Abstract
Previously, we identified increased maternal circulating DAAM2 mRNA in pregnancies complicated by preterm fetal growth restriction (FGR). Here, we assessed whether circulating DAAM2 mRNA could detect FGR, and whether the DAAM2 gene, known to play roles in the Wnt signalling pathway is expressed in human placenta and associated with dysfunction and FGR. We performed linear regression analysis to calculate area under the ROC curve (AUC) for DAAM2 mRNA expression in the maternal circulation of pregnancies complicated by preterm FGR. DAAM2 mRNA expression was assessed across gestation by qPCR. DAAM2 protein and mRNA expression was assessed in preterm FGR placenta using western blot and qPCR. DAAM2 expression was assessed in term cytotrophoblasts and placental explant tissue cultured under hypoxic and normoxic conditions by qPCR. Small interfering RNAs were used to silence DAAM2 in term primary cytotrophoblasts. Expression of growth, apoptosis and oxidative stress genes were assessed by qPCR. Circulating DAAM2 mRNA was elevated in pregnancies complicated by preterm FGR [p < 0.0001, AUC = 0.83 (0.78–0.89)]. Placental DAAM2 mRNA was detectable across gestation, with highest expression at term. DAAM2 protein was increased in preterm FGR placentas but demonstrated no change in mRNA expression. DAAM2 mRNA expression was increased in cytotrophoblasts and placental explants under hypoxia. Silencing DAAM2 under hypoxia decreased expression of pro-survival gene, BCL2 and oxidative stress marker, NOX4, whilst increasing expression of antioxidant enzyme, HMOX-1. The increased DAAM2 associated with FGR and hypoxia implicates a potential role in placental dysfunction. Decreasing DAAM2 may have cytoprotective effects, but further research is required to elucidate its role in healthy and dysfunctional placentas.
Subject terms: Intrauterine growth, Predictive markers, Genetics research, Gene expression
Introduction
The placenta is a unique organ developed in pregnancy, acting as the interface between the maternal and fetal systems. Healthy placental development creates a high flow, low resistance blood delivery system to facilitate the supply of oxygen and nutrients and removal of waste products. This exchange is essential for optimal fetal growth, and as such, appropriate placental development is crucial to establish a healthy pregnancy. Aberrant placentation can progress to serious pregnancy complications such as fetal growth restriction, which is responsible for significant perinatal morbidity and mortality1.
In the first trimester of pregnancy, cytotrophoblasts, a cell type unique to the placenta regulate the remodelling of maternal spiral arteries. The cytotrophoblasts replace endothelial cells, whilst simultaneously expanding the vessel lumen, as well as forming trophoblast plugs2,3. These plugs create a low oxygen environment for initial placental and fetal development. When the trophoblast plugs disintegrate towards the end of the first trimester, the remodelled arteries can provide a high flow, low resistance blood supply to the placenta and fetus, restoring oxygen levels4.
Impaired trophoblast invasion and remodelling of the maternal spiral arteries causes placental insufficiency, resulting in impaired circulation to the placenta. Blood flow can become pulsatile, damaging the fragile placental tissue resulting in placental ischaemia and increased oxidative stress5. Furthermore, oxygen delivery may be compromised, generating variations in oxygen tension and periods of low oxygen referred to as placental hypoxia6. This failure in placental function can severely affect fetal development7.
Fetal growth restriction is a serious pregnancy complication where a fetus fails to reach its growth potential due to inadequate nutrient supply. The most common cause is uteroplacental insufficiency, where dysfunctional placentation and chronic hypoxia impair fetal growth and development5. Fetal growth restriction is associated with an increased risk of major perinatal injury, cardiovascular, respiratory and neurological morbidities and major risk of mortality, with early onset cases being most serious8–10. Despite being one of the most severe complications of pregnancy, with up to 45% of non-anomalous stillbirths associated with fetal growth restriction11, there are still no treatments that improve placental insufficiency nor accurate means of early detection of placental dysfunction preceeding severe impairment to fetal growth. There is an urgent clinical need to develop better tests for placental insufficiency to decrease perinatal morbidity and mortality.
In a recent multi-center cohort study, we used next generation sequencing to measure cell-free RNA in the blood of pregnant women with preterm fetal growth restriction, fetal acidemia in utero, and stillbirth. Our study identified significantly altered mRNA signatures in the maternal circulation where there was placental insufficiency, preterm fetal growth restriction and fetal hypoxia12. Dishevelled Associated Activator of Morphogenesis 2 (DAAM2) was one of the most differentially regulated genes identified. DAAM2 mRNA was elevated in the circulation of women with preterm fetal growth restriction. Whether these circulating mRNAs originated from the dysfunctional placenta was not established in that study.
DAAM2 is known for its role in the Wnt signalling pathway13,14. Recently, the first study to report on Daam2 in the placenta demonstrated a role in placental vascularization and establishment of the maternal–fetal blood supply in mice15. However, there are no published studies investigating DAAM2 expression or function in the human placenta.
In this study, we aimed to assess whether DAAM2 was expressed in the human placenta and whether gene expression or protein production was altered by gestation, or placental dysfunction associated with preterm fetal growth restriction. We also set out to explore possible functional roles for DAAM2 in the placenta related to growth and dysfunction.
Results
DAAM2 is increased in the circulation of pregnancies complicated by fetal growth restriction
Using next-generation sequencing, we initially discovered DAAM2 expression was increased in the maternal circulation of pregnancies complicated by preterm fetal growth restriction (FGR) in the FOX study cohort12. The majority of the fetal growth restricted cases were significantly growth restricted with a median birthweight centile (using intrauterine fetal charts16) of 0.1 (interquartile range 0.0–0.4; see Table 1 for baseline clinical characteristics of women in the FOX study).
Table 1.
Patient characteristics for cases of Fetal Growth Restriction (FGR) and control cohorts as part of the FOX study.
| Characteristics | FGR cases (n = 128) | Controls (n = 42) | P |
|---|---|---|---|
| Maternal age, years | 32 (6) | 30 (6) | 0.13 |
| Nulliparity | 80 (63%) | 38 (45%) | 0.013 |
| Body-mass index, kg/m2 | 27 (6) | 24 (5) | 0.0009 |
| Smoking during pregnancy | 17 (13%) | 6 (7%) | 0.16 |
| Diabetes during pregnancy | 15 (12%) | 8 (10%) | 0.60 |
| Chronic hypertension | 12 (9%) | 0 (0%) | 0.004 |
| Preeclampsia | 63 (49%) | 3 (4%) | < 0.00001 |
| Absent or reversed end diastolic flow in umbilical artery | 58 (45%) | – | – |
| Median gestational age at blood sampling (wks) | 30.5 (28.6–32.1) | 30.0 (28–32.1) | 0.21 |
| Gestational age at delivery (wks) | 30.5 (28.6–32.1) | 39.4 (39–40.2) | < 0.00001 |
| Birthweight, g | 1023 (315) | 3594 (480) | < 0.00001 |
| Birthweight centiles, corrected for gestationa (median and interquartile range) | 0.1 (0.0–0.4) | 40.8 (27.9–60.9) | < 0.00001 |
| Male sex | 71 (55%) | 46 (55%) | 0.92 |
| Umbilical artery pH, median | 7.27 (7.22–7.3) | – | |
| Umbilical artery pH < 7.2 | 22 (17.2%) | – | |
| Neonatal deaths within 42 days of birth | 2 (2%) | 0 (0%) | 0.42 |
Data are n (%), mean (SD), or median (IQR). Comparison between FGR cases and gestation matched controls is by chi squared analysis and t-test. Each control contributed two blood samples for the analysis, at 28 and 32 weeks gestation. This was done to correct for possible changes in RNA concentrations across gestational age.
aWe used fetal weight reference charts to determine centiles (Hadlock formula, except fetal sex was corrected for).
Here, in the current study we have further analysed the quantitative PCR data presented in our previous study12 to specifically determine the ability of DAAM2 to detect preterm FGR. We log-transformed circulating DAAM2 mRNA expression, demonstrating a highly significant increase (p < 0.0001) in circulating DAAM2 mRNA in pregnancies complicated by preterm fetal growth restriction (Fig. 1a) compared to gestation-matched controls. Additionally, we examined whether maternal circulating DAAM2 mRNA was altered between cases of preterm FGR where fetal acidemia (associated with increased risk of perinatal death17) was apparent (determined by an umbilical artery blood pH < 7.2 (indicating acidosis) versus pH ≥ 7.2 (not acidotic)). We did not detect further altered expression of DAAM2 in the circulation with fetal hypoxia (Supplementary Fig. S1).
Figure 1.
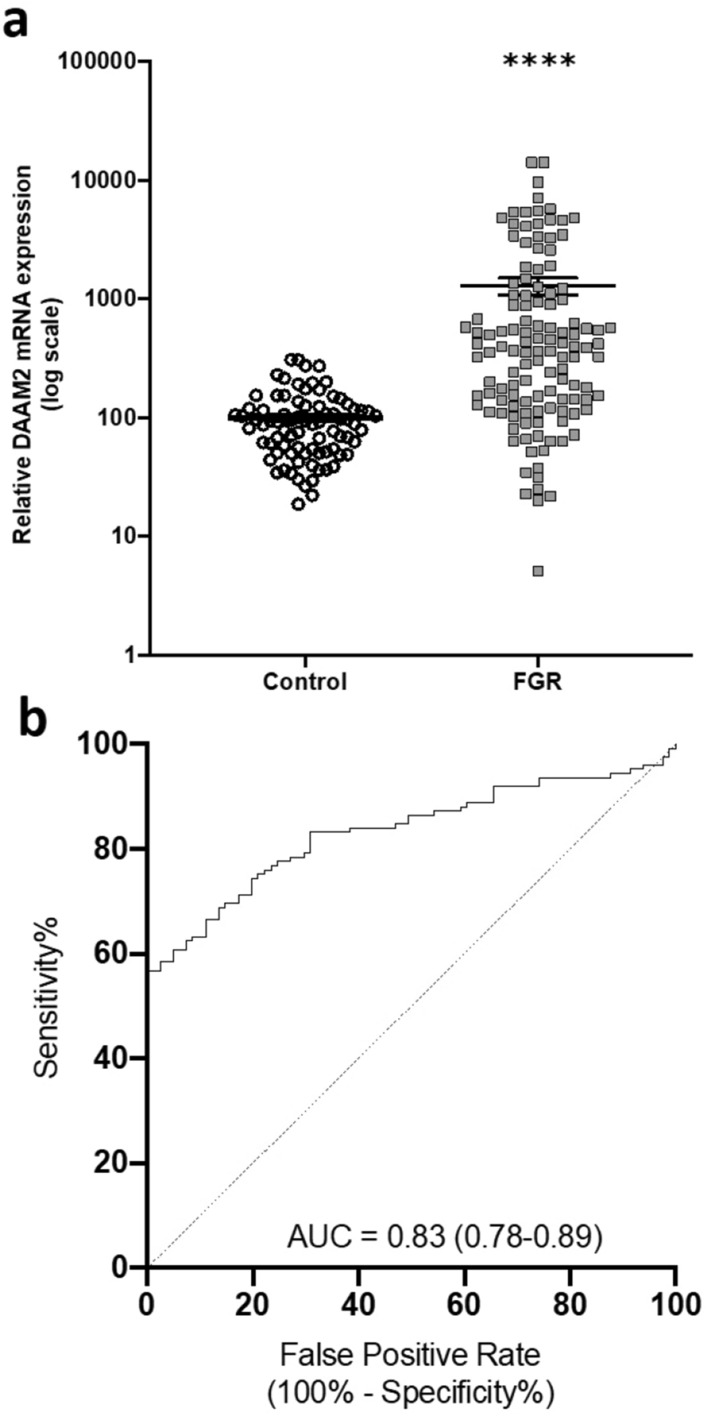
Expression of DAAM2 mRNA in the maternal circulation from pregnancies complicated by preterm fetal growth restriction (FOX Study). DAAM2 mRNA data is expressed as log expression; error bars are median ± IQR. AUC = 0.83; 95% Confidence Interval (0.78–0.89). ****p < 0.0001.
To determine whether DAAM2 RNA could provide a useful test, we performed logistic regression analysis. This provided a test with an area under the receiver operating characteristic (ROC) curve of 0.83 (Fig. 1b). At a specificity of 90.1% (i.e. a 10% screen positive rate) the test had 63.2% sensitivity in identifying preterm fetal growth restriction, with a positive likelihood ratio of 6.4. Thus, circulating DAAM2 mRNA expression demonstrated potential to highlight pregnancies at serious risk of preterm fetal growth restriction and may be useful in a multi-marker test. However, use as a lone marker would require further validation, with comparison to current clinical detection of high-risk pregnancies.
We also found no change in DAAM2 expression when we sub-analysed the cases by coexistent gestational hypertension or preeclampsia (Supplementary Fig. S2). Assessment between 28 and 32 weeks in the control samples revealed no significant increase in DAAM2 expression in the maternal circulation with advancing gestation (data not shown). DAAM2 mRNA was not altered by fetal gender (data not shown).
DAAM2 is expressed in human placenta and increases with advancing gestation
DAAM2 expression was identified in human placental tissue at all gestations examined (first trimester, second trimester and term). There was no significant change in expression between first trimester (7–10 weeks) and second trimester (24–29 weeks) samples. However, a significant increase in expression was observed between the term and first trimester samples (p = 0.008; Fig. 2). DAAM2 expression was also significantly higher at term compared to second trimester gestation (p = 0.034; Fig. 2). Thus, placental DAAM2 expression increases with advancing gestation.
Figure 2.
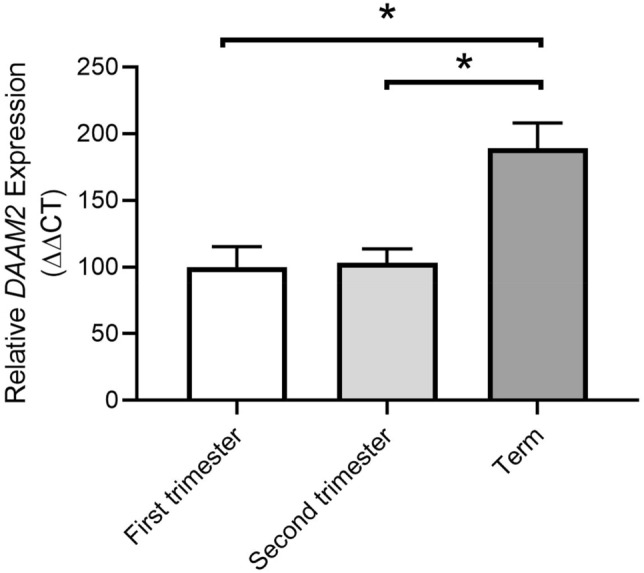
DAAM2 expression in placentas from first trimester, second trimester and term gestation. Expression of DAAM2 is significantly increased at term compared to first trimester and second trimester. There is no change in DAAM2 expression between first trimester and second trimester. Data presented as fold change from first trimester, mean ± SEM. *p < 0.05. First trimester; n = 6, 7–9 weeks. Second trimester; n = 4, 24–29 weeks. Term; n = 9; 38–39 weeks.
DAAM2 protein is increased in placental tissue from pregnancies affected by fetal growth restriction
There was no significant difference in DAAM2 mRNA expression in placental tissue from pregnancies complicated by preterm fetal growth restriction (≤ 34 weeks gestation) compared to gestation-matched preterm control placenta (Fig. 3a). However, placental DAAM2 protein was significantly increased in pregnancies complicated by preterm fetal growth restriction, compared to gestation-matched control placentas (p = 0.049; Fig. 3b,c, Supplementary Fig. S3).
Figure 3.
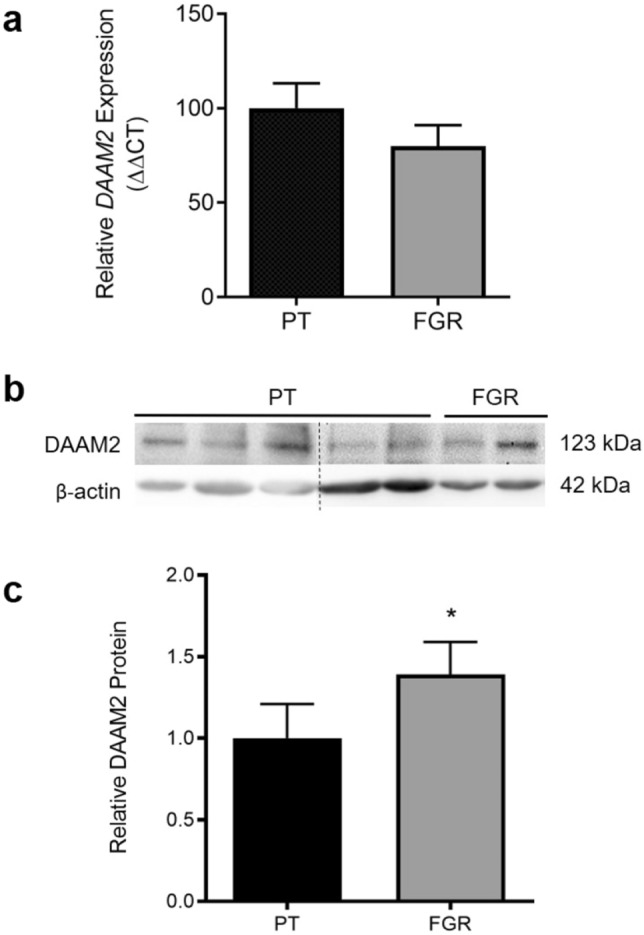
DAAM2 mRNA and protein in preterm placental tissue (≤ 34 weeks). (a) DAAM2 mRNA expression assessed by qPCR. (b) Representative western blot and (c) densitometric analysis of DAAM2 protein. DAAM2 mRNA expression was not significantly changed in fetal growth restricted (FGR; n = 14) tissue compared to preterm control (PT; n = 10) tissue. Relative levels of DAAM2 protein were significantly higher in fetal growth restricted (n = 16) placental tissue compared to preterm control (n = 9) tissue. Protein and mRNA data presented as fold change from control ± SEM. *p < 0.05. Full western blot images are presented in Supplementary Fig. S3.
DAAM2 expression is increased under hypoxia in term placental tissue and isolated cytotrophoblasts
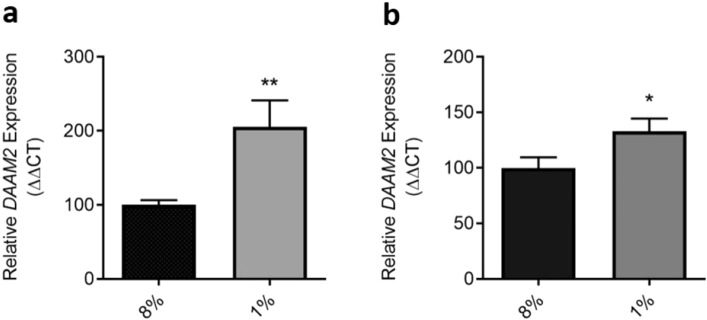
Under hypoxic conditions, DAAM2 mRNA expression was significantly increased in both term cytotrophoblasts (p = 0.005; Fig. 4a) and term placental explants (p = 0.040; Fig. 4b), compared to control cells and tissues cultured under normoxic conditions.
Figure 4.
Expression of DAAM2 mRNA in primary cytotrophoblasts and placental explant tissue under normoxic (8% O2) and hypoxic (1% O2) conditions. Cytotrophoblast (a) and placental explant (b) expression of DAAM2 mRNA is significantly upregulated under hypoxic conditions, compared to normoxic control. Data presented as fold change from control ± SEM. *p < 0.05, **p < 0.01. n = 4–5 experimental replicates (each sample from a different patient), with each experiment performed in triplicate.
Knockdown of DAAM2 in primary cytotrophoblasts
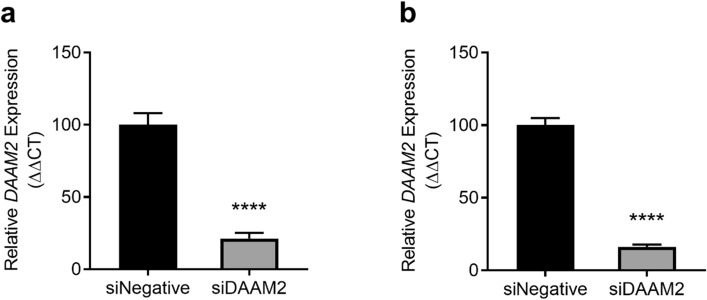
In isolated primary cytotrophoblasts, siRNA directed against DAAM2 significantly decreased expression of DAAM2 mRNA by approximately 80%, under both normoxic (p < 0.0001; Fig. 5a) and hypoxic (p < 0.0001; Fig. 5b) conditions. Importantly, siRNA against DAAM2 did not negatively affect cell survival at either oxygen tension (Supplementary Fig. S4).
Figure 5.
Knockdown of DAAM2 under normoxic (8% O2) and hypoxic (1% O2) conditions. Cytotrophoblast expression of DAAM2 mRNA was significantly knocked down under both normoxic (A) and hypoxic conditions with siRNA against DAAM2 (siDAAM2) compared to the negative control siRNA (siNegative). Data presented as fold change from control ± SEM. ****p < 0.0001. n = 3 experimental replicates (each sample from a different patient), with each experiment performed in triplicate.
Silencing DAAM2 alters expression of apoptosis and oxidative stress markers under hypoxia
To assess the potential functional roles of DAAM2 in the placenta, we assessed expression of important genes in pathways of growth, apoptosis and oxidative stress in cytotrophoblasts where DAAM2 had been silenced under hypoxic conditions.
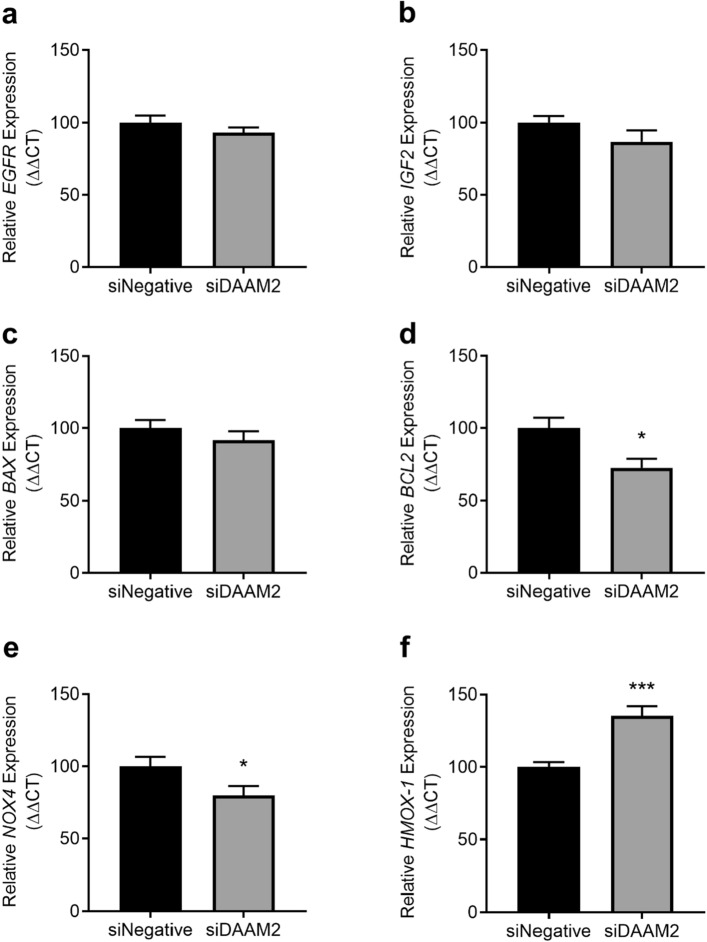
Under hypoxic conditions, knockdown of DAAM2 did not affect the expression of genes involved in placental growth and proliferation: epidermal growth factor receptor (EGFR) and insulin-like growth factor 2 (IGF2) (Fig. 6a,b, respectively). Silencing DAAM2 did not alter expression of the pro-apoptotic gene, BCL2 Associated X (BAX) (Fig. 6c), but significantly decreased pro-survival B-cell lymphoma 2 (BCL2) mRNA expression (p = 0.011; Fig. 6d). mRNA expression of oxidative stress marker, NADPH oxidase 4 (NOX4) was significantly decreased with DAAM2 knockdown compared to the negative siRNA control (p = 0.048; Fig. 6e), whilst the anti-oxidant gene, heme oxygenase 1 (HMOX-1) was significantly increased (p = 0.0002, Fig. 6f).
Figure 6.
Effect of DAAM2 knockdown on expression of cell growth, apoptosis and oxidative stress genes under hypoxia (1% O2). Knockdown of DAAM2 with siRNA (siDAAM2) significantly decreased (d) BCL2, (e) NOX4 and increased (f) HMOX-1 mRNA expression compared to the negative siRNA control (siNegative), but had no effect on (a) EGFR, (b) IGF2 or (c) BAX. Data presented as fold change from control ± SEM. *p < 0.05, ***p < 0.001. n = 3 experimental replicates (each sample from a different patient), with each experiment performed in triplicate.
Silencing DAAM2 did not affect expression of any of these genes under normoxic conditions (Supplementary Fig. S5).
Discussion
In this paper, we identified that circulating DAAM2 mRNA has potential to detect fetal growth restriction (FGR), is expressed in the human placenta throughout gestation and is dysregulated with hypoxia and in disease settings. Furthermore, silencing DAAM2 alters stress markers in the placenta.
DAAM2 is a key regulator of the Wnt signaling pathway, an ancient and evolutionarily conserved pathway that regulates crucial aspects of cell fate determination, cell migration, cell polarity, neural patterning and organogenesis during embryonic development13,14. The first study to report on Daam2 in the placenta was published recently, demonstrating a potential role in placental vascularization and the establishment of the maternal–fetal blood supply in mice15. Importantly, there are no published studies investigating DAAM2 function in the human placenta.
Previously, we identified increased DAAM2 mRNA in the circulation of women whose pregnancies were complicated by early onset fetal growth restriction by next generation sequencing12. However, in that report we did not explore DAAM2 expression further as a marker for fetal growth restriction as we focused on other genes, nor did we perform mechanistic studies in the placenta or explore the potential source for the elevated mRNA in the maternal circulation.
In this study, we performed a further analysis of circulating DAAM2 mRNA concentrations where we characterised the diagnostic potential of DAAM2. We found that circulating DAAM2 mRNA identified preterm FGR with an area under the ROC curve (AUC) of 0.83. This suggests DAAM2 may have potential to predict the risk of preterm FGR. However, as noted in our prior report, a possible limitation as a clinical biomarker is that circulating levels of DAAM2 mRNA may be altered by the administration of corticosteroids12. Thus, further studies are needed to determine whether this differential expression in the circulation remains in a population that has not received corticosteroids.
Regardless of biomarker status, our data (combined with the recent report in animal models showing the gene plays an important role in placental development15) suggest DAAM2 may be involved in placental development, fetal growth and the pathological condition of fetal growth restriction. The current study demonstrates for the first time that DAAM2 is expressed by the human placenta. We confirmed expression of DAAM2 in first trimester, second trimester and term placenta, but interestingly found that DAAM2 expression was highest at term. DAAM2 expression was detected as early as seven weeks gestation, suggesting a possible role for DAAM2 in early placental vascularistion and establishment of the maternal–fetal blood supply, as seen in mice15. Both mouse and human placenta are hemochorial, and both contain fetal capillaries surrounded by layers of trophoblasts directly bathing in maternal blood18. However, there are also key differences between mouse and human placental vascularisation, thus further investigation is needed to explore the potential role for DAAM2 in the first trimester.
Consistent with our discovery in the maternal circulation, we identified an increase in DAAM2 protein in human placental tissue from pregnancies complicated by early onset fetal growth restriction. However, no differences were detected in placental DAAM2 mRNA expression. It is important to note that in the placental samples available to examine DAAM2 mRNA expression, there was a significant difference in gestational age between our controls and fetal growth restriction-complicated pregnancies. It is difficult to ascertain whether this difference is clinically significant, but given we identified a difference in DAAM2 expression across gestation, this may confound interpretation of the mRNA findings. Examination of a larger cohort of early onset cases and gestation-matched controls would be of value to clarify these discrepant findings. Additionally, as with all studies, mRNA expression is not always translated to protein production, hence may be a point of difference. Importantly, in these placental studies we assessed DAAM2 protein and mRNA expression in samples where corticosteroids were given in both the controls and pregnancies complicated by fetal growth restriction. Therefore, the finding of increased production of DAAM2 in the control and fetal growth restricted placentas are not confounded by corticosteroid administration. The control placentas used in these studies were carefully selected, minimising confounding effects. However, given the control placental tissue was obtained from preterm deliveries, it remains that they are not perfect controls.
Placental hypoxia plays an important role in placental dysfunction, and consequently is a contributing factor to the pathophysiology of fetal growth restriction6. Accordingly, we examined DAAM2 expression in the placenta under hypoxic conditions, finding that DAAM2 mRNA expression was increased in both isolated cytotrophoblasts and placental explant tissue. This suggests that hypoxia regulates DAAM2 expression, and is consistent with the dramatically increased DAAM2 mRNA in the maternal circulation of pregnancies complicated by severe fetal growth restriction12.
Given our finding that DAAM2 was increased in the dysfunctional placenta, we examined whether reducing its expression could be beneficial and confer protection or enhance expression of growth associated genes. Silencing DAAM2 expression in primary cytotrophoblasts under hypoxia did not impair cell viability, suggesting that DAAM2 is not essential for trophoblast cell survival. Loss of DAAM2 did not alter expression of IGF2 and EGFR, key genes whose dysregulation has been found to be associated with impaired placental development and fetal growth restriction19–21, thus DAAM2 is unlikely to be driving these pathways. Additionally, the pro-apoptotic gene BAX22 was not altered with DAAM2 suppression. However, silencing DAAM2 decreased expression of the pro-survival gene, BCL2. It is important to note that in addition to its pro-survival role, BCL2 also acts through non-canonical pathways, including regulation of mitrochondrial membrane permeabilization and oxidative stress23,24. It is therefore unsurprising that silencing DAAM2 also altered expression of NOX4, a marker of oxidative stress25. Silencing DAAM2 also increased mRNA expression of the cytoprotective antioxidant enzyme HMOX-126–28. Decreasing oxidative stress in the placenta may have important benefits, especially when hypoxia is driving damage and dysfunction. These findings suggest DAAM2 may have an important role in placental dysfunction, and suppressing DAAM2 in the placenta could be beneficial. Future studies examining the effect of excess DAAM2 may facilitate our understanding of the function of DAAM2 in the placenta.
While a clear role for DAAM2 in the human placenta is not yet apparent, collectively these data and the identification of Daam2 in the murine placenta15 suggest important roles for DAAM2 in the placenta.
In this report, we demonstrated increases in DAAM2 expression in placentas complicated by early onset fetal growth restriction and hypoxia, indicating a potential role in the dysfunctional placenta. Additionally, we identified that placental expression of DAAM2 increases with advancing gestation, and that suppression of DAAM2 enhanced cytoprotective gene pathways in hypoxic cytotrophoblasts. A strength of this study is the use of prized clinical samples, and collaboration with clinical expertise. Another strength of this study is the assessment of DAAM2 in primary placental cells and tissues, rather than cell lines. However, further studies are required to expand these findings and uncover the role of DAAM2 in the healthy and dysfunctional placenta.
Methods
Fetal OXygenation (FOX) Study
Maternal peripheral blood was collected as part of the FOX Study as previously described12. In summary, blood was collected from 128 women with preterm growth restricted fetuses and from 42 women at matched gestations (28 and 34 weeks) with appropriately grown fetuses that progressed to birth at term, across six tertiary hospitals (in Australia and New Zealand). Table 1 provides the baseline clinical characteristics of study participants in the FOX study. Samples were collected directly into PAXgene Blood RNA tubes (Pre-Analytix, Hombrechtikon, Switzerland) to maintain nucleic acid stability and processed according to manufacturer’s instructions. All blood samples were collected after corticosteroid administration, immediately prior to delivery.
Ethical approval was obtained from all institutions (Approval numbers: MHW R11/04, RWH + Sunshine Hospital 10/41, MMH 1928M, RHW 12/240, RNSH 1305-151M, NWH, ACH 12/NTA/96/AM02) and all women provided written, informed consent. Experiments were performed following the relevant institutional guidelines and regulations.
Preterm fetal growth restriction was defined as a customized birthweight < 10th centile (www.gestation.net, Australian parameters) requiring iatrogenic delivery prior to 34 weeks gestation with uteroplacental insufficiency (asymmetrical growth + abnormal artery Doppler velocimetry ± oligohydramnios ± abnormal fetal vessel velocimetry). Fetal growth restriction due to infection, chromosomal or congenital abnormalities, and multiple pregnancy was excluded.
Fetal hypoxic status in the preterm growth restricted cohort was determined by collecting umbilical artery blood at birth and measuring the pH, where hypoxia was defined as pH < 7.2, and normoxia as pH ≥ 7.2.
Placental tissue collection
Ethical approval was obtained from the Mercy Health Human Research Ethics Committee (R11/34) and Austin Health Human Research Ethics Committee (HREC/18/Austin/44). Women presenting to the Mercy Hospital for Women (Heidelberg, Victoria) and The Northern Hospital (Epping, Victoria) gave informed, written consent for the collection of tissue. Women presenting to the Broadmeadows Health Service (Broadmeadows, Victoria) gave informed, written consent for the collection of conceptus samples at surgical termination of pregnancy. Experiments were performed following institutional guidelines and regulations.
First trimester placental tissue was obtained from conceptus material collected at surgical terminations of singleton pregnancies (7–10 weeks gestation) under general anaesthesia via curettage or a combination of aspiration and curettage (according to the surgeon's preference). Placental tissue was identified and isolated from conceptus material, then washed in phosphate buffered saline (PBS). Placental tissue was transferred to RNAlater for 48 h, after which the tissue was snap frozen and stored at − 80 °C for subsequent analysis. Patient characteristics are described in Table 2.
Table 2.
Patient characteristics for placental tissue used to assess DAAM2 expression across gestation.
| First trimester (n = 6) | Second trimester (n = 4) | Term (n = 9) | |
|---|---|---|---|
| Maternal age, years Median (range) | 29 (25–39) | 23.5 (19–27)b | 33 (27–30)b |
| Gestational age at sample collection, weeks Median (IQR) | 8.6 (7.55–8.875)a,c | 27.25 (24.95–28.2)a,b | 39.10 (39.0–39.3)b,c |
| Body mass index (kg/m2) Median (IQR) | 23.47 (21.83–26.94) | 40.90 (24–41) | 26.22 (24.05–29.40) |
| Parity no | |||
| 0 | 2 | 0 | 1 |
| 1 | 3 | 4 | 4 |
| ≥ 2 | 1 | 0 | 4 |
| Mode of delivery | |||
| Vaginal | – | 0 | 0 |
| Caesarean Section | – | 4 | 9 |
| Birth weight (g) Median (IQR) | – | 935 (736.3–1225)b | 3360 (3050–3560)b |
BMI data unavailable for n = 1 second trimester sample.
aSignificant difference between first trimester and second trimester samples.
bSignificant difference between second trimester and term samples.
cSignificant difference between first trimester and term samples.
Placentas were obtained from cases of preterm fetal growth restriction (delivery ≤ 34 weeks gestation), defined as customized birth weight < 10th centile according to Australian population charts29. Cases associated with congenital infection, chromosomal or congenital abnormalities, multiple pregnancies and preeclampsia were excluded.
Control healthy, term (delivery 37–40 weeks gestation) and preterm placentas (delivery ≤ 34 weeks gestation) were collected from normotensive pregnancies where a fetus of normal customized birth weight centile (> 10th centile relative to gestation) was delivered. Placentas with evidence of chorioamnionitis (confirmed by placental histopathology) were excluded.
Term and preterm placental tissue was collected within 30 min of delivery. Preterm delivery in our controls was predominantly for iatrogenic conditions including vasa previa, suspected placental abruption and fetal anaemia. For preterm (fetal growth restriction and control) tissue collection, samples from four sites of the placenta were washed in ice cold PBS and preserved in RNAlater for 48 h, after which the tissue was snap frozen and stored at − 80 °C for subsequent analysis. Patient characteristics are described in Tables 2, 3 and 4.
Table 3.
Patient characteristics of women with fetal growth restriction and control samples for gene (mRNA) expression studies.
| Preterm controls (n = 10) | Fetal growth restriction (n = 14) | |
|---|---|---|
| Maternal age, years Median (IQR) | 34 (26.5–37.5) | 30 (25.3–33.5) |
| Gestational age at delivery, weeks Median (IQR) | 30 (29.4–31.6) | 32.7 (30.9–34.0)* |
| Body mass index (kg/m2) Median (IQR) | 28.4 (24.0–30.0) | 25.8 (18.75–29.5) |
| Parity no. (%) | ||
| 0 | 2 (20.0) | 9 (64.3) |
| 1 | 4 (40.0) | 2 (14.3) |
| ≥ 2 | 4 (40.0) | 3 (21.4) |
| Highest SBP prior to delivery (mmHg) Median (IQR) | 120 (110–126.3) | 120 (115–126.3) |
| Highest DBP prior to delivery (mmHg) Median (IQR) | 70 (67.5–76.25) | 76.5 (70–83.5)* |
| Mode of delivery | ||
| Vaginal (%) | 0 (0) | 0 (0) |
| Caesarean Section (%) | 10 (100) | 14 (100) |
| Birth weight (g) Median (IQR) | 1496 (1322–2011) | 1182 (973–1658) |
BMI data unavailable for n = 3 preterm controls, n = 1 fetal growth restriction sample.
*p < 0.05.
Table 4.
Patient characteristics of women with fetal growth restriction and gestation matched control samples for protein studies.
| Preterm controls (n = 16) | Fetal growth restriction (n = 9) | |
|---|---|---|
| Maternal age, years Median (IQR) | 28.5 (25.25–36.75) | 29 (21–32.5) |
| Gestational age at delivery, weeks Median (IQR) | 30 (29.4–32.08) | 31.4 (30.7–33.3) |
| Body mass index (kg/m2) Median (IQR) | 26 (21.65–32.15) | 20 (18.25–27.7) |
| Parity no. (%) | ||
| 0 | 5 (33.3) | 7 (77.8) |
| 1 | 8 (53.3) | 1 (11.1) |
| ≥ 2 | 2 (13.3) | 1 (11.1) |
| Highest SBP prior to delivery (mmHg) Median (IQR) | 120 (119.3–130.0) | 120 (113.5–127.5) |
| Highest DBP prior to delivery (mmHg) Median (IQR) | 70 (63.5–78.75) | 78 (70–81.5) |
| Mode of delivery | ||
| Vaginal (%) | 3 (20) | 0 (0) |
| Caesarean Section (%) | 12 (80) | 9 (100) |
| Birth weight (g) Median (IQR) | 1587 (1277–1976) | 1000 (893.0–1375)* |
BMI data unavailable for n = 3 preterm control samples.
*p < 0.05.
Placental explant isolation and culture
Placentas were obtained from normal term pregnancies (> 37 weeks gestation) at elective Caesarean section for explant dissection. Placental explants were isolated with maternal and fetal surfaces removed. Three small pieces of placenta totalling 10–15 mg of tissue per well were cultured in 24-well plates, each containing media made up of Gibco Dulbecco's Modified Eagle Medium (DMEM; ThermoFisher Scientific, Scoresby, Vic), supplemented with 10% fetal calf serum (FCS; Sigma-Aldrich, St Louis, USA) and 1% Anti-Anti (AA; Life Technologies, Carlsbad, CA, USA). Explants were cultured under 8% O2, 5% CO2 at 37 °C overnight (16–18 h). After replacement with fresh media (DMEM, 10% FCS, 1%AA), explant tissue was cultured at 37 °C for 24 h under 8% O2 (normoxic conditions) or 1% O2 (hypoxia). Following this, explant tissue was weighed, snap frozen and stored at − 80 °C for subsequent analysis.
Primary cytotrophoblast isolation and culture
Human primary cytotrophoblasts were isolated from normal term placentas from elective Caesarean section as previously described30. The cells were plated in media (DMEM, 10% FCS, 1%AA) on fibronectin (10 µg/mL; BD Bioscience, USA) coated culture plates. Viable cells were incubated under 8% O2, 5% CO2 at 37 °C overnight to equilibrate. After replacement with fresh media, cytotrophoblasts were cultured at 37 °C for 24 h under 8% O2 (normoxic conditions) or 1% O2 (hypoxia). Following this, cells were collected for RNA extraction.
Silencing genes in primary cytotrophoblasts
Small interfering RNA (siRNA) designed against DAAM2 (M-014010-00-0005; Dharmacon, Lafayette, CA, USA) or a negative siRNA control (Qiagen, Valencia, CA, USA) were combined with lipofectamine (RNAiMax; Invitrogen) in optimem (ThermoFisher Scientific) to complex for 20 min at room temperature. After equilibration of the isolated cytotrophoblasts overnight (described above), fresh trophoblast media (DMEM, 10% FCS, no AA) was added to each well and siRNA complexes added in a dropwise manner. Cytotrophoblast cells with siRNA were cultured at 37 °C for 48 h under 8% O2 (normoxic conditions) or 1% O2 (hypoxia). Following this, media and cells were collected for subsequent analysis.
MTS cell viability assay
Cell viability was assessed following siRNA treatment using the MTS assay, CellTiter 96-AQueous One Solution (Promega, Madison WI) according to manufacturer instructions.
Quantitative real time polymerase chain reaction (qPCR)
Total RNA was extracted from whole blood via PAXgene Blood miRNA Kit (Pre-Analytix, Hombrechtikon, Switzerland) according to manufacturer’s instructions, as described previously12. RNA was extracted from placental tissue (collected from first trimester, preterm and term gestations), cultured explants and isolated primary cytotrophoblasts using the Qiagen RNeasy Mini Kit, according to the manufacturer’s instructions. RNA concentration was quantified using a Nanodrop 2000 spectrophotometer (ThermoFisher Scientific, Waltham, MA). RNA was converted to cDNA using the Applied Biosystems High-Capacity cDNA Reverse Transcription Kit (Thermofisher), as per manufacturer’s instructions on the iCycler iQ5 (Biorad) or MiniAmp Thermal Cycler (Applied Biosystems, CA, USA). Quantitative Taqman PCR was performed to quantify mRNA expression of DAAM2, BAX, BCL2, EGFR, IGF2, NOX4 and HMOX-1 (Hs00322497_m1, Hs00180269_m1, Hs00608023_m1, Hs01076078_m1, Hs04188276_m1, Hs00418356_m1, and Hs01110250_m1 respectively; Life Technologies), as well as reference genes for blood: YHWAZ, B2M and GUSB (Hs01122454_m1, Hs00187842_m1, Hs00939627_m1; Life Technologies), cytotrophoblast cells: YWHAZ and explants and placental tissue: TOP1 and CYC1 (Hs01122454_m1, Hs00243257_m1, and Hs00357717_m1 respectively; Life Technologies). Stability of reference genes was confirmed for each tissue type and used appropriately. Taqman qPCR was performed on the CFX384 (Biorad) or QuantStudio 5 (Applied Biosystems) with the following run conditions: 50 °C for 2 min, 95 °C for 10 min, 95 °C for 15 s, 60 °C for 1 min or 50 °C for 2 min, 95 °C for 20 s, 95 °C for 3 s, 60 °C for 30 s (40 cycles). All data were normalized to the appropriate reference gene as an internal control and calibrated against the average Ct of the control samples. All cDNA samples were run in duplicate.
Western blot analysis
Protein lysates were extracted from placental tissue from early onset preterm fetal growth restricted pregnancies (≤ 34 weeks) using RIPA lysis buffer containing proteinase and phosphatase inhibitors (Sigma Aldrich). Protein concentrations were assessed with Pierce BCA Protein Assay Kit (ThermoFisher Scientific). Placental lysates (20 µg) were separated on 10% gels and PVDF membranes (Millipore; Billerica, MA, United States). Membranes were blocked with 5% skim milk, prior to overnight incubation with DAAM2 primary antibody at 1:500 in 5% skim milk/TBS-T (GTX33141, Sapphire Bioscience, NSW, Australia). Blots were incubated with anti-rabbit secondary antibody at 1:2500 in 5% skim milk for 1 h (W401; Promega, VIC, Australia). Membranes were developed with enhanced chemiluminescence reagent (GE Healthcare Life Sciences, NSW, Australia) and detected using the ChemiDoc XRS (BioRad). β-actin acted as the loading control at 1:20,000 in 5% skim milk (Santa Cruz, Texas, USA). Densitometry was performed on images of the blots using ImageJ software (NIH, Bethesda, MD, USA).
Statistical analysis
All in vitro experiments were performed with technical triplicates and repeated with n ≥ 3 different patient samples. Data were tested for normal distribution and statistically tested as appropriate. Either an unpaired t-test (parametric) or Mann–Whitney test (non-parametric) was used. The area under the receiver operating curve (AUC) was calculated to determine the sensitivity/specificity performance for DAAM2. All data are expressed as mean ± SEM. P-values < 0.05 were considered significant. Statistical analysis was performed using GraphPad Prism 8 software (GraphPad Software, Inc.; San Diego, CA, USA).
Supplementary Information
Acknowledgements
The authors acknowledge clinical research midwives Gabrielle Pell, Rachel Murdoch, Genevieve Christophers, Eleanor Johnson, the obstetric clinical and midwifery staff and patients at the Mercy Hospital for Women (Heidelberg), Northern Hospital (Epping) and Broadmeadows Health Service for provision of placental tissue.
Author contributions
N.D.A.—Data generation, literature search, data analysis, interpretation, writing. S.B., N.K.B.—Assistance with data collection. N.P.—Characterisation of clinical cohorts. T.J.K.L.—Intellectual input and interpretation. O.S., K.G., S.P., A.H., J.M.S., S.S., S.C.K., S.P.W.—Sample collection and characterisation of clinical cohorts. L.H.—Intellectual input, sample collection and interpretation. S.T.—Intellectual input, sample collection and characterisation of clinical cohorts. N.J.H.—Study design, data analysis and interpretation, intellectual input, writing, attained funding support. N.D.A wrote the main manuscript text with assistance from N.J.H. All authors reviewed the manuscript.
Funding
The NHMRC provided salary support (#1146128 to NJH, #1159261 to TJKL, and #1136418 to ST). The funders had no role in study design, data collection, analysis, decision to publish or the preparation of the manuscript.
Data availability
The datasets generated during and analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-84785-7.
References
- 1.Sharma D, Shastri S, Sharma P. Intrauterine growth restriction: Antenatal and postnatal aspects. Clin. Med. Insights Pediatr. 2016;10:67–83. doi: 10.4137/CMPed.S40070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Damsky CH, Fisher SJ. Trophoblast pseudo-vasculogenesis: Faking it with endothelial adhesion receptors. Curr. Opin. Cell Biol. 1998;10:660–666. doi: 10.1016/S0955-0674(98)80043-4. [DOI] [PubMed] [Google Scholar]
- 3.Hannan NJ, Jones RL, White CA, Salamonsen LA. The chemokines, CX3CL1, CCL14, and CCL4, promote human trophoblast migration at the feto-maternal interface. Biol. Reprod. 2006;74:896–904. doi: 10.1095/biolreprod.105.045518. [DOI] [PubMed] [Google Scholar]
- 4.Huppertz, B., Gauster, M., Orendi, K., Konig, J. & Moser, G. Oxygen as modulator of trophoblast invasion. J. Anat. 14 (2009). [DOI] [PMC free article] [PubMed]
- 5.Burton GJ, Jauniaux E. Pathophysiology of placental-derived fetal growth restriction. Am. J. Obstet. Gynecol. 2018;218:S745–S761. doi: 10.1016/j.ajog.2017.11.577. [DOI] [PubMed] [Google Scholar]
- 6.Kingdom JC, Kaufmann P. Oxygen and placental villous development: Origins of fetal hypoxia. Placenta. 1997;18:613–621. doi: 10.1016/S0143-4004(97)90000-X. [DOI] [PubMed] [Google Scholar]
- 7.Hutter D, Kingdom J, Jaeggi E. Causes and mechanisms of intrauterine hypoxia and its impact on the fetal cardiovascular system: A review. Int. J. Pediatr. 2010;2010:401323. doi: 10.1155/2010/401323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Malhotra A, et al. Neonatal morbidities of fetal growth restriction: Pathophysiology and impact. Front. Endocrinol. (Lausanne) 2019;10:55. doi: 10.3389/fendo.2019.00055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Caradeux J, Martinez-Portilla RJ, Basuki TR, Kiserud T, Figueras F. Risk of fetal death in growth-restricted fetuses with umbilical and/or ductus venosus absent or reversed end-diastolic velocities before 34 weeks of gestation: A systematic review and meta-analysis. Am. J. Obstet. Gynecol. 2018;218:S774–S782. doi: 10.1016/j.ajog.2017.11.566. [DOI] [PubMed] [Google Scholar]
- 10.Malhotra A, et al. Neonatal morbidities of fetal growth restriction: Pathophysiology and impact. Front. Endocrinol. 2019 doi: 10.3389/fendo.2019.00055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.McCowan LM, Figueras F, Anderson NH. Evidence-based national guidelines for the management of suspected fetal growth restriction: Comparison, consensus, and controversy. Am. J. Obstet. Gynecol. 2018;218:S855–S868. doi: 10.1016/j.ajog.2017.12.004. [DOI] [PubMed] [Google Scholar]
- 12.Hannan NJ, et al. Circulating mRNAs are differentially expressed in pregnancies with severe placental insufficiency and at high risk of stillbirth. BMC Med. 2020;18:145. doi: 10.1186/s12916-020-01605-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lee HK, et al. Daam2-PIP5K is a regulatory pathway for Wnt signaling and therapeutic target for remyelination in the CNS. Neuron. 2015;85:1227–1243. doi: 10.1016/j.neuron.2015.02.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lee HK, Deneen B. Daam2 is required for dorsal patterning via modulation of canonical Wnt signaling in the developing spinal cord. Dev. Cell. 2012;22:183–196. doi: 10.1016/j.devcel.2011.10.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nakaya MA, et al. Placental defects lead to embryonic lethality in mice lacking the Formin and PCP proteins Daam1 and Daam2. PLoS ONE. 2020;15:e0232025. doi: 10.1371/journal.pone.0232025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pritchard NL, et al. Identification of the optimal growth charts for use in a preterm population: An Australian state-wide retrospective cohort study. PLoS Med. 2019;16:e1002923. doi: 10.1371/journal.pmed.1002923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baschat AA, et al. Predictors of neonatal outcome in early-onset placental dysfunction. Obstet. Gynecol. 2007;109:253–261. doi: 10.1097/01.Aog.0000253215.79121.75. [DOI] [PubMed] [Google Scholar]
- 18.Georgiades P, Ferguson-Smith AC, Burton GJ. Comparative developmental anatomy of the murine and human definitive placentae. Placenta. 2002;23:3–19. doi: 10.1053/plac.2001.0738. [DOI] [PubMed] [Google Scholar]
- 19.Constância M, et al. Placental-specific IGF-II is a major modulator of placental and fetal growth. Nature. 2002;417:945–948. doi: 10.1038/nature00819. [DOI] [PubMed] [Google Scholar]
- 20.Fondacci C, et al. Alterations of human placental epidermal growth factor receptor in intrauterine growth retardation. J. Clin. Investig. 1994;93:1149–1155. doi: 10.1172/JCI117067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sibilia M, Wagner EF. Strain-dependent epithelial defects in mice lacking the EGF receptor. Science. 1995;269:234–238. doi: 10.1126/science.7618085. [DOI] [PubMed] [Google Scholar]
- 22.Brady HJM, Gil-Gómez G. Molecules in focus Bax. The pro-apoptotic Bcl-2 family member, Bax. Int. J. Biochem. Cell Biol. 1998;30:647–650. doi: 10.1016/S1357-2725(98)00006-5. [DOI] [PubMed] [Google Scholar]
- 23.Chong, S. J. F. et al. Noncanonical cell fate regulation by Bcl-2 proteins. Trends Cell Biol.10.1016/j.tcb.2020.03.004. [DOI] [PubMed]
- 24.Czabotar PE, Lessene G, Strasser A, Adams JM. Control of apoptosis by the BCL-2 protein family: Implications for physiology and therapy. Nat. Rev. Mol. Cell Biol. 2014;15:49–63. doi: 10.1038/nrm3722. [DOI] [PubMed] [Google Scholar]
- 25.Bedard K, Krause K-H. The NOX family of ROS-generating NADPH oxidases: Physiology and pathophysiology. Physiol. Rev. 2007;87:245–313. doi: 10.1152/physrev.00044.2005. [DOI] [PubMed] [Google Scholar]
- 26.Morse D, Choi AM. Heme oxygenase-1: The “emerging molecule” has arrived. Am. J. Respir. Cell Mol. Biol. 2002;27:8–16. doi: 10.1165/ajrcmb.27.1.4862. [DOI] [PubMed] [Google Scholar]
- 27.Onda K, et al. Proton pump inhibitors decrease soluble fms-like tyrosine kinase-1 and soluble endoglin secretion, decrease hypertension, and rescue endothelial dysfunction. Hypertension. 2017;69:457–468. doi: 10.1161/HYPERTENSIONAHA.116.08408. [DOI] [PubMed] [Google Scholar]
- 28.Onda K, et al. Sofalcone upregulates the nuclear factor (erythroid-derived 2)-like 2/heme oxygenase-1 pathway, reduces soluble fms-like tyrosine kinase-1, and quenches endothelial dysfunction: Potential therapeutic for preeclampsia. Hypertension. 2015;65:855–862. doi: 10.1161/HYPERTENSIONAHA.114.04781. [DOI] [PubMed] [Google Scholar]
- 29.Dobbins TA, Sullivan EA, Roberts CL, Simpson JM. Australian national birthweight percentiles by sex and gestational age, 1998–2007. Med. J. Aust. 2012;197:291–294. doi: 10.5694/mja11.11331. [DOI] [PubMed] [Google Scholar]
- 30.Kaitu’u-Lino, T. U. J. et al. Original Article: Characterization of protocols for primary trophoblast purification, optimized for functional investigation of sFlt-1 and soluble endoglin. Pregnancy Hypertens. Int. J. Women’s Cardiovasc. Health4, 287–295. 10.1016/j.preghy.2014.09.003 (2014). [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and analysed during the current study are available from the corresponding author on reasonable request.