Abstract
Thalassemias are inherited blood disorders that are found in high prevalences in the Mediterranean, Southeast Asia and the Pacific. These diseases provide varying levels of resistance to malaria and are proposed to have emerged as an adaptive response to malaria in these regions. The transition to agriculture in the Holocene has been suggested to have influenced the selection for thalassemia in the Mediterranean as land clearance for farming encouraged interaction between Anopheles mosquitos, the vectors for malaria, and human groups. Here we document macroscopic and microscopic skeletal evidence for the presence of thalassemia in both hunter-gatherer (Con Co Ngua) and early agricultural (Man Bac) populations in northern Vietnam. Firstly, our findings demonstrate that thalassemia emerged prior to the transition to agriculture in Mainland Southeast Asia, from at least the early seventh millennium BP, contradicting a long-held assumption that agriculture was the main driver for an increase in malaria in Southeast Asia. Secondly, we describe evidence for significant malarial burden in the region during early agriculture. We argue that the introduction of farming into the region was not the initial driver of the selection for thalassemia, as it may have been in other regions of the world.
Subject terms: Archaeology, Biological anthropology, Anaemia
Introduction
Thalassemias, a group of inherited hemoglobin blood disorders1, are highly prevalent in present day Southeast Asia and the Pacific. These diseases stand testament to a deep history of genetic adaptation to the parasitic disease malaria2,3. However, it is unknown when thalassemias or malaria originated in the region. The two forms of the disease, alpha and beta thalassemia, cause disruptions to the synthesis of the alpha or beta hemoglobin chains respectively. Malformation of hemoglobin results in an excess of the opposing hemoglobin chain which is the cause of disease in the body4,5. Today, malaria afflicts approximately 8 million people in Southeast Asia. Eradication efforts are sporadic and reliant on access to treatment at a community level to be effective6. Thalassemias, like other hemoglobin disorders, disrupt the mechanism for malarial parasite-binding to red blood cells2. In Southeast Asia where thalassemia genes are observed in high frequencies, in as much as over 75% of the population, malaria is also highly endemic, particularly the most lethal form Plasmodium falciparum3.
Although most variants of alpha thalassemia provide resistance to malaria with little clinical complication, most homozygous beta thalassemia variants (thalassemia major and intermedia) can have significant effects on health. Complications of beta thalassemia include gross changes to the skeleton and death from infection or iron overload3,7,8. In Southeast Asia specifically, a milder form of beta thalassemia, the hemoglobin-E (HbE) variant, is also present in high frequencies, with the gene present in up to 30–50% of the population in some geographical areas9. Homozygous HbE is not associated with clinical symptoms. However, complications can occur with co-inheritance of HbE and classical beta thalassemia variants (HbE beta thalassemia)7,10. HbE beta and classic beta thalassemias are frequently found co-inherited with alpha thalassemia in Southeast Asia and result in a wide spectrum of symptoms and severity11. Modern day research demonstrates that variants within Southeast Asia and the Pacific can vary across short distances, and relate to the ecological ranges of Anopheles mosquitos, the vectors for malarial parasites3. Given the co-evolutionary relationship that continues to drive high frequencies of thalassemia in the Southeast Asian region today, it can be hypothesized that the identification of thalassemia in the prehistoric record can be, at least in part, related to a prior selection pressure of malaria.
To date, the only archaeological evidence for hereditary anemia in the region dates from approximately 4000–3500 BP from Khok Phanom Di, a Neolithic site in central Thailand (Fig. 1)12. In the Mediterranean region, where beta thalassemia is similarly frequent, skeletal evidence in prehistoric assemblages supports the emergence of the disease with the transition to agriculture (the Neolithic) from approximately 7000 years ago13. It is hypothesized that agricultural practices such as land clearance encouraged contact between human groups and Anopheles mosquitos13,14. Did these conditions then emerge alongside the spread of agriculture in Southeast Asia as is proposed to be the case in the Mediterranean?
Figure 1.
Map of Southeast Asia with sites important for this study. Khok Phanom Di in Thailand provides the only prehistoric evidence of thalassemia in the region. Our study includes research from Man Bac and Con Co Ngua in northern Vietnam.
Modified from image by Koba-chan (https://upload.wikimedia.org/wikipedia/commons/1/15/Topographic30deg_N0E90.png) created from DEMIS Mapserver (http://www2.demis.nl/worldmap/mapper.asp). Published under CC BY-SA 3.0 (https://creativecommons.org/licenses/by-sa/3.0/deed.en).
The emergence of farming in Mainland Southeast Asia (MSEA) occurred much later than the Mediterranean, from only after 4500 years ago15, with most Neolithic sites post-dating 4,000 BP. Prior to this time the region was occupied by indigenous Pre-Neolithic foragers descended from the first people out of Africa and into Asia16. By the early seventh millennium BP some forager groups in northern Vietnam and southern China developed large sedentary settlements, at the same time as agriculture was practiced to the north in China17. The subsequent adoption of agriculture in MSEA during the Neolithic (4500–3100 BP) was associated with multiple migration events of farmers from what is now geo-politically southern China, coming into contact with these forager groups15,18,19. These subsistence transition and migration events significantly altered the demography and genetics of this region from this point forward. The aim of this research is to investigate, for the first time, whether thalassemia, as a proxy for malaria burden, was present prior to agriculture in northern Vietnam, and to what degree the transition to agriculture may have contributed to the emergence of thalassemia in Southeast Asia. Additionally, if thalassemia is observed we aim to further investigate which variants may have been present.
We applied a diagnostic protocol based on macroscopically observed dry bone lesions for thalassemia to two archaeological human skeletal assemblages. The Pre-Neolithic site of Con Co Ngua radiocarbon dated to at minimum 6200–6700 cal BP17,18 represented a pre-agricultural but sedentary forager community (n = 155; Fig. 1). The Man Bac site dating to 3,906–3,523 cal BP was occupied during the agricultural transition of Southeast Asia (n = 70). This site captures co-habitation of indigenous forager and migrant farmers during the early stages of the agricultural transition18. To support the strength of the disease diagnosis using the macroscopic methods, we also conducted histological analysis on three Con Co Ngua individuals to assess microscopic pathological changes in affected bone.
Results
Macroscopic results present strong evidence for thalassemia at Man Bac
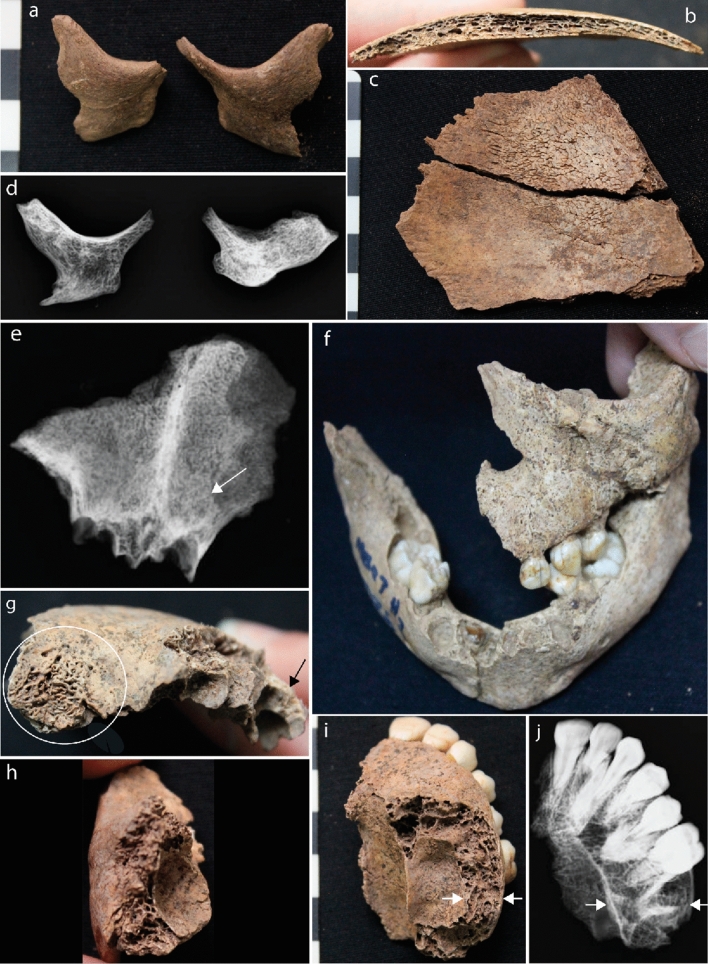
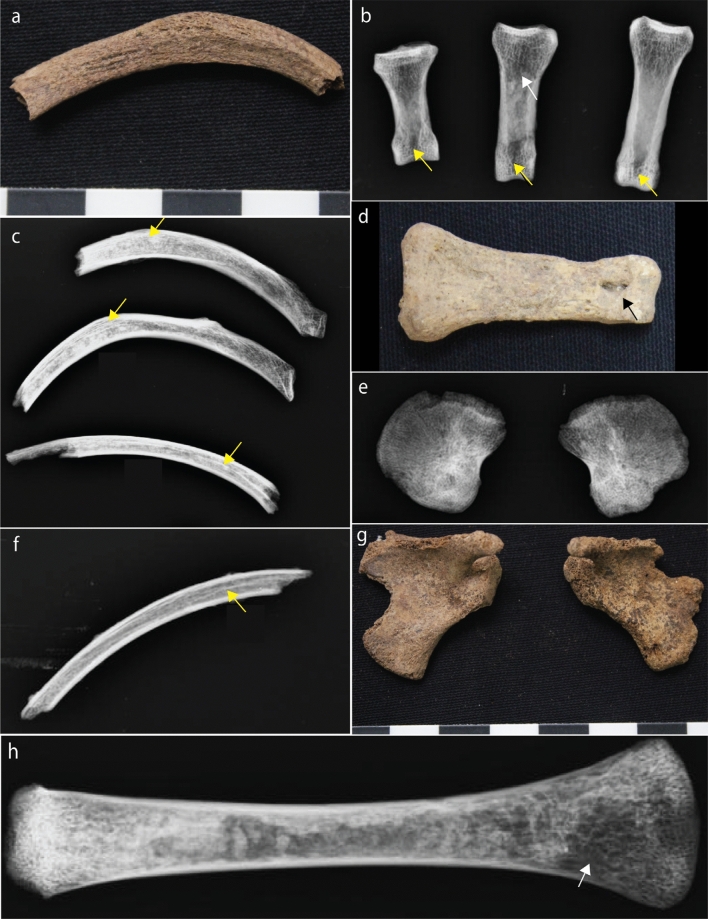
Five children aged between 6 months and 12 years and one adult presented with skeletal changes that are strongly diagnostic (pathognomonic) for thalassemia (Table 1, Supplementary Text S1; Supplementary Table S1; Figs. 2, 3). These changes included rodent facies, a skeletal condition specific to thalassemia, where expansion of the marrow results in a bulbous face and mandible. Radiographs confirm marrow expansion, a clinical consequence of severe thalassemia (Fig. 2). Radiographic ‘rib-within-a-rib sign’, again pathognomonic for thalassemia, was identified in three individuals. This condition results from extensive marrow expansion within the shafts of the ribs. Further evidence for thalassemia included a lack of the development of facial and cranial sinuses, commonly caused by extensive marrow expansion of the face since infancy. Two children also presented with severe porosity of the orbits and the endocranium, likely due to marrow expansion through the cortical margins of the skull, as a result of thalassemia. Additionally, a newborn with multiple lesions suggestive of possible thalassemia, including evidence of marrow expansion throughout the skeleton was identified (Table 1, Figs. 2, 3).
Table 1.
Macroscopic and radiographic diagnosis of thalassemia in Man Bac individuals.
| Individual ID | MB07H1M8 | MB07H1M12* | MB07H2M26 | MB05M12 | MB05M3 | MB07H1M1 |
|---|---|---|---|---|---|---|
| Age (years) | 30–39 | 0 | 1.5 | 2 | 0.5 | 12 |
| Sex | Male | N/A | N/A | N/A | N/A | N/A |
| Diagnostic criteria | ||||||
| Marrow hyperplasia of the facial bones: maxillae-leading to ventral displacement of central incisors, zygomatic bones-leading to orbital displacement, and/or mandible (rodent facies deformity) (SD) | A | N/A | P | P | P | P |
| Radiographic: “rib-within-a-rib” appearance. Radiographically defined sclerotic bands within the ribs due to extramedullary hematopoiesis (SD) | P | A | P | P | A | |
| Poor or lack of pneumatization of the paranasal and cranial sinuses sparing the ethmoid sinuses (D) | A | N/A | P | A** | A | P |
| Enlarged tubular bones of the hands and feet due to marrow hyperplasia (infants) sometimes associated with enlarged nutrient foramina or Radiographic: coarse trabecular patterns of the hands or feet, sometimes associated with cyst-like lucencies due to focal collection of hyperplasic marrow (D) | P | N/A | N/A | N/A | A | N/A |
| Premature fusion of epiphyseal plates particularly of the proximal humerus and distal femur, often causing short long bone maximum length (D) | A | A | A | A | A | N/A |
| Widening of entire rib, or widening of the rib head and neck with pronounced bulbous appearance posteriorly (costal osteomas). Associated with radiograph appearance of erosion of the inner cortex (D) | P | A | A | A | P | A |
| Diagnosis | Probable | Possible | Probable | Probable | Probable | Probable |
P = present, A = absent, N/A = skeletal element missing/unobservable.
*MB07H1M12 presented with multiple suggestive lesions, but no diagnostic (D) or strongly diagnostic (SD) lesions (see Supplementary Table S1).
**Frontal sinuses were not preserved, absence of pneumatization based on preservation of maxillary sinuses only.
Figure 2.
Cranial evidence for thalassemia at Man Bac. (a) Anterior protrusion of the zygomatic bones consistent with rodent facies (MB05M3, approx. 6 months old, antero-lateral aspect). (b) and (c) Diploic expansion of the cranial vault. There is no porosity on the ectocranium but hair-on-end formations are present on the endocranium (MB05M12, approx. 2 years, lateral aspect). (d) Marrow hyperplasia of the zygomatic bones (MB05M12, antero-posterior view). (e) Lack of pneumatization of the frontal sinus (MB07H1M1, approx. 12 years, antero-posterior view). (f) Rodent facies of the maxilla, mandible and zygoma (MB07H2M26, approx. 1.5 years, antero-superior aspect). (g) Severe cribra orbitalia (white circle) and diploic expansion of the crania (black arrow, antero-lateral aspect) (MB07H1M1). (h) Marrow hyperplasia of the left zygoma (MB07H1M1, lateral aspect). (i) and (j) Marrow hyperplasia of the maxilla (MB07H1M1, superior-inferior view). The expanse of the marrow hyperplasia is indicated by the white arrows.
Figure 3.
Postcranial evidence for thalassemia at Man Bac. (a) Enlarged rib (MB05M3, approx. 6 months old, superior aspect). (b) and (d) Expanded foramina of the phalanges (yellow arrows) with marrow hyperplasia (white arrow, MB07H1M8, middle aged adult, antero-posterior view). (c) “Rib-within-a-rib” sign (yellow arrows, MB07H1M8, supero-inferior view). (e) Alteration of the trabecular structure of the ilia. Note the radiating pattern (MB07H1M12, neonate, antero-posterior view). (f) “Rib-within-a-rib” sign (yellow arrow, MB07H2M26, approx. 1.5 years, supero-inferior view). (g) Enlargement of the scapular spines (MB07H1M12, neonate, posterior aspect). (h) Marrow hyperplasia of the humerus (white arrow, MB05M12, approx. 2 years, antero-posterior view).
Macroscopic results present evidence suggestive but not diagnostic for thalassemia at Con Co Ngua
We identified seven adults and adolescents at Con Co Ngua presenting with macroscopic and radiographic ‘bone-within-a-bone’ changes of the limb bones (Supplementary Fig. S1; Supplementary Table S1). We observed a mixture of enlargement and restriction of the medullary canal areas in these individuals. In thalassemia, this skeletal change is caused by marrow expansion within the long bones perforating the outer cortex. As such, these pathologies are suggestive of thalassemia but alone are not diagnostic for thalassemia, as they can also be found in a number of other chronic conditions20 (Supplementary text S1). However, these skeletal changes were similar to those with strong diagnostic evidence of thalassemia at Man Bac. We extracted bone samples from three of the CCN individuals for further microscopic analysis.
Combined microscopic and macroscopic results strongly suggest thalassemia at Con Co Ngua
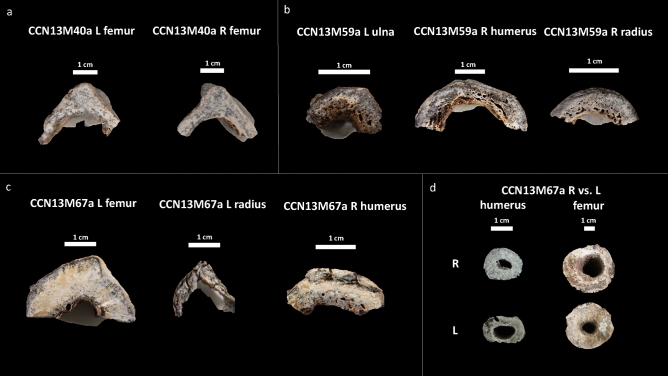
We assessed bone samples of one adolescent (> 16 years) of unknown sex (CCN13M67a), and two adults: CCN13M40a (young adult female), CCN13M59a (old adult male) (Fig. 4). We focused specifically on the endocortex (cortical bone located on the inner third portion of the cross-section) as preliminary macroscopic assessment of the samples prior to microscopy determined this region to be particularly important (Fig. 4). Widespread pores were evident on the endocortical surfaces of samples from the old adult male (CCN13M59a). The porosity consists of ‘giant’ pores that appear to have been created as a result of adjacent coalescing pores (Figs. 4, 5). All three bones of this individual showed a “trabecularization effect”, which essentially transformed cortical bone into a trabeculae-like matrix of bone as a result of prolonged resorption, and possibly marrow expansion.
Figure 4.
Cross section of microscopic samples from Con Co Ngua. (a) Femora sections from CCN13M40a. There is increased cortical thickness, and the cortical width of right femur is asymmetrically wider than the left. (b) Upper limb sections from CCN13M40a. Large endocortical pores are evident. Medullary canal widening is most distinct on the humerus. (c) Upper and lower limb sections from CCN13M67a. While the femur presents with extreme cortical thickness and restriction of the medullary canal area, the upper limb sections present with severe porosity of the endocortical surfaces. (d) Asymmetry of the medullary canals of the humeri and femora of CCN13M67a.
Figure 5.
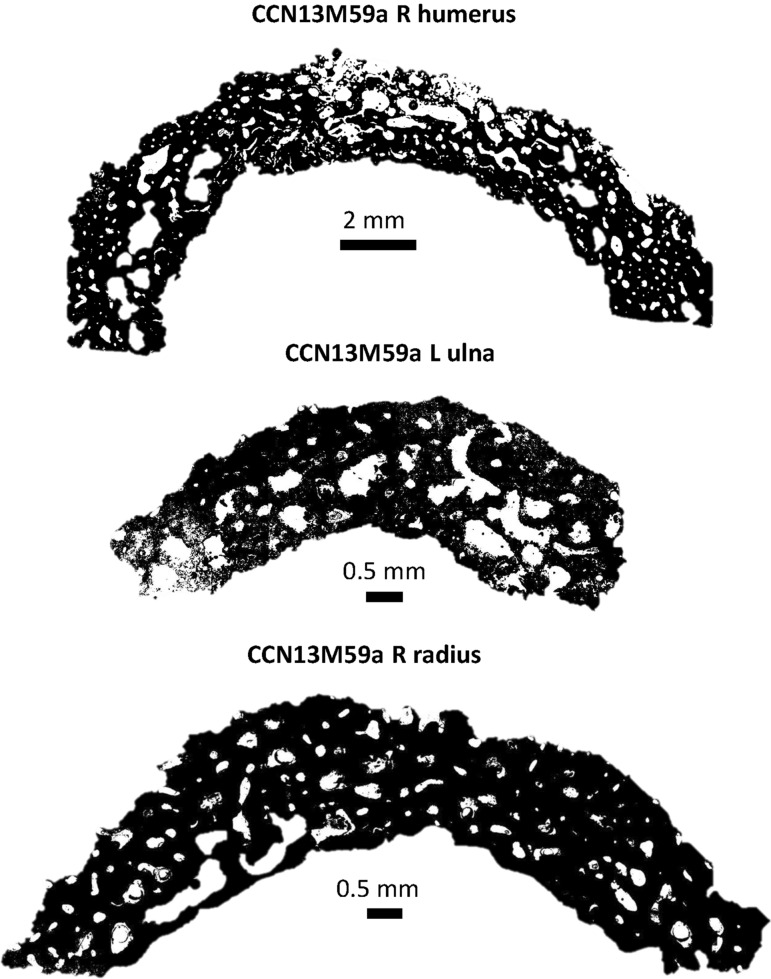
Macroporosity observed in the arm bones of CCN13M59a. The porosity is large and coalescing.
In contrast, the bone samples in the young adult female (CCN13M40a), showed denser and enlarged endocortical surfaces. The adolescent (CCN13M67a) presented with a mix of large porous regions of the endocortex (right humerus) and regions of increased cortical density (femur). Possible beginnings of the ‘trabecularization effect’ such as those observed in the old adult male (CCN13M59a) were found in the right humerus of the old adult. The extent of medullary bone size reduction can be seen, particularly in the adolescent femur section where the cortical wall is unusually enlarged (Fig. 4d). This femur showed evidence for secondary bone remodeling confirming cyclical replacement of old bone with new bone tissue, possibly driven by increased metabolic bone needs (Fig. 6). There was a clear pattern whereby samples from the femur did not show evidence for widespread porosity and coalescing of bone pores, but were observed in the bones of the upper limb (ulna, humerus, and radius) (Fig. 6).
Figure 6.
Regions of bone captured from the endocortical surface in the adolescent (CCN13M67a), allowing to examine the degree of bone modelling and remodeling. (a–d) The femur: Secondary osteons (SO), primary osteons (PO), Haversian canals (HC), and osteocyte lacunae (OL) can be seen. (a) White arrows point to endosteal lamellar layers which are typical for this bone region. (c) White arrows point to a cement line of a secondary osteon that indicates a remodeling event of a fragmentary osteon (FO) underneath, confirming cyclical replacement of old bone with new bone tissue, possibly driven by increased metabolic bone needs. (e) White arrows point to primary lamellar bone layers and an isolated HC. (f) A SO amongst primary bone.
We compared the microscopic results to various diseases of infectious and metabolic etiologies known to cause the macroscopic ‘bone-within-a-bone’ sign (Supplementary text S1). The microscopic outcomes of Con Co Ngua are only consistent with clinical bone histological observations in cases of beta thalassemia. Increased osteoclast-mediated resorptive activity, and decreased osteoblastic activity have both been described as underlying processes that increase porosity of the endocortical margins of the long bones in beta thalassemia21. In thalassemia, skeletal changes can be localized, due to focal deposits of iron during iron overload which is consistent with the variation between the upper and lower limbs we observed across the samples21–23. The overall histological bone pattern observed in the Con Co Ngua individuals therefore supports localized metabolic changes consistent with thalassemia.
Discussion
What hemoglobinopathy variants were present in prehistoric Vietnam?
Our results demonstrate skeletal evidence for thalassemia from the early seventh millennium BP in northern Vietnam. The gross skeletal changes in all but one Man Bac individual are consistent with beta thalassemia4. The clinical consequences, including bone changes, of beta thalassemia only develop following replacement of fetal gamma hemoglobin with adult beta hemoglobin in the months following birth. However, the possible case diagnosed in a Man Bac newborn may be consistent with alpha thalassemia major (Bart’s hydrops fetalis)24. Alpha thalassemia major, caused by deletion of all of the four alpha alleles, is a common variant in Southeast Asia today25. There is little clinical description of the skeletal complications of alpha thalassemia major due to its fatal outcome. However, severe bone marrow expansions occur in utero during growth detected as early as 18 fetal weeks7, and skeletal complications similar to beta thalassemia major can be expected24. Severe growth stunting is a common outcome of alpha thalassemia major, and skeletal deformities have been clinically documented, but not described5,7.
Evidence for co-occurrence of both alpha and beta thalassemia variants at Man Bac is of evolutionary significance. The gross skeletal pathologies of Man Bac post-birth individuals are commonly observed in patients with beta thalassemia major, a severe form that requires removal of excess iron and blood transfusions in order to survive past infancy26. However, 57% (4/7) of individuals macroscopically diagnosed with thalassemia at Man Bac were older than 1-year of age, having survived infancy without treatment. Alpha and beta thalassemia co-inheritance is known to result in a milder form of disease than beta thalassemia major due to a balance of depleted alpha and beta hemoglobins27. Additionally, the Southeast Asian variant HbE beta thalassemia commonly results in severe forms of thalassemia that do not require blood transfusions to survive (known clinically as thalassemia intermedia)28. It is possible that co-inheritance of beta thalassemia with alpha and/or HbE may account for the severe skeletal changes in individuals at Man Bac who survived past infancy.
It is not possible to determine whether the Con Co Ngua individuals had beta or alpha thalassemia. While there is clear clinical recognition of the skeletal changes of beta thalassemia29, HbH alpha thalassemia caused by deletion of three of the four alpha alleles are reported to cause mild to moderate skeletal deformities in some patients5. Significant facial deformity was not recorded at Con Co Ngua. However, infants at this site were very poorly preserved, so more severe forms of thalassemia at Con Co Ngua cannot be ruled out.
Evidence for deep antiquity of thalassemia and malaria in Mainland Southeast Asia
Based on our macroscopic and microscopic observations we suggest that thalassemia was potentially a considerable burden for Southeast Asian populations prior to the adoption of farming. This contrasts with our current knowledge on the emergence of thalassemias in the Mediterranean and demonstrates that the agricultural transition was not the defining factor in the emergence of this disease in Southeast Asia. We note here that the one adult with probable thalassemia from Man Bac had dental and skeletal affinity to Australo-Papuan populations, such as those from Con Co Ngua (and all foraging groups from Vietnam prior to agriculture). The inhabitants from the Neolithic central Thailand site of Khok Phanom Di where thalassemia was previously reported represent mixed Australo-Papuan and East Asian dental affinities30. Therefore, a deep antiquity for thalassemia in the region prior to the Neolithic appears highly probable. However, the complexity of gene flow and stabilizing selection in the region exceeds the capabilities of skeletal morphometric data.
The heterogeneity of mutations that cause beta, HbE beta and alpha thalassemia in Southeast Asia support multiple instances of independent emergence within the region likely tied to a consistent threat of malaria within MSEA31,32. Alpha thalassemia variants in Southeast Asia populations have similarly been identified in modern indigenous Papuan and Austroasiatic (modern Southeast Asian) speaking populations33. The widespread geography of these variants today, and the compounded effect of multiple migration and genetic admixture events since prehistory, mean it is not possible to determine the origins of these variants from the distribution in modern populations. However, different haplotypes do indicate separate founder effects in Australo-Papuan (indigenous Australian and New Guinean Highlanders) and Austroasiatic (Thai) groups33. At least in the case of alpha thalassemia, a pre-agricultural deep history of this disease in MSEA is consistent with present day genetic data. In contrast, selection models of one HbE variant from a Thai population indicates an emergence between 4400 and 1240 years ago, consistent with agricultural intensification between the Neolithic to Iron Age periods (4500–1500 BP) in MSEA34. HbE emergence is consistent with the time period of the Man Bac individuals with thalassemia. Multiple independent events of thalassemia emergence may have occurred due to different social transitions in Southeast Asia’s prehistory.
The presence of thalassemia in prehistoric MSEA, as is the case today, indicates a constant selection pressure of malaria stemming from deep antiquity. It is expected that the gene frequencies of thalassemias in the absence of malaria would not have been maintained given its mortal cost when inherited in its severe forms14. While P. falciparum has a considerably higher mortality rate than P. vivax, thereby incurring a stronger selection pressure, both Plasmodium species are endemic to the region and thought to have had possible selection effects on thalassemia in Southeast Asia35. Both P. falciparum and P. vivax malarias share African origins. It is thought that P. vivax emerged out of Africa with human groups prior to 30,000 years ago36 consistent with the first anatomically modern humans who inhabited MSEA as early as 60,000 years ago16. P. falciparum is thought to have emerged in Africa approximately 60,000–40,000 years ago, and estimated to have undergone a bottle neck approximately 6,000–4,000 years ago that likely favored human infection37. This proposed bottle neck event post-dates the skeletal evidence for thalassemia at Con Co Ngua and pre-dates that of Man Bac. Rather than provide resistance, alpha thalassemia has been proposed to increase susceptibility to infection by P. vivax35. This susceptibility may be an adaptive mechanism to cross vaccinate against P. falciparum35 which signals the complex connection between Plasmodium infections and the emergence of thalassemia. The association between thalassemias and P. vivax morbidity and mortality, however, remains unknown and is a necessary component to understanding the relationship between malarial types and the emergence of thalassemias in the region38. Rarer still, Plasmodium variants commonly found in Southeast Asian forest macaques have also been known to transfer to humans39, an important local reservoir in communities in Vietnam40. Potential human to non-human primate interactions may also factor into the relationship between malaria and thalassemia in the prehistory of the region. Both Man Bac and Con Co Ngua inhabitants exploited non-human primates as a food source17,41.
Lowland flood plains in Vietnam, such as where Man Bac and Con Co Ngua are located have low reported malarial cases and primary vectors today, as is the case with coastal and estuarine environments42. However, the original ecology of the region surrounding these sites in antiquity included movement within fringe forests, brackish water, and riverine areas that may have placed the inhabitants at risk of a number of different Anopheles mosquito species that carry both human and simian Plasmodium species42. In tropical forested areas of MSEA, malarial vectors are common. Tropical forested regions expanded substantially from approximately 14,000 years ago following the Last Glacial Maximum, which would have been opportunistic for the spread of species A. dirus and A. minimus, the primary mosquito vectors of malaria in MSEA42. The tropical climate would have enabled year-round transmission even in seasonal areas such as northern Vietnam that experience dry and wet seasons43. While pre-agricultural, the complex hunter-gathers of Con Co Ngua existed in large sedentary populations, thereby encompassing a suite of factors that have been argued to be associated with the emergence of malaria and thalassemia in the Mediterranean44. Sedentary residential mobility would have enabled completion of the cycle of the disease within one host population42, increasing the selection pressure on these populations. In Southeast Asia today, an increased risk is observed in people who live in villages but actively exploit the forest for resources45. This risk appears to be related to the early night feeding times of these vectors. The exposure to forest Anopheles mosquitos and sedentary residence alone would likely have been sufficient to increase the probability of contracting malaria in the absence of agricultural land clearing.
The agricultural transition and gene flow variation
While Con Co Ngua individuals were less well preserved than those from Man Bac, 10% (7/70) of the latter assemblage presented evidence for thalassemia. Man Bac is contemporaneous with Khok Phanom Di indicating these alleles were widespread through the region at this point in time. Therefore, the transition to agriculture may have considerably altered the gene flow or increased the selection pressure of thalassemia mutations, and shaped the relationship between human groups and malarial vectors in the region. Wet rice agriculture (irrigation) was only developed during the Iron Age (2500–1500 BP) in Southeast Asia46. This form of agriculture has been previously, erroneously, argued to be linked to increased malarial vectors compared to dry rice agriculture in MSEA47. Our results indicate a strong relationship between human groups and malaria prior to wet rice farming. Present day reports of differences in malarial vector prevalence in irrigated versus non-irrigated farming localities vary and depends on other ecological dynamics such as seasonality48. In Mainland Southeast Asia, A. dirus is found in significantly higher densities in forested areas than in villages, and reside in shallow pools42 rather than in deep waters. Therefore, the moated settlements of the Iron Age in Thailand may have made the ecology around villages less attractive to primary malarial vectors or attracted different Anopheles mosquitos such as A. barbirostris with significantly lower vector potential49. Instead, the reliance of dry rice farming on mild seasonal flooding may have increased the exposure of Neolithic inhabitants to malarial vectors42, particularly as they continued to exploit forested areas through mixed farmer-foraging practices50. While Pre-Neolithic foragers in northern Vietnam and southern China were arguably primarily sedentary, an increase in sedentism and significant population growth during the transition to agriculture in MSEA51,52 would have increased the transmission potential within human populations.
Possible interaction between Man Bac inhabitants and other nearby contemporaneous sites further inland, where higher vector densities have been reported42, may have also contributed to the presence of thalassemia at Man Bac. Alternatively, we also recognize that gene flow of thalassemia alleles from other inland Neolithic sites may be responsible, and a direct relationship between thalassemia and malaria presence at Man Bac cannot be determined. A similar argument can be made for Con Co Ngua, as contemporaneous forager sites of northern Vietnam are also archaeologically documented to have maintained interactions through exchange53. Nevertheless, it is apparent that mobility and interactions within and between groups were essential for the spread of thalassemia variants in the prehistory of MSEA, and the prevalence of this disease can be tied directly or indirectly to malarial endemicity in prehistoric MSEA.
Conclusion
Through a combined macroscopic and microscopic approach we demonstrate that thalassemia has been present in northern Vietnam from at least the early seventh millennium BP. This finding indicates that agriculture was not a crucial factor in the emergence of thalassemia in response to malaria in Southeast Asia as appears to be the case in the Mediterranean. In the context of large sedentary forager populations exploiting forested resources, such as the inhabitants of Con Co Ngua, we propose a pre-agricultural origin for the emergence of thalassemia in MSEA as an adaptive response to the threat of malaria. However, the agricultural transition approximately 4500–3500 years ago likely encouraged the spread of malarial vectors, increasing the gene frequencies of thalassemias. The outcomes of our research indicate a deep history of thalassemias which are endemic in MSEA today.
Methods and materials
Ethics and approvals
Paleopathological observation and collection and analysis of samples for histological investigation was approved by the Vietnam Institute of Archaeology in Hanoi, Vietnam on October 12, 2018. Bone samples were exported to the Australian National University in Canberra under approval by Dr. Nguyen Gia Doi (Director), and are currently housed at this institution until further notice. As the samples are of an archaeological nature, no ethical approvals were required for this study.
The sample
We macroscopically assessed 70 individuals from Man Bac and 155 individuals from Con Co Ngua for pathology. All skeletal elements with pathological change with the potential for diagnosis were radiographed. Three individuals from Con Co Ngua were sampled for histological analysis.
The skeletal assemblage of Man Bac was well preserved. While post-mortem breaks were frequent, the skeletons were easily reconstructed. Surfaces were excellently preserved. For most skeletons both adult and subadult were either complete or near complete54. In contrast, the individuals from Con Co Ngua presented with erosion of the cortical surfaces, but not to the degree that pathologies could not be observed. Post-mortem breakage was common, and a large proportion of the assemblage was at most partially complete. Subadults were commonly poorly preserved, with infants highly fragmented. Methods for age and sex of the individuals from Man Bac and Con Co Ngua were previously reported and briefly summarized here50,55,56. Non-metric traits of the os coxae, cranial morphology, and sample-specific post-cranial functions were used to estimate adult sex57–60. Adult age-at-death was estimated by way of pubic symphyseal morphology61 and/or sample-specific molar wear functions developed by Oxenham60. Subadult age-at-death was estimated using dental eruption and calcification standards62,63, and/or skeletal maturity schedules64.
Paleopathological differential diagnosis
The paleopathological diagnosis of thalassemia was based on macroscopic and radiographic observations of pathologies in dry bone. A standardized ‘threshold’ diagnostic protocol for thalassemia was produced following clinical diagnostic standards (Table 2). A possible case was diagnosed when an individual exhibited one diagnostic or two suggestive lesions. A probable case was diagnosed when an individual exhibited one strongly diagnostic or two diagnostic lesions. Such recording methods are consistent with a number of previously published diagnostic protocols in paleopathology, and ensure diagnostic rigour18,65.
Table 2.
Criteria for diagnosis of thalassemia in dry bone (SD = strongly diagnostic, D = diagnostic, and S = suggestive).
| Pathology | Diagnostic strength | Differential diagnosis | References |
|---|---|---|---|
| Marrow hyperplasia of the facial bones: maxillae- leading to ventral displacement of central incisors, zygomatic bones-leading to orbital displacement, and/ or mandible (rodent facies deformity) | SD | 29,66–70 | |
| Radiographic: “rib-within-a-rib” appearance. Radiographically defined sclerotic bands within the ribs due to extramedullary hematopoiesis | SD | Sickle cell anemia, osteomyelitis, leukemia* | 29,66,68,71 |
| Poor or lack of pneumatization of the paranasal and cranial sinuses sparing the ethmoid sinuses | D | Neoplasms, Paget’s disease, trauma, hypopituitarism, hypothyroidism, osteopetrosis, sickle cell anemia | 29,68 |
| Widening of entire rib, or widening of the rib head and neck with pronounced bulbous appearance posteriorly (costal osteomas). Associated with radiograph appearance of erosion of the inner cortex | D | Neuroblastoma, Nieman-Pick disease, Leukemia | 29,66,68,71 |
| Enlarged tubular bones of the hands and feet due to marrow hyperplasia (infants) sometimes associated with enlarged nutrient foramina or Radiographic: coarse trabecular patterns of the hands or feet, sometimes associated with cyst-like lucencies due to focal collection of hyperplasic marrow | D | Treponemal disease, leprosy, tuberculosis | 12,29,66,68 |
| Premature fusion of epiphyseal plates particularly of the proximal humerus and distal femur, often causing short long bone maximum length | D | Scurvy, hypervitaminosis A, trauma, achondroplasia, Morquio’s disease, Ellis-van Creveld disease, peripheral dysostosis, poliomyelitis, mucopolysaccharidosis, rickets, osteomalacia | 12,29,66,68,72 |
| Extensive marrow proliferation of the long bones leading to expansion of the medullary canal, associated with thin cortices (and in extreme circumstances honeycomb-like porosity) resulting in swollen appearance or metaphyseal flasked shaped deformities | S | Other hemolytic anemia, scurvy, rickets, metaphyseal dysplasia, Gaucher’s disease, osteomyelitis | 66,68,73 |
| Severe porotic hyperostosis/ diploic expansions of vault and maxilla with “hair-on-end” appearance and/ or cribra orbitalia | S | After Lagia74: hemolytic anemias and red cell enzyme disorders (including sickle cell disease, iron deficiency anemia and G-6-PD deficiency), cancers (including leukemia, multiple myeloma, meningioma, metastases and secondary to kidney cancers), and polycythemia | 29,66,68,74 |
| Wide dental spacing | S | Skeletal dysplasias, normal variation | 66 |
| Spiculated or scalloped proliferation of subperiosteal reactive new bone on the shafts of the limb bones and the clavicles | S | Infectious diseases, rickets, scurvy, hypertrophic osteoarthropathy, Gaucher’s diseases, Paget’s disease | 12,69,75 |
| Marked osteoporosis and cortical thinning of the vertebrae, with compression fractures in severe cases | S | Age related osteoporosis, osteomalacia, scurvy, trauma | 29 |
| Bone infarction | S | Osteomyelitis, sickle cell anemia, osteosarcoma | 76 |
| Enlargement and alteration of the trabecular pattern in flat bones (pelvis and scapula) | S | Other hemolytic anemia, leukemia | 70 |
A probable case is defined as an individual exhibiting at minimum one strongly diagnostic pathology or two diagnostic pathologies. A possible case is defined as an individual exhibiting at minimum one diagnostic or two suggestive pathologies.
*A strongly diagnostic lesion is one that is considered pathognomonic for that disease and alone stands as evidence of probable disease. In extremely rare instances these pathologies can occur in other diseases which are listed here in the differential diagnosis.
Gross skeletal manifestations of thalassemia are restricted to beta thalassemia only, except in the case of alpha thalassemia major (also called Bart’s hydrops fetalis) a fatal form, where infants die prior to or immediately following birth7. Severe porotic hyperostosis leading to a ‘hair-on-end’ appearance is a common skeletal manifestation of thalassemia (as is the case in other anemias) and has been reported particularly in cases of thalassemia major68,77.
Extensive marrow hyperplasia of the medullary canal and within cancellous bone regions cause considerable thinning of the bone cortex, and expansion of the area of the medullary canal68. The extent of the marrow hyperplasia causes thinning trabeculae. However, subperiosteal new bone response to the trabecular destruction instigates the coarsening of trabeculae which is observed radiographically68. In extreme circumstances cortical thinning can progress to destruction of the cortex and subsequent honeycomb appearance of the bone surface70.
A distinct facial deformity called rodent facies occurs in severe cases particularly in thalassemia major whereby extensive marrow proliferation of the maxillae and zygomatic bones creates anterior bulging of the face, often associated with anterior displacement of the central incisors, and lack of pneumatization of the maxillary, sphenoidal and frontal sinuses29,66–68. This pathological lesion is strongly diagnostic for thalassemia, particularly of thalassemia major (Table 2)29,68. The marrow proliferation also causes anterior teeth protrusion and malocclusion of the remaining teeth66. A strongly diagnostic radiographic feature of this disorder is the ‘rib-within-in-a-rib’ sign. This feature is produced by extensive marrow hypertrophy perforating the thin cortex of the rib associated with a radiodense line in the middle of the medullary canal29,66,69,78. Hypertrophic changes to the hands and feet of infants (with associated expanded foramina in the phalanges) is also characteristic of the disease, as this lesion indicates an extreme form of anemia unlikely to be associated with non-genetic etiologies12,68.
Marrow perforation of the cortex known as extramedullary hematopoiesis occurs. In certain circumstances subperiosteal new bone response similar to the “hair-on-end” appearance of the skull, surrounding thin cortices of postcranial bones, can result as a consequence of marrow perforation69,75. This skeletal response has been documented to lead to premature fusion of the epiphyses of long bones particularly of the proximal humerus and distal femur68,75. The extent of prematurity is dependent on the individual. This lesion is more common in cases of thalassemia intermedia75. Bone infarction, while less common in thalassemia than sickle cell anemia, has also been reported. In this pathological condition, bone death occurs followed by inflammatory new bone response around the region of necrosis, and has a distinct appearance on radiographs76.
Histological sectioning
All sectioning procedures followed standard protocols79,80 and were completed at the Institute of Archaeology, Hanoi, Vietnam. A Dremel Variable-Speed Rotary Tool 3000 with associated Dremel blades was used to cut into the bone shafts. This involved making two longitudinally oriented parallel (along the long bone axis) cuts, followed by a transversely oriented (perpendicular to the longitudinal cuts) cut, so that sections freely detached themselves from the bone. Permissions obtained from the collection curators strictly stipulated that sections were only to be removed from the bone regions already affected by post-mortem breaks to avoid further damage to the specimens. For example, where the long bone was broken in half or had epiphyseal ends missing due to taphonomic issues. Sampling in those regions avoided further damage to the specimens. However, we ensured that the bone sections still derived from the long bone shafts so that we could assess the histological changes on the endocortical surfaces. The three individuals sampled for histological analysis met these criteria. We sampled the left and right femur of CCN13M40a (Fig. 4). The right humerus, left ulna and left radius of CCN13M59a were sampled as they were the only available long bones for this individual (the lower limb only consisted of a fragmented right fibula). The right humerus, left radius, and left femur of the subadult CCN13M67a were sectioned as both the upper and lower limb bones showed a degree of endocortical thickening. The lower (distal direction) third of the left femoral shaft, approximately 4 cm below the linea aspera, and the midshaft of the right femur, were sampled in CCN13M40a. The upper limb bones of CCN13M59a were sectioned at the distal end of the left ulna and the right radius, and the midshaft of the right humerus. The left femur of CCN13M67a was sectioned at the middle of the upper (proximal direction) third of shaft, the distal third of the shaft in the left radius, and the upper (proximal direction) third shaft of the right humerus. We note that the left radius in this individual shows a well remodeled and healed fracture at midshaft (Supplementary Fig. S2), which clearly resulted in a deformation of the bone. This will need to be borne in mind when interpreting the appearance of histology in thin section. A total of eight, approximately 1 cm thick, roughly C-shaped, sections of bone from the posterior aspect were removed for the entire sample (Fig. 4).
Prior to histological preparation, external size measurements of the bones and sections were recorded using standard digital calipers and a soft measuring tape. All measurements were repeated at least two times from which an average was computed and reported here. The measurements79 were: shaft circumference (Circ) at point of sectioning (mm), anterior–posterior diameter in mm (AP dm), medial–lateral diameter in mm (ML dm), cortical width (Ct.W) of the extracted sample measured from both the proximal and distal surfaces in the lateral direction (from the endosteal, referring to the endosteum lining the medullary cavity, to the periosteal surfaces).
Histopathology
No prior information on age, sex or macropathology was provided for the samples to ensure an objective observation. All samples were processed into thin sections following standard methods applicable for histological analysis of archaeological human bone81,82. The processing was completed at the Hard Tissue Histology laboratory in the School of Archaeology and Anthropology at the Australian National University (Canberra, Australia), which is also where the resulting thin sections are curated until further notice. Each sample was embedded in Buehler epoxy resin, cut on a low-speed saw equipped with a diamond wafering blade, and attached to microscope glass slides using Stuck epoxy glue. The attached samples were further trimmed on a cutting saw, and later lapped on a series of grinding pads of various grit sizes that increased from 400 to 1200 in coarseness. The thickness of each section was controlled using a handheld Buehler 28 × 48 mm target holder, allowing to produce sections of approximately 90–100 μm thickness. The sections were polished using a Buehler MicroPolish powder, 0.05 µm, until all scratches created during grinding were removed. This was followed by cleaning the sections in an ultrasonic bath, dehydrating them in a series of ethanol baths, and clearing using xylene for histopathology. The samples were finally covered with glass cover slips glued to the bone surface with DPX.
Imaging and histomorphological analysis
Each thin section was imaged using transmitted and polarized light under a high powered Olympus BX53 microscope equipped with a DP74 high resolution camera81. We used a series of objective lenses, ranging from 4 × to 40 × (40 × to 400 × total magnification), to examine histological features82. Each section was first examined for any overall diagenetic damage, and second for patterns in, and areas of, abnormal bone matrices and micro-organization. This was followed by a targeted examination of bone regions on the endocortical surfaces, which is where we hypothesized abnormal changes would occur as per the macroscopic manifestation (Fig. 4). As the border between intra-cortical and endocortical bone relies on a somewhat arbitrary division, the endocortical area examined in our samples was, on average, 1.5–1.7 cm once the entire section surface was sub-divided into four equal segments (one sub-periosteal, two intra-cortically, and one endocortically). We captured individual regions of interest (ROIs) from within the endocortical areas. It is also where we recorded a full endocortical strip of bone using an automated stitching tool of the Olympus CellSens software.
The microscopic examination revealed an overall moderately good preservation of the samples83. Our histological examination was based on inspecting bone regions for: bone matrix type (woven, lamellar)84; bone growth stage (primary, secondary/ Haversian)85; bone remodeling indicators (poorly remodeled with singular secondary osteon units, well remodeled with several generations of fragmentary and intact secondary osteons)80,85, ‘giant’ resorption canals (porosity effect indicating prolonged osteoclast-mediated resorption, tabularization effect)86,87. Where necessary, we also undertook histomorphometric measurements using ImageJ ‘straight line’ tool to obtain maximum diameter of pores in cases of abnormal porosity.
Supplementary Information
Acknowledgements
We would like to thank Dr. Ngo Anh Son, Mr. Bui Van Khanh and Ms. Nellissa Ling for their assistance with the radiographs. We are grateful to Dr. Nguyen Gia Doi for permission to extract histological samples. This work was supported by a National Geographic Early Career Grant (EC-54332R-18); Royal Society of New Zealand Skinner Fund Grant; University of Otago Doctoral Scholarship; Australian Research Council DP110101097 and FT120100299. Histological processing was funded by the Australian Research Council DE190100068.
Author contributions
M.F.O. directed the excavation of MB. M.F.O. and H.H.T. jointly directed the excavation of CCN. Initial osteological analyses, including sex, age, and pathology were performed by MF.O. and K.D. for MB and M.F.O., H.B., K.D. and A.W. for CCN. M.V. and H.B. designed the hydatid disease study. M.V., H.B., T.M. and M.W. collected the data. M.V., H.B., J.M. and M.W. analyzed the data. All authors contributed input to the manuscript including contextualization of the data and drafting of the manuscript. M.V. prepared Figs. 1, 2, 3 and S1. J.M. prepared Figs. 4, 5 and S2.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Melandri Vlok, Email: Melandri.vlok@otago.ac.nz.
Marc F. Oxenham, Email: marc.oxenham@anu.edu
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-83978-4.
References
- 1.Oxenham, M. F. In The Encyclopedia of Archaeological Sciences (ed López Varela, S. L.) 1–4 (Wiley Online, 2018).
- 2.Weatherall D. Genetic variation and susceptibility to infection: the red cell and malaria. Br. J. Haematol. 2008;141:276–286. doi: 10.1111/j.1365-2141.2008.07085.x. [DOI] [PubMed] [Google Scholar]
- 3.Weatherall DJ. The evolving spectrum of the epidemiology of thalassemia. Hematol. Oncol. Clin. 2018;32:165–175. doi: 10.1016/j.hoc.2017.11.008. [DOI] [PubMed] [Google Scholar]
- 4.Aydinok Y. Thalassemia. Hematology. 2012;17:s28–s31. doi: 10.1179/102453312X13336169155295. [DOI] [PubMed] [Google Scholar]
- 5.Galanello R, Cao A. Alpha-thalassemia. Genet. Med. 2011;13:83–88. doi: 10.1097/GIM.0b013e3181fcb468. [DOI] [PubMed] [Google Scholar]
- 6.World Health Organization. World Malaria Report 2019. (2019).
- 7.Fucharoen S, Winichagoon P. Thalassemia in Southeast Asia: problems and strategy for prevention and control. Southeast Asian J. Trop. Med. Public Health. 1992;23:647–647. [PubMed] [Google Scholar]
- 8.Vichinsky E. Non-transfusion-dependent thalassemia and thalassemia intermedia: epidemiology, complications, and management. Curr. Med. Res. Opin. 2016;32:191–204. doi: 10.1185/03007995.2015.1110128. [DOI] [PubMed] [Google Scholar]
- 9.Antonarakis SE, et al. Evidence for multiple origins of the beta E-globin gene in Southeast Asia. Proc. Natl. Acad. Sci. 1982;79:6608–6611. doi: 10.1073/pnas.79.21.6608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fucharoen S, Weatherall DJ. The hemoglobin E thalassemias. Cold Spring Harbor Perspect. Med. 2012;2:a011734. doi: 10.1101/cshperspect.a011734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Winichagoon P, Fucharoen S, Weatherall D, Wasi P. Concomitant inheritance of α-thalassemia in β°-thalassemia/Hb E disease. Am. J. Hematol. 1985;20:217–222. doi: 10.1002/ajh.2830200303. [DOI] [PubMed] [Google Scholar]
- 12.Tayles N. Anemia, genetic diseases, and malaria in prehistoric mainland Southeast Asia. Am. J. Phys. Anthropol. 1996;101:11–27. doi: 10.1002/(SICI)1096-8644(199609)101:1<11::AID-AJPA2>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 13.Hershkovitz I, et al. Possible congenital hemolytic anemia in prehistoric coastal inhabitants of Israel. Am. J. Phys. Anthropol. 1991;85:7–13. doi: 10.1002/ajpa.1330850103. [DOI] [PubMed] [Google Scholar]
- 14.Viganó C, Haas C, Rühli FJ, Bouwman A. 2000 Year old β-thalassemia case in sardinia suggests malaria was endemic by the Roman period. Am. J. Phys. Anthropol. 2017;164:362–370. doi: 10.1002/ajpa.23278. [DOI] [PubMed] [Google Scholar]
- 15.Bellwood, P. & Oxenham, M. in The Neolithic Demographic Transition and Its Consequences (eds Bocquet-Appel, J. P. & Bar-Yosef, O.) 13–34 (Springer Science, 2008).
- 16.Oxenham, M. & Buckley, H. in The Routledge Handbook of Bioarchaeology in Southeast Asia and the Pacific. (eds Fredrick Oxenham, M. & Buckley, H.) 9–23 (Routledge, 2016).
- 17.Oxenham MF, et al. Between foraging and farming: strategic responses to the Holocene thermal maximum in Southeast Asia. Antiquity. 2018;92:940–957. doi: 10.15184/aqy.2018.69. [DOI] [Google Scholar]
- 18.Vlok M, et al. Two probable cases of infection with Treponema pallidum during the neolithic period in Northern Vietnam (ca. 2000–1500B.C.) Bioarchaeol. Int. 2020;4:15–39. doi: 10.5744/bi.2020.1000. [DOI] [Google Scholar]
- 19.McColl H, et al. The prehistoric peopling of Southeast Asia. Science. 2018;361:88–92. doi: 10.1126/science.aat3628. [DOI] [PubMed] [Google Scholar]
- 20.Williams H, Davies A, Chapman S. Bone within a bone. Clin. Radiol. 2004;59:132–144. doi: 10.1016/S0009-9260(03)00337-4. [DOI] [PubMed] [Google Scholar]
- 21.Perisano C, et al. Physiopathology of bone modifications in β-thalassemia. Anemia. 2012;2012:320737. doi: 10.1155/2012/320737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Thongchote K, et al. Bone microstructural defects and osteopenia in hemizygous βIVSII-654 knockin thalassemic mice: sex-dependent changes in bone density and osteoclast function. Am. J. Physiol. Endocrinol. Metabol. 2015;309:E936–E948. doi: 10.1152/ajpendo.00329.2015. [DOI] [PubMed] [Google Scholar]
- 23.Wong P, Fuller PJ, Gillespie MT, Milat F. Bone disease in thalassemia: a molecular and clinical overview. Endocr. Rev. 2016;37:320–346. doi: 10.1210/er.2015-1105. [DOI] [PubMed] [Google Scholar]
- 24.Tan SL, Tseng AP, Thong PW. Bart's hydrops fetalis—clinical presentation and management—an analysis of 25 cases. Aust. N. Z. J. Obstet. Gynaecol. 1989;29:233–237. doi: 10.1111/j.1479-828X.1989.tb01726.x. [DOI] [PubMed] [Google Scholar]
- 25.Chik K-W, et al. Treatment of hemoglobin Bart's hydrops with bone marrow transplantation. J. Pediatr. 1998;132:1039–1042. doi: 10.1016/S0022-3476(98)70406-8. [DOI] [PubMed] [Google Scholar]
- 26.Jaffe, H. L. Metabolic, Degenerative, and Inflammatory Diseases of Bones and Joints. (Lea and Febiger, Washington, DC, 1972).
- 27.Winichagoon P, Fucharoen S, Chen P, Wasi P. Genetic factors affecting clinical severity in β-thalassemia syndromes. J. Pediatr. Hematol. Oncol. 2000;22:573–580. doi: 10.1097/00043426-200011000-00026. [DOI] [PubMed] [Google Scholar]
- 28.Fessas, P. in Radiology of Thalassemia (eds Papavasiliou, C., Cambouris, T. & Fessas, P.) 1–6 (Springer Science & Business Media, 2012).
- 29.Tunacı M, et al. Imaging features of thalassemia. Eur. Radiol. 1999;9:1804–1809. doi: 10.1007/s003300050926. [DOI] [PubMed] [Google Scholar]
- 30.Matsumura H, Oxenham M. Demographic transitions and migration in prehistoric East/Southeast Asia through the lens of nonmetric dental traits. Am. J. Phys. Anthropol. 2014;155:45–65. doi: 10.1002/ajpa.22537. [DOI] [PubMed] [Google Scholar]
- 31.Wong C, et al. On the origin and spread of beta-thalassemia: recurrent observation of four mutations in different ethnic groups. Proc. Natl. Acad. Sci. 1986;83:6529–6532. doi: 10.1073/pnas.83.17.6529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fucharoen G, Fucharoen S, Jetsrisuparb A, Fukumaki Y. Molecular basis of HbE-β-thalassemia and the origin of HbE in Northeast Thailand: identification of one novel mutation using amplified DNA from buffy coat specimens. Biochem. Biophys. Res. Commun. 1990;170:698–704. doi: 10.1016/0006-291X(90)92147-R. [DOI] [PubMed] [Google Scholar]
- 33.Charoenwijitkul T, et al. Molecular characteristics of α+-thalassemia (3.7 kb deletion) in Southeast Asia: molecular subtypes, haplotypic heterogeneity, multiple founder effects and laboratory diagnostics. Clini. Biochem. 2019;71:31–37. doi: 10.1016/j.clinbiochem.2019.06.005. [DOI] [PubMed] [Google Scholar]
- 34.Ohashi J, et al. Extended linkage disequilibrium surrounding the hemoglobin E variant due to malarial selection. Am. J. Hum. Genet. 2004;74:1198–1208. doi: 10.1086/421330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Weatherall D. Thalassaemia and malaria, revisited. Ann. Trop. Med. Parasitol. 1997;91:885–890. doi: 10.1080/00034983.1997.11813215. [DOI] [PubMed] [Google Scholar]
- 36.Liu W, et al. African origin of the malaria parasite plasmodium vivax. Nat. Commun. 2014;5:3346. doi: 10.1038/ncomms4346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Otto TD, et al. Genomes of all known members of a plasmodium subgenus reveal paths to virulent human malaria. Nat. Microbiol. 2018;3:687–697. doi: 10.1038/s41564-018-0162-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Douglas NM, et al. The anaemia of plasmodium vivax malaria. Malar. J. 2012;11:135. doi: 10.1186/1475-2875-11-135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hartmeyer GN, et al. Plasmodium cynomolgi as cause of malaria in tourist to Southeast Asia, 2018. Emerg. Infect. Dis. 2019;25:1936. doi: 10.3201/eid2510.190448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chinh VD, et al. Prevalence of human and non-human primate Plasmodium parasites in anopheline mosquitoes: a cross-sectional epidemiological study in Southern Vietnam. Trop. Med. Health. 2019;47:1–6. doi: 10.1186/s41182-019-0139-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jones RK, et al. Shifting subsistence patterns from the terminal pleistocene to late holocene: a regional Southeast Asian analysis. Quatern. Int. 2019;529:47–56. doi: 10.1016/j.quaint.2019.01.006. [DOI] [Google Scholar]
- 42.Poolsuwan S. Malaria in prehistoric Southeastern Asia. Southeast Asian J. Trop. Med. Public Health. 1995;26:3–22. [PubMed] [Google Scholar]
- 43.Babiker HA, Lines J, Hill WG, Walliker D. Population structure of Plasmodium falciparum in villages with different malaria endemicity in East Africa. Am. J. Trop. Med. Hyg. 1997;56:141–147. doi: 10.4269/ajtmh.1997.56.141. [DOI] [PubMed] [Google Scholar]
- 44.Angel J. Porotic hyperostosis in the Eastern Mediterranean. MCV/Q Med. Coll. Va. Q. 1978;14:10–16. [Google Scholar]
- 45.Durnez L, et al. Outdoor malaria transmission in forested villages of Cambodia. Malar. J. 2013;12:329. doi: 10.1186/1475-2875-12-329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Castillo C, et al. Social responses to climate change in iron age north-east Thailand: new archaeobotanical evidence. Antiquity. 2018;92:1274–1291. doi: 10.15184/aqy.2018.198. [DOI] [Google Scholar]
- 47.King CL, Halcrow SE, Tayles N, Shkrum S. Considering the palaeoepidemiological implications of socioeconomic and environmental change in Southeast Asia. Archaeol. Res. Asia. 2017;11:27–37. doi: 10.1016/j.ara.2017.05.003. [DOI] [Google Scholar]
- 48.Koudou B, et al. Malaria transmission dynamics in central Côte d'Ivoire: the influence of changing patterns of irrigated rice agriculture. Med. Vet. Entomol. 2005;19:27–37. doi: 10.1111/j.0269-283X.2005.00530.x. [DOI] [PubMed] [Google Scholar]
- 49.Thongsahuan S, et al. Susceptibility of Anopheles campestris-like and Anopheles barbirostris species complexes to Plasmodium falciparum and Plasmodium vivax in Thailand. Memórias do Instituto Oswaldo Cruz. 2011;106:105–112. doi: 10.1590/S0074-02762011000100017. [DOI] [PubMed] [Google Scholar]
- 50.Oxenham, M. F., Matsumura, H. & Kim Dung, N. Man Bac: The Excavation of a Neolithic Site in Northern Vietnam The Biology, Terra Australis 33. (ANU ePress, 2011).
- 51.Huffer, D. & Oxenham, M. F. Investigating activity and mobility patterns during the mid-Holocene in Northern Vietnam. In The Routledge Handbook of Bioarchaeology in Southeast Asia and the Pacific Islands, 110 (2015).
- 52.McFadden C, Buckley H, Halcrow SE, Oxenham MF. Detection of temporospatially localized growth in ancient Southeast Asia using human skeletal remains. J. Archaeol. Sci. 2018;98:93–101. doi: 10.1016/j.jas.2018.08.010. [DOI] [Google Scholar]
- 53.Matsumura, H., Hung, H. -C., Zhen, L. & Shinoda, K. in Bio-anthropological Studies of Early Holocene Hunter-Gatherer Sites at Huiyaotian and Liyupo in Guangxi, China. (National Museum of Nature and Science Tokyo, 2017).
- 54.Buikstra, J. E. & Ubelaker, D. H. Standards for Data Collection from Human Skeletal Remains: Proceedings of a Seminar at the Field Museum of Natural History, Arkansas Archaeological Survey Research Series No. 44., (Arkansas Archaeological Survey, 1994).
- 55.Scott, R. M. et al. Domestication and large animal interactions: skeletal Trauma in Northern Vietnam during the Hunter-gatherer Da But Period. PLoS One14, e0218777 (2019). [DOI] [PMC free article] [PubMed]
- 56.Domett, K. M. & Oxenham, M. F. in Man Bac: The Excavation of a Neolithic Site in Northern Vietnam, The Biology. Terra Australis 33 (eds Fredrick Oxenham, M., Matsumura, H., & Kim Dung, N.) 9–20 (ANU ePress, 2011).
- 57.Phenice TW. A newly developed visual method of sexing the Os Pubis. Am. J. Phys. Anthropol. 1969;30:297–301. doi: 10.1002/ajpa.1330300214. [DOI] [PubMed] [Google Scholar]
- 58.McFadden C, Oxenham MF. Revisiting the Phenice technique sex classification results reported by MacLaughlin and Bruce (1990) Am. J. Phys. Anthropol. 2016;159:182–183. doi: 10.1002/ajpa.22839. [DOI] [PubMed] [Google Scholar]
- 59.Walrath DE, Turner P, Bruzek J. Reliability test of the visual assessment of cranial traits for sex determination. Am. J. Phys. Anthropol. 2004;125:132–137. doi: 10.1002/ajpa.10373. [DOI] [PubMed] [Google Scholar]
- 60.Oxenham, M. F. Bioarchaeology of Ancient Northern Vietnam. Vol. 2781 (Archaeopress, Oxford, 2016).
- 61.Brooks S, Suchey JM. Skeletal age determination based on the Os Pubis: a comparison of the Acsádi-Nemeskéri and Suchey-Brooks Methods. Hum. Evol. 1990;5:227–238. doi: 10.1007/BF02437238. [DOI] [Google Scholar]
- 62.Moorrees CF, Fanning EA, Hunt EE., Jr Age variation of formation stages for ten permanent teeth. J. Dent. Res. 1963;42:1490–1502. doi: 10.1177/00220345630420062701. [DOI] [PubMed] [Google Scholar]
- 63.Ubelaker, D. H. Human Skeletal Remains: Excavation, Analysis, Interpretation 3rd edn, (Taraxacum Press, Cambridge, 1989).
- 64.Scheuer, L. & Black, S. Developmental Juvenile Osteology. (Academic Press, Cambridge, 2000).
- 65.Snoddy AME, et al. Macroscopic features of scurvy in human skeletal remains: a literature synthesis and diagnostic guide. Am. J. Phys. Anthropol. 2018;167:876–895. doi: 10.1002/ajpa.23699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lewis M. Thalassaemia: its diagnosis and interpretation in past skeletal populations. Int. J. Osteoarchaeol. 2012;22:685–693. doi: 10.1002/oa.1229. [DOI] [Google Scholar]
- 67.Scutellari, P., Franceschini, F. & O’rzincolo, C. D. J. W. in Radiology of Thalassemia (eds Papavasiliou, C., Cambouris, T., & Fessas, P.) 50–61 (Springer, 1989).
- 68.Cambouris, T. in Radiology of Thalassemia (ed Papavasiliou, C.) 21–43 (Springer Science & Business Media, 1989).
- 69.Adamopoulos, S. G. & Petrocheilou, G. M. Skeletal Radiological Findings in Thalassemia Major. Journal of Research and Practice on the Musculoskeletal System (in press).
- 70.Ortner, D. J. Identification of Pathological Conditions in Human Skeletal Remains. 2nd edn, (Academic Press, Cambridge, 2003).
- 71.Lawson J, Ablow RC, Pearson HA. The ribs in thalassemia. II. The pathogenesis of the changes. Radiology. 1981;140:673–679. doi: 10.1148/radiology.140.3.7280234. [DOI] [PubMed] [Google Scholar]
- 72.Currarino G, Erlandson M. Premature fusion of epiphyses in Cooley’s anaemia. Radiology. 1964;93:656–664. doi: 10.1148/83.4.656. [DOI] [PubMed] [Google Scholar]
- 73.Lawson JP, Ablow RC, Pearson HA. Premature fusion of the proximal humeral epiphyses in thalassemia. Am. J. Roentgenol. 1983;140:239–244. doi: 10.2214/ajr.140.2.239. [DOI] [PubMed] [Google Scholar]
- 74.Lagia A, Eliopoulos C, Manolis S. Thalassemia: macroscopic and radiological study of a case. Int. J. Osteoarchaeol. 2007;17:269–285. doi: 10.1002/oa.881. [DOI] [Google Scholar]
- 75.Colavita N, Orazi C, Danza S, Falappa P, Fabbri R. Premature epiphyseal fusion and extramedullary hematopoiesis in thalassemia. Skeletal Radiol. 1987;16:533–538. doi: 10.1007/BF00351267. [DOI] [PubMed] [Google Scholar]
- 76.Kanthawang T, Pattamapaspong N, Louthrenoo W. Acute bone infarction: a rare complication in thalassemia. Skeletal Radiol. 2016;45:1013–1016. doi: 10.1007/s00256-016-2387-6. [DOI] [PubMed] [Google Scholar]
- 77.Sfikakis, P. in Radiology of Thalassemia (eds Papavasiliou, C., Cambouris, T., & Fessas, P.) 44–49 (Springer, 1989).
- 78.Lewis, M. E. Paleopathology of Children: Identification of Pathological Conditions in the Human Skeletal Remains of Non-Adults. (Academic Press, Cambridge, 2017).
- 79.Miszkiewicz JJ, Mahoney P. Ancient human bone microstructure in medieval England: comparisons between two socio-economic groups. Anatom. Rec. 2016;299:42–59. doi: 10.1002/ar.23285. [DOI] [PubMed] [Google Scholar]
- 80.Miszkiewicz JJ. Investigating histomorphometric relationships at the human femoral midshaft in a biomechanical context. J. Bone Miner. Metab. 2016;34:179–192. doi: 10.1007/s00774-015-0652-8. [DOI] [PubMed] [Google Scholar]
- 81.Miszkiewicz, J. J. & Mahoney, P. in Human Remains: Another Dimension 29–43 (Academic Press, Cambridge, 2017).
- 82.Schultz M. Paleohistopathology of bone: a new approach to the study of ancient diseases. Am. J. Phys. Anthropol. 2001;116:106–147. doi: 10.1002/ajpa.10024. [DOI] [PubMed] [Google Scholar]
- 83.Trueman C, Martill DM. The long-term survival of bone: the role of bioerosion. Archaeometry. 2002;44:371–382. doi: 10.1111/1475-4754.t01-1-00070. [DOI] [Google Scholar]
- 84.Kuhn G, Schultz M, Müller R, Rühli FJ. Diagnostic value of micro-CT in comparison with histology in the qualitative assessment of historical human postcranial bone pathologies. Homo. 2007;58:97–115. doi: 10.1016/j.jchb.2006.11.002. [DOI] [PubMed] [Google Scholar]
- 85.Pitfield R, Miszkiewicz JJ, Mahoney P. Cortical histomorphometry of the human humerus during ontogeny. Calcif. Tissue Int. 2017;101:148–158. doi: 10.1007/s00223-017-0268-1. [DOI] [PubMed] [Google Scholar]
- 86.Lamm C, et al. Micro-CT analyses of historical bone samples presenting with osteomyelitis. Skeletal Radiol. 2015;44:1507–1514. doi: 10.1007/s00256-015-2203-8. [DOI] [PubMed] [Google Scholar]
- 87.Chappard C, et al. 3D characterization of pores in the cortical bone of human femur in the elderly at different locations as determined by synchrotron micro-computed tomography images. Osteoporos. Int. 2013;24:1023–1033. doi: 10.1007/s00198-012-2044-4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.