Abstract
Up to 15 billion dollars of US health care expenditure each year is consumed by treatment of poorly healing wounds whose etiologies are often associated with aberrancies in tissue oxygenation. To address this issue, several modes of tissue oxygen delivery systems exist, including Hyperbaric Oxygen Therapy (HBOT) and Topical Oxygen Therapy (TOT), but their efficacies have yet to be fully substantiated. Micro/nanobubbles (MNBs), which range anywhere from 100 μm to <1 μm in diameter and are relatively stable for hours, offer a new mode of oxygen delivery to wounds. The aim of this article is to systematically review literature examining the use of TOT for wound healing and use of MNBs for tissue oxygenation using the MEDLINE database. The search yielded 87 articles (12 MNB articles and 75 TOT articles), of which 52 met the inclusion criteria for this literature review (12 MNB articles and 40 TOT articles). Additionally, we present an analysis on the efficacy of our MNB generating technology and propose its use as a wound healing agent.
Keywords: micro/nanobubbles, nanobubbles, tissue oxygenation, topical oxygen therapy, wound healing
1. INTRODUCTION
Oxygen is the most important element of life, playing a part in almost every cellular function. The process of oxidative phosphorylation requires oxygen to produce energy that is needed for the everyday operation of cellular machinery.1 Oxygen is also involved in a host of other processes, including the oxidative killing of bacteria, angiogenesis, collagen formation and the re‐epithelisation of wounds.2, 3, 4, 5 Because oxygen homeostasis is so crucial to life, it is of no surprise that nature has evolved intricate systems for efficiently extracting oxygen from the atmosphere and transporting it to tissues.
The process of normal wound healing consists of three phases: inflammation, proliferation and tissue remodelling. In each of these steps, oxygen plays an important role.1 For example, during the inflammatory phase, reactive oxygen species aid in the oxidative killing of bacteria and, in low concentrations, act as cellular messengers to promote activities involved in wound healing.3 During the proliferative phase, oxygen increases keratinocyte differentiation and migration and fibroblast and endothelial cell production, as well as re‐epithelisation through growth factor activation by reactive oxygen species.5, 6, 7, 8, 9 Oxygen is also required for myofibroblast differentiation, enzyme‐mediated posttranslational hydroxylation and collagen cross‐linking.10, 11, 12, 13 Finally, the process of angiogenesis itself proceeds more efficiently and is maintained by sufficient oxygenation of the tissue through vascular endothelial growth factor (VEGF) production.2, 14, 15
Under normal conditions, the partial pressure of serum oxygen (pO2) is typically around 100 mmHg, and once at the capillary level, oxygen can diffuse up to 64 μm to sufficiently nourish the skin.16 Studies have shown that the necessary transcutaneous oxygen levels (tcPO2) for normal skin is 30 mmHg or greater.17 Levels of tcPO2 lower than 30 mmHg are insufficient for tissue diffusion and lead to abnormal wound healing.18 Under ideal conditions, when tissue oxygen pressure is greater than 30 mmHg, normal granulation tissue develops, and the wound follows the natural course of healing.18 However, wounds with 13 to 30 mmHg of oxygen show an accumulation of necrotic debris and little or no granulation tissue, leading to slower or even stalled healing.18 Finally, in skin with less than 13 mmHg of oxygen tension, the wound becomes gangrenous because there is insufficient oxygenation to support the most basic metabolic activities.18
Researchers once believed that the sole source of oxygen available for biochemical processes in wound healing was derived from the systemic circulation. It is now thought, and supported by several studies, that skin can utilise oxygen from the atmosphere. In vitro studies show that oxygen passes through the stratum corneum and up to 0.30 mm of the superficial dermis.19, 20 Similarly, in vivo oxygen sensing assays, which are still being developed and perfected, demonstrate that oxygen passes up to 0.25 to 0.40 mm below the skin surface.21
Today, chronic wound treatment consumes 6 to 15 billion dollars in US health care costs, much of which is attributed to conditions that are associated with aberrancies in oxygen delivery.22 Whether it is chronic non‐healing wounds caused by peripheral vascular disease, diabetes mellitus, pressure ulcers or chronic venous insufficiency, oxygen is one of the limiting reagents. Thus, since the advent of hyperbaric oxygen therapy (HBOT) by Henshaw in 1662, medicine has sought to improve oxygen delivery to poorly healing tissue.23
HBOT is defined as 100% oxygen delivered systemically at 2 to 3 times atmospheric pressure.24 It is thought that HBOT increases the pO2 of oxygen systemically by 10 to 15 times the normal value, creating a gradient that allows oxygen to diffuse into hypoxic tissue.25 The Undersea and Hyperbaric Medical Society indicates the use of HBOT for several conditions, including decompression sickness, acute thermal burns, arterial insufficiencies, refractory osteomyelitis, and compromised grafts and flaps as well as necrotising and anaerobic soft tissue infections.26 In addition, new studies have revealed a possible regenerative component to HBOT therapy where the release of progenitor cells has been demonstrated with its use.27 While HBOT has advocates and numerous studies, and randomised clinical trials point to its efficacy,28, 29, 30, 31 HBOT occasionally causes vasoconstriction, cellular toxicity and tissue damage.24, 32, 33 HBOT works through oxygenation of the systemic circulation, and thus, conditions that limit blood flow, such as peripheral vascular disease, limit the therapeutic effects of HBOT.32 In addition, new studies demonstrate that healing tissue relies more on oxygen diffusion rather than the blood oxygen‐carrying capacity.32 The paucity of blood vessels in ischaemic wound areas forces oxygen to diffuse over large distances of 150 μm or more in order to reach hypoxic tissue at the base of ulcers, limiting the effects of HBOT.34 Finally, HBOT is expensive, time consuming, and requires patients to leave their homes for treatment, all of which limit its utility and compliance.5
The limitations of HBOT, coupled with recent advances in medical technology over the past several years, necessitated the development of topical oxygen wound therapies (TOT). The purpose of this review is to systematically investigate the basic science and clinical literature of the different modes of TOT currently available and to introduce micro/nanobubbles (MNBs) as a novel therapeutic modality for oxygen delivery to wounds. Recognising that these topical therapies are relatively new and varied, the presented studies are limited in scope and design and cannot be compared with the extent of studies that relate to HBOT. However, it is also recognised that topical therapies may have distinct and excellent advantages both in terms of simplicity of application and in physiological modes of action.
2. STUDY DESIGN
A PubMed literature search was conducted on June 10th to search for articles on TOT and MNBs in relation to wound healing. Keywords included the following: “topical oxygen therapy,” “topical gaseous oxygen,” “topical dissolved oxygen,” “topical oxygen emulsion,” “topical hyperbaric,” “oxygen generating wound dressing,” “microbubbles, nanobubbles,” “nanodroplets,” “wound healing,” “pressure ulcer” and “diabetic foot ulcer”. This search strategy was refined after conducting individual searches for terms previously used to terms used to describe TOT and MNBs. Additionally, our search strategy was reviewed independently by a librarian. Referenced articles from the selected studies were further evaluated for potential inclusion in our analysis. In total, 75 unique studies were identified from our TOT search, and 12 unique articles on MNBs as they pertain to liquid ventilation and wound healing were acquired.
3. SELECTION CRITERIA AND DATA COLLECTION
All abstracts were assessed if they used any form of TOT or MNBs as a source of oxygen delivery. Articles were generally excluded if they considered the use of MNBs or TOT for purposes other than wound healing and oxygen delivery. The full text of each article was read critically by a research assistant (J.P. or Z.O.) to ensure each article met selection criteria. Each summary and article was then read and cross‐referenced for completeness (L.S.). In total, 52 articles (12 MNB and 40 TOT) were included for the review. For additional information, see the PRISMA diagram for TOT and MNB papers (Figures 1 and 2)
Figure 1.
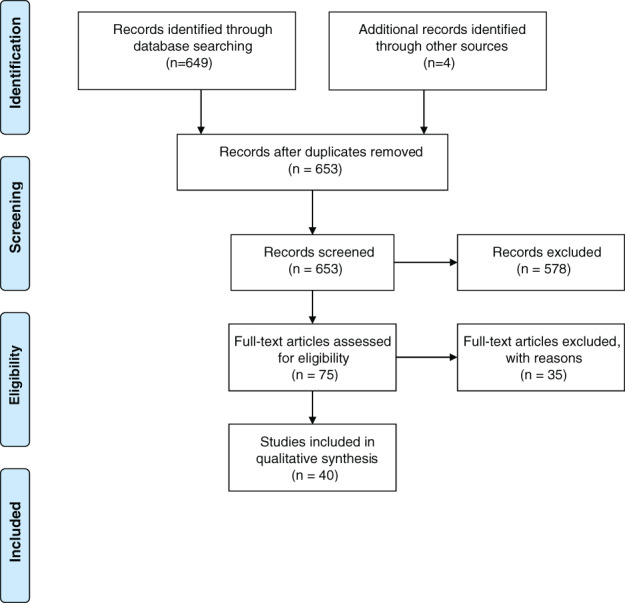
TOT flow diagram: 653 articles were obtained from search terms. After reviewing all 653 abstracts, only 75 articles met the inclusion criteria for complete full‐text assessment. After full‐text assessment, only 40 articles were found to utilise a mode of TOT and were thus incorporated in the review
Figure 2.
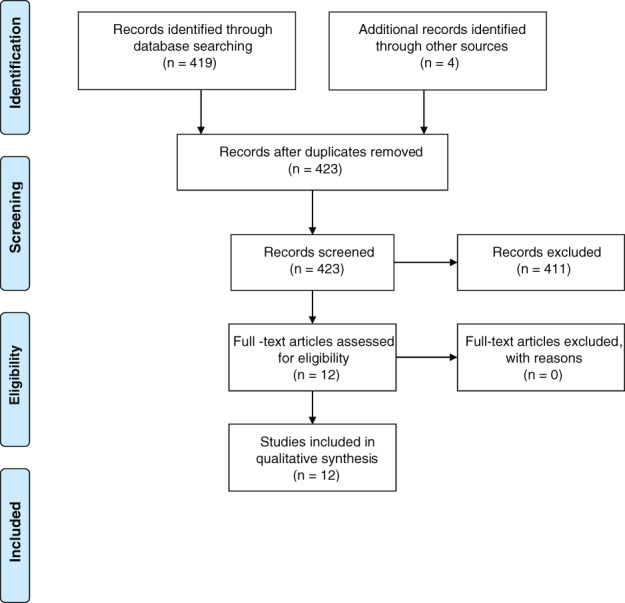
MNB flow diagram: 423 articles were obtained from search terms. After reviewing all 423 abstracts, only 12 articles met the inclusion criteria for complete full‐text assessment. These 12 articles were incorporated in the systematic review
4. A. A GENERAL OVERVIEW OF THERAPEUTIC TOPICAL OXYGEN
With a growing understanding of the limitations of HBOT, a variety of TOT methodologies were developed to provide more effective external oxygen delivery, bypass circulatory issues and increase accessibility. The FDA classifies topical oxygen chambers for extremities as class II medical products.35 As such, TOT may be subject to regulation by the FDA, but it clears the way for future therapies. To be an effective TOT, a product needs oxygen to diffuse through the liquid or solid phase of the tissue and migrate into oxygen‐deficient cells.36 In this review, we will examine literature on the 4 main categories of TOT: (1) Topical pressurise oxygen therapy (TPOT), (2) Topical continuous oxygen therapy (TCOT), (3) Wound dressing that release oxygen, or (4) Topical oxygen emulsion. For each modality, we will first review animal and in vitro literature followed by translational research and case studies. Additionally, we will present a study comparing topical dissolved oxygen and topical gaseous oxygen on tissue oxygenation. We will also review existing literature on MNBs when used for the purpose of tissue oxygenation. Finally, Table 1 further describes the study model and sample size for each TOT article reviewed.
Table 1.
Reviewed topical oxygen therapy study characteristics
| Group | TOT subtype | Study model | Number of Wounds/Patients Treated |
|---|---|---|---|
| Fries et al38 | TPOT | Basic Science: Porcine dermal wound | 20 wounds |
| Gordillo et al41 | TPOT | Basic Science: Porcine dermal wound | 20 wounds |
| Rao et al40 | TPOT | Basic Science: Rat ischemic limb wound | 12 wounds |
| Gordillo et al41 | TPOT vs HBOT | Clinical: Non‐randomised study | HBOT: 32 patients; TPOT: 25 patients |
| Gordillo et al42 | TPOT | Clinical: Retrospective analysis of wounds | 58 wounds/32 patients |
| Blackman et al43 | TPOT vs conventional compression dressing | Clinical: DFU | TPOT: 17 patients; CCD: 11 patients |
| Tawfick et al | TPOT vs conventional compression dressing | Clinical: RVU | TPOT: 67 patients; CCD: 65 patients |
| Tawfick et al | TPOT vs conventional compression dressing | Clinical: RVU | TPOT: 46 patients; CCD: 37 patients |
| Kalliainen et al46 | TPOT | Clinical: Retrospective analysis of complex wounds | 58 wounds/32 patients |
| Copeland et al47 | TPOT | Clinical: Retrospective analysis of wounds | 4127 wounds/3462 patients |
| Fischer48 | TPOT | Clinical: Various wound type: ulcer, sores etc. | 52 patients |
| Ignacio et al49 | TPOT | Clinical: Grade III leg and foot ulcers | 15 patients |
| Heng et al51 | TPOT | Clinical: Leg ulcers of various etiologies | 6 patients |
| Heng17 | TPOT | Clinical: Gangreous wounds | 24 wounds/15 patients |
| Heng et al50 | TPOT | Clinical: Gangreous wounds | 40 patients/79 wounds |
| Upson54 | TPOT | Clinical: 2 Case reports: leg ulcers from ischemia | 2 patient |
| Leslie et al53 | TPOT | Clinical: Prospective randomised control on DFU | 12 patients |
| Agarwal et al52 | TPOT | Clinical: Human necrotising wound and trauma wound | 3 wounds/3 patients |
| Landau and Schattner55 | TPOT + LEL | Clinical: DFU | 100 patients |
| Landau et al56 | TPOT + LEL | Clinical DFU + CVU | 374 patients |
| Nie et al57 | TPOT + bFGF | Clinical: 2 degree burn | 34 wounds/25 patients |
| Kaufman et al58 | TCOT | Basic Science: Guinea pig 3rd degree burns | 8 wounds |
| Tracey et al62 | TCOT: Epiflo | Basic Science: Dermal wounds in horse model | 4 hourses; 16 wounds |
| Kaufman et al59 | TCOT | Basic Science: Guinea pig dermal wound | 4 wounds |
| Woo et al61 | TCOT: Epiflo | Clinical: Chronic wounds | 9 patients |
| Yu et al63 | TCOT | Clinical: DFU | 10 patients |
| Banks and Ho60 | TCOT: Epiflo | Clinical: Stage IV pressure ulcers | 3 patients |
| Roe et al18 | TGO vs TDO | Basic Science: Human skin samples | Skin samples |
| Gueldner et al64 | Wound dressing: Ox66 | Basic Science: Human fibroblasts and human keratinocytes | n/a |
| Chandra et al65 | Wound dressing: TOT oxygen generating wound dressing | Basic Science: Porcine full‐thickness surgical wounds | 5 pigs, 4 wounds per pig, 2 control & 2 treated |
| Lo et al66 | Wound dressing: Microfluidic bandage | Basic Science: 13‐week old mice full‐thickness surgical wound | 5 mice with 5 wounds treated w/ oxygen compared to controls |
| Zellner et al68 | Wound dressing: Dissolved oxygen dressing | Basic Science: Porcine full‐thickness surgical wound | 4 pigs, 8 wounds treated with oxygen vs control |
| Paola et al | Wound dressing: SOS | Clinical: DFU | 218 patients, 110 treated with oxygen vs the rest control |
| Ivins et al70 | Wound dressing: Oxyzyme hydrogel | Clinical: Leg ulcers | 31 patients; only outcome of 5 patients reported |
| Almeleh71 | Wound dressing: Oxyge delivering hydrogel | Clinical: 2nd Degree Burn | 2 wounds/2 patients |
| Kellar et al69 | Wound dressing: Oxygen‐enriched dressing | Clinical: Healthy human subject skin | 50 subjects, no wounds |
| Li et al73 | TOE Perfluorocarbon Oxygen emulsion | Basic Science: Perfluorocarbon Oxygen Emulsion | 6 pigs, 6 wounds treated TOE vs control |
| Li et al74 | TOE Perfluorocarbon Oxygen emulsion | Basic Science: TOE Perfluorocarbon Oxygen Emulsion | 6 pigs, 6 wounds treated TOE vs control |
| Davis et al75 | TOE Perfluorocarbon Oxygen emulsion | Basic Science: 2nd degree partial thickness Porcine wounds | 8 pigs, 40 wounds per pig treated with oxygen vs control |
| Onouye et al | Other: Oxygen mist | Clinical: Post CO2 Laser Resurfacing | 3 wounds/3 patients |
Abbreviations: CVU, chronic venous ulcer; DFU, diabetic foot ulcer; LEL, low energy laser; SOS, super‐oxidised solution; TCOT, topical continuous oxygen therapy; TGO, topical gaseous oxygen; TOE, topical oxygen emulsion; TOT, topical oxygen therapy; TPOT, topical pressuried oxygen therapy; RVU, refractory venous ulcer.
5. B. TOPICAL PRESSURISED OXYGEN THERAPY
Topical Pressurised Oxygen Therapy (TPOT) involves the use of a chamber or bag, which encloses the affected areas while administering 100% oxygen at a pressure that is slightly above 1 atm.37 This reduces costs, allows for in‐home treatment, and minimising the potential side effects associated with HBOT.37 Additionally, TPOT, unlike HBOT, provides no risk of systemic oxygen toxicity and, thus, can be administered until the wound is healed.37
Initial laboratory studies on TPOT were performed by Fries et al and Gordillo et al who investigated the effects of TPOT on excisional dermal wounds in pigs while evaluating wound oxygenation levels.38, 39 The authors demonstrated improved healing, increased VEGF expression and improved neovascularisation compared to the controls.39 Both studies found an increase in wound oxygenation and reported pO2 measurements of 40 mmHg in the TPOT‐treated wounds, which was more than 4 times that which was measured in the control groups.38, 39 Rao et al exposed rats with ischaemic limb wounds to TPOT, showing increased collagen fibre accumulation, better neovascularisation and a significant decrease in healing time in the wounds treated with TPOT compared to the controls.40
Translational research supports literature on TPOT obtained from animal models. Contrasting TPOT with HBOT, Gordillo et al performed a clinical study in 2008 on 32 patients receiving HBOT and compared them with 25 patients receiving TPOT.41 A statistically significant improvement in wound size was observed in the TPOT‐ treated wounds, while no significant improvement was found in the HBOT‐treated wounds.41 Similarly, in a retrospective analysis of 58 wounds in 32 patients who received TPOT, Gordillo et al reported a 75% overall healing rate over 9 months of treatment in 2004.42
A series of clinical studies were performed by Blackman et al to assess the effects of TPOT vs a conventional compression dressing (CCD) on the healing of diabetic foot ulcers (DFUs). Of the 28 patients enrolled in the study, 17 received TPOT 5 times a week, while 11 received CCD.43 The TPOT‐treated wounds were significantly (P = 0.04) more likely to show complete epithelialisation using TPOT (82.4%) than the CCD‐treated wounds (45.5%).43 In addition, there was a significant difference in the median wound closure time in the wounds subjected to TPOT versus compression (56 vs 93 days, respectively).43 Tawfick et al studied the benefits of TPOT over CCD in 83 patients with refractory venous ulcers (RVU).44 After 12 weeks, significantly more ulcers healed using TPOT (80%) than CCD (35%), and the median healing time was significantly shorter using TPOT (45 days) when compared with CCD (182 days).44 Additionally, Tawfick et al reproduced the study with similar results in a larger sample of patients.45
Kalliainen et al retrospectively looked at 58 wounds in 32 patients using TPOT and showed that 65.5% of the wounds healed.46 Copeland et al performed a retrospective chart review examining 4127 wounds.47 Using their TPOT protocol, 59.4% of chronic wounds decreased in size, while 41.6% of wounds showed no healing.47 Chronic wounds that were smaller and younger than 1‐year‐old experienced the greatest benefit from TPOT.47 Both studies concluded that TPOT improved wound healing.46, 47
From a clinical standpoint, Fischer was one of the first to utilise TPOT on patients with pressure sores and skin ulcers. He administered pure oxygen at 22 mmHg at 2 to 8 liters/minute.48 The result revealed that TPOT improved the healing time and suppressed bacterial growth in diabetic foot ulcers, venous stasis ulcers and pressure sores. Similarly, Ignacio et al tested TPOT at 16 to 20 mmHg 2 times a day for 45 minutes on grade‐III ulcers and reported compete healing in 11 of his 15 patients (73.3% healing rate).49
For 15 patients who failed standard‐of‐care treatment for gangrenous wounds, Heng et al administered TPOT for 4 weeks. After seeing a significant decrease in ulcer size, a larger randomised control study was conducted with 40 patients and 79 wounds.17, 50 At 12 months, 90% of the ulcers in the TPOT group healed compared with 22% in the control group.50 Heng et al observed a statistically significant decrease in wound size and an increase in capillary density. Interestingly, in another study using TPOT on 6 patients with leg ulcers of various etiologies, in 1986, Heng et al reported histological signs of endothelial toxicity after 8 weeks of TPOT treatment, which reversed 1 to 2 weeks post‐cessation of the therapy.51 These toxic effects manifested clinically in white areas within the bed of granulation tissue and were attributed to HBOT super‐oxide radical formation, which leads to highly toxic free oxygen radicals.
In order reduce the cost associated with prefabricated TPOT, Agarwal et al used a disposable bag and oxygen cylinder connected to a suction catheter to create a makeshift TPOT delivery device.52 Patients with necrotising soft tissue infections and large post‐traumatic wounds showed improved healing, earlier granulation tissue development and reduced hospital stays with this new technique.52
Leslie et al performed a prospective randomised control study in 28 patients with diabetic foot ulcers (DFUs), 12 of whom received TPOT for 2 weeks.53 No significant difference was seen in the number of microorganisms or healing rates when compared to the controls.53 Upson performed a case study on 2 recalcitrant leg ulcers and found that the use of TPOT with cleanings, debridement and change in positioning improved the rate of wound healing.54
Clinicians have also tested TPOT in combination with other adjunctive modalities. Landau and Schattner studied the combined effect of low‐energy lasers (LEL) and TPOT on DFUs in an open, uncontrolled study. TPOT was administered 2 to 3 times a week, followed by treatment with a helium‐neon low‐energy laser.55 Of the 100 patients who underwent this combined therapy, 81% were cured after 25 ± 13 treatments administered over 3·2 ± 1·7 months.55 A recurrence rate of 4% was found after a median of 18 months.55 Landau et al further studied the combined effect of using LEL and TPOT in 218 patients with DFUs and 156 patients with chronic venous ulcers. Using the same methodology stated above, the combined 347 patients experienced a 78% wound closure rate, leading this group to conclude that TPOT and LEL have a synergistic effect on wound healing.56
Nie et al studied the combined effects of treating deep partial‐thickness burn wounds with basic fibroblast growth factor (bFGF) and TPOT. In their study, 85 patients were assigned to either standard care, TPOT only, bFGF only or combined TPOT and bFGF treatment groups.57 TPOT was used for a total of 90 minutes a day for 3 weeks, and bFGF was applied daily.57 The wound healing rates and the effectiveness of the therapy were significantly improved in the combined TPOT and bFGF group, leading the researchers to conclude that this combined therapy was beneficial.57
In summary, TPOT differs from HBOT in that it localises the delivery of pressurised oxygen to a specific area. As demonstrated above, TPOT appears to have positive effects on wound healing, affecting angiogenesis, collagen formation and re‐epithelialisation rates. Despite these positive findings, certain limitations do exist. For example, the mechanism of TPOT has not been fully substantiated, issues with local wound bed endothelial toxicity have been reported and treatment protocols/setting are not standardised.33, 36, 51 In addition, most TPOT studies are case reports and small non‐randomised clinical studies on treatment‐resistant wounds, which may limit the generalisability of TPOT therapy.36
6. C. TOPICAL CONTINUOUS OXYGEN THERAPY
Another method of TOT is Topical Continuous Oxygen Therapy (TCOT). Unlike TPOT, TCOT does not utilise pressure or require a chamber to be administered.36 Instead, portable units that generate a pure, continuous flow of oxygen for 24 hours a day for 7 days a week are used to promote wound healing for as long as treatment is deemed necessary.36
Kaufman et al was one of the first to use TCOT to treat full‐thickness burns in Guinea pigs. Burns treated with TCOT for 25 days had a high degree of collagen maturation, organisation, synthesis and improved wound healing.58, 59
Furthermore, Banks et al studied the effect of TCOT using Epiflo (Ogenix Corporation, Cleveland, Ohio), which is a portable transdermal oxygen delivery system that takes oxygen from the air, concentrates it to 100% and delivers it to the wound. When Epiflo was applied to 3 patients with stage IV pressure ulcers, there was an improvement in wound healing based on the wound dimensions and volume.60 Similarly, using Epiflo on 9 patients with chronic wounds, Woo et al saw a statistically significant decrease in the mean wound surface area (12.03‐9.60 cm2) and the wound infection checklist score after 4 weeks (5.3‐2.7).61 In contrast, Tracey et al used Epiflo for the treatment of dermal wounds in horses and found no difference in the healing times compared with the controls.62
Finally, Yu et al used TCOT in 9 patients with DFUs and observed a statistically significant decrease in wound size compared with baseline measurements. Moreover, after 8 weeks, 90% of the ulcers in the TCOT group healed in comparison with 30% using standard care.63
In summary, TCOT delivers a continuous supply of oxygen 24 hours a day, 7 days per week at atmospheric pressure. The lack of a pressurised chamber adds to its portability and has allowed engineers to develop TCOT devices ranging anywhere from an occlusive dressing to battery‐operated oxygen extraction units.36 Several small studies demonstrate that TCOT improves wound healing. However, much like TPOT, larger studies are needed to thoroughly investigate and assess its limitations.
7. D. WOUND DRESSINGS AS TOT
Modern wound dressings and coverings that permit the release of oxygen directly to wounds have been developed. For example, Gueldner et al developed a polyoxygenetated aluminium hydroxide compound termed Ox66™ (Hemotek, LLC, Plano, TX, USA. Ox66 organises itself in a true clathrate while capturing oxygen in its lattice.64 Fibroblasts doused in Ox66 facilitate a significant increase in wound closure compared with controls when assessed by scratch assay.64 Toxicity assays using Ox66 show minimal to no toxicity and, at certain doses, improved viability in both previously mentioned cell lines.64 Gueldner et al proposed that Ox66 might serve as a chemical, topical, non‐toxic means of delivering oxygen in a cost‐effective manner to be used in conjunction with other therapies.64
Chandra et al developed a wound dressing film that chemically generates oxygen in situ using sodium percarbonate and calcium peroxide to deliver oxygen to dermal wounds for 3 days.65 They tested the efficacy of their dressing on full‐thickness surgical skin wounds created on 5 to 6‐week‐old pigs.65 After 8 weeks, the wounds treated with this oxygen‐delivery system showed faster wound closure, better reepithelialisation, greater collagen deposition, improved vascularisation and more well‐defined edges.65
Lo et al developed a microfluidic bandage capable of delivering 70% oxygen across a full‐thickness wound in diabetic mice.66 This group did not see any significant difference in the closure rate and angiogenesis between the oxygen‐treated and non‐treated wounds.66 However, collagen maturity was greater in the treated wounds compared to the controls.66 Consequently, their study suggests that oxygen improves collagen maturity but not the total level of collagen when using this microfluidic bandage.66
Furthermore, Paola et al compared the use of a super‐oxidised solution (SOS), which creates oxygen and reactive oxygen species, with a standard treatment (10% povidone iodine solution) in 218 patients with grade II or III foot ulcers. The findings from this study suggest that patients treated with SOS have significantly shorter healing times (43 days vs 55 days for control), fewer bacterial strains in their wound and no adverse effects because of the treatment.67
In addition, experiments performed by Zellner et al on full‐thickness surgical wounds further support the use of dissolved oxygen dressings.68 They applied an oxygen‐dissolved gel or a control oxygen‐free hydrogel to full‐thickness porcine wounds and found that the control wounds failed to heal more often than the treated wounds (6 out of 8 control wounds failed vs 2 out of 8 treated wounds).68 Histologically, the oxygen‐treated wounds displayed significantly less bacterial load, inflammation, dermal fibrosis and necrosis.68
Kellar et al also demonstrated that the topical delivery of oxygen to normal intact skin had histological benefits.69 In this study, patients were asked to apply a topical oxygen solution underneath a wound dressing to one leg while utilising the contralateral leg as a control. At 8 weeks, the legs receiving excess oxygen demonstrated a decrease in inflammatory markers and skin desquamation/roughness and an increase in collagen I and elastin levels.69 A histological examination showed an improved skin texture and hydration when using the solution.69
Ivins et al used Oxzyme: (Oxyzyme™ Sterile Wound Dressing with Iodine; Insense Ltd, UK), an oxygen‐generating hydrogel, for the treatment of 31 patients with venous ulcers over a 6‐week time course.70 The Oxyzyme dressing contains the oxidase enzyme, which reacts with oxygen to produce hydrogen peroxide.70 Hydrogen peroxide serves as carrier of oxygen, and when it reaches the iodine component of the dressing near the wound bed, it is converted into dissolved oxygen. Results of this study should be interpreted with caution, however, as the authors only report on the outcomes of 5 of the patients, all whom experienced complete wound healing using the Oxyzyme dressing.70
TCOT has also been used as a delivery source for the treatment of burns. For example, Almeleh et al used oxygen‐delivering hydrogel (ODH) on 2 patients with second‐degree burns. After treatment, these patients experienced accelerated spontaneous neo‐epithelialisation. This group concluded that autologous epithelial regeneration is stimulated by ODH.71
Finally, topical oxygen delivery methods may also find a use in cosmetic dermatology during postoperative care. Onouye et al studied patients who underwent CO2 laser resurfacing and subjected half of these patients’ faces to an oxygen mist protocol for 5 days, while the other hemiface received a standard occlusive dressing.72 However, they found that only crusting, not wound healing, was improved using the oxygen mist.72
In summary, the classic wound dressing has been updated in many ways to deliver oxygen to tissue. Wound dressings deliver oxygen in different forms, from hydrogels and films to microfluidic bandages. Oxygen‐delivering dressings have different efficacies, with some clearly enhancing the rate of re‐epithelialisation, while others show minimal to no effects. Consequently, wound dressings that deliver oxygen are heterogeneous, and each form has both advantages and limitations that need to be considered with their application.
8. E. TOPICAL OXYGEN EMULSION
Topical oxygen emulsion (TOE) offers a similar ability to deliver oxygen directly to a wound as the dressings discussed above. However, instead of relying on oxygen‐generating materials, an emulsion containing supersaturated oxygen is applied to the wound.73 The emulsion's ability to slowly deliver oxygen over a length of time comes as a result of the perfluorocarbon droplets present in the emulsion that increase the oxygen‐carrying capacity of the solution.73 Similar to the oxygen‐delivering dressings, TOE increases the wound re‐epithelisation rate and improves other wound characteristics (angiogenesis, granulation tissue formation and collagen III deposition).74 Li et al demonstrated all of these effects in a porcine model of partial‐thickness burns.73, 74 Davis et al conducted a similar experiment on second‐degree burns, also showing improved reepithelisation.75 These limited results may support TOE as an adjunct to wound healing.74, 75 However, data on the use of TOE are limited, and more studies are required to determine it efficacy.
9. F. TOPICAL GASEOUS VERSUS TOPICAL DISSOLVED OXYGEN DEVICES
Although comparisons have mostly centred on HBOT vs TPOT in general, topical gaseous and topical dissolved oxygen devices have been compared by Roe et al in 2010. Roe et al used an apparatus with human skin samples to evaluate the depth and magnitude of oxygen penetration from Topical Gaseous Oxygen (TGO) and Topical Dissolved Oxygen (TDO) devices. TGO devices derive 100% oxygen from the air, while TDO devices use a fluid with atmospheric oxygen at 159 mmHg.18 The group found that the TDO device was significantly more effective at transferring oxygen across the human dermis and had a higher peak oxygen level in the tissue compared to TGO devices.18 In addition, TDO had a greater depth of oxygen penetration through the skin (>700 μm) when compared to TGO.18 As gaseous oxygen must overcome the liquid phase before it diffuses into the cell, researchers believe that TDO may be better for treating wounds.18 This comparison is limited to 1 study, and therefore, it requires further investigation to determine whether these conclusions can be generalised.
10. G. MICRO/NANOBUBBLES
MNBs are miniature gaseous voids in fluid. The size cut‐off values for these miniature gas bubbles differ in the literature, with authors classifying microbubbles anywhere between <100 μM and 1 μM and nanobubbles as <1 μM in diameter.76 For the purpose of this review, we will consider microbubbles and nanobubbles as one entity called MNBs. MNBs are used for a wide variety of purposes, including waste water treatment, biofuel production and agriculture.76 In the medical field, their unique properties are harnessed as a contrast agent for ultrasonographic diagnostic scanning as well as drug and gene delivery.77, 78, 79 Recently, there has been an explosion of literature on MNBs; yet, very few studies have looked into their application as an oxygen‐delivery source for wounds as demonstrated by our systematic search, which produced only 12 relevant papers.
The properties that make MNBs unique and contribute to their versatility are their stability in water, slow rise, gradual shrinkage and collapse.76 Because their internal pressure is much higher than their local environment, MNBs promote the solubility of their internal gas, often oxygen, into the liquid in which they are produced.76 When MNBs are produced in water using oxygen or air as gas impregnated in their core, their negatively charged surface not only prevents them from merging together, which would cause them to lose their properties, but also attracts particulate matter and assists in the removal of debris.76 In addition, MNBs generate free radicals as they shrink, which potentially contributes to their antibacterial effects.80, 81 Nanobubbles and microbubbles that shrink to become nanobubbles exist in a solution for different time periods depending on the conditions in which they are formed.82
Upon a review of the literature, several studies using MNBs for application in respiratory compromise were found. For example, Feshitan et al demonstrated 100% survival for at least 2 hours using oxygen microbubbles for the peritoneal oxygenation of rats experiencing acute lung trauma.83 Given the effectiveness and safety of microbubble oxygenation, Feshitan et al proposed using oxygen microbubbles as a therapy for acute respiratory distress syndrome.83 Lundgren et al showed that the use of intravascular microbubbles generated by an I.V. infusion of 2% dodecafluoropentane enhanced the denitrogenation of oxygen‐breathing pigs compared with the controls.84 Lundgren et al proposed that treating decompression sickness using microbubbles and oxygen improves the hypoxia caused by the decompression bubbles.84 Similarly, Tyssebotn et al showed that infused dodecafluoropentane forming oxygen microbubbles improved hypoxia in pigs with right‐to‐left shunts.85 Most importantly, the oxygenation lasted for 2 hours post‐infusion, and no signs of toxicity or pulmonary air embolisation were observed.85
MNBs have also been used for the purpose of total liquid ventilation. In a study conducted by Kakiuchi et al, saline‐based MNBs were used in the total liquid ventilation of anaesthetised rats.86 This group achieved an oxygen content of 45 mg/L using MNBs, which is nearly 8 times the dissolved oxygen content of normal saline (6 mg/L).86 Although the 40 minutes increase in life span using the MNBs was considered impractical in application, Kakiuchi et al are working on solutions with denser MNBs, which they believe can significantly increase life span during total liquid ventilation.86
Matsuki et al was the first to apply MNBs to common infusions in daily medical care.76, 80 They demonstrated that saline is the preferred solution for MNBs and that MNBs formed in dextran showed the highest pO2 at 60 minutes.80 Finally, their results suggest that MNBs in normal saline (pO2 1060 mmHg) improved the pO2 in blood under hypoxic conditions.80
Although literature on the use of MNBs as a topical agent for tissue oxygenation is sparse, several important studies exist that demonstrate its utility. Studies performed by a number of groups demonstrate the topical delivery of microbubbles in vivo to mice, showing a significant increase in the delivery of oxygen to hypoxic skin.87 At the cellular level, nanobubbles not only counteract the hypoxia‐dependent dysregulation of the MMP/TIMP balance in human keratinocytes but also reduce HIF‐1‐alpha signalling, both of which have important implications in wound healing. This may relate to the intermittent application of the technology that prevents a switch off of the hypoxic drive induced by a constant flow of oxygen.88, 89, 90 In addition, through florescent labelling, researchers show that nanobubbles are internalised by cells, including keratinocytes, with a diffuse cytoplasmic distribution.89, 90, 91 Furthermore, studies looking into potential adverse effects of MNBs found no associated cellular toxicity with their application.87, 88, 89, 90
Recent advances in nanotechnology have allowed researchers to modify MNBs, increasing their stability, improving their oxygen delivery and adding additional properties that make them even more suitable for applications in wound therapy.87 Many groups show that the modification of nanobubbles by adding Perfluoropentane (PFP) or 2H, 3H‐decafluoropentane (DFP) to their oxygenated cores and chitosan or dextran to the nanobubble shells increases the amount and the duration of oxygen released to hypoxic tissue.88, 92 In addition, the use of Chitosan in nanobubble shells has healing capabilities as well as anti‐cancer and anti‐microbial properties.93, 94, 95, 96 The net positive charge that the nanobubbles attain, with the addition of chitosan, is also favourable to the skin due to its anionic nature.97, 98 Furthermore, compounds such as PFP and DFP, which are added to the nanobubble core, are inert and non‐toxic to normal human cells.92
Interestingly, ultrasound improves the gas delivery kinetics by nanobubble cavitation and increases the skin's permeability to MNBs through sonophoresis.87, 99, 100, 101, 102, 103 In vitro and in vivo studies demonstrate that an ultrasound improves the ability of both liquid‐ and gel‐coated nanobubble formulations to deliver oxygen through the skin.87, 91 Furthermore, studies investigating liquid vs gel formulations of coated nanobubbles result in higher and longer‐lasting oxygen levels in the tissue using either preparation when compared with the controls. However, compared to the liquid formulations, the gel formulations have lower net oxygen levels and faster release dynamics, which is attributed to the gel preparation process.87
One important theoretical concern that exists for MNB therapy is the potential for a gas embolism, especially after intravenous infusion of the MNBs. However, this concern would be significantly less with their local tissue application.
11. DISCUSSION
Chronic non‐healing wounds constitute a major health problem, amounting up to 15 billion dollars in US health care costs.22 Perturbations in oxygen delivery and its content in tissue are identified as major factors in non‐healing wounds. Local tissue hypoxia is ever so common in wounds and is related to health problems, such as diabetes, venous stasis, and peripheral vascular disease and when coupled with ischemia reperfusion cycles, can lead to even further tissue damage.104 Thus, maintaining an appropriate level of oxygen in these wounds should be a therapeutic goal for clinicians.105
Since the advent of HBOT in 1663, clinicians have sought to improve and maintain oxygen levels in tissue. Therapies such as HBOT and TOT were developed in an attempt to accelerate wound healing. However, both HBOT and TOT have many challenges, including serious complications, cellular toxicity, and the lack of a standardised protocol for administration.5, 106 Furthermore, HBOT requires continuous monitoring, the technology and the equipment are costly and not portable, and the efficacy is limited to certain wounds. On the other hand, TOT has a restricted ability to penetrate the skin, and endothelial toxicity is reported with its use. Finally, TOE, which is a promising new technique that delivers emulsion‐containing supersaturated oxygen to wounds, is dependent on a synthetically manufactured dressing that cannot yet be used as an irrigation solution and continues to face technological challenges.106 Clearly, advances in the delivery of oxygen to wounds have been made, but there are still many aspects of this therapy that require improvement.
MNBs offer a new technology for creating oxygen‐enriched fluids that can bypass many of the issues associated with HBOT and TOT. Using MNB technology, fluids can be saturated with various gases such as oxygen. MNBs have several unique properties as follows: (1) they can remain in an aqueous solution for extended periods of time; (2) they serve as a reservoir for oxygen; and (3) their charged surface attracts debris from the local environment.80, 107 For these reasons, interest in MNBs is growing, yet their implementation in medicine is limited. Given their ability to carry oxygen and their stability in solution, MNBs are an excellent vehicle for external oxygen delivery to tissues, and their application in wound healings still remains to be fully explored.87
Furthermore, recent research suggests that bacterial biofilm and leukocytes consume oxygen in chronic wounds, depleting oxygen from the specialised cells involved in orchestrating wound healing.108 Likewise, anaerobes that flourish in this oxygen‐depleted state are increasingly pathological and resistant to antibacterial treatments.109 To address these issues, surface modifications to MNBs, such as changes to the surface charge, can be engineered to increase the interaction and penetration of MNBs into biofilm.110 Once in the biofilm, MNBs produce reactive oxidative species and can deliver antibiotics and oxygen impregnated in their cores, all of which help fight infection.81
Interestingly, preliminary work performed at our lab at the UC Irvine Center for Tissue Engineering has not only shown that MNBs can hold significantly more oxygen than the controls but also that the high oxygen content of MNB fluid can be sustained over hours (Figure 3). In addition, when exposing adipose tissue to MNBs for 15 to 30 minutes, there is a statistically significant reduction in oxygen radicals, which we attribute to a lesser degree of hypoxia compared to the controls. We are currently working on translating these in vitro results by assessing fat graft retention rates using MNB‐soaked lipoaspirate in a murine model. Our lab is also currently investigating the application of MNBs through the use of our patent‐pending technology for wound healing, as well as for the purposes of tissue transplantation and replantation.
Figure 3.
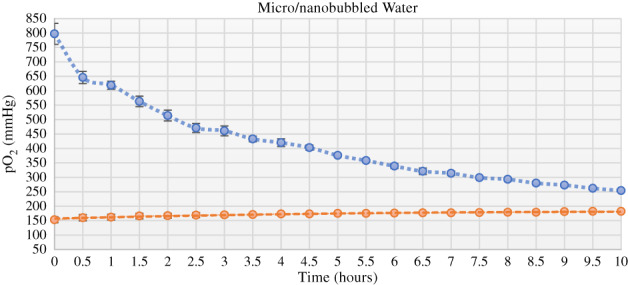
pO2 measurements of micro/nanobubbles (blue) with oxygen‐impregnated core made in water compared with control (orange) composed of dissolved oxygen gas in water. pO2 measured in solution using (ABL800 FLEX Radiometer Medical ApS, Copenhagen, Denmark)
We hypothesise that the MNB technology is a novel and promising alternative to hyperbaric and topical oxygen therapy for the treatment of wounds. With the advent of nanotechnology, the surface and core of MNBs can be modified to increase their stability, efficacy and ability to interact with biofilm. The increased stability of MNBs in solution may allow for the production of off‐the‐shelf solutions and gel formulations. Finally, when used to irrigate wounds, we believe MNBs solutions would: (1) increase wound oxygen tension, (2) provide debridement, and (3) promote healing. Consequently, MNBs may provide an inexpensive and simple method for improving oxygen delivery to wounds that can be use in wards or the clinic without the cost and risks associated with current methods of oxygen delivery.
Sayadi LR, Banyard DA, Ziegler ME, et al. Topical oxygen therapy & micro/nanobubbles: a new modality for tissue oxygen delivery. Int Wound J. 2018;15:363–374. 10.1111/iwj.12873
REFERENCES
- 1. Rodriguez PG, Felix FN, Woodley DT, Shim EK. The role of oxygen in wound healing: a review of the literature. Dermatol Surg. 2008;34(9):1159‐1169. [DOI] [PubMed] [Google Scholar]
- 2. Sheikh AY, Rollins MD, Hopf HW, Hunt TK. Hyperoxia improves microvascular perfusion in a murine wound model. Wound Repair Regen. 2005;13(3):303‐308. [DOI] [PubMed] [Google Scholar]
- 3. Sundaresan M, Yu ZX, Ferrans VJ, Irani K, Finkel T. Requirement for generation of H2O2 for platelet‐derived growth factor signal transduction. Science. 1995;270(5234):296‐299. [DOI] [PubMed] [Google Scholar]
- 4. Allen DB, Maguire JJ, Mahdavian M, et al. Wound hypoxia and acidosis limit neutrophil bacterial killing mechanisms. Arch Surg. 1997;132(9):991‐996. [DOI] [PubMed] [Google Scholar]
- 5. Gordillo GM, Sen CK. Revisiting the essential role of oxygen in wound healing. Am J Surg. 2003;186(3):259‐263. [DOI] [PubMed] [Google Scholar]
- 6. Dimitrijevich SD, Paranjape S, Wilson JR, Gracy RW, Mills JG. Effect of hyperbaric oxygen on human skin cells in culture and in human dermal and skin equivalents. Wound Repair Regen. 1999;7(1):53‐64. [DOI] [PubMed] [Google Scholar]
- 7. Conconi MT, Baiguera S, Guidolin D, Furlan C, Menti AM, Vigolo S. Effects of hyperbaric oxygen on proliferative and apoptotic activities and reactive oxygen species generation in mouse fibroblast 3T3/J2 cell line. J Investig Med. 2003;51(4):227‐232. [DOI] [PubMed] [Google Scholar]
- 8. Tompach PC, Lew D, Stoll JL. Cell response to hyperbaric oxygen treatment. Int J Oral Maxillofac Surg. 1997;26(2):82‐86. [DOI] [PubMed] [Google Scholar]
- 9. Kairuz E, Upton Z, Dawson RA, Malda J. Hyperbaric oxygen stimulates epidermal reconstruction in human skin equivalents. Wound Repair Regen. 2007;15(2):266‐274. [DOI] [PubMed] [Google Scholar]
- 10. Roy S, Khanna S, Wallace WA, et al. Characterization of perceived hyperoxia in isolated primary cardiac fibroblasts and in the reoxygenated heart. J Biol Chem. 2003;278(47):47129‐47135. [DOI] [PubMed] [Google Scholar]
- 11. Hopf HW, Gibson JJ, Angeles AP, et al. Hyperoxia and angiogenesis. Wound Repair Regen. 2005;13(6):558‐564. [DOI] [PubMed] [Google Scholar]
- 12. Prockop DJ, Kivirikko KI, Tuderman L, Guzman NA. The biosynthesis of collagen and its disorders (first of two parts). N Engl J Med. 1979;301(1):13‐23. [DOI] [PubMed] [Google Scholar]
- 13. Tuderman L, Myllyla R, Kivirikko KI. Mechanism of the prolyl hydroxylase reaction. 1. Role of co‐substrates. Eur J Biochem. 1977;80(2):341‐348. [DOI] [PubMed] [Google Scholar]
- 14. Sheikh AY, Gibson JJ, Rollins MD, Hopf HW, Hussain Z, Hunt TK. Effect of hyperoxia on vascular endothelial growth factor levels in a wound model. Arch Surg. 2000;135(11):1293‐1297. [DOI] [PubMed] [Google Scholar]
- 15. Patel V, Chivukula IV, Roy S, et al. Oxygen: from the benefits of inducing VEGF expression to managing the risk of hyperbaric stress. Antioxid Redox Signal. 2005;7(9–10):1377‐1387. [DOI] [PubMed] [Google Scholar]
- 16. Sheffield P. Tissue oxygen measurements with respect to soft tissue wound healing with normobaric and hyperbaric oxygen. HBO Rev. 1985;6:18‐46. [Google Scholar]
- 17. Heng MC, Harker J, Bardakjian VB, Ayvazian H. Enhanced healing and cost‐effectiveness of low‐pressure oxygen therapy in healing necrotic wounds: a feasibility study of technology transfer. Ostomy Wound Manage. 2000;46(3):52‐60. 62. [PubMed] [Google Scholar]
- 18. Roe DF, Gibbins BL, Ladizinsky DA. Topical dissolved oxygen penetrates skin: model and method. J Surg Res. 2010;159(1):e29‐e36. [DOI] [PubMed] [Google Scholar]
- 19. Penneys R, Felder W, Christophers E. The passage of oxygen through isolated sheets of human stratum corneum. Proc Soc Exp Biol Med. 1968;127(4):1020‐1022. [DOI] [PubMed] [Google Scholar]
- 20. Gruber RP, Heitkamp DH, Billy LJ, Amato JJ. Skin permeability to oxygen and hyperbaric oxygen. Arch Surg. 1970;101(1):69‐70. [DOI] [PubMed] [Google Scholar]
- 21. Stucker M, Struk A, Altmeyer P, et al. The cutaneous uptake of atmospheric oxygen contributes significantly to the oxygen supply of human dermis and epidermis. J Physiol. 2002;538(Pt 3):985‐994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Markova A, Mostow EN. US skin disease assessment: ulcer and wound care. Dermatol Clin. 2012;30(1):107‐111. ix. [DOI] [PubMed] [Google Scholar]
- 23. Kindwall EP, Kindwall WH. A history of hyperbaric medicine. Hyperbaric Medicine Practice. 2nd ed. Flagstaff, Ariz: Best Publishing; 2002. [Google Scholar]
- 24. Sen CK, Khanna S, Gordillo G, et al. Oxygen, oxidants, and antioxidants in wound healing: an emerging paradigm. Ann N Y Acad Sci. 2002;957:239‐249. [DOI] [PubMed] [Google Scholar]
- 25. Wasiak J, Bennett M, Cleland HJ. Hyperbaric oxygen as adjuvant therapy in the management of burns: can evidence guide clinical practice? Burns. 2006;32(5):650‐652. [DOI] [PubMed] [Google Scholar]
- 26. UHMS . In: MD LW, ed. Hyperbaric Oxygen Therapy Indications. 13th ed. North Palm Beach, Florida: Best Publishing; 2014. [Google Scholar]
- 27. Heyboer M 3rd, Milovanova TN, Wojcik S, et al. CD34+/CD45‐dim stem cell mobilization by hyperbaric oxygen – changes with oxygen dosage. Stem Cell Res. 2014;12(3):638‐645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Tiaka EK, Papanas N, Manolakis AC, Maltezos E. The role of hyperbaric oxygen in the treatment of diabetic foot ulcers. Angiology. 2012;63(4):302‐314. [DOI] [PubMed] [Google Scholar]
- 29. Thackham JA, McElwain DL, Long RJ. The use of hyperbaric oxygen therapy to treat chronic wounds: a review. Wound Repair Regen. 2008;16(3):321‐330. [DOI] [PubMed] [Google Scholar]
- 30. Thom SR. Hyperbaric oxygen – its mechanisms and efficacy. Plast Reconstr Surg. 2011;127(Suppl 1):131s‐141s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Gothard L, Haviland J, Bryson P, et al. Randomised phase II trial of hyperbaric oxygen therapy in patients with chronic arm lymphoedema after radiotherapy for cancer. Radiother Oncol. 2010;97(1):101‐107. [DOI] [PubMed] [Google Scholar]
- 32. Heng MC. Topical hyperbaric therapy for problem skin wounds. J Dermatol Surg Oncol. 1993;19(8):784‐793. [DOI] [PubMed] [Google Scholar]
- 33. Feldmeier JJ, Hopf HW, Warriner RA 3rd, Fife CE, Gesell LB, Bennett M. UHMS position statement: topical oxygen for chronic wounds. Undersea Hyperb Med. 2005;32(3):157‐168. [PubMed] [Google Scholar]
- 34. Silver I. The measurement of oxygen tension in healing tissue. Prog Respir Res. 1969;3:124‐135. [Google Scholar]
- 35. Medical devices; reclassification of the topical oxygen chamber for extremities. Final rule. Fed Regist. 2011;76(79):22805‐22807. [PubMed] [Google Scholar]
- 36. Dissemond J, Kröger K, Storck M, Risse A, Engels P. Topical oxygen wound therapies for chronic wounds: a review. J Wound Care. 2015;24(2):53‐60. [DOI] [PubMed] [Google Scholar]
- 37. Mutluoglu M, Cakkalkurt A, Uzun G, Aktas S. Topical oxygen for chronic wounds: a PRO/CON debate. J Am Coll Clin Wound Spec. 2013;5(3):61‐65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Fries RB, Wallace WA, Roy S, et al. Dermal excisional wound healing in pigs following treatment with topically applied pure oxygen. Mutat Res. 2005;579(1–2):172‐181. [DOI] [PubMed] [Google Scholar]
- 39. Gordillo GM, Sen CK. Evidence‐based recommendations for the use of topical oxygen therapy in the treatment of lower extremity wounds. Int J Low Extrem Wounds. 2009;8(2):105‐111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Rao C, Xiao L, Liu H, et al. Effects of topical oxygen therapy on ischemic wound healing. J Phys Ther Sci. 2016;28(1):118‐123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Gordillo GM, Roy S, Khanna S, et al. Topical oxygen therapy induces vascular endothelial growth factor expression and improves closure of clinically presented chronic wounds. Clin Exp Pharmacol Physiol. 2008;35(8):957‐964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Gordillo GM, Schlanger R, Wallace WA, et al. Protocols for topical and systemic oxygen treatments in wound healing. Methods Enzymol. 2004;381:575‐585. [DOI] [PubMed] [Google Scholar]
- 43. Blackman E, Moore C, Hyatt J, Railton R, Frye C. Topical wound oxygen therapy in the treatment of severe diabetic foot ulcers: a prospective controlled study. Ostomy Wound Manage. 2010;56(6):24‐31. [PubMed] [Google Scholar]
- 44. Tawfick W, Sultan S. Does topical wound oxygen (TWO2) offer an improved outcome over conventional compression dressings (CCD) in the management of refractory venous ulcers (RVU)? A parallel observational comparative study. Eur J Vasc Endovasc Surg. 2009;38(1):125‐132. [DOI] [PubMed] [Google Scholar]
- 45. Tawfick WA, Sultan S. Technical and clinical outcome of topical wound oxygen in comparison to conventional compression dressings in the management of refractory nonhealing venous ulcers. Vasc Endovasc Surg. 2013;47(1):30‐37. [DOI] [PubMed] [Google Scholar]
- 46. Kalliainen LK, Gordillo GM, Schlanger R, Sen CK. Topical oxygen as an adjunct to wound healing: a clinical case series. Pathophysiology. 2003;9(2):81‐87. [DOI] [PubMed] [Google Scholar]
- 47. Copeland K, Purvis AR. A retrospective chart review of chronic wound patients treated with topical oxygen therapy. Adv Wound Care. 2017;6(5):143‐152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Fischer BH. Topical hyperbaric oxygen treatment of pressure sores and skin ulcers. Lancet. 1969;2(7617):405‐409. [DOI] [PubMed] [Google Scholar]
- 49. Ignacio DR, Pavot AP, Azer RN, Wisotsky L. Topical oxygen therapy treatment of extensive leg and foot ulcers. J Am Podiatr Med Assoc. 1985;75(4):196‐199. [DOI] [PubMed] [Google Scholar]
- 50. Heng MC, Harker J, Csathy G, et al. Angiogenesis in necrotic ulcers treated with hyperbaric oxygen. Ostomy Wound Manage. 2000;46(9):18‐28. 30‐2. [PubMed] [Google Scholar]
- 51. Heng MC, Kloss SG. Endothelial cell toxicity in leg ulcers treated with topical hyperbaric oxygen. Am J Dermatopathol. 1986;8(5):403‐410. [DOI] [PubMed] [Google Scholar]
- 52. Agarwal V, Aroor S, Gupta N, Gupta A, Agarwal N, Kaur N. New technique of applying topical oxygen therapy as a cost‐effective procedure. Indian J Surg. 2015;77(Suppl 3):1456‐1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Leslie CA, Sapico FL, Ginunas VJ, Adkins RH. Randomized controlled trial of topical hyperbaric oxygen for treatment of diabetic foot ulcers. Diabetes Care. 1988;11(2):111‐115. [DOI] [PubMed] [Google Scholar]
- 54. Upson AV. Topical hyperbaric oxygenation in the treatment of recalcitrant open wounds. A clinical report. Phys Ther. 1986;66(9):1408‐1412. [DOI] [PubMed] [Google Scholar]
- 55. Landau Z, Schattner A. Topical hyperbaric oxygen and low energy laser therapy for chronic diabetic foot ulcers resistant to conventional treatment. Yale J Biol Med. 2001;74(2):95‐100. [PMC free article] [PubMed] [Google Scholar]
- 56. Landau Z, Sommer A, Miller EB. Topical hyperbaric oxygen and low‐energy laser for the treatment of chronic ulcers. Eur J Intern Med. 2006;17(4):272‐275. [DOI] [PubMed] [Google Scholar]
- 57. Nie K, Li P, Zeng X, et al. Clinical observation of basic fibroblast growth factor combined with topical oxygen therapy in enhancing burn wound healing. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2010;24(6):643‐646. [PubMed] [Google Scholar]
- 58. Kaufman T, Alexander JW, Nathan P, Brackett KA, MacMillan BG. The microclimate chamber: the effect of continuous topical administration of 96% oxygen and 75% relative humidity on the healing rate of experimental deep burns. J Trauma. 1983;23(9):806‐815. [PubMed] [Google Scholar]
- 59. Kaufman T, Brackett KA, Alexander JW, MacMillan BG. The microclimate chamber: morphological aspects of experimental deep burns treated topically with continuous humidified oxygen. Ann Plast Surg. 1983;10(3):218‐223. [DOI] [PubMed] [Google Scholar]
- 60. Banks PG, Ho CH. A novel topical oxygen treatment for chronic and difficult‐to‐heal wounds: case studies. J Spinal Cord Med. 2008;31(3):297‐301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Woo KY, Coutts PM, Sibbald RG. Continuous topical oxygen for the treatment of chronic wounds: a pilot study. Adv Skin Wound Care. 2012;25(12):543‐547. [DOI] [PubMed] [Google Scholar]
- 62. Tracey AK, Alcott CJ, Schleining JA, et al. The effects of topical oxygen therapy on equine distal limb dermal wound healing. Can Vet J. 2014;55(12):1146‐1152. [PMC free article] [PubMed] [Google Scholar]
- 63. Yu J, Lu S, McLaren AM, Perry JA, Cross KM. Topical oxygen therapy results in complete wound healing in diabetic foot ulcers. Wound Repair Regen. 2016;24(6):1066‐1072. [DOI] [PubMed] [Google Scholar]
- 64. Gueldner J, Zhang F, Zechmann B, Bruce ED. Evaluating a novel oxygenating therapeutic for its potential use in the advancement of wound healing. Toxicol In Vitro. 2017;43:62‐68. [DOI] [PubMed] [Google Scholar]
- 65. Chandra PK, Ross CL, Smith LC, et al. Peroxide‐based oxygen generating topical wound dressing for enhancing healing of dermal wounds. Wound Repair Regen. 2015;23(6):830‐841. [DOI] [PubMed] [Google Scholar]
- 66. Lo JF, Brennan M, Merchant Z, et al. Microfluidic wound bandage: localized oxygen modulation of collagen maturation. Wound Repair Regen. 2013;21(2):226‐234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Paola LB, Senesi E, Merico A, Vido M, Assaloni D, DaRos R. Super‐oxidized solution (SOS) therapy for infected diabetic foot ulcers. Wounds. 2006;18(9):262‐270. [Google Scholar]
- 68. Zellner S, Manabat R, Roe DF. A dissolved oxygen dressing: a pilot study in an ischemic skin flap model. J Int Med Res. 2015;43(1):93‐103. [DOI] [PubMed] [Google Scholar]
- 69. Kellar RS, Audet RG, Roe DF, Rheins LA, Draelos ZD. Topically delivered dissolved oxygen reduces inflammation and positively influences structural proteins in healthy intact human skin. J Cosmet Dermatol. 2013;12(2):86‐95. [DOI] [PubMed] [Google Scholar]
- 70. Ivins N, Simmonds W, Turner A, Harding K. The use of an oxygenating hydrogel dressing in VLU. Wounds. 2007;3:1‐5. [Google Scholar]
- 71. Almeleh R. Spontaneous accelerated epithelialization in deep dermal burns using an oxygen‐delivering hydrogel: a report of two cases. Wounds. 2013;25(10):E18‐E25. [Google Scholar]
- 72. Onouye T, Menaker G, Christian M, Moy R. Occlusive dressing versus oxygen mist therapy following CO2 laser resurfacing. Dermatol Surg. 2000;26(6):572‐576. [DOI] [PubMed] [Google Scholar]
- 73. Li J, Zhang YP, Zarei M, et al. A topical aqueous oxygen emulsion stimulates granulation tissue formation in a porcine second‐degree burn wound. Burns. 2015;41(5):1049‐1057. [DOI] [PubMed] [Google Scholar]
- 74. Li J, Ollague Sierra J, Zhu L, et al. Effects of a topical aqueous oxygen emulsion on collagen deposition and angiogenesis in a porcine deep partial‐thickness wound model. Exp Dermatol. 2013;22(10):674‐676. [DOI] [PubMed] [Google Scholar]
- 75. Davis SC, Cazzaniga AL, Ricotti C, et al. Topical oxygen emulsion: a novel wound therapy. Arch Dermatol. 2007;143(10):1252‐1256. [DOI] [PubMed] [Google Scholar]
- 76. Matsuki N, Ichiba S, Ishikawa T, et al. Blood oxygenation using microbubble suspensions. Eur Biophys J. 2012;41(6):571‐578. [DOI] [PubMed] [Google Scholar]
- 77. Badea R, Seicean A, Diaconu B, et al. Contrast‐enhanced ultrasound of the pancreas‐‐a method beyond its potential or a new diagnostic standard? J Gastrointestin Liver Dis. 2009;18(2):237‐242. [PubMed] [Google Scholar]
- 78. Dijkmans PA, Juffermans LJ, Musters RJ, et al. Microbubbles and ultrasound: from diagnosis to therapy. Eur J Echocardiogr. 2004;5(4):245‐256. [DOI] [PubMed] [Google Scholar]
- 79. Juffermans LJ, Dijkmans PA, Musters RJ, et al. Local drug and gene delivery through microbubbles and ultrasound. Neth Hear J. 2004;12(9):394‐399. [PMC free article] [PubMed] [Google Scholar]
- 80. Matsuki N, Ishikawa T, Ichiba S, Shiba N, Ujike Y, Yamaguchi T. Oxygen supersaturated fluid using fine micro/nanobubbles. Int J Nanomedicine. 2014;9:4495‐4505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Takahashi M, Chiba K, Li P. Free‐radical generation from collapsing microbubbles in the absence of a dynamic stimulus. J Phys Chem B. 2007;111(6):1343‐1347. [DOI] [PubMed] [Google Scholar]
- 82. Swanson EJ, Mohan V, Kheir J, Borden MA. Phospholipid‐stabilized microbubble foam for injectable oxygen delivery. Langmuir. 2010;26(20):15726‐15729. [DOI] [PubMed] [Google Scholar]
- 83. Feshitan JA, Legband ND, Borden MA, Terry BS. Systemic oxygen delivery by peritoneal perfusion of oxygen microbubbles. Biomaterials. 2014;35(9):2600‐2606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Lundgren C, Bergoe G, Olszowka A, Tyssebotn I. Tissue nitrogen elimination in oxygen‐breathing pigs is enhanced by fluorocarbon‐derived intravascular micro‐bubbles. Undersea Hyperb Med. 2005;32(4):215‐226. [PubMed] [Google Scholar]
- 85. Tyssebotn IM, Lundgren CEG, Olszowka AJ, Bergoe GW. Hypoxia due to shunts in pig lung treated with O2 and fluorocarbon‐derived intravascular microbubbles. Artif Cells Blood Substit Immobil Biotechnol. 2010;38(2):79‐89. [DOI] [PubMed] [Google Scholar]
- 86. Kakiuchi K, Matsuda K, Harii N, Sou K, Aoki J, Takeoka S. Establishment of a total liquid ventilation system using saline‐based oxygen micro/nano‐bubble dispersions in rats. J Artif Organs. 2015;18(3):220‐227. [DOI] [PubMed] [Google Scholar]
- 87. Magnetto C, Prato M, Khadjavi A, et al. Ultrasound‐activated decafluoropentane‐cored and chitosan‐shelled nanodroplets for oxygen delivery to hypoxic cutaneous tissues. RSC Adv. 2014;4(72):38433‐38441. [Google Scholar]
- 88. Bisazza A, Giustetto P, Rolfo A, et al. Microbubble‐mediated oxygen delivery to hypoxic tissues as a new therapeutic device. Conf Proc IEEE Eng Med Biol Soc. 2008;2008:2067‐2070. [DOI] [PubMed] [Google Scholar]
- 89. Khadjavi A, Magnetto C, Panariti A, et al. Chitosan‐shelled oxygen‐loaded nanodroplets abrogate hypoxia dysregulation of human keratinocyte gelatinases and inhibitors: new insights for chronic wound healing. Toxicol Appl Pharmacol. 2015;286(3):198‐206. [DOI] [PubMed] [Google Scholar]
- 90. Basilico N, Magnetto C, D'Alessandro S, et al. Dextran‐shelled oxygen‐loaded nanodroplets reestablish a normoxia‐like pro‐angiogenic phenotype and behavior in hypoxic human dermal microvascular endothelium. Toxicol Appl Pharmacol. 2015;288(3):330‐338. [DOI] [PubMed] [Google Scholar]
- 91. Cavalli R, Bisazza A, Giustetto P, et al. Preparation and characterization of dextran nanobubbles for oxygen delivery. Int J Pharm. 2009;381(2):160‐165. [DOI] [PubMed] [Google Scholar]
- 92. Prato M, Magnetto C, Jose J, et al. 2H,3H‐decafluoropentane‐based nanodroplets: new perspectives for oxygen delivery to hypoxic cutaneous tissues. PLoS One. 2015;10(3):e0119769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Cho YW, Cho YN, Chung SH, Yoo G, Ko SW. Water‐soluble chitin as a wound healing accelerator. Biomaterials. 1999;20(22):2139‐2145. [DOI] [PubMed] [Google Scholar]
- 94. Je JY, Cho YS, Kim SK. Cytotoxic activities of water‐soluble chitosan derivatives with different degree of deacetylation. Bioorg Med Chem Lett. 2006;16(8):2122‐2126. [DOI] [PubMed] [Google Scholar]
- 95. Harkins AL, Duri S, Kloth LC, Tran CD. Chitosan‐cellulose composite for wound dressing material. Part 2. Antimicrobial activity, blood absorption ability, and biocompatibility. J Biomed Mater Res B Appl Biomater. 2014;102(6):1199‐1206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Lee DS, Jeong SY, Kim YM, Lee MS, Ahn CB, Je JY. Antibacterial activity of aminoderivatized chitosans against methicillin‐resistant Staphylococcus aureus (MRSA). Bioorg Med Chem. 2009;17(20):7108‐7112. [DOI] [PubMed] [Google Scholar]
- 97. Wu X, Landfester K, Musyanovych A, Guy RH. Disposition of charged nanoparticles after their topical application to the skin. Skin Pharmacol Physiol. 2010;23(3):117‐123. [DOI] [PubMed] [Google Scholar]
- 98. Chen Y, Zhou L, Yuan L, Zhang ZH, Liu X, Wu Q. Formulation, characterization, and evaluation of in vitro skin permeation and in vivo pharmacodynamics of surface‐charged tripterine‐loaded nanostructured lipid carriers. Int J Nanomedicine. 2012;7:3023‐3032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Kogan P, Gessner RC, Dayton PA. Microbubbles in imaging: applications beyond ultrasound. Bubble Sci Eng Technol. 2010;2(1):3‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Bang JH, Suslick KS. Applications of ultrasound to the synthesis of nanostructured materials. Adv Mater. 2010;22(10):1039‐1059. [DOI] [PubMed] [Google Scholar]
- 101. Torchilin V. Multifunctional and stimuli‐sensitive pharmaceutical nanocarriers. Eur J Pharm Biopharm. 2009;71(3):431‐444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Liu Y, Miyoshi H, Nakamura M. Encapsulated ultrasound microbubbles: therapeutic application in drug/gene delivery. J Control Release. 2006;114(1):89‐99. [DOI] [PubMed] [Google Scholar]
- 103. Postema M, Gilja OH. Contrast‐enhanced and targeted ultrasound. World J Gastroenterol. 2011;17(1):28‐41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Hinck D, Franke A, Gatzka F. Use of vacuum‐assisted closure negative pressure wound therapy in combat‐related injuries‐literature review. Mil Med. 2010;175(3):173‐181. [DOI] [PubMed] [Google Scholar]
- 105. Couch KS, Stojadinovic A. Negative‐pressure wound therapy in the military: lessons learned. Plast Reconstr Surg. 2011;127(1):117s‐130s. [DOI] [PubMed] [Google Scholar]
- 106. Maeda Y, Hosokawa S, Baba Y, Tomiyama A, Ito Y. Generation mechanism of micro‐bubbles in a pressurized dissolution method. Exp Thermal Fluid Sci. 2015;60:201‐207. [Google Scholar]
- 107. Ushikubo FY, Furukawa T, Nakagawa R, et al. Evidence of the existence and the stability of nano‐bubbles in water. Colloid Surf. 2010;361(1–3):31‐37. [Google Scholar]
- 108. James GA, Ge Zhao A, Usui M, et al. Microsensor and transcriptomic signatures of oxygen depletion in biofilms associated with chronic wounds. Wound Repair Regen. 2016;24(2):373‐383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Fair RJ, Tor Y. Antibiotics and bacterial resistance in the 21st century. Perspect Med Chem. 2014;6:25‐64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Ikuma K, Decho AW, Lau BL. When nanoparticles meet biofilms‐interactions guiding the environmental fate and accumulation of nanoparticles. Front Microbiol. 2015;6:591. [DOI] [PMC free article] [PubMed] [Google Scholar]


