Abstract
A new method (V.A.C.ULTA™ Therapy System, KCI USA, Inc., San Antonio, TX) combines the benefits of negative pressure wound therapy (NPWT; V.A.C.® Therapy, KCI USA, Inc.) with regulated, periodic instillation of user‐selected topical wound solutions (V.A.C. VeraFlo™ Therapy, KCI USA, Inc.). In simulated wound model studies comparing solution distribution using NPWT with and without a soak phase, the instillation soak phase allowed for uniform solution distribution across the wound bed, whereas continuous (no soak) irrigation resulted in uneven coverage. Additional in vitro work illustrated that bacterial particle aerosolisation during wound cleansing was significantly decreased using NPWT with instillation (NPWTi) versus commercially available low‐pressure wound cleansers (P < 0·05). In porcine studies, NPWT with saline instillation induced 43% more granulation tissue versus NPWT (P < 0·05) and was as effective at wound cleansing as pulsed lavage. These studies have demonstrated that NPWTi may be an effective wound management therapy that provides both wound cleansing and NPWT benefits.
Keywords: Negative pressure wound therapy, Negative pressure wound therapy with instillation, Periodic instillation, Reticulated open‐cell foam, Wound cleansing
Introduction
Negative pressure wound therapy (NPWT) has been used for the past 20 years with a growing body of scientific and clinical research literature supporting its use in various wound types, including chronic wounds and those complex in nature 1, 2. In 1995, Kinetic Concepts, Inc., USA (KCI) was the first to commercialise an NPWT system, commonly referred to as the Vacuum‐Assisted Closure (V.A.C.®) Therapy System. The utilisation of instillation therapy in combination with V.A.C. Therapy via a gravity‐fed method was first reported in 1998 by Fleischmann et al. to manage septic wounds that failed to respond to conventional therapy 3. This cleansing method was initially used in infected wounds using antimicrobial solutions. This combinatory therapy has now expanded to include cleansing regimens that can facilitate the removal of exudates, debris and other inhibitors of healing 4, 5. Examples of such topical wound solutions include isotonic solutions (normal saline or 0·9% sodium chloride), biguanides (polyhexanide), hypochlorite‐based solutions (sodium hypochlorite), sulphur‐based compounds (sulphonamides), cationic solutions (benzalkonium chloride) and silver compounds (silver nitrate) 6. Both standard NPWT and NPWT with instillation therapy (NPWTi; V.A.C. VeraFlo™ Therapy, KCI USA, Inc., San Antonio, TX) have recently been combined into one system (NPWT/NPWTi; V.A.C.ULTA™ Therapy System, KCI USA, Inc.) that allows for automated and controlled delivery of topical instillation solutions to the wound bed, followed by a programmed ‘soak’ time before the subsequent removal of the solution using NPWT (Figure 1). These discrete phases are applied in cycles. Thus, NPWTi combines the benefits of wound cleansing using topical wound solutions with the well‐documented advantages of NPWT.
Figure 1.
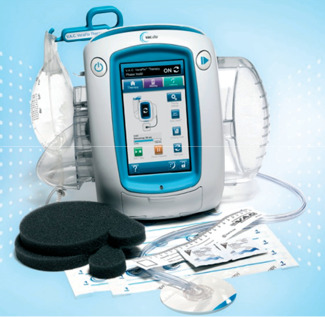
Negative pressure wound therapy (NPWT)/NPWT with instillation (NPWTi) system (V.A.C.ULTA™ Therapy System) that combines NPWT (V.A.C.® Therapy) with the option for instillation therapy using NPWTi (V.A.C. VeraFlo™ Therapy) dressings. (Reprinted with permission from KCI Licensing, Inc.)
This review summarises the 2012 and 2013 International Surgical Wound Forum presentations based on recently published scientific literature evaluating NPWTi. The articles focussed on the results of benchtop and preclinical studies, suggesting the potential benefits of NPWTi. (The following results have not been confirmed in human studies and are not to be considered clinical claims.)
Advancement of dressings
NPWTi uses specially designed reticulated open‐cell foam (ROCF) dressings: ROCF‐V (V.A.C. VeraFlo™ Dressing System, KCI USA, Inc.) and ROCF‐VC (V.A.C. VeraFlo Cleanse™ Dressing System, KCI USA, Inc.), which have a pore size similar to that of the standard ROCF‐G (V.A.C. GranuFoam™ Dressing, KCI USA, Inc.). However, these new dressings have a slightly different chemical composition using a polyurethane ester backbone as opposed to the polyurethane ether found in ROCF‐G. This chemical modification makes ROCF‐V and ROCF‐VC less hydrophobic than ROCF‐G and augments fluid distribution within the wound bed. Thus, ROCF‐V and ROCF‐VC dressings are more conducive to instillation therapy. Lessing et al. 7 compared the wicking capacity of ROCF‐G and ROCF‐V in a compressed state, finding that ROCF‐V distributed more fluid (saline) and had greater wicking capability (P < 0·05) than ROCF‐G (Figure 2). Furthermore, the formulation changes for ROCF‐V and ROCF‐VC offer greater mechanical strength compared with ROCF‐G, thereby increasing their ability to withstand tearing and reducing the likelihood of dressing fragment retention at the wound site during dressing changes 7.
Figure 2.
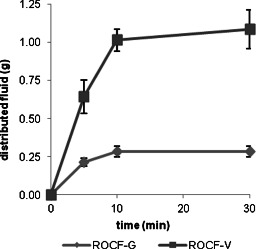
Fluid distribution (wicking) capabilities over time, comparing reticulated open‐cell foam (ROCF)‐G (V.A.C.® GranuFoam Dressing) to ROCF‐V (V.A.C. VeraFlo™ Dressing) in a compressed state 7.
Fluid distribution
To further assess the fluid distribution capabilities of the NPWTi dressings, a benchtop study using agar‐based wound models was performed. Uniformity of the exposure of instillation fluids was evaluated by the addition of a dye to the instillant for enhancement of the visual uptake of the fluid into the agar 8. NPWTi begins with the instillation fluid being slowly introduced to the wound and remaining on the wound bed for a predetermined period of time, or ‘soak time’, prior to its removal by the application of NPWT.
Recently, publications have reported the use of continuous irrigation with continuous application of NPWT 9, 10, 11. This method eliminates the typical soak times that are imparted using NPWTi with periodic instillation. A simulated wound model study was then performed comparing continuous irrigation to periodic instillation, addressing fluid distribution over the wound bed. Continuous irrigation therapy (NPWT at −125 mmHg with simultaneous fluid administration of 30 ml/hour for 3·5 hours) showed limited capability to deliver instillants to the simulated wound bed surface. This is illustrated by the lack of colour distribution across the agar model using this therapy and the staining localised to areas directly below the instillation pad even after 3·5 hours of therapy (Figure 3). Conversely, NPWTi showed more uniform coverage of the entire simulated wound bed after a single 10‐minute soak time and increased exposure with repeated cycles of instillation. Through pixelation analysis of treated agar model images, NPWTi led to significantly greater coverage of the agar wound bed (73·0% ± 3·2%) compared with continuous irrigation method (30·3% ± 10·7%, P < 0·05). In a more complex wound model that contained tunnelling and undermining, qualitative assessment using the continuous irrigation method demonstrated limited distribution throughout the wound bed, whereas NPWTi provided instillation fluid exposure across the entire wound bed including the more difficult‐to‐reach locations (Figure 3). The results of this study suggest that NPWTi allows for a significantly more uniform exposure of topical wound solutions to all areas of the wound bed in comparison to continuous irrigation therapy 8.
Figure 3.
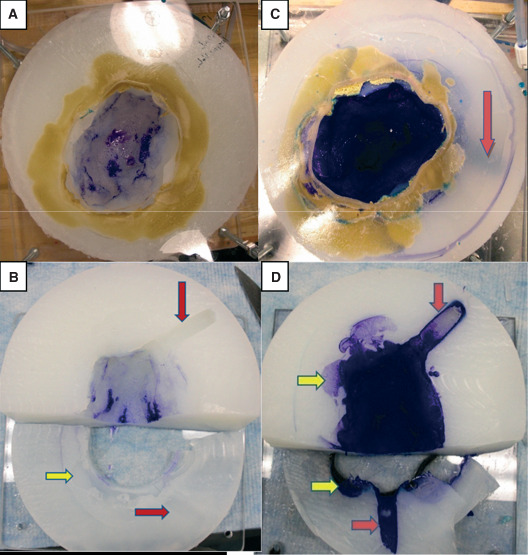
Wound bed coverage by instillation solution via continuous irrigation (A and B) versus periodic‐instillation (negative pressure wound therapy with instillation, NPWTi) (C and D) therapy on an agar‐based complex wound model with undermining (yellow arrows) and tunnelling (red arrows) is indicated 8. (A and C) Full view of agar wound model. (B and D) Lateral sectioning of agar wound model to expose undermining and tunnelling portions. Note the lack of dye exposure in undermined and tunnelled portions of the agar wound model treated with the continuous instillation therapy (B) compared to the definitive staining in undermined and tunnelled portions treated with NPWTi (D). (Reprinted with permission from reference 8.)
Bacterial management: cross‐contamination
Wound cleansing is a crucial step in wound management and healing. The introduction of fluid into the wound bed is a prevalent method used to remove healing impediments such as cellular debris, necrotic tissue, elevated inflammatory and enzymatic elements, as well as infectious agents 12, 13, 14. Standard wound irrigation techniques, such as lavage, may disperse bacteria beyond the wound surface, thus increasing the risk of cross‐contamination to the patient, health care worker and surrounding environment. Allen et al. 15 conducted a benchtop study using an anatomical wound model with a simulated sacral pressure ulcer to assess the cross‐contamination potential of NPWTi using ROCF‐V and ROCF‐VC in comparison to lavage. The simulated sacral pressure ulcer was inoculated with inactivated common wound bacteria such as Escherichia coli and Staphylococcus aureus, fluorescently labelled for quantification purposes. Collection plates around the simulated sacral wound were arranged concentrically in a 15‐cm radius from the centre of the wound. They captured the droplets or splashed fluid when the wound was cleansed using NPWTi or three different commercially available low‐pressure lavage systems (pressurised can and two different spray bottles) following the manufacturer's instructions. In this study, NPWTi consisted of five cycles of instillation with saline followed by a 60‐second soak time and application of negative pressure at −125 mmHg for 20 minutes. As Figure 4 illustrates, the standard wound irrigation techniques led to aerosolisation of bacterial particles with more than 50% of the bacterial particles recovered 15 cm away from the wound with the assumption that the remaining bacterial particles were dispersed beyond the collection plates (P < 0·5). With NPWTi treatments, no bacterial particles were detected outside of the simulated sacral ulcer perimeter with more than 95% of the amount inoculated found in the canister attached to the therapy unit, thereby maintaining containment of the potentially infectious agents. Thus, NPWTi may help to decrease the risk of cross‐contamination to patients and health care personnel in the wound care environment 15.
Figure 4.
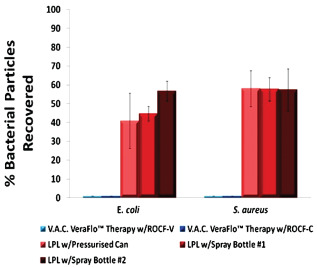
Bacterial particles detected after cleansing treatments. Negative pressure wound therapy with instillation (NPWTi) therapies were significantly different from LPL w/Pressurised Can, LPL w/Spray Bottle #1 and LPL w/Spray Bottle #2, within the bacterial strain group (P < 0·05) 15. LPL, low‐pressure lavage; ROCF‐V, V.A.C. VeraFlo™ Dressing; ROCF‐VC, V.A.C. VeraFlo Cleanse™ Dressing; N/A, not applicable. (Reprinted with permission from reference 15.)
Preclinical outcomes: granulation
NPWT has been demonstrated to stimulate the production of granulation tissue 1, 16; however, the impact of NPWTi on granulation tissue formation wounds remained unclear. To elucidate the influence of NPWTi on granulation tissue formation, Lessing et al. 7 used full‐thickness excisional dorsal wounds created in a porcine wound model, which were subsequently treated with either NPWT (−125 mmHg) or NPWTi with saline (cycles of 5‐minute soak followed by NPWT at −125 mmHg for 2·5 hours). Following 7 days of treatment, histological analyses were performed on wound tissue samples to evaluate granulation thickness and oedema. The wounds treated with NPWTi and saline showed a statistically significant (P < 0·05) 43% increase in granulation tissue thickness compared to wounds treated with standard NPWT (Figure 5). Furthermore, evaluation of the tissues by a histopathologist revealed minimal oedema and inflammation in either treatment group, suggesting that the newly produced granulation tissue was of good quality and not the result of swelling 7.
Figure 5.
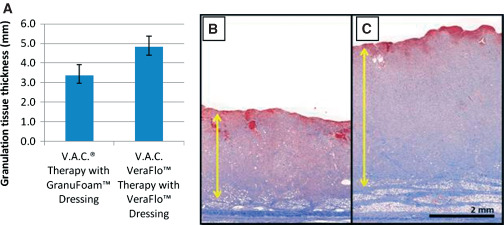
Graphical (A) and histological evaluations (B and C) of granulation tissue production in a porcine full‐thickness excisional wound model comparing negative pressure wound therapy (NPWT) with reticulated open‐cell foam (ROCF)‐G (B) and NPWT with instillation (NPWTi) with ROCF‐V (C) after 7 days of treatment 7. Following the 7 days of therapy, NPWTi resulted in a significant increase (P < 0·05) in granulation tissue thickness compared with traditional NPWT. (B and C: Reprinted with permission from KCI Licensing, Inc.)
Preclinical outcomes: wound cleansing
Regular wound cleansing can help to promote healing by removing devitalised tissue, infectious agents, cellular debris and exudates 17. An established method for wound cleansing is irrigation using low‐pressure lavage with saline 5, and a preclinical study was performed by Allen et al. 15 to compare this technique to NPWTi with saline. Porcine full‐thickness wounds that were allowed to lightly granulate were inoculated with a fluorescein dextran solution, simulating wound debris and cleansed with either low‐pressure lavage or NPWTi with saline. Low‐pressure lavage was used to deliver 1000 ml of saline in a 2‐minute period. NPWTi delivered approximately 40 ml of saline with a 5‐minute soak followed by 5 minutes of NPWT at −125 mmHg, compressing ten instillation cycles into 2 hours of therapy. Fluorescent images were taken prior to and post cleansing to quantify the debris removal, revealing both techniques were equally able to cleanse the wound debris [low‐pressure lavage at 95% ± 1·5% debris cleansed versus NPWTi at 99% ± 0·6% (P < 0·05)] (cleansing with NPWTi is illustrated in Figure 6). However, assessment of wound swelling using a three‐dimensional imaging system showed that low‐pressure lavage increased swelling, suggesting tissue damage, whereas swelling was reduced after NPWTi 15. The results of this preclinical study suggested that NPWTi might be a gentle, yet effective technique for wound cleansing.
Figure 6.
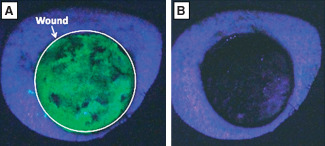
Fluorescent images prior to (A) and post (B) cleansing of the dextran‐inoculated porcine wound via negative pressure wound therapy with instillation (NPWTi) 15. (Reprinted with permission from reference 15.)
Conclusions
In summary, benchtop data suggested that NPWTi (V.A.C. VeraFlo Therapy) allowed for effective distribution and uniform exposure of topical wound solutions across the wound bed. Also, in vitro data with NPWTi indicated that bacterial aerosolisation might be minimised. Furthermore, preclinical work in porcine models suggested that NPWTi cleansed wounds as effectively as low‐pressure lavage while reducing oedema; increased granulation tissue formation compared with traditional NPWT was also observed. These findings indicated the positive potential of the implementation of NPWTi in medical practice; however, these results require confirmation in humans.
Acknowledgements
All authors (AMR, DA and MCL) are employees of Kinetic Concepts, Inc. (KCI). Drs AMR and MCL presented as a faculty members during the International Surgical Wound Forum (ISWF), an annual educational event sponsored by Kinetic Concepts, Inc. (KCI). This article is part of a KCI‐funded educational supplement based on faculty presentations at 2012 and 2013 ISWF sessions related to wound care strategies with a focus on use of negative pressure wound therapy with instillation (i.e. VA.C. Instill® Wound Therapy and V.A.C. VeraFlo™ Therapy, KCI, San Antonio, TX). The authors express special thanks to the KCI Research and Development Department and especially to Dr Amy McNulty and Dr Paul Slack for guidance and execution. They also thank reviewers (specifically Twan Wackers, Alice Goodwin and Dr Sandra Garcia) whose input is greatly appreciated.
Rycerz AM, Allen D, Lessing C. Science supporting negative pressure wound therapy with instillation.
References
- 1. Argenta LC, Morykwas MJ. Vacuum‐assisted closure: a new method for wound control and treatment: clinical experience. Ann Plast Surg 1997;38:563–76. [PubMed] [Google Scholar]
- 2. Banwell PE, Teot L. Topical negative pressure (TNP): the evolution of a novel wound therapy. J Wound Care 2003;12:22–8. [DOI] [PubMed] [Google Scholar]
- 3. Fleischmann W, Russ M, Westhauser A, Stampehl M. Vacuum sealing as carrier system for controlled local drug administration in wound infection. Unfalchirurg 1998;101:649–54[Article in German]. [DOI] [PubMed] [Google Scholar]
- 4. Jerome D. Advances in negative pressure wound therapy: the VAC instill. J Wound Ostomy Continence Nurs 2007;34:191–4. [DOI] [PubMed] [Google Scholar]
- 5. Kaehn K, Eberlein T. In‐vitro test for comparing the efficacy of wound rinsing solutions. Br J Nurs 2009;18:S4–10. [DOI] [PubMed] [Google Scholar]
- 6. Atiyeh BS, Dibo SA, Hayek SN. Wound cleansing, topical antiseptics and wound healing. Int Wound J 2009;6:420–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lessing C, Slack P, Hong KZ, Kilpadi D, McNulty A. Negative pressure wound therapy with controlled saline instillation (NPWTi): dressing properties and granulation response in vivo. Wounds 2011;23:309–19. [PubMed] [Google Scholar]
- 8. Rycerz AM, Slack P, McNulty AK. Distribution assessment comparing continuous and periodic wound instillation in conjunction with negative pressure wound therapy using an agar‐based model. Int Wound J 2013;10:214–20. DOI: 10.1111/j.1742-481X.2012.00968.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Scimeca CL, Bharara M, Fisher TK, Giovinco N, Armstrong DG. Novel use of doxycycline in continuous‐instillation negative pressure wound therapy as “wound chemotherapy”. Foot Ankle Spec 2010;3:190–3. [DOI] [PubMed] [Google Scholar]
- 10. Scimeca CL, Bharara M, Fisher TK, Kimbriel H, Mills JL, Armstrong DG. Novel use of insulin in continuous‐instillation negative pressure wound therapy as “wound chemotherapy”. J Diabetes Sci Technol 2010;4:820–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Giovinco NA, Bui TD, Fisher T, Mills JL, Armstrong DG. Wound chemotherapy by the use of negative pressure wound therapy and infusion. ePlasty 2010;10:e9. [PMC free article] [PubMed] [Google Scholar]
- 12. Barr JE. Principles of wound cleansing. Ostomy Wound Manage 1995;41(7A Suppl.)):15S–21S discussion 22S. [PubMed] [Google Scholar]
- 13. Wysocki AB. Evaluating and managing open skin wounds: colonization versus infection. AACN Clin Issues 2002;13:382–97. [DOI] [PubMed] [Google Scholar]
- 14. Edlich RF, Rodeheaver GT, Thacker JG, Lin KY, Drake DB, Mason SS, Wack CA, Chase ME, Treble C, Long WB 3rd, Vassers RJ. Revolutionary advances in the management of traumatic wounds in the emergency department during the last 40 years: part I. Emerg Med 2010;38:40–50. [DOI] [PubMed] [Google Scholar]
- 15. Allen D, Labarbera LA, Bondre IL, Lessing MC, Rycerz AM Jr, Kilpadi DV, Collins BA, Perkins J, McNulty AK. Comparison of tissue damage, cleansing and cross‐contamination potential during wound cleansing via two methods: lavage and negative pressure wound therapy with instillation. Int Wound J 2012. DOI: 10.1111/j.1742-481X.2012.01073.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Saxena V, Hwang CW, Huang S, Eichbaum Q, Ingber D, Orgill DP. Vacuum‐assisted closure: microdeformations of wounds and cell proliferation. Plast Reconstr Surg 2004;114:1086–98. [DOI] [PubMed] [Google Scholar]
- 17.European Wound Management Association (EWMA). Position document. Wound bed preparation in practice. London: MEP Ltd, 2004. URL www.woundsinternational.com


