Abstract
Pressure ulcers present a major clinical challenge, are physically debilitating and place the patient at risk of serious comorbidities such as septic shock. Recombinant human activated protein C (APC) is an anticoagulant with anti‐inflammatory, cytoprotective and angiogenic effects that promote rapid wound healing. Topical negative pressure wound therapy (TNP) has become widely used as a treatment modality in wounds although its efficacy has not been proven through randomised controlled trials. The aim of this study was to determine the preliminary efficacy and safety of treatment with APC for severe chronic pressure sores with and without TNP. This case presentation describes the history, management and outcome of two patients each with a severe chronic non‐healing pressure ulcer that had failed to respond to conventional therapy. TNP was added to conservative management of both ulcers with no improvement seen. Then local application of small doses of APC was added to TNP and with conservative management, resulted in significant clinical improvement and rapid healing of both ulcers, displaying rapid growth of vascular granulation tissue with subsequent epithelialisation. Patients tolerated the treatment well and improvements suggested by long‐term follow‐up were provided. Randomised placebo‐controlled double blind trials are needed to quantify the efficacy, safety, cost‐effectiveness, optimal dose and quality of life changes seen from treatment with APC.
Keywords: Activated protein C, Chronic, Pressure ulcer, Topical negative pressure, Wound
Introduction
Pressure ulcers (also called pressure sores, decubitus ulcers or bed sores) are commonly encountered in hospitals 1 and are associated with increased morbidity and mortality particularly in those with impaired sensation, prolonged immobility or advanced age 2. They are also the most common result of hospitalisation following spinal cord injury 3. Pressure damage occurs particularly over bony prominences but can affect any area of the skin. Damage varies from superficial lesions (grade I) to bony involvement with necrotic tissue (grade IV).
Acute management demands debridement and wound dressing with supervised bed rest and frequent re‐positioning to avoid physical trauma with surgery as a second line, but common option 4, 5. Bed rest results in risk of pressure sores in other areas and the development of generalised atrophy and weakness resulting in the loss of physical functions such as transferring and sitting.
Various medical complications arise from pressure sores. Pressure ulcers increase the risk of sepsis, which occurs in 26% of patients with non‐healing ulcers 6, and osteomyelitis, which occurs in 86% of stage IV ulcers 7. A study of mortality of inpatients showed a mortality rate of 11·6% in those with secondary pressure ulcers 8. Because of such risks and a tendency for pressure ulcers to become chronic, standard management relies on removing the cause and decreasing the risk of local colonisation by pathogens. However, no definitive treatment exists to speed up the ulcer healing process in order to help patients improve their functional status 9.
The purpose of the study was to assess the impact of treatment with activated protein C (APC) when combined with topical negative pressure wound therapy (TNP) for pressure areas that have not responded to standard care for an extended period of time. This case study describes the treatment of both a grade III and a grade IV ulcer with APC, in conjunction with TNP. Recombinant human APC is a versatile protein exhibiting therapeutic potential in various pathologies including sepsis, lung disorders, kidney injury, diabetic nephropathy, inflammatory bowel disease, systemic lupus erythematosus, amyotrophic lateral sclerosis, cancer metastasis and ischaemic stroke and reperfusion injury following myocardial infarction 10, 11, 12 and has recently shown efficacy in treating chronic wounds 13, 14, 15. TNP has been used increasingly in recent years for various acute and chronic wounds; however, its efficacy is yet to be backed by a strong evidence base 16, 17, 18.
Methods
Two patients were included in this study. Their selection was based on the non‐responsiveness of their wounds to all other therapies, as described in each case. Written informed consent was obtained from both patients and the study was approved by the Northern Sydney Area Human Research Ethics Committee. The primary endpoint was time to complete healing.
APC administration followed a standardised procedure. APC was drawn up from the vial into a syringe with a 30G needle. It was administered first into the base and edges of the wound then the peripheral wall of healthy tissue around the wound with the intention of allowing the APC to penetrate into the soft tissues around the wound. The wound was covered with Tegaderm™ (3M Health Care, St. Paul, MN) (an occlusive transparent dressing that prevents leakage of APC but also allows visualsation of the injection). Any remaining APC volume was injected through the Tegaderm™ into the wound cavity. The dose was different for the two patients. For case 1 the APC was drawn straight from the vial, at a strength of 400 µg/ml. For case 2, the APC was diluted at a ratio of 1:1 with normal saline at a strength of 200 µg/ml. The amount administered varied for each treatment and reflected the change in size of the wound cavity as the dosage reduced. The amount administered is described in each patient's treatment. TNP was administered as previously described 15.
Results
Case 1
A 39‐year‐old female with a past medical history of severe rheumatoid arthritis underwent bilateral hip replacements following degenerative disease. Given that she was unable to lie on either hip postoperatively she remained in the supine position and developed a sacral pressure sore, which progressively worsened over 2 years. The sore was unresponsive to conventional therapies consisting of various wound dressings for the first year, and continued to be unresponsive when TNP was added to the treatment regimen for the second year. Given the patient was bedridden, and would therefore lie on the wound, surgical management was likely to fail. The wound size did not change for 12 months before treatment. The patient had normal blood biochemistry and a review by a dietician showed good nutritional status and no supplementation required.
She was then started on a regimen of locally applied APC and TNP as described in Methods. On presentation to the plastic surgery team she had a circular midline sacral grade III pressure sore measuring 20 mm in diameter and 10 mm at its deepest point. The sacrum and overlying ligaments were palpable at the base of the wound. She was started on twice weekly APC (1 ml, 400 µg/ml) by injection, as described earlier. By day 10 the wound base was covered with healthy granulation tissue and by day 21 the entire wound depth was covered with granulation tissue and the wound size had decreased to a diameter of 12 mm with a negligible depth. Treatment with APC and TNP was stopped on day 24 (6 mm diameter and a negligible depth) and simple dressings were applied as per standard management. The wound continued to epithelialise and was completely healed by day 35 (Figure 1).
Figure 1.
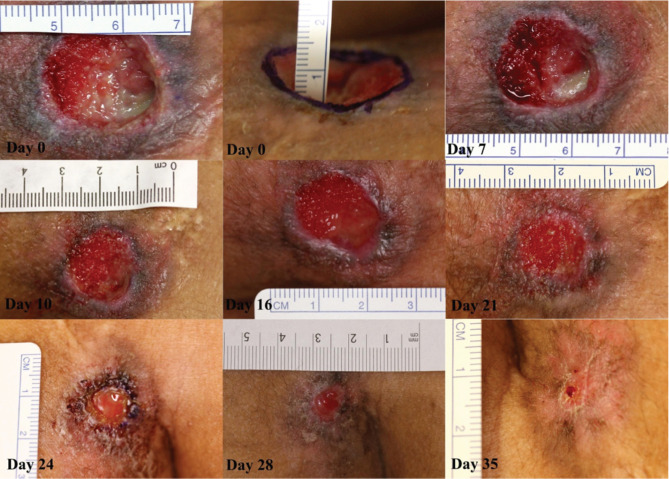
Case 1. Sacral pressure ulcer progression through healing after activated protein C (APC) administration. On day 0 the wound had a central depth of 10 mm. At day 7 the wound was 20 mm in diameter. Rapid growth of granulation tissue is apparent from day 7 to day 16. At day 16 the diameter was 18 mm. On day 21 the diameter was 14 mm and by day 24 the wound was 6 mm in diameter. Therapy was stopped at this point. At day 28 the diameter was 5 mm. Final follow‐up at day 35 showed that the wound had healed completely. No images of follow‐up.
During the first dressing change, the patient reported some pain, possibly due to infiltration of the injected solution into surrounding soft tissue. As a result, oral oxycodone 5–10 mg was given prior to each subsequent treatment, which successfully reduced her pain. There were no other complications or reports of pain associated with the treatment. Long‐term follow‐up in the clinic after 6 months showed that the wound remained closed. After that point she was lost to further follow‐up.
Case 2
A 33‐year‐old male driver involved in a high‐speed motor vehicle accident sustained 40% total body surface area (TBSA) burns, multiple spinal fractures and multiple other trauma‐related soft tissue and visceral injuries and a complete T7 spinal cord injury resulting in paraplegia. He required bilateral above the knee amputation due to trauma sustained in the accident. He had a defunctioning colostomy and had been in hospital for 6 months for management of his injuries and burns. During this time he was bed‐bound and due to the fragility of his skin following the burns he developed a large stage‐IV sacral pressure sore. Stage IV is defined as full thickness loss of skin with extensive destruction 19. He was made to lie on an air mattress and because he was an inpatient, was turned from side to side every 4 hours. The ulcer continued to enlarge despite pressure care and therapy with conventional dressings. After 2 months of this conservative management with dressings he was started on treatment with TNP for a further 1 month, which was also ineffective. In conjunction with the dietetics team as part of normal treatment for burns he was placed on a diet containing high protein, high electrolyte and high minerals; however, there was still no improvement. Grafting was not possible due to the depth of the wound (Figure 2).
Figure 2.
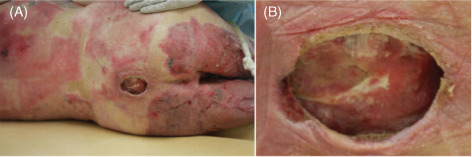
Case 2. Sacral pressure ulcer after 3 months of conventional therapy. (A) Sacral wound and surrounding burn areas. (B) Sacral wound after 3 months of conventional therapy [2 months of dressings and 1 month of topical negative pressure wound therapy (TNP)] measuring 60 × 50 mm2 wide × 20 mm deep. Both the superior and inferior aspects of the wound were undermined, extending superiorly and inferiorly. The lumbosacral spinous processes and their supraspinal ligaments were palpable at the base of the wound.
After a total of 3 months of conventional therapy with dressings and TNP, the wound size had not reduced. The undermined cavities continued to enlarge such that two fingers could be inserted in the cavities, past the superior and inferior margins of the wound. The wound doubled in size over the 4 weeks before treatment.
This patient was then started on APC and TNP and standard pressure care was continued. Bloods tests were unremarkable at the beginning of the study. He received supplemental feeding via nasogastric tube for a month before the start of APC therapy and for the duration of the trial and was reviewed regularly by the dietician. To account for the increased wound size in this patient compared to case 1, a larger dose of APC was applied by injection, as described in Methods section. Treatment consisted of 8 ml of APC (200 g/ml) twice weekly until day 13, followed by 5 ml until day 19, then 3 ml until day 64. The reduced amounts reflected the change in size of the wound cavity (Figure 3). On day 5, the wound base had formed a large amount of granulation tissue with visibly rich vascularity. Treatment was sustained and this granulation tissue continued to expand. By day 13, granulation tissue was accumulating in the superior and inferior undermined cavities of the wound. On day 27, the superior and inferior cavities were almost completely filled with granulation tissue and the wound size was decreased to 50 × 30 × 5 mm3. Up to this point, the patient was an inpatient receiving wound therapy while also being treated for his other injuries. The patient underwent two further APC treatments as an outpatient before being discharged on day 64, when the wound cavity had completely filled with granulation tissue and the wound size was reduced to 20 × 10 × 0 mm3. Therapy was discontinued at this point and the patient continued to have wound dressing changes at a rehabilitation hospital to which he was discharged. He was seen again in the clinic 4 months after discharge and the wound was completely healed and the patient was healthy and independently mobile in a wheelchair.
Figure 3.
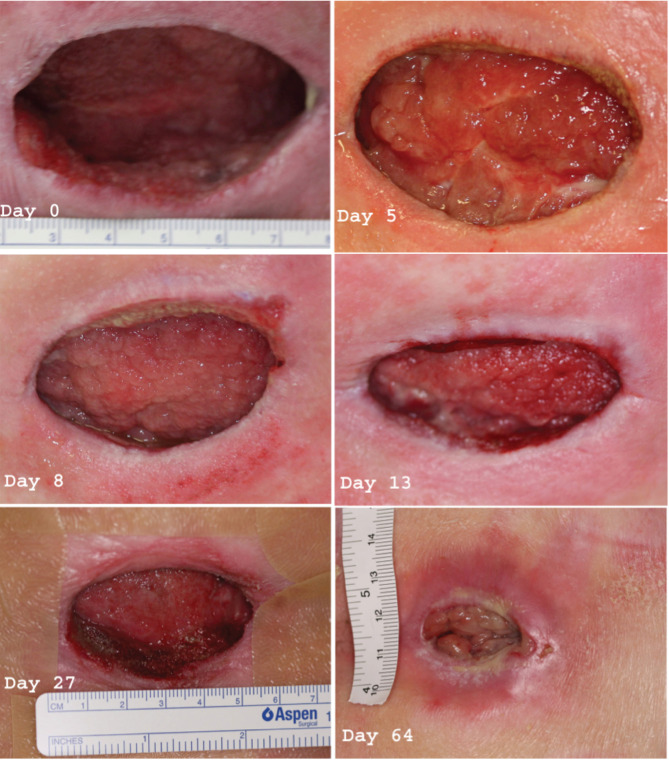
Case 2. Sacral pressure ulcer progression through healing after activated protein C (APC) administration. Day 0 image shows the wound after approximately 6 weeks of topical negative pressure wound therapy (TNP) where the size remained unchanged at 60 × 50 × 20 mm3. The superior and inferior cavities approximately had extended 50 mm past the wound edge. Lower lumbar and sacral vertebral spinous processes and their overlying ligaments were palpable at the base of the wound. Day 5 image shows a significant amount of granulation tissue forming at the base of the wound (arrow). This tissue was vascular with a rapid capillary refill. On day 8 the granulation tissue continued to expand. On day 13 there was granulation tissue filling the superior and inferior cavities. On day 27 wound size had decreased to 50 × 30 × 5 mm3 and the superior and inferior cavities had closed further. On day 64 the wound was almost completely filled with granulation tissue. The size had considerably shrunk to 20 × 10 × 0 mm3. Treatment was stopped at this point and the wound continued to heal up to day 80 (no image) when there was no visible wound.
Discussion
There is growing evidence that APC has potential as a safe and effective treatment option for wound healing. Previous research has shown that APC accelerates healing of recalcitrant orthopaedic 15, diabetic, venous and arteriovenous ulcers 13, 14, but this is the first evidence that APC is effective in healing pressure sores. Left to conservative care, clinical experience suggests these ulcers may have continued to degrade leading to a high risk of sepsis, osteomyelitis and, in case 2, mortality 6, 7, 20. Modern medicine demands better management for such a common debilitating condition. Furthermore, when pressure ulcers are treated, the associated comorbidities that occur with bed rest are also treated. These include the risk of additional pressure sores 21, decreased bone density 22, increased risk of falls and fractures 23, thromboembolic disease, generalised atrophy, joint contractures and insulin resistance 24.
The established mechanisms behind the wound healing capacities of APC fit well within the clinical picture of wound healing seen here in these patients. APC has been shown to support wound healing through various mechanisms. After injection into the tissues surrounding a wound, the APC molecule binds to its membrane receptor on keratinocytes, the endothelial protein C receptor (ECPR). Upon binding to ECPR, APC then links to the G‐protein coupled receptors, protease activated receptors 1 and 2 (PAR‐1 and PAR‐2). APC linkage to PAR‐1 transactivates PAR‐2 to induce a cascade of intracellular events, which activates the PI3K/Src/Akt pathway responsible for keratinocyte proliferation. APC linkage to PAR‐2 induces inhibition of the p38 pathway, which is responsible for decreasing the inflammation 25. APC also acts via PAR‐1 on keratinocytes to transactivate endothelial growth factor receptor (EGFR), which transactivates the angiopoietin receptor Tie2. Tie2 induces phosphorylation of the protein kinase Akt, which induces angiogenesis. Therefore, APC's stimulation of angiogenesis could account for the rapid accumulation of healthy granulation tissue 25. Akt induces cell survival and improved cell barrier function 25. Proliferation is required for wound healing, but just as importantly, inflammation needs to be controlled so that the cells of the dermis and epidermis can progress through each phase necessary for complete wound healing. These phases overlap, but are generally considered to be in the order of inflammation, granulation, angiogenesis, epithelialisation and remodelling. The mechanism of action of APC described above shows how APC provides support through each of these phases of healing. Although we are unable to confirm the molecular signalling mechanisms in these cases, they do provide evidence that APC is effective in clinically reducing inflammation, producing healthy granulation tissue reinforced by angiogenesis and supporting effective epithelialisation leading to wound closure.
The ulcers shown in these cases provide typical examples of complicated, chronic, non‐healing ulcers with poor prognosis. Both patients had characteristics placing them at risk of comorbidities 21, and these characteristics were more for the patient presented in case 2 as he had loss of sensory function and structural changes to skin with significant loss of mobility and activity. Both patients' ulcers had been managed conservatively with no improvement and both faced a risk of significant comorbidity. Both patients were then offered TNP to be added to their management and neither showed clinical improvement or changes in ulcer healing. After starting treatment with APC, however, the condition of both patients improved substantially, correlating with the rapid healing of their ulcers. The well‐defined mechanism of APC and the temporal relationship between clinical improvement and APC administration are consistent with APC being key to reversing the stagnant condition of these ulcers.
In summary, these were severe, non‐healing, chronic wounds not responding to multiple modalities of standard care, including debridement, multiple types of wound dressing, pressure care and TNP, suggesting that the administration of APC directly altered the course of ulcer progression. The limitations of case studies are recognised and a randomised placebo‐controlled double blind trial is required to determine the efficacy of APC in pressure sores and whether TNP enhances the benefits of APC.
References
- 1. Meehan M. Multisite pressure ulcer prevalence survey. Decubitus 1990;3:14–7. [DOI] [PubMed] [Google Scholar]
- 2. Berlowitz DR, Wilking SV. The short‐term outcome of pressure sores. J Am Geriatr Soc 1990;38:748–52. [DOI] [PubMed] [Google Scholar]
- 3. Ditunno JF, Formal CS. Chronic Spinal Cord Injury. N Engl J Med 1994;330:550–6. [DOI] [PubMed] [Google Scholar]
- 4. Bergstrom N, Horn SD, Smout RJ, Bender SA, Ferguson ML, Taler G, Sauer AC, Sharkey SS, Voss AC. The national pressure ulcer long‐term care study: outcomes of pressure ulcer treatments in long‐term care. J Am Geriatr Soc 2005;53:1721–9. [DOI] [PubMed] [Google Scholar]
- 5. Bluestein D, Javaheri A. Pressure ulcers: prevention, evaluation, and management. Am Fam Physician 2008;78:1186–94. [PubMed] [Google Scholar]
- 6. Staas WE Jr, Cioschi HM. Pressure sores – a multifaceted approach to prevention and treatment. West J Med 1991;154:539–44. [PMC free article] [PubMed] [Google Scholar]
- 7. Deloach ED, DiBenedetto RJ, Womble L, Gilley JD. The treatment of osteomyelitis underlying pressure ulcers. Decubitus 1992;5:32–41. [PubMed] [Google Scholar]
- 8. McInnes E, Bell‐Syer SEM, Dumville JC, Legood R, Cullum NA. Support surfaces for pressure ulcer prevention. Cochrane Database Syst Rev 2008;4:CD001735. [DOI] [PubMed] [Google Scholar]
- 9. Werdin F, Tenenhaus M, Rennekampff H‐O. Chronic wound care. Lancet 372:1860‐2. [DOI] [PubMed] [Google Scholar]
- 10. Montes R, Puy C, Molina E, Hermida J. Is EPCR a multi‐ligand receptor? Pros and cons. Thromb Haemost 2012;107:815–26. [DOI] [PubMed] [Google Scholar]
- 11. Montes R, Díez J, Hermida J. Activated protein C: reasons to believe. J Thromb Haemost 2012;10:1733–5. [DOI] [PubMed] [Google Scholar]
- 12. Jackson CJ, Xue M. Activated protein C – an anticoagulant that does more than stop clots. Int J Biochem Cell Biol 2008;40:2692–7. [DOI] [PubMed] [Google Scholar]
- 13. Whitmont K, McKelvey KJ, Fulcher G, Reid ML, Xue M, Cooper A, Jackson CJ. Treatment of chronic diabetic lower leg ulcers with activated protein C: a randomised placebo‐controlled, double‐blind pilot clinical trial. Int Wound J 2013; [Epub 2013/07/15]; doi: 10.1111/iwj.12125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Whitmont K, Reid I, Tritton S, March L, Xue M, Lee M, Fulcher G, Sambrook P, Slobedman E, Cooper A, Jackson C. Treatment of chronic leg ulcers with topical activated protein C. Arch Dermatol 2008;144:1479–83. [DOI] [PubMed] [Google Scholar]
- 15. Wijewardena A, Vandervord E, Lajevardi SS, Vandervord J, Jackson CJ. Combination of activated protein C and topical negative pressure rapidly regenerates granulation tissue over exposed bone to heal recalcitrant orthopedic wounds. Int J Low Extrem Wounds 2011;10:146–51. [DOI] [PubMed] [Google Scholar]
- 16. Ubbink Dirk T, Westerbos Stijn J, Evans D, Land L, Vermeulen H. Topical negative pressure for treating chronic wounds. Cochrane Database Syst Rev 2008;3:CD001898. [DOI] [PubMed] [Google Scholar]
- 17. Thompson JT, Marks MW. Negative pressure wound therapy. Clin Plast Surg 2007;34:673–84. [DOI] [PubMed] [Google Scholar]
- 18. Kim PJ, Attinger CE, Steinberg JS, Evans KK, Lehner B, Willy C, Lavery L, Wolvos T, Orgill D, Ennis W, Lantis J, Gabriel A, Schultz G. Negative pressure wound therapy with instillation: consensus guidelines. Plast Reconstr Surg 2013;132:1569–79. [DOI] [PubMed] [Google Scholar]
- 19. Gupta S, Baharestani M, Baranoski S, de Leon J, Engel SJ, Mendez‐Eastman S, Niezgoda JA, Pompeo MQ. Guidelines for managing pressure ulcers with negative pressure wound therapy. Adv Skin Wound Care 2004;17(2 Suppl):1–16. [DOI] [PubMed] [Google Scholar]
- 20. McInnes E, Dumville JC, Jammali‐Blasi A, Bell‐Syer SE. Support surfaces for treating pressure ulcers. Cochrane Database Syst Rev 2011;12:CD009490. [DOI] [PubMed] [Google Scholar]
- 21. Bergstrom N, Braden BJ, Laguzza A, Holman V. The Braden scale for predicting pressure sore risk. Nurs Res 1987;36:205–10. [PubMed] [Google Scholar]
- 22. Bloomfield SA. Changes in musculoskeletal structure and function with prolonged bed rest. Med Sci Sports Exerc 1997;29:197–206. [DOI] [PubMed] [Google Scholar]
- 23. Campbell AJ, Borrie MJ, Spears GF. Risk factors for falls in a community‐based prospective study of people 70 years and older. J Gerontol 1989;44:M112–7. [DOI] [PubMed] [Google Scholar]
- 24. Brower RG. Consequences of bed rest. Crit Care Med 2009;37:S422–8. [DOI] [PubMed] [Google Scholar]
- 25. Julovi SM, Xue M, Dervish S, Sambrook PN, March L, Jackson CJ. Protease activated receptor‐2 mediates activated protein C‐induced cutaneous wound healing via inhibition of p38. Am J Pathol 2011;179:2233–42. [DOI] [PMC free article] [PubMed] [Google Scholar]


