Abstract
Calciphylaxis is a rare and potentially fatal disease that affects the subcutaneous layer of the skin. It is a calcific vasculopathy induced by a systemic process that causes occlusion of small blood vessels. The mortality rate for individuals diagnosed with calciphylaxis is estimated between 52% and 81% with sepsis being the leading cause of death. Uraemic calciphylaxis and its known effective treatments are well documented in the literature. Unfortunately, there is no known effective treatment for non‐uraemic calciphylaxis. Most of the current treatments for non‐uraemic calciphylaxis are derived from uraemic calciphylaxis treatment protocols. We report a case of a 75‐year‐old female with calciphylaxis on the right lower extremity who was successfully treated with four pamidronate infusions in addition to local wound care. This case represents a non‐uraemic calciphylaxis wound successfully treated with pamidronate infusions and standard wound care, and suggests that IV pamidronate can be an effective treatment option.
Keywords: calcific arteriolopathy, leg ulcer, non‐healing wound, sodium thiosulphate, vascular calcifications
1. INTRODUCTION
Calciphylaxis is a rare and fatal disease that affects the subcutaneous layer of the skin. In calciphylaxis, vascular calcification and thrombosis of small/medium blood vessels lead to skin and subcutaneous tissue necrosis. It is believed to be induced by a systemic process; however, the exact pathophysiology remains unclear. The hallmarks of calciphylaxis that differentiates it from other vascular calcifications is the systematic calcification of the arterial tunica media layer with potential fibrosis and necrosis of surrounding adipose tissue. People who are diagnosed with calciphylaxis are reported to have 1‐year mortality rates from 52% to as high as 81%.1, 2, 3, 4, 5 The leading cause of death from calciphylaxis is sepsis. Although calciphylaxis is most common among Caucasian females in their fifth decade of life, it has also been reported among the younger population including children.5, 6, 7
Calciphylaxis is characterised by calcification, microthrombosis, and fibrointimal hyperplasia of small dermal and subcutaneous arteries and arterioles that lead to ischaemia and septal panniculitis.8 The disease may progress to vascular calcification in skeletal muscles, the brain, lungs, intestines, eyes, and mesentery. It can eventually lead to ischaemia, necrosis, and sepsis. Although the exact pathogenesis is unclear, Au and Crawford hypothesised that calcification is an early and essential process in calciphylaxis plaque development and that the vascular calcification leads to vascular endothelial dysfunction and injury.9, 10
Initially, calciphylaxis was thought to only be associated with end‐stage renal disease (ESRD). However, since it was first described by Dr. Selye in 1961, calciphylaxis is now known to occur in patients without ESRD.11 Calciphylaxis associated with ESRD and chronic kidney disease (CKD) is known as calcific uraemic arteriolopathy (CUA), and calciphylaxis of non‐uraemic aetiology is known as non‐uraemic calciphylaxis (NUC).9 CUA is more prevalent in ESRD patients than CKD patients. Its incidence can be as high as 4.1% in haemodialysis‐dependent patients.12
The diagnosis of calciphylaxis can be difficult to make as it can be easily overlooked and mistaken for other wound aetiologies. The diagnosis is made based on a combination of clinical, biochemical, and histopathological findings. Clinically, the wounds tend to present with intense pain associated with cutaneous lesions with indurated nodules and/or ulcerations with a central necrotic eschar. These findings alone are only suggestive of calciphylaxis because they are very common amongst wounds in general; thus, making calciphylaxis a difficult disease to diagnose. However, the presence of livedo reticularis and severe pain increases the probability of calciphylaxis.1 When combined with biochemical laboratory findings, it can be determined whether the calciphylaxis is uraemic or non‐uraemic in nature. Histopathologically, via skin (punch) biopsy, a pathologist can help confirm the diagnosis of calciphylaxis if the presence of calcifications of small and/or medium blood vessels, ischaemia, and necrosis of subcutaneous fats can be identified.13 Thus, a skin biopsy is required for definitive diagnosis of calciphylaxis. Radiographic findings are not beneficial, and therefore, imaging is not recommended as part of the workup for this pathology.14, 15, 16
Overall, calciphylaxis wounds have poor healing and are often complicated by blistering and superimposed infection, which often lead to hospital admission for intravenous antibiotics. Ulcerations are most commonly covered with black eschar when patients first present to the clinic. Majority of the time, the ulcerations are found on the legs (60%), abdomen (23%), and buttocks (9%).4
Nigwaker et al found that the most common cause of NUC is primary hyperthyroidism (27.8%), followed by malignancy (22.2%), alcoholic liver disease (16.7%), connective diseases (11.1%), and diabetes (5.5%).1, 4 Unfortunately, there is no known effective treatment for NUC. Most of the current treatments are derived from CUA treatment protocols. In this article, we present a case of NUC and a novel treatment approach.
2. CASE REPORT
A 75‐year‐old female with a past medical history significant for diabetes, hypertension, hyperlipidaemia, and atrial fibrillation (on Warfarin). While the patient was being treated for pyoderma gangrenosum on her left lower extremity (LLE), which healed uneventfully, she started developing painful, discoloured lesions with no open wounds on her right lower extremity (Figure 1).
Figure 1.
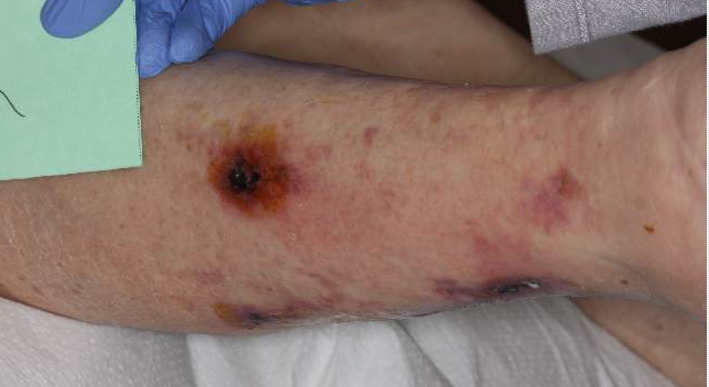
Initial presentation of right lower extremity wounds—Oedematous with multiple discoloured crusted lesions with violaceus borders on the posterior, medial, and lateral aspect of the right distal leg
The initial examination of her RLE showed an oedematous leg with no open wound and discoloured crusted lesions on the posterior, medial, and lateral aspects of the right distal leg. The patient was initially treated with a compression wrap; however, after 1 month of treatment, the lesions had worsened. It became painful and eschar developed over the crusted lesions (Figure 2). Patient rated her pain of the right lower extremity at 10/10. Considering the recent history involving her LLE, a preliminary diagnosis of pyoderma gangrenosum was considered, and the patient was started on a course of oral steroid treatment (weekly tapers starting at 60 mg). Unfortunately, there was no noticeable improvement after one course of oral steroid and the lesions had transformed into extremely tender reticulate purpura with black eschar and violaceous borders (Figure 3). A 4‐mm skin‐punch biopsy was performed along with lab work [complete blood count (CBC), complete metabolic panel (CMP), and parathyroid hormone (PTH)]. The wounds were dressed with Xeroform and Coban 2 Lite compression wrap. Patient's labs returned within normal limits.
Figure 2.
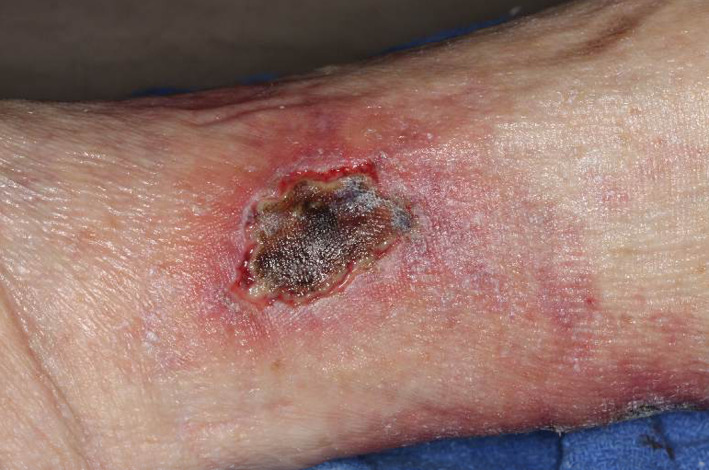
One month after initial presentation. The wound had worsened with an eschar developed over the crusted lesion along increased pain
Figure 3.
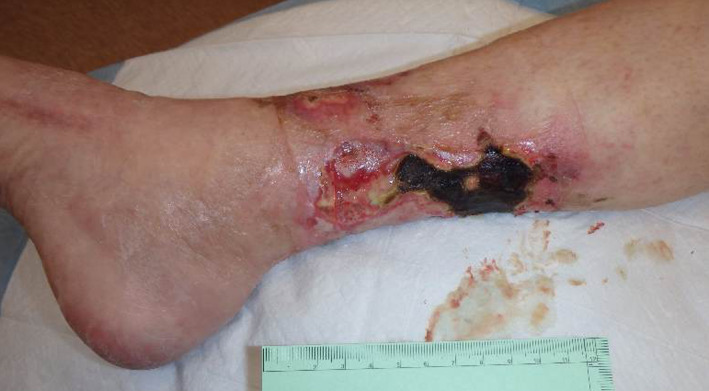
Right lower extremity wounds 3 months after initial presentation. The discoloured lesions had transformed into extremely tender reticulate purpura with black eschar and violaceous borders
At this point, a multidisciplinary approach was taken and pain management was consulted. The patient was started on oxycodone‐acetaminophen 5/325 and 50 mg Fentanyl patch, which was subsequently reduced to 25 mg due to nausea. The patient was switched from warfarin to rivaroxaban because of the possibility that these ulcers were due to warfarin‐induced skin necrosis. Vascular workup did not show any right lower extremity venous insufficiency. The LLE had very minimal reflux at the superficial femoral junction and in the popliteal vein. The patient's arterial studies did not show any occlusive disease.
Patient's right leg wounds were persistently necrotic with no improvement after discontinuing warfarin. Further lab work was ordered to evaluate for rheumatological and haematological disease that could be contributing to the lack of healing. Rheumatology workup was unyielding and the patient was found to have no autoimmune disease. Haematological workup showed an IgG Kappa monoclonal gammopathy of unknown significance (MGUS). Skin biopsy report was consistent with a diagnosis of calciphylaxis (Figure 4).
Figure 4.
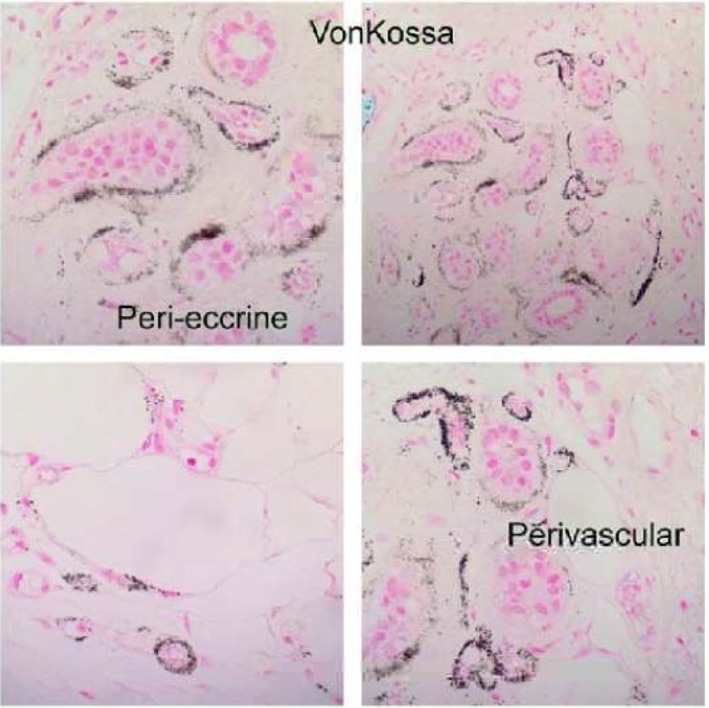
Histology of right lower extremity wound. The specimen shows small vessels in the interlobular septa and at the periphery of a lobule that show some calcific deposits in the vessel walls. One of the superficial dermal‐dilated small vessels contains a small amount of fibrin thrombus. A von Kossa stain for calcium was carried out and fine granular calcific deposits were found in the vessels in the dermal subcutaneous layer and in smaller capillary size vessels in the superficial subcutaneous tissue
Given the working diagnosis of NUC, the patient received pamidronate disodium 30 mg infusions every 2 weeks along with local wound care, which consisted of weekly compression wrap. With each infusion, the patient's right leg wounds improved and eventually resolved after 6 months of treatment. The patient received a total of four pamidronate infusions (Figures 5, 6, 7). At the last visit, patient's wounds had completely closed with complete re‐epithelisation (Figure 8). There was no tenderness, no periwound erythema, and minimal oedema. She was discharged with a knee‐high compression stocking and has not experienced a recurrence for over 1.5 years.
Figure 5.
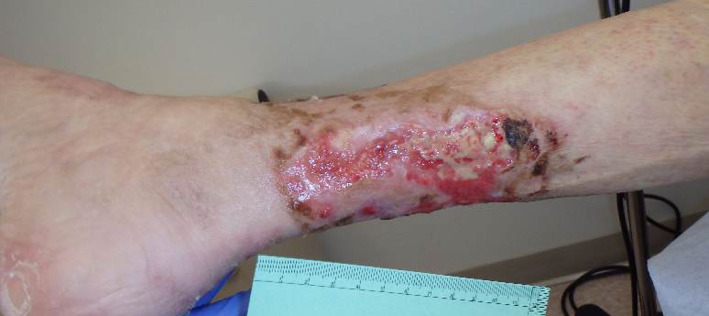
Four months since treatment started. Two weeks status post the first pamidronate infusion. The eschars had almost completely resolved with improvement in ulcer size and wound base appearance
Figure 6.
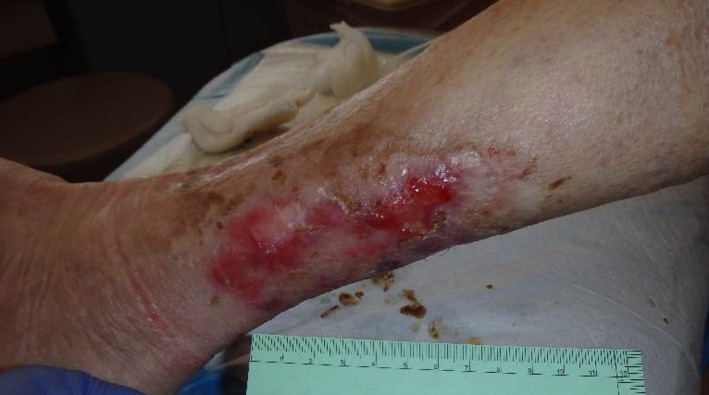
After third infusion of pamidronate. The eschars and fibrotic wounds base had completely resolved. Epithelisation was noted with improvement in wound size and pain
Figure 7.
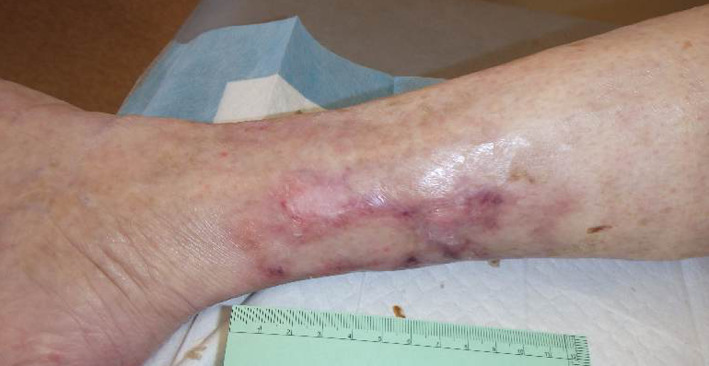
After fourth pamidronate infusion. Wounds had completely closed
Figure 8.
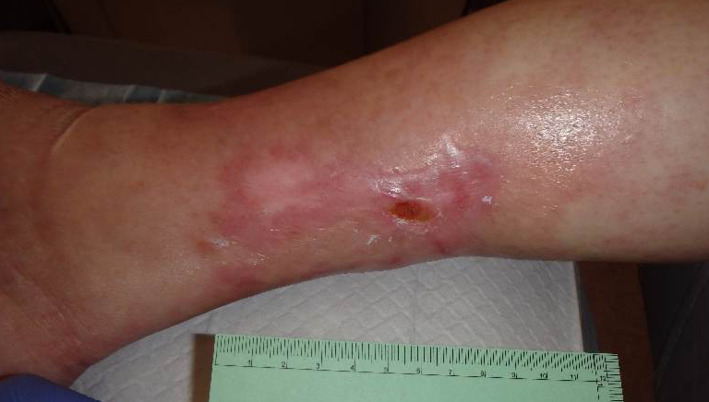
One month since the last pamidronate infusion. The wound remained closed
3. DISCUSSION
NUC wounds represent a rare pathology and can be difficult to treat. Given the rarity of the disease, the literature is limited. Despite the rarity of the disease, NUC can be a fatal disease and must be treated as such. A thorough workup is recommended to determine the aetiology of the disease (Figure 9). In suspected cases of NUC, the authors recommend treating the underlying condition and considering bisphosphonate infusion in addition to local wound care. Based on the article by Nigwekar et al, malignancy is the second most common causes of NUC after hyperparathyroidism.4 Thus, it is not only recommended, but necessary to rule out for the possibility of malignancy.
Figure 9.
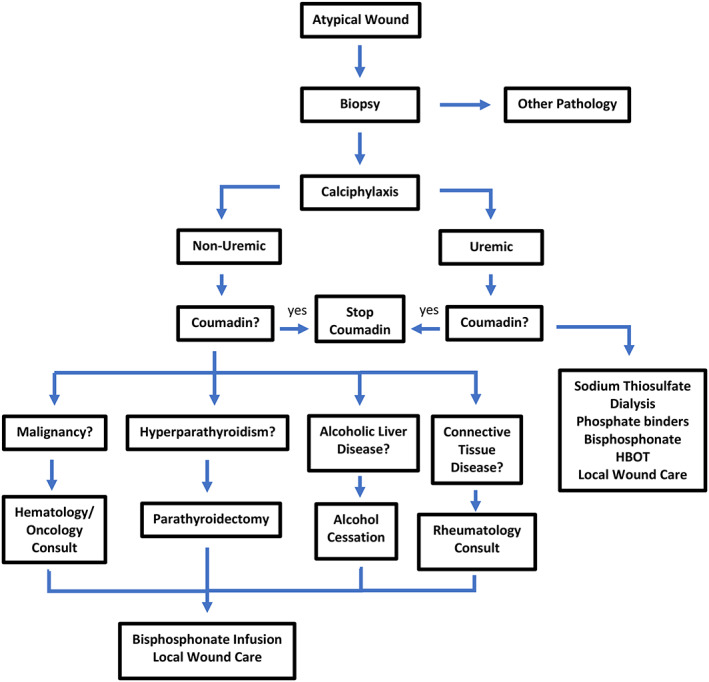
Suggested initial treatment guideline for non‐uraemic calciphylaxis (NUC). Once etiology is determined, treatment of the underlying condition is recommended
There have been case reports of resolution of NUC with sodium thiosulphate infusion. This case represents NUC wounds that were successfully treated with pamidronate infusions. To our knowledge, only two other case series reported the successful treatment of NUC with pamidronate, which were performed in Europe.17, 18 The first reported case was in Germany by Schliep et al; he was able to treat calciphylaxis of unknown origin with 6 pamidronate infusion and a repeated skin biopsy showed that the arterial calcification had vanished.17 The second reported case was in France by Lorriaux et al.18 In that case series, both patients were initially treated with sodium thiosulphate, but that was discontinued because of side effects. They were then started on pamidronate disodium 30 mg infusions every 2 weeks, and a marked improvement was noted after eight infusions.18
Most of the current treatment modalities for NUC are modelled from CUA treatment. Hammadah et al reported a case of successful resolution of NUC in a patient with acral gangrene with treatment using sodium thiosulphate.19 Vicco et al2 reported a case of NUC associated with congestive heart failure secondary to Chagas disease. Vicco found that as the cardiac function improved, so did the NUC wounds. Thus, suggesting a possible connection between poor wound healing and cardiac function.2 However, Vicco also mentioned that because of the lack of evidence indicating association between cardiac function and wound healing, the coincidence of improving NUC wounds with improved cardiac function could simply be due to the resolution of Chagas disease itself. The parasite Trypanosoma cruzi is known to cause several immunological disturbances and could result in the development of NUC.20
Differentiating calciphylactic wounds from other aetiologies can be challenging without a skin biopsy, especially in patients on warfarin. Both warfarin‐induced skin necrosis and calciphylaxis wounds can have similar clinical presentations. A biopsy can help distinguish between warfarin‐induced skin necrosis and calciphylaxis.21
Pamidronate disodium is a nitrogen‐containing bisphosphonate drug. It is commonly used to treat hypercalcaemia of malignancy. Its mechanism of action consists of inhibition of bone resorption and a decrease in mineralisation by disrupting osteoclast activity. Pamidronate is thought to be beneficial in NUC because arterial calcification may be due to increased calcium levels in the blood. Pamidronate upregulates the calcium receptors in the body and can help regulate PTH levels in the blood. These effects can directly regulate the serum calcium levels.22, 23, 24, 25
Calciphylaxis affects multiple organ systems; thus, it is a disease that requires a multidisciplinary approach. Patients often initially present to a wound care physician for evaluation of a wound. It is the responsibility of the wound care physician to involve other appropriate specialties for appropriate management of the patient. Other specialties that are commonly involved in the workup and treatment of NUC include dermatology, pain management, vascular surgery, general surgery, haematology, and rheumatology (Table 1).
Table 1.
Recommended specialists to be involved in patient care
| Specialty | Roles |
|---|---|
| Primary care provider | Management of overall health |
| Switch patient from warfarin to another anticoagulation (if possible) | |
| Management of infusion (sodium thiosulphate/pamidronate) | |
| Management of the PTH level | |
| Podiatrist/wound care physician | Wound care |
| Surgical management of wound | |
| Antibiotic recommendation | |
| Hyperbaric oxygen therapy | |
| Pain management | Management of pain |
| Dermatopathologist | Biopsy review |
| Histopathology analysis | |
| General surgery | Parathyroidectomy (if indicated) |
| Haematology/oncology | Evaluation for possible underlying malignancy |
| Switch patient from warfarin to another anticoagulation (if possible) | |
| Nephrologist | Not recommended as nephrologists do not treat NUC |
| Radiologist | Not recommended |
| Radiographic study has little benefit in diagnosing NUC |
NUC, non‐uraemic calciphylaxis; PTH, parathyroid hormone.
Like CUA wounds, prophylactic antibiotics for NUC wounds are not indicated nor recommended unless there are signs of active infection. Adjunctive hyperbaric oxygen therapy (HBOT) may be beneficial as there are several case reports showing positive results. Arenas et al successfully healed two patients with CUA with the use of HBOT in addition to medical management.26 Podymow et al also found similar results in five patients with CUA.27 However, its benefits in NUC is still undetermined because there is limited research in this field.
NUC is a complicated condition with high mortality and morbidity that can clinically mimic venous leg ulcers. This can create a significant delay in diagnosis and subsequent treatment. It is important to remember that malignancy is the second most common cause of NUC, and a delay in diagnosis can put the patient at a higher mortality risk. The authors understand that proposing a treatment algorithm with such limited information is unconventional (Figure 9). However, based on our literature review, we believe that the treatment algorithm we proposed will help clinician better manage this rare disease. To the author's best knowledge, to date, there has been no agreed upon standard treatment for NUC. This is the first case of successful treatment of NUC with only pamidronate infusion in the United States. Furthermore, our case report suggests and agrees with both Schliep and Lorriaux that NUC can successfully be treated with a single agent (bisphosphonate); thus, minimising additional medications, which can increase compliance and decrease potential side effects and drug interactions. In the past, most NUC conditions have been treated with sodium thiosulphate infusion. Pamidronate offers a viable alternative treatment option for these patients.
CONFLICTS OF INTEREST
The authors have no conflicts of interest to report.
Disclosure
None reported.
Truong DH, Riedhammer MM, Zinszer K. Non‐uraemic calciphylaxis successfully treated with pamidronate infusion. Int Wound J. 2019;16:250–255. 10.1111/iwj.13019
REFERENCES
- 1. Ning MS, Dahir KM, Castellanos EH, McGirt LY. Sodium thiosulfate in the treatment of non‐uremic calciphylaxis. J Dermatol. 2013;40(8):649‐652. [DOI] [PubMed] [Google Scholar]
- 2. Vicco MH, Ferini F, Rodeles L, Baretta M, Marcipar I, Bottasso OA. Non‐uremic calciphylaxis and chagas disease. Rev Clin Esp (Barc). 2014;214(5):55. [DOI] [PubMed] [Google Scholar]
- 3. Nigwekar SU. Multidisciplinary approach to calcific uremic arteriolopathy. Curr Opin Nephrol Hypertens. 2015;24(6):531‐537. [DOI] [PubMed] [Google Scholar]
- 4. Nigwekar SU, Wolf M, Sterns RH, Hix JK. Calciphylaxis from nonuremic causes: a systematic review. Clin J Am Soc Nephrol. 2008;3(4):1139‐1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Nigwekar SU, Kroshinsky D, Nazarian RM, et al. Calciphylaxis: risk factors, diagnosis, and treatment. Am J Kidney Dis. 2015;66(1):133‐146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Araya CE, Fennell RS, Neiberger RE, Dharnidharka VR. Sodium thiosulfate treatment for calcific uremic arteriolopathy in children and young adults. Clin J Am Soc Nephrol. 2006;1(6):1161‐1166. [DOI] [PubMed] [Google Scholar]
- 7. Feng J, Gohara M, Lazova R, Antaya RJ. Fatal childhood calciphylaxis in a 10‐year‐old and literature review. Pediatr Dermatol. 2006;23(3):266‐272. [DOI] [PubMed] [Google Scholar]
- 8. Dauden E, Onate MJ. Calciphylaxis. Dermatol Clin. 2008;26(4):68 ix. [DOI] [PubMed] [Google Scholar]
- 9. Au S, Crawford RI. Three‐dimensional analysis of a calciphylaxis plaque: clues to pathogenesis. J Am Acad Dermatol. 2002;47(1):53‐57. [DOI] [PubMed] [Google Scholar]
- 10. Hayden MR, Kolb LG, Khanna R. Calciphylaxis and the cardiometabolic syndrome. J Cardiometab Syndr. 2006;1(1):76‐79. [DOI] [PubMed] [Google Scholar]
- 11. Selye H, Gentile G, Prioreschi P. Cutaneous molt induced by calciphylaxis in the rat. Science. 1961;134(3493):1876‐1877. [DOI] [PubMed] [Google Scholar]
- 12. Maroz N, Mohandes S, Field H, Kabakov Z, Simman R. Calciphylaxis in patients with preserved kidney function. J Am Coll Clin Wound Spec. 2015;6(1–2):24‐28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Reiter N, El‐Shabrawi L, Leinweber B, Berghold A, Aberer E. Calcinosis cutis: part I. diagnostic pathway. J Am Acad Dermatol. 2011;65(1):4. [DOI] [PubMed] [Google Scholar]
- 14. Han MM, Pang J, Shinkai K, Franc B, Hawkins R, Aparici CM. Calciphylaxis and bone scintigraphy: case report with histological confirmation and review of the literature. Ann Nucl Med. 2007;21(4):235‐238. [DOI] [PubMed] [Google Scholar]
- 15. Norris B, Vaysman V, Line BR. Bone scintigraphy of calciphylaxis: a syndrome of vascular calcification and skin necrosis. Clin Nucl Med. 2005;30(11):725‐727. [DOI] [PubMed] [Google Scholar]
- 16. Shmidt E, Murthy NS, Knudsen JM, et al. Net‐like pattern of calcification on plain soft‐tissue radiographs in patients with calciphylaxis. J Am Acad Dermatol. 2012;67(6):1296‐1301. [DOI] [PubMed] [Google Scholar]
- 17. Schliep S, Schuler G, Kiesewetter F. Successful treatment of calciphylaxis with pamidronate. Eur J Dermatol. 2008;18:554‐556. [DOI] [PubMed] [Google Scholar]
- 18. Lorriaux A, Chaby G, Dhaille F, et al. Nonuraemic calciphylaxis: response to treatment with pamidronate and negative pressure therapy. Clin Exp Dermatol. 2015;40(1):52‐55. [DOI] [PubMed] [Google Scholar]
- 19. Hammadah M, Chaturvedi S, Jue J, et al. Acral gangrene as a presentation of non‐uremic calciphylaxis. Avicenna J Med. 2013;3(4):109‐111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Ouaissi A, Da Silva AC, Guevara AG, Borges M, Guilvard E. Trypanosoma cruzi‐induced host immune system dysfunction: a rationale for parasite immunosuppressive factor(s) encoding gene targeting. J Biomed Biotechnol. 2001;1(1):11‐17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Danziger J. Vitamin K‐dependent proteins, warfarin, and vascular calcification. Clin J Am Soc Nephrol. 2008;3(5):1504‐1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Raymond CB, Wazny LD. Sodium thiosulfate, bisphosphonates, and cinacalcet for treatment of calciphylaxis. Am J Health Syst Pharm. 2008;65(15):1419‐1429. [DOI] [PubMed] [Google Scholar]
- 23. Hanafusa T, Yamaguchi Y, Tani M, Umegaki N, Nishimura Y, Katayama I. Intractable wounds caused by calcific uremic arteriolopathy treated with bisphosphonates. J Am Acad Dermatol. 2007;57(6):1021‐1025. [DOI] [PubMed] [Google Scholar]
- 24. Price PA, Faus SA, Williamson MK. Bisphosphonates alendronate and ibandronate inhibit artery calcification at doses comparable to those that inhibit bone resorption. Arterioscler Thromb Vasc Biol. 2001;21(5):817‐824. [DOI] [PubMed] [Google Scholar]
- 25. Torregrosa JV, Duran CE, Barros X, et al. Successful treatment of calcific uraemic arteriolopathy with bisphosphonates. Nefrologia. 2012;32(3):329‐334. [DOI] [PubMed] [Google Scholar]
- 26. Arenas MD, Gil MT, Gutierrez MD, et al. Management of calcific uremic arteriolopathy (calciphylaxis) with a combination of treatments, including hyperbaric oxygen therapy. Clin Nephrol. 2008;70(3):261‐264. [DOI] [PubMed] [Google Scholar]
- 27. Podymow T, Wherrett C, Burns KD. Hyperbaric oxygen in the treatment of calciphylaxis: a case series. Nephrol Dial Transplant. 2001;16(11):2176‐2180. [DOI] [PubMed] [Google Scholar]


