Abstract
Radiation-induced skin injury (RISI) is one of the common serious side effects of radiotherapy (RT) for patients with malignant tumors. Mesenchymal stem cells (MSCs) are applied to RISI repair in some clinical cases series except some traditional options. Though direct replacement of damaged cells may be achieved through differentiation capacity of MSCs, more recent data indicate that various cytokines and chemokines secreted by MSCs are involved in synergetic therapy of RISI by anti-inflammatory, immunomodulation, antioxidant, revascularization, and anti-apoptotic activity. In this paper, we not only discussed different sources of MSCs on the treatment of RISI both in preclinical studies and clinical trials, but also summarized the applications and mechanisms of MSCs in other related regenerative fields.
Keywords: Mesenchymal stem cells, Radiation, Radiotherapy, Skin injury, Treatment
Introduction
Radiation-induced skin injury (RISI) is a common side effect of radiotherapy (RT) for malignant tumors or bone marrow transplant. Seventy percent of cancer patients especially with head and neck, skin, anogenital, and breast cancer would receive RT alone or as an adjuvant to surgeries at different stages and almost 95% patients with RT would occur with RISI [1, 2].
RISI is divided into acute RISI (occurring within hours to weeks after radiation) with clinical manifestations of skin erythema, blister, dermatitis, and necrotic ulcer and chronic RISI (occurrence from months to years post-radiation) with clinical syndromes of fibrosis, leathery skin, atrophy, and pigmentation [3]. These clinical symptoms mentioned above would not only reduce quality of life, but also interrupt tumor treatment progress (Fig. 1). Though traditional methods including topical steroids, creams, ointments, and hydrogel dressings have been applied to treat RISI, no criterion standard exists for the treatment of RISI [4–6].
Fig. 1.
Four typical cases of radiation-induced skin injury. a Radiation-induced dermatitis appeared in a 61-year-old female patient who was diagnosed with esophageal cancer (T3N0M0) after the third RT. b Radiation-induced dermatitis appeared in a 59-year-old female patient suffering from mammary cancer (T1N0M0) after four times of RT. c Radiation-induced skin ulcer appeared in a 57-year-old male patient who suffered from esophageal cancer (T4N0M0) after 9 times of RT and conventional dressing change. d Radiation-induced skin ulcer appeared in a 44-year-old male patient with diagnosis of mycosis fungoides after 6 times of RT together with conventional dressing change
In this paper, we discussed the applications and potential mechanisms of MSCs in RISI and summarized the applications of MSCs in other regenerative fields.
The mechanism of RISI
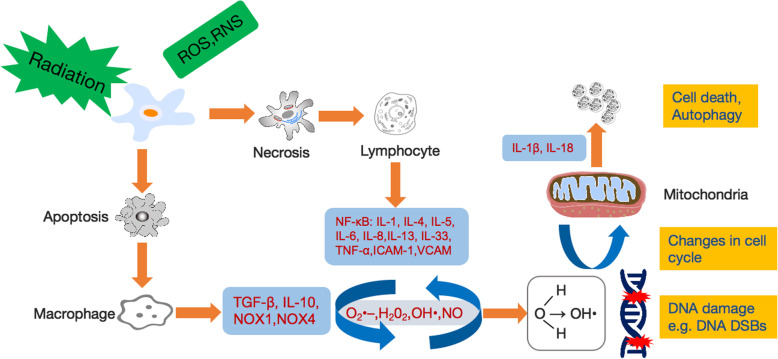
The mechanisms involved in RISI include oxidative stress and inflammation. The specific mechanisms of RISI are not fully understood but may be related to DNA damage, changes in cell cycle progression, and/or cell death [7, 8]. Chemical bond of DNA is particularly vulnerable to break while directly be exposed to radiation and the DNA double-strand breaks (DSBs) are the most serious type of DNA damage, from which γ-H2AX and MRN complex contains Mre11 are early indexes to reflect DNA damage and summarized in Fig. 2 [9–11].
Fig. 2.
The molecular mechanisms of RISI
Oxidative stress
Oxidative stress is the combination of cell damage and stress response signals that has been taken into account as one of the causes of acute and chronic radiation damage to the body. Reactive oxygen species (ROS) are additional products of aerobic metabolism varieties [10, 11]. Active oxygen-containing metabolites include superoxide anions (O2•–), hydrogen peroxide (H2O2), hydroxyl radicals (OH•), nitric oxide (NO), and its free radicals produced by its reaction with superoxide or oxygen that are known as reactive nitrogen species (RNS) [12–14]. Many reactions mediated by ROS protect cells from oxidative stress and then re-establish “redox homeostasis”. The increase of glutathione, an intracellular antioxidant, or the increase of protein/enzyme expression ability of scavenging ROS in cells is called oxidative stress reaction to maintain redox homeostasis in cells and tissues [15–17].
Some studies show that radiation damage to a single cell could trigger some signals and amplify the production of free radicals in irradiated cells that last for hours to days or even months [18, 19]. In addition, signals generated by irradiated cells could also be transferred to neighboring non-irradiated cells through bystander effects and activate REDOX metabolism [20]. Cytokines, miRNAs, vesicles, high mobility group box 1 (HMGB1), and free DNA fragments are those intracellular mediators by which chronic ROS/RNS released from irradiated cells, resulting in cell damage [21–23].
Inflammation
ROS/RNS could injure biological molecules such as nucleic acids, proteins, and lipids which would trigger a series of inflammatory reactions, result in apoptosis, and inhibit cell proliferation [24, 25]. Epithelial cells are extremely sensitive and susceptible to radiation as well as blood vessel and cutaneous appendages [26]. Free radicals generated from the interaction between radiation and water molecules may cause damage to biologically active ingredients. In the early stage of radiation injury, epithelial dysplasia, cell necrosis, nuclear pyknosis, and tissue fibrosis could be observed under the microscope [27, 28]. What is more, DNA DSBs caused by radiation would trigger changes in cell cycle progression, including the disturbing G1/S process and manifesting G1 cyclin inhibition. Some experiments showed that G1 inhibition was related to the survival state of p53 gene [29–31]. Upon cell exposure to radiation during DNA replication, it would extend the length of or lead to a block in S phase during DNA replication. Furthermore, ataxia telangiectasia mutated (ATM) and DNA-dependent protein kinase catalytic subunit (DNA-PKcs) are activated by DNA DSBs, followed by phosphorylation of checkpoint kinase 2 (CHK2) and lead to a block in G2/M phase [32–34]. Cell death events caused by radiation include apoptosis, necrosis, autophagy, mitotic catastrophe, and ferroptosis are related to the sensitivity of different cell types to radiation. Although the common mechanism may be related to damage of nuclear DNA, the specific mechanism involved in cell death still needs further research [35–37].
Conventional treatment for RISI
Treatment details containing pros and cons of conventional methods for RISI are summarized in Table 1.
Table 1.
Summary of the pros and cons of conventional treatments for RISI
| Conventional treatments | Options | Pros | Cons | Reference |
|---|---|---|---|---|
| Systemic therapy | Antibiotics | Control and eliminate bacterial infection | Potential risk of antibiotic resistance occurrence | [38–40] |
| Water-electrolyte balance | Supplement of loss of water-electrolyte by wounds | Cardiopulmonary load by excessive rehydration | [39, 41] | |
| Nutritional support | Reduce autologous protein degradation and enhance immunity | Burden to kidney and liver | [40, 42] | |
| Local treatment | Topical corticosteroids | Anti-inflammation, immunosuppression and anti-proliferation | Decrease histamine and deplete mast cells on skin | [41, 43] |
| Creams and ointments | Anti-inflammatory and promote macrophages recruitment | Less effective in managing patient-reported symptoms | [44, 45] | |
| Hydrogel dressings | Induce healing time, reduce pain and infection | Less frequent occurrence of severe skin reactions | [46, 47] | |
| Hyperbaric oxygen therapy | Increase the oxygen supply, inhibit inflammatory reaction, and reduce exudation | Arterial insufficiency, refractory osteomyelitis and bone necrosis | [48, 49] | |
| Superoxide dismutase | Antioxidant enzymes for scavenging free radicals | Not mentioned | [50, 51] | |
| Low-intensity laser | Reduction of edema and analgesia induction | Not mentioned | [50, 52] |
Systemic therapy
Systematic treatment is mainly based on different stages of RISI development, including antibiotics, water-electrolyte balance, and nutritional support. Experience has shown that infusion of fresh-frozen plasma (FFP), proteinase inhibitors, free-radical scavenger, and drugs of increasing the immunity could alleviate symptoms of RISI patients [38, 39].
Local treatment
Topical corticosteroids
Corticosteroids are widely used to treat dermatitis induced by acute radiation [40]. Shukla et al. reported that the use of corticosteroids could prevent radiation-induced skin toxicity effectively and the incidence of wet desquamation was 13% in the corticosteroid group than 37% in control group [42]. Another clinical trial with 120 breast cancer patients enrolled showed that (mometasone furoate) MFF group significantly reduced radiation dermatitis and improved health-related quality of life [41].
Creams and ointments
Some non-steroidal anti-inflammatory creams and ointments have been applied to attempt to relieve the symptoms of RISI. It has been shown that topical application of triethylamine has a significant advantage in the prevention of radiation dermatitis by early recruitment of macrophages and stimulation of granulation tissue in comparison with the control group [43, 44].
Hydrogel dressings
Hydrogel is a form of highly cross-linked polymer that is polymerized by hydrophilic and hydrophobic groups and exhibits an outstanding characteristic of biocompatibility [45]. Censabella et al. indicated that patients with breast cancer received hydrogel dressings during radiotherapy period and significantly reduced the incidence of radiation-induced moist desquamation in comparison with the D-panthenol group [46].
Hyperbaric oxygen therapy (HBOT)
Hyperbaric oxygen therapy (HBOT) could be applied to form a hyperoxia environment in wound surface, keep the wound skin dry, and improve local microenvironment. The use of HBOT directly destroys the living environment of anaerobic bacteria, reduces the chance of bacterial infection, and accelerates the regression of redness and swelling [47, 48].
Superoxide dismutase (SOD)
SOD is a kind of antioxidant enzymes for scavenging free radicals in tissues by scavenging superoxide anion to reduce the adverse reactions [49]. Campan et al. reported that skin fibrosis induced by RT in breast cancer patients could be alleviated effectively with topical use of SOD [50].
Low-intensity laser
Main mechanisms of low-intensity laser for treating radiation dermatitis mainly are related to the expansion in local capillary, increase in vascular permeability, promotion of blood circulation, and proliferation of wound fibroblasts [51, 52].
MSCs for applications in regenerative medicine
MSCs are multipotent stem cells that can differentiate into a variety of cell types to maintain tissue integrity and intracellular homeostasis [53]. On-going clinical applications involved adipose-derived mesenchymal stem cells (ADMSCs), stromal vascular fractions (SVFs), and platelet-rich plasma (PRP) have raised in regenerative fields enrolling wound healing, scar management, breast augmentation, and soft tissue defects reconstruction [54, 55]. MSC actions through anti-inflammatory and immune-modulatory capabilities by secreting large number of anti-inflammatory cytokines, cell growth factors, antibacterial peptides, and proteins such as IL-17, indoleamine 2,3-dioxygenase (IDO), and other functional molecules [56, 57]. The application of MSCs to patients after the SARS-CoV-2 infection could alleviate the over-activation of the immune system, improve the lung microenvironment, and reduce the risk of cytokine storm syndrome (CSS) and acute respiratory distress syndrome (ARDS) of the body [58–60]. Animal experiments and clinical applications using MSCs in the treatment of RISI were summarized in Tables 2 and 3.
Table 2.
Summary of MSC source, injury type, and main findings from the treatment of RISI using MSCs in preclinical studies
| Publication | Animal model | MSC source | Injury type | Main findings | Reference |
|---|---|---|---|---|---|
| Rong et al. 2019 | Sprague-Dawley rat | Human fetal skin-derived stem cell | Radiation-induced skin injury | Enhanced radiation dermatitis angiogenesis | [1] |
| Kakabadze et al. 2019 | Lewis inbred rats | Rat bone marrow | Nonhealing wounds of radiation | Closing the burn wound increased the rate of healing | [4] |
| Myung et al. 2019 | SKH-1 hairless mice | Umbilical cord blood-derived | Combined radiation and wound injury | UCB-MSCs+PRP improve regeneration efficacy by enhancing angiogenesis | [7] |
| Wu et al. 2018 | Nude mice | Adipose-derived stem | Cutaneous punch wounds with radiation exposure | Revitalized irradiated tissues and accelerated wound healing | [10] |
| Sun et al. 2018 | Sprague-Dawley rat | WJ-MSCs | Radiation-induced skin injury | Accelerated wound closure and enhanced the wound healing quality | [16] |
| Liu et al. 2018 | SD rats | Human umbilical cord | Irradiation-induced skin ulcers | Promoted neovascularization and reepithelization, and improved healing of irradiation-induced skin ulcers | [13] |
| Lee et al. 2017 | Male C57BL/6 mice | Human umbilical cord blood-derived | Combined radiation wound | Enhanced wound healing and angiogenesis in the wound site | [15] |
| Riccobono et al. 2016 | Minipigs | Adipose tissue-derived stromal/stem cells | Cutaneous radiation syndrome | Have an effect on both muscle inflammation and regeneration | [20] |
| Jin et al. 2016 | Sprague-Dawley rat | Rat MSCs+PDGF | Radiation-induced skin ulceration | Radiation-induced skin ulceration | [22] |
| Rodriguez-Menocal et al. 2015 | C57BL/6 mice | Whole bone marrow, whole bone marrow cultured cells, MSCs | Radiation-induced delayed wound | Mixed bone marrow cell preparations may be superior to a more purified stem cell formulation in stimulating wound healing | [23] |
| Horton et al. 2013 | C3H/HeN mice | Syngeneic or allogeneic BMSC | IR-induced fibrosis | Alter the progression of radiation-induced fibrosis by altering macrophage phenotype and suppressing local inflammation | [61] |
| Huang et al. 2013 | Sprague-Dawley rat | Adipose-derived stem cells | Acute radiation ulcers | Contributed to vascularization by acting as angiogenesis-promoting cells | [26] |
| Forcheron et al. 2012 | Minipigs | Autologous adipose MSCs | Cutaneous radiation syndrome | Attracted numerous immune cells and accumulated at the dermis/subcutis barrier | [27] |
| Agay et al. 2010 | Minipig | Autologous bone marrow | Cutaneous radiation syndrome | Led to local accumulation of lymphocytes and improved vascularization | [30] |
| Riccobono et al. 2012 | Minipig | Adipocyte-derived stem cells | Cutaneous radiation syndrome | Improved wound healing | [32] |
| Horton et al. 2013 | Female C3H/HEN mice | Mouse bone marrow | Cutaneous radiation syndrome | Reduced inflammation and fibrosis | [33] |
| Zheng et al. 2015 | Sprague-Dawley rat | Rat bone marrow | Radiation-induced skin injury | Reduced inflammation, decreased expression of PGE2 and TGF-β1 | [38] |
| Xia et al. 2014 | Sprague-Dawley rat | Rat bone marrow MSCs expressing human VEGF and beta-defensin-3 | Radiation and excisional injury | Improved skin appendage regeneration and collagen deposition; shortened wound healing duration | [45] |
| Xie et al. 2013 | C3Hf/Kam female and C57Bl/6 male gnotobiotic mice | Murine bone | Radiation-induced wound | Haptized collagen strips to reduce radiation wound-healing deficits | [54] |
| Kotenko et al. 2012 | Male Wistar rats | Rats bone marrow | Radiation skin damage | Normalize intercellular interaction and influence the processes of proliferation and differentiation of skin cells | [8] |
| Yan et al. 2011 | Minipigs | MSCs expressing hPDGF loaded onto acellular amniotic membrane | Radiation and excisional injury | Improved granulation, re-epithelialization and angiogenesis | [62] |
| Ebrahimian et al. 2009 | C57Bl/6 male mice | Homogenous ADSCs | Skin punched wounds and then irradiated | Promoted dermal wound healing and enhanced wound closure | [63] |
| Hao et al. 2008 | Sprague-Dawley rat | Rat bone marrow MSCs expressing human PDGF and beta-defensin-2 | Radiation and excisional injury | Improved granulation tissue formation and reduced bacterial load | [64] |
| Francois et al. 2007; Mouiseddine et al. 2007 | NOD/SCID mice | Human bone marrow | Radiation dermatitis | Improved wound healing | [65] |
Table 3.
Summary of MSC source, injury type, and main findings from the treatment of RISI using MSCs in clinical applications
| Publication | MSC source | Combined treatment | Injury type | Main findings | Reference |
|---|---|---|---|---|---|
| Portas et al. 2016 | Allogenic cadaveric bone marrow | Surgical methods, hyperbaric oxygen treatment | Radiation-induced chronic skin lesions | Ulcer dimensions were reduced and remission of signs and symptoms | [2] |
| Guo et al. 2014 | Allogeneic bone marrow | HLA-mismatched peripheral blood stem cell transplantation | Radioactive skin ulceration | Suggesting a potential benefit of MSCs in radiation treatments | [5] |
| Kotenko et al. 2012 | Human bone marrow | Surgery and traditional conservative therapy | Severe local radiation ulcers | Rapid growth of granulation tissue and marginal epithelization and reduction in the ulcer | [8] |
| Bey et al. 2010 | Autologous bone marrow | Skin autograft | Severe radiation burn and radiation dermatitis | Modulating radiation inflammatory processes | [12] |
| Benderitter et al. 2010 | Autologous bone marrow | Plastic surgery or skin graft | Severe radiological burn | Being driven by the quality and the rapidity of the wound healing | [14] |
| Akita et al. 2010 | Autologous adipose tissue | Temporal artificial dermis impregnated+BFGF | Chronic radiation injuries | Wound was healed and no sign of recurrence appeared | [17] |
| Lataillade et al. 2007 | Autologous bone marrow | Surgery | Radiation burn | Open new prospects in radiotherapy complications | [19] |
Adipose-derived mesenchymal stem cells (ADMSCs)
ADMSC is one of the sources of stem cells which could secrete a variety of cytokines with multiple differentiation potential and immune exemption features. ADMSCs are extracted from adipose tissue and have biological characteristics of convenient obtain access [66–68]. Pietro et al. identified the phenotype of MSCs and elaborated that transcription factors (including OCT4, SOX2, NANOG, NEUROD1, PAX6, and SOX3) were involved in self-renewal capacity and multi-lineage differentiation potential. They verified the safety and efficacy of human ADMSCs localize in SVFs in breast reconstruction and assessed the volumetric persistence of breast augmentation by fat graft enhanced with ADMSCs in a 5-year follow-up retrospective case series [8, 69–73]. Besides, classic nanofat methods were innovatively modified by Pietro et al. and presented better outcomes of SVFs for the treatment of scars [62, 74–76]. They also proposed the maintenance and survival of SVF-enhanced autologous fat grafts were promoted by mixing with PRP and showed better outcomes of scars on the face compared with traditional surgical methods [63–65, 77]. Maria et al. combined PRP/insulin by improving chondrogenic and osteogenic differentiation of ADMSCs in a 3D collagen scaffold for treating osteochondral defects [78, 79].
Results of current applications with MSCs especially with ADMSCs indicated ADMSCs being new, alternative while bright approaches for treating those who suffer from the COVID-19 pandemic caused by coronavirus 2 (SARS-CoV-2) because of its easy access from autologous abdominal subcutaneous fat with low immunogenicity and little ethical disputes [80–82].
Bone marrow-derived mesenchymal stem cells (BMMSCs)
BMMSCs own the ability to differentiate into osteoblasts, chondroblasts, tendon cells, endothelial cells, glial cells, and hepatocyte. In view of the unique clinical and biological characteristics such as secretion of various hematopoietic growth factors, reconstruction of hematopoietic microenvironment, low immunogenicity together with vulnerable to transfection, and expression of foreign gene products, BMMSCs could partly make up for the deficiency of traditional treatment [83, 84]. BMMSCs still maintain its multi-directional differentiation potential and induce amplification in a destroyed tissue environment to participate in tissue repair or regeneration process [70, 85].
Umbilical cord mesenchymal stem cells (UCMSCs)
In recent years, UCMSCs have attracted more attention because of the stronger ability of proliferation and differentiation compared with BMMSCs. Apart from anti-inflammatory properties, UCMSCs play numerous roles in promoting systemic and local tissue repair, enhancing the ability of autophagy and self-repair together with increasing oxidative stress resistance in order to promote angiogenesis [86, 87]. Fang et al. studied that UCMSC-derived exosomes could reduce the myogenesis fibroblast accumulation and scar formation in a mouse wound defect model [88]. Wang et al. reported that human UCMSCs present positive treatment effects on acute radiation enteritis in rats [89].
Discussions
Various reasons include RT, occupational exposure, nuclear leakage accident, or/and nuclear war would lead to RISI that characterized by potential progressiveness and difficult to heal [90, 91]. Stewart et al. indicated that radiation-induced changes of signal molecule levels and formation of oxygen free radicals would cause DNA single-strand breaks, resulting in incomplete repair, premature senility, and accelerated differentiation [92].
The mechanisms of MSCs involved in wound progression are mainly through direct differentiation, immunomodulation, paracrine actions, and other related mechanisms such as recruitment of endogenous stem/progenitor cells, antibacterial and antioxidant effects with the intent of improving the wound microenvironment, shortening the inflammatory phase, and accelerating the proliferation phase to regulate matrix deposition and collagen remodeling, so as to arrive the accelerated healing and less scar formation [41, 54, 93] (Fig. 3). The inflammatory cytokines (e.g., anti-inflammatory cytokines like interleukin-10 (IL-10) and interleukin-4 (IL-4) and inflammatory factors such as tumor necrosis factor α (TNF-α) and interferon β (IFN-β)) and immune cells of wounds could activate MSCs that associated with proliferation and secretion of different immune cell subsets. Besides, MSCs promote the transformation of macrophages from M1-type to M2-type to accelerate the process of wound repair [94–96].
Fig. 3.
The mechanisms of MSCs in the field of regenerative medicine (e.g., in wound repair)
MSCs can be differentiated into keratinocytes, endothelial cells, sweat gland cells, or dermal papilla cells to participate in wound repair after direct induction in vitro. ADMSCs were obtained from the adipose tissues and are characterized by abundant sources and easy access compared to other sources of MSCs such as BMMSCs and UCMSCs. Altman et al. indicated that ADMSCs with fluorescent protein marked to dermal scaffolds could detect the expression of heat shock protein 47 (HSP-47) through fibroblast differentiation [97]. Ebrahimian et al. detected the expression of keratin 5 (K5) and keratin 14 (K14) which are signs of keratinocytes in epidermis after injection of ADMSCs into the back muscles of mice and indicated the differentiation potential of ADMSCs to epidermal keratinocytes [63]. Because of their lower immunogenicity, with no or low expression of major histocompatibility complex (MHC), host immune reaction is rarely caused after transplantation of ADMSCs [60, 91].
In recent years, BMMSCs have been one of the research hotspots for regenerative medicine with the following biological characteristics: (1) BMMSCs were able to grow adhering to the wall of the culture dishes; (2) the cell surface phenotype were CD3, CD4, CD8, CD14, CD19, CD34, CD45, and HLA-DR negative and CD29, CD44, CD71, CD73, CD90, CD105, and CD124 positive; (3) medium levels of positive MHC I expression and no expression of MHC II; (4) no expression of costimulatory molecules like B7-1, CD40, and CD86 [60, 92–94, 98, 99]. Silva et al. reported that BMMSCs could differentiate into vascular endothelial cells in vivo microenvironment and result in re-epithelialization of the wounded tissues [100]. Recent research has shown that MSCs participate in wound repair through paracrine growth factors, cytokines, and chemokines which have been described before. Some researchers reported that BMMSCs could be induced in radioactive environments that lead to further amplification to participate in tissue repair and regeneration process, resulting in achieving the purpose of MSCs transplantation in the treatment of radiation injury [101–103]. HGF and PGE2 that secreted by MSCs could, on the one hand, regulate the balance of TGF-β1 and TGF-β2 on the wound surface and, on the other hand, inhibit the EMT process to inhibit the formation of myofibroblasts and lead to less scar outcomes.
Though BMMSCs have been widely studied in recent years, obtaining BMMSCs would bring risks to patients because of the invasive operation which might restrict the application of BMMSCs [104, 105]. Umbilical cord blood contains a large number of MSCs with multi-lineage differentiation potential and homing properties. UCMSCs could be derived from a wide range of sources with easier access and lower immunogenicity compared with other sources of MSCs [106, 107]. Human UCMSCs promote neovascularization and reepithelization to enhance the healing of radiation-induced skin ulcer might be activated by PI3K/Akt signaling pathway [108]. In addition, the result in the study indicated the regulatory effect UCMSCs to Flt-3L and TGF-β1. What is more, VEGF/SDF-1-CXCR4, inflammatory cytokines (e.g., IL-6 and IL-8), and growth factor receptor axis (e.g., HGF-c-met and PDGF axis) are involved in the recruitment of endogenous stem/progenitor cells of MSCs to promote tissue repair [83, 109–111].
Conclusion
Many studies have confirmed the effects of MSCs on tissue repair as an acellular therapy in recent years. This review focused on the latest application of MSCs from various sources as a promising agent for RISI both in animal models and clinical trials and discussed related mechanism. The specific mechanism of MSCs on tissue repair and regenerative medicine need to be further studied in the future.
Acknowledgements
We thank Haoruo Zhang and Caixiang Chen for their help to the manuscript.
This work was financed by National Natural Science Foundation of China (grant No. 81971855, 81671930), Joint funding Project of Science and Technology Innovation in Fujian Province (grant No. 2017Y9101), Special Financial Funds of Fujian Province (grant No. 2018B054), and Industrial Technology Joint Innovation Project of Fujian Provincial Development and Reform Commission (grant No. 2011601).
Abbreviations
- RISI
Radiation-induced skin injury
- RT
Radiotherapy
- MSCs
Mesenchymal stem cells
- ROS
Reactive oxygen species
- NO
Nitric oxide
- RNS
Reactive nitrogen species
- HMGB1
High mobility group box 1
- ATM
Ataxia telangiectasia mutated
- DNA-PKcs
DNA-dependent protein kinase catalytic subunit
- FFP
Fresh-frozen plasma
- HBOT
Hyperbaric oxygen therapy
- CHK2
Checkpoint kinase 2
- SVFs
Stromal vascular fractions
- PRP
Platelet-rich plasma
- SOD
Superoxide dismutase
- VEGF
Vascular endothelial growth factor
- IDO
Indoleamine 2,3-dioxygenase
- PDGF
Platelet-derived growth factor
- CSS
Cytokine storm syndrome
- ARDS
Acute respiratory distress syndrome
- ADMSCs
Adipose-derived mesenchymal stem cells
- BMMSCs
Bone marrow-derived mesenchymal stem cells
- UCMSCs
Umbilical cord mesenchymal stem cells
- SARS-CoV-2
Coronavirus 2
- IL-10
Interleukin-10
- IL-4
Interleukin-4
- TNF-α
Tumor necrosis factor α
- IFN-β
Interferon β
- HSP-47
Heat shock protein 47
- K5
Keratin 5
- K14
K14 keratin
- MHC
Major histocompatibility complex
Authors’ contributions
XC designed the manuscript. ZF wrote the text. PC, ST, and AC revised the format of the figures. CZ, GP, and ML revised the manuscript. XC approved the final submission. All authors discussed the results and reviewed the manuscript. The authors read and approved the final manuscript.
Funding
This work was financed by National Natural Science Foundation of China (grant No. 81971855, 81671930), Joint funding Project of Science and Technology Innovation in Fujian Province (grant No. 2017Y9101), Special Financial Funds of Fujian Province (grant No. 2018B054), and Industrial Technology Joint Innovation Project of Fujian Provincial Development and Reform Commission (grant No. 2011601).
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Rong X, Li J, Yang Y, Shi L, Jiang T. Human fetal skin-derived stem cell secretome enhances radiation-induced skin injury therapeutic effects by promoting angiogenesis. Stem Cell Res Ther. 2019;10:383. doi: 10.1186/s13287-019-1456-x. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 2.Portas M, Mansilla E, Drago H, Dubner D, Radl A, Coppola A, Di Giorgio M. Use of human cadaveric mesenchymal stem cells for cell therapy of a chronic radiation-induced skin lesion: a case report. Radiat Prot Dosim. 2016;171(1):99–106. [DOI] [PubMed]
- 3.Carter S, Zahs A, Palmer J, Wang L, Ramirez L, Gamelli R, Kovacs E. Intestinal barrier disruption as a cause of mortality in combined radiation and burn injury. Shock. 2013;40(4):281–9. [DOI] [PMC free article] [PubMed]
- 4.Kakabadze Z, Chakhunashvili D, Gogilashvili K, Ediberidze K, Chakhunashvili K, Kalandarishvili K, Karalashvili L. Bone marrow stem cell and decellularized human amniotic membrane for the treatment of nonhealing wound after radiation therapy. Exp Clin Transplant. 2019;17(null):92–98. doi: 10.6002/ect.MESOT2018.O29. [DOI] [PubMed] [Google Scholar]
- 5.Guo M, Dong Z, Jianhui Q, Yu C, Qiyun S, Hu K, Guangxian L, Li W, Bo Y, Qiuhong M, et al. Severe acute radiation syndrome: treatment of a lethally 60Co-source irradiated accident victim in China with HLA-mismatched peripheral blood stem cell transplantation and mesenchymal stem cells. J Radiat Res. 2014;55(2):205–209. doi: 10.1093/jrr/rrt102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mendoza A, Neely C, Charles A, Kartchner L, Brickey W, Khoury A, Sempowski G, Ting J, Cairns B, Maile R. Radiation combined with thermal injury induces immature myeloid cells. Shock. 2012;38(5):532–542. doi: 10.1097/SHK.0b013e31826c5b19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Myung H, Jang H, Myung JK, Lee C, Lee J, Kang JH. Platelet-rich plasma improves the therapeutic efficacy of mesenchymal stem cells by enhancing their secretion of angiogenic factors in a combined radiation and wound injury model. Exp Dermatol. 2020;29:158-167. [DOI] [PubMed]
- 8.Kotenko K, Moroz B, Nadezhina N, Galstyan I, Eremin I, Deshevoy J, Lebedev V, Slobodina T, Grinakovskaya D, Zhgutov Y, et al. Successful treatment of localised radiation lesions in rats and humans by mesenchymal stem cell transplantation. Radiat Prot Dosim. 2012;151(4):661–665. doi: 10.1093/rpd/ncs177. [DOI] [PubMed] [Google Scholar]
- 9.Ablordeppey E, Drewry A, Theodoro D, Tian L, Fuller BM, Griffey R. Current practices in central venous catheter position confirmation by point of care ultrasound: a survey of early adopters. Shock. 2019;51(5):613–618. doi: 10.1097/SHK.0000000000001218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wu S, Shirado T, Mashiko T, Feng J, Asahi R, Kanayama K, Mori M, Chi D, Sunaga A, Sarukawa S. Therapeutic effects of human adipose-derived products on impaired wound healing in irradiated tissue. Plast Reconstr Surg. 2018;142(2):383–391. doi: 10.1097/PRS.0000000000004609. [DOI] [PubMed] [Google Scholar]
- 11.Kalns J, Ryan K, Mason P, Bruno J, Gooden R, Kiel J. Oxidative stress precedes circulatory failure induced by 35-ghz microwave heating. Shock. 2000;13(1):52–59. doi: 10.1097/00024382-200013010-00010. [DOI] [PubMed] [Google Scholar]
- 12.Eric B, Marie P, Patrick D, Marc B, Michel B, François T, Pierre B, Isabelle E, Laetitia B, Muriel G. Emerging therapy for improving wound repair of severe radiation burns using local bone marrow-derived stem cell administrations. Wound Repair Regen. 2010;18(1):50–58. doi: 10.1111/j.1524-475X.2009.00562.x. [DOI] [PubMed] [Google Scholar]
- 13.Liu Z, Yu D, Xu J, Li X, Wang X, He Z, Zhao T. Human umbilical cord mesenchymal stem cells improve irradiation-induced skin ulcers healing of rat models. Biomed Pharmacother. 2018;101:729. doi: 10.1016/j.biopha.2018.02.093. [DOI] [PubMed] [Google Scholar]
- 14.Benderitter M, Gourmelon P, Bey E, Chapel A, Clairand I, Prat M, Lataillade J. New emerging concepts in the medical management of local radiation injury. Health Phys. 2010;98:851–857. doi: 10.1097/HP.0b013e3181c9f79a. [DOI] [PubMed] [Google Scholar]
- 15.Lee C, Shim S, Jang H, Myung H, Lee J, Bae C, Myung J, Kim M, Lee S, Jang W. Human umbilical cord blood–derived mesenchymal stromal cells and small intestinal submucosa hydrogel composite promotes combined radiation-wound healing of mice. Cytotherapy. 2017;19(9):1048–1059. doi: 10.1016/j.jcyt.2017.06.007. [DOI] [PubMed] [Google Scholar]
- 16.Sun J, Zhang Y, Song X, Zhu J, Zhu Q. The healing effects of conditioned medium derived from mesenchymal stem cells on radiation-induced skin wounds in rats. Cell Transplant. 2018;28:105–15. [DOI] [PMC free article] [PubMed]
- 17.Akita S, Akino K, Hirano A, Ohtsuru A, Yamashita S. Noncultured autologous adipose-derived stem cells therapy for chronic radiation injury. Stem Cells Int. 2010;2010:532704. doi: 10.4061/2010/532704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Goessler U, Bugert P, Kassner S, Stern-Straeter J, Bran G, Sadick H, Hörmann K, Riedel Frank: In vitro analysis of radiation-induced dermal wounds. Otolaryngol Head Neck Surg, 2010,142(6), 845–850. [DOI] [PubMed]
- 19.Lataillade J, Doucet C, Bey E, Carsin H, Huet C, Clairand I, Bottollier J, Chapel A, Ernou I, Gourven M, et al. New approach to radiation burn treatment by dosimetry-guided surgery combined with autologous mesenchymal stem cell therapy. Regen Med. 2007;2(5):785–794. doi: 10.2217/17460751.2.5.785. [DOI] [PubMed] [Google Scholar]
- 20.Riccobono D, Agay D, François S, Scherthan H, Drouet M, Forcheron F. Contribution of intramuscular autologous adipose tissue-derived stem cell injections to treat cutaneous radiation syndrome: preliminary results. Health Phys. 2016;111(2):117–26. [DOI] [PubMed]
- 21.Biju P, Garg S, Wang W, Choudhry M, Kovacs E, Fink L, Hauer J. Procalcitonin as a predictive biomarker for total body irradiation-induced bacterial load and lethality in mice. Shock. 2012;38(2):170–176. doi: 10.1097/SHK.0b013e31825b2db3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jin I, Kim J, Wu H, Hwang S. Effect of mesenchymal stem cells and platelet-derived growth factor on the healing of radiation induced ulcer in rats. Tissue Eng Regen Med. 2016;13(1):78–90. doi: 10.1007/s13770-015-0055-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rodriguez M, Shareef S, Salgado M, Shabbir A, Van B. Role of whole bone marrow, whole bone marrow cultured cells, and mesenchymal stem cells in chronic wound healing. Stem Cell Res Ther. 2015;6(1):24. doi: 10.1186/s13287-015-0001-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Frei M, Ryan K, Berger R, Jauchem J. Sustained 35-GHz radiofrequency irradiation induces circulatory failure. Shock. 1995;4(4):289–293. doi: 10.1097/00024382-199510000-00010. [DOI] [PubMed] [Google Scholar]
- 25.Simon P, Lorton F, Guiziou N, Levieux K, Vrignaud B, Masson D, Dupas B, Gras L. Serum S100β neuroprotein reduces use of cranial computed tomography in children after minor head trauma. Shock. 2015;44(5):410–416. doi: 10.1097/SHK.0000000000000442. [DOI] [PubMed] [Google Scholar]
- 26.Huang S, Huang C, Shyu J, Lee H, Chen S, Chan J, Huang S. Promotion of wound healing using adipose-derived stem cells in radiation ulcer of a rat model. J Biomed Sci. 2013;20(1):51. doi: 10.1186/1423-0127-20-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ryan K, Frei M, Berger R, Jauchem J. Does nitric oxide mediate circulatory failure induced by 35-GHz microwave heating? Shock. 1996;6(1):71–76. doi: 10.1097/00024382-199607000-00015. [DOI] [PubMed] [Google Scholar]
- 28.Forcheron F, Agay D, Scherthan H, Riccobono D, Herodin F, Meineke V, Drouet M. Autologous adipocyte derived stem cells favour healing in a minipig model of cutaneous radiation syndrome. PLoS One. 2012;7:e31694. doi: 10.1371/journal.pone.0031694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ryan K, Frei M, Jauchem J. Circulatory failure induced by 35 GHz microwave heating: effects of chronic nitric oxide synthesis inhibition. Shock. 1997;7(1):70–76. doi: 10.1097/00024382-199701000-00010. [DOI] [PubMed] [Google Scholar]
- 30.Diane A, Harry S, Fabien F, Nancy G, Francis H, Viktor M, Michel D. Multipotent mesenchymal stem cell grafting to treat cutaneous radiation syndrome: development of a new minipig model. Exp Hematol. 2010;38(10):945–956. doi: 10.1016/j.exphem.2010.06.008. [DOI] [PubMed] [Google Scholar]
- 31.Tharakan B, Hunter F, Smythe W, Childs E. Alpha-lipoic acid attenuates hemorrhagic shock-induced apoptotic signaling and vascular hyperpermeability. Shock. 2008;30(5):571–577. doi: 10.1097/SHK.0b013e31816a7308. [DOI] [PubMed] [Google Scholar]
- 32.Diane R, Diane A, Harry S, Fabien F, Mylène V, Bruno B, Viktor M, Michel D. Application of adipocyte-derived stem cells in treatment of cutaneous radiation syndrome. Health Phys. 2012;103(2):120. doi: 10.1097/HP.0b013e318240595b. [DOI] [PubMed] [Google Scholar]
- 33.Jason A, Kathryn E, Eun J, Ayla O, Bradley T, Jeffrey F, Deborah E. Mesenchymal stem cells inhibit cutaneous radiation-induced fibrosis by suppressing chronic inflammation. Stem Cells. 2013;31(10):2231–41. [DOI] [PMC free article] [PubMed]
- 34.Patel D, Mittal S, Tiwari N, Maurya A, Singh D, Pandey A, Pal A. Plasmodium-salmonella coinfection induces intense inflammatory response, oxidative stress, and liver damage: a mice model study for therapeutic strategy. Shock. 2018;50(6):741–749. doi: 10.1097/SHK.0000000000001111. [DOI] [PubMed] [Google Scholar]
- 35.Tânia G, Song Y, Brede D, Xie L, Gutzkow K, Salbu B. Gamma radiation induces dose-dependent oxidative stress and transcriptional alterations in the freshwater crustacean daphnia magna. Sci Total Environ. 2018;620–629(JUL.1):206–216. doi: 10.1016/j.scitotenv.2018.02.039. [DOI] [PubMed] [Google Scholar]
- 36.Aura R, Bibiana V, Tautgirdas R, Sebastian B. The effect of UVB irradiation and oxidative stress on the skin barrier-a new method to evaluate sun protection factor based on electrical impedance spectroscopy. Sensors (Basel) 2019;19(10):2376. doi: 10.3390/s19102376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hsu D, Li Y, Chien S, Liu M. Effects of sesame oil on oxidative stress and hepatic injury after cecal ligation and puncture in rats. Shock. 2004;21(5):466–469. doi: 10.1097/00024382-200405000-00011. [DOI] [PubMed] [Google Scholar]
- 38.Zheng K, Wu W, Yang S, Huang L, Chen J, Gong C, Fu Z, Zhang L, Tan T. Bone marrow mesenchymal stem cell implantation for the treatment of radioactivityinduced acute skin damage in rats. Mol Med Rep. 2015;12:7065–7071. doi: 10.3892/mmr.2015.4270. [DOI] [PubMed] [Google Scholar]
- 39.Ahmed N, Radwan N, Aboul E, Salama N. The antioxidant effect of Green Tea Mega EGCG against electromagnetic radiation-induced oxidative stress in the hippocampus and striatum of rats. Electromagn Biol Med. 2017;36(1):63–73. doi: 10.1080/15368378.2016.1194292. [DOI] [PubMed] [Google Scholar]
- 40.Singh K, Gautam R, Meena R, Nirala J, Jha S, Rajamani P. Effect of mobile phone radiation on oxidative stress, inflammatory response, and contextual fear memory in wistar rat. Environ Sci Pollut Res Int. 2020;27(16):19340–19351. doi: 10.1007/s11356-020-07916-z. [DOI] [PubMed] [Google Scholar]
- 41.Holmes C, Plichta J, Gamelli R, Radek K. Burn injury alters epidermal cholinergic mediators and increases HMGB1 and Caspase 3 in autologous donor skin and burn margin. Shock. 2017;47(2):175–183. doi: 10.1097/SHK.0000000000000752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cejka C, Kossl J, Hermankova B, Holan V, Kubinova S, Zhang J. Therapeutic effect of molecular hydrogen in corneal uvb-induced oxidative stress and corneal photodamage. Sci Rep. 2017;7(1):18017. doi: 10.1038/s41598-017-18334-6. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 43.Oliveirag G, Shimoda K, Enkhbaatar P, Jodoin J, Burke A, Chinkes D. Skin nitric oxide and its metabolites are increased in nonburned skin after thermal injuries. Shock. 2004;22(3):278–282. doi: 10.1097/01.shk.0000135259.90311.33. [DOI] [PubMed] [Google Scholar]
- 44.Breslin J, Wu M, Guo M, Reynoso R, Yuan S. Toll-like receptor 4 contributes to microvascular inflammation and barrier dysfunction in thermal injury. Shock. 2008;29(3):1. doi: 10.1097/SHK.0b013e3181454975. [DOI] [PubMed] [Google Scholar]
- 45.Xia Z, Zhang C, Zeng Y, Wang T, Ai G. Transplantation of BMSCs expressing hVEGF165/hBD3 promotes wound healing in rats with combined radiationwound injury. Int Wound J. 2014;11:293–303. doi: 10.1111/j.1742-481X.2012.01090.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kruse J, Strootman E, Bart C, Visser A, Leer J, Wondergem J. Radiation-induced changes in gene expression and distribution of atrial natriuretic peptide (ANP) in different anatomical regions of the rat heart. Int J Radiat Biol. 2002;78(4):297–304. doi: 10.1080/09553000110102021. [DOI] [PubMed] [Google Scholar]
- 47.Yarnold J, Brotons M. Pathogenetic mechanisms in radiation fibrosis. Radiother Oncol. 2010;97(1):149–161. doi: 10.1016/j.radonc.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 48.Etulain J. Platelets in wound healing and regenerative medicine. Platelets. 2018;29(6):556–68. [DOI] [PubMed]
- 49.Miron R, Dard M, Weinreb M. Enamel matrix derivative, inflammation and soft tissue wound healing. J Periodontal Res. 2015;50(5):555–69. [DOI] [PubMed]
- 50.Litvin F, Vasilieva G, Baranov V. Functional features of microcirculation system and its possibilities of oxygen transport in children and youths of Bryansk region in conditions of radioecological pressure. Radiats Biol Radioecol. 2016;56(5):514–522. [PubMed] [Google Scholar]
- 51.Wei L, Zhang J, Xiao X, Mai H, Zheng K, Sun W, Ke Z, Lei W, Feng L, Yuan L, et al. Multiple injections of human umbilical cord-derived mesenchymal stromal cells through the tail vein improve microcirculation and the microenvironment in a rat model of radiation myelopathy. J Transl Med. 2014;12(1):246. [DOI] [PMC free article] [PubMed]
- 52.Michael W, Raphael G, Ewa D, Natalia C, Elena G, Lilia L, William H. Marrow-derived stromal cell delivery on fibrin microbeads can correct radiation-induced wound-healing deficits. J Investig Dermatol. 2013;133(2):553–561. doi: 10.1038/jid.2012.326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Okazaki S, Funasaka Y, Wakamatsu K, Kawana S, Saeki H. Effect of infrared radiation a on photoaged hairless mice harboring eumelanin and pheomelanin in the epidermis. J Dermatol. 2015;42(4):382–390. doi: 10.1111/1346-8138.12790. [DOI] [PubMed] [Google Scholar]
- 54.Hasoun L, Bailey S, Outlaw K, Ayling J. Rearrangement and depletion of folate in human skin by ultraviolet radiation. Br J Dermatol. 2015;173(4):1087–1090. doi: 10.1111/bjd.13885. [DOI] [PubMed] [Google Scholar]
- 55.Johnson Jodi L., Koetsier Jennifer L., Sirico Anna., Agidi Ada T., Antonini Dario., Missero C, Green K: The desmosomal protein desmoglein 1 aids recovery of epidermal differentiation after acute UV light exposure. J Invest Dermatol, 2014,134(8), 2154–2162. [DOI] [PMC free article] [PubMed]
- 56.Auerswald S, Schreml S, Meier R, Soares A, Haubner F. Wound monitoring of pH and oxygen in patients after radiation therapy. Radiat Oncol. 2019;14(1):199. [DOI] [PMC free article] [PubMed]
- 57.Alhasaniah A, Sherratt M, O'Neill C. The impact of ultraviolet radiation on barrier function in human skin: molecular mechanisms and topical therapeutics. Curr Med Chem. 2018;25(40):5503–5511. doi: 10.2174/0929867324666171106164916. [DOI] [PubMed] [Google Scholar]
- 58.Jörg R, Reichrath S. Hope and challenge: the importance of ultraviolet (UV) radiation for cutaneous vitamin D synthesis and skin cancer. Scand J Clin Lab Invest Suppl. 2012;243:112–119. doi: 10.3109/00365513.2012.682876. [DOI] [PubMed] [Google Scholar]
- 59.Hart P, Mary N, Byrne S, Lesley E. Exposure to ultraviolet radiation in the modulation of human diseases. Ann Rev Pathol Mech Dis. 2019;14(1):421058260. doi: 10.1146/annurev-pathmechdis-012418-012809. [DOI] [PubMed] [Google Scholar]
- 60.Christopher I, Kimberly P, Roger E, Nicholas J, Michael K, Raymond S, Steven E. Efficacy of corticosteroids and external beam radiation in the management of moderate to severe thyroid eye disease. J Neuroophthalmol. 2007;27(3):205. doi: 10.1097/WNO.0b013e31814a5ef8. [DOI] [PubMed] [Google Scholar]
- 61.Horton J, Hudak K, Chung E, White A, Scroggins B, Burkeen J, Citrin D. Mesenchymal stem cells inhibit cutaneous radiation-induced fibrosis by suppressing chronic inflammation. Stem Cells. 2013;31(10):2231–41. [DOI] [PMC free article] [PubMed]
- 62.Pietro G, Claudio C, Barbara DA, et al. Impact of the different preparation methods to obtain human adipose-derived stromal vascular fraction cells (AD-SVFs) and human adipose-derived mesenchymal stem cells (AD-MSCs): enzymatic digestion versus mechanical centrifugation. Int J Mol Sci. 2019;20(21):5471. [DOI] [PMC free article] [PubMed]
- 63.Cervelli V, Bocchini I, Di Pasquali C, et al. P.R.L. platelet rich lipotransfert: our experience and current state of art in the combined use of fat and PRP. Biomed Res Int. 2013;2013:434191. doi: 10.1155/2013/434191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Giovanna SM, Alessandra B, Pietro G, et al. Combined treatment with platelet-rich plasma and insulin favours chondrogenic and osteogenic differentiation of human adipose-derived stem cells in three-dimensional collagen scaffolds. J Tissue Eng Regen Med. 2017;11:2398–2410. doi: 10.1002/term.2139. [DOI] [PubMed] [Google Scholar]
- 65.Pietro G, Giovanna SM, Alessandra B, et al. Concise review: the use of adipose-derived stromal vascular fraction cells and platelet rich plasma in regenerative plastic surgery. Stem Cells. 2017;35:117–134. doi: 10.1002/stem.2498. [DOI] [PubMed] [Google Scholar]
- 66.Shukla T, Sands B. Novel non-biologic targets for inflammatory bowel disease. Curr Gastroenterol Rep. 2019;21(5):22. [DOI] [PubMed]
- 67.Liao Y, Feng G, Dai T, Long F, Tang J, Pu Y, Zheng X, Cao S, Xu S, Du X. Randomized, self-controlled, prospective assessment of the efficacy of mometasone furoate local application in reducing acute radiation dermatitis in patients with head and neck squamous cell carcinomas. Medicine (Baltimore) 2019;98(52):e18230. doi: 10.1097/MD.0000000000018230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Abbas H, René-Jean B. Trolamine emulsion for the prevention of radiation dermatitis in patients with squamous cell carcinoma of the head and neck. Support Care Cancer. 2012;20(1):185–190. doi: 10.1007/s00520-011-1110-3. [DOI] [PubMed] [Google Scholar]
- 69.Elliott E. Phase III trial of an emulsion containing trolamine for the prevention of radiation dermatitis in patients with advanced squamous cell carcinoma of the head and neck: results of Radiation Therapy Oncology Group Trial 99-13. J Clin Oncol. 2006;24(13):2092–2097. doi: 10.1200/JCO.2005.04.9148. [DOI] [PubMed] [Google Scholar]
- 70.William J, Steve W, Geert W, David H, Raul C, Tony E. Characterization of two classes of pancreatic shock factors: functional differences exhibited by hydrophilic and hydrophobic shock factors. Shock. 2003;20(4):356–362. doi: 10.1097/01.shk.0000082442.66379.90. [DOI] [PubMed] [Google Scholar]
- 71.Censabella S, Claes S, Orlandini M, Braekers R, Bulens P. Efficacy of a hydroactive colloid gel versus historical controls for the prevention of radiotherapy-induced moist desquamation in breast cancer patients. Eur J Oncol Nurs. 2017;29:1–7. doi: 10.1016/j.ejon.2017.04.003. [DOI] [PubMed] [Google Scholar]
- 72.Legeza V, Galenko Y, Zinov'ev E, Paramonov B, Kreichman G, Turkovskii I, Gumenyuk E, Karnovich A, Khripunov A. Effects of new wound dressings on healing of thermal burns of the skin in acute radiation disease. Bull Exp Biol Med. 2004;138(3):311–315. doi: 10.1007/s10517-005-0029-4. [DOI] [PubMed] [Google Scholar]
- 73.Gentile Pietro, Scioli Maria Giovanna, Bielli Alessandra et al. Comparing different nanofat procedures on scars: role of the stromal vascular fraction and its clinical implications. Regen Med. 2017;12:939–52. [DOI] [PubMed]
- 74.Pietro G, Donato C, Enza P, et al. Engineered fat graft enhanced with adipose-derived stromal vascular fraction cells for regenerative medicine: clinical, histological and instrumental evaluation in breast reconstruction. J Clin Med. 2019;8(4):504. [DOI] [PMC free article] [PubMed]
- 75.Pietro G, Ashutosh K, Donato C, et al. Fat graft enhanced with adipose-derived stem cells in aesthetic breast augmentation: clinical, histological, and instrumental evaluation. Aesthet Surg J. 2020;40:962–977. doi: 10.1093/asj/sjz292. [DOI] [PubMed] [Google Scholar]
- 76.Pietro G, Serena PM, Claudio C. Characteristics and potentiality of human adipose-derived stem cells (hASCs) obtained from enzymatic digestion of fat graft. Cells. 2019;8(3):282. [DOI] [PMC free article] [PubMed]
- 77.Pietro G, Simone G, Review C. Adipose-derived stem cells (ASCs) and adipocyte-secreted exosomal microRNA (A-SE-miR) modulate cancer growth and proMote wound repair. J Clin Med. 2019;8(6):855. [DOI] [PMC free article] [PubMed]
- 78.Pietro G, Barbara DA, Methap P, et al. Adipose-derived stromal vascular fraction cells and platelet-rich plasma: basic and clinical evaluation for cell-based therapies in patients with scars on the face. J Craniofac Surg. 2014;25:267–272. doi: 10.1097/01.scs.0000436746.21031.ba. [DOI] [PubMed] [Google Scholar]
- 79.Pietro G, Aris S, Jacopo P, et al. Research progress on mesenchymal stem cells (MSCs), adipose-derived mesenchymal stem cells (AD-MSCs), drugs, and vaccines in inhibiting COVID-19 disease. Aging Dis. 2020;11:1191–1201. doi: 10.14336/AD.2020.0711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pietro G, Aris S. Adipose stem cells (ASCs) and stromal vascular fraction (SVF) as a potential therapy in combating (COVID-19)-disease. Aging Dis. 2020;11:465–469. doi: 10.14336/AD.2020.0422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pietro G, Aris S. Adipose-derived stromal stem cells (ASCs) as a new regenerative immediate therapy combating coronavirus (COVID-19)-induced pneumonia. Expert Opin Biol Ther. 2020;20:711–716. doi: 10.1080/14712598.2020.1761322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Nandra K, Takahashi K, Collino M, Benetti E, Wong W, Goh F, Suzuki K, Patel N, Thiemermann C. Acute treatment with bone marrow-derived mononuclear cells attenuates the organ injury/dysfunction induced by hemorrhagic shock in the rat. Shock. 2012;37(6):592. doi: 10.1097/SHK.0b013e31824e4c0d. [DOI] [PubMed] [Google Scholar]
- 83.Mao M. Intravenous delivery of bone marrow-derived endothelial progenitor cells improves survival and attenuates lipopolysaccharide-induced lung injury in rats. Shock. 2010;34:196–204. [DOI] [PubMed]
- 84.Liu Y, Chen J, Zhang Z, Ma X, Chen Y, Du X, Liu H, Zong Y, Lu G. NLRP3 inflammasome activation mediates radiation-induced pyroptosis in bone marrow-derived macrophages. Cell Death Dis. 2017;8(2):e2579. doi: 10.1038/cddis.2016.460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Liu D, Kong F, Yuan Y, Seth P, Xu W, Wang H, Xiao F, Wang L, Zhang Q, Yang Y, et al. Decorin-modified umbilical cord mesenchymal stem cells (MSCs) attenuate radiation-induced lung injuries via regulating inflammation, fibrotic factors, and immune responses. Int J Radiat Oncol Biol Phys. 2018;101(4):945–956. doi: 10.1016/j.ijrobp.2018.04.007. [DOI] [PubMed] [Google Scholar]
- 86.Guo D, Zhu X, Li Q, Liu G, Ruan G, Pang R, Chen Y, Wang Q, Wang J, Liu J, et al. Efficacy and mechanisms underlying the effects of allogeneic umbilical cord mesenchymal stem cell transplantation on acute radiation injury in tree shrews. Cytotechnology. 2018;70(5):1–22. doi: 10.1007/s10616-018-0239-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Fang S, Xu C, Zhang Y, Xue C, Yang C, Bi H, Qian X, Wu M, Ji K, Zhao Y, et al. Umbilical cord-derived mesenchymal stem cell-derived Exosomal microRNAs suppress myofibroblast differentiation by inhibiting the transforming growth factor-β/SMAD2 pathway during wound healing. Stem Cells Transl Med. 2016;5(10):1425–1439. doi: 10.5966/sctm.2015-0367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lee J, Choi K, Park J, Moon H, Choi G, Lee J, Suh E, Kim H, Eun S, Kim G, et al. Bisphenol A distribution in serum, urine, placenta, breast milk, and umbilical cord serum in a birth panel of mother–neonate pairs. Sci Total Environ. 2018;1(626):1494–1501. doi: 10.1016/j.scitotenv.2017.10.042. [DOI] [PubMed] [Google Scholar]
- 89.Wang R, Zhu C, Qiao P, Liu J, Zhao Q, Wang K, Zhao T. Experimental treatment of radiation pneumonitis with human umbilical cord mesenchymal stem cells. Asian Pac J Trop Med. 2014;7(4):262–266. doi: 10.1016/S1995-7645(14)60034-1. [DOI] [PubMed] [Google Scholar]
- 90.Silva V, Hilly O, Strenov Y, Tzabari C, Hauptman Y, Feinmesser R. Effect of cell phone-like electromagnetic radiation on primary human thyroid cells. Int J Radiat Biol. 2016;92:107–115. doi: 10.3109/09553002.2016.1117678. [DOI] [PubMed] [Google Scholar]
- 91.Narayanan S, Jetti R, Kesari K, Kumar R, Nayak S, Bhat P. Radiofrequency electromagnetic radiation-induced behavioral changes and their possible basis. Environ Sci Pollut Res Int. 2019;26(30):30693–710. [DOI] [PubMed]
- 92.Xiang M, Yuan Y, Fan L, Li Y, Li A, Yin L, Scott M, Xiao G, Billiar T, Wilson M, et al. Role of macrophages in mobilization of hematopoietic progenitor cells from bone marrow after hemorrhagic shock. Shock. 2012;37:518–523. doi: 10.1097/SHK.0b013e318249b81d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Xuan W, Wang Y, Tang Y, Ali A, Hu H, Maienschein-Cline M, Ashraf M, et al. Cardiac progenitors induced from human induced pluripotent stem cells with cardiogenic small molecule effectively regenerate infarcted hearts and attenuate fibrosis. Shock. 2018;50:627–639. doi: 10.1097/SHK.0000000000001133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Zhao Y, Wang J, Yano N, Zhang L, Wang H, Zhang S, Qin G, Dubielecka P, Zhuang S, Liu P. Irisin promotes cardiac progenitor cell-induced myocardial repair and functional improvement in infarcted heart. J Cell Physiol. 2019;234(2):1671–1681. doi: 10.1002/jcp.27037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Qiao Z, Greven J, Horst K, Pfeifer R, Kobbe P, Pape H, Hildebrand F. Fracture healing and the underexposed role of extracellular vesicle-based crosstalk. Shock. 2018;49(5):486–496. doi: 10.1097/SHK.0000000000001002. [DOI] [PubMed] [Google Scholar]
- 96.Altman A, Abdul K, Alt E, Butler C. Adipose tissue-derived stem cells enhance bioprosthetic mesh repair of ventral hernias. Plast Reconstr Surg. 2010;126(3):855–857. doi: 10.1097/PRS.0b013e3181e6044f. [DOI] [PubMed] [Google Scholar]
- 97.Forsythe R, Xu D, Lu Q, Deitch E. Lipopolysaccharide-induced enterocyte-derived nitric oxide induces intestinal monolayer permeability in an autocrine fashion. Shock. 2002;17(3):180. doi: 10.1097/00024382-200203000-00004. [DOI] [PubMed] [Google Scholar]
- 98.Xiu F, Jeschke M. Perturbed mononuclear phagocyte system in severely burned and septic patients. Shock. 2013;40(2):81–88. doi: 10.1097/SHK.0b013e318299f774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Stewart F, Seemann I, Hoving S, Russellet N. Understanding radiation-induced cardiovascular damage and strategies for intervention. Clin Oncol (R Coll Radiol) 2013;25:617–624. doi: 10.1016/j.clon.2013.06.012. [DOI] [PubMed] [Google Scholar]
- 100.Laura B, John F, Jenny U, Coombs C, Zehir A, Ptashkin R, Berger M, Levine R, Fagin J. Radioactive iodine-related clonal hematopoiesis in thyroid cancer is common and associated with decreased survival. J Clin Endocrinol Metab. 2018;103(11):4216–4223. doi: 10.1210/jc.2018-00803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Tycho I, Joe C, Krijn V, Joost P, Maarten J, Pieter A, Eric B, Steven A. Non-surgical stem cell delivery strategies and in vivo cell tracking to injured myocardium. Int J Cardiovasc Imaging. 2011;27(3):367–383. doi: 10.1007/s10554-010-9658-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Yang S, Chung C, Ayala A, Chaudry I, Wang P. Differential alterations in cardiovascular responses during the progression of polymicrobial sepsis in the mouse. Shock. 2002;17(1):55. doi: 10.1097/00024382-200201000-00010. [DOI] [PubMed] [Google Scholar]
- 103.Ince E, Temiz A, Ezer S. Poorly understood and often miscategorized congenital umbilical cord hernia: an alternative repair method. Hernia. 2017;21(3):449–454. doi: 10.1007/s10029-016-1544-0. [DOI] [PubMed] [Google Scholar]
- 104.Fordyce A, Timms L, Stalder K, Tyler H. Short communication: the effect of novel antiseptic compounds on umbilical cord healing and incidence of infection in dairy calves. J Dairy Sci. 2018;101(6):5444–5448. doi: 10.3168/jds.2017-13181. [DOI] [PubMed] [Google Scholar]
- 105.Dabrowski F, Burdzinska A, Kulesza A, Sladowska A, Zolocinska A, Gala K, Paczek L, Wielgos M. Comparison of the paracrine activity of mesenchymal stem cells derived from human umbilical cord, amniotic membrane and adipose tissue. J Obstet Gynaecol Res. 2017;43(11):1758–1768. doi: 10.1111/jog.13432. [DOI] [PubMed] [Google Scholar]
- 106.Maillacheruvu P, Engel L, Crum I, Devendra K, Eric S. From cord to caudate: characterizing umbilical cord blood stem cells and their paracrine interactions with the injured brain. Pediatr Res. 2018;83(1–2):205–213. doi: 10.1038/pr.2017.251. [DOI] [PubMed] [Google Scholar]
- 107.Shohara R, Yamamoto A, Takikawa S, Iwase A, Hibi H, Kikkawa F, Ueda M. Mesenchymal stromal cells of human umbilical cord Wharton's jelly accelerate wound healing by paracrine mechanisms. Cytotherapy. 2012;14(10):1171–1181. doi: 10.3109/14653249.2012.706705. [DOI] [PubMed] [Google Scholar]
- 108.Liu F, Luo X, Shen H, Dong J, Yang J. Using human hair follicle-derived keratinocytes and melanocytes for constructing pigmented tissue-engineered skin. Skin Res Technol. 2011;17:373–379. doi: 10.1111/j.1600-0846.2011.00510.x. [DOI] [PubMed] [Google Scholar]
- 109.Shim W, Mehta A, Lim S, Zhang G, Lim C, Chua T. G-CSF for stem cell therapy in acute myocardial infarction: friend or foe? Cardiovasc Res. 2011;89(1):20. doi: 10.1093/cvr/cvq301. [DOI] [PubMed] [Google Scholar]
- 110.Wang J, Hauer-Jensen M. Neuroimmune interactions: potential target for mitigating or treating intestinal radiation injury. Br J Radiol. 2007;80(1):S41–S48. doi: 10.1259/bjr/33057885. [DOI] [PubMed] [Google Scholar]
- 111.Ra J, Shin I, Kim S, Kang S, Kang B, Lee H, Kim Y, Jo J, Yoon E, Choi H, et al. Safety of intravenous infusion of human adipose tissue-derived mesenchymal stem cells in animals and humans. Stem Cells Dev. 2011;20(8):1297–1308. doi: 10.1089/scd.2010.0466. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.