Abstract
The literature date estimated that about 5% of all oral cavity cancers are hard palate cancers while soft palate cancers account for about 5-12% of oropharyngeal cancers. Although rare, usually these tumors had a more aggressively behavior than other oral cancer sites. That is why our study aimed to investigate comparatively the epidemiological, clinical and histopathological peculiarities of the two palatal sites of oral squamous cell carcinomas. We conducted a retrospective study limited to a period of 10 years in a single medical institution to investigate the morphoclinical profile of such tumors. We found that patients with hard palate SCCs had an average age slightly larger compared to those who developed soft palate tumors. Also, those with hard palate tumors are mostly diagnosed in less advanced stages compared to those at the level of the soft palate, and implicitly the former had a longer survival time. Histopathologically the most encountered hard palate SCC were the conventional well-differentiated tumor, and from the peculiar SCC variant the papillary and verrucous forms while for the soft palate SCC prevailed the moderate and poor differentiated conventional SCC and from the peculiar SCC variant the basaloid and acantholytic forms. In conclusion hard palate tumors differ in many aspects from those of the soft palate, and thus specification of the origin tumor site become important for the assessment of prognosis, treatment and survival outcome of such patients.
Keywords: Epidemiology, histopathology, oral cavity, palate, squamous cell carcinomas
Introduction
In 2018, according to the GLOBOCAN the world wide estimated incidence of oral cancers was of 354,864 new cases, being ranked as the 18th most encountered cancer sites with approximatively 50% of these patients dying, representing 1.9% of all cancer deaths in all sites [1].
Variations in the incidence of oral cancer was noticed not only geographically but also by anatomic site, sex, race/ethnicity, and age group [2].
Thus, U.S. Cancer Statistics analyzing trends in incidence of oral cancers during 2007-2016 showed increases in the incidence rate for the base of tongue, anterior tongue, gum, and tonsil cancers, while for the lip, floor of mouth, soft and hard palate was recorded a decrease in the incidence rate.
Differences were also noticed regarding the distribution of these cancers in hard and soft palate.
At least 50% of all hard palate cancers seems to be squamous cell carcinomas (SCC), while in the soft palate the incidence of this tumor is around 70% [3].
Even rare compared with other oral cancer sites by its locally aggressiveness palate squamous cell carcinomas may cause significant disabling functional morbidities involving speech, mastication and swallowing [4,5].
According to these our study aimed to investigate the epidemiological and morphological profile of palate SCCs diagnosed in the last decade at the Emergency Clinical County Hospital from Craiova.
Materials and Methods
We retrospectively reviewed data of 45 cases with palate SCC, treated in the Department of Oral and Maxillofacial Surgery from Emergency Clinical County Hospital from Craiova, from January 2010 to December 2019.
From the medical records were reviewed data regarding age, gender, tumor topography (hard versus soft palate), clinical TNM stage, clinical appearance, the presence or absence of clinically detectable lymph node metastases, data related to the loco regional extension of the tumor and survival period after surgery.
From the archive of the Pathology Laboratory we reviewed the corresponding histopathological slides according to the diagnostic criteria established by WHO classification (2005) [6].
The study has been approved by the Ethics Comitee of the University of Medicine and Pharmacy of Craiova, and all patients signed a written informed consent regarding their participation in the study.
Besides histopathological variants of SCC other variables of interest were: the malignancy degree, presence and absence of bone invasion, presence and absence of muscle invasion, presence and absence of perineural invasion, presence and absence of lymph node metastasis.
For statistical analysis we used the SPSS version 12 software and descriptive statistics presenting the quantitative variable in form of absolute and relative frequencies.
The Chi-squared test was used to compare the variables, with p<0.05 as significant level.
Results
Our casuistry includes 45 cases of palate SCCs most of them being males, with gender ratios of 5.43:1 in favor of males.
Patients ranged in age from 35 through 81 years with a peak incidence occurred during the six and seventh decade, and with the average age of 60.17 years.
Most patients came from urban areas, respectively 62.22% of all investigated cases. The most involved anatomic region of the palate was the hard palate with 24 cases, the remainder tumors developed in the soft palate.
Histopathologically the most encountered lesion was the conventional SCC with 38 cases, from which 17 cases were well differentiated, 13 cases moderate differentiated and 8 cases were grouped as poor differentiated forms.
The remaining 7 cases were classified as peculiar histological subtypes of SCC with 2 cases of basaloid SCC, 2 cases of papillary SCC, 2 cases of acantholytic SCC and one cases of verrucous SCC.
Regarding tumor topography in hard palate developed more frequently the well differentiated conventional SCC (52%), followed by the moderate differentiated form (20%), and the rarest were the poor differentiated form (12%) and the peculiar SCC variant (12%).
From the last category being part 2 cases of papillary SCC and one cases of verrucous SCC.
The soft palate lesions were dominated by moderate conventional SCC (38.1%), followed poor differentiated variant (23.8%), well differentiated tumors (19.05%) and peculiar SCC variant (19.05%), represented by 2 cases of basaloid SCC, and 2 cases of acantholytic SCC.
Histologically the conventional SCC presented as invasive islands and cords of malignant squamous epithelial cells (Figure 1A).
Figure 1.
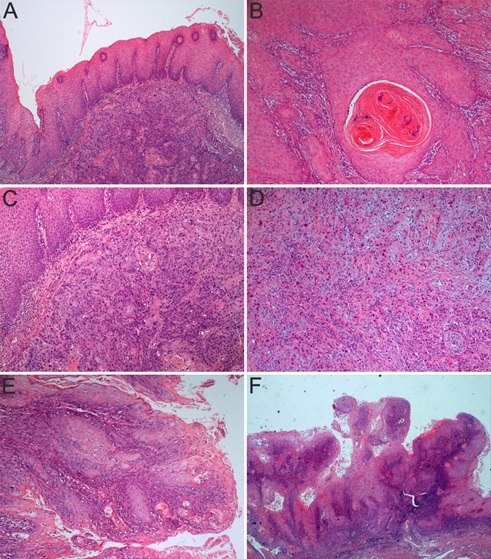
Palate Squamous Cell Carcinoma (SCC). A. Palate mucosa with SCC composed of invasive islands and cords of malignant squamous epithelial cells. HE staining, x50; B. Well-differentiated SCC, neoplastic proliferations with squamous epithelial architecture and dyskeratosis with keratin pearls formation. HE staining, x100; C. Moderate- differentiated SCC, neoplastic proliferation with obvious malignancy appearances and few keratin pearls formation. HE staining, x100; D. Poor-differentiated SCC, the neoplastic proliferations do no longer resembles squamous epithelium, keratinization is minimal and nuclear atypia are evident. HE staining, x100; E. Papillary SCC, a papillary tumor growth composed of numerous, thin, finger-like papillary projections. HE staining, x100; F. Verrucous SCC, neoplastic proliferation with abundant keratosis and parakeratosis in an acanthotic squamous epithelium accomplishing the “church spires” appearance. HE staining, x25
In well differentiated forms, tumor proliferations accomplish almost completely the cytoarchitecture of normal squamous epithelium, but with obvious characteristics of malignancy, such as: rupture of the basement membrane and invasion at least in the underlying chorion, loss of cellular polarity, disorganized growth, dyskeratosis (more than 50% of tumor cells) with keratin pearls formation (Figure 1B), minimal atypical cells (with increased nuclear-to-cytoplasmic ratio, large nuclei, irregular contour and uneven staining, distinct eosinophilic nucleoli and typical and atypical mitotic figures).
In moderately differentiated lesions the characteristics of squamous epithelium are less obvious to the detriment of those of malignancy, dyskeratosis being present in 20-50% of tumor cells, the nuclear pleomorphism is increased as well as the number of mitosis (Figure 1C).
In poorly differentiated lesions the resemblance to the squamous epithelium is ambiguous, predominating immature cells, with numerous typical and atypical mitoses, minimal keratinization and necrosis (Figure 1D).
Papillary SCC variant was diagnosed in two cases, both with origin in hard palate.
The tumors developed in men aged 61 and respectively 71 years from urban areas.
In the older patient the lesion was diagnosed in clinical stage II while in the other patient the clinical stage was I.
At the time of diagnosis, no lymph node metastases were detected, nor underlying bone invasion and perineural invasion.
Clinically these lesions presented as exophytic mass of 1.5cm and respectively of 2.4cm in in the largest diameter.
Both patients are still alive with more than 5 years from the date of surgery.
Histologically appear as a papillary tumor growth composed of numerous, thin, finger-like papillary projections (Figure 1E).
These projections are made up of delicate fibrovascular core lined by the neoplastic epithelium that showed dyskeratosis, surface keratinization, loss of polarity, increased nuclear cytoplasmic (N/C) ratio, nuclear enlargement and irregularities, prominent nucleoli and numerous mitotic figures. Stromal invasion was present and also an associated rich chronic inflammatory response.
Verrucous SCC variant was observed in only one case, respectively in a 76-year-old man from an urban area, that was diagnosed with clinical stage II.
The patient presented with a cauliflower mass developed at the level of the hard palate of about 3.1cm in diameter.
No lymph node metastases were detected, nor underlying bone invasion and perineural invasion.
The patient is still alive with more than 5 years from the date of surgery.
Histologically the lesions present abundant keratosis and parakeratosis in an acanthotic squamous epithelium accomplishing the “church spires” appearance (Figure 1F).
The neoplastic epithelium is well differentiated and the advancing tumor margins were broad with pushing appearance.
Near the front of tumor invasion was present an abundant inflammatory infiltrate.
Basaloid SCC variant was diagnosed in two cases, one developed in a 69-year-old man and the other in a 50-year-old man. Both originate from soft palate and at the time of diagnosis, clinically they were in stage 4 of the disease.
The older patient had bone invasion and lymph node dissemination. Clinically they present as firm, nodular mass with 4.7cm and respectively 6.3cm in diameter, both with invasion of adjacent muscles. Both patients did not survive more than 12 months from the date of surgery. Histologically we noticed invasive solid, trabecular, cords, and nests tumor proliferations that sometimes present obvious peripheral palisading (Figure 2A).
Figure 2.
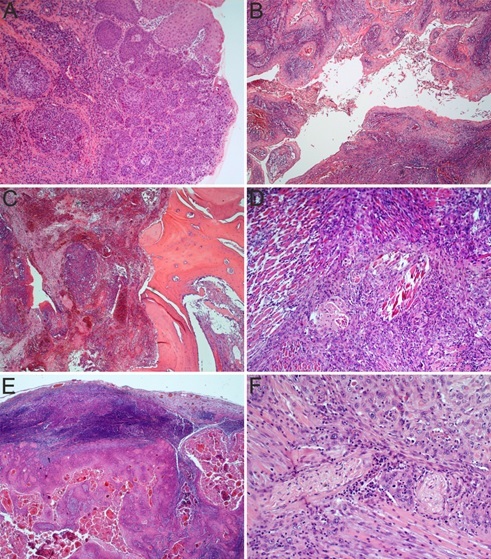
Palate Squamous Cell Carcinoma (SCC). A. Basaloid SCC, neoplastic proliferation consisting of invasive solid, trabecular, cords, and nests tumor proliferations that sometimes present obvious peripheral palisading. HE staining, x100; B. Acantholytic SCC, with islands and sheets of neoplastic squamous cell carcinoma with central prominent acantholysis that create a false appearance of glandular differentiation. HE staining, x50; C. SCC invading the palate bone. HE staining, x50; D. Moderate- differentiated SCC invasive in muscle fibers. HE staining, x100; E. Lymph node with SCC metastasis. HE staining, x25; F. SCC invading nerve bundles. HE staining, x200
These neoplastic proliferations were composed by small packed cells with hyperchromatic nuclei without nucleoli, and scant cytoplasm. Also we noticed that the basaloid proliferations were in direct continuity with areas of squamous differentiation including abrupt keratinization in the form of squamous pearls. Small foci of squamous differentiation were also present inside the basaloid proliferations. In one cases we noticed the surface ulceration and none showed perineural invasion.
Acantholytic SCC variant was diagnosed in two men aged 38 and respectively 46 years, both developed from the soft palate. Clinically appeared as a soft, exophytic mass, one of 3.7cm and respective 5.3cm in diameter. The youngest patient at the time of diagnosis was classified as stage IV of the disease while the other case was enclosing as stage III disease. Both cases showed clinically detectable lymph node metastases, adjacent muscle invasion and a survival rate of less than 12 months after surgery. Histologically were observed islands and sheets of neoplastic squamous cell carcinoma with central prominent acantholysis that create a false appearance of glandular differentiation (Figure 2B).
Usually, these pseudolumina contain cellular debris, acantholytic and dyskeratotic cells, with bizarre shapes and even as large multinucleated cells.
Patients with hard palate lesions had a peak incidence in the six and eight decades with an average age of 61.16 years. Most of them were man (76%) mainly from urban areas (56%). At the time of diagnosis these patients complained of palatal ulcerating lesion (17 cases), initial painless. The second common presentation was as a hard palate mass (vegetative type-5 cases) and the last one was as an ulcerovegetative lesions (3 cases). In more than half of the cases the patients also complained of pain (13 cases), especially those in the advanced stages. As clinical stage 64% cases from the hard palate lesions were presented in advanced stages (II and III), respectively 8 cases in both stage II and stage III. Most of the lesions developed in the hard palate (76%) they were larger than 2cm at the time of diagnosis, with 44% of lesions being classified as T2 lesions. Also in 14 cases there was bone invasion (Figure 2C), and in 10 cases perineural invasion. At the first presentation the neck lymph node enlargement was obvious in 7 cases that were histopathologically confirmed.
The soft palate lesions prevailed in the 6th decade of life (33.33%), patients with such tumors had an average age of 59.04 years. More than 90% were man and two-thirds of them came from urban areas. At the first presentation to the doctor, two thirds of the patients complained of soft palate mass and in the rest of the cases they present ulcerovegetative lesions (7 cases). Also in 71.42% cases patients complained of pain. Most of these patients presented in advanced stages (90.47%), respectively 8 cases in stage III and 11 cases in stage IV. As T stage most patients (71.5%) had bulky lesions larger than 4 cm and presence of muscle invasion was noticed in about 81% of these cases (Figure 2D).
In 15 cases a neck lymph node enlargement was clinical observed that were histopathologically confirmed (Figure 2E) and the perineural invasion was noticed in more than half lesions (52.38%) (Figure 2F).
Regardless of tumor topography in terms of the survival period after surgery we noticed that most of the patients (68.88%) did not survive more than 12 months. In less than 18% of cases the survival was longer than 5 years.
Related to tumor topography we noticed a better survival among patients with tumors developed in the hard palate compared to the soft palate, the percentage of survivors over 5 years after the operation was of 28% for hard palate tumors versus 4.76% in soft palate tumors.
The number of patients who died in the first 12 months after surgery it was almost double for soft palate tumors compared to that developed in the hard palate (48% compared with 90.47%).
Clinical evaluation of the lymph node adenopathy revealed a higher predisposition for the soft palate (14 positive cases out of 21 cases) versus the hard palate (8 positive cases out of 24 patients), λ2(1, N=45)=4.9802, p=0.0256 (Figure 3A).
Figure 3.
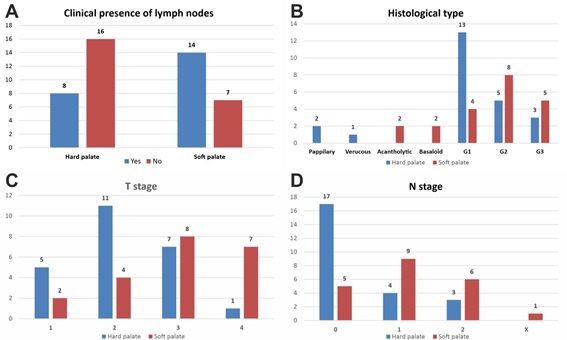
Statistical difference ratios for Palate Squamous Cell Carcinoma (SCC) tumors of the soft palate and hard palate in what it regards adenopathy (A), histopathological type (B), T and N stages (C and D). All values on the OY axe represent the actual number of cases
When considering the histopathological subtypes, there was no clear-cut predominance of a specific subtype for any of the two localizations, λ2 (6, N=45)=5.598, p=0.0608 (Figure 3B).
Regarding the tumor stage, there was a preponderance of more aggressive tumors at the level of the soft palate compared to the hard palate, λ2 (3, N=45)=8.958, p=0.02984 (Figure 3C).
Ulceration of the tumors was mostly observed for hard palate tumors, λ2 (1, N=45)=24.716, p<0.0001. Regarding the lymph ganglia histopathological invasion, there was again a preponderance of more aggressive tumors at the level of the soft palate compared to the hard palate, λ2 (3, N=45)=9.1902, p=0.02686 (Figure 3D).
Regarding survival times, there were no statistically significant differences for the two localizations, λ2 (3, N=45)=7.3785, p=0.117188.
Discussions
Globally, carcinomas with oral topography was ranked as the 18th to 26th most common human cancer [7]. In 2018, about 354,864 cases of oral cavity carcinoma were newly diagnosed, representing 2% of all cancer sites and among them 228,389 deaths were registered, which represented 1.9% of the total deaths by all cancer sites [1].
Wide geographical variations in the incidence of oral cancer have been reported with the Southern Asia and Pacific Islands as regions with the worldwide highest incidence and mortality rates among both genders. According to the U.S. Cancer Statistics data reported during 2007-2016 regarding cancers of oral cavity and pharynx the incidence rates varied by anatomic site, sex, race/ethnicity, and age group [2].
Thus an increase of incidence rates was recorded for cancers of the oral cavity and pharynx combined, base of tongue, anterior tongue, gum, tonsil, oropharynx, and other oral cavity and pharynx.
At the same time, it was noticed a decline in the incidence rates for cancers of the lip, floor of mouth, soft palate and uvula (-3.7%), hard palate (-0.9%), hypopharynx, and nasopharynx, and were stable for cancers of the cheek and other mouth and salivary gland. Related to gender differences in female was found a decrease of incidences for cancers of the floor of mouth, soft palate and uvula, nasopharynx, and hypopharynx, and an increasing for cancers of the anterior tongue, gum, and tonsil while among males was noticed a stable incidence for hard palate, cheek and other mouth, and salivary gland, but an increased frequency for cancers of the base of tongue, oropharynx, and other oral cavity and pharynx and a decreased incidence for cancer of the lip. By age group among sites with increasing rate trends, the increases were mainly driven by increases among persons aged 50-79 years, while among persons aged 20-49 years those rates were stable or decreased [2].
Unlike U.S. in China, at Beijing, a 10-year continuous single-center study conducted between 2004 and 2013, showed a decrease in incidence of oral SCCs at the sites of tongue, gingiva, buccal mucosa, oral floor, oropharynx, lip and palate [7].
Also, a series of studies proved that in regions of China, South East Asia, and India the most frequent oral SCC developed at the buccal mucosa site [8,9,10].
This appears to be the consequence of the endemic regional practice of chewing betel quid.
It has been estimated that about 90% of oral cancers are SCCs [11].
The rest of the tumors could develop not only from the oral epithelium but also from connective tissue, minor salivary glands, lymphoid tissue, and melanocytes. Thus minor salivary gland carcinomas may account for less than 5% of oral cancers, with about 60% developing in the hard palate, and as the most common tumors are quoted: mucoepidermoid carcinoma (54%), low-grade adenocarcinoma (17%), and adenoid cystic carcinoma (15%) [12].
Other types of cancers of the oral cavity include melanomas, maxillary bones malignancies and odontogenic tumors [13,14].
In the Department of Oral and Maxillofacial Surgery from Emergency Clinical County Hospital Craiova, from January 2010 to December 2019 were treated 45 patients with palate SCC. Most of them were men, respectively 84.44% of cases with a peak incidence occurring during the six and seventh decade, and with the average age of 60.17 years. 62.22% of cases came from urban areas, and 53.33% developed in hard palate.
It has been estimated that about 5% of all oral cavity cancers are hard palate cancers [15], and about two-thirds up to 90% of these cancers are SCCs [16,17].
Other hard palate malignancies include: adenoid cystic carcinoma, mucoepidermoid carcinomas, and adenocarcinomas [15].
About 80% of the soft palate cancers are SCCs and these could account for about 5-12% of oropharyngeal cancers [18], being rarer than tonsil or tongue base SCCs [19,20].
The remainder of soft palate malignancies are more common of minor salivary gland origin [20,21,22].
Between 1973 and 2015 in the Surveillance, Epidemiology, and End Results (SEER) database were recorded 1,489 hard palate SCCs [23] and between 1973 and 2014 the same database noticed 4,366 cases of soft palate SCCs [4].
According to the gender and age group the hard palate SCCs was estimated to develop at 60-70 year-old patients with a slight female predominance [23,24,25], while the soft palate SCCs are more frequently present in 6th to 7th decade, among males, who are twice as likely to be affected as women [26,27,28,29].
As the common risk factors for palate SCCs were cited tobacco use and alcohol consumption, the carcinogenic effects being dose-dependent and with a synergistic effect [30].
As major hard palate SCC risk factors, developed among patients from India and Philippines, were quoted poor oral hygiene, mechanical irritation, poorly fitting dentures, and syphilis [17].
The influences of human papilloma virus (HPV) infections in the pathogenesis of the soft palate SCC it is a very small one since the HPV-positivity rate among these patients was as low as 3.1 [31].
Histopathologically, in our casuistry predominate the conventional SCC with 84.44% cases, from which the most encountered were well-differentiated tumors (44.73%) followed by moderate differentiated forms (34.21%) and poor differentiated tumors (21.06%). In 15.55% cases the histopathologic investigation classified them as peculiar histological subtypes of SCC, represented by 2 cases of basaloid SCC, 2 cases of papillary SCC, 2 cases of acantholytic SCC and one cases of verrucous SCC.
Topographically, in hard palate developed more frequently the well differentiated conventional SCC (52%), and the rarest were the poor differentiated form (12%) and the peculiar SCC variant (12%), represented by 2 cases of papillary SCC and one cases of verrucous SCC. In soft palate lesions were dominated by moderate conventional SCC (38.1%), and at the opposite pole they were well differentiated tumors (19.05%) and peculiar SCC variant (19.05%), represented by 2 cases of basaloid SCC, and 2 cases of papillary SCC.
The current WHO classification of tumors of the head and neck identify the conventional oral SCCs and several peculiar histological subtypes, namely basaloid SCC, spindle cell SCC, adenosquamous carcinoma, carcinoma cuniculatum, verrucous SCC, lymphoepithelial carcinoma, papillary SCC and acantholytic SCC [32].
Conventional SSC is the most frequent malignancies arising from the oral epithelium and according to the degree of keratinization, cellular and nuclear pleomorphism and mitotic activity is divided in well, moderately and poor differentiated forms. Most of the oral SCC are moderately and well differentiated forms while the poor differentiated SCC are less frequent. However, it seems that this malignancies grading of oral SCCs correlates poorly with outcomes and treatment response [33].
On the other hand, in the last 10 years were describe several histological variants of SCCs that have peculiar histomorphological appearance with certain impact on the prognosis, metastatic potential, survival rate and treatment of such patients [34,35].
Although rare, these histological variants may account for 10-15% of all SCCs [36] and need an accurate diagnosis because many of these can be easily misdiagnosed with a real impact on the prognosis and treatment of such patients.
In our casuistry the hard palate SCCs prevail in the six and eight decades with an average age of 61.16 years. Most patients were man (76%) and at the time of diagnosis they especially complained about painless ulcerating lesions (70.83%). About 64% cases presented in advanced stages (II and III), with 76% of lesions being larger than 2cm at the time of diagnosis. In more than half of the cases (58.33%) it was recorded bone invasion and about 30% had clinical detectable neck lymph node enlargement at the first presentation. The peak incidence for soft palate lesions was recorded in 6th decade of life (33.33%), the average age of these patients being of 59.04 years. More than 90% were man and they mostly complain to have soft palate mass (66.66%) at first presentation. The great majority (90.47%) presented in advanced stages (clinical stage III and IV) with 71.42 of them having clinical detectable neck lymph node enlargement at the first presentation. About 71.5% had bulky lesions, larger than 4cm, and with muscle invasion noticed in about 81% of these cases, and perineural invasion in more than half lesions (52.38%).
More common the hard palate SCC debuts as an ulcerating asymptomatic lesion that in more advance stage become painful [20].
Other reported presenting symptoms include palatal mass, bleeding, ill-fitting dentures, or loose teeth. In up to 70% of cases hard palatal tumors extend to the soft palate, gingiva, maxillary sinus, sphenopalatine foramen, pterygopalatine fossa, and skull base [20].
Usually, the soft palate SCC present as an asymptomatic leukoplakia or erythroplakia [37] but which over time will develop by extension hypernasal speech, dysphagia, trismus, referred otalgia, hemoptysis, or neck mass in adults [20].
About 50% of soft palate SCC at the first presentation to the clinician are already extended beyond soft palate, at the level of: the tonsil, posterior tonsillar pillar, oropharyngeal wall, retromolar trigone, inferior alveolar process, and base of the tongue [19].
Lymph node metastases are rare for patients with hard palate SCCs [38], but these are present in up to 50% of soft palate SCCs at the first presentation [39].
For T1-T2 soft palate SCCs the nodal metastases rate at presentation is about 20%, and this percentage may increase up to 60-70% for T3 and T4 lesions [40].
Also up to 25% of soft palate SCCs may present as tumors with synchronous or metachronous cancers especially on the floor of the mouth [19,40].
Oral papillary SCC is a rare variant of SCC with favorable prognosis [41].
It develops in the 6-7 decades of life, slightly affecting men [42,43,44].
The most common tumor topography is larynx [45], the oral cavity being rarely affected with gums as the most common site, followed by buccal mucosa, tongue, palate, lower lip, floor of mouth and oropharynx [46].
The palate papillary SCC are rare, until now in English literature being reported about 15 cases, more common in hard palate [42,43,44,45,46,47].
Clinically, they present as soft, exophytic growth mass with size ranging from 2mm up to 4cm [43].
It was described two variants of oral papillary SCC: a broad based exophytic form and a papillary form [48].
The main differential diagnosis is with squamous papilloma in which lack the high grade epithelial atypia. Other differential diagnoses that can come into question are verrucous carcinoma (with obvious hyperkeratosis, broad pushing retepegs showing no evidence of invasion) and exophytic CSS (with high grade atypia and a rather cauliflower-appearance than celery stalk-looking). Generally oral papillary SCC has an invasive behavior, Argyris et al. reported tumor invasion in 91% of studied cases [42].
However, the vascular, perineural, or osseous invasion are usually absent [48].
Overall this variant of oral SCC has better prognosis than the conventional SCC of similar clinical stage but worse than verrucous carcinoma [47,48,49].
Oral verrucous carcinoma seems to be the most common variant of SCC in oral cavity, peculiar involving the buccal mucosa, alveolar ridge, and oral tongue. The palate is rarer involved, the English literature reporting less than 100 cases [50,51,52,53].
Alonso JE et al. reviewing the oral verrucous carcinoma cases recorded between 1973 to 2012 in the SEER database found 79 cases with hard palate location, representing 5.3% from all oral sites origin of this tumor [50].
Usually occurs in elderly patients with an average age at diagnosis of 69.5 years [50].
Its etiology is controversial, many authors incriminating as the main cause chronic irritation from extensive tobacco use, alcohol consumption or chewing betel nuts [54,55].
Clinically, oral verrucous carcinoma presents as a cauliflower-like exophytic growth with whitish-to-gray surface that sometime may have erythematous areas [35,55].
First oral verrucous carcinoma must be differentiated from verrucous hyperplasia which does not extend more deeply than adjacent uninvolved epithelium. Also the verruca vulgaris may be considered, but in these lesions are seen peculiar aspects as a prominent keratohyalin granules and parakeratosis with sharply defined acanthotic rete ridges. As mentioned above another differential diagnosis can be made with papillary SCC. In terms of biological behavior this tumor must be considered as a non-metastasizing variant of well differentiated SCC. Although in the vast majority of cases the lesions have a superficial involvement, in the advanced stages can be recorded bone, salivary glands, muscles and cartilage invasion. The metastatic potential of these lesions is low, with very few cases described in the literature [50,56,57].
Oral verrucous carcinoma it is a recurrent lesion, some authors recoding a 38.5% local recurrence rate [35].
Basaloid SCC is a rare high grade variant of SCC that has the ability for metastasis to distant sites. It develops in elderly man with a history of heavy smoking and alcohol consumption [58].
Basaloid SCC shows predilection head and neck region, particularly in larynx and hypopharynx [59,60].
Regarding oral localization, this SCC variant seems to have predilection for tongue, followed by floor of the mouth, retromolar trigone, and gingival mucosa [35,58,61].
Palate basaloid SCC are very rare in the English literature being reported a few dozen cases [58,59,61,62,63].
Clinically, usually the tumor appears as a firm, exophytic, poorly defined mass, with surface ulceration [35,64].
Furthermore, some cases could present as multifocal lesions that comprise other mucosal sites of the head and neck, as well as the gastrointestinal tract and lower respiratory tract or even as metastatic even at the initial presentation [61].
In the differential diagnoses, especially in the palate sites could be included adenoid cystic carcinoma (with origin in salivary gland has a peculiar growth pattern-the cribriform one, no squamous component and perineural invasion) or adenosquamous carcinoma (with prominent squamous component, absence of basaloid cells with peripheral nuclear palisading, and presence of glandular differentiation). Most patient present with higher stage and with nodal metastases, and their prognosis is worse than those with conventional SCC, having a very high mortality [59,60,65,66].
This aggressive behavior is due to early local or regional recurrences and distant metastasis. The literature date reported a neck lymph node metastasis rate of 64% and a distant metastasis rate of 44%, especially in lung, liver, bone, brain and skin [60,66,67,68].
With such behavior it was estimated that patients with basaloid SCC have a survival rate at least half of those with the conventional version [68].
Acantholytic SCC (adenoid SCC) is a rare variant of SCC characterized by marked acantholysis in the tumor islands leading to a pseudoglandular appearance, but no evidence of glandular differentiation, secretory activity or products [69].
Usually it develops in the sun exposed skin of the head and neck region [70] and less frequently in the oral cavity, in the literature having been reported less than 50 cases [71,72].
The palate origin is even rarer with less than 10 reported cases [73].
The patients age with oral acantholytic SCC varies widely from the 4th to the 8th decade, with 51.9 years mean age [69].
A slight prevalence of cases has been reported in men. Oral acantholytic SCC should be differentiated from adenosquamous carcinoma (which contain a true adenocarcinoma component positive to mucin stains and to glandular immunomarkers), intraoral primary or metastatic adenocarcinoma (in these cases the molecular marker related to adenocarcinoma are very useful) and oral angiosarcomas (with which it shares some clinical features and a similar histopathological pattern and even more overlaps of cytokeratin-expression and of expression of vascular differential markers) [72].
Of a real use in differentiation of oral acantholytic SCC from the last entity proved to be the expression of Fli-1 in angiosarcoma and cytoplasmatic immunoreaction for γ2-chain of ln-5 in the acantholytic tumors [71].
The intraoral cases are more aggressive than those of the skin and lips [74], the former having a certain tendency to aggressive invasion and metastasis to the lymph nodes [71,75].
With such behavior, oral acantholytic SCC have a worse prognosis than conventional SCC [73].
Overall, in our casuistry about 69% of patients did not survive more than 12 months after surgery. As percentage the number of patients who died in the first 12 months after surgery it was almost double for soft palate tumors compared to that developed in the hard palate (48% compared with 90.47%).
A longer survival period was recorded especially among patients with SCCs developed in the hard palate compared to the soft palate, the percentage of survivors over 5 years after surgery was of 28% for hard palate tumors versus 4.76% in soft palate tumors.
The literature data show for the overall 5-year survival rates of hard palate SCCs values varying between 33 to 86% depending on tumor presentation and treatment modality [23,76].
Thus patients treated with surgery have a higher 5-year disease-specific survival than those who were treated with radiation therapy alone (71% versus 29%) [23].
Also it was showed that more advanced T stage is associated with worse overall 5-year survival rates for hard palate SCCs [23,25,77].
Moreover, it was stated that for hard palate SCCs in any T stage but without clinically evident neck lymph node metastases should be done elective neck management with radiation, neck dissection, or sentinel lymph node biopsy due to the risk of metastases and potential for improved survival [14].
For the soft palate SCC the overall 5-year survival rates range between 21-37.2% in stage IV-A [18,78] to 40-66% in stage I-II [18,78,79].
Also it was showed a higher survival rate up to 70.8% for the unilateral soft palate tumors compared to those that cross the midline or with bilateral lesions, in which the survival rate falls below 51% due to a higher incidence of regional metastasis [40].
Higher T stage [80], presence of residual tumor cells in neck lymph nodes after radiation therapy [81], and recurrence in the parapharyngeal space [82] were associated with lower survival rates.
Conclusions
Our ten years’ single institute experience proved that hard palate tumors differ in many aspects from those of the soft palate.
Patients with hard palate SCCs had an average age slightly larger compared to those who developed soft palate tumors.
Also, those with hard palate tumors are mostly diagnosed in less advanced stages compared to those at the level of the soft palate, and implicitly the former had a longer survival time.
Soft palate tumors showed more frequent muscle and perineural invasion, and lymph node dissemination, while hard palate tumors were rather invasive in adjacent bone.
Histopathologically the most encountered hard palate SCC were the conventional well-differentiated tumor, and from the peculiar SCC variant the papillary and verrucous forms while for the soft palate SCC prevailed the moderate and poor differentiated conventional SCC and from the peculiar SCC variant the basaloid and acantholytic forms.
Thus it becomes important to establish from the beginning the exact location of the tumor since it have impact on the prognosis, treatment and survival outcome of such patients.
Conflict of interests
None to declare.
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Ellington TD, Henley SJ, Senkomago V, O'Neil ME, Wilson RJ, Singh S, Thomas CC, Wu M, Richardson LC. Trends in Incidence of Cancers of the Oral Cavity and Pharynx - United States 2007-2016. MMWR Morb Mortal Wkly Rep. 2020;69(15):433–438. doi: 10.15585/mmwr.mm6915a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Abu Rass N, Surougi E, Baheydarah S, Baroom A, ALGhamdi H, AlTuwayjiri H, AlMansour N. Neoplasms of the Palate: A Review. The Egyptian Journal of Hospital Medicine. 2018;70(8):1393–1400. [Google Scholar]
- 4.Chan CK, Han AY, Alonso JE, Xu MJ, Mallen-St Clair J, Heaton CM, Ryan WR, Kuan EC, St John MA. Squamous Cell Carcinoma of the Soft Palate in the United States: A Population-Based Study. Otolaryngol Head Neck Surg. 2018;159(4):662–668. doi: 10.1177/0194599818771931. [DOI] [PubMed] [Google Scholar]
- 5.Zuydam AC, Lowe D, Brown JS, Vaughan ED, Rogers SN. Predictors of speech and swallowing function following primary surgery for oral and oropharyngeal cancer. Clin Otolaryngol. 2005;30(5):428–437. doi: 10.1111/j.1365-2273.2005.01061.x. [DOI] [PubMed] [Google Scholar]
- 6.Johnson N, Franceschi S, Ferlay J, Ramadas K, Schmid S, MacDonald DG, Bouquot JE, Slootweg PJ. In: World Health Organization classification of tumours: pathology and genetics of head and neck tumours. Barnes L, Eveson JW, Reichart P, Sidransky D, editors. Lyon: IARC; 2005. Squamous cell carcinoma; pp. 168–175. [Google Scholar]
- 7.Bai XX, Zhang J, Wei L. Analysis of primary oral and oropharyngeal squamous cell carcinoma in inhabitants of Beijing, China-a 10-year continuous single-center study. BMC Oral Health. 2020;20(1):208–208. doi: 10.1186/s12903-020-01192-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bobdey S, Sathwara J, Jain A, Saoba S, Balasubramaniam G. Squamous cell carcinoma of buccal mucosa: An analysis of prognostic factors. South Asian J Cancer. 2018;7(1):49–54. doi: 10.4103/sajc.sajc_317_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huang CH, Chu ST, Ger LP, Hou YY, Sun CP. Clinicopathologic evaluation of prognostic factors for squamous cell carcinoma of the buccal mucosa. J Chin Med Assoc. 2007;70(4):164–170. doi: 10.1016/S1726-4901(09)70351-X. [DOI] [PubMed] [Google Scholar]
- 10.Sahu PK, Kumar S. Epidemiological Aspects of Oral Cancer in North Indian Population. Indian J Otolaryngol Head Neck Surg. 2019;71(Suppl 1):944–948. doi: 10.1007/s12070-019-01629-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Warnakulasuriya S. Global epidemiology of oral and oropharyngeal cancer. Oral Oncol. 2009;45(4-5):309–316. doi: 10.1016/j.oraloncology.2008.06.002. [DOI] [PubMed] [Google Scholar]
- 12.Gross JH, Mazul AL, Zevallos JP. In: Oral Cancer: Evaluation Therapy and Rehabilitation. Fakhry C, Pitman KT, Kiess AP, Eisele DW, editors. New York: Thieme; 2020. Epidemiology of Oral Cavity Cancer; pp. 2–13. [Google Scholar]
- 13.Montero PH, Patel SG. Cancer of the oral cavity. Surg Oncol Clin N Am. 2015;24(3):491–508. doi: 10.1016/j.soc.2015.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tharakan T, Mehta V. In: Oral Cancer: Evaluation Therapy and Rehabilitation. Fakhry C, Pitman KT, Kiess AP, Eisele DW, editors. New York: Thieme; 2020. Squamous Cell Cancer of the Palate; pp. 192–199. [Google Scholar]
- 15.Truitt TO, Gleich LL, Huntress GP, Gluckman JL. Surgical management of hard palate malignancies. Otolaryngol Head Neck Surg. 1999;121(5):548–552. doi: 10.1016/S0194-5998(99)70084-7. [DOI] [PubMed] [Google Scholar]
- 16.Petruzzelli GJ, Myers EN. Malignant neoplasms of the hard palate and upper alveolar ridge. Oncology (Williston Park) 1994;8(4):43–48. [PubMed] [Google Scholar]
- 17.Vosler PS, Myers EN. In: Operative Otolaryngology. Myers EN, Snyderman CH, editors. Philadelphia: Elsevier; 2018. Transoral inferior maxillectomy; pp. 227–236. [Google Scholar]
- 18.Iyer NG, Nixon IJ, Palmer F, Kim L, Whitcher M, Katabi N, Ghossein R, Shah JP, Patel SG, Ganly I. Surgical management of squamous cell carcinoma of the soft palate: factors predictive of outcome. Head Neck. 2012;34(8):1071–1080. doi: 10.1002/hed.21878. [DOI] [PubMed] [Google Scholar]
- 19.Leemans CR, Engelbrecht WJ, Tiwari R, Devillé WL, Karim AB, van der Waal I, Snow GB. Carcinoma of the soft palate and anterior tonsillar pillar. Laryngoscope. 1994;104(12):1477–1481. doi: 10.1288/00005537-199412000-00009. [DOI] [PubMed] [Google Scholar]
- 20.Sadeghi N, Panje WR. In: Surgical Oncology: An Algorithmic Approach. Saclarides TJ, Millikan K, Godellas C, editors. New York: Springer-Verlag; 2003. Cancer of the soft palate; pp. 32–39. [Google Scholar]
- 21.Osborne RF, Brown JJ. Carcinoma of the oral pharynx: an analysis of subsite treatment heterogeneity. Surg Oncol Clin N Am. 2004;13(1):71–80. doi: 10.1016/S1055-3207(03)00117-0. [DOI] [PubMed] [Google Scholar]
- 22.Zini A, Czerninski R, Sgan-Cohen HD. Oral cancer over four decades: epidemiology, trends, histology, and survival by anatomical sites. J Oral Pathol Med. 2010;39(4):299–305. doi: 10.1111/j.1600-0714.2009.00845.x. [DOI] [PubMed] [Google Scholar]
- 23.Alonso JE, Han AY, Kuan EC, Strohl M, Clair JM, St John MA, Ryan WR, Heaton CM. The survival impact of surgical therapy in squamous cell carcinoma of the hard palate. Laryngoscope. 2018;128(9):2050–2055. doi: 10.1002/lary.27080. [DOI] [PubMed] [Google Scholar]
- 24.Binahmed A, Nason RW, Hussain A, Abdoh AA, Sándor GK. Treatment outcomes in squamous cell carcinoma of the maxillary alveolus and palate: a population-based study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;105(6):750–754. doi: 10.1016/j.tripleo.2007.09.001. [DOI] [PubMed] [Google Scholar]
- 25.Morris LG, Patel SG, Shah JP, Ganly I. High rates of regional failure in squamous cell carcinoma of the hard palate and maxillary alveolus. Head Neck. 2011;33(6):824–830. doi: 10.1002/hed.21547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chung CK, Constable WC. Squamous cell carcinoma of the soft palate and uvula. Int J Radiat Oncol Biol Phys. 1979;5(6):845–850. doi: 10.1016/0360-3016(79)90069-5. [DOI] [PubMed] [Google Scholar]
- 27.Evans JF, Shah JP. Epidermoid carcinoma of the palate. Am J Surg. 1981;142(4):451–455. doi: 10.1016/0002-9610(81)90373-1. [DOI] [PubMed] [Google Scholar]
- 28.Fee WE, Schoeppel SL, Rubenstein R, Goffinet DR, Goode RL, Boles R, Tuschman M. Squamous cell carcinoma of the soft palate. Arch Otolaryngol. 1979;105(12):710–718. doi: 10.1001/archotol.1979.00790240024006. [DOI] [PubMed] [Google Scholar]
- 29.Russ JE, Applebaum EL, Sisson GA. Squamous cell carcinoma of the soft palate. Laryngoscope. 1977;87(7):1151–1156. doi: 10.1288/00005537-197707000-00017. [DOI] [PubMed] [Google Scholar]
- 30.Forastiere A, Koch W, Trotti A, Sidransky D. Head and neck cancer. N Engl J Med. 2001;345(26):1890–1900. doi: 10.1056/NEJMra001375. [DOI] [PubMed] [Google Scholar]
- 31.Gelwan E, Malm IJ, Khararjian A, Fakhry C, Bishop JA, Westra WH. Nonuniform Distribution of High-risk Human Papillomavirus in Squamous Cell Carcinomas of the Oropharynx: Rethinking the Anatomic Boundaries of Oral and Oropharyngeal Carcinoma From an Oncologic HPV Perspective. Am J Surg Pathol. 2017;41(12):1722–1728. doi: 10.1097/PAS.0000000000000929. [DOI] [PubMed] [Google Scholar]
- 32.Sloan P, Gale N, Hunter K, Lingen M, Nylander K, Reibel J, Salo T, Zain RB. In: WHO classification of head and neck tumours. EI-Naggar AK, Chan JKC, Grandis JR, Takata T, Slootweg PJ, editors. Lyon: International Agency for Research on Cancer; 2017. Malignant surface epithelial tumours; pp. 109–111. [Google Scholar]
- 33.Barnes L, Eveson JW, Reichart P, Sidransky D. In: World Health Organization classification of tumours. Pathology and genetics of head and neck tumours. Barnes L, Everson JW, Reichart P, Sidransky D, editors. Lyon: International Agency for Research on Cancer Press; 2005. Oral cavity and oropharynx; pp. 164–208. [Google Scholar]
- 34.Pathak J, Swain N, Patel S, Poonja L. Histopathological variants of oral squamous cell carcinoma-institutional case reports. J Oral Maxillofac Pathol. 2014;18(1):143–145. doi: 10.4103/0973-029X.131945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pereira MC, Oliveira DT, Landman G, Kowalski LP. Histologic subtypes of oral squamous cell carcinoma: prognostic relevance. J Can Dent Assoc. 2007;73(4):339–344. [PubMed] [Google Scholar]
- 36.Thompson LD. Squamous cell carcinoma variants of the head and neck. Curr Diagn Pathol. 2003;9:384–396. [Google Scholar]
- 37.Johnson NW, Jayasekara P, Amarasinghe AA. Squamous cell carcinoma and precursor lesions of the oral cavity: epidemiology and aetiology. Periodontol 2000. 2011;57(1):19–37. doi: 10.1111/j.1600-0757.2011.00401.x. [DOI] [PubMed] [Google Scholar]
- 38.Adelstein D, Gillison ML, Pfister DG, Spencer S, Adkins D, Brizel DM, Burtness B, Busse PM, Caudell JJ, Cmelak AJ, Colevas AD, Eisele DW, Fenton M, Foote RL, Gilbert J, Haddad RI, Hicks WL, Hitchcock YJ, Jimeno A, Leizman D, Lydiatt WM, Maghami E, Mell LK, Mittal BB, Pinto HA, Ridge JA, Rocco J, Rodriguez CP, Shah JP, Weber RS, Witek M, Worden F, Yom SS, Zhen W, Burns JL, Darlow SD. NCCN Guidelines Insights: Head and Neck Cancers Version 2.2017. J Natl Compr Canc Netw. 2017;15(6):761–770. doi: 10.6004/jnccn.2017.0101. [DOI] [PubMed] [Google Scholar]
- 39.Tshering Vogel DW, Zbaeren P, Thoeny HC. Cancer of the oral cavity and oropharynx. Cancer Imaging. 2010;10(1):62–72. doi: 10.1102/1470-7330.2010.0008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cohan DM, Popat S, Kaplan SE, Rigual N, Loree T, Hicks WL. Oropharyngeal cancer: current understanding and management. Curr Opin Otolaryngol Head Neck Surg. 2009;17(2):88–94. doi: 10.1097/moo.0b013e32832984c0. [DOI] [PubMed] [Google Scholar]
- 41.Alotaiby F, Song F, Boyce BJ, Cao D, Zhao Y, Lai J. Unusual Papillary Squamous Cell Carcinoma of the Tip of Tongue Presenting in a Patient Status Post Heart Transplant. Anticancer Res. 2018;38(7):4203–4206. doi: 10.21873/anticanres.12715. [DOI] [PubMed] [Google Scholar]
- 42.Argyris PP, Kademani D, Pambuccian SE, Nguyen R, Tosios KI, Koutlas IG. Comparison between p16 INK4A immunohistochemistry and human papillomavirus polymerase chain reaction assay in oral papillary squamous cell carcinoma. J Oral Maxillofac Surg. 2013;71(10):1676–1682. doi: 10.1016/j.joms.2013.04.004. [DOI] [PubMed] [Google Scholar]
- 43.Ding Y, Ma L, Shi L, Feng J, Liu W, Zhou Z. Papillary squamous cell carcinoma of the oral mucosa: a clinicopathologic and immunohistochemical study of 12 cases and literature review. Ann Diagn Pathol. 2013;17(1):18–21. doi: 10.1016/j.anndiagpath.2012.04.005. [DOI] [PubMed] [Google Scholar]
- 44.Ferrer MJ, Estellés E, Villanueva A, López R. Papillary squamous cell carcinoma of the oropharynx. Eur Arch Otorhinolaryngol. 2003;260(8):444–445. doi: 10.1007/s00405-003-0602-2. [DOI] [PubMed] [Google Scholar]
- 45.Russell JO, Hoschar AP, Scharpf J. Papillary squamous cell carcinoma of the head and neck: a clinicopathologic series. Am J Otolaryngol. 2011;32(6):557–563. doi: 10.1016/j.amjoto.2010.09.010. [DOI] [PubMed] [Google Scholar]
- 46.Bao Z, Yang X, Shi L, Feng J, Liu W, Zhou Z. Clinicopathologic features of oral squamous papilloma and papillary squamous cell carcinoma: a study of 197 patients from eastern China. Ann Diagn Pathol. 2012;16(6):454–458. doi: 10.1016/j.anndiagpath.2012.04.001. [DOI] [PubMed] [Google Scholar]
- 47.Karthik KP, Balamurugan R, Pushpa TS. Oral papillary squamous cell carcinoma (OPSCC): a rare presentation in the palate. J Oral Med Oral Surg. 2020;26(2):1–3. [Google Scholar]
- 48.Fitzpatrick SG, Neuman AN, Cohen DM, Bhattacharyya I. Papillary variant of squamous cell carcinoma arising on the gingiva: 61 cases reported from within a larger series of gingival squamous cell carcinoma. Head Neck Pathol. 2013;7(4):320–326. doi: 10.1007/s12105-013-0444-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Patankar SR, Poornima V, Avdhani V, Bhatia S. Papillary squamous cell carcinoma of antrum-clear cell variant. J Oral Maxillofac Pathol. 2014;18(1):146–148. doi: 10.4103/0973-029X.131946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Alonso JE, Kuan EC, Arshi A, St John MA. A population-based analysis of verrucous carcinoma of the oral cavity. Laryngoscope. 2018;128(2):393–397. doi: 10.1002/lary.26745. [DOI] [PubMed] [Google Scholar]
- 51.McDonald JS, Crissman JD, Gluckman JL. Verrucous carcinoma of the oral cavity. Head Neck Surg. 1982;5(1):22–28. doi: 10.1002/hed.2890050106. [DOI] [PubMed] [Google Scholar]
- 52.Medina JE, Dichtel W, Luna MA. Verrucous-squamous carcinomas of the oral cavity. A clinicopathologic study of 104 cases. Arch Otolaryngol. 1984;110(7):437–440. doi: 10.1001/archotol.1984.00800330019003. [DOI] [PubMed] [Google Scholar]
- 53.Rajendran R, Varghese I, Sugathan CK, Vijayakumar T. Ackerman's tumour (verrucous carcinoma) of the oral cavity: a clinico-epidemiologic study of 426 cases. Aust Dent J. 1988;33(4):295–298. doi: 10.1111/j.1834-7819.1988.tb04180.x. [DOI] [PubMed] [Google Scholar]
- 54.Koch BB, Trask DK, Hoffman HT, Karnell LH, Robinson RA, Zhen W, Menck HR, Commission on Cancer, American College of Surgeons. American Cancer Society National survey of head and neck verrucous carcinoma: patterns of presentation, care, and outcome. Cancer. 2001;92(1):110–120. doi: 10.1002/1097-0142(20010701)92:1<110::aid-cncr1298>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- 55.Rekha KP, Angadi PV. Verrucous carcinoma of the oral cavity: a clinico-pathologic appraisal of 133 cases in Indians. Oral Maxillofac Surg. 2010;14(4):211–218. doi: 10.1007/s10006-010-0222-0. [DOI] [PubMed] [Google Scholar]
- 56.Asproudis I, Gorezis S, Aspiotis M, Tsanou E, Kitsiou E, Merminga E, Peschos D, Charalabopoulos K, Agnantis NJ. Orbital metastasis from verrucous carcinoma of the oral cavity: case report and review of the literature. In Vivo. 2007;21(5):909–912. [PubMed] [Google Scholar]
- 57.Schrader M, Laberke HG, Jahnke K. Lymphknotenmetastasen beim verrukösen Karzinom (Ackerman-Tumor) [Lymphatic metastases of verrucous carcinoma (Ackerman tumor)] HNO. 1987;35(1):27–30. [PubMed] [Google Scholar]
- 58.Jayasooriya PR, Tilakaratne WM, Mendis BR, Lombardi T. A literature review on oral basaloid squamous cell carcinomas, with special emphasis on etiology. Ann Diagn Pathol. 2013;17(6):547–551. doi: 10.1016/j.anndiagpath.2013.09.001. [DOI] [PubMed] [Google Scholar]
- 59.Gupta B, Bhattacharyya A, Singh A, Sah K, Gupta V. Basaloid squamous cell carcinoma-A rare and aggressive variant of squamous cell carcinoma: A case report and review of literature. Natl J Maxillofac Surg. 2018;9(1):64–68. doi: 10.4103/njms.NJMS_14_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Soriano E, Faure C, Lantuejoul S, Reyt E, Bolla M, Brambilla E, Righini CA. Course and prognosis of basaloid squamous cell carcinoma of the head and neck: a case-control study of 62 patients. Eur J Cancer. 2008;44(2):244–250. doi: 10.1016/j.ejca.2007.11.008. [DOI] [PubMed] [Google Scholar]
- 61.Karatzanis AD, Fragkiadakis GM, Prokopakis EP, Koutsopoulos AV, Helidonis ES, Velegrakis GA. Basaloid squamous cell carcinoma of the soft palate: case report. Auris Nasus Larynx. 2008;35(4):592–596. doi: 10.1016/j.anl.2007.11.010. [DOI] [PubMed] [Google Scholar]
- 62.Hellquist HB, Dahl F, Karlsson MG, Nilsson C. Basaloid squamous cell carcinoma of the palate. Histopathology. 1994;25(2):178–180. doi: 10.1111/j.1365-2559.1994.tb01576.x. [DOI] [PubMed] [Google Scholar]
- 63.Lovejoy HM, Matthews BL. Basaloid-squamous carcinoma of the palate. Otolaryngol Head Neck Surg. 1992;106(2):159–162. doi: 10.1177/019459989210600206. [DOI] [PubMed] [Google Scholar]
- 64.Wain SL, Kier R, Vollmer RT, Bossen EH. Basaloid-squamous carcinoma of the tongue, hypopharynx, and larynx: report of 10 cases. Hum Pathol. 1986;17(11):1158–1166. doi: 10.1016/s0046-8177(86)80422-1. [DOI] [PubMed] [Google Scholar]
- 65.Banks ER, Frierson HF, Mills SE, George E, Zarbo RJ, Swanson PE. Basaloid squamous cell carcinoma of the head and neck. A clinicopathologic and immunohistochemical study of 40 cases. Am J Surg Pathol. 1992;16(10):939–946. doi: 10.1097/00000478-199210000-00003. [DOI] [PubMed] [Google Scholar]
- 66.Raslan WF, Barnes L, Krause JR, Contis L, Killeen R, Kapadia SB. Basaloid squamous cell carcinoma of the head and neck: a clinicopathologic and flow cytometric study of 10 new cases with review of the English literature. Am J Otolaryngol. 1994;15(3):204–211. doi: 10.1016/0196-0709(94)90006-x. [DOI] [PubMed] [Google Scholar]
- 67.Chaidas K, Koltsidopoulos P, Kalodimos G, Skoulakis C. Basaloid squamous cell carcinoma of the tonsil. Hippokratia. 2012;16(1):74–75. [PMC free article] [PubMed] [Google Scholar]
- 68.Winzenburg SM, Niehans GA, George E, Daly K, Adams GL. Basaloid squamous carcinoma: a clinical comparison of two histologic types with poorly differentiated squamous cell carcinoma. Otolaryngol Head Neck Surg. 1998;119(5):471–475. doi: 10.1016/S0194-5998(98)70104-4. [DOI] [PubMed] [Google Scholar]
- 69.Nayak SD, Jose M, Sequeira J. Oral adenoid/acantholytic squamous cell carcinoma: a report of two cases with review of literature. Kathmandu Univ Med J (KUMJ) 2012;10(39):83–87. doi: 10.3126/kumj.v10i3.8028. [DOI] [PubMed] [Google Scholar]
- 70.Kerawala CJ. Acantholytic squamous cell carcinoma of the oral cavity: a more aggressive entity. Br J Oral Maxillofac Surg. 2009;47(2):123–125. doi: 10.1016/j.bjoms.2008.04.018. [DOI] [PubMed] [Google Scholar]
- 71.Driemel O, Müller-Richter UD, Hakim SG, Bauer R, Berndt A, Kleinheinz J, Reichert TE, Kosmehl H. Oral acantholytic squamous cell carcinoma shares clinical and histological features with angiosarcoma. Head Face Med. 2008;4:17–17. doi: 10.1186/1746-160X-4-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Mardi K, Singh N. Acantholytic squamous cell carcinoma of the oral cavity: A rare entity. J Oral Maxillofac Pathol. 2014;18(Suppl 1):S128–130. doi: 10.4103/0973-029X.141359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lin JS, Lin HP, Liu CJ. Acantholytic squamous cell carcinoma of soft palate-case report and literature review. J Dent Sci. 2016;11(2):207–209. doi: 10.1016/j.jds.2015.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Garcia C, Crowson AN. Acantholytic squamous cell carcinoma: is it really a more-aggressive tumor. Dermatol Surg. 2011;37(3):353–356. doi: 10.1111/j.1524-4725.2011.01886.x. [DOI] [PubMed] [Google Scholar]
- 75.Papadopoulou E, Tosios KI, Nikitakis N, Papadogeorgakis N, Sklavounou-Andrikopoulou A. Acantholytic squamous cell carcinoma of the gingiva: report of a case and review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;109(6):e67–71. doi: 10.1016/j.tripleo.2010.01.019. [DOI] [PubMed] [Google Scholar]
- 76.Givi B, Eskander A, Awad MI, Kong Q, Montero PH, Palmer FL, Xu W, De Almeida JR, Lee N, O'Sullivan B, Irish JC, Gilbert R, Ganly I, Patel SG, Goldstein DP, Morris LG. Impact of elective neck dissection on the outcome of oral squamous cell carcinomas arising in the maxillary alveolus and hard palate. Head Neck. 2016;38 Suppl 1(Suppl 1):E1688–1694. doi: 10.1002/hed.24302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Os AD, Karakullukcu B, Leemans CR, Halmos GB, Roodenburg JL, Weert SV, Karagozoglu KH, Witjes MJ. Management of the clinically N0 neck in squamous cell carcinoma of the maxillary alveolus and hard palate. Head Neck. 2016;38(12):1794–1798. doi: 10.1002/hed.24511. [DOI] [PubMed] [Google Scholar]
- 78.Erkal HS, Serin M, Amdur RJ, Villaret DB, Stringer SP, Mendenhall WM. Squamous cell carcinomas of the soft palate treated with radiation therapy alone or followed by planned neck dissection. Int J Radiat Oncol Biol Phys. 2001;50(2):359–366. doi: 10.1016/s0360-3016(00)01578-9. [DOI] [PubMed] [Google Scholar]
- 79.Chera BS, Amdur RJ, Hinerman RW, Morris CG, Villaret DB, Werning JW, Mendenhall WM. Definitive radiation therapy for squamous cell carcinoma of the soft palate. Head Neck. 2008;30(8):1114–1119. doi: 10.1002/hed.20848. [DOI] [PubMed] [Google Scholar]
- 80.Nixon IJ, Palmer FL, Lakin P, Kattan MM, Lee NY, Ganly I. Pathologically determined tumor volume vs pathologic T stage in the prediction of outcome after surgical treatment of oropharyngeal squamous cell carcinoma. JAMA Otolaryngol Head Neck Surg. 2013;139(11):1151–1155. doi: 10.1001/jamaoto.2013.4973. [DOI] [PubMed] [Google Scholar]
- 81.Simon C, Goepfert H, Rosenthal DI, Roberts D, El-Naggar A, Old M, Diaz EM, Myers JN. Presence of malignant tumor cells in persistent neck disease after radiotherapy for advanced squamous cell carcinoma of the oropharynx is associated with poor survival. Eur Arch Otorhinolaryngol. 2006;263(4):313–318. doi: 10.1007/s00405-005-1016-0. [DOI] [PubMed] [Google Scholar]
- 82.Douglas WG, Rigual NR, Giese W, Bauer J, Wiseman SM, Loree TR, Schwarz J, Alrawi S, Hicks WL. Advanced soft palate cancer: the clinical importance of the parapharyngeal space. Otolaryngol Head Neck Surg. 2005;133(1):66–69. doi: 10.1016/j.otohns.2005.03.007. [DOI] [PubMed] [Google Scholar]


