Abstract
An evidence-based triage plan for cellular therapy distribution is critical in the face of emerging constraints on healthcare resources. We evaluated the impact of treatment delays related to COVID-19 on patients scheduled to undergo hematopoietic cell transplantation (HCT) or chimeric antigen receptor T-cell (CAR-T) therapy at our center. Data were collected in real time between March 19 and May 11, 2020, for patients who were delayed to cellular therapy. We evaluated the proportion of delayed patients who ultimately received cellular therapy, reasons for not proceeding to cellular therapy, and changes in disease and health status during delay. A total of 85 patients were delayed, including 42 patients planned for autologous HCT, 36 patients planned for allogeneic HCT, and 7 patients planned for CAR-T therapy. Fifty-six of these patients (66%) since received planned therapy. Five patients died during the delay. The most common reason for not proceeding to autologous HCT was good disease control in patients with plasma cell dyscrasias (75%). The most common reason for not proceeding to allogeneic HCT was progression of disease (42%). All patients with acute leukemia who progressed had measurable residual disease (MRD) at the time of delay, whereas no patient without MRD at the time of delay progressed. Six patients (86%) ultimately received CAR-T therapy, including 3 patients who progressed during the delay. For patients with high-risk disease such as acute leukemia, and particularly those with MRD at the time of planned HCT, treatment delay can result in devastating outcomes and should be avoided if at all possible.
Key Words: Cellular therapy, COVID-19, Hematopoietic cell transplantation, Chimeric antigen receptor T cell therapy
INTRODUCTION
Cellular therapy, including hematopoietic cell transplantation (HCT) and chimeric antigen receptor T cell (CAR-T) therapy, is routinely used in the treatment of patients with hematologic malignancies and some solid tumors, such as germ cell tumors. In many aggressive malignancies, including the majority of acute leukemias and relapsed/refractory lymphomas or germ cell tumors, cellular therapy offers the best or only chance of cure 1, 2, 3. Importantly, the timing of cancer treatment impacts outcomes [4]; in the case of allogeneic HCT (allo-HCT), earlier referral and shorter time from diagnosis to transplantation are associated with reduced risk of relapse and improved survival 5, 6, 7.
Coronavirus disease (COVID-19) is an infectious disease caused by the 2019 novel severe acute respiratory syndrome coronavirus 2 (SARS-COV-2). The COVID-19 pandemic swept through New York City in the spring of 2020 and rapidly overwhelmed its healthcare systems. This had immediate implications for patients undergoing cellular therapies, whose hospital stays are routinely measured in weeks [8,9]. For patients undergoing allo-HCT, donor-related considerations added another layer of complexity. In response to anticipated challenges with collection and delivery, the National Marrow Donor Program (NMDP) required advanced graft collection and cryopreservation of stem cell products for patients undergoing HCT.
Memorial Sloan Kettering Cancer Center (MSKCC), like other medical centers in New York City, substantially restricted its ambulatory and surgical services during the height of the pandemic. Owing to immense constraints on healthcare resources, including intensive care unit (ICU) beds and blood products [10] in particular, coupled with concerns about the safety of patients undergoing cellular treatment because of their immunocompromised status and increased vulnerability to infectious complications, the number of cellular therapies performed between late March and May 2020 was dramatically reduced. We implemented a triage plan prioritizing cellular therapy in younger patients with minimal comorbidities and high-risk diseases, for whom the potential benefit of transplantation was felt to outweigh the risk of exposure to COVID-19, in line with that used at other major transplantation centers [8,9]. Specifically, we prioritized allo-HCT for young patients with acute leukemia with measurable residual disease (MRD) or primary induction failure or with high-risk myelodysplastic syndrome, autologous HCT (auto-HCT) done with curative intent for patients with high-risk myelomas and lymphomas, and CAR-T therapy in patients with progressive disease or substantial risk of progression who lacked other treatment options. This resulted in many HCT and CAR-T treatment delays.
Before and beyond the pandemic, natural disasters and mass emergencies that threaten normal operations are inevitable; in 2012, Superstorm Sandy jeopardized power, transportation, and telecommunication in the northeastern United States and led to the evacuation and closure of a neighboring transplantation unit in New York City [11]. In such resource-constrained settings, an evidence-based triage plan is a critical component of preparedness in the face of potential disaster. Our center has previously reported on determinants of COVID-19 disease severity in patients with cancer and on the outcomes of cellular therapy recipients who sustained COVID-19 infection [12,13]. As the pandemic has persisted and grown, we have sought to evaluate the early outcomes of patients delayed to cellular therapy in an effort to better prepare for a second and subsequent waves, as well as for other potentially catastrophic events.
METHODS
We aimed to evaluate the impact of treatment delays related to COVID-19 on patients who were scheduled to undergo cellular therapy. Data were collected prospectively between March 19 and May 11, 2020. All patients who either had an admission date scheduled or were undergoing workup with a plan for admission during this time of restricted cellular therapy were included in this analysis. We also followed patients who proceeded to cellular therapy during this timeframe without delay, to assess the risk of nosocomial COVID-19 infection and any other implications for their treatment during this time period. COVID-19 infection was diagnosed by real-time reverse transcription PCR as described previously [12,13].
Three months after cellular treatment activity resumed, we evaluated the proportion of delayed patients who ultimately received cellular therapy, reasons for not proceeding to cellular therapy, changes in disease and health status during the period of delay, and therapeutic strategies used while waiting. Decisions regarding postremission or maintenance therapy were made by each patient's physician on a case-by-case basis.
Baseline patient and disease characteristics and clinical outcome data were retrieved from the institutional database. Approval for this retrospective study was obtained from the MSKCC’s Institutional Review Board.
RESULTS
Total Cohort
We identified 85 patients who experienced delay of HCT or CAR-T during the initial COVID-19 pandemic surge. This included 42 patients (49.4%) scheduled to undergo auto-HCT, 36 (42.4%) patients planned for allo-HCT, and 7 (8.2%) patients planned for CAR-T therapy. Patient characteristics are displayed in Table 1 . At the time of this analysis, 56 patients (66%) had received the planned therapy, including 62% planned for auto-HCT, 67% planned for allo-HCT, and 86% planned for CAR-T therapy (Figure 1 ).
Table 1.
Characteristics of Patients Delayed to Cellular Therapy
| Characteristic | Value |
|---|---|
| Number of patients | 85 |
| Age, yr, median (range) | 63 (18-80) |
| Male sex, n (%) | 49 (58) |
| Autologous HCT, n | 42 |
| Diagnosis, n (%) | |
| Amyloid | 1 (2) |
| Germ cell tumor | 3 (7) |
| Multiple myeloma | 25 (60) |
| Lymphoma | 13 (31) |
| Disease status at delay, n (%)*,† | |
| CR | 19 (49) |
| VGPR | 15 (38) |
| PR | 3 (8) |
| SD | 1 (3) |
| PD | 1 (3) |
| Allogeneic HCT, n | 36 |
| Diagnosis, n (%)† | |
| Acute leukemia | 18 (50) |
| MDS | 5 (14) |
| MPN | 8 (22) |
| Other‡ | 5 (14) |
| Disease status at delay, n (%)† | |
| CR | 22 (61) |
| VGPR | 1 (3) |
| SD | 11 (31) |
| PD | 2 (6) |
| CAR-T therapy, n | 7 |
| Diagnosis, n (%) | |
| DLBCL | 6 (86) |
| Multiple myeloma | 1 (14) |
| Disease status at delay, n (%) | |
| CR | 2 (29) |
| PR | 3 (43) |
| SD | 1 (14) |
| PD | 1 (14) |
CR indicates complete remission; VGPR, very good partial response; PR, partial remission; SD, stable disease; PD, progressive disease;; MDS, myelodysplastic syndrome; MPN, myeloproliferative neoplasm; DLBCL, diffuse large B-cell lymphoma.a Germ cell tumor excluded.
Numbers may sum to >100% due to rounding.
Aplastic anemia, follicular lymphoma, multiple myeloma, monomorphic epitheliotropic intestinal T cell lymphoma, and T cell prolymphocytic leukemia.
Figure 1.
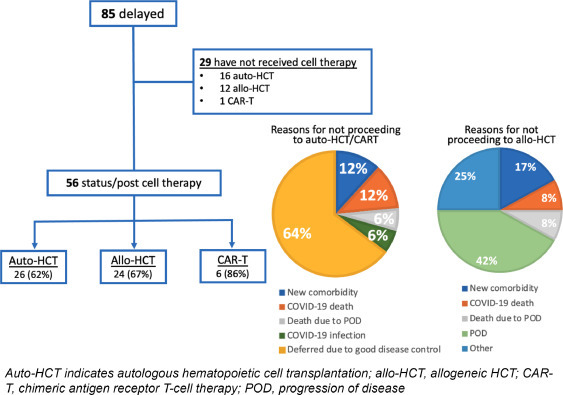
Outcomes of patients delayed to cellular therapy.
Six patients were infected with COVID-19 during the delay. Of the 85 patients, 5 died during the delay, 3 due to complications from COVID-19 and 2 from disease progression.
Auto-HCT
Forty-two patients planned for auto-HCT experienced delays, 26 (62%) of whom have since received the planned therapy. The median delay was 9 weeks (range, 1 to 21 weeks). In 34 patients (81%), maintenance strategies were used during this time. Details of the maintenance regimens are provided in Supplementary Table S1.
Among 26 patients with plasma cell dyscrasias, the median delay was 10 weeks (range, 4 to 21 weeks). Disease status at time of delay was complete remission (CR) or very good partial response (VGPR) in 22 patients (85%), partial response (PR) in 2 patients (8%), stable disease in 1 patient (4%),m and progressive disease in 1 patient (4%). MRD analysis was performed in nearly all patients, and decisions regarding delay were ultimately individualized based on age, comorbidities, MRD, disease status and kinetics, and patient and physician preference. Two patients with multiple myeloma, both of whom received maintenance treatment during the delay, experienced progression of disease. One of these patients progressed from VGPR and then underwent auto-HCT. The second patient experienced minimal progression in serum myeloma markers, but auto-HCT was ultimately deferred in light of the pandemic, and their disease remains in VGPR on maintenance.
Among the 13 patients with lymphoma, median delay was 7 weeks (range, 3 to 14 weeks). Disease status at time of delay was CR in 12 (92%) and PR in 1 (8%). No patient progressed during the delay.
All 3 patients with germ cell tumors had progressive disease at the time of delay. Because of these patients’ young age and the outpatient nature of these transplantations, delays were minimal, ranging from 0.5 to 1.5 weeks. All 3 patients were delayed as a precaution due to COVID-19 exposure (without infection). Two patients experienced an increase in tumor markers during a 1-week delay, but ultimately all patients received planned cellular therapy.
Among the 16 patients who had not received cellular therapy by the time of this analysis, 2 patients died of complications related to COVID-19. There were no deaths related to disease. Twelve patients, all with plasma cell dyscrasias, deferred cellular therapy because of good disease control. Two additional patients have been deferred for cellular therapy owing to ongoing pneumonitis related to COVID-19 in 1 patient, and the development of a new comorbidity in 1 patient (Figure 1).
Allo-HCT
Thirty-six patients planned for allo-HCT experienced delays, 24 (67%) of whom have since received the planned therapy. The median delay was 11 weeks (range, 5 to 24 weeks). Twenty-four patients (67%) received treatment or postremission therapy during this time aimed at preventing disease progression or relapse (Supplementary Table S1). The most common regimen received was a hypomethylating agent with venetoclax.
All 18 patients with acute leukemia were in morphologic remission at the time of delay, including 3 patients who were candidates for a second allo-HCT. In 12 patients (67%), there was no evidence of disease progression during the delay, and all have since undergone allo-HCT. The majority (11 of 12) received postremission therapy during the delay. Two patients (11%) experienced disease progression during the delay but eventually underwent allo-HCT after additional treatment. Four patients (22%) did not undergo HCT, including 1 patient who died due to complications related to COVID-19, 1 patient who developed prohibitive organ toxicity as a result of consolidation chemotherapy given during the delay, 1 patient who experienced disease progression precluding allo-HCT, and 1 patient who transferred care elsewhere and was lost to follow-up.
MRD was evaluated in 16 patients with acute leukemia (89%) using 10-color multiparameter flow cytometry with a sensitivity of approximately in 1 in 1000 events. Nine of these 16 patients were MRD-negative, none of whom progressed during the delay. In contrast, all 3 patients with acute leukemia who progressed during the delay were among the 7 patients with MRD. Of note, the patients with MRD, although recognized to be at high risk of disease progression, were delayed if they were considered at high risk for complications from COVID-19 or HCT given the limited number of ICU beds. Two of these 3 patients did not receive postremission therapy while waiting, both because of COVID-19 infection.
All 5 patients with myelodysplastic syndrome (MDS) had ≤5% blasts at the time of transplantation delay. Only 1 patient proceeded to allo-HCT without disease progression. Of the other 4, 2 underwent allo-HCT, 1 transitioned to hospice care owing to disease progression, and 1 died of disease. Three of the 5 patients with MDS received treatment during the delay, 2 of whom progressed despite treatment.
Among the 8 patients with myeloproliferative neoplasms, 6 (75%) have since undergone allo-HCT, only 1 of whom experienced progression during the delay. This patient, who had chronic myelogenous leukemia, experienced molecular progression despite treatment with a tyrosine kinase inhibitor. One patient with myelofibrosis has since deferred allo-HCT, and 1 patient transferred care to another center and was lost to follow-up.
Among the remaining 5 patients whose allo-HCT was delayed, 1 patient has since undergone allo-HCT without interim disease progression. Three patients experienced relapse or disease progression during the delay and are currently receiving further noncellular therapies, and 1 patient developed prohibitive organ toxicity and has not undergone allo-HCT.
As a result of the NMDP's early pandemic policy of graft collection and cryopreservation before the initiation of conditioning, a graft was collected from the donor and ultimately not used for 4 patients. Three of these grafts were from related donors and 1 was from an unrelated donor.
CAR-T Therapy
Seven patients planned for CAR-T therapy experienced delays. Six patients ultimately received cellular treatment, with a median delay of 5 weeks (range, 2 to 15 weeks). In 5 patients (71%), maintenance strategies were used during this time (Supplementary Table S1). One patient who would have proceeded to therapy under normal circumstances was deferred because of advanced age (80 years) and comorbidities in light of the circumstances and subsequently died of disease.
Five of the 7 patients were treated during the delay period. Three patients experienced disease progression during the delay, including 2 patients with diffuse large B cell lymphoma (DLBCL) who had been receiving chemotherapy during the delay and 1 patient with multiple myeloma who had been closely monitored.
Patients Who Were Not Delayed
Between March 19 and May 11, 2020, the period during which cellular therapy was severely restricted at MSKCC, 19 patients underwent cellular treatment as initially planned (Table 2 ). Four patients underwent allo-HCT (3 for acute leukemia or MDS with excess blasts and 1 for multiply relapsed T cell lymphoma), 3 patients received CAR-T therapy (all for relapsed DLBCL), and 12 patients underwent auto-HCT (5 for germ cell tumor, 4 for lymphoma and 3 for myeloma). Four of the auto-HCTs were performed on an outpatient basis.
Table 2.
Characteristics of Patients who Proceeded to Cellular Therapy without Delay
| Characteristic | Value |
| Number of patients | 19 |
| Age, yr, median (range) | 46 (22-73) |
| Male sex, n (%) | 10 (53) |
| Autologous HCT, n | 12 |
| Diagnosis, n (%) | |
| Germ cell tumor | 5 (42) |
| Multiple myeloma | 3 (25) |
| Lymphoma | 4 (33) |
| Disease status at cell therapy, n (%)* | |
| CR | 4 (57) |
| PR | 2 (29) |
| PD | 1 (14) |
| Allogeneic HCT, n | 4 |
| Diagnosis, n (%) | |
| Acute leukemia | 2 (50) |
| MDS | 1 (25) |
| Lymphoma | 1 (25) |
| Disease status at cell therapy, n (%) | |
| CR | 3 (75) |
| SD | 1 (25) |
| CAR-T therapy, n | 3 |
| Diagnosis, n (%) | |
| DLBCL | 3 (100) |
| Disease status at cell therapy, n (%) | |
| PR | 2 (67) |
| PD | 1 (33) |
MDS indicates myelodysplastic syndrome; MPN, myeloproliferative neoplasm; DLBCL, diffuse large B-cell lymphoma, CR, complete remission; PR, partial remission; SD, stable disease; PD, progressive disease.a Germ cell tumor excluded.
Nosocomial COVID-19
One concern leading to delayed cellular therapy was the risk of exposing patients, particularly those of older age and/or with significant comorbidities, to COVID-19. At the height of the initial COVID-19 epidemic in New York City in March 2020, contact tracing was performed for all possible staff and patient exposures to assess the risk of nosocomial transmission. Three COVID-19-positive staff members exposed 27 patients on the inpatient transplant units in March, with an overall attack rate of secondary transmission from health care worker to patient of 3.7% (1 of 27). The patient who tested positive for SARS-CoV2 RNA by PCR at 12 days after the last exposure was considered a probable transmission, because the patient had been admitted several weeks earlier to visitor access restrictions. Among outpatients in the apheresis center, 9 patients were exposed to 2 COVID-19 positive staff members. Two patients subsequently tested positive at 9 and 13 days from last staff contact, but patient-staff interactions in the ambulatory setting were thought to be minimal, with staff using surgical masks for direct patients care, and wide-spread regional community transmission precludes definite conclusions on the risk of transmission.
Importantly, all of these exposures occurred before initiation of visitation restrictions, surveillance swabs for inpatients every 72 hours, regular asymptomatic testing for staff, and universal staff masking and eye protection, both inpatient and outpatient. Similar to the literature [14], since April 2020 when these strategies were implemented, we have not had any known nosocomial transmission among the HCT and CAR T cell patients.
DISCUSSION
Cases of COVID-19 were first reported in the United States in January 2020, and by March 2020, the pandemic forced rapid and drastic changes to New York City's healthcare systems to accommodate the rapidly rising numbers of patients in need of hospital beds in general and ICU beds in particular and the decreased availability of blood products. In addition, clinical research activity shut down, adding another layer of complexity for patients who were only eligible for treatment on a clinical trial [15]. Early on, data on outcomes of COVID-19 infection in immunocompromised patients and on the impact of cellular therapy treatment delays were lacking, and decisions regarding cellular therapy policies and practices had little evidence to guide them. Our center has since reported overall favorable clinical outcomes in recipients of cellular therapy who sustained COVID-19 infection while in remission [12], although recognizing the limitation of a small and heterogenous cohort. Now, after several months of close monitoring of outcomes in patients in whom life-saving cellular therapy was delayed, we report our findings and recommendations as further viral waves emerge throughout the country.
While acknowledging the limitations of a single-center analysis, we observed the following among our cohort of patients delayed to cellular therapy: (1) the majority of patients with plasma cell dyscrasias in CR or VGPR at time of delay successfully deferred cellular therapy in favor of maintenance therapy until the city-wide positivity rate improved; (2) all patients with acute leukemia who progressed had MRD at the time of delay, whereas none of the patients without MRD at the time of delay progressed; (3) among patients with acute leukemia who did not progress during the delay and subsequently underwent allo-HCT, nearly all received postremission therapy during the delay, but conversely, 2 of the 3 patients with acute leukemia who progressed during the delay did not receive maintenance therapy; (4) the majority of patients awaiting allo-HCT who progressed during the delay did not undergo allo-HCT, regardless of the underlying diagnosis; (5) there were instances in this cohort in which consolidation therapy given during the delay was deleterious and resulted in organ toxicity that prevented cellular therapy once we resumed normal operations, demonstrating that maintenance strategies are not without cost; and (6) with the proper use of personal protective equipment and aggressive surveillance, the risk of nosocomial COVID-19 infection was extremely low.
From this, we conclude that for patients with high-risk malignancies, particularly those with leukemia and MRD at the time of planned HCT, treatment delay resulted in devastating outcomes and should be avoided if at all possible. When delays are absolutely necessary, this experience suggests that interventions to keep disease control (postremission therapy or continued treatment when applicable) enabled patients to bridge to cellular therapy and thus should be considered. We find the low risk of nosocomial transmission reassuring and believe that fear of contracting COVID-19 in the healthcare setting should not be a barrier to timely cellular therapy. Furthermore, given the lack of known nosocomial transmissions in our apheresis center since the implementation of rigorous infection control measures, and considering that apheresis yield can be compromised in patients receiving chemotherapy, we recommend upfront collection of peripheral blood stem cells and/or apheresis for CAR-T therapy even when cellular treatment is delayed.
Long-term follow-up will be needed to evaluate whether patients who experienced delays but ultimately received cellular therapy experienced inferior outcomes compared with patients who were never delayed, and whether patients with plasma cell dyscrasias who deferred auto-HCT achieved similar outcomes as patients who proceeded with consolidative auto-HCT without delay. It also must be noted that our institution, a dedicated cancer center, does not have an open emergency room and thus was uniquely positioned, with significantly fewer patient admissions compared with neighboring hospitals. Nonetheless, we believe that our findings will prove especially useful to institutions facing severe constraints on healthcare resources and needing a triage plan for cellular therapy distribution.
The adverse outcomes observed in our series among patients with delayed cellular therapy underscores that the impact of the COVID-19 pandemic on mortality extends far beyond the reported deaths directly attributable to the virus.
ACKNOWLEDGMENTS
The authors thank Theodore and Laura Hromadka for their support.
Financial disclosure: This research was supported in part by National Institutes of Health Grant P01 CA23766 and National Cancer Institute Cancer Center Support Grant P30 CA008748. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Conflict of interest statement: We know of no conflicts of interest associated with this publication, however all financial disclosures for authors are listed as follows: DRF - Astellas, Decibel, Novartis, Seattle Genetics and UpToDate; CC – AlloVir; PBD – Kite Pharmaceuticals; SAG – Amgen, CRISPR Therapeutics AG, CSL Behring, Caladrius, Celgene, Ceramedix; ExpertConnect; GlaxoSmithKline; Janssen Research & Development, LLC, Karyopharm, Kite Pharmaceuticals, Mangolia Innovation, Novartis, Omeros, Pfizer Inc, Physicians' Education Resource, Sanofi US Services Inc, TRM Oncology, and Xcenda; BG – Actinium; OBL – MorphoSys AG; HJL – Takeda, Celgene, Janssen, and Caelum; SM – June, Celgene, Bristol-Myers Squibb, Janssen Oncology, Takeda Oncology, Allogene Therapuetics, Elsevier Publications, and Physician Education Resource; MLP – Novartis, Merck & Co Inc, Pharmacyclics, and Kite; IP – ExCellThera and Merck & Co Inc; DMP – CareDx, Generon (Shanghai) Corporation Ltd, Gerson Lehrman Group, Guidepoint Global Advisors, Kadmon Holdings, Inc, and Slingshot Insights; CSS – Juno Therapeutics, Sanofi-Genzyme, Spectrum Pharmaceuticals, Novartis, Genmab, Precision Biosciences, Kite – A Gilead Company, Celgene, and GlaxoSmithKline; MS – Angiocrine Bioscience, Kite Pharmaceuticals, McKinsey & Company, Omeros, and i3 Health; MRMV – CRYOSTEM, DKMS, Duke-NUS Medical School, European Federation for Immunogenetics, European Society for blood and Marrow Transplantation, Falk Foundation e.V., Fred Hutchinson Cancer Research Center, French National Research Agency, Juno Therapeutics, King Faisal Specialist Hospital & Research Centre, Lille University Hospital (Chu De Lille), Magenta, Ospedale Pediatrico Bambino Gesu, Parker Institute for Cancer Immunotherapy, Seres Therapeutics, Technical University of Denmark, the Korean Society of Blood and Marrow Transplantation, The Meredith A. Cowden Foundation, University of Copenhagen, and Xinqiao Hospital; MAP: AbbVie, Bristol-Myers Squibb, Cidara Therapeutics, Inc., Incyte, Merck & Co Inc., Miltenyi Biotec Incorporated, Nektar Therapeutics, NexImmune, Novartis, and Omeros. MTN, GLS, JDR, EVR, AAA, MD, MK, JNB, DJC, RJL, EBP, BCS, and RT have no disclosures.
Authorship statement: M.T.N. and R.T. contributed to the literature search and study design; M.T.N., R.T., and J.D.R. performed data collection; M.T.N., M.A.P., and R.T. created the figures; M.T.N., J.N.B., D.J.C., P.B.D., S.A.G., H.J.L., B.G., M.A.P., G.J.L., E.V.R., A.A.A., M.D., M.K., and R.T. contributed to data analysis and interpretation; M.T.N., R.T., G.L.S., D.R.F., E.V.R., M.K., A.A.A., and M.D. wrote the original manuscript; O.B.L., S.M., M.L.P., D.M.P., C.S.S., M.R.M.V., R.J.L., E.B.P., I.P., B.C.S., M.S., C.C., and M.A.P. reviewed and edited the manuscript; and M.T.N., R.T., and J.D.R. verified the underlying data.
Footnotes
Financial disclosure: See Acknowledgments on page 438.e5.
Supplementary material associated with this article can be found in the online version at doi:10.1016/j.jtct.2021.02.011.
Appendix. Supplementary materials
Supplemental Table 1: Therapeutic Regimens Utilized During Delay.
REFERENCES
- 1.Appelbaum FR. Hematopoietic-cell transplantation at 50. N Engl J Med. 2007;357:1472–1475. doi: 10.1056/NEJMp078166. [DOI] [PubMed] [Google Scholar]
- 2.Cappell KM, Sherry RM, Yang JC, et al. Long-term follow-up of anti-CD19 chimeric antigen receptor T-cell therapy. J Clin Oncol. 2020;38:3805–3815. doi: 10.1200/JCO.20.01467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Feldman DR, Sheinfeld J, Bajorin DF, et al. TI-CE high-dose chemotherapy for patients with previously treated germ cell tumors: results and prognostic factor analysis. J Clin Oncol. 2010;28:1706–1713. doi: 10.1200/JCO.2009.25.1561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hanna TP, King WD, Thibodeau S, et al. Mortality due to cancer treatment delay: systematic review and meta-analysis. BMJ. 2020;371:m4087. doi: 10.1136/bmj.m4087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee SJ, Klein J, Haagenson M, et al. High-resolution donor-recipient HLA matching contributes to the success of unrelated donor marrow transplantation. Blood. 2007;110:4576–4583. doi: 10.1182/blood-2007-06-097386. [DOI] [PubMed] [Google Scholar]
- 6.Pidala J, Lee SJ, Ahn KW, et al. Nonpermissive HLA-DPB1 mismatch increases mortality after myeloablative unrelated allogeneic hematopoietic cell transplantation. Blood. 2014;124:2596–2606. doi: 10.1182/blood-2014-05-576041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kim H, Lee KH, Yoon SS, et al. Allogeneic hematopoietic stem cell transplant for adults over 40 years old with acquired aplastic anemia. Biol Blood Marrow Transplant. 2012;18:1500–1508. doi: 10.1016/j.bbmt.2012.03.015. [DOI] [PubMed] [Google Scholar]
- 8.Bachanova V, Bishop MR, Dahi P, et al. Chimeric antigen receptor T cell therapy during the COVID-19 pandemic. Biol Blood Marrow Transplant. 2020;26:1239–1246. doi: 10.1016/j.bbmt.2020.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Algwaiz G, Aljurf M, Koh M, et al. Real-world issues and potential solutions in hematopoietic cell transplantation during the COVID-19 pandemic: perspectives from the Worldwide Network for Blood and Marrow Transplantation and Center for International Blood and Marrow Transplant Research Health Services and International Studies Committee. Biol Blood Marrow Transplant. 2020;26:2181–2189. doi: 10.1016/j.bbmt.2020.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pagano MB, Hess JR, Tsang HC, et al. Prepare to adapt: blood supply and transfusion support during the first 2 weeks of the 2019 novel coronavirus (COVID-19) pandemic affecting Washington State. Transfusion. 2020;60:908–911. doi: 10.1111/trf.15789. [DOI] [PubMed] [Google Scholar]
- 11.Thewes K, Wuckovich M, DeRiso R, Blitzstein S. Emergency evacuation of a bone marrow transplant unit during Superstorm Sandy: this was not a drill. Biol Blood Marrow Transplant. 2014;20:S310–S311. [Google Scholar]
- 12.Shah GL, DeWolf S, Lee YJ, et al. Favorable outcomes of COVID-19 in recipients of hematopoietic cell transplantation. J Clin Invest. 2020;130:6656–6667. doi: 10.1172/JCI141777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Robilotti EV, Babady NE, Mead PA, et al. Determinants of COVID-19 disease severity in patients with cancer. Nat Med. 2020;26:1218–1223. doi: 10.1038/s41591-020-0979-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rhee C, Baker M, Vaidya V, et al. Incidence of nosocomial COVID-19 in patients hospitalized at a large US academic medical center. JAMA Netw Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.20498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Unger JM, Blanke CD, LeBlanc M, Hershman DL. Association of the coronavirus disease 2019 (COVID-19) outbreak with enrollment in cancer clinical trials. JAMA Netw Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.10651. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Table 1: Therapeutic Regimens Utilized During Delay.


