Abstract
The PTH/PTHrP receptor (PTHR1) mediates the actions of parathyroid hormone (PTH) and PTH-related peptide (PTHrP) by coupling this G protein-coupled receptor (GPCR) to the alpha-subunit of the heterotrimeric stimulatory G protein (Gsα) and thereby to the formation of cAMP. In growth plates, PTHrP-dependent activation of the cAMP/PKA second messenger pathway prevents the premature differentiation of chondrocytes into hypertrophic cells resulting in delayed growth plate closure. Heterozygous mutations in GNAS, the gene encoding Gsα, lead to a reduction in cAMP levels in growth plate chondrocytes that is sufficient to cause shortening of metacarpals and/or -tarsals, i. e. typical skeletal aspects of Albright’s Hereditary Osteodystrophy (AHO). However, heterozygous mutations in other genes, including those encoding PTHrP, PRKAR1A, PDE4D, and PDE3A, can lead to similar or even more pronounced acceleration of skeletal maturation that is particularly obvious in hands and feet, and reduces final adult height. Genetic mutations other than those resulting in Gsα haploinsufficiency thus reduce intracellular cAMP levels in growth plate chondrocytes to a similar extent and thereby accelerate skeletal maturation.
Keywords: PTH, PTHrP, PTH/PTHrP receptor, GNAS, alpha-subunit of the stimulatory G protein, genetic disease, acrodysostosis, cAMP signaling pathway, PRKAR1A, PDE4D, PDE3A, HDAC4
Introduction
Shortening of one or several metacarpals and/or -tarsals, a well-established aspect of Albright’s Hereditary Osteodystrophy (AHO), is frequently associated with genetic defects that affect signaling events down-stream of the PTH/PTHrP receptor. This G protein-coupled receptor (GPCR, class B) is abundantly expressed in kidneys, bones, and growth plates, and at lower levels in a large variety of other tissues. The PTH/PTHrP receptor couples predominantly to the alpha-subunit of heterotrimeric stimulatory G protein (Gsα) and mediates the actions of two ligands, parathyroid hormone (PTH; gene name, PTH) and PTH-related peptide (PTHrP; also referred to as PTH-like peptide, PTHLP; gene name, PTHLH).
PTH, which is synthesized in mammals almost exclusively by the parathyroid glands, serves as the most prominent peptide hormone involved in the endocrine regulation of calcium and phosphate homeostasis [1]. In contrast to PTH, whose expression is restricted to the parathyroids, PTHrP is expressed in numerous tissues where it serves different paracrine and/or -autocrine functions that include the regulation of chondrocyte growth and differentiation (for review see [2, 3]). Upon binding to the PTH/PTHrP receptor, PTH and PTHrP stimulate the generation of cAMP, the most prominent second messenger down-stream of the PTH/PTHrP receptor, that is required for activation of protein kinase A (PKA). The subsequent phosphorylation of SIK2 and SIK33 inhibits the cellular activity of these salt-inducible kinases, thereby reducing phosphorylation of SIK substrates, including class IIa HDACs and CRTC family proteins (for review see [4–6]). Here, we review several rare genetic defects that can lead to similar shortening of metatarsals and/or -tarsals high-lighting the particularly sensitive involvement of the cAMP/PKA signaling pathway in these skeletal elements.
Albrights Hereditary Osteodystrophy features in pseudohypoparathyroidism type Ia and pseudopseudohypoparathyroidism
In 1942, Fuller Albright and colleagues described several patients with a constellation of physical features, including a rounded face, short stature, brachydactyly type E, and heterotopic ossifications, as well as early-onset obesity and variable neurocognitive deficiencies that occurred in the presence of hypocalcemia and hyperphosphatemia. He furthermore showed that these patients revealed no phosphaturic response to parathyroid extracts, leading him and his co-authors to conclude that there is renal resistance to PTH thus coining the term pseudohypoparathyroidism (PHP) [7].
Since the late 1960s, it is well established that kidney- and bone-derived tissues increase cAMP formation in response to PTH [8–11]. Furthermore, it was later shown that patients affected by the PHP variant first described by Albright et al. [7] (now referred to as pseudohypoparathyroidism type Ia (PHP1A)), revealed an impaired increase in urinary cAMP excretion when challenged with PTH, thus linking this second messenger to the lack of PTH-induced phosphaturia [12]. Readily accessible cells from PHP1A patients were subsequently shown to have an approximately 50 % reduction in G protein activity [13, 14]. Such cells from other patients revealed no in vitro abnormality, despite clinical and laboratory findings consistent with PHP1A; this disease variant was initially referred to as PHP type Ic (PHP1C) [15]. However, individuals affected by PHP1C showed inactivating genetic mutations in the same gene (see below) and thus belong to the PHP1A group of patients. While PTH resistance is usually the most obvious endocrine abnormality in PHP1A, which leads to hypocalcemia, hyperphosphatemia, and low or inappropriately normal 1,25(OH)2D formation despite elevated PTH levels, patients affected by this disorder frequently show also resistance to various other ligands that mediate there actions through GPCRs (for example, TSH, calcitonin, GHRH, epinephrine, LH, and FSH) (for a review see [16–19]).
An indistinguishable reduction of G protein activity is also observed for cells derived from patients affected by pseudopseudohypoparathyroidism (PPHP), first described by Fuller Albright and colleagues in 1952 [20], or its variant progressive osseous heteroplasia (POH) [13, 14, 21]. PPHP and POH patients show no hormonal resistance, yet most of the clinically obvious AHO features, including shortening of metacarpals and -tarsals.
Heterozygous GNAS mutations are the cause of shortened metacarpals and –tarsals in PHP1A and PPHP
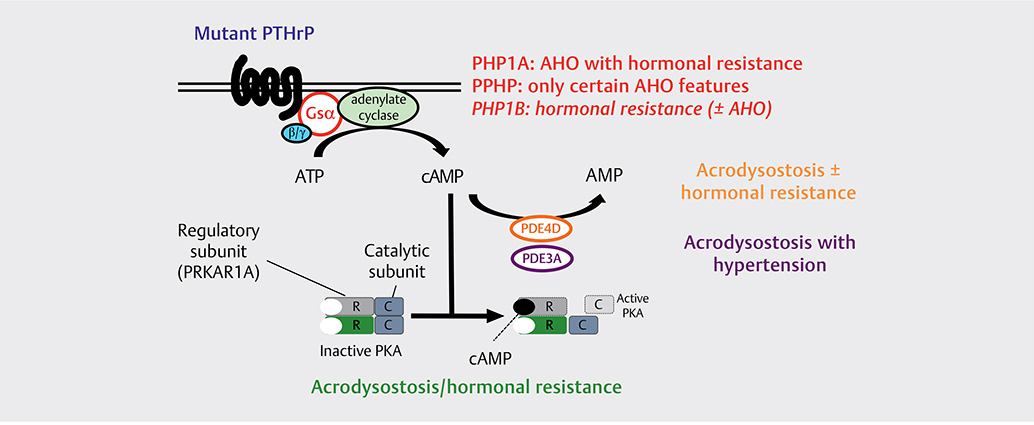
PHP1A and PPHP are both caused by inactivating mutations involving those GNAS exons that encode Gsα, the alpha-subunit of the stimulatory G protein [22, 23]. Gsα, which is ubiquitously expressed, couples a large number of GPCRs, including the PTH/PTHrP receptor, to adenylyl cyclase, the enzyme required for the intracellular generation of cAMP, the second messenger required for PKA activation (Fig. 1).
Fig. 1.
Human disorders associated with short metacarpals and -tarsals due to impaired cAMP formation, impaired cAMP actions, or accelerated cAMP degradation. The PTH/PTHrP receptor is abundantly expressed in kidney and bone where it mediates the PTH-dependent regulation of calcium and phosphate homeostasis. It is also expressed in numerous other tissues, particularly in the growth plate chondrocytes, where it slows, following activation by PTHrP, hypertrophic differentiation. Genetic diseases with reduced activation of the cAMP/PKA pathway are indicated.
Gsα is derived from the GNAS locus, a complex differentially methylated region located on chromosome 20q13.3, which encodes besides Gsα several other alternatively spliced transcripts [17, 24, 25]. In some tissues such as renal proximal tubules, thyroid or pituitary gland, Gsα expression occurs predominantly or exclusively from the maternal allele; expression from the paternal allele is silenced in these tissues through as yet unknown mechanisms. Consequently, inactivating maternal GNAS mutations involving the exons encoding Gsα are the cause of shortened metacarpals and -tarsals (and other known AHO features), as well as hormonal resistance, i. e. PHP1A (see Fig. 1; red text and symbols). The same or similar mutations on the paternal GNAS allele do not cause hormonal resistance, i. e. PPHP, yet skeletal abnormalities equivalent to those encountered in PHP1A [21, 26, 27]. These findings suggest that Gsα haploinsufficiency is likely responsible for the skeletal AHO features in PHP1A and PPHP.
Epigenetic GNAS abnormalities cause PHP1B
Patients affected by another PHP variant, namely PHP type Ib (PHP1B), were initially thought to develop only PTH-resistant hypocalcemia and hyperphosphatemia, but it is now well established that resistance to other hormones, particularly to TSH, can occur also in this PHP variant [17, 24, 25]. The autosomal dominant form of PHP1B (AD-PHP1B) is associated with a loss-of-methylation at one (exon A/B) or several (exons A/B, XL and AS) maternal exons of the GNAS complex locus that can be due to genetic deletions, duplications, or inversions (see Fig. 1; red text in italics). Sporadic PHP1B (sporPHP1B), the most frequent form of PHP1B, is typically associated with loss-of-methylation at all differentially methylated regions (DMR) on the maternal GNAS allele (exons A/B, XL and AS) and gain-of-methylation at the DMR on the paternal GNAS allele (exon NESP). With the exception of the sporPHP1B variant that is caused by paternal uniparental isodisomy or heterodisomy involving the chromosome 20q13.3 region comprising GNAS, sporPHP remains unresolved at the molecular level [17, 24, 25]. Loss-of-methylation at the maternal GNAS exon A/B is shared among all PHP1B variants and this epigenetic change is thus likely responsible for the much reduced or absent Gsα expression from the maternal allele.
Gsα deficiency in PHP1B can cause shortened metacarpals and -tarsals
A considerable number of PHP1B patients revealed shortening of metacarpals and -tarsals, which can be indistinguishable from the findings in patients affected by PHP1A or PPHP [25, 28–32], thereby highlighting once again the importance of bi-allelic Gsα expression for normal skeletal development. Based on the observations in PPHP and the different forms of PHP, shortening of metacarpals and -tarsals occurs when Gsα is derived from only one parental GNAS allele. This suggests that haploinsufficiency for this signaling protein in growth plate chondrocytes is sufficient to accelerate the maturation of these cells thereby leading to premature growth plate closure and shortening of some bones. Metacarpals and -tarsals thus seem to be particularly sensitive to a decline in intracellular cAMP accumulation. However, skeletal abnormalities similar to those caused by GNAS mutations occur also in other patients, who carry mutations in genes encoding proteins up-stream or down-stream of the PTH/PTHrP receptor and are thus likely to lead to an equivalent reduction of cAMP levels in growth plate chondrocytes (see Fig. 1; red text and symbols).
PTHLH mutations
Shortening of one or several metacarpals and -tarsals, or of all phalanges can also be caused by heterozygous mutations in PTHLH, the gene encoding PTHrP [33] (Fig. 1; blue text). These molecular defects include translocations [34], microdeletions [35, 36], and heterozygous missense, nonstop, or nonsense mutations [35–39]. Furthermore, a recently identified heterozygous splice-site mutation leads to the formation of a truncated PTHrP that is most likely biologically inactive [40]. When larger families were studied, the mutations segregated with the disorder and were not found in control alleles, thus supporting the conclusion that PTHrP haploinsufficiency alone can accelerate the maturation of growth plate chondrocytes. Accelerated chondrocyte maturation occurs not only in metatarsals and -carpals that appear to be particularly sensitive to reduced cAMP formation, but is likely to affect the entire skeleton thus leading to small reductions in adult height or to obvious short stature. Interestingly, a de novo duplication comprising the PTHLH gene was found to be associated in one patient with symmetrical enchondromatosis indicating that increased PTHrP expression can also lead to disease by slowing chondrocyte maturation [41, 42].
PRKAR1A mutations
Acrodysostosis refers to a group of rare skeletal dysplasias that share characteristic clinical, biochemical, and radiological features, particularly severe brachydactyly [43, 44]. As indicated above, the most commonly used effector system downstream of cAMP is protein kinase A (PKA). In the absence of cAMP, PKA exists as a tetramer that comprises dimers of one of four different regulatory subunits (PRKAR1A and PRKAR2A are ubiquitously expressed; PRKAR1B is found only in brain and testis, and PRKAR2B only adrenal and adipose tissue). These regulatory dimers lock the two catalytic (C) subunits, which are ubiquitously or brain-specifically expressed, into the inactive state. Cyclic AMP triggers the sequential release of the catalytic PKA subunits thus allowing enzyme activation [45–47]. Several different heterozygous PRKAR1A mutations were identified as the cause of acrodysostosis ACRDYS1 [48–51] (see Fig. 1; green text and symbols). These mutations all impair cAMP binding to PRKAR1A, thereby repressing constitutively the activity of the catalytic subunit and impairing the PKA response to cAMP [48, 52]. Consequently, there is little or no phosphaturic effect of PTH, despite normal urinary cAMP excretion. Likewise, PRKAR1A mutations are not predicted to impair PTHrP-dependent cAMP formation in chondrocytes, but PKA activation is most likely reduced thereby accelerating growth plate maturation. Heterozygous “activating” mutations involving PRKAR1A and heterozygous inactivating mutations involving the GNAS exons encoding Gsα thus lead to similar skeletal phenotypes.
PDE4D mutations
Genetic PDE4D defects cause acrodysostosis without obvious evidence for hormonal resistance in most cases [53–56]. PDE4D belongs to the class IV of Camp-specific phosphodiesterases that hydrolyze cAMP. In humans, a single PDE4D gene gives rise, through alternate splicing to at least nine different isoforms [57]. Numerous heterozygous missense PDE4D mutations have been identified in patients affected by acrodysostosis (ACRDYS2) [49–51, 56, 58–60] (see Fig. 1; orange text and symbols). The location and functional analysis of PDE4D mutations occurring in ACRDYS2 indicate that these mutations can influence the regulation of PDE activity by PKA resulting in increased hydrolytic activity [61, 62]. Increased expression of other PDE4 isoforms may also occur, as suggested by observations in some patients [58]. Although the clinical and radiographic findings in acrodysostosis can be similar to those observed in the different PHP variants [43, 44], GNAS-related skeletal abnormalities appear to be more variable and are usually less severe than acrodysostosis caused by PDE4D mutations. In contrast to typically severe skeletal dysplasia in ACRDYS2, resistance toward PTH and other hormones is less pronounced than in PHP1A and PHP1B, or not detectable [48, 50, 55, 56, 60].
PDE3A mutations
PDE3A is a widely expressed member of the PDE family, which hydrolyzes both cAMP and cGMP with high efficacy. Heterozygous mutations that activate PDE3A have been identified in patients in whom brachydactyly is associated with hypertension and moderately short stature; these autosomal dominant features occurred in the absence of other AHO features and mineral ion abnormalities [63, 64] (see Fig. 1; purple text and symbols). The mutations identified in this disorder are clustered in the N-terminal catalytic domain of PDE3A and increase protein kinase A-mediated PDE3A phosphorylation, thereby increasing its activity [60, 64–66]. The resulting increase in cAMP hydrolysis likely reduces the intracellular concentration of this second messenger, which is generated in the vasculature in response to PTHrP, thus potentially limiting the hypotensive activity of this peptide [64]. Likewise, a reduction in PTHrP-dependent accumulation of intracellular cAMP in growth plate chondrocytes could be responsible for brachydactyly and short stature, just like in the disorders caused by genetic mutations outlined above.
Brachydactyly caused by other genetic mutations
Shortening of metacarpals and -tarsals can be encountered also in several other disorders, including Turner syndrome, tricho-rhino-phalangeal syndrome, and Leri-Weill syndrome [67–69]; the genes mutated in these disorders are thus likely to affect chondrocyte growth and differentiation. Interestingly, Madelung deformities, similar to those due to mutations in the short stature homeobox containing gene (SHOX), can be observed also in some PHP1B patients [32]. Furthermore, mutations in HDAC4, a histone deacetylase involved in the regulation of genes important in the development of several different tissues, including bone, cause brachydactyly type E associated with mental retardation (BDMR). Initially, large deletions involving one 2q37 allele were discovered in this syndrome [70]. The clinical and molecular analysis of additional patients with different overlapping deletions involving the 2q37.3 region subsequently defined a critically important region, which then led to the identification of de novo mutations in HDAC4 [71]. Consistent with phenotype of Hdac4−/− mice [72], HDAC4 deletions or mutations are thus the cause of the BDMR syndrome. Interestingly, two individuals out of a large cohort of patients with PHP1B due to GNAS methylation changes revealed AHO-like features, including brachydactyly, that were associated with heterozygous deletions involving the HDAC4 locus [73]. It remains uncertain, however, whether PHP1B patients with GNAS methylation changes and thus hormonal resistance develop AHO-like features only when combined with HDAC4 variants.
PTH/PTHrP receptor haploinsufficiency does not shorten metacarpals or -tarsals
The mRNA encoding the PTH/PTHrP receptor is found in a large variety of fetal and adult tissues [74, 75]. Abundant concentrations are detected in proximal renal tubular cells, in osteoblasts, and in the prehypertrophic chondrocytes of metaphyseal growth plate (for review, see [3]). Mice lacking both Pthlh alleles display a dramatic acceleration of chondrocyte differentiation resulting in premature mineralization of all bones that are formed through an endochondral process thus leading to a completely mineralized skeleton at birth. From these and other studies, it is now well established that PTHrP is required for the continuous proliferation of chondrocytes in the growth plate, and that it postpones their programmed differentiation into hypertrophic chondrocytes. Consistent with this role of PTHrP in chondrocyte biology, inactivating homozygous (or compound heterozygous) PTH/PTHrP receptor mutations cause a lethal chondrodysplasia characterized much accelerated maturation of all bones that are formed through an endochondral process [76]. Surprisingly, however, inactivating heterozygous PTH/PTHrP receptor mutations are not associated with premature growth plate closure, although the R150C mutation, which is presumably activating, was found to have a small effect on growth [77].
Conclusions
Mutations in the genes encoding PTHrP, Gsα, or down-stream proteins involved in cAMP signaling or degradation are associated with accelerated chondrocyte differentiation and thus lead to premature growth plate closure, thereby limiting bone growth.
Footnotes
Conflict of Interest
The authors declare that they have no conflict of interest.
References
- [1].Potts JT Jr., Gardella TJ Progress, paradox, and potential: Parathyroid hormone research over five decades. Ann NY Acad Sci 2007; 1117: 196–208 [DOI] [PubMed] [Google Scholar]
- [2].Suva LJ,, Freeman A, Martin TJ. Eds. Parathyroid hormone-related protein: Gene structure, biosynthesis, metabolism and regulation. San Diego: Adademic Press; 2015: 45–64 [Google Scholar]
- [3].Maes C, Kronenberg HM. In DeGroot LJ, and Jameson JL, Eds. Endocrinology. Philadelphia, PA: W.B. Saunders Company; 2016: 1038–1062 [Google Scholar]
- [4].Gardella TJ, Jüppner H, Brown EM et al. In DeGroot LJ, and Jameson JL, Eds. Endocrinology. Philadelphia, PA: W.B. Saunders Company; 2016: 969–990 [Google Scholar]
- [5].Wein MN. Parathyroid hormone signaling in osteocytes. JBMR Plus 2018; 2: 22–30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Wein MN, Foretz M, Fisher DE et al. Salt-inducible kinases: Physiology, regulation by cAMP, and therapeutic potential: (Trends Endocrinol. Metab. 29, 723–735, 2018). Erratum: Trends Endocrinol Metab 2019; 30: 407. [DOI] [PubMed] [Google Scholar]
- [7].Albright F, Burnett CH, Smith PH et al. Pseudohypoparathyroidism - an example of “Seabright-Bantam syndrome”. Endocrinology. 1942; 30: 922–932 [Google Scholar]
- [8].Chase LR, Aurbach GD. Parathyroid function and the renal excretion of 3’5’-adenylic acid. Proc Natl Acad Sci USA 1967; 58: 518–525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Chase LR, Fedak SA, Aurbach GD. Activation of skeletal adenyl cyclase by parathyroid hormone in vitro. Endocrinology 1969; 84: 761–768 [DOI] [PubMed] [Google Scholar]
- [10].Chase LR, Aurbach GD. The effect of parathyroid hormone on the concentration of adenosine 3’,5’-monophosphate in skeletal tissue in vitro. J Biol Chem 1970; 245: 1520–1526 [PubMed] [Google Scholar]
- [11].Marcus R, Aurbach GD. Adenyl cyclase from renal cortex. Biochim Biophys Acta 1971; 242: 410–421 [DOI] [PubMed] [Google Scholar]
- [12].Chase LR, Melson GL, Aurbach GD. Pseudohypoparathyroidism: Defective excretion of 3’,5’-AMP in response to parathyroid hormone. J Clin Invest 1969; 48: 1832–1844 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Levine MA, Downs RW Jr., Singer M et al. Deficient activity of guanine nucleotide regulatory protein in erythrocytes from patients with pseudohypoparathyroidism. Biochem Biophys Res Commun 1980; 94: 1319–1324 [DOI] [PubMed] [Google Scholar]
- [14].Farfel Z, Brickman AS, Kaslow HR et al. Defect of receptor-cyclase coupling protein in pseudohypoparathyroidism. N Engl J Med 1980; 303: 237–242 [DOI] [PubMed] [Google Scholar]
- [15].Linglart A, Carel JC, Garabedian M et al. GNAS1 lesions in pseudohypoparathyroidism Ia and Ic: Genotype phenotype relationship and evidence of the maternal transmission of the hormonal resistance. J Clin Endocrinol Metab 2002; 87: 189–197 [DOI] [PubMed] [Google Scholar]
- [16].Levine MA.In LR JP Bilezikian, Rodan GA ed, Principles of Bone biology. New York: Academic Press; 2002: 1137–1159 [Google Scholar]
- [17].Bastepe M, Jüppner H. In: DeGroot LJ, and Jameson JL, Eds. Endocrinology. Philadelphia, PA: W.B. Saunders Company; 2016: 1147–1159 [Google Scholar]
- [18].Linglart A, Maupetit-Mehouas S, Silve C. GNAS -related loss-of-function disorders and the role of imprinting. Horm Res Paediatr 2013; 119–129 [DOI] [PubMed] [Google Scholar]
- [19].Elli FM, Linglart A, Garin I et al. The prevalence of GNAS deficiency-related diseases in a large cohort of patients characterized by the EuroPHP Network. J Clin Endocrinol Metab 2016; 101: 3657–3668 [DOI] [PubMed] [Google Scholar]
- [20].Albright F, Forbes AP, Henneman PH. Pseudo-pseudohypoparathyroidism. Trans Assoc Am Physicians 1952; 65: 337–350 [PubMed] [Google Scholar]
- [21].Shore E, Ahn J, Jan de Beur S et al. Paternally-inherited inactivating mutations of the GNAS1 gene in progressive osseous heteroplasia. New Engl J Med 2002; 346: 99–106 [DOI] [PubMed] [Google Scholar]
- [22].Patten JL, Johns DR, Valle D et al. Mutation in the gene encoding the stimulatory G protein of adenylate cyclase in Albright’s hereditary osteodystrophy. New Engl J Med 1990; 322: 1412–1419 [DOI] [PubMed] [Google Scholar]
- [23].Weinstein LS, Gejman PV, Friedman E et al. Mutations of the Gs a-subunit gene in Albright hereditary osteodystrophy detected by denaturing gradient gel electrophoresis. Proc Natl Acad Sci USA 1990; 87: 8287–8290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Tafaj O, Jüppner H. Pseudohypoparathyroidism: One gene, several syndromes. J Endocrinol Invest 2017; 40: 347–356 [DOI] [PubMed] [Google Scholar]
- [25].Mantovani G, Bastepe M, Monk D et al. Diagnosis and management of pseudohypoparathyroidism and related disorders: First International Consensus Statement. Nat Rev Endocrinol 2018; 14: 476–500 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Davies SJ, Hughes HE. Imprinting in Albright’s hereditary osteodystrophy. J Med Genet 1993; 30: 101–103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Long DN, McGuire S, Levine MA et al. Body mass index differences in pseudohypoparathyroidism type 1a vs. pseudopseudohypoparathyroidism may implicate paternal imprinting of Galpha(s) in the development of human obesity. J Clin Endocrinol Metab 2007; 92: 1073–1079 [DOI] [PubMed] [Google Scholar]
- [28].Schipani E, Weinstein LS, Bergwitz C et al. Pseudohypoparathyroidism type Ib is not caused by mutations in the coding exons of the human parathyroid hormone (PTH)/PTH-related peptide receptor gene. J Clin Endocrinol Metab 1995; 80: 1611–1621 [DOI] [PubMed] [Google Scholar]
- [29].Bastepe M, Lane AH, Jüppner H. Paternal uniparental isodisomy of chromosome 20q (patUPD20q) - and the resulting changes in GNAS1 methylation - as a plausible cause of pseudohypoparathyroidism. Am J Hum Genet 2001; 68: 1283–1289 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Mantovani G, Bondioni S, Linglart A et al. Genetic analysis and evaluation of resistance to thyrotropin and growth hormone-releasing hormone in pseudohypoparathyroidism type Ib. J Clin Endocrinol Metab 2007; 92: 3738–3742 [DOI] [PubMed] [Google Scholar]
- [31].Sharma A, Phillips AJ, Jüppner H. Hypoplastic metatarsals–beyond cosmesis. N Engl J Med 2015; 373: 2189–2190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Sanchez J, Perera E, Jan de Beur S et al. Madelung-like deformity in pseudohypoparathyroidism type 1b. J Clin Endocrinol Metab 2011; 96: E1507–E1511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Mundlos S The brachydactylies: A molecular disease family. Clin Genet 2009; 76: 123–136 [DOI] [PubMed] [Google Scholar]
- [34].Maass PG, Wirth J, Aydin A et al. A cis-regulatory site downregulates PTHLH in translocation t(8;12)(q13;p11.2) and leads to Brachydactyly Type E. Hum Mol Genet 2010; 19: 848–860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Klopocki E, Hennig BP, Dathe K et al. Deletion and point mutations of PTHLH cause brachydactyly type E. Am J Hum Genet 2010; 86: 434–439 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Thomas-Teinturier C, Pereda A, Garin I et al. Report of two novel mutations in PTHLH associated with brachydactyly type E and literature review. Am J Med Genet A 2016; 170: 734–742 [DOI] [PubMed] [Google Scholar]
- [37].Wang J, Wang Z, An Y et al. Exome sequencing reveals a novel PTHLH mutation in a Chinese pedigree with brachydactyly type E and short stature. Clin Chim Acta 2015; 446: 9–14 [DOI] [PubMed] [Google Scholar]
- [38].Jamsheer A, Sowinska-Seidler A, Olech EM et al. Variable expressivity of the phenotype in two families with brachydactyly type E, craniofacial dysmorphism, short stature and delayed bone age caused by novel heterozygous mutations in the PTHLH gene. J Hum Genet 2016; 61: 457–461 [DOI] [PubMed] [Google Scholar]
- [39].Pereda A, Garzon-Lorenzo L, Garin I et al. The p.R56 * mutation in PTHLH causes variable brachydactyly type E. Am J Med Genet A 2017; 173: 816–819 [DOI] [PubMed] [Google Scholar]
- [40].Reyes M, Bravenboer B, Jüppner H. A Heterozygous splice-site mutation in PTHLH causes autosomal dominant shortening of metacarpals and metatarsals. J Bone Miner Res 2019; 34: 482–489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Collinson M, Leonard SJ, Charlton J et al. Symmetrical enchondromatosis is associated with duplication of 12p11.23 to 12p11.22 including PTHLH. Am J Med Genet A 2010; 152A: 3124–3128 [DOI] [PubMed] [Google Scholar]
- [42].Silve C A Cup Half-Full or Half-Empty? When PTHrP Levels Matter. IBMS BoneKEy. 2010; 7: http://www.bonekey-ibms.org/cgi/content/full/ibmske;7/9/325 325–32 [Google Scholar]
- [43].Ablow RC, Hsia YE, Brandt IK. Acrodysostosis coinciding with pseudohypoparathyroidism and pseudo-pseudohypoparathyroidism. AJR Am J Roentgenol 1977; 128: 95–99 [DOI] [PubMed] [Google Scholar]
- [44].Davies SJ, Hughes HE. Familial acrodysostosis: can it be distinguished from Albright’s hereditary osteodystrophy? Clin Dysmorphol. 1992; 1: 207–215 [PubMed] [Google Scholar]
- [45].Tasken K, Skalhegg BS, Tasken KA et al. Structure, function, and regulation of human cAMP-dependent protein kinases. Adv Second Messenger Phosphoprotein Res 1997; 31: 191–204 [DOI] [PubMed] [Google Scholar]
- [46].Taylor SS, Buechler JA, Yonemoto W. cAMP-dependent protein kinase: Framework for a diverse family of regulatory enzymes. Annu Rev Biochem 1990; 59: 971–1005 [DOI] [PubMed] [Google Scholar]
- [47].Taylor SS, Kim C, Cheng CY et al. Signaling through cAMP and cAMP-dependent protein kinase: diverse strategies for drug design. Biochim Biophys Acta 2008; 1784 (1): 16–26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Linglart A, Menguy C, Couvineau A et al. Recurrent PRKAR1A mutation in acrodysostosis with hormone resistance. N Engl J Med 2011; 364: 2218–2226 [DOI] [PubMed] [Google Scholar]
- [49].Silve C, Clauser E, Linglart A. Acrodysostosis. Horm Metab Res 2012; 44: 749–758 [DOI] [PubMed] [Google Scholar]
- [50].Silve C, Le-Stunff C, Motte E et al. Acrodysostosis syndromes. BoneKEy; 2012; 11: 21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Elli FM, Bordogna P, de Sanctis L et al. Screening of PRKAR1A and PDE4D in a Large Italian series of patients clinically diagnosed with Albright hereditary osteodystrophy and/or pseudohypoparathyroidism. J Bone Miner Res 2016; 31: 1215–1224 [DOI] [PubMed] [Google Scholar]
- [52].Rhayem Y, Le Stunff C, Abdel Khalek W et al. Functional characterization of PRKAR1A mutations reveals a unique molecular mechanism causing acrodysostosis but multiple mechanisms causing carney complex. J Biol Chem 2015; 290: 27816–27828 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Lee H, Graham JM Jr., Rimoin DL et al. Exome sequencing identifies PDE4D mutations in acrodysostosis. Am J Hum Genet 2012; 90: 746–751 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Michot C, Le Goff C, Goldenberg A et al. Exome sequencing identifies PDE4D mutations as another cause of acrodysostosis. Am J Hum Genet 2012; 90: 740–745 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Linglart A, Fryssira H, Hiort O et al. PRKAR1A and PDE4D mutations cause acrodysostosis but two distinct syndromes with or without GPCR-signaling hormone resistance. J Clin Endocrinol Metab 2012; 97: E2328–E2338 [DOI] [PubMed] [Google Scholar]
- [56].Lindstrand A, Grigelioniene G, Nilsson D et al. Different mutations in PDE4D associated with developmental disorders with mirror phenotypes. J Med Genet 2014; 51: 45–54 [DOI] [PubMed] [Google Scholar]
- [57].Houslay MD, Adams DR. PDE4 cAMP phosphodiesterases: Modular enzymes that orchestrate signalling cross-talk, desensitization and compartmentalization. Biochem J 2003; 370: 1–18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [58].Kaname T, Ki CS, Niikawa N et al. Heterozygous mutations in cyclic AMP phosphodiesterase-4D (PDE4D) and protein kinase A (PKA) provide new insights into the molecular pathology of acrodysostosis. Cell Signal 2014; 26: 2446–2459 [DOI] [PubMed] [Google Scholar]
- [59].Lynch DC, Dyment DA, Huang L et al. Identification of novel mutations confirms PDE4D as a major gene causing acrodysostosis. Hum Mutat 2013; 34: 97–102 [DOI] [PubMed] [Google Scholar]
- [60].Michot C, Le Goff C, Blair E et al. Expanding the phenotypic spectrum of variants in PDE4D/PRKAR1A: From acrodysostosis to acroscyphodysplasia. Eur J Hum Genet 2018; 26: 1611–1622 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Cedervall P, Aulabaugh A, Geoghegan KF et al. Engineered stabilization and structural analysis of the autoinhibited conformation of PDE4. Proc Natl Acad Sci USA 2015; 112: E1414–E1422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Briet C, Pereda A, Le Stunff C et al. Mutations causing acrodysostosis-2 facilitate activation of phosphodiesterase 4D3. Hum Mol Genet 2017; 26: 3883–3894 [DOI] [PubMed] [Google Scholar]
- [63].Bilginturan N, Zileli S, Karacadag S et al. Hereditary brachydactyly associated with hypertension. J Med Genet 1973; 10: 253–259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [64].Maass PG, Aydin A, Luft FC et al. PDE3A mutations cause autosomal dominant hypertension with brachydactyly. Nat Genet 2015; 47: 647–653 [DOI] [PubMed] [Google Scholar]
- [65].Boda H, Uchida H, Takaiso N et al. A PDE3A mutation in familial hypertension and brachydactyly syndrome. J Hum Genet 2016; [DOI] [PubMed] [Google Scholar]
- [66].Renkema KY, Westermann JM, Nievelstein RAJ et al. PDE3A gene screening improves diagnostics for patients with Bilginturan syndrome (hypertension and brachydactyly syndrome). Hypertens Res 2018; 41: 981–988 [DOI] [PubMed] [Google Scholar]
- [67].Pereda A, Garin I, Garcia-Barcina M et al. Brachydactyly E: Isolated or as a feature of a syndrome. Orphanet J Rare Dis 2013; 8: 141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Karaca A, Reyes M, Shumate LT et al. Severe brachydactyly and short stature resulting from a novel pathogenic TRPS1 variant within the GATA DNA-binding domain. Bone 2019; 123: 153–158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [69].Grigelioniene G, Eklof O, Ivarsson SA et al. Mutations in short stature homeobox containing gene (SHOX) in dyschondrosteosis but not in hypochondroplasia. Hum Genet 2000; 107: 145–149 [DOI] [PubMed] [Google Scholar]
- [70].Phelan MC, Rogers RC, Clarkson KB et al. Albright hereditary osteodystrophy and del(2) (q37.3) in four unrelated individuals. Am J Med Genet 1995; 58: 1–7 [DOI] [PubMed] [Google Scholar]
- [71].Williams SR, Aldred MA, Der Kaloustian VM et al. Haploinsufficiency of HDAC4 causes brachydactyly mental retardation syndrome, with brachydactyly type E, developmental delays, and behavioral problems. Am J Hum Genet 2010; 87: 219–228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [72].Wein MN, Liang Y, Goransson O et al. SIKs control osteocyte responses to parathyroid hormone. Nat Commun 2016; 7: 13176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [73].Elli FM, deSanctis L, Maffini MA et al. Association of GNAS imprinting defects and deletions of chromosome 2 in two patients: Clues explaining phenotypic heterogeneity in pseudohypoparathyroidism type 1B/iPPSD3. Clin Epigenetics 2019; 11: 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [74].Lee K, Deeds JD, Segre GV. Expression of parathyroid hormone-related peptide and its receptor messenger ribonucleic acid during fetal development of rats. Endocrinology 1995; 136: 453–463 [DOI] [PubMed] [Google Scholar]
- [75].Riccardi D, Lee WS, Lee K et al. Localization of the extracellular Ca(2 + )-sensing receptor and PTH/PTHrP receptor in rat kidney. Am J Physiol 1996; 271: F951–F956 [DOI] [PubMed] [Google Scholar]
- [76].Silve C, Jüppner H. In: Bilezikian J, ed Principles of Bone Biology. San Diego: Academic Press; 2019 [Google Scholar]
- [77].Marouli E, Graff M, Medina-Gomez C et al. Rare and low-frequency coding variants alter human adult height. Nature 2017; 542: 186–190 [DOI] [PMC free article] [PubMed] [Google Scholar]