Main text
RNA-based vaccine technology received a significant boost this past year with the approval of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) mRNA vaccines developed by Biotech/Pfizer and Moderna. In addition to such mRNA-based vaccines, self-amplifying RNA (sa-RNA) can also be used for vaccination.1 In addition to the transgene product, the sa-mRNA encodes an RNA-dependent RNA polymerase, which permits self-replication of the transcript, thereby increasing the level and duration of protein expression and allowing application of lower doses. This longer-lasting effect may even obviate the need for a booster vaccine. The aim of any vaccine is to induce potent immune responses. Paradoxically, innate immune responses elicited by sa-mRNA vaccines may lower their effectiveness. In this issue of Molecular Therapy, Zhong et al.2 show that inhibition of the type 1 interferon (IFN I) response to sa-RNA using corticosteroids and other inhibitors substantially increased production of the sa-mRNA-encoded protein antigen but, unfortunately, was detrimental to the vaccine effect. However, modification of the purification method to eliminate double-stranded RNA (dsRNA) byproducts during sa-mRNA production reduced innate immunity and improved the efficacy of a Zika virus vaccine, resulting in higher antibody titers and T cell responses against Zika target antigen.
To manufacture the sa-RNA vaccine, linearized plasmid DNA is used as a template for transcription by T7 RNA polymerase. dsRNA is generated as a byproduct during this process. Following delivery of the vaccine, the dsRNA induces IFN I production through activation of innate immune receptors that can reside in endosomes or in the cytoplasm, such as toll-like receptor 3 or retinoic acid-inducible gene I (RIG-I). Induction of antigen-specific immune responses—the goal of vaccination—requires activation signals provided by such innate immune sensors. The latter recognize pathogen-associated molecular patterns (PAMPs), such as those generated during infection by dsRNA viruses, and help alert the immune system to an infection. One can mimic these effects by the inclusion of adjuvant molecules that are similar to naturally occurring PAMPs in vaccine formulations. Innate immune signals induce cytokine production and prompt dendritic cells and other antigen-presenting cells to provide co-stimulation to T cells, all of which aid in the generation of an adaptive immune response. One could therefore speculate that contamination of the sa-mRNA vaccine with innate immune activating dsRNA might enhance the vaccine. However, this view is overly simplistic.
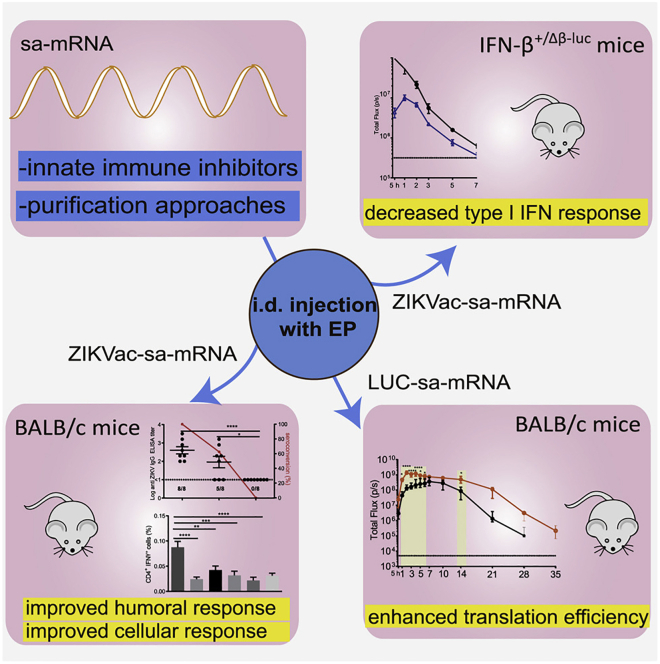
The type, strength, and timing of innate signals all critically influence the vaccination outcome. In the context of sa-mRNA vaccines, dsRNA molecules, in particular, induce IFN I responses. IFN I has been described as a critical regulator of mRNA-based vaccines and also plays a crucial role in antiviral T cell responses. For instance, sensing of IFN I by conventional dendritic cells is a major factor in their ability to activate CD8+ T cell responses in adeno-associated viral gene transfer, resulting in unwanted immune responses in gene therapy.3 However, this does not necessarily mean that dsRNA-induced IFN I will have the desired immune stimulatory effect on RNA-based vaccines. As is the case with other cytokines, IFNα and IFNβ produce pleiotropic effects. IFN I contributes to activation of dendritic cells, enhances co-stimulation, and also directly stimulates T cells. However, IFN I cytokines can also induce an antiviral state in cells to limit gene expression and thereby suppress viral replication. For a vaccine, the consequence may be reduced expression of the vaccine antigen, which ironically lowers vaccine efficacy. To address this issue, Zhong et al.2 topically applied corticosteroids at the site of vaccine administration in mice prior to intradermal electroporation. The steroids suppressed IFN I production, and the approach was ultimately successful in raising expression, as measured by a luciferase reporter coexpressed from the sa-mRNA (see Figure 1). The effect could be further enhanced by inclusion of inhibitors of specific innate immune signaling pathways. At the same time, however, the immune-suppressive effect of the steroids completely prevented vaccination against Zika virus, possibly by suppressing the required immune stimulatory effects of IFN I and/or suppression of other components of the immune response.
Figure 1.
Inhibition of the type 1 interferon (IFN I) response to sa-RNA using corticosteroids and other inhibitors increases production of the sa-mRNA-encoded transgene but is detrimental to the vaccine effect
The application of corticosteroids at the site of vaccine administration in mice prior to intradermal electroporation suppresses IFN I production and vaccine efficacy (top right panel) but increases expression of a luciferase reporter coexpressed from the sa-mRNA (bottom right panel). Modification of the saRNA purification protocol to eliminate most of the dsRNA led to reduced activation of innate immunity but increased vaccine efficacy against Zika virus (bottom left panel).
In an alternative approach to the problem, the authors aimed to find ways to eliminate dsRNA molecules from their vaccine preparations rather than suppressing their effects. This was accomplished by switching from silica- to cellulose-based purification of the produced RNA. An optimized purification technique eliminated most of the dsRNA, resulting in multiple beneficial effects on the vaccine. Not only was innate immunity reduced, but vaccine efficacy against Zika virus was much improved by multiple measures, including increased seroconversion, antibody titers, and the magnitude of CD4+ and CD8+ T cell responses. These observations should serve as a guide for general improvements in development of sa-RNA vaccines. Since electroporation is not easily scalable for delivery, it would be interesting to determine whether delivery using lipid nanoparticles (LNPs) is similarly affected. Certain liposomes, themselves, can activate dendritic cells, so it should be possible to deliver sa-mRNA vaccines that are dsRNA-free without the need for additional adjuvants.4 However, one needs to keep in mind that the magnitude of the resulting T cell response is also dependent on the route of mRNA-lipoplex administration and that regulation of the immune response by IFN I does not entirely take place at the dendritic cell level.5
References
- 1.Bloom K., van den Berg F., Arbuthnot P. Self-amplifying RNA vaccines for infectious diseases. Gene Ther. 2020 doi: 10.1038/s41434-020-00204-y. Published online October 22, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhong Z., McCafferty S., Opsomer L., Wang H., Huysmans H., De Temmerman J., Lienenklaus S., Portela Catani J.P., Combes F., Sanders N.N. Corticosteroids and cellulose purification improve, respectively, the in vivo translation and vaccination efficacy of sa-mRNAs. Mol. Ther. 2021;29:1370–1381. doi: 10.1016/j.ymthe.2021.01.023. this issue. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shirley J.L., Keeler G.D., Sherman A., Zolotukhin I., Markusic D.M., Hoffman B.E., Morel L.M., Wallet M.A., Terhorst C., Herzog R.W. Type I IFN Sensing by cDCs and CD4+ T Cell Help Are Both Requisite for Cross-Priming of AAV Capsid-Specific CD8+ T Cells. Mol. Ther. 2020;28:758–770. doi: 10.1016/j.ymthe.2019.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Vitor M.T., Bergami-Santos P.C., Cruz K.S., Pinho M.P., Barbuto J.A., De La Torre L.G. Dendritic Cells Stimulated by Cationic Liposomes. J. Nanosci. Nanotechnol. 2016;16:270–279. doi: 10.1166/jnn.2016.10714. [DOI] [PubMed] [Google Scholar]
- 5.Van Hoecke L., Roose K., Ballegeer M., Zhong Z., Sanders N.N., De Koker S., Saelens X., Van Lint S. The Opposing Effect of Type I IFN on the T Cell Response by Non-modified mRNA-Lipoplex Vaccines Is Determined by the Route of Administration. Mol. Ther. Nucleic Acids. 2020;22:373–381. doi: 10.1016/j.omtn.2020.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]