatology 2016;63:1817–27. [DOI] [PubMed] [Google Scholar]
- 24.Cassinotto C, Lapuyade B, Mouries A, et al. Non-invasive assessment of liver fibrosis with impulse elastography: comparison of supersonic shear imaging with ARFI and FibroScan®. J Hepatol 2014;61:550–7. [DOI] [PubMed] [Google Scholar]
- 25.Fraquelli M, Rigamonti C, Casazza G, et al. Reproducibility of transient elastography in the evaluation of liver fibrosis in patients with chronic liver disease. Gut 2007;56:968–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cassinotto C, Lapuyade B, Aït-Ali A, et al. Liver fibrosis: noninvasive assessment with acoustic radiation force impulse elastography-comparison with FibroScan M and XL probes and FibroTest in patients with chronic liver disease. Radiology 2013;269:283–92. [DOI] [PubMed] [Google Scholar]
- 27.Boursier J, Cassinotto C, Hunault G, et al. Criteria to determine reliability of noninvasive assessment of liver fibrosis with virtual touch quantification. Clin Gastroenterol Hepatol 2019;17:164–71. [DOI] [PubMed] [Google Scholar]
- 28.Fraquelli M, Baccarin A, Casazza G, et al. Liver stiffness measurement reliability and main determinants of point shear-wave elastography in patients with chronic liver disease. Aliment Pharmacol Ther 2016;44:356–65. [DOI] [PubMed] [Google Scholar]
- 29.Leung VY-fong, Shen J, Wong VW-sun, et al. Quantitative elastography of liver fibrosis and spleen stiffness in chronic hepatitis B carriers: comparison of shear-wave elastography and transient elastography with liver biopsy correlation. Radiology 2013;269:910–8. [DOI] [PubMed] [Google Scholar]
- 30.Zhuang Y, Ding H, Zhang Y, et al. Two-Dimensional shear-wave elastography performance in the noninvasive evaluation of liver fibrosis in patients with chronic hepatitis B: comparison with serum fibrosis indexes. Radiology 2017;283:873–82. [DOI] [PubMed] [Google Scholar]
- 31.Ferraioli G, Tinelli C, Zicchetti M, et al. Reproducibility of real-time shear wave elastography in the evaluation of liver elasticity. Eur J Radiol 2012;81:3102–6. [DOI] [PubMed] [Google Scholar]
- 32.Yoon JH, Lee JM, Han JK, et al. Shear wave elastography for liver stiffness measurement in clinical sonographic examinations: evaluation of intraobserver reproducibility, technical failure, and unreliable stiffness measurements. J Ultrasound Med 2014;33:437–47. [DOI] [PubMed] [Google Scholar]
- 33.Boursier J, Cassinotto C, Hunault G, et al. Criteria to determine reliability of noninvasive assessment of liver fibrosis with virtual touch quantification. Clin Gastroenterol Hepatol 2019;17:164–171.e5. [DOI] [PubMed] [Google Scholar]
- 34.Paparo F, Corradi F, Cevasco L, et al. Real-time elastography in the assessment of liver fibrosis: a review of qualitative and semi-quantitative methods for elastogram analysis. Ultrasound Med Biol 2014;40:1923–33. [DOI] [PubMed] [Google Scholar]
- 35.Herrmann E, Ledinghen de V, Cassinotto C, et al. Assessment of biopsy-proven liver fibrosis by two-dimensional shear wave elastography: an individual patient data-based meta-analysis. Hepatology 2018;67:260–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chow JC-L, Wong GL-H, Chan AW-H, et al. Repeating measurements by transient elastography in non-alcoholic fatty liver disease patients with high liver stiffness. J Gastroenterol Hepatol 2019;34:241–8. [DOI] [PubMed] [Google Scholar]
- 37.de Franchis R, Baveno VI Faculty. Expanding consensus in portal hypertension: report of the Baveno VI consensus workshop: Stratifying risk and individualizing care for portal hypertension. J Hepatol 2015;63:743–52. [DOI] [PubMed] [Google Scholar]
- 38.Thiele M, Madsen BS, Hansen JF, et al. Accuracy of the enhanced liver fibrosis test vs FibroTest, elastography, and indirect markers in detection of advanced fibrosis in patients with alcoholic liver disease. Gastroenterology 2018;154:1369–79. [DOI] [PubMed] [Google Scholar]
- 39.Lee MS, Bae JM, Joo SK, et al. Prospective comparison among transient elastography, supersonic shear imaging, and ARFI imaging for predicting fibrosis in nonalcoholic fatty liver disease. PLoS One 2017;12:e0188321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wagner M, Corcuera-Solano I, Lo G, et al. Technical failure of Mr elastography examinations of the liver: experience from a large single-center study. Radiology 2017;284:401–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Singh S, Venkatesh SK, Wang Z, et al. Diagnostic performance of magnetic resonance elastography in staging liver fibrosis: a systematic review and meta-analysis of individual participant data. Clin Gastroenterol Hepatol 2015;13:440–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Singh S, Venkates SK, Keaveny A, et al. Diagnostic accuracy of magnetic resonance elastography in liver transplant recipients: a pooled analysis. Ann Hepatol 2016;15:363–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mariappan YK, Dzyubak B, Glaser KJ, et al. Application of modified Spin-Echo–based sequences for hepatic Mr elastography: evaluation, comparison with the conventional gradient-echo sequence, and preliminary clinical experience. Radiology 2017;282:390–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Yin M, Glaser KJ, Talwalkar JA, et al. Hepatic Mr elastography: clinical performance in a series of 1377 consecutive examinations. Radiology 2016;278:114–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chen J, Yin M, Talwalkar JA, et al. Diagnostic performance of Mr elastography and Vibration-controlled transient elastography in the detection of hepatic fibrosis in patients with severe to morbid obesity. Radiology 2017;283:418–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Trout AT, Serai S, Mahley AD, et al. Liver stiffness measurements with Mr elastography: agreement and repeatability across imaging systems, field strengths, and pulse sequences. Radiology 2016;281:793–804. [DOI] [PubMed] [Google Scholar]
- 47.Runge JH, Bohte AE, Verheij J, et al. Comparison of interobserver agreement of magnetic resonance elastography with histopathological staging of liver fibrosis. Abdom Imaging 2014;39:283–90. [DOI] [PubMed] [Google Scholar]
- 48.Kennedy P, Wagner M, Castéra L, et al. Quantitative elastography methods in liver disease: current evidence and future directions. Radiology 2018;286:738–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Singh S, Venkatesh SK, Loomba R, et al. Magnetic resonance elastography for staging liver fibrosis in non-alcoholic fatty liver disease: a diagnostic accuracy systematic review and individual participant data pooled analysis. Eur Radiol 2016;26:1431–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wu W-P, Chou C-T, Chen R-C, et al. Non-invasive evaluation of hepatic fibrosis: the diagnostic performance of magnetic resonance elastography in patients with viral hepatitis B or C. PLoS One 2015;10:e0140068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ichikawa S, Motosugi U, Nakazawa T, et al. Hepatitis activity should be considered a confounder of liver stiffness measured with Mr elastography. J Magn Reson Imaging 2015;41:1203–8. [DOI] [PubMed] [Google Scholar]
- 52.Imajo K, Kessoku T, Honda Y, et al. Magnetic resonance imaging more accurately classifies steatosis and fibrosis in patients with nonalcoholic fatty liver disease than transient elastography. Gastroenterology 2016;150:626–37. [DOI] [PubMed] [Google Scholar]
- 53.Cui J, Heba E, Hernandez C, et al. Magnetic resonance elastography is superior to acoustic radiation force impulse for the diagnosis of fibrosis in patients with biopsy-proven nonalcoholic fatty liver disease: a prospective study. Hepatology 2016;63:453–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Xiao H, Shi M, Xie Y, et al. Comparison of diagnostic accuracy of magnetic resonance elastography and Fibroscan for detecting liver fibrosis in chronic hepatitis B patients: a systematic review and meta-analysis. PLoS One 2017;12:e0186660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hsu C, Caussy C, Imajo K, et al. Magnetic resonance vs transient elastography analysis of patients with nonalcoholic fatty liver disease: a systematic review and pooled analysis of individual participants. Clin Gastroenterol Hepatol 2019;17:630–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wu W-P, Hoi C-I, Chen R-C, et al. Comparison of the efficacy of Gd-EOB-DTPA-enhanced magnetic resonance imaging and magnetic resonance elastography in the detection and staging of hepatic fibrosis. Medicine 2017;96:e8339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wang Q-B, Zhu H, Liu H-L, et al. Performance of magnetic resonance elastography and diffusion-weighted imaging for the staging of hepatic fibrosis: a meta-analysis. Hepatology 2012;56:239–47. [DOI] [PubMed] [Google Scholar]
- 58.Degos F, Perez P, Roche B, et al. Diagnostic accuracy of FibroScan and comparison to liver fibrosis biomarkers in chronic viral hepatitis: a multicenter prospective study (the FIBROSTIC study). J Hepatol 2010;53:1013–21. [DOI] [PubMed] [Google Scholar]
- 59.Boursier J, Vergniol J, Guillet A, et al. Diagnostic accuracy and prognostic significance of blood fibrosis tests and liver stiffness measurement by FibroScan in non-alcoholic fatty liver disease. J Hepatol 2016;65:570–8. [DOI] [PubMed] [Google Scholar]
- 60.Petta S, Vanni E, Bugianesi E, et al. The combination of liver stiffness measurement and NAFLD fibrosis score improves the noninvasive diagnostic accuracy for severe liver fibrosis in patients with nonalcoholic fatty liver disease. Liver Int 2015;35:1566–73. [DOI] [PubMed] [Google Scholar]
- 61.Wong VW-S, Vergniol J, Wong GL-H, et al. Diagnosis of fibrosis and cirrhosis using liver stiffness measurement in nonalcoholic fatty liver disease. Hepatology 2010;51:454–62. [DOI] [PubMed] [Google Scholar]
- 62.Nguyen-Khac E, Chatelain D, Tramier B, et al. Assessment of asymptomatic liver fibrosis in alcoholic patients using fibroscan: prospective comparison with seven non-invasive laboratory tests. Aliment Pharmacol Ther 2008;28:1188–98. [DOI] [PubMed] [Google Scholar]
- 63.Crespo G, Fernández-Varo G, Mariño Z, et al. ARFI, FibroScan®, ELF, and their combinations in the assessment of liver fibrosis: a prospective study. J Hepatol 2012;57:281–7. [DOI] [PubMed] [Google Scholar]
- 64.Kim BK, Kim HS, Park JY, et al. Prospective validation of ELF test in comparison with Fibroscan and FibroTest to predict liver fibrosis in Asian subjects with chronic hepatitis B. PLoS One 2012;7:e41964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Trembling PM, Lampertico P, Parkes J, et al. Performance of enhanced liver fibrosis test and comparison with transient elastography in the identification of liver fibrosis in patients with chronic hepatitis B infection. J Viral Hepat 2014;21:430–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Cui J, Ang B, Haufe W, et al. Comparative diagnostic accuracy of magnetic resonance elastography vs. eight clinical prediction rules for non-invasive diagnosis of advanced fibrosis in biopsy-proven non-alcoholic fatty liver disease: a prospective study. Aliment Pharmacol Ther 2015;41:1271–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Shi Y, Xia F, Li Q-J, et al. Magnetic resonance elastography for the evaluation of liver fibrosis in chronic hepatitis B and C by using both gradient-recalled echo and spin-echo echo planar imaging: a prospective study. Am J Gastroenterol 2016;111:823–33. [DOI] [PubMed] [Google Scholar]
- 68.Ichikawa S, Motosugi U, Ichikawa T, et al. Magnetic resonance elastography for staging liver fibrosis in chronic hepatitis C. Magn Reson Med Sci 2012;11:291–7. [DOI] [PubMed] [Google Scholar]
- 69.Huwart L, Sempoux C, Vicaut E, et al. Magnetic resonance elastography for the noninvasive staging of liver fibrosis. Gastroenterology 2008;135:32–40. [DOI] [PubMed] [Google Scholar]
- 70.Boursier J, de Ledinghen V, Zarski J-P, et al. Comparison of eight diagnostic algorithms for liver fibrosis in hepatitis C: new algorithms are more precise and entirely noninvasive. Hepatology 2012;55:58–67. [DOI] [PubMed] [Google Scholar]
- 71.Sebastiani G, Halfon P, Castera L, et al. Comparison of three algorithms of non-invasive markers of fibrosis in chronic hepatitis C. Aliment Pharmacol Ther 2012;35:92–104. [DOI] [PubMed] [Google Scholar]
- 72.Petta S, Wong VW-S, Cammà C, et al. Serial combination of non-invasive tools improves the diagnostic accuracy of severe liver fibrosis in patients with NAFLD. Aliment Pharmacol Ther 2017;46:617–27. [DOI] [PubMed] [Google Scholar]
- 73.Castéra L, Sebastiani G, Le Bail B, et al. Prospective comparison of two algorithms combining non-invasive methods for staging liver fibrosis in chronic hepatitis C. J Hepatol 2010;52:191–8. [DOI] [PubMed] [Google Scholar]
- 74.Boursier J, de Ledinghen V, Leroy V, et al. A stepwise algorithm using an at-a-glance first-line test for the non-invasive diagnosis of advanced liver fibrosis and cirrhosis. J Hepatol 2017;66:1158–65. [DOI] [PubMed] [Google Scholar]
- 75.Patel YA, Gifford EJ, Glass LM, et al. Identifying nonalcoholic fatty liver disease advanced fibrosis in the Veterans health administration. Dig Dis Sci 2018;63:2259–66. [DOI] [PubMed] [Google Scholar]
- 76.Unalp-Arida A, Ruhl CE. Liver fibrosis scores predict liver disease mortality in the United States population. Hepatology 2017;66:84–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Morling JR, Fallowfield JA, Guha IN, et al. Using non-invasive biomarkers to identify hepatic fibrosis in people with type 2 diabetes mellitus: the Edinburgh type 2 diabetes study. J Hepatol 2014;60:384–91. [DOI] [PubMed] [Google Scholar]
- 78.Roulot D, Costes J-L, Buyck J-F, et al. Transient elastography as a screening tool for liver fibrosis and cirrhosis in a community-based population aged over 45 years. Gut 2011;60:977–84. [DOI] [PubMed] [Google Scholar]
- 79.Cheng P-N, Chiu H-C, Chiu Y-C, et al. Comparison of FIB-4 and transient elastography in evaluating liver fibrosis of chronic hepatitis C subjects in community. PLoS One 2018;13:e0206947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Harman DJ, Ryder SD, James MW, et al. Direct targeting of risk factors significantly increases the detection of liver cirrhosis in primary care: a cross-sectional diagnostic study utilising transient elastography. BMJ Open 2015;5:e007516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Caballería L, Pera G, Arteaga I, et al. High prevalence of liver fibrosis among European adults with unknown liver disease: a population-based study. Clin Gastroenterol Hepatol 2018;16:1138–45. [DOI] [PubMed] [Google Scholar]
- 82.Chinnaratha MA, Jeffrey GP, MacQuillan G, et al. Prediction of morbidity and mortality in patients with chronic hepatitis C by non-invasive liver fibrosis models. Liver Int 2014;34:720–7. [DOI] [PubMed] [Google Scholar]
- 83.Sanyal AJ, Harrison SA, Ratziu V, et al. The natural history of advanced fibrosis due to nonalcoholic steatohepatitis: data from the Simtuzumab trials. Hepatology 2019;70:1913–27. [DOI] [PubMed] [Google Scholar]
- 84.Lee DH, Lee JM, Chang W, et al. Prognostic role of liver stiffness measurements using magnetic resonance elastography in patients with compensated chronic liver disease. Eur Radiol 2018;28:3513–21. [DOI] [PubMed] [Google Scholar]
- 85.Asrani SK, Talwalkar JA, Kamath PS, et al. Role of magnetic resonance elastography in compensated and decompensated liver disease. J Hepatol 2014;60:934–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hézode C, Castéra L, Roudot-Thoraval F, et al. Liver stiffness diminishes with antiviral response in chronic hepatitis C. Aliment Pharmacol Ther 2011;34:656–63. [DOI] [PubMed] [Google Scholar]
- 87.Mauro E, Crespo G, Montironi C, et al. Portal pressure and liver stiffness measurements in the prediction of fibrosis regression after sustained virological response in recurrent hepatitis C. Hepatology 2018;67:1683–94. [DOI] [PubMed] [Google Scholar]
- 88.Chalasani N, Abdelmalek MF, Loomba R, et al. Relationship between three commonly used non-invasive fibrosis biomarkers and improvement in fibrosis stage in patients with non-alcoholic steatohepatitis. Liver Int 2019;39:924–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Jayakumar S, Middleton MS, Lawitz EJ, et al. Longitudinal correlations between MRE, MRI-PDFF, and liver histology in patients with non-alcoholic steatohepatitis: analysis of data from a phase II trial of selonsertib. J Hepatol 2019;70:133–41. [DOI] [PubMed] [Google Scholar]
- 90.Vilar-Gomez E, Calzadilla-Bertot L, Friedman SL, et al. Serum biomarkers can predict a change in liver fibrosis 1 year after lifestyle intervention for biopsy-proven NASH. Liver Int 2017;37:1887–96. [DOI] [PubMed] [Google Scholar]
- 91.Ajmera VH, Liu A, Singh S, et al. Clinical utility of an increase in magnetic resonance elastography in predicting fibrosis progression in NAFLD. Hepatology 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Siddiqui MS, Yamada G, Vuppalanchi R, et al. Diagnostic accuracy of noninvasive fibrosis models to detect change in fibrosis stage. Clin Gastroenterol Hepatol 2019;17:1877–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.D’Ambrosio R, Aghemo A, Fraquelli M, et al. The diagnostic accuracy of Fibroscan® for cirrhosis is influenced by liver morphometry in HCV patients with a sustained virological response. J Hepatol 2013;59:251–6. [DOI] [PubMed] [Google Scholar]
- 94.D’Ambrosio R, Degasperi E, Aghemo A, et al. Serological tests do not predict residual fibrosis in hepatitis C cirrhotics with a sustained virological response to interferon. PLoS One 2016;11:e0155967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Tachi Y, Hirai T, Kojima Y, et al. Liver stiffness measurement using acoustic radiation force impulse elastography in hepatitis C virus-infected patients with a sustained virological response. Aliment Pharmacol Ther 2016;44:346–55. [DOI] [PubMed] [Google Scholar]
- 96.Jacobson IM, Lim JK, Fried MW. American gastroenterological association Institute clinical practice Update—Expert review: care of patients who have achieved a sustained virologic response after antiviral therapy for chronic hepatitis C infection. Gastroenterology 2017;152:1578–87. [DOI] [PubMed] [Google Scholar]
- 97.Kim BS, Seo YS, Kim YS, et al. Reduced risk of hepatocellular carcinoma by achieving a subcirrhotic liver stiffness through antiviral agents in hepatitis B virus-related advanced fibrosis or cirrhosis. J Gastroenterol Hepatol 2018;33:503–10. [DOI] [PubMed] [Google Scholar]
- 98.Charlton M, Viker K, Krishnan A, et al. Differential expression of lumican and fatty acid binding protein-1: new insights into the histologic spectrum of nonalcoholic fatty liver disease. Hepatology 2009;49:1375–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Pan JJ, Bao F, Du E, et al. Morphometry confirms fibrosis regression from sustained virologic response to direct-acting antivirals for hepatitis C. Hepatol Commun 2018;2:1320–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Loomba R, Cui J, Wolfson T, et al. Novel 3D magnetic resonance elastography for the noninvasive diagnosis of advanced fibrosis in NAFLD: a prospective study. Am J Gastroenterol 2016;111:986–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Allen AM, Shah VH, Therneau TM, et al. The role of three-dimensional magnetic resonance elastography in the diagnosis of nonalcoholic steatohepatitis in obese patients undergoing bariatric surgery. Hepatology 2018. [Epub ahead of print: 24 Dec 2018]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Yin Z, Murphy MC, Li J, et al. Prediction of nonalcoholic fatty liver disease (NAFLD) activity score (NAS) with multiparametric hepatic magnetic resonance imaging and elastography. Eur Radiol 2019;29:5823–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Decaris ML, Emson CL, Li K, et al. Turnover rates of hepatic collagen and circulating collagen-associated proteins in humans with chronic liver disease. PLoS One 2015;10:e0123311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Lawitz EJ, Coste A, Poordad F, et al. Acetyl-CoA carboxylase inhibitor GS-0976 for 12 weeks reduces hepatic de novo lipogenesis and steatosis in patients with nonalcoholic steatohepatitis. Clin Gastroenterol Hepatol 2018;16:1983–91. [DOI] [PubMed] [Google Scholar]
- 105.Decaris ML, Li KW, Emson CL, et al. Identifying nonalcoholic fatty liver disease patients with active fibrosis by measuring extracellular matrix remodeling rates in tissue and blood. Hepatology 2017;65:78–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Daniels SJ, Leeming DJ, Eslam M, et al. Adapt: an algorithm incorporating pro-C3 accurately identifies patients with NAFLD and advanced fibrosis. Hepatology 2019;69:1075–86. [DOI] [PubMed] [Google Scholar]
- 107.Boyle M, Tiniakos D, Schattenberg JM, et al. Performance of the pro-C3 collagen neo-epitope biomarker in non-alcoholic fatty liver disease. JHEP Reports 2019;1:188–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Loomba R, Schork N, Chen C-H, et al. Heritability of hepatic fibrosis and steatosis based on a prospective twin study. Gastroenterology 2015;149:1784–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Krawczyk M, Rau M, Schattenberg JM, et al. Combined effects of the PNPLA3 rs738409, TM6SF2 rs58542926, and MBOAT7 rs641738 variants on NAFLD severity: a multicenter biopsy-based study. J Lipid Res 2017;58:247–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Huang H, Shiffman ML, Friedman S, et al. A 7 gene signature identifies the risk of developing cirrhosis in patients with chronic hepatitis C. Hepatology 2007;46:297–306. [DOI] [PubMed] [Google Scholar]
- 111.Hardy T, Zeybel M, Day CP, et al. Plasma DNA methylation: a potential biomarker for stratification of liver fibrosis in non-alcoholic fatty liver disease. Gut 2017;66:1321–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Eslam M, Hashem AM, Romero-Gomez M, et al. FibroGENE: a gene-based model for staging liver fibrosis. J Hepatol 2016;64:390–8. [DOI] [PubMed] [Google Scholar]
- 113.Caussy C, Tripathi A, Humphrey G, et al. A gut microbiome signature for cirrhosis due to nonalcoholic fatty liver disease. Nat Commun 2019;10:1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Loomba R, Seguritan V, Li W, et al. Gut microbiome-based metagenomic signature for non-invasive detection of advanced fibrosis in human nonalcoholic fatty liver disease. Cell Metab 2017;25:1054–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Tokushige K, Hashimoto E, Kodama K, et al. Serum metabolomic profile and potential biomarkers for severity of fibrosis in nonalcoholic fatty liver disease. J Gastroenterol 2013;48:1392–400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Nallagangula KS, Shashidhar KN, Lakshmaiah V, et al. Evolution of proteomic biomarker for chronic liver disease: promise into reality. J Circ Biomark 2018;7:1849454418777186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Pirola CJ, Fernández Gianotti T, Castaño GO, et al. Circulating microRNA signature in non-alcoholic fatty liver disease: from serum non-coding RNAs to liver histology and disease pathogenesis. Gut 2015;64:800–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Poynard T, Morra R, Halfon P, et al. Meta-analyses of FibroTest diagnostic value in chronic liver disease. BMC Gastroenterol 2007;7:40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Patel K, Gordon SC, Jacobson I, et al. Evaluation of a panel of non-invasive serum markers to differentiate mild from moderate-to-advanced liver fibrosis in chronic hepatitis C patients. J Hepatol 2004;41:935–42. [DOI] [PubMed] [Google Scholar]
- 120.Loomba R, Jain A, Diehl AM, et al. Validation of serum test for advanced liver fibrosis in patients with nonalcoholic steatohepatitis. Clin Gastroenterol Hepatol 2019;17:1867–76. [DOI] [PubMed] [Google Scholar]
- 121.Parkes J, Roderick P, Harris S, et al. Enhanced liver fibrosis test can predict clinical outcomes in patients with chronic liver disease. Gut 2010;59:1245–51. [DOI] [PubMed] [Google Scholar]
- 122.Bertot LC, Jeffrey GP, de Boer B, et al. Diabetes impacts prediction of cirrhosis and prognosis by non-invasive fibrosis models in non-alcoholic fatty liver disease. Liver Int 2018;38:1793–802. [DOI] [PubMed] [Google Scholar]
- 123.Munteanu M, Pais R, Peta V, et al. Long-term prognostic value of the FibroTest in patients with non-alcoholic fatty liver disease, compared to chronic hepatitis C, B, and alcoholic liver disease. Aliment Pharmacol Ther 2018;48:1117–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Chan W-K, Treeprasertsuk S, Goh GB-B, et al. Optimizing use of nonalcoholic fatty liver disease fibrosis score, Fibrosis-4 score, and liver stiffness measurement to identify patients with advanced fibrosis. Clin Gastroenterol Hepatol 2019;17:2570–80. [DOI] [PubMed] [Google Scholar]
- 125.Shaheen AAM, Myers RP. Diagnostic accuracy of the aspartate aminotransferase-to-platelet ratio index for the prediction of hepatitis C–related fibrosis: a systematic review. Hepatology 2007;46:912–21. [DOI] [PubMed] [Google Scholar]
- 126.Angulo P, Bugianesi E, Bjornsson ES, et al. Simple noninvasive systems predict long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology 2013;145:782–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Guillaume M, Moal V, Delabaudiere C, et al. Direct comparison of the specialised blood fibrosis tests FibroMeter V2G and Enhanced Liver Fibrosis score in patients with non-alcoholic fatty liver disease from tertiary care centres. Aliment Pharmacol Ther 2019;50:1214–22. [DOI] [PubMed] [Google Scholar]
- 128.Mulabecirovic A, Mjelle AB, Gilja OH, et al. Repeatability of shear wave elastography in liver fibrosis phantoms—Evaluation of five different systems. PLoS One 2018;13:e0189671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Thiele M, Detlefsen S, Sevelsted Møller L, et al. Transient and 2-dimensional shear-wave elastography provide comparable assessment of alcoholic liver fibrosis and cirrhosis. Gastroenterology 2016;150:123–33. [DOI] [PubMed] [Google Scholar]
- 130.Eddowes PJ, Sasso M, Allison M, et al. Accuracy of FibroScan controlled attenuation parameter and liver stiffness measurement in assessing steatosis and fibrosis in patients with nonalcoholic fatty liver disease. Gastroenterology 2019;156:1717–30. [DOI] [PubMed] [Google Scholar]
- 131.Liang X, Xie Q, Tan D, et al. Interpretation of liver stiffness measurement-based approach for the monitoring of hepatitis B patients with antiviral therapy: a 2-year prospective study. J Viral Hepat 2018;25:296–305. [DOI] [PubMed] [Google Scholar]
- 132.Dong X-Q, Wu Z, Li J, et al. Declining in liver stiffness cannot indicate fibrosis regression in patients with chronic hepatitis B: A 78-week prospective study. J Gastroenterol Hepatol 2019;34:755–63. [DOI] [PubMed] [Google Scholar]
- 133.Tachi Y, Hirai T, Toyoda H, et al. Predictive ability of laboratory indices for liver fibrosis in patients with chronic hepatitis C after the eradication of hepatitis C virus. PLoS One 2015;10:e0133515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Tachi Y, Hirai T, Kojima Y, et al. Liver stiffness reduction correlates with histological characteristics of hepatitis C patients with sustained virological response. Liver Int 2018;38:59–67. [DOI] [PubMed] [Google Scholar]