Abstract
Severe acute respiratory syndrome coronavirus 2 causes coronavirus disease 2019 (COVID-19), which has become a global pandemic. Apart from the mild features of the disease, long-term complications involve many systems including both endocrine and cardiovascular systems. Myocarditis, secondary to COVID-19, has become a well-known complication of the disease. However, endocrine complications are generally not common, particularly isolated pituitary abnormalities. There is one other report of diabetes insipidus developing as a late sequela of COVID-19. In this article, we report a case of a young male who presented with features of myocarditis but developed diabetes insipidus on day 7 of admission as a long-term complication after recovery from COVID-19 infection. His laboratory test results at the time of developing the complication revealed a high serum sodium level and low urine osmolality. The patient recovered on administration of desmopressin and was discharged after 16 days of hospitalization.
Keywords: diabetes insipidus, myocarditis, complication, COVID-19
Introduction
Coronavirus disease 2019 (COVID-19) is a global pandemic disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus. COVID-19 pandemic has resulted in millions of cases and unfortunate deaths worldwide till now, as we undergo the second wave of the disease. It has shown to affect several organ systems in the human body, the most common and most severely affected being the pulmonary system. As we learn more about the disease every day and as time passes, we are coming across different manifestations and long-term systemic effects of this extraordinary disease.
In the coming months and years, we might see long-term consequences of COVID-19 on the endocrine system as well. We present a unique case of a young male who developed diabetes insipidus as a long-term complication after recovering from COVID-19 infection.
Case Report
A 28-year-old healthy male with no significant past medical history presented with a cough, worsening shortness of breath, and chest pain for 2 days. He described his chest pain as intense sharp, stabbing pain in the chest with breathing. Review of systems was positive for mild headache and nausea. He was diagnosed with COVID-19 infection a month ago via nasopharyngeal reverse transcription polymerase reaction (RT-PCR) at an outside facility after being exposed at work. He had mild symptoms of COVID-19 initially, which included loss of smell, change in taste, dry cough, fatigue, and headache. He completed a 14-day quarantine period without requiring any supplemental oxygen and was treated symptomatically. He denied any fevers, chills, heat or cold intolerance, blurry visions, or any other related symptoms. The patient reported no history of tobacco, alcohol, or recreational drug abuse.
On physical examination, he was found to be alert and oriented. Vitals signs showed a blood pressure of 107/72 mm Hg, a pulse of 103 beats per minute, temperature was 98.4 °F, respiratory rate of 36 per minute, and oxygen saturation of 87% on room air. The patient was put on 2 L of supplemental oxygen immediately. Pupils were about 4 mm bilaterally and reactive to light. On auscultation, he had dual heart sounds with no murmurs noted. Breath sounds were bilaterally equal. The abdomen was soft and nontender, with active bowel sounds. No focal neurological deficits were noted. The investigations are presented in Table 1.
Table 1.
Laboratory Work.
| On admission | On discharge | Normal value | |
|---|---|---|---|
| White blood cells, mm3/µL | 10.1 | 6.8 | 4-11 |
| Hemoglobin, g/dL | 13.5 | 13.5 | 13.5-17.7 |
| Platelets, per mm3/µL | 150 | 542 | 150-450 |
| Serum sodium, mmol/L | 137 | 135 | 134-144 |
| Serum potassium, mmol/L | 3.1 | 3.3 | 3.5-5.1 |
| Serum creatinine, mg/dL | 1.1 | 1.2 | 0.6-1.7 |
| TSH, IU/mL | 0.13 | N/A | 0.36-3.7 |
| Free T4, pg/mL | 1.2 | N/A | 0.7-1.6 |
| AST, unit/L | 97 | 39 | 6-58 |
| ALT, unit/L | 212 | 108 | 14-67 |
| ALP, unit/L | 135 | 98 | 38-150 |
| Total bilirubin, mg/dL | 5.6 | 0.8 | 0.3-1.2 |
| Albumin, g/dL | 2.7 | 2.8 | 3.4-4.7 |
| Direct bilirubin, mg/dL | 4.2 | 0.4 | 0.1-0.4 |
| Troponin, ng/mL | 0.43 | N/A | <0.06 |
| Brain natriuretic peptide, pg/mL | 19600 | N/A | 0-125 |
| COVID-19 RT-PCR NP | Negative | Negative | |
| Inflammatory markers | |||
| C-reactive protein, mg/dL | 32.5 | 9 | 0-0.8 |
| ESR, mm/h | 88 | 57 | 0-21 |
| Procalcitonin, ng/mL | 1.4 | N/A | <0.10 |
| Lactate, mmol/L | 3.5 | 2 | 0.4-2.0 |
Abbreviations: TSH, thyroid-stimulating hormone; AST, aspartate transaminase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; COVID-19, coronavirus disease 2019; RT-PCR, reverse transcription polymerase chain reaction; NP, nasopharyngeal; ESR, erythrocyte sedimentation rate.
Electrocardiogram showed accelerated junctional rhythm with retrograde conduction with nonspecific T wave changes. Chest X-ray showed patchy bibasilar opacities, right greater than left. Computed tomography angiogram of chest was negative for pulmonary emboli but revealed tree-in-bud opacities predominantly in the right lung (Figure 1). The vasculitis panel was ordered, which was negative. Abdominal ultrasound showed mild hepatomegaly with normal common bile duct diameter. The acute hepatitis panel was sent, which returned negative.
Figure 1.
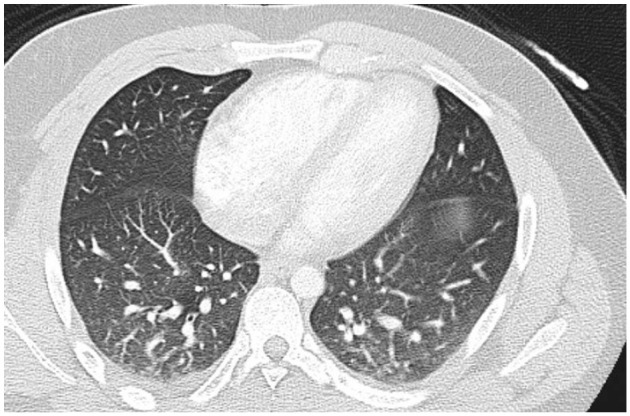
Computed tomography revealing tree-in-bud opacities predominantly in the right lung.
He was started on antibiotic treatment for community-acquired pneumonia. A transthoracic echocardiogram showed left ventricular dysfunction with an ejection fraction of 30% without evidence of clinical heart failure. The patient was diagnosed with myocarditis secondary to post COVID-19 viral infection and he was started on guideline-directed medical therapy with metoprolol, lisinopril, and low-dose aspirin.
On the night before discharge, day 7 of his admission, the patient developed sudden onset of polyuria, polydipsia, and increased thirst. His documented 24-hour urine volume was 2.5 L before he developed the complaints. At the time of these complaints, his urine output was approximately 7 L within 24 hours and the serum sodium level increased to 153 mEq/L. His urine studies revealed a low urine osmolality (93 mOsm/kg) and urine sodium of 16 mOsm/kg. On the administration of 2 µg of desmopressin, the patient improved clinically and symptomatically. He had a decreased urine output, increased urine osmolarity, urine sodium, and improved serum sodium levels (Table 2).
Table 2.
Desmopressin Test.
| Urine osmolarity (mOsm/kg) | Urine sodium (mOsm/kg) | Serum sodium (mmol/L) | Urine output (L/24 h) | |
|---|---|---|---|---|
| Before desmopressin | 93 | 16 | 153 | 7 |
| After 2 µg of desmopressin | 229 | 41 | 145 | 5 |
A magnetic resonance imaging of the brain was performed, which did not reveal any pituitary mass, acute hemorrhage, or malignancy. A diagnosis of diabetes insipidus as a sequelae of COVID-19 viral infection was made. Apart from close monitoring for serum sodium levels and urine output, the patient’s desmopressin dose was optimized and hydrochlorothiazide was also administered. He was discharged home after 16 days of hospitalization on desmopressin 0.3 mg thrice daily and hydrochlorothiazide 12.5 mg daily with close follow-up with endocrinology, nephrology, cardiology, and primary care physician. After 2 weeks, the patient followed-up with the primary care physician and his inflammatory markers, electrolytes, and brain natriuretic peptide were all within normal limits. His urine output was controlled on medications and therefore, his follow-up will continue till recovery.
Discussion
Most of the patients recover fully from COVID-19 after 2 to 6 weeks, but a small percentage of patients may develop post-COVID-19 complications. The long-term effects of COVID-19, its clinical course, and the likelihood of full recovery are still unknown. Some of the well-reported complications associated with post-COVID-19 infection in literature include myocarditis, pericarditis, restrictive lung disease, thromboembolic events such as stroke, pulmonary embolism, myocardial infarction, permanent or partial loss of smell and taste, anxiety, depression, posttraumatic stress disorder, persistent fatigue, and myalgias.1
Involvement of the hypothalamic-pituitary axis secondary to SARS-CoV-2 has been suggested by previous studies. The involvement was first described in a study in which 40% of 61 patients had developed such sequelae; the commonest being central hypocortisolism. However, the manifestations were transient and resolved within a year in 87.5% of the patients.2 In another study focusing on COVID-19, central hypothyroidism had also been noted. Additionally, in a few male survivors, dysfunction of the hypothalamic-pituitary-testicular axis was also noted owing to immune-mediated damage.3
The possibility of pituitary dysfunction was described in a recent case report of a 68-year-old man with severe COVID-19 infection requiring critical care management. In the report, the patient developed altered mental status, polyuria, and other features suggestive of diabetes insipidus, over a span of 23 days and was COVID-19 RT-PCR positive on admission. Despite developing similar symptoms over a similar period, our patient was comparatively younger, had no medical history, and did not require invasive mechanical ventilation.4 Additionally, our patient was COVID-19 RT-PCR negative twice on current admission pointing toward the development of post-COVID-19 complication.
Two hypotheses, inflammation-mediated reversible hypophysitis and direct immune-mediated damage, could be possible explanations for the damage.5 SARS-CoV-2 can directly target the hypothalamic and pituitary tissues because of a high expression of ACE-2 receptors.6 Additionally, SARS-CoV-2 genome-mediated edema and degeneration has also been found in autopsy analysis of patients that could explain the hypophyseal damage.2 However, the question of the damage being reversible remains unanswered.
A similar mechanism can be postulated for the presence of myocarditis. ACE-2 receptors are also present on cardiomyocytes.7 The receptor fraction is upregulated in case of heart failure that could potentially increase the number of entry points for viral invasion.8 In a study focusing on myocarditis, the clinical presentation varied from mild symptoms of fatigue and dyspnea, as noticed in our patient, to full-blown heart failure.9 Furthermore, in such cases, elevations in troponin and BNP reflect both myocardial injury and ventricular dilation. These results were also present in our patient. As suggested by earlier studies, electrocardiogram findings are nonspecific and might not present the classical findings of diffuse ST-elevations.10
To our knowledge, this is the second case of new-onset diabetes insipidus associated with COVID-19 infection as well as the first case of concomitant myocarditis and diabetes insipidus as a post-COVID-19 complication. Desmopressin had been used as the therapy to treat diabetes insipidus in both cases. More studies with long-term follow-up of recovered COVID-19 patients are needed to better determine post-COVID-19 complications, clinical course, and recovery.
Footnotes
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval: Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent: Written informed consent was obtained from the patient’s parent for their anonymized information to be published in this article.
ORCID iDs: Nismat Javed  https://orcid.org/0000-0002-8756-927X
https://orcid.org/0000-0002-8756-927X
Abdul Ahad Ehsan Sheikh  https://orcid.org/0000-0002-6075-5769
https://orcid.org/0000-0002-6075-5769
References
- 1. World Health Organization. What we know about long-term effects of COVID-19. The latest on the COVID-19 global situation & long-term sequelae. Accessed February 18, 2021. https://www.who.int/docs/default-source/coronaviruse/risk-comms-updates/update-36-long-term-symptoms.pdf?sfvrsn=5d3789a6_2; 2020
- 2. Leow MKS, Kwek DSK, Ng AWK, Ong KC, Kaw GJL, Lee LSU. Hypocortisolism in survivors of severe acute respiratory syndrome (SARS). Clin Endocrinol (Oxf). 2005;63:197-202. doi: 10.1111/j.1365-2265.2005.02325.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lazartigues E, Qadir MMF, Mauvais-Jarvis F. Endocrine significance of SARS-CoV-2’s reliance on ACE2. Endocrinology. 2020;161:bqaa108. doi: 10.1210/endocr/bqaa108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Rajevac H, Bachan M, Khan Z. Diabetes insipidus as a symptom of Covid-19 infection: case report. Chest. 2020;158:A2576. doi: 10.1016/j.chest.2020.09.172 [DOI] [Google Scholar]
- 5. Pal R, Banerjee M. COVID-19 and the endocrine system: exploring the unexplored. J Endocrinol Invest. 2020;43:1027-1031. doi: 10.1007/s40618-020-01276-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Marazuela M, Giustina A, Puig-Domingo M. Endocrine and metabolic aspects of the COVID-19 pandemic. Rev Endocr Metab Disord. 2020;21:495-507. doi: 10.1007/s11154-020-09569-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Goulter AB, Goddard MJ, Allen JC, Clark KL. ACE2 gene expression is up-regulated in the human failing heart. BMC Med. 2004;2:19. doi: 10.1186/1741-7015-2-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Guo J, Wei X, Li Q, et al. Single-cell RNA analysis on ACE2 expression provides insights into SARS-CoV-2 potential entry into the bloodstream and heart injury. J Cell Physiol. 2020;235: 9884-9994. doi: 10.1002/jcp.29802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Siripanthong B, Nazarian S, Muser D, et al. Recognizing COVID-19-related myocarditis: the possible pathophysiology and proposed guideline for diagnosis and management. Heart Rhythm. 2020;17:1463-1471. doi: 10.1016/j.hrthm.2020.05.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Zeng JH, Liu YX, Yuan J, et al. First case of COVID-19 complicated with fulminant myocarditis: a case report and insights. Infection. 2020;48:773-777. doi: 10.1007/s15010-020-01424-5 [DOI] [PMC free article] [PubMed] [Google Scholar]


