Abstract
Purpose
The purpose of this meta-analysis was to evaluate the overall safety and effectiveness of perioperative intravenous dexamethasone to facilitate postoperative rehabilitation in patients after total knee arthroplasty (TKA).
Methods
A comprehensive literature search was performed using the Embase, PubMed, Cochrane Library, and China National Knowledge Infrastructure (CNKI) databases for relevant randomized controlled trials (RCTs) from inception to 2020. Methodological quality of the trials was assessed using the Cochrane Risk of Bias Tool, and the relevant data were extracted using a predefined data extraction form.
Results
Ten RCTs with 1100 knees were included. Our study showed a significant reduction in pain using a postoperative pain visual analog scale (VAS) at 24 hours and 48 hours, total opioid consumption at 24 hours and 48 hours, postoperative nausea and vomiting (PONV), active range of motion (ROM) limitation, and passive ROM limitation at 72 hours in dexamethasone-treated groups compared with controls.
Conclusion
Intravenous low-dose dexamethasone is potentially useful in the perioperative setting for reducing postsurgical immediate ROM limitations, pain, opioid consumption, and PONV. There are no data that directly attribute an increase in postoperative complications to intravenous dexamethasone. More high-quality studies are necessary to draw these conclusions.
Keywords: Intravenous dexamethasone, rapid recovery, low-dose, total knee arthroplasty, meta-analysis, pain, range of motion, opioid consumption, postoperative nausea and vomiting
Introduction
Over the past decades, the number of total knee arthroplasty (TKA) surgeries that have been performed has greatly increased, and future demands for this surgery are predicted to continue to rise rapidly.1–3 However, moderate to severe pain and poor postoperative range of motion (ROM) in the early postoperative period occur frequently as a result of soft tissue injury, extensive bone resection, and surgical trauma.4,5 Moreover, postoperative nausea and vomiting (PONV) is also an ongoing problem. Postoperative pain, poor postoperative immediate ROM, and PONV can cause delayed discharge and patient dissatisfaction.6,7 Therefore, the perioperative period should focus on pain management, improving postoperative immediate ROM, and preventing PONV.
Dexamethasone is a highly efficient, long-acting glucocorticoid steroid with an anti-inflammatory effect, and it has been used extensively during the TKA perioperative period. Dexamethasone can inhibit the lipoxygenase and cyclooxygenase pathways in the inflammatory chain reaction, which decreases the level of inflammatory mediators including leukotrienes and prostaglandins.8 However, it can also block the release of neuropeptides from nerve endings and decrease the level of bradykinin in tissues,9,10 both of which increase the sense of injury in surgical wounds and inflammatory tissues. Heterogeneity among studies regarding the dose and administration protocol of dexamethasone, and the perioperative management of TKA makes it difficult to determine the practical value of dexamethasone. To date, no large-scale dose-finding studies have been performed to examine the analgesic effects. To the best of our knowledge, all current high-quality prospective studies use low-dose (no more than 20 mg) of dexamethasone. Additionally, through its central antiemetic effect, this steroid reduces PONV by systemic application rather than topical application.11 Thus, assessing the role of intravenous low-dose dexamethasone in rapid recovery after TKA using a meta-analysis should be a top priority.
Statistical heterogeneity, which was high in previous meta-analyses,12,13 likely originated from different doses and routes of administration. Additionally, the sample size in previous meta-analyses was also small, and they did not report postoperative knee ROM, which was an important parameter for early postoperative recovery. Furthermore, new high-quality randomized controlled trials (RCTs)14,15,17 have been published with seemingly mixed results. Two RCTs16,18 that were written in Chinese were not part of the previous reviews, which caused publication bias and statistical bias.
Thus, a new meta-analysis is needed to evaluate the overall safety and effectiveness of perioperative intravenous dexamethasone for a rapid recovery in patients undergoing TKA.
Methods
Search strategy
The Embase, PubMed, Cochrane Library, and China National Knowledge Infrastructure (CNKI) databases were searched from their inception to 2020 by two of the authors. Prospective randomized trials were searched using combinations of the following terms: dexamethasone, total knee arthroplasty, TKA, and total knee replacement (TKR). Language restrictions were not applied, and the reference lists of searched articles were carefully reviewed to identify additional studies for inclusion.
Inclusion and exclusion criteria
RCTs comparing the clinical efficacy between intravenous low-dose (no more than 20 mg) dexamethasone with placebo or nothing in patients who underwent primary TKA were included in our study. The relevant outcomes included postoperative immediate ROM, visual analog scale (VAS), opioid consumption, length of stay (LOS), adverse effects, and complications during follow-up. Exclusion criteria included the following: studies that were not RCTs; non-intravenous dexamethasone administration; or studies involving revision knee arthroplasty, bilateral TKA, and other surgical approaches such as total hip arthroplasty. Ethics approval and informed consent are not applicable because the data are from articles that have been previously published.
Data extraction
We carefully extracted related data from all eligible studies using a standard extraction worksheet, and disagreements were resolved through discussion. We extracted only the data of interest when the articles had multiple comparisons. If there were no data available in the original article, the authors were contacted by e-mail for further information, or the data were calculated from the available coefficients. Milligrams dispensed for all the named narcotics were converted into morphine milligram equivalent using the following multiplication factors to convert one dispensed milligram of the named narcotic to its morphine milligram equivalent: tramadol × 0.10; oxycodone × 1.50; morphine × 1.00; and fentanyl × 0.01. These potency ratios have been validated in clinical studies.19–21
Quality assessment and risk of bias
The included RCTs were assessed by two authors in accordance with the Cochrane Handbook for Systematic Reviews of Interventions version.22 The following specific items were included: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, selective reporting, incomplete outcome data, and other bias. Disagreements were resolved through discussions with a third author.
Statistical analysis
Statistical analysis was performed using Stata 14.0 software (College Station, TX, USA), and a P value <0.05 was considered to be a significant difference. For continuous outcomes, such as ROM, LOS, VAS, and blood glucose, the standard mean difference (SMD) or the weighted mean difference (WMD) with 95% confidence interval (CI) was calculated. For dichotomous variables such as PONV and complications, a risk ratio (RR) with 95% CI was used to measure the risk evaluation. The primary assessment for statistical heterogeneity was performed using chi-squared and I2 tests. If P >0.05 or I2 < 50%, the heterogeneity was not considered. Our study used the random-effects model for all outcomes. In addition, publication bias was evaluated using a Begg’s funnel plot, and P >0.1 indicated that there was no significant publication bias.
Results
Search results
There were 378 related studies that were initially identified through database searching. Among these, 109 duplicates were excluded using Endnote X8 (Clarivate, Philadelphia, PA, USA), and 250 studies were removed on the basis of the title and abstract. Ten files14–18,23–27 that met the inclusion criteria were ultimately identified after carefully and completely reading each of the 19 remaining studies. The specific process of including studies is shown in Figure 1.
Figure 1.
Flow diagram of the literature search.
THA, total hip arthroplasty.
The sample size of the included studies ranged from 40 to 269 participants, and average age range in the different studies was 58.5 to 72 years. Basic characteristics of the included studies are described in Table 1.
Table 1.
Basic characteristics of the included studies.
| Studies | Year | Country | BMI | Gender (M:F) (D/C) |
Surgical approach | Sample size (D/C) | Mean age (D/C) | Primary diagnosis | Dexamethasone group | Control group | Postoperative medication | Relevant outcome | Follow-up |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fujii and Nakayama27 | 2005 | Japan | 23.7/22.3 | 11:29/10:30 | TKA | 40/40 | 59/58.5 | Unknown | Postoperative IV 8 mg or 16 mg DXM | vehicle | A continuous epidural infusion of combination bupivacaine 0.125% 100 mL plus morphine 0.1 mg/kg. Indomethacin 50 mg was given as rescue analgesia. | adverse effects | 24 hours |
| Koh et al.24 | 2013 | Korea | 26.3/26.1 | 18:117/15:119 | TKA with a standard medial parapatellar arthrotomy | 135/134 | 72.0/72.0 | OA | Preoperative IV 10 mg DXM | NS | PCA (1 mL of a 100-mL solution containing 1500 g or 2000 g fentanyl), 200 mg celecoxib, 75 mg pregabalin, and 650 mg acetaminophen every 12 hours, and IV ketoprofen (100 mg) or IV metoclopramide (10 mg) as required. | VAS, adverse effects, and complications | 12 months |
| Deng et al.18 | 2016 | China | 25.7/24.9 | 13:7/9:11 | TKA | 20/20 | 68.3/66.9 | OA | Preoperative IV 10 mg DXM | NS | PCA (sufentanil 250 μg + droperidol 10 mg + 250 mL NS), a routine anti-infection and anticoagulant treatment. | VAS, ROM, adverse effects, and complications | 6 months |
| Morales-Munoz et al.25 | 2017 | Spain | 31.1/30.2 | 8:19/6:21 | TKA | 27/27 | 68.8/68.8 | Unknown | Preoperative IV 8 mg DXM | NS | Postoperative analgesia for the first 48 hours consisted of continuous perfusion of metamizol (5 mg/minute) and ondansetron (16 g/minute) together with 3-mg boluses of morphine delivered intermittently using a PCA infusion pump | VAS, ROM, adverse effects, and complications | 48 hours |
| Xu et al.26 | 2017 | China | 24.2/24.3 | 8:46/9:45 | TKA with a midline skin incision, medialparapatellar approach | 54/54 | 64.6/64.6 | OA | Pre-and post-operative IV 10 mg DXM respectively | NS | MOAD (50 mg diclofenac every 12 hours, 75 mg pregabalin every 8 hours), oral oxycodone (10 mg every 8 hours) or an IV parecoxib (40 mg) with IV metoclopramide (10 mg) as required. A routine anticoagulant treatment with LMWH. | VAS, ROM, adverse effects, and complications | 2 months |
| Dissanayake et al.23 | 2018 | Australia | Unknown | 41:45/42:36 | TKA | 86/78 | 68.7/66.7 | Unknown | Preoperative IV 8 mg DXM, and another 8 mg DXM was injected when the BGL was less than 8 mmol/L postoperatively | NS | Oxycodone/naloxone 10/5 mg twice daily for 4 to 5 days after surgery, paracetamol 1 g every 6 hours, gabapentin 300 mg daily for 2 to 3 days after surgery, ibuprofen 200–400 mg every 8 hours or celecoxib 100 mg twice daily, oxycodone 5–10 mg every 3 hours and tramadol slow release 100–200 mg twice daily as required. | VAS, adverse effects, and complications | 6 weeks |
| Wu et al.14 | 2018 | China | 26.1/25.9 | 17:33/18:42 | TKA with a midlineskin incision, medial parapatellar approach | 50/50 | 66.9/67.4 | OA | Preoperative IV 10 mg DXM | NS | MOAD (200 mg celecoxib every 12 hours, 75 mg pregabalin every 8 hours), oral oxycodone (10 mg every 8 hours) or IV pethidine hydrochloride (100 mg) with IV metoclopramide (10 mg) as required. Routine anticoagulant treatment with LMWH. | VAS, ROM, adverse effects, and complications | 72 hours |
| Xu et al.15 | 2018 | China | 25.7/28.8 | 11:49/8:53 | TKA with a midline skin incision, medial parapatellar approach | 60/61 | 64.5/65.8 | OA | Preoperative IV 20 mg DXM | NS | Oral diclofenac (50 mg every 12 hours), oral mosapride (5 mg 3 times daily), oral oxycodone HCl (10 mg) or IV morphine (10 mg) with an IV metoclopramide (10 mg) as required. | VAS, ROM, adverse effects, and complications | 72 hours |
| Wang et al.16 | 2019 | China | 26.5/26.9 | 12:20/13:19 | TKA with a midline skin incision, medialparapatellar approach | 32/32 | 65.3/65.9 | OA | Preoperative IV 8 mg DXM | NS | Anticoagulant with rivaroxaban 12 hours after surgery, analgesic treatment with oral and IV paroxib for the first 3 days after surgery, oral celecoxib after 3 days, and IV tramadol as required. | VAS and adverse effects | 72 hours |
| Tammachote and Kanitnate17 | 2020 | Thailand | 27.0/27.0 | 8:42/6:44 | TKA with a standard medial parapatellar arthrotomy | 50/50 | 67.0/69.0 | OA | Preoperative IV 0.15 mg/kg of DXM, the maximum dose of DEX was 12 mg. | NS | An IV 15 or 30 mg of ketorolac every 6 hours and 3 mg of IV morphine every 3 hours as required for the first 2 days after surgery; 250 mg of oral naproxen twice daily, 1300 mg of extended-release acetaminophen every 8 hours, 25 mg of nortriptyline plus 75 mg of pregabalin and 50 mg of tramadol as required on postoperative day 3. | VAS, ROM, adverse effects | 12 weeks |
BGL, blood glucose level; DXM, dexamethasone; IV, intravenous; LMWH, low-molecular-weight heparin; LOS, length of stay; MOAD, multimodal oral analgesic drugs; NS, normal saline; PCA, patient-controlled analgesia; TKA, total knee arthroplasty; VAS, visual analog scale; OA, osteoarthritis; ROM, range of motion; D, dexamethasone group; C, control group.
The results of the risk of bias assessment are presented in Figure 2.
Figure 2.

Risk of bias summary for the included studies. (+ represents yes; – represents no; ? represents unclear).
Primary outcomes
Pain visual analog scale
A pain VAS following TKA was reported in nine studies.14–18,23–26 All of these nine studies assessed VAS at 24 hours,14–18,23–26 and the VAS at 48 hours was evaluated in six studies.15–17,23–25 The pooled results demonstrated that perioperative intravenous low-dose dexamethasone decreased the postoperative pain score at 24 hours (SMD = −0.430; 95%CI: −0.570 to −0.291; P < 0.001) and 48 hours (SMD = −0.250; 95%CI: −0.405 to −0.096; P = 0.002). The statistical heterogeneity among trials was low in the VAS at 24 hours (I2 = 15.6%,) and 48 hours (I2 = 12.0%) (Figure 3).
Figure 3.
Forest plot for the comparison of VAS at 24 hours and 48 hours after surgery.
VAS, visual analog scale.
Immediate range of motion
Four studies14,15,18,26 provided ROM data at 72 hours after TKA. Among these four studies, three14,15,26 assessed passive ROM at 72 hours, and one study18 assessed active ROM at 72 hours. The overall results showed that intravenous low-dose dexamethasone improved the active ROM (SMD = 1.254; 95%CI: 0.573 to 1.935; P < 0.001) and passive ROM (SMD = 0.371; 95%CI: 0.152 to 0.589; P = 0.001). Heterogeneity was not identified for ROM between articles (I2 = 0) (Figure 4).
Figure 4.
Forest plot for the comparison of ROM at 72 hours after surgery among the two subgroups.
ROM, range of motion.
Postoperative nausea and vomiting
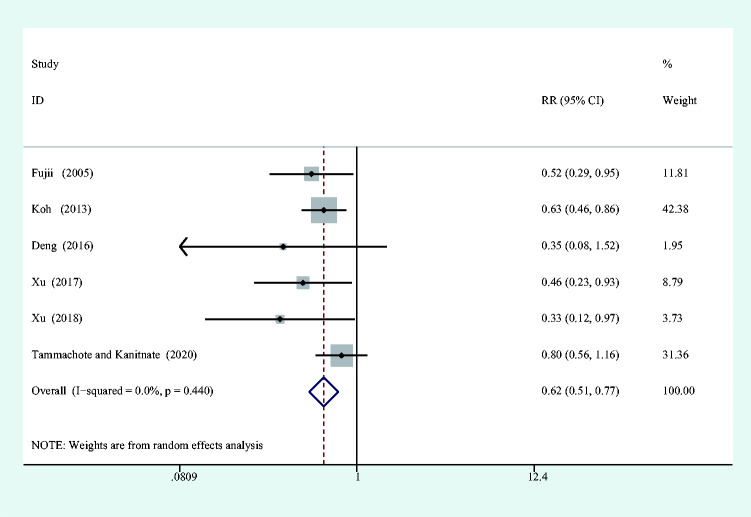
Data from six studies15,17,18,24,26,27 investigated the PONV. The overall results clarified that perioperative intravenous dexamethasone (10 to 20 mg) decreased the incidence of PONV (RR = 0.623; 95%CI: 0.508 to 0.765; P < 0.001). Statistical heterogeneity in PONV was not identified between articles (I2 = 0) (Figure 5).
Figure 5.
Forest plot for the comparison of PONV.
PONV, postoperative nausea and vomiting.
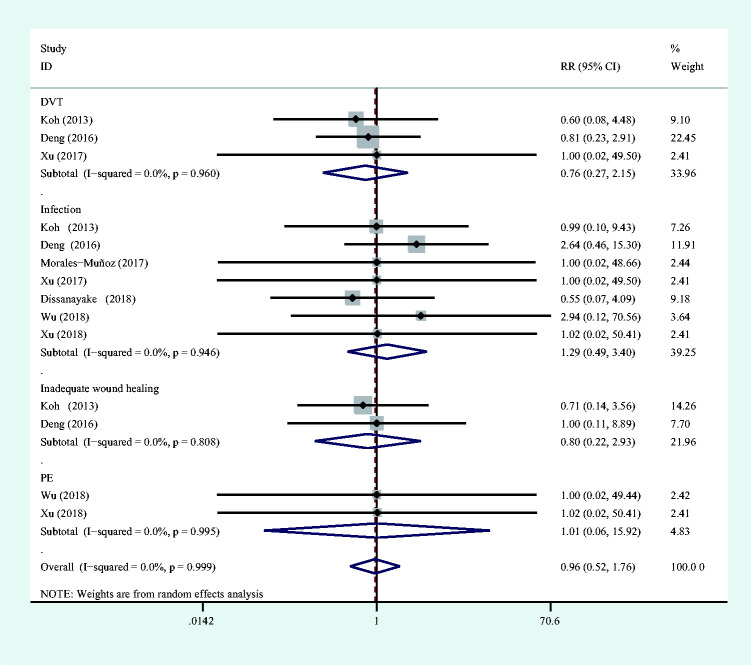
Complications
Seven studies14,15,18,23–26 reported the postoperative complications including deep vein thrombosis (DVT), infection, inadequate wound healing, and pulmonary embolism (PE). The pooled outcomes showed that perioperative intravenous dexamethasone at doses of 8 to 20 mg did not increase the risk of postoperative early complications compared with the control groups (RR = 0.964; 95%CI: 0.527 to 1.762). There was no statistical heterogeneity found between trials (I2 = 0) (Figure 6).
Figure 6.
Forest plot for the comparison of complications.
Secondary outcomes
Total opioid consumption at 24 and 48 hours
Four studies17,23–25 reported the total opioid consumption at 24 hours and 48 hours after surgery. The pooled results demonstrated that perioperative intravenous low-dose dexamethasone decreased the total opioid consumption at 24 hours (SMD = −0.474; 95%CI: −0.850 to −0.098; P = 0.013) and 48 hours (SMD = −0.323; 95%CI: −0.533 to −0.114; P = 0.003). The statistical heterogeneity among trials was high for the total opioid consumption at 24 hours (I2 = 77.8%, P = 0.004) and low at 48 hours (I2 = 32.7%) (Figure 7).
Figure 7.
Forest plot for the comparison of total opioid consumption at 24 hours and 48 hours after surgery.
Length of stay
Five studies14,15,17,23,26 reported the outcome of LOS after TKA. The overall pooled outcomes illustrated that there was no significant difference in LOS between the dexamethasone group and the control group (WMD = −0.110; 95%CI: −0.245 to 0.025). No statistical heterogeneity was detected for LOS between the analyzed articles (I2 = 0) (Figure 8).
Figure 8.
Forest plot for the comparison of LOS.
LOS, length of stay.
Blood glucose
Postoperative blood glucose at 24 hours was reported in four studies,15–17,23 and two studies15,16 recorded blood glucose at 72 hours. Compared with the control group, perioperative intravenous administration of low-dose dexamethasone remarkably increased the postoperative blood glucose levels at 24 hours (WMD = 0.750; 95%CI: 0.408 to 1.092; P < 0.001), but there was not much difference in the postoperative blood glucose at 72 hours (WMD = −0.188; 95%CI: −0.595 to 0.359). The degree of statistical heterogeneity among trials was low for blood glucose at 24 hours (I2 = 0) and 72 hours (I2 = 33.9%) (Figure 9).
Figure 9.
Forest plot for the comparison of blood glucose at 24 hours and 72 hours after surgery.
Publication bias
Publication bias was evaluated using a Begg’s funnel plot for VAS at 24 hours, which showed a low risk of publication bias. However, the publication bias cannot be eliminated because of the limited number (less than 10) of studies that were included in each analysis (Figure 10).
Figure 10.
Begg’s funnel plot of the current meta-analysis of VAS at 24 hours after surgery.
VAS, visual analog scale.
Discussion
The main findings of this meta-analysis were that perioperative intravenous low-dose dexamethasone may be useful for improving the immediate ROM, lowering pain, and decreasing opioid consumption and the incidence of PONV in the early postoperative period without increasing related adverse events during the follow-up period.
Pain management is the key to quick postoperative rehabilitation and satisfactory recovery of patients.28 Moreover, postoperative pain can contribute to postponement of discharge, prolonged stiffness, immobility-related complications, inability to perform rehabilitation exercise, poor outcome, delayed recovery, and greater consumption of care resources.29,30 In this study, we observed that, compared with placebo, perioperative use of dexamethasone was an effective approach to decrease the VAS score at 24 hours and 48 hours, which was consistent with two previous meta-analyses.12,13 Inflammation markers including C-reactive protein (CRP) and interleukin (IL)-6 achieved effective control during the corresponding period. Thus, we believe that surgical trauma after undergoing TKA often leads to severe postoperative inflammatory reactions, which are concurrent with postoperative pain.31 Opioid consumption is another important component of postoperative pain assessment. Our meta-analysis stated that perioperative intravenous low-dose dexamethasone could decrease the consumption of opioids at 24 hours and 48 hours after TKA, which were also consistent with studies by Li et al.12 and Fan et al.13 However, Zhou et al.32 found that there was no significant difference in terms of opioid requirements at 24 hours and 48 hours, but the p-value was very close to 0.05. For these distinctions, we believe that one of the most likely reasons is the different routes of administration. Zhou et al.’s study included two articles33,34 that were not listed in this study or in Li et al.’s12 or Fan et al.’s13 study. The intervention in these two studies33,34 was periarticular injection of dexamethasone, which has been shown to have no effect on opioid consumption at 24 hours and 48 hours postoperatively compared with intravenous administration.35 Therefore, the difference is greatly reduced in the pooled results when these two articles33,34 are included.
ROM is a significant parameter for postoperative functional recovery of patients who are undergoing TKA. An angle of 67° of knee flexion is required for the swing phase of gait, 80° is required to walk up stairs, 90° is required to walk down stairs, 93° is required to stand up from a chair after TKA,36 and 106° is necessary for related activities such as tying shoelaces.37 Lei et al.38 found that two intravenous doses of 10 mg dexamethasone improved the functional outcomes of total hip arthroplasty. However, it is still uncertain whether dexamethasone is effective for improving knee function after primary TKA. A few studies reported an improvement in intravenous dexamethasone groups,14,15,18 while another study did not show a difference.26 This is the first systematic review and meta-analysis to provide important evidence that intravenous low-dose dexamethasone may improve early postoperative ROM. The most likely reason for this improvement is the increased willingness of the patient to perform early functional exercises due to the relief of immediate postoperative pain. However, many other variables may influence immediate postoperative ROM including the surgical technique, presurgical training and education, and implant design, as well as postoperative care within the hospital, which was not reported in the included papers. Because of these limitations and because only four trials reported the immediate postoperative ROM, further investigation is required.
Through a central antiemetic effect, steroids reduce PONV via systemic application. However, the links between PONV control and the dose of systemic dexamethasone have not been investigated thoroughly. Several reviews have confirmed and documented the efficacy of perioperative dexamethasone at doses of 4 to 10 mg as PONV prophylaxis, and there was no risk of side effects.39–41 However, our study found that 10 to 20 mg of intravenous dexamethasone is also effective in preventing PONV. Bustos et al.42 found that two scheduled doses of 8 mg of dexamethasone seemed to be a safe adjunct to the perioperative protocol that reduced PONV. This seems to be in agreement with perioperative intravenous low-dose dexamethasone for prophylactic PONV.
Because of the low incidence rate of complications such as DVT, infection, inadequate wound healing, and symptomatic PE after TKA, it is difficult to obtain sufficient power in RCTs to show any potential superiority of intravenous dexamethasone for these results. Vuorinen et al.43 revealed that use of a low dose of dexamethasone did not increase the incidence of postoperative prosthetic joint infections. To date, there are no data that reliably and directly attribute the result of an increase in postoperative complications to intravenous dexamethasone. In addition, our study illustrated that perioperative intravenous dexamethasone might contribute to postoperative hyperglycemia. Thus, we suggest that blood glucose monitoring and management should be strengthened after surgery, especially in diabetics.
Current rapid recovery protocols have changed total joint replacement by reducing the hospital stay, and there was no increase in the readmission rate.44 As mentioned above, perioperative intravenous low-dose dexamethasone was associated with an immediate improvement in the ROM and a lower pain score and PONV. However, these benefits did not seem to translate into a shorter LOS in this study. One possible explanation for this was that five related studies14,15,17,23,26 had different discharge criteria, which might greatly reduce the clinical significance of this parameter.
Our meta-analysis has several advantages compared with meta-analyses that were previously published.12,13,32 First, our study included three additional high-quality RCTs14,15,17 and two RCTs16,18 that were written in Chinese and had been excluded from previous meta-analyses, and this would reduce statistical bias and publication bias. Second, we reported the postoperative knee ROM, which is an important parameter for early postoperative recovery and discharge criteria, and it was not included in the previous study. Third, we excluded studies with different routes of administration such as periarticular and perineural dexamethasone, which significantly reduced the heterogeneity in the pooled results. Our study also has several limitations. Only ten RCTs with 1100 knees were included. The follow-up varied from 24 hours to 12 months, and the absence of mid-term and long-term follow-up did not allow robust assessment of the incidence of complications. Statistical heterogeneity was inevitable because of the different dexamethasone doses. There was no standardization for concomitant painkillers, which also introduces statistical bias when evaluating the efficacy of dexamethasone. The results of this study cannot be applied to other types of steroids because there were significant differences in pharmacological properties such as half-life and anti-inflammatory effects in different steroids.
Conclusion
Perioperative intravenous low-dose dexamethasone may play a critical role in rapid recovery after TKA by reducing the immediate ROM limitation, pain, opioid consumption, and PONV. There are no data that reliably and directly attribute intravenous dexamethasone to an increase in postoperative complications, and high-quality studies with a large sample size are required to explore the relationship between complications and the dose and response before dexamethasone is extensively used.
Acknowledgements
We are incredibly grateful for the many helpful comments from anonymous reviewers of an earlier version of this manuscript.
Data availability: The data that support the findings of this study are available upon request from the corresponding author.
Declaration of conflicting interest: The authors declare that there is no conflict of interest.
Funding: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Authors’ contributions: YGZ and YYZ designed the study. YTH, RGY, CLW, and JY participated in the literature search and data extraction. YGZ, HYW, and RGY oversaw quality assessment and statistical analysis. YGZ was responsible for manuscript review.
ORCID iD: Yiyuan Zhang https://orcid.org/0000-0002-7886-6926
References
- 1.Kim S. Changes in surgical loads and economic burden of hip and knee replacements in the US: 1997–2004. Arthritis Rheum 2008; 59: 481–488. [DOI] [PubMed] [Google Scholar]
- 2.Koh IJ, Kim TK, Chang CB, et al. Trends in use of total knee arthroplasty in Korea from 2001 to 2010. Clin Orthop Relat Res 2013; 471: 1441–1450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kurtz S, Ong K, Lau E, et al. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am 2007; 89: 780–785. [DOI] [PubMed] [Google Scholar]
- 4.Sinatra RS, Torres J, Bustos AM. Pain management after major orthopaedic surgery: current strategies and new concepts. J Am Acad Orthop Surg 2002; 10: 117–129. [DOI] [PubMed] [Google Scholar]
- 5.Lakra A, Murtaugh T, Shah RP, et al. Early postoperative pain predicts 2-year functional outcomes following knee arthroplasty [published online ahead of print, 2019 Jul 3]. J Knee Surg 2019; 10.1055/s-0039-1692650. [DOI] [PubMed] [Google Scholar]
- 6.Clement ND, Bardgett M, Weir D, et al. The rate and predictors of patient satisfaction after total knee arthroplasty are influenced by the focus of the question: a standard satisfaction question is required. Bone Joint J 2018; 100-B: 740–748. [DOI] [PubMed] [Google Scholar]
- 7.Dorr LD, Chao L. The emotional state of the patient after total hip and knee arthroplasty. Clin Orthop Relat Res 2007; 463: 7–12. [PubMed] [Google Scholar]
- 8.Romundstad L, Stubhaug A. Glucocorticoids for acute and persistent postoperative neuropathic pain: what is the evidence?. Anesthesiology 2007; 107: 371–373. [DOI] [PubMed] [Google Scholar]
- 9.Hargreaves KM, Costello A. Glucocorticoids suppress levels of immunoreactive bradykinin in inflamed tissue as evaluated by microdialysis probes. Clin Pharmacol Ther 1990; 48: 168–178. [DOI] [PubMed] [Google Scholar]
- 10.Hong D, Byers MR, Oswald RJ. Dexamethasone treatment reduces sensory neuropeptides and nerve sprouting reactions in injured teeth. Pain 1993; 55: 171–181. [DOI] [PubMed] [Google Scholar]
- 11.Lunn TH, Kehlet H. Perioperative glucocorticoids in hip and knee surgery benefit vs. harm? A review of randomized clinical trials. Acta Anaesthesiol Scand 2013; 57: 823–834. [DOI] [PubMed] [Google Scholar]
- 12.Li X, Xu G, Xie W, et al. The efficacy and safety of dexamethasone for pain management after total knee arthroplasty: a systematic review and meta-analysis. Int J Surg 2018; 53: 65–71. [DOI] [PubMed] [Google Scholar]
- 13.Fan Z, Ma J, Kuang M, et al. The efficacy of dexamethasone reducing postoperative pain and emesis after total knee arthroplasty: a systematic review and meta-analysis. Int J Surg 2018; 52: 149–155. [DOI] [PubMed] [Google Scholar]
- 14.Wu Y, Lu X, Ma Y, et al. Perioperative multiple low-dose dexamethasones improves postoperative clinical outcomes after total knee arthroplasty. BMC Musculoskelet Disord 2018; 19: 428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Xu H, Zhang S, Xie J, et al. Multiple doses of perioperative dexamethasone further improve clinical outcomes after total knee arthroplasty: a prospective, randomized, controlled study. J Arthroplasty 2018; 33: 3448–3454. [DOI] [PubMed] [Google Scholar]
- 16.Wang YF, Liu HM, Wu JM, et al. Effect of methylprednisolone and dexamethasone on perioperative condition of total knee arthroplasty. The Orthopedic Journal of China 2019; 27: 794–798. DOI:10.3977/j.issn.1005-8478.2019.09.06 [Google Scholar]
- 17.Tammachote N, Kanitnate S. Intravenous dexamethasone injection reduces pain from 12 to 21 hours after total knee arthroplasty: a double-blind, randomized, placebo-controlled trial. J Arthroplasty 2020; 35: 394–400. [DOI] [PubMed] [Google Scholar]
- 18.Deng B, Wang JC, Yan LQ, et al. A study of clinical effects of dezocine combined with dexamethasone in total knee arthroplasty. Chinese Journal of Bone and Joint 2016; 5: 859–863. DOI:10.3969/j.issn.2095-252X.2016.11.013 [Google Scholar]
- 19.Woodhouse A, Hobbes AF, Mather LE, et al. A comparison of morphine, pethidine and fentanyl in the postsurgical patient-controlled analgesia environment. Pain 1996; 64: 115–121. [DOI] [PubMed] [Google Scholar]
- 20.Woodhouse A, Ward ME, Mather LE. Intra-subject variability in post-operative patient-controlled analgesia (PCA): is the patient equally satisfied with morphine, pethidine and fentanyl?. Pain 1999; 80: 545–553. [DOI] [PubMed] [Google Scholar]
- 21.Lee CR, McTavish D, Sorkin EM. Tramadol. A preliminary review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in acute and chronic pain states. Drugs 1993; 46: 313–340. [DOI] [PubMed] [Google Scholar]
- 22.Higgins JPT, Green S. (eds). Cochrane handbook for systematic reviews of interventions. Wiley-Blackwell, 2011, pp.102–108 version 5.1.0 Available from http://www.cochrane-handbook.org. [Google Scholar]
- 23.Dissanayake R, Du HN, Robertson IK, et al. Does dexamethasone reduce hospital readiness for discharge, pain, nausea, and early patient satisfaction in hip and knee arthroplasty? A randomized, controlled trial. J Arthroplasty 2018; 33: 3429–3436. [DOI] [PubMed] [Google Scholar]
- 24.Koh IJ, Chang CB, Lee JH, et al. Preemptive low-dose dexamethasone reduces postoperative emesis and pain after TKA: a randomized controlled study. Clin Orthop Relat Res 2013; 471: 3010–3020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Morales-Muñoz C, Sánchez-Ramos JL, Díaz-Lara MD, et al. Analgesic effect of a single-dose of perineural dexamethasone on ultrasound-guided femoral nerve block after total knee replacement. Rev Esp Anestesiol Reanim 2017; 64: 19–26. [DOI] [PubMed] [Google Scholar]
- 26.Xu B, Ma J, Huang Q, et al. Two doses of low-dose perioperative dexamethasone improve the clinical outcome after total knee arthroplasty: a randomized controlled study. Knee Surg Sports Traumatol Arthrosc 2017; 26: 1549–1556. [DOI] [PubMed] [Google Scholar]
- 27.Fujii Y, Nakayama M. Effects of dexamethasone in preventing postoperative emetic symptoms after total knee replacement surgery: a prospective, randomized, double-blind, vehicle-controlled trial in adult Japanese patients. Clin Ther 2005; 27: 740–745. [DOI] [PubMed] [Google Scholar]
- 28.Myles PS, Williams DL, Hendrata M, et al. Patient satisfaction after anaesthesia and surgery: results of a prospective survey of 10,811 patients. Br J Anaesth 2000; 84: 6–10. [DOI] [PubMed] [Google Scholar]
- 29.Pang WW, Hsu TC, Tung CC, et al. Is total knee replacement more painful than total hip replacement? Acta Anaesthesiol Sin 2000; 38: 143–148. [PubMed] [Google Scholar]
- 30.Puntillo KA. Acute pain management: operative or medical procedures and trauma. Nurs Dyn 1993; 2: 14. [PubMed] [Google Scholar]
- 31.Kehlet H, Jensen TS, Woolf CJ. Persistent postsurgical pain: risk factors and prevention. Lancet 2006; 367: 1618–1625. [DOI] [PubMed] [Google Scholar]
- 32.Zhou G, Ma L, Jing J, et al. A meta-analysis of dexamethasone for pain management in patients with total knee arthroplasty. Medicine (Baltimore) 2018; 97: e11753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chu W, Li WZ. Effect of dexamethasone used in periarticular infiltration analgesia for total knee arthroplast. J Clin Orthop 2017; 20: 568–571. [Google Scholar]
- 34.Liu DM, Zhou XS. A study of dexamethasone used in periarticular infiltration analgesia for total knee arthroplasty. Chin J bone Joint Injury 2016; 31: 1161–1163. [Google Scholar]
- 35.Li D, Wang Q, Zhao X, et al. Comparison of intravenous and topical dexamethasone for total knee arthroplasty: a randomized double-blinded controlled study of effects on dexamethasone administration route and enhanced recovery. J Arthroplasty 2020: S0883-5403(20)31210-9. [DOI] [PubMed] [Google Scholar]
- 36.Laubenthal KN, Smidt GL, Kettelkamp DB. A quantitative analysis of knee motion during activities of daily living. Phys Ther 1972; 52: 34–43. [DOI] [PubMed] [Google Scholar]
- 37.Jevsevar DS, Riley PO, Hodge WA, et al. Knee kinematics and kinetics during locomotor activities of daily living in subjects with knee arthroplasty and in healthy control subjects. Phys Ther 1993; 73: 229–242. [DOI] [PubMed] [Google Scholar]
- 38.Lei YT, Xu B, Xie XW, et al. The efficacy and safety of two low-dose peri-operative dexamethasone on pain and recovery following total hip arthroplasty: a randomized controlled trial. Int Orthop 2018; 42: 499–505. [DOI] [PubMed] [Google Scholar]
- 39.Henzi I, Walder B, Tramer MR. Dexamethasone for the prevention of postoperative nausea and vomiting: a quantitative systematic review. Anesth Analg 2000; 90: 186–194. [DOI] [PubMed] [Google Scholar]
- 40.Holte K, Kehlet H. Perioperative single-dose glucocorticoid administration: pathophysiologic effects and clinical implications. J Am Coll Surg 2002; 195: 694–712. [DOI] [PubMed] [Google Scholar]
- 41.Apfel CC, Korttila K, Abdalla M, et al. A factorial trial of six interventions for the prevention of postoperative nausea and vomiting. N Engl J Med 2004; 350: 2441–2451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bustos FP, Coobs BR, Moskal JT. A retrospective analysis of the use of intravenous dexamethasone for postoperative nausea and vomiting in total joint replacement. Arthroplasty Today 2019; 5: 211–215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Vuorinen MA, Palanne RA, Mäkinen TJ, et al. Infection safety of dexamethasone in total hip and total knee arthroplasty: a study of eighteen thousand, eight hundred and seventy two operations. Int Orthop 2019; 43: 1787–1792. [DOI] [PubMed] [Google Scholar]
- 44.Stambough JB, Nunley RM, Curry MC, et al. Rapid recovery protocols for primary total hip arthroplasty can safely reduce length of stay without increasing readmissions. J Arthroplasty 2015; 30: 521–526. [DOI] [PMC free article] [PubMed] [Google Scholar]